Abstract
A telomere consists of repeated DNA sequences (TTAGGG)n as part of a nucleoprotein structure at the end of the linear chromosome, and their progressive shortening induces DNA damage response (DDR) that triggers cellular senescence. The telomere can be maintained by telomerase activity (TA) in the majority of cancer cells (particularly cancer stem cells) and pluripotent stem cells (PSCs), which exhibit unlimited self-proliferation. However, some cells, such as telomerase-deficient cancer cells, can add telomeric repeats by an alternative lengthening of the telomeres (ALT) pathway, showing telomere length heterogeneity. In this review, we focus on the mechanisms of the ALT pathway and potential clinical implications. We also discuss the characteristics of telomeres in PSCs, thereby shedding light on the therapeutic significance of telomere length regulation in age-related diseases and regenerative medicine.
Keywords: alternative lengthening of telomeres, telomerase, DNA damage, pluripotent stem cells, telomere maintenance mechanism, genome stability
1. Introduction
Telomeres consist of tandem TTAGGG repeats, ending with an approximate 50–500 nt G-rich 3’-strand overhang [1,2]. Telomeres are enclosed by the shelterin protein complex, which contains the double-strand telomeric DNA binding factors TRF1 and TRF2, the TRF2-interacting protein RAP1, the bridging factor TIN2, and the telomeric ssDNA-binding protein POT1 and its direct interactor TPP1 [3]. Shelterin proteins bind to the telomere structure and mediate the formation of a telomeric loop (T-loop) in which the single-strand 3’ overhang is concealed into a D-loop structure [4,5]. In somatic cells, telomeres continuously shorten with cell division, resulting in an accumulation of DNA damage that triggers cellular senescence [6,7,8]. In stem cells and about 85% of cancer cells, the telomeres could be elongated by telomerase through adding TTAGGG repeats to the chromosome ends. However, in the other 15% of cancer cells, telomeres are maintained by an alternative lengthening of telomeres (ALT) mechanism, which mainly relies on homologous recombination (HR) between sister chromatids. This review focuses on the mechanisms of ALT in tumors and pluripotent stem cells as well as the implications for related potential therapies.
2. Telomere Function in Maintaining Genomic Stability
During lagging strand synthesis, the 5’ end cannot be replicated entirely due to the end replication problem, resulting in gradual loss of chromosomal end at each round of cell division [9]. Telomeres located at chromosome ends serve as an extendable DNA structure to solve this problem. Moreover, telomeres are recognized as difficult-to-replicate sites which are sensitive to replication stress and additional replication factors, including helicase, DNase, topoisomerase, and other DNA binding proteins that are required to properly replicate the telomeric dsDNA [10]. The mutation of these proteins induces telomere dysfunction or damage, and eventually leads to premature aging [11,12,13]. The telomere binding protein deficiency leads to “telomere uncapping” and eventually chromosomal instability and cell death. For example, the loss of telomeric dsDNA-binding protein TRF2 would cause ATM-dependent chromosomal fusion by NHEJ (Non-homologous end joining) [14]. In addition, the depletion of POT1 leads to aberrant RPA (replication protein A) accumulation and homologous recombination at telomeres, and consequently genomic instability and replicative senescence [15]. Both telomere shortening and telomere deprotection significantly increase the risk of tumorigenesis, especially when the tumor suppressor gene p53 is ablated. Consistently, mice with hyper-long telomere live longer and have less spontaneous tumor incidence [16].
Other telomere accessory factors and telomeric structures have also been identified to play essential roles in telomere integrity. For instance, telomere-repeat-encoding RNA (also referred to as TERRA) transcribed from the telomeric DNA, has been shown as a potential component of eukaryotic telomeres and to play essential roles in telomere homeostasis. TERRA transcription is repressed upon telomere elongation, mediated by the increased HP1 α and trimethylated H3K9 density [17]. Abnormal heterochromatin status induces TERRA expression deficiency and augmentation of telomere dysfunction-induced foci (TIF) [18]. Telomere 3’-strand overhang is thought to invade double-stranded telomeric DNA, resulting in a bulging duplex lariat construction known as T-loop that masks the 3’ end of the telomere from being recognized as a single strand or double strand DNA break. Thus, the generation of G-tail and T-loop is essential for telomere length and integrity maintenance [19,20]. G-quadruplex is another structure arisen from the self-stacking of two or more guanine quartets, frequently observed at the telomere region [21,22]. Also, loss-of-function mutations in DNA helicases, which have G-quadruplex unwinding activity link to telomere maintenance deficiency and genome instability [23,24,25].
