Abstract
Background
Mycobacterium tuberculosis (Mtb) is the causative agent of tuberculosis (TB), affecting approximately one third of the world’s population. Development of an adequate immune response will determine disease progression or progress to chronic infection. Risk of developing TB among human immunodeficiency virus (HIV)-coinfected patients (HIV-TB) is 20–30 times higher than those without HIV infection, and a synergistic interplay between these two pathogens accelerates the decline in immunological functions. TB treatment in HIV-TB coinfected persons is challenging and it has a prolonged duration, mainly due to the immune system failure to provide an adequate support for the therapy. Therefore, we aimed to study the role of the hormone 7-oxo-dehydroepiandrosterone (7-OD) as a modulator of anti-tuberculosis immune responses in the context of HIV-TB coinfection.
Methods
A cross-sectional study was conducted among HIV-TB patients and healthy donors (HD). We characterized the ex vivo phenotype of CD4 + T cells and also evaluated in vitro antigen-specific responses by Mtb stimulation of peripheral blood mononuclear cells (PBMCs) in the presence or absence of 7-OD. We assessed lymphoproliferative activity, cytokine production and master transcription factor profiles.
Results
Our results show that HIV-TB patients were not able to generate successful anti-tubercular responses in vitro compared to HD, as reduced IFN-γ/IL-10 and IFN-γ/IL-17A ratios were observed. Interestingly, treatment with 7-OD enhanced Th1 responses by increasing Mtb-induced proliferation and the production of IFN-γ and TNF-α over IL-10 levels. Additionally, in vitro Mtb stimulation augmented the frequency of cells with a regulatory phenotype, while 7-OD reduced the proportion of these subsets and induced an increase in CD4 + T-bet+ (Th1) subpopulation, which is associated with clinical data linked to an improved disease outcome.
Conclusions
We conclude that 7-OD modifies the cytokine balance and the phenotype of CD4 + T cells towards a more favorable profile for mycobacteria control. These results provide new data to delineate novel treatment approaches as co-adjuvant for the treatment of TB.
Keywords: Human, Infectious immunity-bacteria, Hormone modulatory mechanisms, Cytokines, Effector T cells
Background
HIV and Mycobacterium tuberculosis (HIV-TB) coinfection represents a challenge for the study of its physiology, since the presence of both pathogens is characterized by persistent immune dysregulation and altered cytokine profile. Although highly active antiretroviral therapy impedes HIV replication and leads to increased CD4 + T cell numbers, Mtb-specific responses do not change substantially over the first six months on antiretroviral therapy [1]. This situation unravels the need for developing additional immunotherapies to protect from and control Mtb infection, especially in HIV+ individuals. The identification of host factors that promote disease progression or control may lead to the discovery of new host-directed therapies (HDT). In the context of HIV-TB coinfection, these treatments should aim to enhance antigen-specific immune responses, reduce excess inflammation, preserve cell function or improve the effectiveness of conventional therapies. HDT could offer additional advantages for coinfected patients since they may reduce the length of treatments, achieving better outcomes and/or decreasing the chances of relapse or reinfection [2, 3].
Different cell subpopulations are involved in active protection against M. tuberculosis (Mtb) infection. In particular, CD4 + T cells restrict bacterial growth through the production of cytokines that activate macrophages [4]. The variability in the quality and quantity of cytokines produced at the onset of T cell activation depends on the nature of the threat encountered [5]. IFN-γ mediates macrophage activation through the induction of phagocytosis, autophagy and antigenic presentation, and contributes to the resistance and eradication of intracellular pathogens [6, 7]. Additionally, TNF-α is essential for the prevention of Mtb infection and maintenance of latent TB infection [8, 9]. In contrast, IL-10 is a regulatory cytokine that protects the host from excessive inflammation and tissue damage and also inhibits immune responses [10, 11]. Lastly, IL-17A contributes both to the protection and the pathology of TB because it is involved in the formation of mature granuloma [12], but also it mediates the recruitment of neutrophils, which are related to pathological damage of the lung [13].
To date, few studies have explored the effects of immunomodulatory compounds on the function of T cell effectors in the context of TB, particularly during HIV coinfection [14]. Our research group has published several data on this subject, since we have studied the role of DHEA in the context of HIV-TB coinfection for years [15, 16]. In a recent report, we demonstrated the presence of a hormonal imbalance in HIV-TB patients, who exhibited higher plasma levels of DHEA and its androstenetriol (AET) and 7-oxo-DHEA (7-OD) metabolites. Remarkably, we found that higher concentrations of 7-OD positively correlated with absolute CD4 + T cell counts and nadir CD4 + T cell values and also was associated to lung-restricted TB infection [17].
Given the importance of CD4 + T cells over the control of mycobacterial growth and the reported effect of DHEA as an immune-modulating hormone, this work aimed to study the effect of 7-OD on Mtb-induced cell proliferation, cytokine production and master transcription factors expression in CD4 + T cells. Our results showed that Mtb induced a response with a regulatory phenotype instead of an effector response. On the contrary, 7-OD modified the effect caused by Mtb-stimulation, inducing a Th1 type response by increasing lymphoproliferation, the production of IFN-γ and TNF-α over IL-10 levels and induced an augment in CD4+ T-bet+ (i.e., Th1) subpopulation. Therefore, we conclude that 7-OD modifies the balance of cytokines and the profile of CD4 + T cells towards a more favorable profile for mycobacteria control. These results provide new data to delineate novel HDT approaches as co-adjuvants for the treatment of TB.
Methods
Study population
This is a cross-sectional study. A total of 62 subjects (Table 1) were recruited and classified into two groups: 1) chronic HIV-1+ infected individuals with active TB (HIV-TB), who received none or less than two weeks of anti-TB therapy at the moment of sample collection; 2) healthy donors (HD), with no history of TB, HIV or systemic infections. HIV-TB patients were evaluated at the Hospital de Infecciosas “Dr. Francisco Javier Muñiz”, Buenos Aires, Argentina. HIV and TB diagnosis was determined following current guidelines. Some individuals were on anti-retroviral treatment [18]. Peripheral blood buffy-coats from HD were kindly provided by the Hemotherapy Division of Sanatorio “Dr. Julio Mendez”, Buenos Aires, Argentina. None of the subjects had metabolic or endocrine disorders or received DHEA or glucocorticoids. The entire group of individuals had been BCG (Bacillus Calmette-Guerin)-vaccinated at birth.
Table 1.
Characteristics of the subjects enrolled in the study. IQR: interquartile range. Mann-Whitney U test, **p < 0.01 and ****p < 0.001
| Characteristic | HIV-TB (n = 34) | HD (n = 28) |
|---|---|---|
|
Median CD4 + T cell count (cell/ml) |
162.5 (IQR: 34.8–270.0) | 789.0 (IQR: 627.2–1031.0) **** |
|
Median viral load (copies/ml) |
86,637 (IQR: 9536–241,280) | N/A |
| Median DHEA plasma concentration (ng/ml) | 4.4 (IQR: 2.3–8.7) | 0.8 (IQR: 0.7–4.4) ** |
|
Median 7-OD plasma concentration (ng/ml) |
10.9 (IQR: 4.7–45.7) | 5.0 (IQR: 3.0–7.1) ** |
Antigens, mitogens and steroids
Pre-titrated Mycobacterium tuberculosis (Mtb) H37Rv gamma-irradiated whole cells were obtained from BEI Resources, NIAID, NIH (NR-14819); Phytohemmaglutinin (PHA) and DHEA were acquired from Sigma-Aldrich, and 7-OD was purchased from Steraloids Inc.
Culture conditions
EDTA-anticoagulated blood samples were drawn during morning hours. PBMCs were isolated by density gradient centrifugation on Fycoll-Paque® and cultured in RPMI medium (Sigma-Aldrich) supplemented with 10% fetal bovine serum (PAA), 2mML-glutamine (Gibco BRL), 100 U/mL penicillin (PAA) and 100 mg/mL streptomycin (Gibco BRL) at 37 °C in a humidified CO2 containing atmosphere. PBMCs were stimulated with Mtb H37Rv gamma-irradiated whole cells (10 μg/mL) and treated with DHEA or 7-OD (at 1 × 10−9M, 1 × 10−8M, 1 × 10−7M and 1 × 10−6M). We chose steroid concentrations that encompass the spectra ranging from sub-physiological to pharmacological levels, according to our previous work [17].
Cell proliferation assay and cytokine production
PBMCs were plated at 2.5 × 105 cells/well in a U-bottom 96-well plate, as described above. Specific lymphoproliferation was assessed by measuring [methyl-3H] thymidine (GE Healthcare) incorporation. PBMCs were cultured for 96 h, when [methyl-3H] thymidine (0.5 μCi per well) was added and incubated for another 24-h period. Cells were harvested by pouring them onto glassfiber filters (Whatman GF/A). Filters were placed into vials containing liquid scintillation cocktail (2 ml/disc, Optiphase Hisafe 2, PerkinElmer) to determine radiation (counts per minute, CPM) in a liquid scintillation counter (LKB Wallac 1214 RackBeta). To assay cytokine production, cell-free culture medium was obtained from cultures assayed at 120 h. Supernatants were centrifuged for 3 min at 13000 rpm, collected and stored at − 20 °C until cytokine measurement was performed by ELISA, according to the manufacturer’s instructions. IFN-γ, IL-10, TNF-α (BD OptEIA™) and IL-17A (BioLegend) were quantified.
Flow cytometry
Ex-vivo and in-vitro analysis were conducted. Recently thawed or freshly isolated PBMCs were stained with fluorochrome-conjugated antibodies (Abs) for ex vivo study: PE-Cy7 anti-CD3, BV510 anti-CD4, FITC anti-CD25, PE anti-FoxP3, Alexa Fluor 647 anti-ROR-γt, (BD Biosciences) and PerCP/Cy5.5 anti-T-bet (BioLegend). For the in vitro tests, PBMCs were first plated at 1 × 106 cells/ml in a 48-well plate, and incubated as described above. Cells were treated with 7-OD at 1 × 10−6M or DHEA at 1 × 10−7M. After 72 h, cells were harvested, stained, and analyzed by flow cytometry. FoxP3 staining protocol was used according to the manufacturer’s instructions (BD Biosciences). Negative control samples were incubated with irrelevant isotype-matched mAbs in parallel with experimental samples. Samples acquisition and analysis were carried out on a FACSCanto flow cytometer using the BD FACSDiva software (BD Biosciences). Data were analyzed with FlowJo Software (Version 10.4, Tree Star) after gating on the lymphocyte population in the FSC/SSC window and excluding cell aggregates (doublets) by FSC-A/FSC-H and FSC-H/FSC-W. Dead cells were excluded using Live/Dead viability probe (Life Technologies). Gating strategy is shown in Additional file 1: Figure S1. Boolean gating was used to define individual non-overlapping functional subsets or CD4 + T cells that expressed only two transcription factors. Frequencies were normalized to unstimulated control. Mean fluorescence intensity (MFI) was depicted as the ratio of the geometric mean MFI of the marker of interest over the MFI of the corresponding negative population. MFI is expressed as median ± interquartile range (IQR).
Statistical analyses
Statistical analyses were conducted using GraphPad Prism 7 (GraphPad software Inc.). D’Agostino & Pearson normality test was used to assess normal distribution. Comparisons of the two groups for paired samples were assessed by the paired t test or Wilcoxon matched-pairs signed rank test, as appropriate. Alternatively, unpaired t test or Mann-Whitney U test were used to evaluate unpaired samples. Comparisons of three or more variables were done using the Friedman test, followed by post-hoc comparisons: Fisher’s LSD or Dunn’s test. Correlation analyses were determined using the Spearman’s rank test. p values < 0.05 were considered significant.
Results
Description of study cohorts
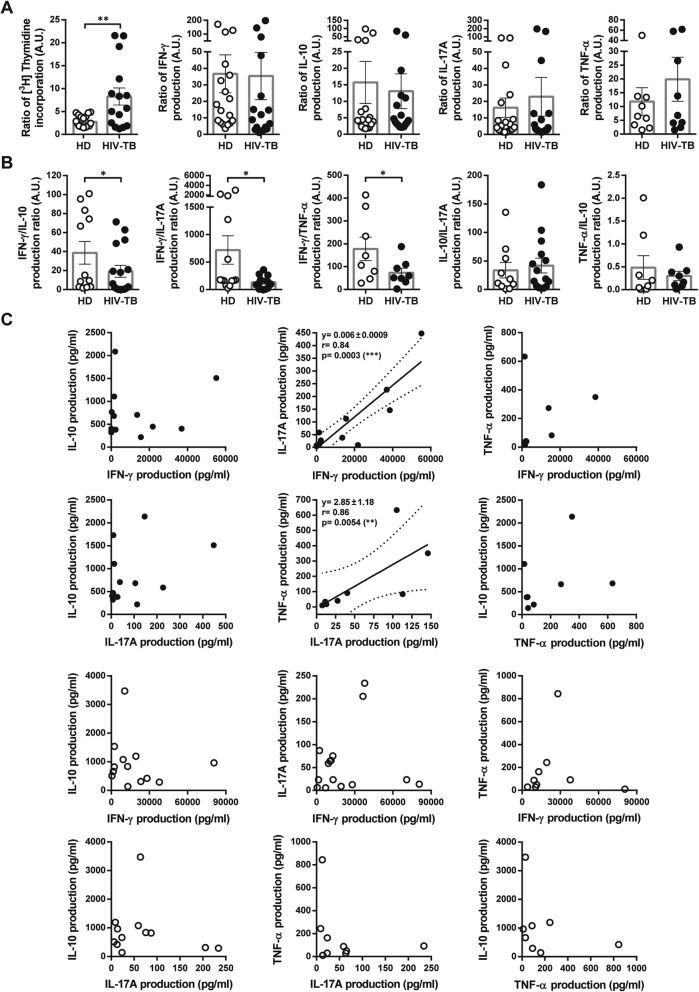
We initially analyzed memory responses to Mtb on PBMCs, studying cell proliferation and cytokine production (IFN-γ, IL-10, IL-17A and TNF-α). Cytokine production ratio and correlation analyses were also performed (Fig. 1). We observed that Mtb-induced lymphoproliferation in HIV-TB was higher than in HD (Fig. 1a). However, compared to HIV-TB individuals, HD produced increased levels of IFN-γ with respect to those of IL-10, IL-17A and TNF-α (Fig. 1b), suggesting a predominant Th1-type response. Alternatively, a positive association between the secretion of IL-17A with IFN-γ and TNF-α was observed in HIV-TB individuals (Fig. 1c). These data suggest that HIV-TB patients are not able to generate an adequate response for the containment of the bacteria, as a result of the reduction in IFN-γ/IL-10 and IFN-γ/IL-17A ratios and the combined production of pro-inflammatory cytokines (TNF-α and IL-17A) which unrestrained levels are associated with immunopathology [19, 20].
