Abstract
Bivalve filter feeders, such as oysters, filter large volumes of water and are particularly exposed to microplastics (MP). Consequently, these animals digest and assimilate high levels of MP in their bodies that may likely impact their physiology, and potentially affect shellfish stocks, benthic habitats and, indirectly, the health status of the marine ecosystem and human consumers. In this study we exposed juvenile oysters, Crassostrea gigas, to 3 different MP concentrations (104, 105 and 106 particles L−1), represented by 6μm Polystyrene (PS) microbeads, compared to a control treatment receiving no MP. The study ran for a period of 80 days to test for the impacts of MP on growth, Condition Index and Lysosomal Stability. From histological analysis, microbeads were detected in the intestines of exposed oysters and in the digestive tubules, but no cellular inflammatory features were observed over time. Weight and shell length remained comparable between the different treatments and control. We found that Condition Index in the highest concentration increased initially but significantly reduced over time. The oysters in the highest MP exposure also showed the lowest mean Lysosomal Stability score throughout the experiment. Lysosomes play a vital role in the cells defense mechanisms and breakdown of constituents, crucial for the oysters’ wellbeing. Most importantly, we detected an increased mortality in those oysters who were chronically exposed to the highest loads of MP.
Keywords: Aquaculture, Aquatic ecology, Ecological health, Environmental hazard, Environmental health, Environmental science, Environmental toxicology, Wastewater management, Water pollution, Microplastics, Pacific oyster, Long term exposure, Lysosomal stability, Condition index, Histology, Polystyrene microbeads
Aquaculture; Aquatic ecology; Ecological health; Environmental hazard; Environmental health; Environmental science; Environmental toxicology; Wastewater management; Water pollution; Microplastics; Pacific oyster; Long term exposure; Lysosomal stability; Condition index; Histology; Polystyrene microbeads
1. Introduction
It is well established that the marine environment is widely polluted with MPs (<5 mm) and that this issue poses a serious threat to marine biota [1, 2]. Bivalve filter feeders living in coastal waters, such as oysters, are particularly exposed to MPs because of their feeding mode and enormous filtration capacity; individual oysters can filter ~5–25L of seawater h-1 [3, 4, 5], making them likely to ingest MPs [4, 6]. Many specimens have been found to contain high loads of MPs in the field [1]. Microplastics in oysters are directly related to the population density within the watershed. Hooded oysters, Saccostrea cucullata, along the Pearl River Estuary (China) near urban areas contained statistically significantly more MPs than those near rural areas [7]. Bivalves ingest and assimilate high levels of MPs in their bodies that may likely impact their physiology, and potentially affect both shellfish stocks, habitats and, indirectly, the health status of the marine ecosystem and human consumers [3, 8, 9, 10, 11]. Bivalves are recommended as ideal sentinel species in several marine monitoring programmes, including those supported by international bodies such as ICES and OSPAR [12]. As a result, bivalves have been recommended as a bioindicator for monitoring MP pollution [13]. They are typically chosen for exposure experiments due to their important role in the economy and the ecosystem. Several experimental studies have shown cellular responses (e.g. loss of lysosomal membrane integrity, oxidative stress, DNA damage) or negative effects on feeding, growth and reproduction of adult bivalves, such as oysters, mussels and clams, after exposure to relatively high concentrations of certain types of MPs, mostly PS spheres [6, 14, 15, 16, 17]. Yet another study found no statistically significant effect on development or feeding capacity of Pacific oyster larvae (Crassostrea gigas) after ingestion of micro- and nanoplastics [18]. More evidence is needed, as previous studies mostly used acute and subchronic treatments and exposure concentrations exceeding environmental concentrations, and thus being of indirect relevance [1, 19]. The Pacific oyster is the most cosmopolitan of all oyster species and a successful aquaculture species. They have a wide global distribution, are hardy and grow rapidly, and thrive in temperatures ranging from 8-22 °C with a salinity between 24 and 28 ppt [20]. As a result, the Pacific oyster has also become the leading species in world shellfish culture, with an estimated production of 573 617 t in 2016 [21]. They are relatively straightforward to culture and handle in the laboratory and bioaccumulate toxins by filtration, making them an ideal species for studying biological processes. In this study, we tested the hypothesis that a model MP (fluorescently stained spherical PS; 6 μm) impacts the physiology and health of juvenile Pacific oysters during their growing phase. To detect impacts of 6 μm PS microplastics in juvenile oysters we opted for three different MP concentrations and a long-term treatment of 80 days. To demonstrate exposure to and effects of PS microbeads, a set of generic biomarkers and endpoints, showed to be responsive in earlier laboratory studies with bivalves, were used. These were: Condition Index (CI), Lysomal Stability (LMS) and growth. In addition, we performed a histological analysis to identify the distribution of PS microbeads in digestive tissues and to screen for potential pathology.
2. Material & methods
2.1. Tested organism
The oysters were supplied from Guernsey oyster hatchery and were considered healthy and uncontaminated by biological agents other than normal flora. The oysters were sent directly from the hatchery and transferred in crates to Cefas’ Weymouth laboratory. To avoid biological contamination, the study was conducted in a room where no further studies with other bivalve species were taking place. The seed oysters were held in 15L flow-through glass tanks. Oysters were rinsed and acclimatised for 1 day prior to the start of the study. All oysters were fed a diet consisting of live algae (Tetraselmis suecica & Isochrysis galbana mixture from Guernsey Sea Farms delivered weekly) supplemented with artificial food (SD1800 - Shellfish Diet 1800 from Reed Mariculture) at predefined feeding times. They were dosed with 6 μm PS microbeads once daily. The uptake of the microbeads was optimised by feeding the oysters algae mixtures within a similar size range as the microbeads. During weekdays, oysters were fed twice a day, once with the live algae mixture supplemented with preserved algae (SD1800) and once with pure preserved algae (SD1800). During the weekend, oysters were fed only once a day, with a mixture of live and preserved algae. Food concentrations were calculated as: 5% wet weight of live algae and 8% dry weight of preserved algae per g dry weight of oyster tissue. The body weight used in this feeding calculation was increased weekly, with 5% as a measure of predicted growth in the absence of real data during the first 10 days and revised body weight predictions after each sampling point using collected data.
The tanks in the study were bespoke glass aquaria, semi static 15 L tanks, all of which could be easily emptied via a bottom valve. Daily, the seawater in the tanks was drained and refilled with clean seawater. All used seawater was UV treated and filtered via a series of three sequential ceramic filter units (20μm, 10μm, 0.2μm – Deltaqua International). The PS microbeads were added to the tanks and then corresponding live algal suspensions were added. Further details of the feeding regime can be found in the supplementary material.
2.2. Tested MP
The MPs used in this study are chemically inert 6μm Red Fluorescent PS Microbeads (Fluoresbrite Polysciences Cat #19111-2 Lot 653002 (day 0–58) & Lot # 660155 (day 59–80)). Fluoresbrite particles are routinely used in a wide range of applications, including as tracer particles and in phagocytosis assays. The initial stock solution, 1 × 106 particles L−1, was made according to Table A (see supplementary material) using the manufacturer's supplied solution. The MPs stock, as supplied by the manufacturer and stored in the fridge, was removed in the morning and sonicated in a water bath for 5 min prior to use to disperse any aggregates formed. Solutions for the 1 × 105 and 1 × 104 particles L−1 were prepared by serial solutions (1:10) of the stock solution in reverse osmosis water (rH2O). PS microbeads were added to filtered seawater and suspended in the water column by using a filtered air lift.
