Five-hundred-fold enhanced fluorescence emission from fluorophore-protein coated silver fractal-like structures has been observed. These results further serve to complement our recent work on the effects of nobel metal particles with fluorophores, a relatively new phenomenon we have termed both metal-enhanced fluorescence [1] and radiative decay engineering [2,3]. We believe that this new silver-surface preparation, which results in ultrabright and photostable fluorophores, offers a new generic technology platform for increased fluorescence signal levels, with widespread potential applications to the analytical sciences, imaging, and medical diagnostics.
Silver fractal-like structures have been grown on both silver electrodes and on glass slides. These structures were coated with a monolayer of human serum albumin. (HSA) protein that had been labeled with two fluorescent probes, indocyanine green (ICG) and fluorescein (FITC), respectively. Remarkable increases in fluorescence intensities were observed, typically 500-fold on the glass–silver fractal substrate and some 100-fold on the roughened silver electrodes, as compared with a control with the same substrates but with no fractal structures. In addition, dramatically decreased lifetimes and significantly increased photostabilities of the fluorescent labels were observed. Our results show that we can detect a 160-fold increase in the number of detectable photons from the labeled protein, prior to photobleaching. These exciting results are explained by the metallic surfaces modifying the radiative decay rate, G, of the fluorescent labels.
Roughened silver electrodes are widely used for surface-enhanced Raman scattering (SERS) [4,5]. Although the large increases in Raman signals observed with roughened silver electrodes are thought to be due to surface-contact interactions, we questioned their use in metal-enhanced fluorescence, given that metal-enhanced fluorescence is thought to be a through-space phenomenon. We therefore reasoned that a monolayer protein spacer (≈4 nm) would provide for the through-space distance requirement of metal-enhanced fluorescence [1–3].
A constant current was passed between two silver electrodes separated by 10 mm in pure water (Fig. 1). We typically observed the growth of the fractal-like structures on the silver cathode electrode (Fig. 1, bottom). We incubated the electrode in 30 mM ICG, 60 mM HSA for 24 hr, then rinsied it with water to remove the unbound material. As a control sample, an unused electrode was also coated with ICG–HSA. Figure 2 shows the fluorescence intensity of ICG–HSA-coated roughened electrodes. The remarkable increase in fluorescence intensity, ≈100-fold, was also accompanied by a significantly reduced ICG lifetime, reduced >100-fold. An increase in fluorescence intensity coupled with a reduction in lifetime can only be explained by an increase in the radiative decay rate:
| (1) |
| (2) |
where knr are the nonradiative rates.
Fig. 1.
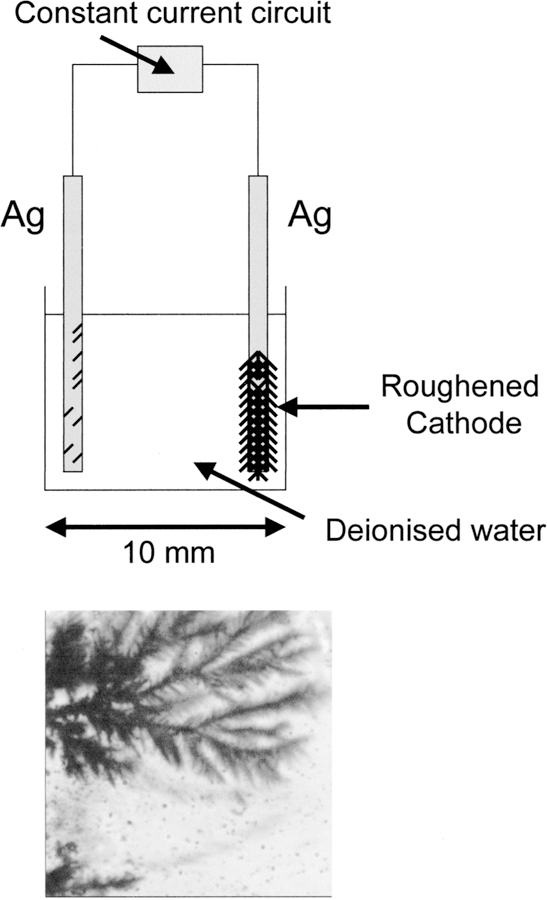
Top: constant-current apparatus for silver fractal growth. Bottom: silver growth on the silver cathode after 6 s. Bright field image.
Fig. 2.