Fig. 1.
In vitro responses to M. tuberculosis of PBMCs from HIV-TB and HD individuals. PBMCs from HD and HIV-TB individuals were stimulated with gamma-irradiated Mtb (10 μg/ml) for 5 days. Each graph depicts values from HIV-TB patients (black circles) and HD (open circles). Each symbol represents an individual subject. a Mtb-induced proliferation and cytokine secretion. The Y axis shows cell proliferation or cytokine production. Values are relativized to unstimulated cells. b Cytokine production profiles from HD and HIV-TB in response to Mtb. The Y axis indicates cytokine ratio. Error bars show mean ± SEM. Unpaired t test or Mann-Whitney U test, as appropriated *p < 0.05, **p < 0.01. c Correlation analysis among secretion of IFN-γ, IL-10, IL-17A and TNF-α from HIV-TB and HD groups. Continuous lines represent linear regression curves, and dashed lines represent 95% confidence intervals. Spearman’s rank correlation coefficient (r), p value and slope are indicated
7-oxo-DHEA boosts Mtb-specific lymphoproliferation
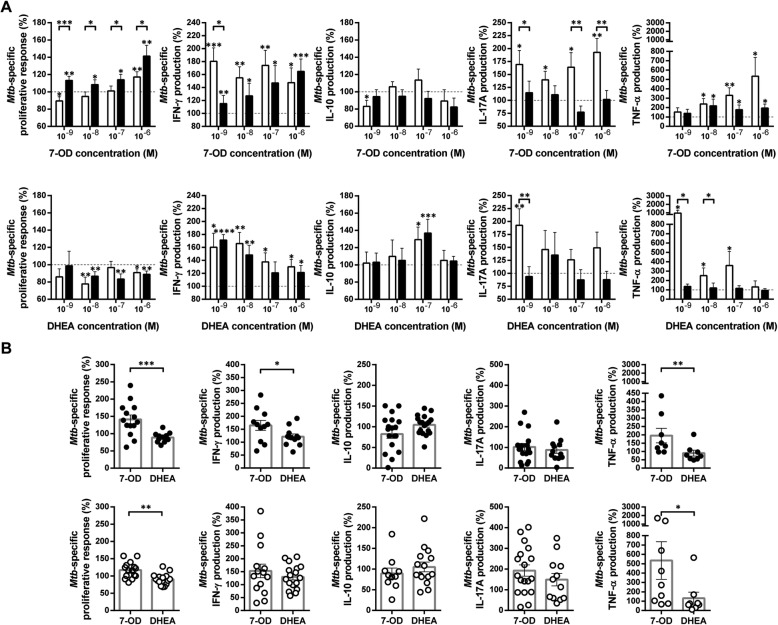
It is generally accepted that pathogen-specific T cells exhaustion prevents lymphoproliferation and the ability to mediate effector functions during chronic infections [21]. In effect, the proliferative capacity of HIV-specific CD4 + T cells is impaired in individuals with higher viral load [22]. However, Mtb-specific CD4+ T cells from both of both HIV-infected and uninfected individuals, preserve a robust proliferative response [23]. Therefore, we studied the effect of 7-OD and DHEA on Mtb-induced cell proliferation. We observed that 7-OD significantly promoted proliferative activity, with a differential outcome on cells from HIV-TB individuals when compared with HD (Fig. 2a, left upper panel). Otherwise, DHEA induced anti-proliferative activity, in both HIV-TB individuals and HD (Fig. 2a, left lower panel). We contrasted the modulatory effect of 7-OD and DHEA at the highest dose (1 × 10−6M) and found that 7-OD significantly promotes Mtb-specific cell proliferation (Fig. 2b).
Fig. 2.
7-OD and DHEA modulate Mtb-specific immune responses. PBMCs from HIV-TB and HD were stimulated with gamma-irradiated Mtb as indicated in methods. a Effect of 7-OD and DHEA at different concentrations on Mtb-specific responses. The X axis indicates hormone concentrations. The Y axis shows cell proliferation or cytokine production. Values are relativized to cells stimulated with Mtb (100%). The results are plotted for HD (open bars) and HIV-TB patients (black bars). Error bars show mean ± SEM. Statistical analyses were performed using absolute values. Unpaired t test or Mann-Whitney U test *p < 0.05, **p < 0.01, ***p < 0.005 and ****p < 0.001. Numbers of individuals per group were from 8 to 18, according to sample availability. b Comparison of the immunomodulatory capacity of 7-OD and DHEA at 1 × 10−6M on PBMCs stimulated with Mtb. Values are relativized to the cells stimulated only with antigen (100%). The results are plotted for HIV-TB patients (black circles) and HD (open circles). Each symbol represents an individual subject. The statistical analysis was performed using absolute values. Unpaired t test or Mann-Whitney U test, as appropriated *p < 0.05, **p < 0.01, and ***p < 0.005. a and b To estimate the immunomodulatory effect of steroids, the response of Mtb-stimulated cells was defined as 100%. The calculation was: Modulation (%) = (Mtb-stimulated and hormone treated - unstimulated) / (Mtb-stimulated - unstimulated) × 100
7-OD modifies Mtb-induced cytokine profile to a Th1-type
We aimed to study the effect of 7-OD over the cytokine pattern (IFN-γ, IL-10, IL-17A and TNF-α) secreted by Mtb-stimulated PBMCs. The results showed that 7-OD significantly stimulated the production of IFN-γ and TNF-α in both study groups (Fig. 2a, upper panel). No modulation by 7-OD on IL-10 and IL-17A secretion was observed in the HIV-TB group, while this hormone downregulated the production of IL-10 (1 × 10− 9 M) and stimulated the secretion of IL-17A in HD (Fig. 2a, upper panel). On the other hand, DHEA increased the production of IFN-γ and IL-10 in HD and HIV-TB (1 × 10− 7 M) (Fig. 2a, lower panel). DHEA also enhanced IL-17A and TNF-α levels in HD at low doses (Fig. 2a, lower panel), coinciding with its physiological plasma concentration [17]. 7-OD (1 × 10−6M) was more effective than DHEA in increasing the secretion of IFN-γ in HIV-TB and TNF-α in both groups. In contrast, no differences were observed between both hormone effects on the production of IL-10 or IL-17A (Fig. 2b).
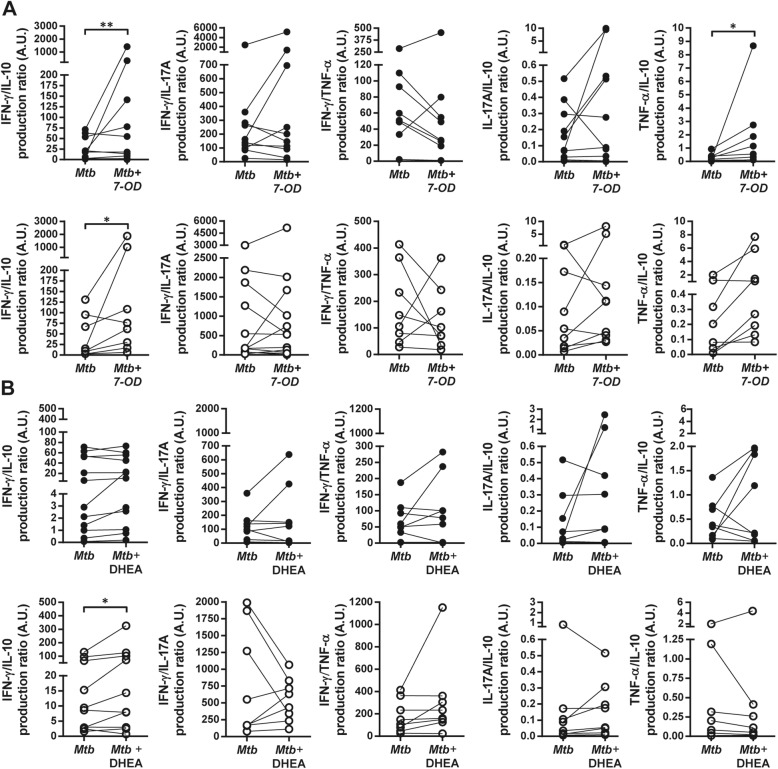
As Mtb modulate the production of cytokines in favor of a regulatory response, an important feature of an adjuvant compound is the ability to promote a Th1 profile. Thus, Mtb-induced cytokine ratio was analyzed. Remarkably, 7-OD (1 × 10−6M) managed to significantly upregulate IFN-γ/IL-10 and TNF-α/IL-10 ratios only in patients (Fig. 3, upper panel). This effect was not observed with DHEA (Fig. 3, lower panel), but both hormones induced an increment of IFN-γ/IL-10 ratio in HD. These data indicate that 7-OD was able to modulate Mtb-specific cell proliferation and a Th1 type cytokine profile during HIV-TB co-infection, by increasing the levels of IFN-γ and TNF-α, without suppressing the necessary action of IL-10 and IL-17A, which would mediate an optimal resistance to the mycobacteria [24].
Fig. 3.
