Abstract
Introduction
Orally and daily levothyroxine (LT4) is the treatment of choice for hypothyroidism. In the majority of cases, the lack of effectiveness by this way may be due to poor adherence; however, gastrointestinal malabsorption may explain more cases of thyroxine refractoriness than previously reputed, due to the number of occult forms of these disorders.
Case Presentation
A 55-year-old white man with a diagnosis of low risk of recurrence of follicular variant of papillary thyroid carcinoma was treated with total thyroidectomy, 30 mCi iodine 131, and oral LT4. A year before he presented a gastric adenocarcinoma that required a partial gastrectomy. He evolved with multiple episodes of intestinal subocclusion that had to be treated with enterectomy in the first instance, then digestive rest and total parenteral nutrition. In spite of having made increases in oral LT4 dose (3 µg/kg), the patient persisted with a thyroid-stimulating hormone level >100 mIU/L. For this reason, we decided to administer intramuscular LT4.
Conclusion
Since there are no guidelines or consensus of intramuscular LT4 use, our experience and how we decided the dose and way of administration are presented in this article to contribute to future cases.
Keywords: Refractory hypothyroidism, Intramuscular levothyroxine, Levothyroxine therapy
What Is Known about This Topic?
True malabsorption is a cause of refractory hypothyroidism. Treatment with intramuscular levothyroxine (LT4) is an alternative for the treatment of these patients.
What Does This Case Report Add?
This case report is useful to the reader in that it provides evidence that intramuscular LT4 may solve some cases of extreme refractoriness.
Introduction
Primary hypothyroidism is a very common disease; its predominant causes are autoimmune disease and thyroid surgery. Currently, the first-line treatment is oral levothyroxine (LT4). However, there are cases in whom the dose of oral LT4 is not sufficient to reverse the symptoms or thyroid-stimulating hormone (TSH) levels (TSH >4.5 mIU/L after a 6-week interval of having increased the dose the last time) despite having increased the dose above 1.9 μg/kg/day [1, 2, 3]. In patients who require high doses of LT4, adherence to medical treatment should be confirmed in the first instance and the causes associated with malabsorption of LT4 should be evaluated [1, 4]. Since LT4 malabsorption is infrequent, we present a clinical case with a diagnosis of LT4 malabsorption that raised the need for alternatives to oral administration.
Case Report
A 55-year-old white man with a history of seminoma, surgery, and external beam radiotherapy in 1990 presented a gastric adenocarcinoma in July 2013, treated by partial gastrectomy. Three months after surgery, he suffered several episodes of intestinal subocclusion treated in the first instance with enterectomy and then with gastric repose on several subsequent occasions. In December 2014, a total thyroidectomy was performed due to a follicular-variant papillary carcinoma. He received treatment with 30 mCi of iodine 131 and oral LT4. During the following 3 years he had an excellent response receiving oral LT4 150 μg/day (2.14 μg/kg/day).
In November 2017, he was hospitalized for intestinal subocclusion and secondary malabsorptive syndrome (he had lost 12 kg within 3 months) presenting the following laboratory values: TSH >100 mIU/L (reference range [RR]: 0.35–4.94), free thyroxine (FT4) <5 pmol/L (RR: 9–19), total thyroxine (TT4) 1.8 μg/dL (RR: 4.9–11.7), and total triiodothyronine (TT3) <25 ng/dL (RR: 5–159). The patient was receiving oral LT4 150 μg/day (2.58 μg/kg/day). It was interpreted that the cause of his LT4 malabsorption was subacute subocclusion which improved within <48 h, so we took the decision of increase oral LT4 to 175 μg/day (3 μg/kg) plus oral vitamin C to improve LT4 absorption. After 2 weeks, the patient was readmitted for malabsorptive syndrome with severe intestinal subocclusion, which was interpreted as secondary to peritonitis due to a history of cobalt therapy and previous surgeries [5, 6]. The laboratory values at admission were TSH >100 mIU/L, FT4 6 pmol/L, TT4 2.2 μg/dL, and TT3 <25 ng/dL.
