Abstract
Purpose
Pulmonary complications occur frequently in primary antibody deficiency (PAD). While the impact of antibody deficiency may appear implicit for certain respiratory infections, immunoglobulin replacement therapy does not completely ameliorate pulmonary complications in PAD. Thus, there may be antibody-independent factors influencing susceptibility to respiratory disease in PAD, but these remain incompletely defined.
Methods
We harnessed the multicenter US Immunodeficiency Network primary immunodeficiency registry to compare prevalence of asthma, bronchiectasis, interstitial lung disease (ILD), and respiratory infections between two forms of PAD: common variable immunodeficiency (CVID) and x-linked agammaglobulinemia (XLA). We also defined the clinical and immunological characteristics associated with ILD and asthma in CVID.
Results
Asthma, bronchiectasis, ILD, pneumonia, and upper respiratory infections were more prevalent in CVID than XLA. ILD was associated with autoimmunity, bronchiectasis, and pneumonia as well as fewer B and T cells in CVID. Asthma was the most common chronic pulmonary complication and associated with lower IgA and IgM in CVID. Age of symptom onset or CVID diagnosis was unrelated with ILD or asthma.
Conclusion
Despite having less severe immunoglobulin deficiency than XLA, respiratory infections, ILD, and asthma were more common in CVID. Among CVID patients, ILD was associated with autoimmunity and reduced lymphocytes and asthma with lower immunoglobulins. Though our results are tempered by registry limitations, they provide evidence that factors beyond lack of antibody promote pulmonary complications in PAD. Efforts to understand how genetic etiology, nature of concurrent T cell deficiency, and propensity for autoimmunity shape pulmonary disease may improve treatment of PAD.
Keywords: Primary antibody deficiency, interstitial lung disease, asthma, common variable immunodeficiency, x-linked agammaglobulinemia
Introduction
Pulmonary complications occur frequently in primary antibody deficiency (PAD), with these patients having a heightened risk of acute respiratory infections as well as chronic lung disease [1–3]. While the role of antibody deficiency in susceptibility to infection and bronchiectasis may be inherent, other pulmonary complications, like asthma and interstitial lung disease (ILD), may be a consequence of infectious as well as non-infectious factors [4–7]. Importantly, lung disease remains a frequent manifestation and leading cause of morbidity and mortality in PAD despite immunoglobulin (Ig) replacement therapy for reasons that are incompletely defined [8–12]. Better understanding of the antibody-independent factors that influence the progression of lung disease in PAD may help improve treatment.
Common variable immunodeficiency (CVID) and x-linked agammaglobulinemia (XLA) are distinct forms of PAD that share profound antibody deficiency, requirement for Ig replacement therapy, and susceptibility to lung disease [11–15]. Despite these similarities, differences in the occurrence of some forms of lung disease between CVID and XLA were reported in a single-center study from Iran [16]. Moreover, CVID patients have variable susceptibility to pulmonary disease [17, 18]. Uncovering the reasons why lung disease manifestations vary so greatly among patients with PAD on Ig replacement therapy may help our understanding of the antibody-independent factors that influence pulmonary complications.
Few multicenter studies have been conducted to study factors influencing pulmonary disease in PAD, and these have not focused on the chronic lung complications of asthma or ILD [14, 19, 20]. We harnessed the multicenter US Immunodeficiency Network (USIDNET) registry to compare prevalence of pulmonary complications in CVID and XLA as well as identify associated clinical and laboratory characteristics of asthma and ILD. Our results indicate that pulmonary complications in PAD are shaped by factors that extend beyond antibody deficiency, as acute and chronic lung disease were significantly more common in CVID even though Ig deficiency was more profound in XLA. In CVID, ILD was linked with autoimmune cytopenias and T cell deficiency, while asthma was associated with greater Ig deficiency, despite this complication being less common in XLA. Further exploration of these factors influencing pulmonary complications may improve clinical management of PAD.
Methods
Patient Registry Query
Data were obtained from USIDNET, a program of the Immune Deficiency Foundation, supported by a cooperative agreement, U24AI86837, from the National Institute of Allergy and Infectious Diseases. A query was submitted to the USIDNET registry requesting demographics, age at symptom onset, age at diagnosis, respiratory and autoimmune conditions, Ig levels, complete blood count, and lymphocyte screen for patients with a diagnosis of CVID and XLA. Patients with a diagnosis of CVID were included in the analysis if they had an IgA and/or IgM value which was less than two standard deviations below the standard mean for age [21]. We did not exclude patients with IgG levels within two standard deviations of the laboratory mean since patients may have been on Ig replacement at the time of enrollment. CVID patients included in this study were not linked with a specific genetic diagnosis. For the purpose of this manuscript, the following diagnoses were included as ILD: bronchiolitis obliterans, bronchiolitis obliterans organizing pneumonia, cryptogenic organizing pneumonia, diffuse infiltrative lung disease, fibrosis of lung, follicular bronchiolitis, interstitial lung disease, interstitial pneumonia, lymphoid interstitial pneumonia, multiple hyalinizing granulomas of lung, multiple nodules of lung, pulmonary granuloma, pulmonary nodular lymphoid hyperplasia, pulmonary sarcoidosis, respiratory bronchiolitis-associated interstitial lung disease, restrictive lung disease, and sarcoidosis. The following diagnoses were included as autoimmune disease: Addison’s disease, antiphospholipid syndrome, antisynthetase syndrome, autoimmune encephalitis, autoimmune enteropathy, autoimmune hepatitis, Behcet’s syndrome, celiac disease, Crohn’s disease, dermatomyositis, diabetes mellitus type 1, discoid lupus erythematosus, essential mixed cryoglobulinemia, Graves’ disease, Hashimoto’s thyroiditis, Henoch-Schoenlein purpura, iritis/uveitis, juvenile rheumatoid arthritis, mixed connective tissue disease, psoriasis, myasthenia gravis, optic neuritis, primary biliary sclerosis, Raynaud’s disease, rheumatoid arthritis, Sjogren’s syndrome, systemic lupus erythematosus, systemic sclerosis, ulcerative colitis, vasculitis, and vitiligo,
Statistical Analysis
Statistical analysis was performed using R studio® version 1.1.49 and Microsoft® Excel© 2016. Chi-square test of independence was used to compare categorical values and Mann-Whitney test was used for continuous variables. Odds ratio (unadjusted) was used to compare the prevalence of various respiratory conditions between XLA and CVID patients. Continuous variables were summarized using median (interquartile range, IQR).