Ratios of in vitro Mtb-specific cytokine production are modified by 7-OD and DHEA. PBMCs from HIV-TB individuals (black circles) and HD (open circles) were stimulated with gamma-irradiated Mtb in the presence/absence of hormones, as specified in methods. The Y axis indicates cytokine production ratio in response to Mtb and its modulation by (a) 7-OD or (b) DHEA, both at 1 × 10−6M. Each symbol represents an individual subject. The statistical analysis was performed using absolute values. Paired t test or Wilcoxon matched-pairs signed rank test, as appropriated *p < 0.05
Characterization of CD4 + Th subpopulations in HIV-TB and HD individuals
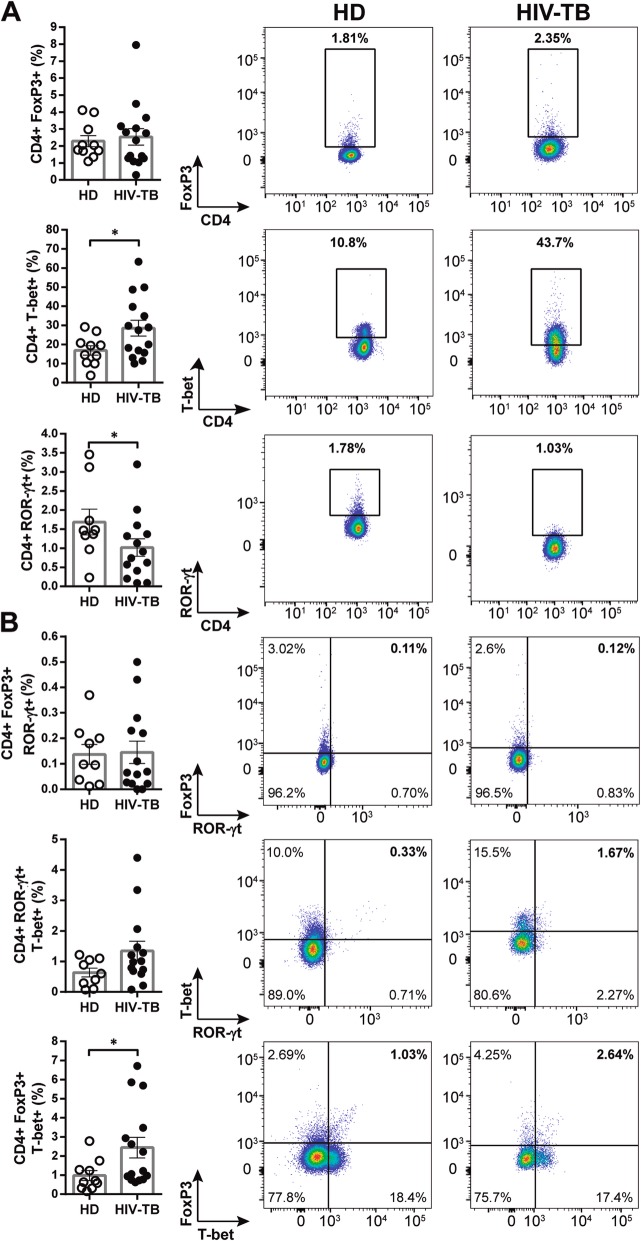
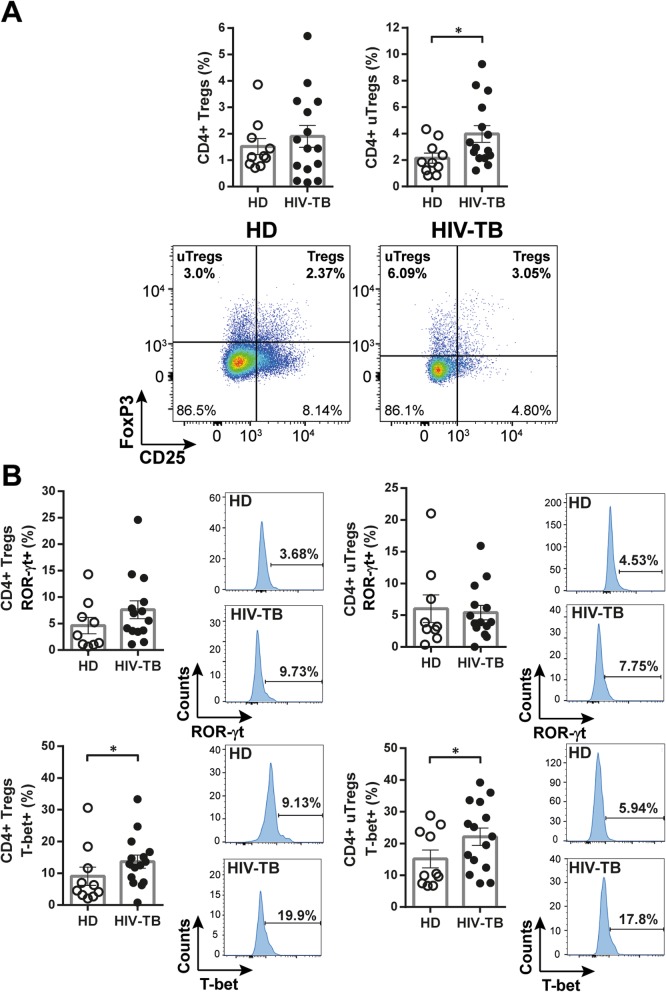
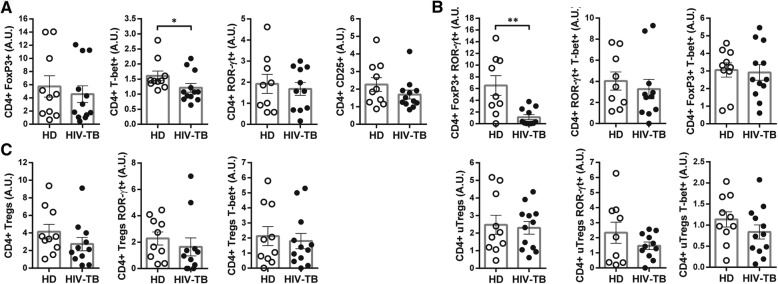
Determination of the expression of transcription factors (TF) FoxP3, ROR-γt and T-bet and of CD25 in PBMCs from HIV-TB patients and HD was performed. In ex-vivo analyses we found that HIV-TB patients presented higher frequencies of CD4+ T-bet+ and CD4 + FoxP3 + T-bet+ cells, and exhibited lower numbers of CD4+ ROR-γt + subpopulation compared to HD (Fig. 4a and b). Besides conventional regulatory CD4 + T cells (i.e. Tregs, CD25 + FoxP3+), the frequency of “unconventional” regulatory CD4 + T cells (i.e. uTregs, CD25-FoxP3+) was also analyzed. This is a CD4 + FoxP3+ T cell population with undetectable levels of CD25 [25] that exerts an inhibitory capacity supressing proliferation and cytokine production from effector T cells [26]. We found that in HIV-TB patients uTregs were expanded, as reported elsewhere (Fig. 5a and [25]). Moreover, within this cohort a larger frequency of T-bet+ cells were observed in both Tregs and uTregs populations (Fig. 5 b).
Fig. 4.
Phenotype of CD4 + T cells from HD and HIV-TB. Recently thawed or freshly isolated PBMCs were stained and analyzed by flow cytometry. Figure shows the percentage of CD4+ cells that express (a) FoxP3, T-bet or ROR-γt and (b) the co-expression of transcription factors using a Boolean gating strategy. Representative flow cytometry examples are shown. The results are plotted for HD (open circles) and HIV-TB patients (black circles). Each symbol represents an individual subject. Unpaired t test or Mann-Whitney U test, as appropriated *p < 0.05
Fig. 5.
Analysis of Treg and uTreg subsets within CD4 + T cells from HIV-TB patients and HD. Recently thawed or freshly isolated PBMCs were stained and analyzed by flow cytometry. Figure shows the percentage of (a) CD4+ regulatory T cells (Tregs, CD25 + FoxP3+) and CD4+ unconventional regulatory T cells (uTregs, CD25-FoxP3+) and (b) the co-expression of the transcription factors ROR-γt or T-bet within each population. The results are plotted for HD (open circles) and HIV-TB patients (black circles). Each symbol represents an individual subject. Unpaired t test or Mann-Whitney U test, as appropriated *p < 0.05. Representative flow cytometry examples are shown
The balance Th1/Tregs may play an essential role in clinical outcome
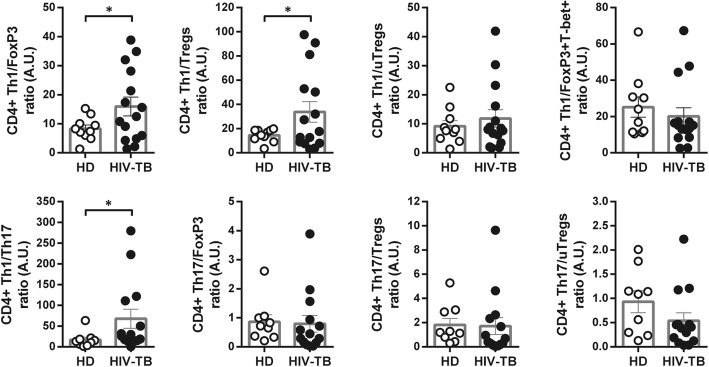
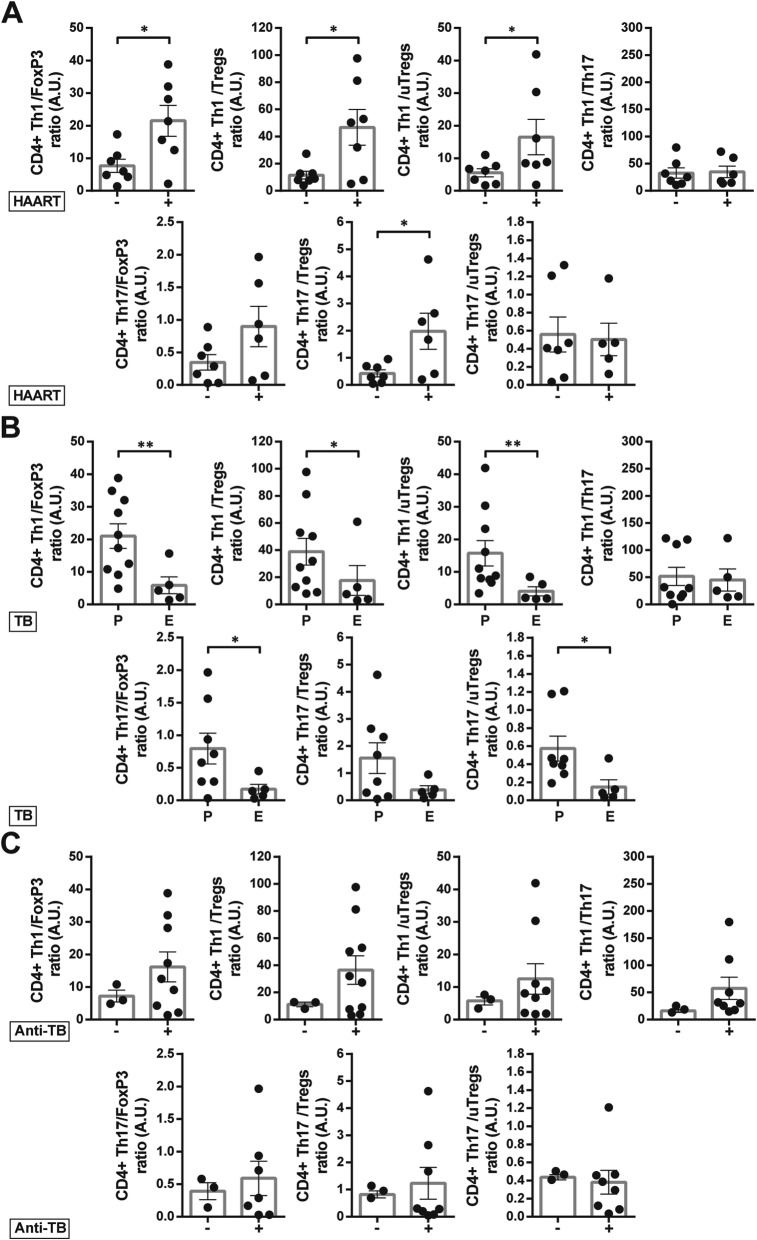
The balance among Th1, Th17, Tregs and uTregs phenotypes was analyzed. Unexpectedly, data revealed that HIV-TB individuals presented a higher proportion of Th1 cells over Tregs and Th17 populations, when compared to HD (Fig. 6). We contrasted our findings with clinical data from the patients enrolled. The frequency of CD25+, FoxP3+ or FoxP3 + T-bet+ cells was negatively associated with CD4 + T cell count. The same results were observed for uTregs and Tregs, which also correlated negatively with CD4/CD8 T cell ratio. CD4 + FoxP3+ cells were associated with lower nadir CD4 + T cell count and CD4/CD8 T cell ratio, whereas this subset presented a positive correlation with viral load (Table 2A). In addition, greater frequencies of CD4 + FoxP3+ T cells were found in patients who were neither on highly active antiretroviral therapy (HAART) nor TB treatment and in those with extrapulmonary TB. Similarly, higher proportions of Tregs were found in patients who were not receiving HAART, and uTregs were present to a greater extent in those individuals with disseminated TB (Fig. 7a and b). These results suggest that the expression of FoxP3 in CD4 + T cells may be associated with an unfavorable disease outcome in HIV-TB patients. Conversely, T-bet expression could be related to a favorable clinical outcome, since it was associated with greater CD4 + T cell counts and nadir value (Table 2A) and with the development of a lung-restricted TB instead of a disseminated infection. Moreover, uTregs which express T-bet were present in greater frequency in individuals with pulmonary TB (Fig. 7b). Finally, in individuals who had not received TB treatment, higher frequencies of CD4 + T cells expressing CD25 or ROR-γt were found (Fig. 7c).
Fig. 6.
Changes in the balance between Th1, Tregs, uTregs and Th17 cells in patients with HIV-TB compared with HD. Recently thawed or freshly isolated PBMCs were stained and analyzed by flow cytometry. Figure shows CD4 + T cell subset ratios. The results are plotted for HD (open circles) and HIV-TB patients (black circles). Each symbol represents an individual subject. Unpaired t test or Mann-Whitney U, as appropriated *p < 0.05
Table 2.