At two different stages, water samples were taken during one full cycle (0h, 1h, 4h, 12h & 24h) from tanks with different concentrations to improve our understanding of the actual exposure conditions and processes involved within the tanks. These samples were analysed for PS microbead concentrations using a fluorescent cytoflow counter. Two additional tests were run in duplicate tanks, one set containing seawater and PS microbeads, the other set containing seawater, PS microbeads and algae. A 1 × 10−4 dilution of the Fluoresbrite polychromatic 6.0 μm Microspheres (Polysciences) was prepared to identify the position of the bead cluster on the cytogram. This cluster reference was used in further analysis to identify the number of beads in the samples. Each water sample was placed in an ultrasonic bath for 10 min and homogenised before being passed through a 200 μm mesh and a 20 μm mesh, removing excessive organic matter prior to the flow cytometer analysis. Each sample was run using the following cytoflow counter settings: Forward scatter; Trigger level of 25mV; Maximum flow speed; 10-minute runtime. Forward scatter was selected as trigger level and used to remove noise from the cytogram. Ten minutes of runtime was allocated to analyse the maximum number of beads/volume.
2.3. Experimental design and treatment
In this study we exposed C. gigas to 3 different concentrations of MPs (104, 105 and 106 particles L−1; PS microbeads; 6μm) compared to a control treatment receiving no plastics for a period of 80 days, to test for the impacts of MP on growth, CI and LMS. We reviewed available microplastic field concentrations [22] in combination with model outputs [23, 24] to select three concentrations for this size of microplastics, representing potential short-term and long-term environmental exposure scenarios. Histology was conducted at the start and during the sampling points to locate the PS microbeads in the oyster tissue. Samples (growth, CI, LMS and histology) were taken on days 0, 10, 20, 40 and 80.
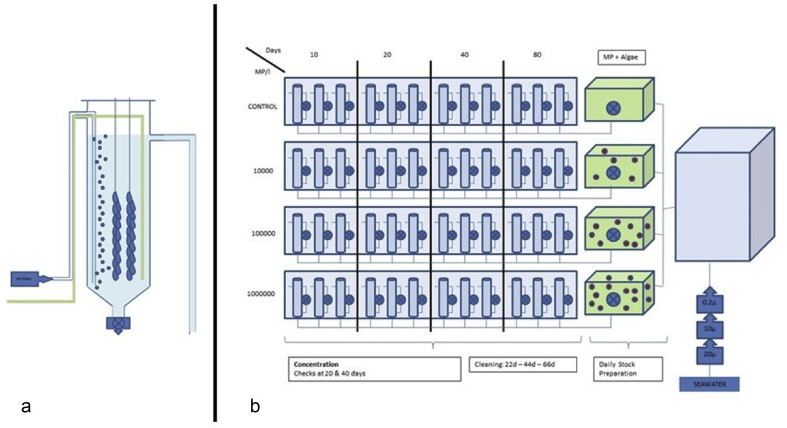
Each of the 4 treatments was replicated 12 times (48 tanks in total) (Figure 1). Each tank contained 30 oysters, 2 glass strips to which 15 juvenile oysters were attached. All 1440 oysters were weighed and measured at the start. At each sampling day (10, 20, 40 and 80) all the animals were removed from the 3 replicate tanks for each treatment and processed (Table B – see supplementary material).
Figure 1.
a. Individual tank setup with 30 juvenile oysters. b. Experimental setup showing the 48 tanks and the PS microbead concentrations, the green tanks represent the stock preparation made daily for each exposure concentration (MP + Algae). All seawater was UV treated and ran through three sequential ceramic filtration units (20μm, 10μm, 0.2μm) before use.
The water temperature was dependent on ambient air temperature, around 18 +/- 2 °C throughout. The oysters were acclimatised in the experimental tanks, under these conditions, for a minimum of 1 day before the start of the study. The following parameters were logged to ensure consistency between the tanks during the entire experiment: temperature (daily), pH (twice weekly; AM Monday & PM Friday), DO2 (twice weekly; AM Monday & PM Friday), Salinity (twice weekly; AM Monday & PM Friday). Light levels were set on a 12h day cycle and total Lux was measured at the end of the study.
All tanks were covered during the entirety of the experiment, access to the laboratory was limited and appropriate laboratory ware (cotton) was worn to avoid contamination from the air. The tanks were manually cleaned on day 22, 44 and 66. Biofilm scrapes from the side of the tank and pseudofaeces were collected and smeared on microscopic slides to determine microbead presence. The wastewater was sand filtered and treated by the facility Ozone plant before discharge. Any other whole animal or tissue waste was discharged as clinical waste and incinerated.
2.4. Sampling procedure
A specific bench area in the biocontainment experimental tank facility was prepared and cleaned before and after sampling. Sampling of oysters was done inside this area to avoid contamination. Oysters were weighed and measured prior to fixing them onto the glass rods. On the selected sampling dates (10, 20, 40 and 80) the oysters were weighed and measured again, after which CI was determined on a subset of 15 oysters (Figure 2). Another sub-selection of 10 oysters was taken to determine LMS (Figure 2). Live samples were taken to the postmortem room and prepared for biomarker assays on site. Tissues were frozen for biomarker analysis, fixed for histology or dried for final dry weight. The digestive gland was removed, embedded in OCT in cryotomes and frozen in liquid nitrogen, after which the blocks were stored at -80C for later analysis. The remaining 5 oysters were used for histology (Figure 2); for this, 8–10μm soft tissue slides were made and stored at -80C.
Figure 2.
Oyster number and sampling procedure, length and wet weight were measured for all oysters, before the experiment and at all sampling points. The abbreviations stand for: LMS = wet weight, shell length, flesh removed and cryopreserved for further lysosomal stability analysis. G- CI = Wet weight, length, dry shell weight, dry tissue weight for Condition Index determination. Histo = Wet weight, shell length, shell removed and tissue fixed in Davidsons for further histology analysis.
2.5. Biological parameters, biomarkers and histology
2.5.1. Shell length & weight
Shell height, the maximum dimension from hinge to growth edge, is commonly referred to as shell length, which will be used to describe this dimension here. The shell length of every oyster was measured to the nearest mm. Additional dimensions were measured to account for irregular oyster shapes (e.g., long and thin). All measurements (±1.0 mm) were taken using a digital calliper system that enabled the rapid recording of data. In the weighing technique, oysters were air dried at room temperature for 5 min and weighed to the nearest 0.0001g. Oyster meat was oven dried to constant weight (68C for 48 h) and then meat and shell were weighed separately to the nearest 0.0001g, after a short cooling period.
2.5.2. Condition index
The CI of bivalves is measured by relating either the weight or volume of the meat to some aspect of the shell. In the current study, oyster shell length and weight measurements were standardized using the following formula: Condition Index = (dry meat weight in g) * 100/(shell weight in g). This widely-used condition index, because of the nature of the measurements involved, is easily standardized and is thus used globally [25]. In addition, the use of dry tissue weights eliminates the bias due to water content fluctuations of whole tissue. A low value for this index indicates that a major biological effort has been expended, either as maintenance energy under poor environmental conditions or disease, or in the production and release of gametes. Thus, as an indicator of stress, or sexual activity, this index gives meaningful information about the physiological state of the animal [26].