Results
Clinical and Laboratory Comparison between USIDNET Patients with CVID and XLA
To determine whether the form of PAD influenced the prevalence of specific pulmonary complications, we compared data from patients with CVID and XLA in the USIDNET registry. Data from 1647 patients with a diagnosis of CVID or XLA were obtained from the registry. Five hundred and seventy-eight patients were excluded because they were documented as cases of CVID without an IgA or IgM less than two standard deviations below the mean for age and thus did not meet consensus guidelines for this diagnosis [22]. Of the 1069 remaining patients, 749 had a diagnosis of CVID and 320 had a diagnosis of XLA. The data were entered from 209 contributors, with a median of two patients entered per contributor (interquartile range 1 to 4).
Differences in patient demographics and immunological lab values between the CVID and XLA cohorts were generally as expected (Table 1). The XLA cohort was entirely male compared to 43% of CVID patients. CVID patients were older, had later symptom onset, were diagnosed later, and had fewer CD4+ and CD8+ T cells than XLA patients (P < 0.001). IgA and IgM as well as circulating CD19+ B cells were all significantly lower in XLA compared to CVID (P < 0.001). Pre-diagnostic IgG levels were also lower in XLA compared to CVID (P = 0.015), but values were only available in 179 with CVID and 11 with XLA. Use of the registry data did not allow us to match the cohorts for length of time of IgG replacement therapy or follow-up.
Table 1.
Clinical and laboratory comparison between patients with CVID and XLA
| CVID N = 749 |
XLA N = 320 |
P value* | |
|---|---|---|---|
| Sex (male) | 322 (43.0) | 320 (100.0) | < 0.001 |
| Age at last visit | 39.50 [24.90, 50.30] | 17.40 [8.43, 28.25] | < 0.001 |
| Age of symptom onset | 16.00 [6.00, 30.00] | 1.00 [0.50, 2.15] | < 0.001 |
| Age of diagnosis | 29.90 [15.20, 42.70] | 2.40 [1.00, 5.60] | < 0.001 |
| CD4 | 664.00 [470.00, 927.00] | 1576.50 [1120.00, 2426.75] | < 0.001 |
| CD8 | 390.00 [246.50, 681.00] | 795.84 [484.56, 1241.10] | < 0.001 |
| CD19 | 176.00 [80.25, 309.25] | 0.00 [0.00, 13.00] | < 0.001 |
| Pre-diagnostic IgG | 353 [186–514] | 65 [25.5–325] | 0.015 |
| IgA | 9.99 [6.64, 31.00] | 6.66 [0.99, 8.14] | < 0.001 |
| IgM | 23.00 [10.99, 44.00] | 10.00 [4.99, 22.00] | < 0.001 |
Chi-square test for categorical variables and Mann-Whitney test for continuous variables
Continuous variables were summarized as median [IQR] and categorical variables were summarized as counts (percentage)
Comparison of Pulmonary Complications between Patients with CVID and XLA
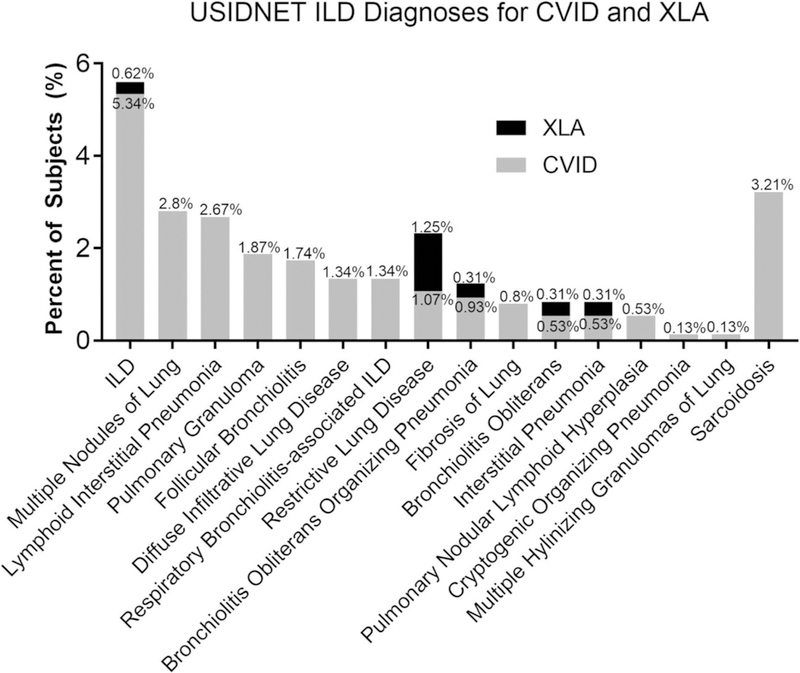
We next compared the prevalence of specific pulmonary complications between the CVID and XLA patients from the USIDNET registry. The prevalence of ILD was significantly higher in patients with CVID compared to XLA (14.3% vs. 2.2%, P < 0.001) with an odds ratio of 7.45 (Table 2). The different subtypes and terminology of lung disease in the USIDNET registry that was included as ILD and their relative proportions are shown in Fig. 1. The general term ILD was the most common designation in CVID (5.34%), with sarcoidosis (3.21%), multiple