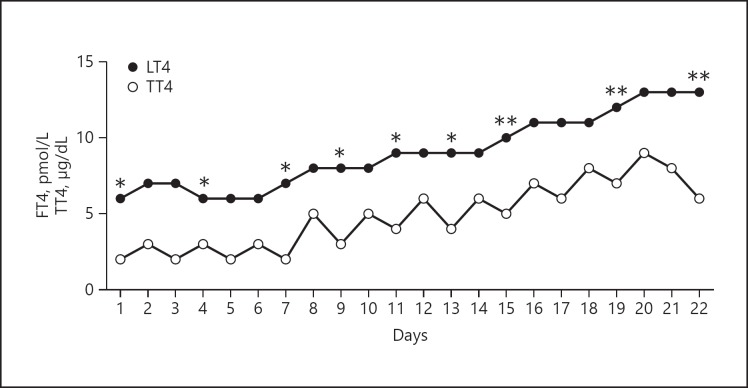
Upon physical examination the patient was lucid, without myxedema, thin, with a blood pressure of 110/60 mm Hg, a heart rate of 60 bpm, a weight of 58 kg, thyroid absent due to surgical history, without adenopathies in the neck. Due to the malabsorption syndrome secondary to intestinal subocclusion, the gastroenterologist ordered suspension of all oral drugs and food administration and started parenteral nutrition. In consequence, we needed to choose another therapeutic option for the administration of LT4. Based on the availability in our country we chose intramuscular LT4, administered at a dose of 200 μg twice a week with a progressively increasing dose according to clinical tolerance (max: 1,200 μg/week) as well as TT4 and FT4 levels (Fig. 1). The patient had very good tolerance and did not present any systemic or local adverse effects to intramuscular LT4. To avoid any local effect we decided the following: Only one nurse performed injections, rotating the application site every week. The hormone doses were evaluated daily during the first 20 days (Fig. 1). The hormone levels were normalized after 1 month of treatment (TSH: 1.99 mIU/L; FT4: 18 pmol/L; TT4: 8.6 μg/dL; TT3: 76 ng/dL) (Table 1). The LT4 formulation used in our patient was 200 μg/mL (each vial was 1 mL).
Fig. 1.
FT4 and TT4 levels after administration of 200 and 400 μg of intramuscular LT4. *200 μg of intramuscular LT4. **400 μg of intramuscular LT4. FT4, free thyroxine; LT4, levothyroxine; TT4, total thyroxine.
Table 1.
FT4 and TSH levels after administration of intramuscular LT4
| 15/12/2017 | 22/12/2017 | 10/01/2018 | 15/01/2018 | 06/08/2018 | |
|---|---|---|---|---|---|
| TSH, mIU/L (RR: 0.35–4.94) | 45 | >100 | 27 | 1.99 | 0.21 |
| FT4, pmol/L (RR: 9–19) | 7 | 8 | 14 | 18 | 13 |
| TT4, µg/dL (RR: 4.9–11.7) | 3.6 | 3.6 | 7.3 | 8.6 | 8.3 |
| TT3, ng/dL (RR: 58–159) | <25 | 45 | 54 | 76 | 75 |
FT4, free thyroxine; LT4, levothyroxine; RR, reference range; TSH, thyroid-stimulating hormone; TT3, total triiodothyronine; TT4, total thyroxine.
As permanent malabsorption diagnosis was performed, gastroenterologist continued with parenteral feeding at home. After 2 months the intramuscular LT4 dose was maintained at 600 μg/week (administered in 2 doses, 400 μg on Mondays and 200 μg on Thursdays, because the formulation is 200 μg/vial), remaining clinically and biochemically euthyroid until today.