2.5.3. Lysosomal membrane stability
A series of solutions and reagents were used to test LMS. A lysosomal membrane labilising buffer (Solution A) was made with 0.1M Na-citrate Buffer - 2.5% NaCl w:v (pH 4.5). The substrate incubation medium (Solution B) consisted of 20 mg of N-Acetyl-β-hexosaminidase (Sigma, N4006) or Napthol AS-BI phosphate (Sigma N2125), dissolved in 2.5 mL of 2-methoxyethanol (Merck, 859) and made up to 50 mL with solution A. This solution contained 3.5 g of collagen-derived polypeptide (POLYPEP, P5115 Sigma) as low viscosity polypeptide to act as a section stabiliser. This solution was prepared 5 min before use. The diazoniumdye (Solution C) contained 0.1M Na-phosphate buffer (pH 7.4) containing 1 mg mL−1 of diazonium dye Fast Violet B salts (Sigma, F1631). The fixative (Solution D) was made from Baker's calcium formol containing 2.5% NaCl (w:v). An aqueous mounting medium (Vector Laboratories H1000, Kaiser glycerine gelatine, Difco, Sigma) was used.
The lysosomal membrane stability was cytochemically determined using N-Acetyl-β-hexosaminidase [27, 28, 29]. Cryostat sections were cut at 8–10μm (in duplicate on the same slide) and left in the cryostat chamber until just before use. Seven slides were prepared in this manner. Solution A was placed into a water bath at 37 °C to acclimatise. The slides were placed into pre-treatment solution A so that each slide had a different pre-treatment time of 30, 25, 20, 15, 10, 5, and 2 min i.e. slide 7 = 30 min, slide 6 = 25 min, slide 5 = 20 min, etc. Following pre-treatment, slides were transferred to solution B for 20 min at 37 °C in a staining jar in a shaking water-bath. The slides were rinsed with a saline solution (3.0% NaCl) at 37 °C for 2–3 min. The slides were then transferred to solution C at room temperature for 10 min. Following this, slides were rinsed rapidly in running tap water for 5 min. Sections were fixed for 10 min in Solution D pre-cooled to 4 °C. Finally, slides were rinsed in distilled water, mounted in aqueous mounting medium and analysed.
The labilisation period (LP) is the time of pre-treatment required to labilise the lysosomal membranes fully, resulting in maximal staining intensity for the enzyme being assayed. The staining intensity was assessed visually using microscopic examination. The labilisation period can be effectively measured by microscopic assessment of the maximum staining intensity in the pre-treatment series, a microdensitometer is not completely necessary for accurate determination. All assessments were carried out on duplicate sections for each digestive gland at each pre-treatment time. Lysosomes will stain reddish-purple due to the reactivity of the substrate with N-acetyl-β-hexosaminidase. The LP for each section corresponds to the average incubation time in the acid buffer that produces maximal staining reactivity. LP for the other replicate is similarly obtained. Finally, a mean value of LMS of the sample was calculated utilizing the data obtained from the 10 animals analysed [27].
Determination of the LP is usually quite straightforward, but a complicating situation occasionally arises in which the pre-treatment series shows two peaks of staining intensity, possibly due to differential latent properties of the subpopulations of lysosomes. In this situation, the first peak of activity was used to determine labilisation period, as it is the most responsive to staining [27].
2.5.4. Histology
Histological analyses were conducted on paraffin-embedded tissues sectioned at 8–10μm thickness and stained using a pentachrome staining procedure to determine the prevalence and intensity of the fluorescent PS microbeads by histological examination. Slides were examined using a Nikon Eclipse E800 microscope equipped with fluorescent filters. Images were captured using the Lim Lucia G Screen Measurement™ image analysis system (Nikon, UK) and Nikon DXM1200F video camera. The microbeads used in this experiment are suitable for fluorescence microscopy and yield intense fluorescence. Microscopic viewing using a 475–490nm filter shows an extremely bright red fluorescence, while use of a 545–610nm filter yields a yellow fluorescence with excitation maxima of 491nm and 512nm and emission maxima at 554nm. The main aim was to confirm the uptake and presence of the microbeads but, where possible, the occurrence and extent of tissue pathologies, and the intensity of anomalities were recorded using quantitative or semi-quantitative measures. Measures of prevalence or occurrence, however, do not give a true indication of the health of an organism [30].
2.5.4.1. Statistics
The statistical importance of the apparent difference of Condition Index, Shell Length and Shell Weight were tested by fitting linear mixed models. These were fitted using the lmer function in the R package lme4. Details of the precise models fitted are shown in the Results section. When investigating the dead oysters, because of the low numbers, the deaths were not modelled with mixed models as above. Instead, Fisher's exact test [31] was used based on two-way contingency tables of treatments vs the control.
Mixed models, as used for the CI analysis, were not used for the LMS data because the LMS scores could take only one of eight different values (including zero for the dead ones). In addition, oysters within a tank often had similar LMS scores and so a normally distributed random error – or indeed a tank random effect - wasn't appropriate. A priori, a central interest is in comparing the LMS for the control and the treatment groups. Thus, we performed our comparisons by comparing the tank means of the control against each of the three treatment levels. This allowed us to compare groups where each contained ten tank means. We performed two-sided, non-parametric randomisation tests of the mean levels, using the permute.groups function in the R library emon [32].
3. Results
3.1. PS MP concentrations
All experimental parameters remained stable and within acceptable limits for optimal oyster cultivation over the entire period: daily temperature (18 +/- 2 °C), pH (8.1), DO2 (~7.0 mg L−1) and Salinity (32–35‰). Light levels mimicked a normal day cycle. All the collected water samples, biofilm glass tank scrapes, pseudofaeces and faeces contained PS microbeads. Microplastic concentrations in the water column appeared to be much lower than expected (1 × 104 particle L−1, 1 × 105 particle L−1, 1 × 106 particle L-1). On average, the detected concentrations of microplastics in the water were a factor 10 lower from the start onwards and dropped to about 1000 times lower 24 h later across all exposures. A similar effect, although much lower, was observed in the tanks containing no oysters (concentration dropped on average with a factor 100 after 24 h) and almost no difference was observed in the tanks containing only seawater and PS microbeads (concentration dropped on average with a factor 10 after 24 h). These concentrations drops are most likely the result of the removal via the oysters, algae and biofilms and the static interaction between microbeads. Microbeads were clearly present in the scrapes from the glass ware and in the pseudofaeces and faeces.
3.2. Uptake of PS microbeads
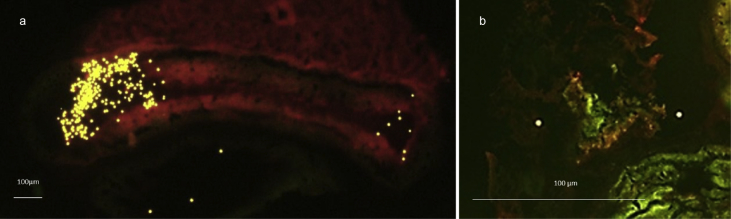
From histological analysis, microbeads were detected in the intestines of exposed oysters (Figure 3a) and in the digestive tubules (Figure 3b). No cellular inflammatory features, including granulomas were observed in exposed animals. No microbeads were observed in control oysters.
Figure 3.