nodules (2.9%), lymphocytic interstitial pneumonia (LIP) (2.67%), pulmonary granuloma (1.87%), and follicular bronchiolitis (FB) (1.74%) being the other most prevalent ILD diagnoses input into the USIDNET registry for these patients. In XLA, 1.25% were denoted as having restrictive lung disease and 0.62% had the general term ILD associated with them, but none had diagnoses of sarcoidosis, multiple nodules, LIP, pulmonary granuloma, or FB. Asthma was also much more prevalent in patients with CVID (31.2% compared to 10.3% of XLA, P < 0.001) with an odds ratio of 3.95. Additionally, sinusitis was significantly higher in patients with CVID compared to those with XLA (76.9% vs. 48.8%, odds ratio 3.5, P < 0.001) as were other upper respiratory tract infections (56.5% vs. 18.8%, odds ratio 5.62, P < 0.001). Bronchiectasis (13.5% vs. 8.4%, P = 0.02) and pneumonia (63.3% vs. 49.1%, P < 0.001) were also higher in CVID, but the odds ratio was lower for these conditions at 1.68 and 1.79, respectively. Thus, despite the fact that antibody deficiency is more profound in XLA, acute respiratory infections as well as the chronic pulmonary complications of asthma, bronchiectasis, and ILD were all more common in CVID.
Table 2.
Comparison of pulmonary complications between CVID and XLA
| Diagnosis N (%) |
CVID N = 749 |
XLA N = 320 |
OR | 95% CI | P value |
|---|---|---|---|---|---|
| ILD (%) | 107 (14.3) | 7 (2.2) | 7.45 | (3.4–16.2) | < 0.001 |
| URT (%) | 423 (56.5) | 60 (18.8) | 5.62 | (4.1–7.7) | < 0.001 |
| Sinusitis (%) | 576 (76.9) | 156 (48.8) | 3.5 | (2.7–4.6) | < 0.001 |
| Bronchiectasis (%) | 101 (13.5) | 27 (8.4) | 1.69 | (1.1–2.6) | 0.02 |
| Pneumonia (%) | 474 (63.3) | 157 (49.1) | 1.79 | (1.4–2.3) | < 0.001 |
| Asthma (%) | 234 (31.2) | 33 (10.3) | 3.95 | (2.7–5.8) | < 0.001 |
ILD interstitial lung disease, URT upper respiratory tract infection
Fig. 1.
Interstitial lung disease (ILD) diagnoses for common variable immunodeficiency (CVID) and x-linked agammaglobulinemia (XLA) in the USIDNET registry. Percentage of CVID and XLA patients with specific ILD diagnoses in USIDNET
Clinical and Laboratory Comparison between CVID Patients with and without ILD
To further identify factors associated with ILD in PAD, characteristics of patients with CVID with ILD (+ILD) and without ILD (−ILD) were compared (Table 3). There were no differences in age at symptom onset, diagnosis, or last clinical visit between CVID patients with or without ILD. CVID patients with ILD had significantly lower CD4+ (P = 0.005) and CD8+ T cells (P < 0.001) as well as CD19+ B cells (P < 0.001). Pre-diagnostic IgG (P = 0.009) and serum IgA (P = 0.05) were also lower in patients with ILD, while IgM (P = 0.631) was similar between groups. CVID patients with ILD more frequently had a history of pneumonia (78.5% vs. 60.7%, P < 0.001) and bronchiectasis (25.2% vs. 11.5%, P < 0.001) compared to those without ILD. There was no significant difference for sinusitis or upper respiratory tract infections between CVID patients with or without ILD. Thus, reductions of lymphocyte subsets and Ig levels as well as infections of the lower respiratory tract were associated with the presence of ILD in CVID.
Table 3.