Discussion
As mentioned before, the usual treatment of hypothyroidism is through oral LT4. Thus, the success of this treatment depends strongly on the gastrointestinal absorption of LT4. The stomach, the duodenum, as well as the upper and lower jejunum are the sites where drug absorption takes place, which happens similarly in all segments of the small intestine, but to a lesser extent in the colon.
Patients with impaired gastric acid secretion require higher doses of LT4, suggesting that normal acid secretion is necessary for effective absorption of oral LT4 [7, 8]. For those patients with oral LT4 malabsorption, parenteral LT4 could be used as an alternative therapy. However, there are no guidelines or consensus about the correct use of parenteral LT4, and there are very few studies assessing the effectiveness and safety of parenteral LT4 as an alternative to oral LT4. On the other hand, there are novel preparations of LT4, such as liquid and softgel LT4, available in many countries whose use have been proven useful in patients with gastrointestinal disorders similar to those of our patient, but currently they are not available in our country [9].
Kalathil et al. [10] presented the case of a patient with hypothyroidism and a severe gastrointestinal absorption disorder of unknown cause, where parenteral treatment with LT4 (500 μg weekly) was reported to have good biochemical response and no side effects.
Taylor et al. [11] presented an 82-year-old patient with a history of hypothyroidism who was hospitalized for myxedema coma. The cause was lack of adherence to the oral LT4 treatment. In this case the use of intramuscular LT4 proved to be an effective alternative and did not produce any adverse effects in the elder patient.
The first series of pediatric cases with successful intramuscular LT4 treatment of refractory primary hypothyroidism was published by Alba et al. [12]. The authors showed that a weekly dose of intramuscular LT4 can be a safe, successful, and well-tolerated treatment regime for pediatric patients.
Table 2 summarizes our patient's treatment with parenteral LT4. The present case report shows the results of using intramuscular LT4 to treat hypothyroidism in a patient subjected to partial gastrectomy and enterectomy, who presented LT4 malabsorption. The most similar cases found in the literature were those of bariatric surgery, where the stomach resection can cause LT4 malabsorption. However, there is no consensus in the literature on whether or not bariatric surgery causes oral LT4 malabsorption [13, 14, 15, 16, 17]. In this case, multiple factors may have led to the patient's LT4 malabsorption: gastrectomy, fibrosis secondary to cobalt therapy (received almost 30 years before), intestinal resection, and secondary acute intestinal occlusion.
Table 2.
Reported cases in the literature
| Reference | Publication year | Age, years | Probable cause | Parenteral dose, µg/week | Time to biochemical euthyroidism |
|---|---|---|---|---|---|
| Alba et al. [12] | 2016 | 16 | isolated thyroxine malabsorption? | 500 | 6 months |
| Alba et al. [12] | 2016 | 12 | isolated thyroxine malabsorption? | 500 | 8 months |
| Alba et al. [12] | 2016 | 6 | isolated thyroxine malabsorption | 300 | 8 months |
| Taylor et al. [11] | 2015 | 82 | poor adherence | 500 | not reported |
| Kalathil et al. [10] | 2012 | 47 | thyroxine malabsorption | 500 | 12 months |
| Anderson et al.[20] | 2009 | 18 | isolated thyroxine malabsorption | 1,000 | not reported |
The dose of intramuscular LT4 to be administered to the patient was calculated with Hays' [18] mathematical model, which predicts serum levels of thyroxine after parenteral administration of LT4 once and twice a week by the subcutaneous, intramuscular, and intravenous routes. The intramuscular route was assumed to have an absorption rate twice that of the subcutaneous route. Intramuscular and intravenous administration of properly adjusted doses of LT4, either once or twice a week, seemed to be acceptable options.
In this case, the definite general malabsorption and indication of total enteral nutrition led to ruling out oral LT4 as a treatment option. Furthermore, due to available options and reported cases [10, 11, 12, 13, 19, 20], intramuscular LT4 treatment was chosen. The dose used was initially conservative with daily titration and according adjustment within the first 2 weeks and three times a week for another 2 weeks until obtaining biochemical euthyroidism.