Histology slides of the oyster intestines (a) and digestive tubules (b) showing the fluorescent PS microbeads (yellow) excited at 491 & 512nm.
3.3. Effects of exposure to PS microbeads
The effects of a range of microbead concentrations on growing juvenile oyster across the 80-day period were determined using various measurements and endpoints: Condition Index, Shell Length, Weight, Lysosomal Membrane Stability and Mortality.
3.4. Condition index
Four observations were excluded because of missing information. This left 716 observations. Of these, 22 were for dead oysters and 694 for live ones. For the first part of the analysis below, only the live oysters were used. The numbers of dead oysters were analysed separately.
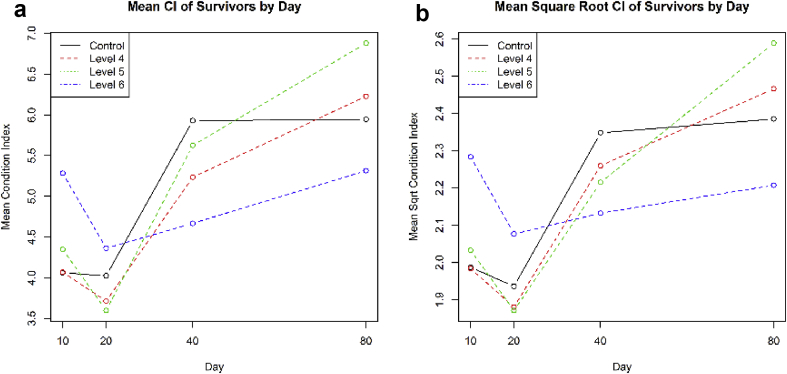
The mean CI was plotted by treatment and day. This is shown in Figure 4a. The plot suggests that there is little noticeable difference between the means for the control and two lowest MP exposure concentrations. However, the mean of the highest exposure concentration is initially the highest (days 10 and 20) but then becomes the lowest (days 40 and 80). This is perhaps even more clearly illustrated in Figure 4b, which has the CI transformed by square root (this transformation will downplay the influence of some of the extreme, high CI values).
Figure 4.
a. Plot of CI means by treatment and day; b. Plot of CI means of square root CI, by treatment and day.
The statistical importance of the apparent difference between the highest exposure concentration and the other ones was tested by fitting linear mixed models. Initially, the full model was fitted
| (1) |
where SCI = CI0.5, TREAT is a factor representing a fixed effect of microbead concentration, DAY is a factor representing a fixed effect due to the duration (0, 10, 20, 40, 80 days) of the experiment, TANK is a random effect and ROD|TANK is a random effect of ROD, nested within TANK. For model comparisons, parameter estimates were obtained by maximum likelihood.
A new treatment factor (TREAT2) was created which contained a single value if the treatment was Control, 104 particles L−1 or 105 particles L−1 and a second value if it was 106 particles L−1. Thus, the new factor has two levels: 106 particles L−1 and ‘the other exposure concentrations’. A similar model to (1) was fitted of the form
| (2) |
but with TREAT2 replacing TREAT. When comparing the fit of models (1) and (2) using a likelihood ratio test we obtain a p-value of 0.88, suggesting little difference between the models. Thus, our modelling suggests that the lowest three levels of MP are behaving similarly.
We now turn to assessing whether the two treatment levels defined by TREAT2 are different. We do this by fitting a model without a treatment effect
| (3) |
and then comparing its fit with model (2). That is, how important is it to distinguish the two treatment levels or can we assume that there is no difference between the treatments? When models (2) and (3) are compared using a likelihood ratio test we get a larger difference in the log-likelihood than before and a p-value of 0.006. Thus, this gives statistical evidence that highest exposure (106 particles L−1) is acting differently to the other exposures. From observation of Figures 4a and 4b, this difference manifests itself in increased growth for the highest exposure treatment for days 10 and 20 but then reduced growth for days 40 and 80.
The analysis above was done on oysters that were alive, however, we also found some dead oysters at the different sampling points. Table 1 below shows the number of deaths by day and treatment. It should be noted that these numbers are all relatively small compared to the 716 oysters analysed for CI. However, the results are interesting in that they show that there were more deaths for the highest MP concentration and, perhaps not surprisingly, there were more deaths on day 80 than the other days.
Table 1.
Number of deaths by day and treatment (out of 716 oysters).
| Treat | DAY |
||||
|---|---|---|---|---|---|
| 10 | 20 | 40 | 80 | Total | |
| Control | 2 | 0 | 1 | 2 | 5 |
| 104 particles L−1 | 1 | 0 | 2 | 0 | 3 |
| 105 particles L−1 | 0 | 1 | 1 | 0 | 2 |
| 106 particles L−1 | 1 | 3 | 1 | 7 | 12 |
| Total | 4 | 4 | 5 | 9 | 22 |
Because of the low numbers, we did not model the deaths with formal models as above. However, we did consider two-way contingency tables of highest MP concentration vs the control (Table 2a) and highest concentration vs the other treatments (Table 2b). As with the modelling above, we need to be careful with implicit multiple comparison tests because we have, to some extent, used the data to guide our testing. Having said that, a priori, we might expect to be comparing the highest levels of MP against either the control or the lower treatment levels.
Table 2a.
Number alive and dead by treatment 106 particles L−1 and control.
| 106 particles L−1 | Control | Total | |
|---|---|---|---|
| Dead | 12 | 5 | 17 |
| Alive | 167 | 174 | 341 |
| Total | 179 | 179 | 358 |
Table 2b.
Number alive and dead by treatment 106 particles L−1 and the other three treatments.
| 106 particles L−1 | Control, 104 particles L−1, 105 particles L−1 | Total | |
|---|---|---|---|
| Dead | 12 | 10 | 22 |
| Alive | 167 | 527 | 694 |
| Total | 179 | 537 | 716 |
For the two-way contingency tables, Fisher's exact test was used to investigate whether there were more deaths from the 106 particles L−1 concentration than there were from the (i) control and (ii) control, 104 particles L−1, 105 particles L−1 treatments (Tables 2a and 2b). For comparison (i), p = 0.13 if we assume an alternative hypothesis that 106 particles L−1 is different to the control and p = 0.07 if we assume that 106 particles L−1 would result in greater deaths than the control. For (ii), corresponding p-values are 0.004 and 0.002 respectively. Thus, whilst there is a suggestion that there are more deaths for 106 particles L−1 than for the control, the small numbers mean that any difference is not quite statistically significant. However, when comparing the 106 particles L−1 with the larger group of treatments, we easily attain the 5% level of statistical significance – suggesting a greater probability of death at 106 particles L−1 than for the other three treatments.
Overall, analysis of the CI data provides strong evidence that the highest PS microbead concentration is having a different/adverse effect on oysters.
3.5. Analysis of shell length and weight
Only live oysters were analysed. There were originally 2,873 data points, 1,440 length and weight values from the oysters at the start of the experiment and 360 data values for the length and weight of the oysters at each of days 10, 20, 40 and 80 (7 values were excluded due to data oddities). At the end of the experiment, 27 oysters were found dead and 1,406 alive.