Clinical and laboratory comparison between CVID with and without ILD
| +ILD N = 107 |
−ILD N = 642 |
P value* | |
|---|---|---|---|
| Sex = male (%) | 47 (43.9) | 275 (42.8) | 0.916 |
| Age at last visit | 38.20 [28.35, 49.10] | 39.55 [24.05, 50.80] | 0.764 |
| Age of symptom onset | 16.00 [11.10, 27.00] | 16.00 [6.00, 31.00] | 0.747 |
| Age of diagnosis | 29.80 [20.92, 37.75] | 29.90 [14.65, 43.10] | 0.968 |
| CD4 | 565.00 [366.00, 773.00] | 687.00 [474.50, 944.75] | 0.005 |
| CD8 | 244.50 [160.75, 409.50] | 433.00 [290.00, 697.00] | < 0.001 |
| CD19 | 85.50 [19.25, 226.00] | 198.00 [91.50, 323.00] | < 0.001 |
| Pre-diagnostic IgG | 277 [97.5–389.5] | 374.4 [194.5–535.75] | 0.009 |
| IgA | 7.00 [6.00, 21.00] | 10.00 [6.67, 33.00] | 0.05 |
| IgM | 21.45 [9.00, 42.50] | 23.00 [11.00, 44.25] | 0.631 |
| Pneumonia | 84 (78.5%) | 390 (60.7%) | < 0.001 |
| Bronchiectasis | 27 (25.2%) | 74 (11.5%) | < 0.001 |
| Sinusitis | 82 (76.6) | 494 (76.9%) | 1 |
| URT | 59 (55.1%) | 364 (56.7%) | 0.845 |
Statistical analysis was performed using Chi-square test for categorical variables and Mann-Whitney test for continuous variables
Continuous variables were summarized as median [IQR] and categorical variables were summarized as counts (percentage)
To gain insight into whether generalized immune dysregulation is associated with ILD in CVID, the prevalence of autoimmune conditions was compared between CVID patients with and without ILD. A diagnosis of autoimmune disorders was significantly higher in patients with ILD compared to patients without ILD (OR = 1.57, P = 0.041). This difference was mainly due to the higher prevalence of cytopenic autoimmune disease in patients with ILD compared to patients without ILD (OR = 2.68, P < 0.001) as there was no significant difference in the prevalence of non-cytopenic autoimmune disease between the two groups (Table 4). Results showed that the prevalence of autoimmune thrombocytopenia and autoimmune hemolytic anemia was higher in patients with ILD compared to patients without ILD (P = 0.001 and P = 0.016, respectively). There was no significant difference in the prevalence of autoimmune neutropenia between both groups, but only eight patients in total had autoimmune neutropenia. These findings indicate that ILD is associated with autoimmune cytopenias in CVID.
Table 4.
Autoimmune disease between patients with and without ILD
| +ILD N = 107 |
−ILD N = 642 |
OR | P value* | |
|---|---|---|---|---|
| AID (%) | 48 (44.9) | 219 (34.1) | 1.57 [1.03–2.4] | 0.041 |
| Cytopenic AID (%) | 28 (26.2) | 75 (11.7) | 2.68 [1.6–4.3] | < 0.001 |
| Non-cytopenic AID (%) | 25 (23.4) | 167 (26.0) | 0.87 [0.5–1.4] | 0.645 |
| Autoimmune thrombocytopenia (%) | 22 (20.6) | 59 (9.2) | 2.56 [1.5–4.4] | 0.001 |
| Autoimmune hemolytic anemia (%) | 13 (12.1) | 35 (5.5) | 2.4 [1.9–4.6] | 0.016 |
| Autoimmune neutropenia (%) | 2 (1.9) | 6 (0.9) | 2.15 [0.3–9.7] | 0.717 |
AID autoimmune disease
Clinical and Laboratory Comparison between CVID Patients with and without Asthma
Finally, we examined whether the parameters associated with ILD in CVID were also shared with asthma, as asthma was the most common chronic lung disease reported in CVID and, like ILD, the role of PAD in its pathogenesis remains unclear. CVID patients with asthma (+asthma) were more likely to be male (46.4% vs. 35.5%, P < 0.007), but otherwise there was no difference in patient demographics between the two groups (Table 5). CVID +asthma had lower IgA and IgM (P < 0.001 and < 0.009, respectively) compared to CVID patients without a diagnosis of asthma (−asthma). Notably, IgE was not statistically different between CVID patients with or without asthma. Unlike ILD, there was no difference in CD19+ B cells, CD4+ T cells, or CD8+ T cells between the two groups. Together these results demonstrate that asthma is associated with male sex and lower levels of IgA and IgM in CVID, factors distinct from those associated with ILD in this same patient cohort.
Table 5.