Correlation analysis between the transcriptional profile of Mtb-Specific CD4 + T cells and clinical data from HIV-TB patients. Table shows the results of CD4 + T cells expressing (A) CD25, FoxP3, ROR-γt or T-bet and the co-expression of FoxP3, ROR-γt and T-bet. Additionally, (B) Tregs (CD25 + FoxP3+) and uTregs (CD25-FoxP3+) subsets and the co-expression of the transcription factors ROR-γt or T-bet within each subpopulation were analyzed. A comparison of the percentage of different CD4+ Th cells was performed with total and nadir CD4 + T cell counts, CD4/CD8 ratio and viral load. Number of values (n), Spearman’s rank correlation coefficient (r), and p value are indicated
| Clinical parameter | FoxP3+ | ROR-γt+ | T-bet+ | CD25+ | FoxP3+ ROR-γt+ | ROR-γt + T-bet+ | FoxP3+ T-bet+ |
|---|---|---|---|---|---|---|---|
| A | |||||||
| CD4 + T cell count (cell/ml) | n = 14 | n = 14 | n = 15 | n = 15 | n = 14 | n = 14 | n = 15 |
| r = −0.66 | r = 0.14 | r = 0.48 | r = − 0.45 | r = 0.29 | r = 0.37 | r = − 0.49 | |
| p = 0.0035 | p = 0.31 | p = 0.041 | p = 0.045 | p = 0.16 | p = 0.19 | p = 0.029 | |
| Nadir CD4 T cell count (cell/ml) | n = 15 | n = 14 | n = 14 | n = 15 | n = 14 | n = 14 | n = 15 |
| r = −0.65 | r = 0.55 | r = 0.65 | r = − 0.41 | r = 0.091 | r = 0.61 | r = − 0.29 | |
| p = 0.021 | p = 0.45 | p = 0.014 | p = 0.22 | p = 0.40 | p = 0.033 | p = 0.28 | |
| CD4/CD8 T cell ratio | n = 15 | n = 14 | n = 15 | n = 15 | n = 14 | n = 14 | n = 15 |
| r = −0.55 | r = − 0.12 | r = 0.23 | r = − 0.27 | r = − 0.034 | r = 0.13 | r = − 0.17 | |
| p = 0.018 | p = 0.35 | p = 0.21 | p = 0.16 | p = 0.49 | p = 0.63 | p = 0.27 | |
| Viral load (copies/ml) | n = 15 | n = 14 | n = 15 | n = 15 | n = 14 | n = 14 | n = 15 |
| r = 0.56 | r = 0.25 | r = − 0.14 | r = 0.41 | r = 0.15 | r = 0.013 | r = 0.071 | |
| p = 0.015 | p = 0.19 | p = 0.32 | p = 0.13 | p = 0.30 | p = 0.97 | p = 0.80 | |
| B | |||||||
| Clinical parameter | Tregs | Tregs ROR-γt+ |
Tregs T-bet+ |
uTregs | uTregs ROR-γt+ |
uTregs T-bet+ |
|
| CD4 + T cell count (cell/ml) | n = 15 | n = 14 | n = 15 | n = 15 | n = 14 | n = 15 | |
| r = −0.71 | r = 0.33 | r = − 0.17 | r = − 0.48 | r = 0.059 | r = − 0.22 | ||
| p = 0.015 | p = 0.25 | p = 0.54 | p = 0.036 | p = 0.84 | p = 0.42 | ||
| Nadir CD4 T cell count (cell/ml) | n = 15 | n = 15 | n = 14 | n = 14 | n = 14 | n = 14 | |
| r = − 0.43 | r = 0.16 | r = − 0.19 | r = − 0.29 | r = − 0.27 | r = − 0.22 | ||
| p = 0.090 | p = 0.66 | p = 0.58 | p = 0.19 | p = 0.44 | p = 0.49 | ||
| CD4/CD8 T cell ratio | n = 15 | n = 14 | n = 15 | n = 15 | n = 14 | n = 15 | |
| r = − 0.53 | r = 0 | r = − 0.17 | r = − 0.27 | r = − 0.059 | r = − 0.16 | ||
| p = 0.022 | p = 0.50 | p = 0.55 | p = 0.15 | p = 0.84 | p = 0.55 | ||
| Viral Load (copies/ml) | n = 14 | n = 14 | n = 14 | n = 14 | n = 14 | n = 14 | |
| r = 0.42 | r = 0.0083 | r = − 0.23 | r = 0.17 | r = − 0.033 | r = − 0.14 | ||
| p = 0.14 | r = 0.98 | p = 0.43 | p = 0.27 | p = 0.92 | p = 0.63 | ||
Fig. 7.
Analisys of different CD4 + T cell subsets and different clinical parameters from HIV-TB patients. CD4 + T cell phenotype from HIV-TB were associated with clinical data: (a) patients on HAART (+) vs. untreated patients (−) at the moment of sample collection, (b) diagnosis of pulmonary (P) or extrapulmonary (E) TB, and (c) individuals on anti-tuberculous treatment -less than two weeks- (+) vs. untreated patients (−) at the moment of sample collection. Each symbol represents an individual subject. Unpaired t test or Mann-Whitney U test, as appropriated *p < 0.05 and **p < 0.01
The association between clinical data and the ratio among Th1, Th17, Tregs, uTregs and CD4 + Foxp3+ T subsets was also analyzed. A higher Th1/FoxP3 ratio was related with greater CD4 + T cell counts and nadir values. Furthermore, Th1/FoxP3, Th1/Tregs and Th1/uTregs ratios were increased in patients on HAART and diagnosed with lung-restricted TB infection (Fig. 8a and b). In addition, a greater Th1/Tregs balance positively correlated with total and nadir CD4 + T cell counts, CD4/CD8 T cell ratio and had negative association with viral load (Table 3). On the other hand, Th17/FoxP3 ratio positively correlated with greater values of total and nadir CD4 + T cell counts and with pulmonary TB (Table 3). Moreover, Th17/Treg ratio was associated with higher CD4 + T cell counts and individuals on HAART. Lastly, Th17/uTregs ratio was greater in HIV-TB patients with pulmonary TB compared with those with a disseminated disease. No associations were found between the balance of CD4+ Th subpopulations and anti-TB treatment, probably due to the low number of untreated patients at the time of recruitment (Fig. 8b).
Fig. 8.
Association between the CD4 + T cell subsets ratio with clinical parameters in HIV-TB patients. Phenotype of CD4 + T cell subset ratios from HIV-TB were correlated with clinical data: (a) patients on HAART (+) vs. untreated patients (−) at the moment of sample collection, (b) diagnosis of pulmonary (P) or extrapulmonary (E) TB, and (c) individuals on anti-tuberculous treatment -less than two weeks- (+) vs. untreated patients (−) at the moment of sample collection. Each symbol represents an individual subject. Unpaired t test or Mann-Whitney U test, as appropriated *p < 0.05 and **p < 0.01
Table 3.
Correlation between clinical parameters and ratios of different CD4+ subpopulations. A comparison of different CD4+ subsets ratios was performed with total and nadir CD4 + T cell counts, CD4/CD8 ratio and viral load. Number of values (n), Spearman’s rank correlation coefficient (r), and p value are indicated
| Clinical parameter | Th1/ FoxP3 |
Th1/ Tregs |
Th1/ uTregs |
Th1/ Th17 |
Th17/ FoxP3 |
Th17/ Tregs |
Th17/ uTregs |
|---|---|---|---|---|---|---|---|
| CD4 + T cell count (cell/ml) | n = 15 | n = 14 | n = 15 | n = 14 | n = 13 | n = 13 | n = 13 |
| r = 0.59 | r = 0.70 | r = 0.55 | r = 0.055 | r = 0.57 | r = 0.65 | r = 0.24 | |
| p = 0.011 | p = 0.0026 | p = 0.018 | p = 0.43 | p = 0.024 | p = 0.18 | p = 0.44 | |
| Nadir CD4 T cell count (cell/ml) | n = 15 | n = 14 | n = 15 | n = 14 | n = 14 | n = 14 | n = 14 |
| r = 0.60 | r = 0.66 | r = 0.36 | r = 0.37 | r = 0.56 | r = 0.48 | r = 0.20 | |
| p = 0.025 | p = 0.018 | p = 0.14 | p = 0.15 | p = 0.048 | p = 0.16 | p = 0.58 | |
| CD4/CD8 T cell ratio | n = 15 | n = 14 | n = 15 | n = 14 | n = 13 | n = 13 | n = 13 |
| r = 0.25 | r = 0.49 | r = 0.20 | r = 0.23 | r = 0.22 | r = 0.33 | r = 0.14 | |
| p = 0.19 | p = 0.036 | p = 0.23 | p = 0.21 | p = 0.23 | p = 0.26 | p = 0.65 | |
| Viral Load (copies/ml) | n = 15 | n = 15 | n = 15 | n = 14 | n = 14 | n = 14 | n = 14 |
| r = −0.37 | r = −0.51 | r = − 0.38 | r = − 0.34 | r = − 0.25 | r = − 0.28 | r = 0.19 | |
| p = 0.087 | p = 0.027 | p = 0.081 | p = 0.12 | p = 0.19 | p = 0.33 | p = 0.52 |
Mtb alters CD4+ T cell phenotype in vitro
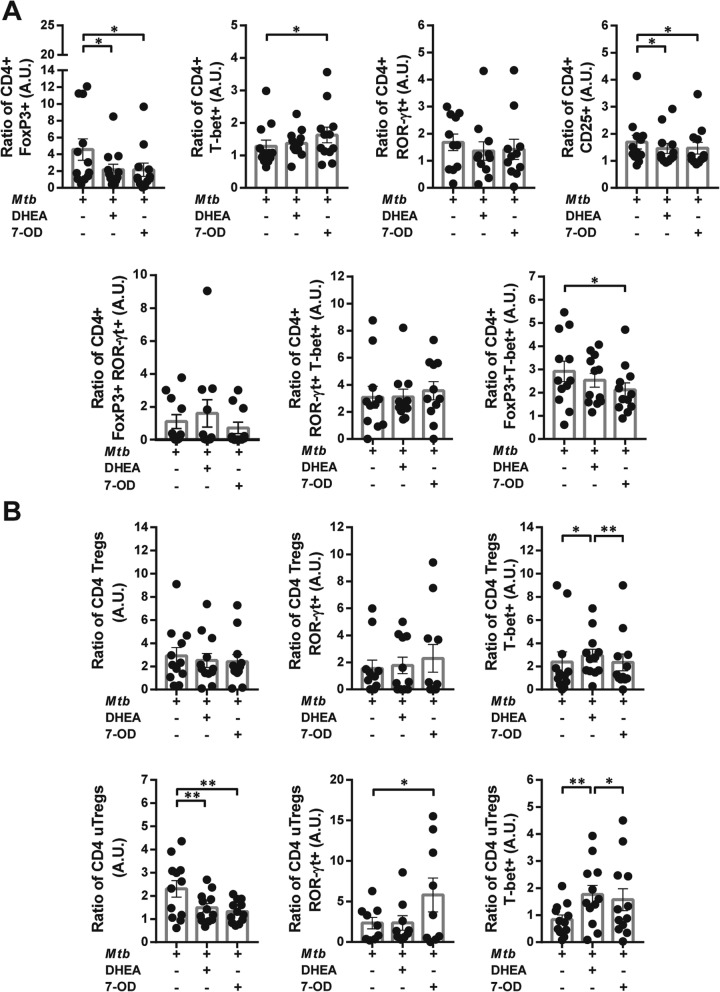
The capacity of Mtb to modulate the profile of CD4+ T cell subpopulations in vitro was analyzed. Mtb stimulation increased the frequency of CD4 + CD25+ and CD4 + FoxP3 cells, but it did not modify the proportion of ROR-γt and T-bet in HIV-TB patients (Fig. 9 a). The same pattern was detected in HD, with the exception of greater proportion of CD4 + T-bet+ (Additional file 2: Figure S2 A). Besides, Mtb increased the frequency of CD4 + T cells that expressed two transcription factors simultaneously in both HIV-TB and HD (Fig. 9 b and Additional file 2: Figure S2 B). In addition, Mtb-stimulation induced a higher expression of CD25 on a per cell basis and downregulated ROR-γt expression (Additional file 5: Table S1) in both study cohorts.
Fig. 9.
Mtb induces changes in CD4+ T-cell phenotype from HIV-TB patients. Recently thawed or freshly isolated PBMCs from HIV-TB individuals were stimulated with an antigen of Mtb, stained and analyzed by flow cytometry, as indicated in methods. Figure shows the percentage of CD4 + T cells that express (a) FoxP3, T-bet, ROR-γt or CD25 and (b) the co-expression of transcription factors using a Boolean gating strategy. c Tregs (CD4+ CD25+ FoxP3+) and uTregs (CD4+ CD25- FoxP3+) subsets were also analyzed. Representative flow cytometry examples are shown. Each symbol represents an individual subject. Unpaired t test or Mann-Whitney U test, as appropriated *p < 0.05, **p < 0.01 and ***p < 0.005
Mtb stimulation also augmented the frequency of Tregs and uTregs in both HIV-TB individuals and HD (Fig. 9c and Additional file 2: Figure S2 C). However, there were no changes in the proportion of cells that expressed ROR-γt and T-bet within these populations (data not shown). Finally, we contrasted the changes induced by Mtb between groups and observed significant differences in the frequency of CD4+ T-bet+ and CD4 + FoxP3 + ROR-γt + cells, finding a greater proportion of these populations in HD (Fig. 10).
Fig. 10.
Comparative CD4 + T cell response from HD and HIV-TB cohorts to Mtb. Recently thawed or freshly isolated PBMCs from HD and HIV-TB individuals were stimulated with an antigen of Mtb, stained and analyzed by flow cytometry, as indicated in methods. The results are plotted for HIV-TB (black circles) patients and HD (open circles). Each symbol represents an individual subject. The figure shows the percentage of CD4 + T cells that express (a) FoxP3, T-bet, ROR-γt or CD25 and (b) the co-expression of transcription factors using a Boolean gating strategy. (c) Tregs (CD4 + CD25 + FoxP3+) and uTregs (CD4 + CD25-FoxP3+) subsets and the co-expression of ROR-γt or T-bet within each subpopulation were analyzed. Values are relativized to unstimulated cells. Error bars show mean ± SEM. Unpaired t test or Mann-Whitney U test, as appropriated *p < 0.05 and **p < 0.01
7-OD modifies Mtb-specific immune response in the context of HIV and Mtb infection
As 7-OD modulated Mtb-specific lymphoproliferation and cytokine production, we aimed to study its effect on CD4+ Th cell phenotype. Remarkably, 7-OD increased the frequency of CD4+ T-bet and reduced the frequency of CD4 + FoxP3 + T-bet+ in HIV-TB patients. Furthermore, both 7-OD and DHEA diminished the percentage of cells expressing CD25+ or FoxP3+, and restored the effect of Mtb on CD4+ lymphocytes (Fig. 11a). Within the cohort of HD, while 7OD raised the frequency of CD4+ T-bet+ cells, DHEA expanded the CD4 + FoxP3 + RORγ-t + population and declined the number of cells that co-expressed FoxP3+ and T-bet after antigen (Ag)-exposure (Additional file 3: Figure 3A).
Fig. 11.