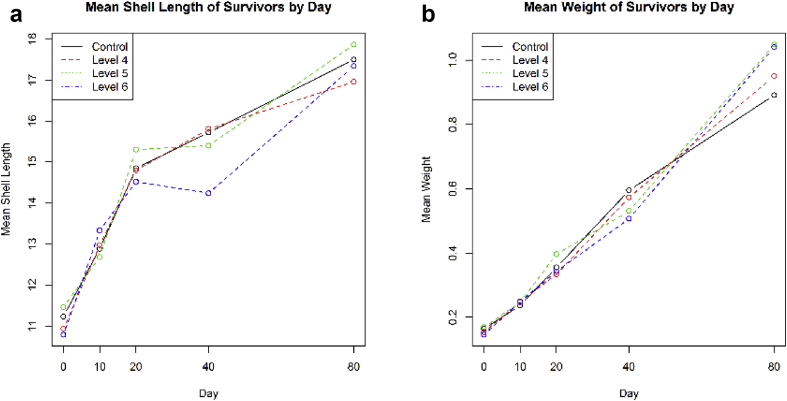
The plot of the shell length means is shown in Figure 5a. There is no obvious pattern amongst the treatments – apart from a reduction for the mean shell length for the highest concentration at day 40, but this is not continued at day 80. Using mixed models of the form in (1) and with the square root of shell length as the dependent variable, likelihood ratio tests confirm that there is no statistically significant interaction between DAY and TREAT (p = 0.18), that there is a statistically significant effect of DAY (p < 0.001) and the effect is close to statistical significance for TREAT (p = 0.052).
Figure 5.
a. Plot of means of shell length by treatment and day; b. Plot of means of weight by treatment and day (the figures are for survivors only).
The plot of the weight means by day and treatment is shown in Figure 5b. Formal statistical modelling suggests that both the DAY by TREAT interaction, the DAY effect and the TREAT main effect are statistically significant (p = 0.007, p < 0.001 and 0.015 respectively).
3.6. Analysis of lysosomal membrane stability
An LMS score was obtained for 342 out of the 480 oysters, there were 14 dead oysters and 124 oysters for which no LMS score could be obtained due to sampling or analytical issues. If we consider that the 14 confirmed dead oysters should have a score of 0, then there are scores for 356 oysters.
Figure 6a shows the mean LMS scores for the 342 alive oysters by treatment and day. There is no obvious pattern with respect to treatment dose here. It is interesting to see that the mean for the highest microbead exposure is always less than the control mean and that the mean for the highest microbead exposure has the lowest mean for the first three measured periods. Figure 6b shows a similar plot to that in Figure 6a, except that in Figure 6b the dead oysters are included, with their LMS scores of 0. This figure perhaps creates a clearer picture in that the mean for the highest MP exposure has the lowest mean score throughout the experiment, tentatively suggesting that the higher dose of microbeads is having a detrimental effect on the lysomal membrane stability.
Figure 6.
a. Mean LMS score of survivors by day; b. Mean LMS of all oysters (dead & alive) by day.
For the analysis excluding and including the dead oysters, the comparison results are shown in Table 3. These confirm the impression given by Figure 6, that the tanks with the highest MP concentration have lower LMS scores than the control tanks (p-value = 0.017).
Table 3.
P-values for comparing LMS score tank means for control and each level of treatment dose. Comparisons have been done twice, without and with the 14 dead oysters.
| Comparison | p-value: Alive oysters only | p-value: Alive and 14 dead oysters |
|---|---|---|
| Control vs Dose 4 | 0.86 | 0.74 |
| Control vs Dose 5 | 0.29 | 0.35 |
| Control vs Dose 6 | 0.34 | 0.017 |
4. Discussion
Our results showed that oysters will accumulate MPs from the water column. Shellfish containing MPs consumed whole not only pose concern for human exposure [3, 9, 11, 13, 33], but also for the animals themselves. The highest PS microbead concentration in our study has been found to increase mortality amongst juvenile oysters. Other studies have shown that exposure to relatively high densities of MPs alter the respiration rates, immunology, reproductive capacity and filtration rates of bivalves [15, 34, 35, 36, 37]. Owing to their role as ecosystem engineers (e.g. reef formation, benthic – pelagic coupling, biodepositioning), such effects are likely to permeate beyond the individual organism into benthic and pelagic food webs [36]. If MPs alter the ability of these filter feeders, there may be wider impacts on their associated communities and on the functioning of coastal ecosystems [35]. More studies are urgently needed to determine the effects of MPs on these key marine species and habitats.
In this study, the MPs deployed were hard and smooth microbeads; however, actual MPs can have any form or shape. To date, there are few peer-reviewed publications on suspected microbeads collected in the field. We could not immediately observe a practically meaningful effect of PS microbeads on the length and weight of the individual oysters. In the same way, Cole et al. [38] found no measurable effects on the development or feeding capacity of oyster larvae exposed to plastic concentrations exceeding those observed in the marine environment. The condition index data provided evidence that the highest PS microbead concentration resulted in lower mean CI levels when compared to the lower treatment levels. These data also showed that a disproportionate number of oysters in the highest dose group die compared to the other groups.
Our results also indicate that the highest dose of microbeads is having a detrimental effect on the lysomal membrane stability. Lysosomes are responsible for the breakdown of all the constituents of the cells and macromolecules derived from the extracellular space via endocytosis [39]. They are also involved in cell defense mechanisms, in the protection against the toxic agents and infection by viruses and bacteria [39, 40]. The physicochemical modifications which lead to the loss of the integrity of the membranes of diverse components of the lysosomes are almost always associated with cellular dysfunction, inflammatory and degenerative diseases as well as apoptosis and cell death [39, 41]. These findings suggest that environmental concentrations of MPs are harmful to the wellbeing of oysters in the long term.
We found no histological evidence of damage to the digestive tissue structures, suggesting that these low concentrations of PS microbeads do not provoke any inflammatory reactions. It is difficult to define the exact underlying mechanisms from the selected endpoints in this study, but other studies have highlighted that PS microbeads in high-dose, short-term experiments caused feeding modifications and reproductive disruption in oysters, with significant impacts on offspring. Dynamic energy budget modeling, supported by transcriptomic profiles, suggested a significant shift of energy allocation from reproduction to structural growth, and elevated maintenance costs in exposed oysters, which is thought to be caused by interference with energy uptake. Molecular signatures of endocrine disruption were also revealed, but no endocrine disruptors were found in the biological samples [15]. In a study by Ribeiro et al. [16] the effects of PS MPs were assessed in tissues of the clam Scrobicularia plana. Clams were exposed for 14 days to 4 particles mL−1, comparable to our lowest (104 particles L−1) concentration, followed by 7 days of depuration. The results revealed that MPs caused reduced antioxidant capacity, DNA damage, neurotoxicity and oxidative damage [16]. A two-month study of the black-lip pearl oyster, P. margaritifera, has shown that environmental concentrations of PS microbeads significantly impact the assimilation efficiency and more broadly the energy balance, with negative repercussions on reproduction. Gonads may have provided the missing energy to maintain animals’ metabolism through the production of metabolites derived from germ cells phagocytosis [17].