3. Telomere Maintenance by Telomerase or ALT
To counteract the telomere loss and bypass replicative senescence, cells must establish a telomere maintenance mechanism (TMM), which permits prolonged proliferative potential [26,27,28]. The vast majority of the cancer cells and stem cells use the telomerase to lengthen telomeres [7]. Extension of telomeres by the telomerase catalytic process consists of several sequential stages. Telomerase is recruited to telomeric DNA via interaction with TPP1 OB-fold then taken to the 3’ terminal end via paring of the alignment fragment of TERC (telomerase RNA template) to the telomere [29,30,31]. Subsequently, the G-strand is reserve-transcribed by TERT (telomerase reverse transcriptase) using TERC as a template. The newly synthesized long G-overhang is covered by the CST complex, which displaces telomerase and recruits primase-Pol α to fill in the C-strand and produce the entire double strand telomeric DNA [32,33].
Approximately 10–15% of tumor cells elongate their telomeres using a recombination-based alternative lengthening mechanism. Interestingly, the correlation between alternative lengthening of telomeres (ALT) and prognosis varies among different cancer types and patients. In soft tissue sarcomas, ALT is associated with lower survival than telomerase activation [34]. In osteosarcomas, there is no difference in the clinical outcome [35]. However, ALT is associated with a better patient outcome in glioblastoma [36]. Differential prognosis of ALT may rely on the different genetic and epigenetic events responsible for TMMs. The preference to use one TMM rather than the other varies among tumor types [37]. Cancers with mesenchymal origin are more likely to use ALT, while cancers with an epithelial origin are more likely to activate telomerase [38,39]. However, the underlying reasons remain poorly understood. The bladder cancer cells epithelial-to-mesenchymal transition (EMT), which prevents cells from undergoing senescence during tumor development, promotes telomerase-to-ALT-like conversion, suggesting the factors involved in EMT may play an essential role in telomerase activity repression [40]. Moreover, telomerase activity is inhibited upon hybridization with ALT cells, indicating the existence of telomerase repressors in ALT-positive cells [41]. Additionally, telomerase positive tumors exhibit ALT hallmark upon anti-telomerase therapies [42,43]. Thus, ALT is considered as an intrinsic mechanism that coexists with telomerase as a back-up TMM during evolution in mammals [44]. Combined inhibition of telomerase and ALT may be needed to achieve extensive clinical efficacy.
Telomeric chromatin accessibility is thought to be essential for telomerase/ALT determination. Loss of telomeric heterochromatin markers, including H3K9me3 and H4K20me3, might induce ALT by promoting chromatin decondensation [45]. Additionally, loss of H3.3-specific chaperone ATRX or its cofactor DAXX progressively induces ALT activation via regulating the level of DNA methylation and heterochromatin [46]. During this process, heterochromatin protein HP1 may serve as a protein scaffold for ATRX recruitment [46,47]. Moreover, depletion of ATRX induced telomeric replication stress and DNA damage response [48]. Thus, ALT activation may be an adaptive response to ATRX depletion-induced telomere replication dysfunction [49].
4. Characteristics and Diagnosis of ALT
ALT in cancers is characterized by heterogeneous telomere sizes, which vary from extremely short (<1 kb) to abnormally long (>20 kb) in human cells [50,51]. Moreover, ALT cells are also featured with the presence of ALT-associated PML bodies (APBs), which are specialized promyelocytic leukemia (PML) protein bodies containing telomeric DNA, shelterin complex, and proteins related to DNA recombination (for example MRN, BLM, and WRN) [52,53,54,55]. Additionally, the appearance of extrachromosomal telomeric circular DNA, which may be the products of telomere trimming or self-replicating templates for telomeric DNA lengthening, is also assumed to be an exclusive feature of ALT-positive cell lines and tumors [56,57]. Telomeric sister chromatin exchange and abnormal insertion at chromosome ends are frequently observed in ALT-positive cells and considered a hallmark of ALT as well [58,59]. Lastly, telomere clustering, which offers a new platform for telomere recombination, was also known as a unique feature of ALT [60].