Mtb-specific response in HIV-TB patients is modulated differentially by 7-OD and DHEA. Recently thawed or freshly isolated PBMCs from HIV-TB patients were stimulated with an antigen of Mtb in the presence/absence of 7-OD at 1 × 10−6M or DHEA at 1 × 10−7M. Then, cells were stained and analyzed by flow cytometry, as described before. This figure shows the percentage of CD4 + T cells that express (a) FoxP3, T-bet, ROR-γt or CD25 and the co-expression of transcription factors using a Boolean gating strategy or (b) CD4+ Tregs and uTregs, with the co-expression of the transcription factors ROR-γt or T-bet within each population. Values are relativized to unstimulated cells. The results are plotted for HIV-TB patients. Each symbol represents an individual subject. Friedman test followed by post-hoc comparisons: Fisher’s LSD or Dunn’s test, as appropriated *p < 0.05, **p < 0.01 and ***p < 0.005
Concerning regulatory populations, treatment with both 7-OD and DHEA decreased the frequency of uTregs without modifying Tregs in HIV-TB individuals (Fig. 11b), while both Tregs and uTregs in HD declined in the presence of DHEA (Additional file 3 Fig. S3B). Besides, DHEA enhanced the expression of T-bet in Tregs and uTregs in the group of HIV-TB (Fig. 11b), while 7-OD augmented the proportion of uTregs expressing ROR-γt in HIV-TB and HD (Fig. 11b and Additional file 3: Figure S3B). Together, these data suggest that both 7-OD and DHEA are able to modify the effect of Mtb on CD4+ Th cell phenotype, but only 7-OD modulates it toward a Th1-like profile.
The expression of each TF was also analyzed on a per cell basis. Our data reveals that 7-OD enhanced the levels of T-bet in CD4 + T cells from HIV-TB (Additional file 5: Table S1). However, this did not occur in HD individuals (data not shown). In conclusion, 7-OD not only modified the percentage of cells expressing different TF, but also changed their levels of expression in HIV-TB patients, partially restoring the effect of Mtb on these populations.
The balance among different CD4 + T cell subpopulations is disturbed by Mtb and modified by 7-OD and DHEA
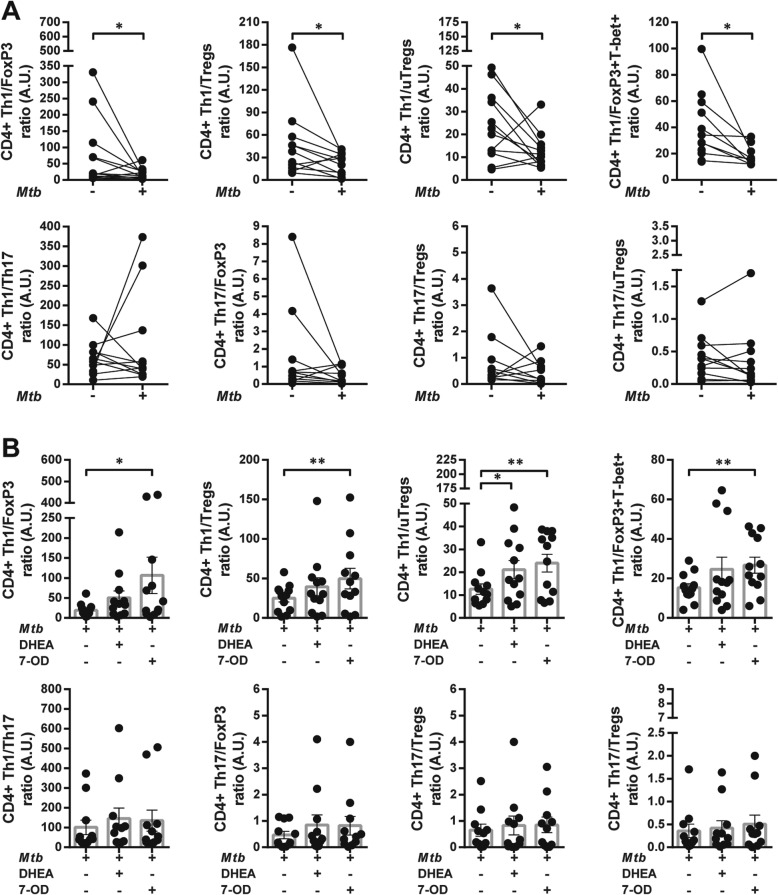
In order to study the changes that Mtb induced on the CD4 + T cell phenotype, TF ratio among different subpopulations was analyzed. The stimulation of PBMCs with Mtb reduced Th1/FoxP3, Th1/Tregs and Th1/FoxP3 + T-bet+ ratios in both HD and HIV-TB cohorts (Fig. 12a and Additional file 4: Figure S4A). Additionally, in co-infected individuals, Th1/uTregs ratio was also decreased (Fig. 12a). No variations in Th1/Th17 and Th17/regulatory ratios were observed in coinfected patients (Fig. 12a), but we found that Mtb reduced Th17/FoxP3 balance in HD (Additional file 4: Fig. S4A).
Fig. 12.
The equilibrium between Th1, Tregs, uTregs and Th17 subsets are disrupted by Mtb and modified by 7-OD and DHEA. Recently thawed or freshly isolated PBMCs from HIV-TB individuals were stained and analyzed by flow cytometry, as indicated in methods. Figure shows CD4 + T cell subset ratios. The results are plotted for HIV-TB patients (black circles) comparing (a) Control vs. stimulated cells. Unpaired t test or Mann-Whitney U test, as appropriated *p < 0.05. (b) Distinct CD4+ Th subset ratios of Mtb-stimulated PBMCs treated with 7-OD and DHEA. Each symbol represents an individual subject. Friedman test followed by post-hoc comparisons: Fisher’s LSD or Dunn’s test, as appropriated *p < 0.05, **p < 0.01 and ***p < 0.005
We wondered if the unbalance in CD4+ Th cells elicited by Mtb may be restored by in vitro treatment with 7-OD and/or DHEA. Remarkably, 7-OD induced an increase in Th1 population over CD4+ FoxP3+, Tregs, uTregs and FoxP3 + T-bet+ T cells in HIV-TB patients. Additionally, DHEA also raised Th1/uTregs ratio in co-infected individuals (Fig. 12b). On the other hand, within the group of HD, 7-OD augmented Th1/FoxP3 and Th1/Tregs ratios while DHEA had the same effect on Th1/uTregs balance. Lastly, in the same cohort, DHEA diminished Th1/Th17 ratio (Additional file 4: Figure S4B). These data demonstrate that CD4 + T cells develop a specific immunity toward certain Th phenotype through interactions with Mtb antigens, associated with a regulatory pathway. Nevertheless, this immunosuppressive milieu was overturn by the influence of 7-OD, which allowed the appropriate expansion of Th1 CD4 + T cells, an important subset for disease resolution.
Discussion
HIV deleterious effects over TB infection are diverse, involving the specific and lasting effect on Mtb-specific immunity. The type of individual immune response will determine disease progression or its development into a chronic stage. Novel host-directed therapies (HDT) that potentiate immunity could be discovered by studying and identifying factors that lead to disease progression or resolution. Therefore, the aim of this research was to scrutinize the role of 7-OD on lymphocyte phenotype and function during HIV-TB coinfection.
The ability to proliferate and differentiate into cytokine-producing effector cells is an important characteristic to pathogen control [27]. We found that patients exhibited higher proliferative response compared to HD, which can be explained by Mtb-specific T cell clones in HIV-TB. HD responded to Ag-stimulation owing to previous BCG immunization or exposure to environmental mycobacteria [28]. Previous reports also demonstrated HIV-TB subjects maintain Mtb-specific proliferative capacity, regardless of the low absolute CD4 + T cells counts and the rapid depletion of Mtb-specific responses after HIV infection [23, 29]. The current results show that 7-OD induces an augmented lymphoproliferation, while DHEA seems to suppress this activity. The lack of modulation by DHEA was reported formerly in PBMCs from TB patients [30]. Thus, our data indicate that concentrations of 7-OD superior to a physiologic range may enhance Mtb-specific immune lymphoproliferative response during HIV-TB coinfection.
Because of inter-individual variability, the use of cytokine ratios rather than single cytokine measurements would improve the study of immune responses. We demonstrate that the production of the protective cytokines IFN-γ and TNF-α is accompanied by the secretion of IL-17A in confected individuals, suggesting a heightened cytokine inflammatory response. Interestingly, HIV-TB individuals displayed reduced IFN-γ/IL-10, IFN-γ/IL-17A and IFN-γ/TNF-α ratios compared to HD. Other authors described a lower ratio of IFN-γ/IL-10 in drug-resistant patients during TB-Immune Reconstitution Inflammatory Syndrome (IRIS), which correlated with higher bacterial load [31]. In systemic lupus erythematosus, it was reported that TNF-α could be protective, since its levels and TNF-α/IL-10 ratio were lower in patients with active disease [32]. Furthermore, it was found higher IFN-γ/TNF-α ratio in household contacts when compared to TB patients [33]. Remarkably, 7-OD modified the impaired immune response against Mtb in HIV-TB patients, with an augment in IFN-γ and TNF-α secretion as well as an increment in IFN-γ/IL-10 and TNF-α/IL-10 ratios. The effect of 7-OD in lymphoproliferation and cytokine production in response to Mtb may induce a protective Th1-like response in the context of HIV-TB coinfection.
Then, we investigated CD4 + T cell phenotype of PBMCs from HD and patients ex vivo. We observed that HIV-TB individuals exhibited lower frequencies of CD4+ ROR-γt + T cells but greater proportions of CD4 + T-bet+, CD4 + FoxP3 + T-bet+ and uTregs (CD4 + CD25-FoxP3+). We found that higher proportions of the CD4 + ROR-γt + subset were associated with those patients on TB treatment vs. untreated individuals. Although its role in human pathology has not yet been fully elucidated, a mouse model revealed that cells expressing ROR-γt + were critical to the induction of an Mtb-specific memory response [34, 35]. Additionally, we observed larger frequencies of the CD4 + T-bet+ population that were associated with better clinical status and propitious disease outcome. These results are in line with the role of T-bet as the master regulator of Th1 cells, involved in the protective and inflammatory responses during Mtb infection [7, 24]. Conversely, we associated both uTregs and CD4 + FoxP3 + T-bet+ T cells with an unfavorable outcome. Furthermore, uTregs were found in greater proportion in those individuals with disseminated TB. A recent study revealed that a cohort of HIV+ individuals with latent TB exhibited a reduction in CD4 + FoxP3 + T-bet+ subset, which correlated inversely with viral load [27]. Our results indicated that this population is associated with an unfavorable result, but differences may be due to a different recruited cohort and that the authors studied Ki67+ cells CD4 + T cells [36]. Otherwise, our previous results observed an increased frequency of uTregs during HIV-TB disease [25] and described that this population exerts regulatory functions, in spite of an altered surface expression of markers and differences in cytokine production [37]. In line with this, lower Th1/uTregs ratio was observed in those patients who were not on HAART and who developed a disseminated TB infection. Our data therefore suggest that the expansion of uTregs population may be involved in the pathogenesis of TB.
Ex vivo analysis also showed that Th1/FoxP3 and Th1/Tregs ratios were higher in HIV-TB cohort. However, superior proportion of Th1 cells as well as Th1/FoxP3 and Th1/Tregs ratios positively correlated with total and nadir CD4 + T cell counts and with those individuals who presented pulmonary instead of disseminated TB. Similarly, higher proportions of Th1 population over CD4 + FoxP3+ and Tregs were associated with patients on HAART, while Th1/Tregs were related with a higher CD4/CD8 T cell ratio and negatively correlated with viral load. We hypothesize that the balance between Th1 and FoxP3+ or Tregs cells may have a critical role in the immune response during HIV-TB, as these parameters are related with clinical data associated with disease consequences, as we discussed in a previous report [17].
Afterwards, we analyzed in vitro Mtb stimulation on CD4+ Th phenotype. Ag-stimulation raised the proportion of CD4 + FoxP3+, Tregs and uTregs in HIV-TB patients. The proportion of Treg in pleural fluid inversely correlated with Mtb-specific immunity at the site of infection [38]. Higher frequencies of Treg suggest that these cells participate in immunosuppression observed in individuals with more severe active disease [38, 39] and persist beyond completion of anti-TB therapy [40]. Furthermore, in HIV-TB patients we observed a significant drop in frequencies of uTregs in response to TB treatment (unpublished data). While a Th1 response orchestrated immunity to intracellular pathogens, CD4 + T cells with a regulatory phenotype were associated with long-term adverse outcomes [27, 41]. Therefore, the balance between effector and suppressive immune responses is essential to control Mtb infection.
Hormone effect on CD4 + T cell lineage in a HIV-TB milieu was studied. Of note, 7-OD reduced the frequency of CD4 + T cells with a regulatory function and led to an augment in the frequency of CD4 + T-bet+ subset. This hormone also increased the proportion of cells expressing ROR-γt within uTregs. Although we could not find any association between the former population and clinical data, studies performed in mice and humans demonstrated that Tregs can be reprogrammed into IL-17 + Foxp3+ T cells, which are believed to control pathological inflammation, potentially antagonizing pro-inflammatory IL-17-producing cells [42].
There may be an association between a transcription factor or marker expression levels and a functional capacity [43, 44]. The assessment of MFI revealed that Mtb-stimulation induced higher levels of CD25 and downregulated ROR-γt expression on total CD4 + T cells. A research work found that ROR-γt expression was diminished in tuberculin skin test (TST) positive individuals, suggesting that lack of Th17 cells predisposes to latent infection [45]. At the same time, an augment in the expression of CD25 was associated with a regulatory phenotype in the context of TB [40, 46] and HIV [47]. Our data demonstrated that 7-OD modified the effect of Mtb on CD4+ lymphocytes by augmenting the expression of ROR-γt and T-bet, potentially enhancing their immune functions. In summary, Mtb boosts the frequency of CD4 + T cells that expresses FoxP3 and modified the expression of CD25 and ROR-γt on a per cell basis. However, 7-OD could reverse this effect by changing the frequency of effector Th1 cells and regulatory CD4 + T cells and the levels of transcription factors, improving thus its functionality.