Our data shows little noticeable difference between the CI of the oysters in the control and those in the two lowest MP concentrations (104 particles L−1 & 105 particles L−1). Similar results were observed in other bivalve studies using low concentrations of PS microparticles (4 particles mL−1), where no statistically significant changes were observed between control and exposed clams (S. plana) after 14 days and in the 7day elimination period. A low value for this index indicates that a major biological effort has been expended, either as maintenance energy under poor environmental conditions. The oysters dosed with the highest concentrations (106 particles L−1) were, however, in a better condition than the oysters in all other treatments for days 10 and 20 but then their condition plummeted for days 40 and 80. We can only speculate on the reasons for this. Initially, the oysters seem to be boosted by MPs and it is only later in time that adverse health effects due to the high MP diet may manifest. This might be related to a higher filtration rate. The European flat oyster, O. edulis, exposed for 2 h per day to MPs filters more algae h-1 than without MPs [36]. Likewise, an increase in filtration rates of the Pacific oyster was found in response to constant exposure to 6 μm PS microbeads [15]. This suggests that oysters filter more in response to plastic particles.
A study in clams, Atactodea striata, has shown that ingestion and retention of MPs were limited by the production of pseudofaeces and faeces [42]. We detected similar stress effects and found pseudofaeces containing high amounts of microbeads, a mechanism known to be a cleaning mechanism, preventing the gills being blocked by particulate matter [43], and as a rejection mechanism for inedible particles [44]. We found rather low amounts of PS microbeads in the faeces. In a similar study, the detoxification of PS MPs in clam tissues was inefficient for the 7 day duration tested [16]. Although this indicates that oysters have the ability to egest MPs via faeces, there is still potential for accumulation and trophic transfer [16, 45] and/or effects of long-term exposure [46].
Continuous augmented filtration without improved food uptake may lead to biomass losses in the long term. Green et al. [35] reported that the biomass of the peppery furrow shell clam, Scrobicularia plana was ~1.5 times lower in mesocosms with the high dose of MPs compared to controls. This indicates that repeated exposure to high concentrations of MPs may lead to “MP fatigue” in oysters, altering the condition of important ecosystem engineers and the formation of benthic assemblages.
All polymer particles with a diameter between 0.1 μm and 5mm are defined as microplastics. This creates several issues in relation to microplastic sampling, analytical and reporting procedures. Most field studies include only particles in a narrower range as microplastic, often determined by their sampling methodology or the detection limit of devices they used [24]. A commonly used lower limit due to mesh size lies between 300 – 800 mm, while the upper limit is often set between 2.16 and 4.75mm or up to 5mm [47]. Likewise, studies differ as to whether all particle shapes are included, distinguishing between fragments, spheres, sheets, pellets, ropes and fibers [24]. The present literature also reports highly variable metrics of concentration, such as averages, medians, maximum averages, average maxima and maxima [24]. To make matters worse, several studies report microplastics in different units. Microplastic concentrations are variably reported as mass or as particle numbers per mass, per volume or per surface area of water or sediment, or even per study site [24]. These differences in units and lack of complete quantification make it difficult to determine realistic concentrations. It makes comparisons between field observations very difficult and limits their usefulness for ecotoxicological experiments. Globally, the highest reported microplastic concentrations in the water column using a mesh size of ~300 μm is 102 particles L-1 [24,48], measured near a harbour, close to a polymer production plant. Up to 100 000 times higher concentrations of small plastic fibres were retained on a 80μm mesh compared to a 450μm mesh [48]. Estuarine studies in South Korea reported high MP concentrations up to 23 particles L−1 between 0.2 and 1 mm in contaminated regions [49]. Applying smaller mesh sizes will retain a larger fraction of MPs [50]. The limits set in these field studies thus result in microplastic numbers being underestimated compared to the definition. Furthermore, concentrations of microplastics in the water column are known to be very heterogenous and variable [51]. For example, the abundance of plastic particles in the water column increased 6-fold shortly after a storm in California coastal waters [52]. Taking into account that amounts of microplastic are also underestimated by up to a factor of 30 when based on surface sampling [53], microplastic concentrations, especially the smallest fraction, might be much higher in reality and present a risk to the most sensitive species at hotspot locations in near-shore regions. Microplastics in sediment are also expected to affect organisms feeding in the water column, via resuspension or transfer through the food chain [24]. Due to increased water turbulence or defouling, originally settled plastic particles are expected to become resuspended in the water column (especially in shallow and near shore environments) and lead to exposure of organisms feeding of the water column [54]. Considering size distributions of particles, it is clear that abundance increases with a decrease in size probably due to fragmentation processes [55]. Just based on mass conservation principles, fragmentation of spherical microplastic particles with a size of >0.1 μm – 5mm into 100nm nanoplastic particles would lead to particle concentrations that are ultimately >1014 times higher than the currently found microplastic particle concentrations [24]. Detecting these smaller fractions of microplastics (<10μm) proves rather problematic and costly with current methodologies and are thus often overlooked and unreported.
Considering the above, we exposed juvenile oysters for almost three-months to what the authors believe to be a series of potential environmental concentrations of a prototypical MP of that size. The concentrations of 6 μm PS microbeads in this study, although seemingly high, could well represent a range of potential scenarios for current, short-term and long-term concentrations of microplastics to which invertebrates might get exposed in the environment. Furthermore, the PS microbead concentrations in our tanks quickly dropped off, especially in the presence of oysters (about a factor 1000 lower after 24h), leading to a steady state concentration over 24h, the situation where the overall input of MPs is fairly in dynamic equilibrium with their elimination via the oysters uptake and removal in the form of faeces and pseudofaeces, interaction with algae and biofilms and/or static clumping.
Most importantly, we observed an increased mortality in oysters exposed chronically for 80 days to 6 μm PS microbeads, dosed at concentrations of 106 particles L−1. Such concentrations are currently not frequently reported in the marine environment but could be found near inputs such as harbours [48], rivers [23], sewage outlets [56] or estuaries [49]. The biological responses and increased mortality, however, seem rather specific to MP and less distinguished in bivalves exposed to suspended sediment plumes [57]. More research, detailing diverse experimental setups, testing different endpoints in a wide range of marine key species and ecosystems, including studies combining realistic mixtures of polymer types and different stressors (e.g. temperature increase, ocean acidification, contaminant & microbiological load) are all needed to allow for future comparisons and greater insights.
Declarations
Author contribution statement
Thomas Maes: Conceived and designed the experiments; Performed the experiments; Analyzed and interpreted the data; Contributed reagents, materials, analysis tools or data; Wrote the paper.
Jon Barry: Analyzed and interpreted the data.
Craig Stenton, Edward Roberts, Ruth Hicks: Performed the experiments.
John Bignell: Contributed reagents, materials, analysis tools or data.
Dick Vethaak, Heather Leslie, Matthew Sanders: Conceived and designed the experiments.
Funding statement
This work was supported by the MICRO EU Interreg-funded project (MICRO 09-002-BE), the OceanWise (EAPA 353/2016) European Regional Development Fund (ERDF) INTERREG Atlantic Area, Priority Axis 4 project and the UK DEFRA projects SLA26, ME5415 and ME5425.
Competing interest statement
The authors declare no conflict of interest.
Additional information
Data associated with this study has been deposited at Cefas under the accession number https://doi.org/10.14466/CefasDataHub.91.
Supplementary content related to this article has been published online at https://doi.org/10.1016/j.heliyon.2019.e03103.
Acknowledgements
We thank Marieke Desender, John Thain, Brett Lyons, Grant Stentiford, Ioanna Katsiadaki, Matthew Green, Michelle Fox, Ewen Bell, Chris Firmin, Myra Van der Meulen, Lisa Devriese, Ika Paul-Pont, Philippe Soudant, Arnaud Huvet, Rossana Sussarellu, Christophe Lambert, David Mazurais and Johan Robbens for technical assistance and advice.