The patient achieved biochemical euthyroidism within 1 month and did not present any side effects during the treatment, even during the period of 1,200 μg/week. Currently, he is clinically and biochemically stable, presenting good tolerance of two applications of intramuscular LT4 per week, which allows him to do his usual work without difficulty. Intramuscular LT4 is an excellent option for future patients.
Statement of Ethics
All diagnostic and therapeutic procedures were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was obtained from the individual participant included in the study.
Disclosure Statement
The authors have no conflicts of interest to declare. Química Montpellier Laboratory provides LT4 only for parenteral use because this compound is not for sale for patients in Argentina. The laboratory delivers it to the Buenos Aires British Hospital pharmacy free of charge and given the clinical condition of the patient, without requesting any kind of compensation from the attending physicians.
Funding Sources
Financial support was provided by Buenos Aires British Hospital.
Author Contributions
All authors made substantial contributions to the conception and design and/or acquisition of data and/or analysis and interpretation of data, participated in drafting the article or revising it critically for important intellectual content, and gave their final approval of the version to be submitted and any revised version.
Acknowledgment
We appreciate the selfless collaboration of Química Montpellier Laboratory.
References
- 1.Lips DJ, van Reisen MT, Voigt V, Venekamp W. Diagnosis and treatment of levothyroxine pseudomalabsorption. Neth J Med. 2004 Apr;62((4)):114–8. [PubMed] [Google Scholar]
- 2.Virili C, Bassotti G, Santaguida MG, Iuorio R, Del Duca SC, Mercuri V, et al. Atypical celiac disease as cause of increased need for thyroxine: a systematic study. J Clin Endocrinol Metab. 2012 Mar;97((3)):E419–22. doi: 10.1210/jc.2011-1851. [DOI] [PubMed] [Google Scholar]
- 3.Cellini M, Santaguida MG, Gatto I, Virili C, Del Duca SC, Brusca N, et al. Systematic appraisal of lactose intolerance as cause of increased need for oral thyroxine. J Clin Endocrinol Metab. 2014 Aug;99((8)):E1454–8. doi: 10.1210/jc.2014-1217. [DOI] [PubMed] [Google Scholar]
- 4.Centanni M, Benvenga S, Sachmechi I. Diagnosis and management of treatment-refractory hypothyroidism: an expert consensus report. J Endocrinol Invest. 2017 Dec;40((12)):1289–301. doi: 10.1007/s40618-017-0706-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Travis LB, Curtis RE, Storm H, Hall P, Holowaty E, Van Leeuwen FE, et al. Risk of second malignant neoplasms among long-term survivors of testicular cancer. J Natl Cancer Inst. 1997 Oct;89((19)):1429–39. doi: 10.1093/jnci/89.19.1429. [DOI] [PubMed] [Google Scholar]
- 6.Zhang L, Hemminki O, Chen T, Yu H, Zheng G, Chattopadhyay S, et al. Second cancers and causes of death in patients with testicular cancer in Sweden. PLoS One. 2019 Mar;14((3)):e0214410. doi: 10.1371/journal.pone.0214410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hays MT, Nielsen KR. Human thyroxine absorption: age effects and methodological analyses. Thyroid. 1994;4((1)):55–64. doi: 10.1089/thy.1994.4.55. [DOI] [PubMed] [Google Scholar]