Given the above features, several approaches were developed to investigate ALT in tumors and cell lines. Telomere Restriction Fragment (TRF) southern blotting was regularly used to detect the heterogeneous telomere [61]. In line with the evidence that PML bodies localize with telomere DNA, the ALT-associated PML bodies (APBs) were used for ALT determination [35,62]. Bromodeoxyuridine (BrdU) labeling of telomeres in G2-phase cells and the examination of mitotic DNA synthesis (MiDAS) at telomeres, allowing the visualization of telomere DNA synthesis outside of S phase, is another straightforward assay to distinguish the ALT formation [60]. Additionally, the C-circle (CC) assay is a widely applied approach to detect ALT via rolling circle amplification for the extrachromosomal telomeres. The level of ALT activation can be quantified through the dot-blot analysis using 32P-labeled telomeric probes [56]. Moreover, the CC assay can detect the number of C-circles within just 30 ng of DNA, which allows the broad usage for the prevalence and prognostic analysis in ALT tumor samples [63].
5. How Is ALT Activated?
It has been widely accepted that DSBs at chromosomal ends trigger BIR (break-induced replication) -mediated telomeric DNA synthesis. Due to the extensive homology at telomere ends, a break-induced replication process is initiated where the broken end invades to the donor telomere then serves as a primer for initiation of DNA replication, relying on POLD3/4 [64]. Moreover, the replication stress at telomeres is also assumed to prime ALT as well, and the replication stress response protein SMARCAL1 was reported to associate with telomeres to inhibit the repair of DSBs and ensure ALT telomere maintenance, demonstrating that resolution of replication stress is a crucial step in the ALT mechanism [65,66]. In the absence of telomerase, telomere shortening leads to ssDNA accumulation at the telomere region, and HR (homologous recombination) machinery counteracts the ssDNA and elongates the telomere through the DNA damage response pathway [67]. In addition, the telomere length maintaining mechanism of telomerase-negative cells requires genes encoding HR-related proteins (including MRN complex, Rad51, Rad52, FANC proteins), suggesting HR are essential for conservative replication of telomeres, besides their DNA repair functions [68]. There are three routes of telomeres being HR-sorted by this type of telomeric DNA exchange: Equivalent telomeric sister chromatin exchange (T-SCE), inequivalent T-SCE, and non-sister chromatid exchange (No-SCE) [69]. Inequivalent T-SCE mainly leads to telomere length heterogeneity without a net gain of telomere length, whereas No-SCE, which may be induced by interchromatin HR and break-induced replication, results in increased telomere length [69].
Two distinct pathways are identified during ALT elevation [70]. One pathway requires RAD52, which binds DNA and facilitates the annealing process between complementary ssDNA strands [71,72]. Recruitment of RAD52 to the telomere is an SLX4-dependent process [73]. Furthermore, the replication intermediates generated in the absence of RAD52 can be processed by SLX4. Simultaneous deletion of RAD52 and SLX4 results in deficiency in mitotic fidelity and telomere dysfunctions, suggesting the accumulation of unresolved stalled forks and recombination intermediates, which may serve as barriers to DNA synthesis and lead to gradual telomere shortening [71]. The other ALT mechanism is independent of RAD52 but requires BLM and POLD3/4, suggesting that activation of this ALT pathway is mediated by a BIR-related process [64,70]. Nevertheless, both processes take place within APBs, which offer a “recombinogenic microenvironment” to facilitate ALT, and these two different repair syntheses rely on the nature of telomere lesions and cell cycle phases [70,71].
BIR functions via an RFC–PCNA–Pol δ axis, independent of other canonical replisome components such as ATM, ATR and Rad51 [74]. Additionally, BLM-TOP3A-RMI (BTR) complex is necessary for ALT-mediated telomere synthesis. In this process, recombination intermediates can initiate large-scale POLD3-dependent telomere synthesis, followed by dissolution, without inducing T-SCE. However, this process is inhibited by the SLX4-SLX1-ERCC4 complex, which promotes the resolution of recombination intermediates, leading to telomere exchange without telomere extension [75] (Figure 1). The complexity of the ALT mechanism leads to different behaviors of ALT tumors in terms of disease progression and prognosis. Hence, a deep understanding of the molecular mechanisms of ALT pathways seems to be essential for diagnosis of ALT and discovery of novel drugs targeting this pathway.