Appendix A. Supplementary data
The following is the supplementary data related to this article:
References
- 1.GESAMP Sources, fate and effects of microplastics in the marine environment: a global assessment. Rep. Stud. - Jt. Group Experts Sci. Aspects Mar. Pollut. (GESAMP) 2015;90 [Google Scholar]
- 2.Avio C.G., Gorbi S., Regoli F. Plastics and microplastics in the oceans: from emerging pollutants to emerged threat. Mar. Environ. Res. 2017 doi: 10.1016/j.marenvres.2016.05.012. [DOI] [PubMed] [Google Scholar]
- 3.Van Cauwenberghe L., Janssen C.R. Microplastics in bivalves cultured for human consumption. Environ. Pollut. 2014;193:65–70. doi: 10.1016/j.envpol.2014.06.010. [DOI] [PubMed] [Google Scholar]
- 4.Van Cauwenberghe L., Claessens M., Vandegehuchte M.B., Janssen C.R. Microplastics are taken up by mussels (Mytilus edulis) and lugworms (Arenicola marina) living in natural habitats. Environ. Pollut. 2015;199:10–17. doi: 10.1016/j.envpol.2015.01.008. [DOI] [PubMed] [Google Scholar]
- 5.Setälä O., Norkko J., Lehtiniemi M. Feeding type affects microplastic ingestion in a coastal invertebrate community. Mar. Pollut. Bull. 2016;102:95–101. doi: 10.1016/j.marpolbul.2015.11.053. [DOI] [PubMed] [Google Scholar]
- 6.Von Moos N., Burkhardt-Holm P., Köhler A. Uptake and effects of microplastics on cells and tissue of the blue mussel Mytilus edulis L. after an experimental exposure. Environ. Sci. Technol. 2012 doi: 10.1021/es302332w. [DOI] [PubMed] [Google Scholar]
- 7.Li H.X. Microplastics in oysters Saccostrea cucullata along the Pearl River estuary, China. Environ. Pollut. 2018 doi: 10.1016/j.envpol.2018.01.083. [DOI] [PubMed] [Google Scholar]
- 8.Li J., Yang D., Li L., Jabeen K., Shi H. Microplastics in commercial bivalves from China. Environ. Pollut. 2015 doi: 10.1016/j.envpol.2015.09.018. [DOI] [PubMed] [Google Scholar]
- 9.Smith M., Love D.C., Rochman C.M., Neff R.A. Microplastics in seafood and the implications for human health. Curr. Environ. Heal. Rep. 2018 doi: 10.1007/s40572-018-0206-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rochman C.M. Anthropogenic debris in seafood: plastic debris and fibers from textiles in fish and bivalves sold for human consumption. Sci. Rep. 2015 doi: 10.1038/srep14340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Barboza L.G.A., Dick Vethaak A., Lavorante B.R.B.O., Lundebye A.K., Guilhermino L. Marine microplastic debris: an emerging issue for food security, food safety and human health. Mar. Pollut. Bull. 2018 doi: 10.1016/j.marpolbul.2018.05.047. [DOI] [PubMed] [Google Scholar]
- 12.Vethaak A.D. Integrated indicator framework and methodology for monitoring and assessment of hazardous substances and their effects in the marine environment. Mar. Environ. Res. 2017;124 doi: 10.1016/j.marenvres.2015.09.010. [DOI] [PubMed] [Google Scholar]
- 13.Li J. Microplastics in mussels along the coastal waters of China. Environ. Pollut. 2016 doi: 10.1016/j.envpol.2016.04.012. [DOI] [PubMed] [Google Scholar]
- 14.Browne M.A., Dissanayake A., Galloway T.S., Lowe D.M., Thompson R.C. Ingested microscopic plastic translocates to the circulatory system of the mussel, Mytilus edulis (L.) Environ. Sci. Technol. 2008;42:5026–5031. doi: 10.1021/es800249a. [DOI] [PubMed] [Google Scholar]
- 15.Sussarellu R. Oyster reproduction is affected by exposure to polystyrene microplastics. Proc. Natl. Acad. Sci. 2016 doi: 10.1073/pnas.1519019113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ribeiro F. Microplastics effects in Scrobicularia plana. Mar. Pollut. Bull. 2017 doi: 10.1016/j.marpolbul.2017.06.078. [DOI] [PubMed] [Google Scholar]
- 17.Gardon T., Reisser C., Soyez C., Quillien V., Le Moullac G. Microplastics affect energy balance and gametogenesis in the pearl oyster Pinctada margaritifera. Environ. Sci. Technol. 2018;52:5277–5286. doi: 10.1021/acs.est.8b00168. [DOI] [PubMed] [Google Scholar]
- 18.Cole M., Galloway T.S. Ingestion of nanoplastics and microplastics by pacific oyster larvae. Environ. Sci. Technol. 2015 doi: 10.1021/acs.est.5b04099. [DOI] [PubMed] [Google Scholar]
- 19.Lenz R., Enders K., Nielsen T.G. Microplastic exposure studies should be environmentally realistic. Proc. Natl. Acad. Sci. 2016 doi: 10.1073/pnas.1606615113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Shatkin G., Shumway S.E., Hawes R. Considerations regarding the possible introduction of the pacific oyster (Crassostrea gigas) to the Gulf of Maine: a review of global experience. J. Shellfish Res. 1997 [Google Scholar]
- 21.FAO . FAO; 2018. FAO yearbook. Fisheries and Aquaculture Statistics 2016/FAO annuaire. Statistiques des pêches et de l’aquaculture 2016/FAO anuario. Estadísticas de pesca y acuicultura 2016. [Google Scholar]
- 22.Erni-Cassola G., Gibson M.I., Thompson R.C., Christie-Oleza J.A. Lost, but found with nile red: a novel method for detecting and quantifying small microplastics (1 mm to 20 μm) in environmental samples. Environ. Sci. Technol. 2017;51:13641–13648. doi: 10.1021/acs.est.7b04512. [DOI] [PubMed] [Google Scholar]
- 23.Siegfried M., Koelmans A.A., Besseling E., Kroeze C. Export of microplastics from land to sea. A modelling approach. Water Res. 2017;127:249–257. doi: 10.1016/j.watres.2017.10.011. [DOI] [PubMed] [Google Scholar]
- 24.Besseling E., Redondo-Hasselerharm P., Foekema E.M., Koelmans A.A. Quantifying ecological risks of aquatic micro- and nanoplastic. Crit. Rev. Environ. Sci. Technol. 2019;49:32–80. [Google Scholar]
- 25.Lawrence D.R., Scott G.I. The determination and use of condition index of oysters. Estuaries. 1982 [Google Scholar]
- 26.Lucas A., Beninger P.G. The use of physiological condition indices in marine bivalve aquaculture. Aquaculture. 1985 [Google Scholar]
- 27.Moore M.N. Cytochemical demonstration of latency of lysosomal hydrolases in digestive cells of the common mussel, Mytilus edulis, and changes induced by thermal stress. Cell Tissue Res. 1976 doi: 10.1007/BF00218706. [DOI] [PubMed] [Google Scholar]
- 28.Moore M.N., Viarengo A. Lysosomal membrane fragility and catabolism of cytosolic proteins: evidence for a direct relationship. Experientia. 1987 doi: 10.1007/BF01945568. [DOI] [PubMed] [Google Scholar]