Clinical and laboratory comparison between CVID patients with and without asthma
| +Asthma N = 476 |
−Asthma N = 234 |
P value* | |
|---|---|---|---|
| n | 476 | 234 | |
| Sex (male) | 221 (46.4) | 83 (35.5) | 0.007 |
| Age at last visit | 39.85 [26.87, 49.73] | 39.65 [23.10, 51.75] | 0.722 |
| Age of symptom onset | 17.00 [7.00, 31.00] | 14.00 [4.30, 27.60] | 0.07 |
| Age of diagnosis | 30.25 [16.60, 42.00] | 27.50 [13.40, 43.80] | 0.35 |
| CD4 | 661.50 [462.00, 929.00] | 674.00 [499.50, 944.00] | 0.402 |
| CD8 | 405.92 [251.00, 729.00] | 398.25 [245.25, 549.75] | 0.208 |
| CD19 | 175.50 [77.25, 326.00] | 164.00 [91.00, 296.00] | 0.684 |
| Pre-diagnostic IgG | 314 [121.5–490] | 379.5 [165.25–566] | 0.269 |
| IgA | 7.00 [6.00, 24.00] | 16.00 [7.00, 46.00] | < 0.001 |
| IgM | 21.00 [10.00, 41.50] | 28.00 [11.30, 52.00] | 0.009 |
| IgE | 2.00 [2.00, 5.00] | 4.00 [1.50, 8.00] | 0.347 |
Statistical analysis was performed using Chi-square test for categorical variables and Mann-Whitney test for continuous variables
Continuous variables were summarized as median [IQR] and categorical variables were summarized as counts (percentage)
Discussion
While Ig deficiency may influence susceptibility to pulmonary complications in PAD, acute respiratory infections, like sinusitis, and chronic lung disease, such as asthma and ILD, often occur despite IgG replacement therapy [23]. There are significant differences in immune dysfunction within the spectrum of PAD, including variability of immunological factors other than the IgG deficit that is therapeutically corrected. Likewise, susceptibility to lung disease fluctuates significantly among PAD patients for reasons incompletely defined. We leveraged the USIDNET registry to find that susceptibility to specific pulmonary complications varied between CVID and XLA and were differentially associated with clinical and laboratory characteristics in CVID.
We found upper respiratory infections, pneumonia, and bronchiectasis to be more common in CVID, despite Ig levels and B cell counts being lower in XLA. Correspondingly, we found CD4+ and CD8+ T cells reduced in CVID compared to XLA, as reported in smaller PAD cohorts [24–27]. This deficit of T cells may contribute to the increase of pulmonary infections in CVID. Community-acquired respiratory infections are more common in HIV+ patients with lower CD4+ T cell counts [28], and deficiency of mucosal-associated invariant T cells has also been associated with increased respiratory infections [29, 30]. Additionally, we previously found bronchiectasis to be associated with reduced CD4+ T cells in a 61 CVID patient cohort from Mount Sinai [31]. Future studies should be conducted to elucidate how T cell deficiency may underlie the increase in respiratory infections seen in CVID relative to XLA or provide an alternative to explain differences in respiratory infections between these two severe forms of PAD.
ILD was far more commonly reported in CVID compared to XLA. The ILD that occurs in CVID is defined by pulmonary lymphoid hyperplasia, which manifests as follicular bronchiolitis (FB) when limited to the peribronchial areas and lymphocytic interstitial pneumonia (LIP) with more diffuse lung involvement (31–36). FB and LIP were among the most common ILD diagnoses for CVID in USIDNET, while no XLA patients had these diagnoses. B cells may be a key factor for CVID ILD as it is ameliorated by rituximab, potentially explaining its absence in XLA [37]. Infections have been associated with this ILD [31, 38–41] which did occur more frequently in CVID patients with history of pneumonia or bronchiectasis. However, this result differed from our previous work from a single center that did not find ILD to be associated with bronchiectasis or pneumonia [31]. This may reflect demographic differences or less stringent means used by the USIDNET registry to categorize patients. Chronic lung disease is frequently misdiagnosed in these patients, as evidenced by the fact that sarcoidosis was reported in 3.21% of subjects but is typically a misdiagnosis of LIP and/or FB in CVID [42, 43]. Rather than infection, inborn immune dysregulation may underlie ILD, as shown by specific genetic variants of CVID [44–48]. Indeed, this study and others found patients with CVID and ILD more likely to have autoimmune cytopenias than other CVID patients [20, 31, 49]. Thus, our results indicate possible roles for infectious as well as non-infectious causes of ILD in CVID and further work is needed to better understand the pathogenesis of this pulmonary complication.
Among CVID patients, we found ILD to be associated with reduced CD4+ and CD8+ T cell counts. While lower CD4+ T cell counts have not been previously linked with ILD in CVID, reduced levels of CD8+ T cells have [32]. Considering that lower CD4+ T cell counts have been linked with bronchiectasis in CVID [31] and pulmonary diagnoses in USIDNET are not necessarily confirmed by imaging or pathology, bronchiectasis cases may have been listed as ILD in USIDNET. Alternatively, the prior single-center studies may not have been adequately powered to detect the association of lower CD4+ T cells with ILD. Another explanation could be that CD4+ T cell counts may have been reduced by immunosuppressive therapy used to manage ILD, which would have been accounted for in the single-center study but not in USIDNET. Further efforts to understand how deficiency of T cell subsets contribute to ILD in CVID is needed.
Rather than being associated with autoimmunity and reduced T cells like ILD, asthma was linked with reduced IgA and IgM in CVID. There was no association with IgE levels and asthma, but specific IgE against allergens may be absent in PAD patients with allergic asthma [42, 50]. Lower Ig levels in CVID were associated with increased respiratory infections which could predispose to more asthma exacerbations. IgM that protects against respiratory infection may be absent in CVID patients with more profound Ig deficiency [51]. However, asthma was actually higher in CVID relative to XLA despite Ig deficiency being less severe in these patients. The higher burden of respiratory infections reported in CVID compared to XLA might be contributing to this higher prevalence of asthma. Alternatively, reduction of regulatory T cells in CVID may increase susceptibility to asthma relative to XLA. Numerous studies have identified reductions of regulatory T cells in CVID [52–55]. While the mechanisms that underlie asthma in PAD remain elusive, our results suggest that investigations into the role of T cells as well as other differences between CVID and XLA may prove informative.