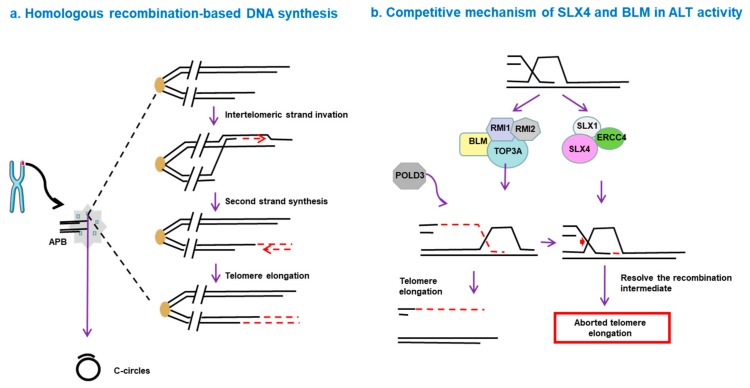
Figure 1.
Homologous recombination-based telomere DNA synthesis. (a) DNA double-strand breaks can trigger telomere synthesis. A break-induced replication process is initiated when the broken end invades a donor telomere, followed by replication of the donor DNA sequence and invading DNA, resulting in increased telomere length. (b) Competitive mechanism of SLX4 and BLM in alternative lengthening of telomeres (ALT) activity. The BLM-TOP3A-RMI (BTR) complex is essential for ALT-mediated telomere synthesis. In this process, recombination intermediates can initiate POLD3-dependent telomere synthesis, followed by dissolution, without inducing telomere sister-chromatid exchange (T-SCE). However, this process is inhibited by the SLX4-SLX1-ERCC4 complex, which promotes the resolution of the recombination intermediates and leads to telomere exchange without telomere elongation.
6. ALT in Pluripotent Stem Cells (PSCs)
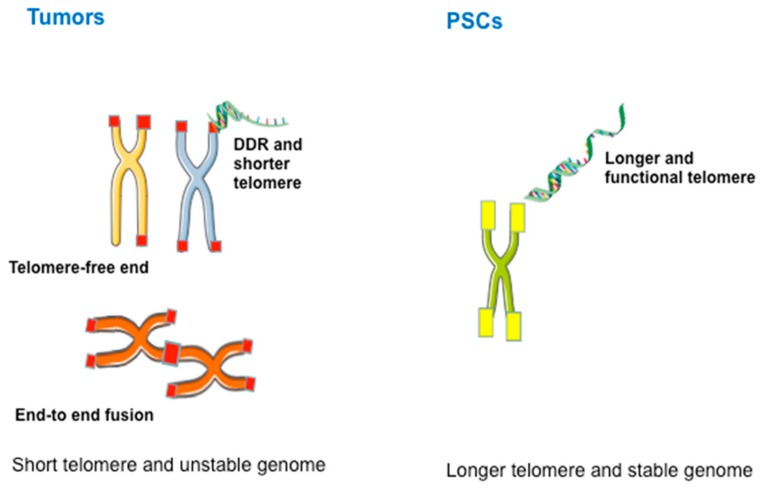
Telomere maintenance is critical for the unlimited self-renewal, stemness, and genomic homeostasis of PSCs [76]. Telomere length represents another important criterion for defining stem cell pluripotency, and modulation of telomere length may present great potential in the application of PSCs in regenerative medicine [77]. Sufficient telomere length is also a requirement for the functionality of adult stem cells [78]. PSCs commonly express telomerase to maintain telomeres, and increasing evidence shows that the ALT-like pathway also plays a crucial role in telomere maintenance [77,79]. Both cancer cells (especially cancer stem cells) and PSCs rely on telomere maintenance for cell proliferation. However, telomeres and their length regulation show apparent differences between these two cell types. For example, the genomes of tumors with ALT are unstable, exhibiting heterogeneous telomeres, extrachromosomal DNA circles, APBs, frequent T-SCE, and dysfunctional telomeres. In contrast, PSCs maintain longer telomeres and stable genomes (Figure 2). The underlying mechanism remains unclear, but ALT in PSCs is mainly triggered by changes in epigenetic reprogramming [79], which provides an “open” chromatin state for activating ALT, rather than the harmful mutations that frequently occur in cancer cells. Additionally, ALT in cancer cells, but not in PSCs, involves mechanisms that negatively regulate telomere length by trimming telomeric DNA, resulting in the formation of t-circles [80].