- 8.Cellini M, Santaguida MG, Virili C, Capriello S, Brusca N, Gargano L, et al. Hashimoto's Thyroiditis and Autoimmune Gastritis. Front Endocrinol (Lausanne) 2017 Apr;8:92. doi: 10.3389/fendo.2017.00092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Virili C, Trimboli P, Romanelli F, Centanni M. Liquid and softgel levothyroxine use in clinical practice: state of the art. Endocrine. 2016 Oct;54((1)):3–14. doi: 10.1007/s12020-016-1035-1. [DOI] [PubMed] [Google Scholar]
- 10.Kalathil D, Rajeev S, Chattington P. Hypothyroidism treated with intramuscular thyroxine injections. Endocrine Abstracts. 2012;28:359. [Google Scholar]
- 11.Taylor PN, Tabasum A, Sanki G, Burberry D, Tennant BP, White J, et al. Weekly Intramuscular Injection of Levothyroxine following Myxoedema: A Practical Solution to an Old Crisis. Case Rep Endocrinol. 2015;2015:169194. doi: 10.1155/2015/169194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Alba P, Mitre N, Feldt M. More than one way to skin a thyroid. Managing pediatric hypothyroidism with weekly intramuscular levothyroxine. J Pediatr Endocrinol Metab. 2016 Jun;29((6)):745–8. doi: 10.1515/jpem-2015-0382. [DOI] [PubMed] [Google Scholar]
- 13.Virili C, Antonelli A, Santaguida MG, Benvenga S, Centanni M. Gastrointestinal Malabsorption of Thyroxine. Endocr Rev. 2019 Feb;40((1)):118–36. doi: 10.1210/er.2018-00168. [DOI] [PubMed] [Google Scholar]
- 14.Skelin M, Lucijanić T, Amidžić Klarić D, Rešić A, Bakula M, Liberati-Čizmek AM, et al. Factors Affecting Gastrointestinal Absorption of Levothyroxine: A Review. Clin Ther. 2017 Feb;39((2)):378–403. doi: 10.1016/j.clinthera.2017.01.005. [DOI] [PubMed] [Google Scholar]
- 15.Pedro J, Cunha F, Souteiro P, Neves JS, Guerreiro V, Magalhães D, et al. The Effect of the Bariatric Surgery Type on the Levothyroxine Dose of Morbidly Obese Hypothyroid Patients. Obes Surg. 2018 Nov;28((11)):3538–43. doi: 10.1007/s11695-018-3388-4. [DOI] [PubMed] [Google Scholar]
- 16.Azran C, Porat D, Fine-Shamir N, Hanhan N, Dahan A. Oral levothyroxine therapy postbariatric surgery: biopharmaceutical aspects and clinical effects. Surg Obes Relat Dis. 2019 Feb;15((2)):333–41. doi: 10.1016/j.soard.2019.01.001. [DOI] [PubMed] [Google Scholar]
- 17.Julià H, Benaiges D, Mollà P, Pedro-Botet J, Villatoro M, Fontané L, et al. Changes in Thyroid Replacement Therapy after Bariatric Surgery: Differences between Laparoscopic Roux-en-Y Gastric Bypass and Laparoscopic Sleeve Gastrectomy. Obes Surg. 2019 Aug;29((8)):2593–9. doi: 10.1007/s11695-019-03890-9. [DOI] [PubMed] [Google Scholar]
- 18.Hays MT. Parenteral thyroxine administration. Thyroid. 2007 Feb;17((2)):127–9. doi: 10.1089/thy.2006.0283. [DOI] [PubMed] [Google Scholar]
- 19.Peynirci H, Taskiran B, Erturk E, Sisman P, Ersoy C. Is Parenteral Levothyroxine Therapy Safe in Intractable Hypothyroidism? J Natl Med Assoc. 2018 Jun;110((3)):245–9. doi: 10.1016/j.jnma.2017.05.007. [DOI] [PubMed] [Google Scholar]
- 20.Anderson L, Joseph F, Goenka N, Patel V. Isolated thyroxine malabsorption treated with intramuscular thyroxine injections. Am J Med Sci. 2009 Feb;337((2)):150–2. doi: 10.1097/MAJ.0b013e31817ee556. [DOI] [PubMed] [Google Scholar]