- 29.Bayne B.L., Livingstone D.R., Moore M.N., Widdows J. A cytochemical and a biochemical index of stress in Mytilus edulis L. Mar. Pollut. Bull. 1976 [Google Scholar]
- 30.Wilson-Ormond E.A., Ellis M.S., Powell E.N., Kim Y., Li S.I. Effects of gas-producing platforms on continental shelf megafauna in the northwest Gulf of Mexico: reproductive status and health. Int. Rev. Hydrobiol. 2000 [Google Scholar]
- 31.Fisher R.A. On the interpretation of χ 2 from contingency tables, and the calculation of P. J. R. Stat. Soc. 1922;85:87. [Google Scholar]
- 32.Barry J., Maxwell D., Jennings S., Walker D., Murray J. Emon. An R-package to support the design of marine ecological and environmental studies, surveys and monitoring programmes. Meth. Ecol. Evol. 2017 [Google Scholar]
- 33.Devriese L.I. Microplastic contamination in brown shrimp (Crangon crangon, Linnaeus 1758) from coastal waters of the southern north sea and channel area. Mar. Pollut. Bull. 2015;98:179–187. doi: 10.1016/j.marpolbul.2015.06.051. [DOI] [PubMed] [Google Scholar]
- 34.Bean T.P. A review of the tools used for marine monitoring in the UK: combining historic and contemporary methods with modeling and socioeconomics to fulfill legislative needs and scientific ambitions. Front. Mar. Sci. 2017;4 [Google Scholar]
- 35.Green D.S. Effects of microplastics on European flat oysters, Ostrea edulis and their associated benthic communities. Environ. Pollut. 2016 doi: 10.1016/j.envpol.2016.05.043. [DOI] [PubMed] [Google Scholar]
- 36.Green D.S., Boots B., O’Connor N.E., Thompson R. Microplastics affect the ecological functioning of an important biogenic habitat. Environ. Sci. Technol. 2017 doi: 10.1021/acs.est.6b04496. [DOI] [PubMed] [Google Scholar]
- 37.Paul-Pont I. Exposure of marine mussels Mytilus spp. to polystyrene microplastics: toxicity and influence on fluoranthene bioaccumulation. Environ. Pollut. 2016 doi: 10.1016/j.envpol.2016.06.039. [DOI] [PubMed] [Google Scholar]
- 38.Cole M., Lindeque P., Fileman E., Halsband C., Galloway T.S. The impact of polystyrene microplastics on feeding, function and fecundity in the marine copepod Calanus helgolandicus. Environ. Sci. Technol. 2015 doi: 10.1021/es504525u. [DOI] [PubMed] [Google Scholar]
- 39.Nazar M.L., Rodrigues L.E.A., Nascimento I. The lysosomal stability as a biomarker for the determination of pollution in aquatic environments. Braz. Arch. Biol. Technol. 2008;51:1071–1077. [Google Scholar]
- 40.Cuervo A.M., Dice J.F. Age-related decline in chaperone-mediated autophagy. J. Biol. Chem. 2000 doi: 10.1074/jbc.M002102200. [DOI] [PubMed] [Google Scholar]
- 41.van Nierop K. Lysosomal destabilization contributes to apoptosis of germinal center B-lymphocytes. J. Histochem. Cytochem. 2006;54:1425–1435. doi: 10.1369/jhc.6A6967.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Xu X.-Y. Microplastic ingestion reduces energy intake in the clam Atactodea striata. Mar. Pollut. Bull. 2017;124:798–802. doi: 10.1016/j.marpolbul.2016.12.027. [DOI] [PubMed] [Google Scholar]
- 43.Barker Jørgensen C. Feeding and cleaning mechanisms in the suspension feeding bivalve Mytilus edulis. Mar. Biol. 1981 [Google Scholar]
- 44.Wegner A., Besseling E., Foekema E.M., Kamermans P., Koelmans A.A. Effects of nanopolystyrene on the feeding behavior of the blue mussel ( Mytilus edulis L.) Environ. Toxicol. Chem. 2012;31:2490–2497. doi: 10.1002/etc.1984. [DOI] [PubMed] [Google Scholar]
- 45.Farrell P., Nelson K. Trophic level transfer of microplastic: Mytilus edulis (L.) to Carcinus maenas (L.) Environ. Pollut. 2013 doi: 10.1016/j.envpol.2013.01.046. [DOI] [PubMed] [Google Scholar]
- 46.Anderson J.C., Park B.J., Palace V.P. Microplastics in aquatic environments: implications for Canadian ecosystems. Environ. Pollut. 2016;218:269–280. doi: 10.1016/j.envpol.2016.06.074. [DOI] [PubMed] [Google Scholar]
- 47.Lechner A. The Danube so colourful: a potpourri of plastic litter outnumbers fish larvae in Europe’s second largest river. Environ. Pollut. 2014;188:177–181. doi: 10.1016/j.envpol.2014.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Noren F. 2007. Small Plastic Particles in Coastal Swedish Waters. [Google Scholar]
- 49.Song Y.K., Hong S.H., Jang M., Han G.M., Shim W.J. Occurrence and distribution of microplastics in the sea surface microlayer in Jinhae bay, South Korea. Arch. Environ. Contam. Toxicol. 2015;69:279–287. doi: 10.1007/s00244-015-0209-9. [DOI] [PubMed] [Google Scholar]
- 50.Hidalgo-ruz V., Gutow L., Thompson R.C., Thiel M. Microplastics in the marine environment: a review of the methods used for identification and quantification. Environ. Sci. Technol. 2012;46:3060–3075. doi: 10.1021/es2031505. [DOI] [PubMed] [Google Scholar]
- 51.Maes T. Microplastics baseline surveys at the water surface and in sediments of the north-east Atlantic. Front. Mar. Sci. 2017;4:135. [Google Scholar]
- 52.Moore C., Moore S., Weisberg S., Lattin G., Zellers A. A comparison of neustonic plastic and zooplankton abundance in southern California’s coastal waters. Mar. Pollut. Bull. 2002;44:1035–1038. doi: 10.1016/s0025-326x(02)00150-9. [DOI] [PubMed] [Google Scholar]
- 53.Kooi M. The effect of particle properties on the depth profile of buoyant plastics in the ocean. Sci. Rep. 2016;6:33882. doi: 10.1038/srep33882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kooi M., Nes E. H. van, Scheffer M., Koelmans A.A. Ups and downs in the Ocean: effects of biofouling on vertical transport of microplastics. Environ. Sci. Technol. 2017;51:7963–7971. doi: 10.1021/acs.est.6b04702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Eriksen M. Plastic pollution in the world’s oceans: more than 5 trillion plastic pieces weighing over 250,000 tons afloat at sea. PLoS One. 2014;9 doi: 10.1371/journal.pone.0111913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Magnusson K., Norén F., Swedish I.V.L. Screening of microplastic particles in and down-stream a wastewater treatment plant. IVL Swedish Environ. Res. Inst. 2014:1–22. [Google Scholar]
- 57.Wilber D.H., Clarke D.G. Biological effects of suspended sediments: a review of suspended sediment impacts on fish and shellfish with relation to dredging activities in estuaries. N. Am. J. Fish. Manag. 2001 [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.