It is known that Mtb infection induces highly polyfunctional responses [48], stimulating the co-expression of FoxP3, T-bet and/or ROR-γt in CD4 + T lymphocytes and that HIV infection alters the transcriptional profiles of these cells [27]. Our findings showed that exposure to Mtb antigens promoted the expansion of those populations in HD and coinfected patients. Cells producing both IL-17 and IL-10 were considered to play a protective regulatory homeostatic role, while CD4+ Th17 population co-secreting IFN-γ were associated with pathology [49]. CD4 + FoxP3 + ROR-γt + lymphocytes have the capacity to produce IL-17 and exhibit suppressive functions via a cell-cell contact mechanism and the secretion of IL-10 [42, 50]. In subjects with latent TB significantly higher frequencies of regulatory IL10 + Th17 were found compared to subjects with extrapulmonary TB when PBMCs were stimulated with latency antigens. Conversely, the response to ESAT6/CFP10 was predominantly managed by IFN-γ + Th17 T cells [49]. In this report, we found that higher frequencies of ROR-γt + T-bet+ CD4 + T cells in the cohort of HIV-TB patients were associated with greater levels of nadir CD4 + T cell counts, although we could not find any effects after the addition of hormones.
Recent studies demonstrated that CD4 + T cells expressing both, T-bet and FoxP3, exert regulatory rather than pro-inflammatory functions. T-bet is required for the expansion of these immunosuppressive cells to limit Th1-mediated immune responses [51, 52]. We hypothesized this subset might be involved in pathogen-specific loss of host resistance. In this report we found that Mtb augmented the proportion of CD4 + FoxP3 + T-bet+ cells, which was negatively associated with peripheral CD4 + T cell numbers, whereas treatment with 7-OD reduced the frequency of this subpopulation. Thus, the results indicate that 7-OD promotes an immunoprotective profile, not only by increasing the number of Th1 cells and the expression of T-bet per cell, but also by decreasing specific Th1 inhibitory (i.e., CD4 + FoxP3 + T-bet+ cells) responses.
Finally, we examined Th profile ratios inasmuch as the balance between Th1 and regulatory pathways might define resolution of Mtb infection and disease outcome. The role of Th cells has been investigated from active disease to post-treatment status [41, 53]. One report showed a reduction in Th1 cells in active-TB patients, followed by an increase in these cells after clinical cure [54]. Another report informed about an increase in the percentage of Mtb-specific Tregs during active TB, with lesser Th1/Treg and Th17/Treg ratios [54]. We observed that Mtb stimulation increased the numbers of lymphocytes with a regulatory phenotype over Th1 CD4 + T cells (lower Th1/FoxP3, Th1/Tregs, Th1/uTregs and Th1/FoxP3 + T-bet+ ratios in patients). Other authors suggested that it would be useful for monitoring progression of infection to focus on the study of the relative balance between Th1/regulatory cells rather than on the analysis of a specific cell subset [55]. We discovered 7-OD and DHEA augmented Th1/uTregs ratio, but only 7-OD could reverse the effect of Mtb in terms of the balance of Th1/FoxP3, Th1/Tregs and Th1/FoxP3 + T-bet+ in HIV-TB individuals.
There is little prior information about the mechanism of action of 7-OD on eukaryotic cells, but no data about its effects on CD4+ T lymphocytes. It was reported that 7-OD modulated cell metabolic activity by rising the levels of mitochondrial glycerol-3-phosphate dehydrogenase (GPD2) and malic enzyme in rats [56, 57]. Also, it was demonstrated that GPD2 participates in glycolysis, gluconeogenesis, glycerol and triacylglycerol metabolism [58]. Moreover, GPD2 is an alternative source of mitochondrial reactive oxygen species (mROS) [59], which are required for CD4+ T cell activation and expansion [60]. It was shown that reduction of GPD2 levels leads to decrease in ROS-mediated oxidative signal and the induction of NF-κB-dependent gene expression [59]. Likewise, NF-κB activation in T cells promoted clonal expansion during the Th1 response, Th1 differentiation and production of IL-2, IFN-γ and TNF-α [61, 62]. We therefore hypothesize that 7-OD stimulates GPD2 abundance and/or activity conducing to ROS production, NF-κB genes expression and the differentiation of CD4+ T cells to a Th1 subset. However, further research is needed to corroborate this hypothesis, given the unexplored nature of the effects of 7-OD on this cell type. Taking into account our results and the information from other authors, we can infer that both mechanisms are working in patients, since the effect at the cellular as well as the systemic level are operating. Thus, we hypothesize that 7-OD-induced modulation of 11β-HSD1 activity [63] from peripheral tissues could regulate cortisol levels and, in parallel, 7-OD stimulation of GPD2 on CD4+ T cells could impact on its metabolism, enhancing T cell activation, proliferation and Th1 differentiation. The differential effect of 7-OD in patients and HD may be caused by a higher number of Mtb-specific clones in HIV-TB individuals and/or a different metabolic state in CD4+ T cells that make them suitable to be modulated by 7-OD.
Conclusions
In summary, our results suggest that Mtb modulate CD4 + T cell phenotype and functionality in HIV-TB patients. Besides, we described the effect of 7-OD on lymphoproliferation, cytokine production profile and CD4+ Th cell subpopulations in vitro. Firstly, 7-OD augmented the Mtb-induced proliferative capacity of PBMCs from the study population. Secondly, this hormone enhanced IFN-γ and TNF-α production, without suppressing the necessary action of IL-10 and IL-17A. Moreover, 7-OD promoted the secretion of IFN-γ and TNF-α over IL-10, therefore modifying the cytokine balance towards a more favorable profile for Mtb control. Third, while Mtb promoted a regulatory CD4 + T cell phenotype, 7-OD expanded CD4 + T-bet+ subpopulation and enhanced T-bet expression, which were related to improved clinical outcomes. Furthermore, 7-OD decreased the percentage of CD4 + T cells with immunosuppressive functions. Finally, 7-OD increased Th1/regulatory ratio, indicating the predominance of a Th1 response which may contribute to TB protection by secreting IFN-γ and activating anti-mycobacterial mechanisms. These results provide new data to delineate novel strategies that accompany the treatment of TB, by modulating T cell-mediated immunity against Mycobacterium tuberculosis.
Supplementary information
Additional file 1: Figure S1. A representative analysis of the gating strategy developed during this study. Recently thawed or freshly isolated PBMCs were stained and analyzed by flow cytometry. The results are plotted for a sample from a HD labelled with (A) fluorochrome-conjugated Abs or (B) isotype control mAbs. Lymphocyte subset data were generated using a FSC-A/SSC-A gate. After doublet exclusion (FSC-A/FSC-H and FSC-H/FSC-W), live cells were selected using Live/Dead viability probe. Then, CD3 + CD4+ cells were gated and within this group, a gating on CD25+, FoxP3+, ROR-γt + or T-bet+ was done. CD4 + T cells that expressed one or two transcription factors were evaluated using Boolean combination gates. Representative flow cytometry examples are shown
Additional file 2: Figure S2. CD4+ T-cell phenotype is modified by Mtb. Recently thawed or freshly isolated PBMCs from HD were stimulated with Mtb, stained and analyzed by flow cytometry, as indicated in methods. Figure shows the percentage of CD4 + T cells that express (A) FoxP3, T-bet, ROR-γt or CD25 and (B) the co-expression of transcription factors using a Boolean gating strategy. (C) Tregs (CD4 + CD25 + FoxP3+) and uTregs (CD4 + CD25-FoxP3+) subsets were also analyzed. Representative flow cytometry examples are shown. Each symbol represents an individual subject. Unpaired t test or Mann-Whitney U test, as appropriated *p < 0.05, **p < 0.01 and ***p < 0.005
Additional file 3: Figure S3. Mtb-specific response in HD is modulated by 7-OD and DHEA. Recently thawed or freshly isolated PBMCs from HD were stimulated with Mtb in the presence/absence of 7-OD at 1 × 10−6M or DHEA at 1 × 10−7M. Then, cells were stained and analyzed by flow cytometry, as described before. Figure shows the percentage of CD4 + T cells expressing (A) FoxP3, T-bet, ROR-γt or CD25 and the co-expression of transcription factors using a Boolean gating strategy or (B) CD4+ Tregs and uTregs, with the co-expression of the transcription factors ROR-γt or T-bet within each population. Values are relativized to unstimulated cells. The results are plotted for HD. Each symbol represents an individual subject. Friedman test followed by post-hoc comparisons: Fisher’s or Dunn’s test, as appropriated *p < 0.05, **p < 0.01 and ***p < 0.005
Additional file 4: Figure S4. The equilibrium between Th1, Tregs, uTregs and Th17 subsets are disrupted by Mtb and modified by 7-OD and DHEA. Recently thawed or freshly isolated PBMCs from HD individuals were stained and analyzed by flow cytometry, as indicated in methods. Figure shows CD4 + T cell subset ratios. The results are plotted for HD (open circles) comparing (A) Control vs. stimulated cells. Unpaired t test (normal distribution) or Mann-Whitney U test (non-normal variables) *p < 0.05. (B) Distinct CD4+ Th subset ratios of Mtb-stimulated PBMCs treated with 7-OD and DHEA. Each symbol represents an individual subject. Friedman test followed by post-hoc comparisons: Fisher’s LSD or Dunn’s test, as appropriated *p < 0.05, **p < 0.01 and ***p < 0.005
Additional file 5: Table S1. Expression of FoxP3, T-bet, ROR-γt and CD25 on a per cell basis in CD4 + T lymphocytes from HIV-TB patients. Recently thawed or freshly isolated PBMCs from HIV-TB were stimulated with Mtb in the presence/absence of 7-OD at 1 × 10−6M or DHEA at 1 × 10−7M. Then, cells were stained and analyzed by flow cytometry, as described before. Table shows median fluorescence intensity (MFI), which was calculated as the ratio of the geometric mean MFI of the marker of interest over MFI of the corresponding negative population. MFI is expressed as median ± interquartile range (IQR). Friedman test followed by Fisher’s LSD or by Dunn’s test, as appropriate *p < 0.05, **p < 0.01 and ***p < 0.005. * indicates significant differences with Mtb-stimulated cells (Mtb)
Acknowledgements
The authors would like to thank the study subjects for their participation, Dr. Gabriela Turk and Dr. Pedro Cahn for continuous support, Dr. Virginia Gonzalez Polo and Dr. Carla A. Pascuale for technical assistance on flow cytometry experiments, and Mr. Sergio Mazzini for revising the manuscript.
Abbreviations
- 7-OD
7-oxo-dehydroepiandrosterone, 7-oxo-DHEA
- AET
Androstenetriol
- Ag
Antigen
- Anti-TB
Antituberculosis treatment
- CPM
Counts per minute
- DHEA
Dehydroepiandrosterone
- HAART
Highly active antiretroviral therapy
- HD
Healthy donors
- HDT
Host-directed therapies
- HIV
Human immunodeficiency virus
- HIV-TB
HIV-Mtb coinfected patients or HIV-Mtb coinfection
- IRIS
Immune Reconstitution Inflammatory Syndrome
- MFI
Mean fluorescence intensity
- Mtb
Mycobacterium tuberculosis
- PBMCs
Peripheral blood mononuclear cells
- SEM
Standard error of the mean
- TB
Tuberculosis
- TF
Transcription factor
- Tregs
Conventional regulatory CD4 + T cells (i.e., CD25 + FoxP3)
- TST
Tuberculin skin test
- uTregs
Unconventional regulatory CD4 + T cells (i.e., CD25-FoxP3+)
Authors’ contributions
MBV and MFQ conceived and designed experiments, analyzed and interpreted the data and wrote the manuscript. MBV processed samples and performed experiments. NL, OS and ML recruited patients, collected samples and obtained clinical data. HS contributed reagents/materials and analyzed and interpreted the data. All authors contributed to the refinement of the report and approved the final version.
Funding
This work was supported by Agencia Nacional de Promoción Científica y Tecnológica (PICT 2012–0059 to MFQ) and by Universidad de Buenos Aires (UBACyT 20020120200050BA to MFQ).
Availability of data and materials
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Ethics approval and consent to participate
The study was carried out in line with the World Medical Association Declaration of Helsinki and approved by the Ethics Committee of Fundación Huésped, Buenos Aires, Argentina. Written informed consent was provided by all the participants.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information accompanies this paper at 10.1186/s12929-019-0604-z.