Despite the fact that both are forms of PAD defined by profound Ig deficiency, we found that pulmonary complications were more common in CVID than XLA. Moreover, we found that ILD and asthma were associated with divergent clinical and laboratory characteristics in CVID. While USIDNET registry data is limited in its ability to account for differences in age at start of Ig replacement, diagnostic delay, IgG trough, or other related variables, the data were consistent with known features of CVID and XLA. There was no differences between age of symptom onset and age of diagnosis between CVID patients with and without ILD or with and without asthma, suggesting that age or diagnostic delay may not contribute to these pulmonary complications. Thus, our results indicate that susceptibility to specific pulmonary complications in PAD is determined by factors that extend beyond antibody deficiency and include PAD diagnosis as well as differences in T cells and association with autoimmune disease. Further exploration of antibody-independent mechanisms of pulmonary disease may help identify novel treatment to complement Ig replacement therapy.
Acknowledgements
The authors thank Tara Caulder, the USIDNET program director, and Marla Goldsmith, the registry manager, for assistance with this project as well as all physicians who enrolled patients for their contributions to USIDNET.
Funding This work was supported by National Institutes of Health grant AI137183 (to P.J.M.).
Abbreviations
- FB
Follicular bronchiolitis
- Ig
Immunoglobulin
- CVID
Common variable immunodeficiency
- ILD
Interstitial lung disease
- LIP
Lymphocytic interstitial pneumonia
- PAD
Primary antibody deficiency
- USIDNET
US Immunodeficiency Network
- XLA
X-linked agammaglobulinemia
Footnotes
Disclosure of Conflicts of Interest The authors declare that they have no conflict of interests.
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Schussler E, Beasley MB, Maglione PJ. Lung disease in primary antibody deficiencies. J Allergy Clin Immunol Pract. 2016;4(6): 1039–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wood P, Network UKPI. Primary antibody deficiencies: recognition, clinical diagnosis and referral of patients. Clin Med (Lond). 2009;9(6):595–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Verma N, Grimbacher B, Hurst JR. Lung disease in primary antibody deficiency. Lancet Respir Med. 2015;3(8):651–60. [DOI] [PubMed] [Google Scholar]
- 4.Hewitt R, Farne H, Ritchie A, Luke E, Johnston SL, Mallia P. The role of viral infections in exacerbations of chronic obstructive pulmonary disease and asthma. Ther Adv Respir Dis. 2016;10(2):158–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Beigelman A, Bacharier LB. Early-life respiratory infections and asthma development: role in disease pathogenesis and potential targets for disease prevention. Curr Opin Allergy Clin Immunol. 2016;16(2):172–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.van Meel ER, Jaddoe VWV, Bonnelykke K, de Jongste JC, Duijts L. The role of respiratory tract infections and the microbiome in the development of asthma: a narrative review. Pediatr Pulmonol. 2017;52(10):1363–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Caminati M, Pham DL, Bagnasco D, Canonica GW. Type 2 immunity in asthma. World Allergy Organ J. 2018;11(1):13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Busse PJ, Razvi S, Cunningham-Rundles C. Efficacy of intravenous immunoglobulin in the prevention of pneumonia in patients with common variable immunodeficiency. J Allergy Clin Immunol. 2002;109(6):1001–4. [DOI] [PubMed] [Google Scholar]
- 9.Orange JS, Grossman WJ, Navickis RJ, Wilkes MM. Impact of trough IgG on pneumonia incidence in primary immunodeficiency: a meta-analysis of clinical studies. Clin Immunol. 2010;137(1):21–30. [DOI] [PubMed] [Google Scholar]
- 10.Walsh JE, Gurrola JG 2nd, Graham SM, Mott SL, Ballas ZK. Immunoglobulin replacement therapy reduces chronic rhinosinusitis in patients with antibody deficiency. Int Forum Allergy Rhinol. 2017;7(1):30–6. [DOI] [PubMed] [Google Scholar]
- 11.Chapel H, Lucas M, Lee M, Bjorkander J, Webster D, Grimbacher B, et al. Common variable immunodeficiency disorders: division into distinct clinical phenotypes. Blood. 2008;112(2):277–86. [DOI] [PubMed] [Google Scholar]
- 12.Resnick ES, Moshier EL, Godbold JH, Cunningham-Rundles C. Morbidity and mortality in common variable immune deficiency over 4 decades. Blood. 2012;119(7):1650–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cunningham-Rundles C, Maglione PJ. Common variable immunodeficiency. J Allergy Clin Immunol. 2012;129(5):1425–6 e3. [DOI] [PubMed] [Google Scholar]
- 14.Quinti I, Soresina A, Spadaro G, Martino S, Donnanno S, Agostini C, et al. Long-term follow-up and outcome of a large cohort of patients with common variable immunodeficiency. J Clin Immunol. 2007;27(3):308–16. [DOI] [PubMed] [Google Scholar]