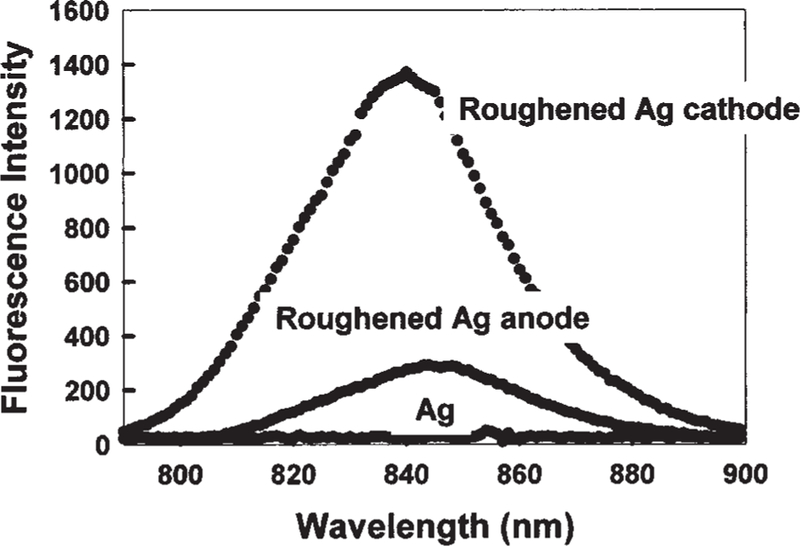
Fluorescence intensity of ICG–HSA-coated roughened silver electrodes and an unused silver electrode, Ag, Ex = 760 nm. It should be noted that the normal fluorescence emission maximum (H2O, pH 7) of 810 nm is shifted in this regard because of the choice of filters than were required to discriminate against a highly scattering surface.
Figure 3 shows the enhanced photostability of ICG on the roughened silver cathode. At the onset of illumination, the relative intensity of ICG-HSA decays more rapidly on the fractal structure than on the control sample (Fig. 3, top). However, the effect is small and the rates become comparable after ≈2 min. Interestingly, if the illumination intensity is adjusted so that both samples yield the same steady-state intensity at the onset of illumination, then the ICG–HSA on the fractal-like silver cathode photobleaches at a much slower rate (Fig. 3, bottom). Given that the detectable signal from the ICG– HSA is given by the area under these photobleaching curves, then substantially more emission can be obtained from the roughened electrodes as compared with the control sample. Intuitively, the ICG intensity remains higher after the initial decrease in intensity (Fig. 3, top), which suggests that ICG will display higher intensities for longer times when bound to fractal silver electrodes. Such effects are most desirable in many analytical applications, such as in high-sensitivity surface assays, in which signal intensity and photostability are paramount concerns.
Fig. 3.
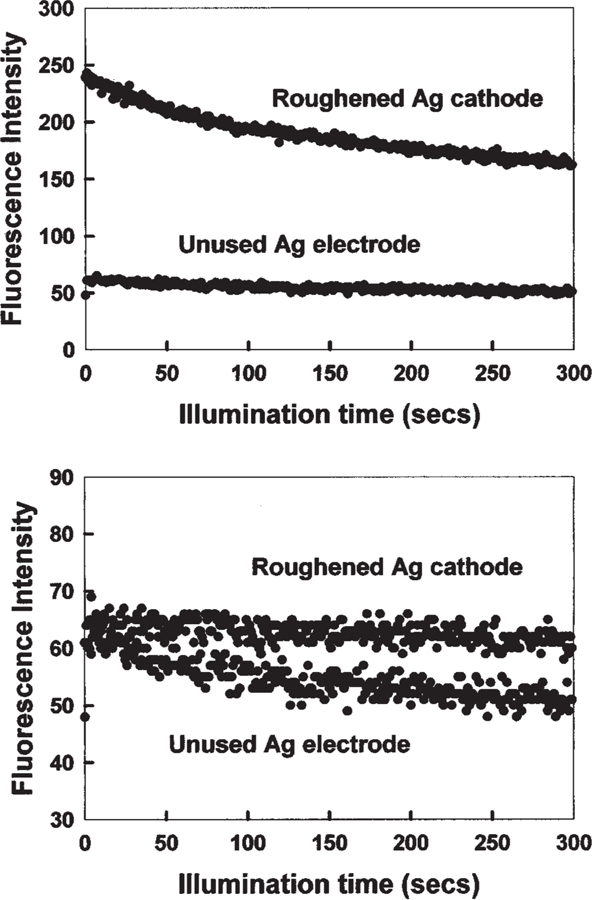
Top: photostability of ICG–HSA on a roughened silver electrode and on an unused silver electrode, measured using the same excitation power at 760 nm, and (bottom) with the laser power adjusted to give the same initial fluorescence intensities. In all measurements, vertically polarized excitation was used, whereas fluorescence emission was observed at the magic angle; i.e., 54.70. It should be noted there is a background of about 40 cps that has not been subtracted in these intensity plots.