References
- 1.Geldmacher Christof, Schuetz Alexandra, Ngwenyama Njabulo, Casazza Joseph P., Sanga Erica, Saathoff Elmar, Boehme Catharina, Geis Steffen, Maboko Leonard, Singh Mahavir, Minja Fred, Meyerhans Andreas, Koup Richard A., Hoelscher Michael. Early Depletion ofMycobacterium tuberculosis–Specific T Helper 1 Cell Responses after HIV‐1 Infection. The Journal of Infectious Diseases. 2008;198(11):1590–1598. doi: 10.1086/593017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tiberi S, du Plessis N, Walzl G, Vjecha MJ, Rao M, Ntoumi F, et al. Tuberculosis: progress and advances in development of new drugs, treatment regimens, and host-directed therapies. Lancet Infect Dis [Internet] 2018;18:e183–e198. doi: 10.1016/S1473-3099(18)30110-5. [DOI] [PubMed] [Google Scholar]
- 3.Wallis RS, Maeurer M, Mwaba P, Chakaya J, Rustomjee R, Migliori GB, et al. Tuberculosis—advances in development of new drugs, treatment regimens, host-directed therapies, and biomarkers. Lancet infect dis [Internet] Elsevier Ltd. 2016;16:e34–e46. doi: 10.1016/S1473-3099(16)00070-0. [DOI] [PubMed] [Google Scholar]
- 4.Ernst JD. The immunological life cycle of tuberculosis. Nat Rev Immunol [Internet]. Nature Publishing Group; 2012 [cited 2013 Feb 28];12:581–591. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22790178. [DOI] [PubMed]
- 5.Sallusto Federica. Heterogeneity of Human CD4+T Cells Against Microbes. Annual Review of Immunology. 2016;34(1):317–334. doi: 10.1146/annurev-immunol-032414-112056. [DOI] [PubMed] [Google Scholar]
- 6.Flynn J L, Chan J, Triebold K J, Dalton D K, Stewart T A, Bloom B R. An essential role for interferon gamma in resistance to Mycobacterium tuberculosis infection. The Journal of Experimental Medicine. 1993;178(6):2249–2254. doi: 10.1084/jem.178.6.2249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Vinicius M, Gomes M, Tiburcio S, Machado JR, Alexandre D, Silva A, et al. Complexity and controversies over the cytokine profiles of T helper cell subpopulations in Tuberculosis. J Immunol Res. 2015;2015:1–13. doi: 10.1155/2015/639107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lin Philana Ling, Plessner Hillarie L., Voitenok Nikolai N., Flynn JoAnne L. Tumor Necrosis Factor and Tuberculosis. Journal of Investigative Dermatology Symposium Proceedings. 2007;12(1):22–25. doi: 10.1038/sj.jidsymp.5650027. [DOI] [PubMed] [Google Scholar]
- 9.Stenger S. Immunological control of tuberculosis: role of tumour necrosis factor and more. Annals of the Rheumatic Diseases. 2005;64(suppl_4):iv24–iv28. doi: 10.1136/ard.2005.042531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Philips J A, Ernst JD. Tuberculosis pathogenesis and immunity. Annu Rev Pathol [Internet]. 2012 [cited 2013 mar 7];7:353–384. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22054143. [DOI] [PubMed]
- 11.Garg Neeraj K., Dwivedi Priya, Jain Ashay, Tyagi Shikha, Sahu Tejram, Tyagi Rajeev K. Development of novel carrier(s) mediated tuberculosis vaccine: More than a tour de force. European Journal of Pharmaceutical Sciences. 2014;62:227–242. doi: 10.1016/j.ejps.2014.05.028. [DOI] [PubMed] [Google Scholar]
- 12.Okamoto Yoshida Yuko, Umemura Masayuki, Yahagi Ayano, O’Brien Rebecca L., Ikuta Koichi, Kishihara Kenji, Hara Hiromitsu, Nakae Susumu, Iwakura Yoichiro, Matsuzaki Goro. Essential Role of IL-17A in the Formation of a Mycobacterial Infection-Induced Granuloma in the Lung. The Journal of Immunology. 2010;184(8):4414–4422. doi: 10.4049/jimmunol.0903332. [DOI] [PubMed] [Google Scholar]
- 13.Cruz Andrea, Fraga Alexandra G., Fountain Jeffrey J., Rangel-Moreno Javier, Torrado Egídio, Saraiva Margarida, Pereira Daniela R., Randall Troy D., Pedrosa Jorge, Cooper Andrea M., Castro António G. Pathological role of interleukin 17 in mice subjected to repeated BCG vaccination after infection with Mycobacterium tuberculosis. The Journal of Experimental Medicine. 2010;207(8):1609–1616. doi: 10.1084/jem.20100265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Suarez GV, Vecchione MB, Angerami MT, Sued O, Bruttomesso AC, Bottasso OA, et al. Immunoendocrine interactions during HIV-TB coinfection : implications for the design of new adjuvant therapies. Biomed Res Int. 2015;2015. [DOI] [PMC free article] [PubMed]
- 15.Angerami Matias, Suarez Guadalupe, Pascutti Maria Fernanda, Salomon Horacio, Bottasso Oscar, Quiroga Maria Florencia. Modulation of the phenotype and function of Mycobacterium tuberculosis-stimulated dendritic cells by adrenal steroids. International Immunology. 2013;25(7):405–411. doi: 10.1093/intimm/dxt004. [DOI] [PubMed] [Google Scholar]
- 16.Suarez GV, Angerami MT, Vecchione MB, Laufer N, Turk G, Ruiz MJ, et al. HIV–TB coinfection impairs CD8+ T-cell differentiation and function while dehydroepiandrosterone improves cytotoxic antitubercular immune responses. Eur J Immunol. 2015;45:2529–2541. doi: 10.1002/eji.201545545. [DOI] [PubMed] [Google Scholar]
- 17.Vecchione MB, Eiras J, Suarez GV, Angerami MT, Marquez C, Sued O, et al. Determination of dehydroepiandrosterone and its biologically active oxygenated metabolites in human plasma evinces a hormonal imbalance during HIV-TB coinfection. Sci Rep [Internet] 2018;8:6692. doi: 10.1038/s41598-018-24771-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Department of Health and Human Services. Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the Use of Antiretroviral Agents in Adults and Adolescents Living with HIV. [Internet]. 2018. Available from: http://www.aidsinfo.nih.gov/ContentFiles/ AdultandAdolescentGL.pdf. Accessed.
- 19.Cruz Andrea, Khader Shabaana A., Torrado Egidio, Fraga Alexandra, Pearl John E., Pedrosa Jorge, Cooper Andrea M., Castro António G. Cutting Edge: IFN-γ Regulates the Induction and Expansion of IL-17-Producing CD4 T Cells during Mycobacterial Infection. The Journal of Immunology. 2006;177(3):1416–1420. doi: 10.4049/jimmunol.177.3.1416. [DOI] [PubMed] [Google Scholar]
- 20.Bekker L.-G., Moreira A. L., Bergtold A., Freeman S., Ryffel B., Kaplan G. Immunopathologic Effects of Tumor Necrosis Factor Alpha in Murine Mycobacterial Infection Are Dose Dependent. Infection and Immunity. 2000;68(12):6954–6961. doi: 10.1128/IAI.68.12.6954-6961.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wherry E John. T cell exhaustion. Nature Immunology. 2011;12(6):492–499. doi: 10.1038/ni.2035. [DOI] [PubMed] [Google Scholar]
- 22.Iyasere C., Tilton J. C., Johnson A. J., Younes S., Yassine-Diab B., Sekaly R.-P., Kwok W. W., Migueles S. A., Laborico A. C., Shupert W. L., Hallahan C. W., Davey R. T., Dybul M., Vogel S., Metcalf J., Connors M. Diminished Proliferation of Human Immunodeficiency Virus-Specific CD4+ T Cells Is Associated with Diminished Interleukin-2 (IL-2) Production and Is Recovered by Exogenous IL-2. Journal of Virology. 2003;77(20):10900–10909. doi: 10.1128/JVI.77.20.10900-10909.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Siddiqui S., Sarro Y., Diarra B., Diallo H., Guindo O., Dabitao D., Tall M., Hammond A., Kassambara H., Goita D., Dembele P., Traore B., Hengel R., Nason M., Warfield J., Washington J., Polis M., Diallo S., Dao S., Koita O., Lane H.C., Catalfamo M., Tounkara A. Tuberculosis specific responses following therapy for TB: Impact of HIV co-infection. Clinical Immunology. 2015;159(1):1–12. doi: 10.1016/j.clim.2015.04.002. [DOI] [PubMed] [Google Scholar]
- 24.Lyadova I V., Panteleev A V. Th1 and Th17 Cells in Tuberculosis: Protection, Pathology, and Biomarkers. Mediators Inflamm. Hindawi Publishing Corporation; 2015;2015. [DOI] [PMC free article] [PubMed]
- 25.Quiroga MF, Angerami MT, Santucci N, Ameri D, Francos JL, Wallach J, et al. Dynamics of adrenal steroids are related to variations in Th1 and Treg populations during Mycobacterium tuberculosis infection in HIV positive persons. PLoS One [Internet]. 2012 [cited 2013 Mar 13];7:e33061. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3303789&tool=pmcentrez&rendertype=abstract [DOI] [PMC free article] [PubMed]
- 26.Angerami MT, Suarez GV, Vecchione MB, Laufer N, Ameri D, Ben G, et al. Expansion of CD25-negative forkhead box P3-positive T cells during HIV and mycobacterium tuberculosis infection. Front Immunol. 2017. [DOI] [PMC free article] [PubMed]
- 27.Riou C, Strickland N, Soares AP, Corleis B, Kwon DS, Wherry EJ, et al. HIV skews the lineage-defining transcriptional profile of Mycobacterium tuberculosis-specific CD4+ T cells. J Immunol. 2016;196:3006–3018. doi: 10.4049/jimmunol.1502094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Von Reyn CF, Horsburgh CR, Olivier KN, Barnes PF, Waddell R, Warren C, et al. Skin test reactions to Mycobacterium tuberculosis purified protein derivative and Mycobacterium avium sensitin among health care workers and medical students in the United States SUMMARY. Int J Tuberc Lung Dis. 2001;5:1122–1128. [PubMed] [Google Scholar]
- 29.Hammond Abdulrahman S., McConkey Samuel J., Hill Philip C., Crozier Sarah, Klein Michèl R., Adegbola Richard A., Rowland‐Jones Sarah, Brookes Roger H., Whittle Hilton, Jaye Assan. Mycobacterial T Cell Responses in HIV‐Infected Patients with Advanced Immunosuppression. The Journal of Infectious Diseases. 2008;197(2):295–299. doi: 10.1086/524685. [DOI] [PubMed] [Google Scholar]
- 30.Mahuad C., Bay M. L., Farroni M. A., Bozza V., Del Rey A., Besedovsky H., Bottasso O. A. Cortisol and Dehydroepiandrosterone Affect the Response of Peripheral Blood Mononuclear Cells to Mycobacterial Antigens during Tuberculosis. Scandinavian Journal of Immunology. 2004;60(6):639–646. doi: 10.1111/j.0300-9475.2004.01514.x. [DOI] [PubMed] [Google Scholar]
- 31.Skolimowska Keira H., Rangaka Molebogeng X., Meintjes Graeme, Pepper Dominique J., Seldon Ronnett, Matthews Kerryn, Wilkinson Robert J., Wilkinson Katalin A. Altered Ratio of IFN-γ/IL-10 in Patients with Drug Resistant Mycobacterium tuberculosis and HIV- Tuberculosis Immune Reconstitution Inflammatory Syndrome. PLoS ONE. 2012;7(10):e46481. doi: 10.1371/journal.pone.0046481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gómez D, Correa PA, Gómez LM, Cadena J, Molina JF, Anaya J-M. Th1/Th2 cytokines in patients with systemic lupus erythematosus: is tumor necrosis factor α protective? Semin Arthritis Rheum [Internet] 2004;33:404–413. doi: 10.1016/j.semarthrit.2003.11.002. [DOI] [PubMed] [Google Scholar]
- 33.Prabhavathi Maddineni, Pathakumari Balaji, Raja Alamelu. IFN-γ/TNF-α ratio in response to immuno proteomically identified human T-cell antigens of Mycobacterium tuberculosis – The most suitable surrogate biomarker for latent TB infection. Journal of Infection. 2015;71(2):238–249. doi: 10.1016/j.jinf.2015.04.032. [DOI] [PubMed] [Google Scholar]
- 34.Umemura M, Yahagi A, Hamada S, Begum MD, Watanabe H, Kawakami K, et al. IL-17-mediated regulation of innate and acquired immune response against pulmonary Mycobacterium bovis Bacille Calmette-Guerin infection. J Immunol. 2014;178:3786–3796. doi: 10.4049/jimmunol.178.6.3786. [DOI] [PubMed] [Google Scholar]
- 35.Khader SA, Bell GK, Pearl JE, Fountain JJ, Rangel-Moreno J, Cilley GE, et al. IL-23 and IL-17 in the establishment of protective pulmonary CD4+ T cell responses after vaccination and during Mycobacterium tuberculosis challenge. Nat Immunol [Internet] 2007;8:369–377. doi: 10.1038/ni1449. [DOI] [PubMed] [Google Scholar]
- 36.Riou Catherine, Strickland Natalie, Soares Andreia P., Corleis Björn, Kwon Douglas S., Wherry E. John, Wilkinson Robert J., Burgers Wendy A. HIV Skews the Lineage-Defining Transcriptional Profile of Mycobacterium tuberculosis–Specific CD4+ T Cells. The Journal of Immunology. 2016;196(7):3006–3018. doi: 10.4049/jimmunol.1502094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Angerami MT, Suarez G V., Vecchione MB, Laufer N, Ameri D, Ben G, et al. Expansion of CD25-Negative Forkhead Box P3-Positive T Cells during HIV and Mycobacterium tuberculosis Infection. Front Immunol [Internet]. 2017;8. Available from: http://journal.frontiersin.org/article/10.3389/fimmu.2017.00528/full [DOI] [PMC free article] [PubMed]
- 38.Chen X, Zhou B, Li M, Deng Q, Wu X, Le X, et al. CD4+CD25+FoxP3+ regulatory T cells suppress Mycobacterium tuberculosis immunity in patients with active disease. Clin Immunol. 2007;123:50–59. doi: 10.1016/j.clim.2006.11.009. [DOI] [PubMed] [Google Scholar]