- 15.Winkelstein JA, Marino MC, Lederman HM, Jones SM, Sullivan K, Burks AW, et al. X-linked agammaglobulinemia: report on a United States registry of 201 patients. Medicine (Baltimore). 2006;85(4):193–202. [DOI] [PubMed] [Google Scholar]
- 16.Aghamohammadi A, Allahverdi A, Abolhassani H, Moazzami K, Alizadeh H, Gharagozlou M, et al. Comparison of pulmonary diseases in common variable immunodeficiency and X-linked agammaglobulinaemia. Respirology. 2010;15(2):289–95. [DOI] [PubMed] [Google Scholar]
- 17.Jolles S The variable in common variable immunodeficiency: a disease of complex phenotypes. J Allergy Clin Immunol Pract. 2013;1(6):545–56 quiz 57. [DOI] [PubMed] [Google Scholar]
- 18.Maglione PJ. Autoimmune and lymphoproliferative complications of common variable immunodeficiency. Curr Allergy Asthma Rep. 2016;16(3):19. [DOI] [PubMed] [Google Scholar]
- 19.Quinti I, Soresina A, Guerra A, Rondelli R, Spadaro G, Agostini C, et al. Effectiveness of immunoglobulin replacement therapy on clinical outcome in patients with primary antibody deficiencies: results from a multicenter prospective cohort study. J Clin Immunol. 2011;31(3):315–22. [DOI] [PubMed] [Google Scholar]
- 20.Gathmann B, Mahlaoui N, Gerard L, Oksenhendler E, Warnatz K, Schulze I, et al. Clinical picture and treatment of 2212 patients with common variable immunodeficiency. J Allergy Clin Immunol. 2014;134(1):116–26. [DOI] [PubMed] [Google Scholar]
- 21.Jolliff CR, Cost KM, Stivrins PC, Grossman PP, Nolte CR, Franco SM, et al. Reference intervals for serum IgG, IgA, IgM, C3, and C4 as determined by rate nephelometry. Clin Chem. 1982;28(1):126–8. [PubMed] [Google Scholar]
- 22.Picard C, Bobby Gaspar H, Al-Herz W, Bousfiha A, Casanova JL, Chatila T, et al. International Union of Immunological Societies: 2017 primary immunodeficiency diseases committee report on inborn errors of immunity. J Clin Immunol. 2018;38(1):96–128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cunningham-Rundles C Key aspects for successful immunoglobulin therapy of primary immunodeficiencies. Clin Exp Immunol. 2011;164(Suppl 2):16–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Giovannetti A, Pierdominici M, Mazzetta F, Marziali M, Renzi C, Mileo AM, et al. Unravelling the complexity of T cell abnormalities in common variable immunodeficiency. J Immunol. 2007;178(6): 3932–43. [DOI] [PubMed] [Google Scholar]
- 25.Liu Y, Wu Y, Lam KT, Lee PP, Tu W, Lau YL. Dendritic and T cell response to influenza is normal in the patients with X-linked agammaglobulinemia. J Clin Immunol. 2012;32(3):421–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bateman EA, Ayers L, Sadler R, Lucas M, Roberts C, Woods A, et al. T cell phenotypes in patients with common variable immunodeficiency disorders: associations with clinical phenotypes in comparison with other groups with recurrent infections. Clin Exp Immunol. 2012;170(2):202–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Azizi G, Rezaei N, Kiaee F, Tavakolinia N, Yazdani R, Mirshafiey A, et al. T-cell abnormalities in common variable immunodeficiency. J Investig Allergol Clin Immunol. 2016;26(4):233–43. [DOI] [PubMed] [Google Scholar]
- 28.Lamas CC, Coelho LE, Grinsztejn BJ, Veloso VG. Community-acquired lower respiratory tract infections in HIV-infected patients on antiretroviral therapy: predictors in a contemporary cohort study. Infection. 2017;45(6):801–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hinks TS, Wallington JC, Williams AP, Djukanovic R, Staples KJ, Wilkinson TM. Steroid-induced deficiency of mucosal-associated invariant T cells in the chronic obstructive pulmonary disease lung. Implications for nontypeable Haemophilus influenzae infection. Am J Respir Crit Care Med. 2016;194(10):1208–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pincikova T, Paquin-Proulx D, Moll M, Flodstrom-Tullberg M, Hjelte L, Sandberg JK. Severely impaired control of bacterial infections in a patient with cystic fibrosis defective in mucosal-associated invariant T cells. Chest. 2018;153(5):e93–e6. [DOI] [PubMed] [Google Scholar]
- 31.Maglione PJ, Overbey JR, Radigan L, Bagiella E, Cunningham-Rundles C. Pulmonary radiologic findings in common variable immunodeficiency: clinical and immunological correlations. Ann Allergy Asthma Immunol. 2014;113(4):452–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bates CA, Ellison MC, Lynch DA, Cool CD, Brown KK, Routes JM. Granulomatous-lymphocytic lung disease shortens survival in common variable immunodeficiency. J Allergy Clin Immunol. 2004;114(2):415–21. [DOI] [PubMed] [Google Scholar]
- 33.Carrillo J, Restrepo CS, Rosado de Christenson M, Ojeda Leon P, Lucia Rivera A, Koss MN. Lymphoproliferative lung disorders: a radiologic-pathologic overview. Part I: reactive disorders. Semin Ultrasound CT MR. 2013;34(6):525–34. [DOI] [PubMed] [Google Scholar]
- 34.Tian X, Yi ES, Ryu JH. Lymphocytic interstitial pneumonia and other benign lymphoid disorders. Semin Respir Crit Care Med. 2012;33(5):450–61. [DOI] [PubMed] [Google Scholar]