To test this technology on other useful surfaces, such as on glass, we grew silver fractal structures on glass between two silver electrodes (Fig. 4). The emission intensity of FITC–HSA was enhanced between 100- and 500-fold when absorbed to the silver fractal structures as compared with the glass control. As was observed with the fractal-silver electrodes, the amplitude-weighted life-time was reduced to near 3 ps as compared with about 80 ps for FITC–HSA-coated glass. This confirms that increased rates of excitation [1–3], or greater coverage of FITC–HSA on the increased surface area fractal structures cannot account for the increased fluorescence signals because these would invariably leave the lifetime unchanged. For both fractal structures, the increase in fluorescence signal is attributed to increases in the radiative decay rate of the fluorophores, Γ, as shown in Eqs. (1) and (2).
Fig. 4.
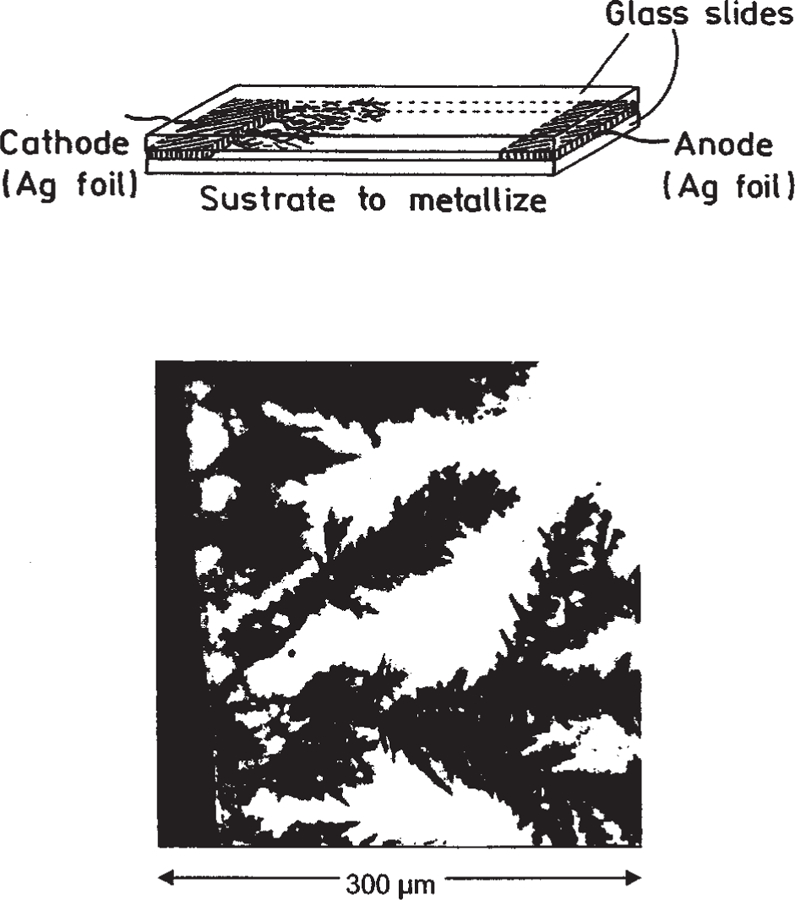
Top: configuration for creation of fractal silver surfaces on silver electrodes and glass. Bottom: silver structures on or near the silver cathode. Bright field image.
APPLICATIONS AND ADVANTAGES OF FRACTAL-METALLIC SURFACES
Our recent reports of metal-enhanced fluorescence have used sodium borohydride or sodium citrate to reduce silver ions to metallic islands on glass substrates [6,7]. We have even employed additional chemistries to immobilize silver colloids to glass [8]. Here silver deposition is accomplished in pure water and localized to regions were the electrodes are placed. In addition, the silver can be generated in situ, on demand, by low-current flow and yet still retains its enhancing properties. As a result, we believe that it will be possible to use proteins and DNA, that are appropriately labeled for ultrabright immunoassays and DNA arrays.