- 39.Urdahl Kevin B. Understanding and overcoming the barriers to T cell-mediated immunity against tuberculosis. Seminars in Immunology. 2014;26(6):578–587. doi: 10.1016/j.smim.2014.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ribeiro-Rodrigues R., Resende Co T., Rojas R., Toossi Z., Dietze R., Boom W. H., Maciel E., Hirsch C. S. A role for CD4+CD25+ T cells in regulation of the immune response during human tuberculosis. Clinical and Experimental Immunology. 2006;144(1):25–34. doi: 10.1111/j.1365-2249.2006.03027.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Garra AO, Redford PS, Mcnab FW, Bloom CI, Wilkinson RJ, Berry MPR. The immune response in Tuberculosis. Annu Rev Immunol. 2013;32:475–527. doi: 10.1146/annurev-immunol-032712-095939. [DOI] [PubMed] [Google Scholar]
- 42.Lochner Matthias, Peduto Lucie, Cherrier Marie, Sawa Shinichiro, Langa Francina, Varona Rosa, Riethmacher Dieter, Si-Tahar Mustapha, Di Santo James P., Eberl Gérard. In vivo equilibrium of proinflammatory IL-17+ and regulatory IL-10+ Foxp3+ RORγt+ T cells. The Journal of Experimental Medicine. 2008;205(6):1381–1393. doi: 10.1084/jem.20080034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Buggert Marcus, Tauriainen Johanna, Yamamoto Takuya, Frederiksen Juliet, Ivarsson Martin A., Michaëlsson Jakob, Lund Ole, Hejdeman Bo, Jansson Marianne, Sönnerborg Anders, Koup Richard A., Betts Michael R., Karlsson Annika C. T-bet and Eomes Are Differentially Linked to the Exhausted Phenotype of CD8+ T Cells in HIV Infection. PLoS Pathogens. 2014;10(7):e1004251. doi: 10.1371/journal.ppat.1004251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Knox JJ, Cosma GL, Betts MR, McLane LM. Characterization of T-bet and Eomes in peripheral human immune cells. Front Immunol. 2014;5:1–13. doi: 10.3389/fimmu.2014.00217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Babu Subash, Bhat Sajid Q., Kumar N. Pavan, Kumaraswami V., Nutman Thomas B. Regulatory T Cells Modulate Th17 Responses in Patients with Positive Tuberculin Skin Test Results. The Journal of Infectious Diseases. 2010;201(1):20–31. doi: 10.1086/648735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zeng Chun, Shi Xiaoyun, Zhang Baojun, Liu He, Zhang Lianjun, Ding Wenjun, Zhao Yong. The imbalance of Th17/Th1/Tregs in patients with type 2 diabetes: relationship with metabolic factors and complications. Journal of Molecular Medicine. 2011;90(2):175–186. doi: 10.1007/s00109-011-0816-5. [DOI] [PubMed] [Google Scholar]
- 47.Kwon D. S., Angin M., Hongo T., Law K. M., Johnson J., Porichis F., Hart M. G., Pavlik D. F., Tighe D. P., Kavanagh D. G., Streeck H., Addo M. M., Kaufmann D. E. CD4+ CD25+ Regulatory T Cells Impair HIV-1-Specific CD4 T Cell Responses by Upregulating Interleukin-10 Production in Monocytes. Journal of Virology. 2012;86(12):6586–6594. doi: 10.1128/JVI.06251-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lin L, Finak G, Ushey K, Seshadri C, Hawn TR, Frahm N, et al. COMPASS identifies T-cell subsets correlated with clinical outcomes. Nat Biotechnol [Internet] 2015;33:610–616. doi: 10.1038/nbt.3187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Rakshit S, Adiga V, Nayak S, Sahoo PN, Sharma PK, van Meijgaarden KE, et al. Circulating Mycobacterium tuberculosis DosR latency antigen-specific, polyfunctional, regulatory IL10+ Th17 CD4 T-cells differentiate latent from active tuberculosis. Sci Rep [Internet] 2017;7:11948. doi: 10.1038/s41598-017-10773-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Voo KS, Wang Y-H, Santori FR, Boggiano C, Wang Y-H, Arima K, et al. Identification of IL-17-producing FOXP3+ regulatory T cells in humans. Proc Natl Acad Sci U S A. 2009;106:4793–4798. doi: 10.1073/pnas.0900408106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kachler K, Holzinger C, Trufa DI, Sirbu H, Finotto S. The role of Foxp3 and Tbet co-expressing Treg cells in lung carcinoma. Oncoimmunol [Internet]. Taylor & Francis; 2018;7:e1456612. Available from: https://doi.org/10.1080/2162402X.2018.1456612. [DOI] [PMC free article] [PubMed]
- 52.Koch Meghan A, Tucker-Heard Glady's, Perdue Nikole R, Killebrew Justin R, Urdahl Kevin B, Campbell Daniel J. The transcription factor T-bet controls regulatory T cell homeostasis and function during type 1 inflammation. Nature Immunology. 2009;10(6):595–602. doi: 10.1038/ni.1731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Simmons JD, Stein CM, Seshadri C, Campo M, Alter G, Fortune S, et al. Immunological mechanisms of human resistance to persistent Mycobacterium tuberculosis infection. Nat Rev Immunol [Internet] Springer US. 2018;18:575–589. doi: 10.1038/s41577-018-0025-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.da Silva MV, Massaro Junior VJ, Machado JR, Silva DAA, Castellano LR, Alexandre PBD, et al. Expression Pattern of Transcription Factors and Intracellular Cytokines Reveals That Clinically Cured Tuberculosis Is Accompanied by an Increase in Mycobacterium -Specific Th1, Th2, and Th17 Cells. Biomed Res Int [Internet] 2015;2015:1–14. doi: 10.1155/2015/591237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Singh Alpana, Vajpayee Madhu, Ali Sharique A., Chauhan Neeraj Kumar. Cellular interplay among Th17, Th1, and Treg cells in HIV-1 subtype “C” infection. Journal of Medical Virology. 2013;86(3):372–384. doi: 10.1002/jmv.23810. [DOI] [PubMed] [Google Scholar]
- 56.Lardy H, Kneer N, Wei Y, Partridge B, Marwah P. Ergosteroids II: Biologically active metabolites and synthetic derivatives of Dehydroepiandrosterone. Steroids [Internet] 1998;63:158–165. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0039128X97001591 [DOI] [PubMed]
- 57.Bobyleva V, Bellei M, Kneer N, Lardy H. The effects of the Ergosteroid 7-Oxo-dehydroepiandrosterone on mitochondrial membrane potential: possible relationship to thermogenesis. Arch Biochem Biophys [Internet] 1997;341:122–128. doi: 10.1006/abbi.1997.9955. [DOI] [PubMed] [Google Scholar]
- 58.Saheki T, Moriyama M, Kuroda E, Funahashi A, Yasuda I, Setogawa Y, et al. Pivotal role of inter-organ aspartate metabolism for treatment of mitochondrial aspartate-glutamate carrier 2 (citrin) deficiency, based on the mouse model. Sci Rep [Internet]. Springer US; 2019;9:4179. Available from: http://dx.doi.org/10.1038/s41598-019-39627-y [DOI] [PMC free article] [PubMed]
- 59.Kamiński MM, Sauer SW, Kamiński M, Opp S, Ruppert T, Grigaravičius P, et al. T cell activation is driven by an ADP-dependent Glucokinase linking enhanced glycolysis with mitochondrial reactive oxygen species generation. Cell Rep. 2012;2:1300–1315. doi: 10.1016/j.celrep.2012.10.009. [DOI] [PubMed] [Google Scholar]
- 60.Sena Laura A., Li Sha, Jairaman Amit, Prakriya Murali, Ezponda Teresa, Hildeman David A., Wang Chyung-Ru, Schumacker Paul T., Licht Jonathan D., Perlman Harris, Bryce Paul J., Chandel Navdeep S. Mitochondria Are Required for Antigen-Specific T Cell Activation through Reactive Oxygen Species Signaling. Immunity. 2013;38(2):225–236. doi: 10.1016/j.immuni.2012.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Corn Radiah A., Aronica Mark A., Zhang Fuping, Tong Yingkai, Stanley Sarah A., Kim Se Ryoung Agnes, Stephenson Linda, Enerson Ben, McCarthy Susan, Mora Ana, Boothby Mark. T Cell-Intrinsic Requirement for NF-κB Induction in Postdifferentiation IFN-γ Production and Clonal Expansion in a Th1 Response. The Journal of Immunology. 2003;171(4):1816–1824. doi: 10.4049/jimmunol.171.4.1816. [DOI] [PubMed] [Google Scholar]
- 62.McCracken Sharon A., Hadfield Katrina, Rahimi Zolaikha, Gallery Eileen D., Morris Jonathan M. NF-κB-regulated suppression of T-bet in T cells represses Th1 immune responses in pregnancy. European Journal of Immunology. 2007;37(5):1386–1396. doi: 10.1002/eji.200636322. [DOI] [PubMed] [Google Scholar]
- 63.Balázs Z, Nashev LG, Chandsawangbhuwana C, Baker ME, Odermatt A. Hexose-6-phosphate dehydrogenase modulates the effect of inhibitors and alternative substrates of 11beta-hydroxysteroid dehydrogenase 1. Mol Cell Endocrinol [Internet]. 2009 [cited 2013 may 1];301:117–122. Available from: http://www.ncbi.nlm.nih.gov/pubmed/19010388. [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Figure S1. A representative analysis of the gating strategy developed during this study. Recently thawed or freshly isolated PBMCs were stained and analyzed by flow cytometry. The results are plotted for a sample from a HD labelled with (A) fluorochrome-conjugated Abs or (B) isotype control mAbs. Lymphocyte subset data were generated using a FSC-A/SSC-A gate. After doublet exclusion (FSC-A/FSC-H and FSC-H/FSC-W), live cells were selected using Live/Dead viability probe. Then, CD3 + CD4+ cells were gated and within this group, a gating on CD25+, FoxP3+, ROR-γt + or T-bet+ was done. CD4 + T cells that expressed one or two transcription factors were evaluated using Boolean combination gates. Representative flow cytometry examples are shown
Additional file 2: Figure S2. CD4+ T-cell phenotype is modified by Mtb. Recently thawed or freshly isolated PBMCs from HD were stimulated with Mtb, stained and analyzed by flow cytometry, as indicated in methods. Figure shows the percentage of CD4 + T cells that express (A) FoxP3, T-bet, ROR-γt or CD25 and (B) the co-expression of transcription factors using a Boolean gating strategy. (C) Tregs (CD4 + CD25 + FoxP3+) and uTregs (CD4 + CD25-FoxP3+) subsets were also analyzed. Representative flow cytometry examples are shown. Each symbol represents an individual subject. Unpaired t test or Mann-Whitney U test, as appropriated *p < 0.05, **p < 0.01 and ***p < 0.005
Additional file 3: Figure S3. Mtb-specific response in HD is modulated by 7-OD and DHEA. Recently thawed or freshly isolated PBMCs from HD were stimulated with Mtb in the presence/absence of 7-OD at 1 × 10−6M or DHEA at 1 × 10−7M. Then, cells were stained and analyzed by flow cytometry, as described before. Figure shows the percentage of CD4 + T cells expressing (A) FoxP3, T-bet, ROR-γt or CD25 and the co-expression of transcription factors using a Boolean gating strategy or (B) CD4+ Tregs and uTregs, with the co-expression of the transcription factors ROR-γt or T-bet within each population. Values are relativized to unstimulated cells. The results are plotted for HD. Each symbol represents an individual subject. Friedman test followed by post-hoc comparisons: Fisher’s or Dunn’s test, as appropriated *p < 0.05, **p < 0.01 and ***p < 0.005
Additional file 4: Figure S4. The equilibrium between Th1, Tregs, uTregs and Th17 subsets are disrupted by Mtb and modified by 7-OD and DHEA. Recently thawed or freshly isolated PBMCs from HD individuals were stained and analyzed by flow cytometry, as indicated in methods. Figure shows CD4 + T cell subset ratios. The results are plotted for HD (open circles) comparing (A) Control vs. stimulated cells. Unpaired t test (normal distribution) or Mann-Whitney U test (non-normal variables) *p < 0.05. (B) Distinct CD4+ Th subset ratios of Mtb-stimulated PBMCs treated with 7-OD and DHEA. Each symbol represents an individual subject. Friedman test followed by post-hoc comparisons: Fisher’s LSD or Dunn’s test, as appropriated *p < 0.05, **p < 0.01 and ***p < 0.005
Additional file 5: Table S1. Expression of FoxP3, T-bet, ROR-γt and CD25 on a per cell basis in CD4 + T lymphocytes from HIV-TB patients. Recently thawed or freshly isolated PBMCs from HIV-TB were stimulated with Mtb in the presence/absence of 7-OD at 1 × 10−6M or DHEA at 1 × 10−7M. Then, cells were stained and analyzed by flow cytometry, as described before. Table shows median fluorescence intensity (MFI), which was calculated as the ratio of the geometric mean MFI of the marker of interest over MFI of the corresponding negative population. MFI is expressed as median ± interquartile range (IQR). Friedman test followed by Fisher’s LSD or by Dunn’s test, as appropriate *p < 0.05, **p < 0.01 and ***p < 0.005. * indicates significant differences with Mtb-stimulated cells (Mtb)
Data Availability Statement
All data generated or analyzed during this study are included in this published article and its supplementary information files.