- 35.Maglione PJ, Ko HM, Beasley MB, Strauchen JA, Cunningham-Rundles C. Tertiary lymphoid neogenesis is a component of pulmonary lymphoid hyperplasia in patients with common variable immunodeficiency. J Allergy Clin Immunol. 2014;133(2):535–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rao N, Mackinnon AC, Routes JM. Granulomatous and lymphocytic interstitial lung disease: a spectrum of pulmonary histopathologic lesions in common variable immunodeficiency—histologic and immunohistochemical analyses of 16 cases. Hum Pathol. 2015;46(9):1306–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Maglione PJ, Gyimesi G, Cols M, Radigan L, Ko HM, Weinberger T, Lee BH, Grasset EK, Rahman AH, Cerutti A, Cunningham-Rundles C BAFF-driven B cell hyperplasia underlies lung disease in common variable immunodeficiency. JCI insight 2019;4(5). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tashtoush B, Okafor NC, Ramirez JF, Smolley L. Follicular bronchiolitis: a literature review. J Clin Diagn Res. 2015;9(9):Oe01–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Thalanayar PM, Holguin F. Follicular bronchiolitis in primary ciliary dyskinesia. Australas Med J. 2014;7(7):294–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Panchabhai TS, Farver C, Highland KB. Lymphocytic interstitial pneumonia. Clin Chest Med. 2016;37(3):463–74. [DOI] [PubMed] [Google Scholar]
- 41.Wheat WH, Cool CD, Morimoto Y, Rai PR, Kirkpatrick CH, Lindenbaum BA, et al. Possible role of human herpesvirus 8 in the lymphoproliferative disorders in common variable immunodeficiency. J Exp Med. 2005;202(4):479–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Agondi RC, Barros MT, Rizzo LV, Kalil J, Giavina-Bianchi P. Allergic asthma in patients with common variable immunodeficiency. Allergy. 2010;65(4):510–5. [DOI] [PubMed] [Google Scholar]
- 43.Verbsky JW, Routes JM. Sarcoidosis and common variable immunodeficiency: similarities and differences. Semin Respir Crit Care Med. 2014;35(3):330–5. [DOI] [PubMed] [Google Scholar]
- 44.Schubert D, Bode C, Kenefeck R, Hou TZ, Wing JB, Kennedy A, et al. Autosomal dominant immune dysregulation syndrome in humans with CTLA4 mutations. Nat Med. 2014;20(12):1410–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Gamez-Diaz L, August D, Stepensky P, Revel-Vilk S, Seidel MG, Noriko M, et al. The extended phenotype of LPS-responsive beige-like anchor protein (LRBA) deficiency. J Allergy Clin Immunol. 2016;137(1):223–30. [DOI] [PubMed] [Google Scholar]
- 46.Kuehn HS, Ouyang W, Lo B, Deenick EK, Niemela JE, Avery DT, et al. Immune dysregulation in human subjects with heterozygous germline mutations in CTLA4. Science. 2014;345(6204):1623–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Milner JD, Vogel TP, Forbes L, Ma CA, Stray-Pedersen A, Niemela JE, et al. Early-onset lymphoproliferation and autoimmunity caused by germline STAT3 gain-of-function mutations. Blood. 2015;125(4):591–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lucas CL, Kuehn HS, Zhao F, Niemela JE, Deenick EK, Palendira U, et al. Dominant-activating germline mutations in the gene encoding the PI(3)K catalytic subunit p110delta result in T cell senescence and human immunodeficiency. Nat Immunol. 2014;15(1):88–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wehr C, Kivioja T, Schmitt C, Ferry B, Witte T, Eren E, et al. The EUROclass trial: defining subgroups in common variable immunodeficiency. Blood. 2008;111(1):77–85. [DOI] [PubMed] [Google Scholar]
- 50.Urm SH, Yun HD, Fenta YA, Yoo KH, Abraham RS, Hagan J, et al. Asthma and risk of selective IgA deficiency or common variable immunodeficiency: a population-based case-control study. Mayo Clin Proc. 2013;88(8):813–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Micol R, Kayal S, Mahlaoui N, Beaute J, Brosselin P, Dudoit Y, et al. Protective effect of IgM against colonization of the respiratory tract by nontypeable Haemophilus influenzae in patients with hypogammaglobulinemia. J Allergy Clin Immunol. 2012;129(3): 770–7. [DOI] [PubMed] [Google Scholar]
- 52.Fevang B, Yndestad A, Sandberg WJ, Holm AM, Muller F, Aukrust P, et al. Low numbers of regulatory T cells in common variable immunodeficiency: association with chronic inflammation in vivo. Clin Exp Immunol. 2007;147(3):521–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Horn J, Manguiat A, Berglund LJ, Knerr V, Tahami F, Grimbacher B, et al. Decrease in phenotypic regulatory T cells in subsets of patients with common variable immunodeficiency. Clin Exp Immunol. 2009;156(3):446–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Melo KM, Carvalho KI, Bruno FR, Ndhlovu LC, Ballan WM, Nixon DF, et al. A decreased frequency of regulatory T cells in patients with common variable immunodeficiency. PLoS One. 2009;4(7):e6269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Arumugakani G, Wood PM, Carter CR. Frequency of Treg cells is reduced in CVID patients with autoimmunity and splenomegaly and is associated with expanded CD21lo B lymphocytes. J Clin Immunol. 2010;30(2):292–300. [DOI] [PubMed] [Google Scholar]