We also anticipate that fractal–metallic surfaces for metal-enhanced fluorescence will find widespread use in medical diagnostics and biotechnology, in an analogous manner to the widespread use of roughened silver electrodes and surfaces for SERS [4,5].
ABOUT THE AUTHORS
Chris D. Geddes Ph.D., is an assistant professor at the University of Maryland Biotechnology Institute, Center for Fluorescence Spectroscopy, in Baltimore. He has a B.Sc. from Lancaster University in England and a Ph.D. in physical chemistry (fluorescence spec-troscopy) from the University of Wales Swansea. He is the editor of the Journal of Fluorescence, Who’s Who in Fluorescence and Annual Reviews in Fluores-cence. He is also executive director of the Society of Fluorescence.
Alexandr Parfenov Ph.D., MD, is a visiting profes-sor from the Research Institute of Physical Chemical Med-icine, Moscow, in Russia. He has a degree of Medicine from the Russian State Medical University (Medical School) and a Ph.D. also, from Department of Cardiology at the same University.
Ignacy Gryczynski Ph.D., is a research professor at the University of Maryland School of Medicine. He has an MS and Ph.D. in physics from the University of Gdansk Institute of Physics in Poland.
David Roll Ph.D., is a visiting professor from the Department of Chemistry, Roberts Wesleyan College in Rochester, New York. He has a BS in chemistry from Harding University and an MS and Ph.D. in Biochem-istry from the University of Illinois at Urbana.
Joanna Malicka Ph.D., is a postdoctoral fellow at the Center for Fluorescence Spectroscopy. She has an MS and a Ph.D. in chemistry from the University of Gdansk.
Joseph R. Lakowicz Ph.D., is director of the Cen-ter for Fluorescence Spectroscopy at the University of Maryland School of Medicine in Baltimore. He has a BS in chemistry from LaSalle University and an MS and Ph.D. in Biochemistry from the University of Illinois at Urbana. He is also the founding editor of the Journal of Biomedical Optics and the Journal of Fluorescence. He is a co-founder and co-President of the Society of Flu-orescence. He is also co-editor of the Who’s Who in Fluorescence and Annual Reviews in Fluorescence.
REFERENCES
- 1.Geddes CD and Lakowicz JR (2002) Metal-Enhanced Fluorescence. J. Fluoresc 12(2), 121–129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lakowicz JR (2001) Radiative Decay Engineering: Biophysical and Biomedical Applications. Anal. Biochem 298, 1–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lakowicz JR, Shen Y, D’Auria S, Malicka J, Fang J, Gryczynski Z, and Gryczynski I (2002) Radiative Decay Engineering 2. Effects of silver island films on fluorescence intensity Lifetimes and Resonance energy transfer. Anal. Biochem 301, 261–277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fleischmann M, Hendra PJ, and McQuillan AJ (1974) Raman spectra of pyradine absorbed at a silver electrode. Chem. Phys. Lett 26(2), 163–166. [Google Scholar]
- 5.Roth E, Hope GA, Schweinsberg DP, Kiefer W, and Fredericks PM (1993) Simple technique for measuring surface enhanced fourier transform Raman spectra of organic compounds. Appl. Spectrosc 47(11), 1794–1800. [Google Scholar]
- 6.Gryczynski I, Malika J, Gryczynski Z, Geddes CD, and Lakowicz JR (2002) The CFS engineers the intrinsic radiative decay rate of low quantum yield fluorophores. J. Fluoresc, 12(1), 11–13. [Google Scholar]
- 7.Malicka J, Gryczynski I, Geddes CD, and Lakowicz JR (2003) Metal enhanced emission from indocyanine Green: A new approach to In Vivo imaging. J. Biomed. Opt, in press. [DOI] [PMC free article] [PubMed]
- 8.Geddes CD, Parfenov A, Gryczynski I, Malicka J, and Lakowicz JR (2003) Metal Enhanced Fluorescence (MEF) Due to Silver Colloids on a Planar Surface: Potential Applications of Indocyanine Green to In Vivo Imaging. J. Phys. Chem. B, submitted. [DOI] [PMC free article] [PubMed]