Figure 2.
Distinctions between telomeres in tumor cells and pluripotent stem cells (PSCs). The genome of tumors is unstable and is characterized by heterogeneous telomeres, extrachromosomal DNA circles, ALT-associated promyelocytic leukemia (PML) bodies (APBs), and frequent T-SCEs, whereas PSCs exhibit longer functional telomeres and stable genomes.
Telomere length homeostasis is crucial for the genomic integrity of embryonic stem cells (ESCs) and must be maintained to prevent excessive telomere elongation. ESCs cultured under standard conditions in the presence of leukemia inhibitory factor (LIF) as well as feeders, can shuttle back and forth from a state that resembles a two-cell embryo-like state [81]. Zscan4, a two-cell embryonic gene expressed during zygotic genome activation, is essential for telomere extension in mESCs by T-SCE-dependent HR [82]. Expression of Zscan4 is activated upon telomere shortening and reaches to the maximum level at the G2 phase of the cell cycle, which may represent a stage in which telomere extension can occur [83]. It is likely that two-cell genes, including Zscan4, are controlled by several regulators to maintain their appropriate activation in a small fraction of ESC populations, leading to efficient elongation of telomeres, followed by a telomerase-dependent mechanism for telomere maintenance [84]. Rif1, a telomere-associated protein, was initially identified in budding yeast that negatively regulates telomere length and plays an essential role in yeast telomere length homeostasis [85]. For differentiated mammalian cells, Rif1 was identified to play roles in DNA damage response and replication timing control but presents no functions related to telomeres [86,87]. In mouse ESCs, Rif1 regulates expression of Zscan4 and other 2-cell genes by maintaining heterochromatic H3K9me3 histone methylation levels at subtelomeric regions. Thus, Rif1 acts as an essential factor for telomere length homeostasis by negatively regulating 2-cell genes [84]. Depletion of Rif1 results in heterogeneous telomere elongation and shortening, similar to what is observed in ALT cancer cells [84].
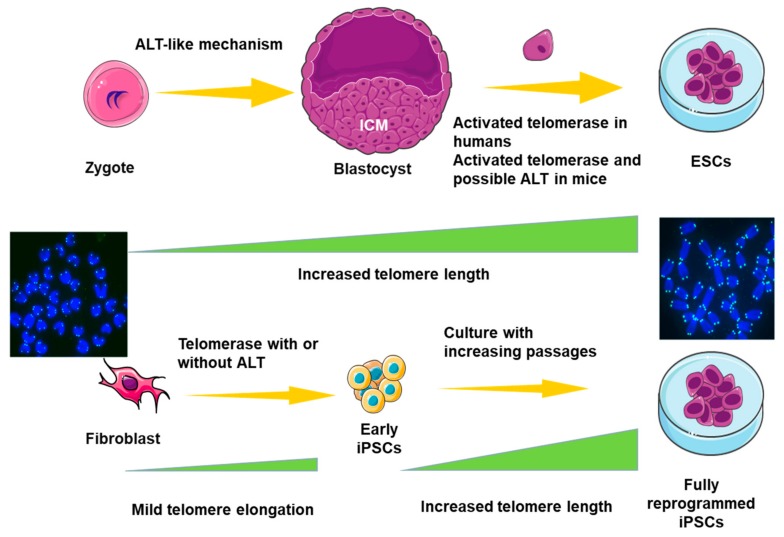
Telomeres of human ESCs are extended by telomerase during the early stage of derivation and reach a relatively stable level [88]. Interestingly, unlike human ESCs, many ALT features are also detected in telomerase-negative mouse ESCs with short telomeres [89]. Telomeres are extended by a mechanism resembling ALT-like activity during early embryo cleavage stage [90]. Despite telomere elongation during cleavage stages, the inner cell mass (ICM) of blastocysts still presents shorter telomeres compared to ESCs, and ESCs derived from the ICM in vitro exhibit elongation of telomeres to a relatively stable level depending on both telomerase [91] and an ALT-like mechanism [77] (Figure 3). This difference may be illustrated by the fact that ALT can emerge in naïve mouse ESCs but not in human ESCs, which more closely resemble the primed state of ESCs or epiblast stem cells in mice. Nevertheless, this assumption requires further evidence [79].
Figure 3.
Telomere extension mechanism during acquisition of pluripotency. Telomeres of embryonic stem cells (ESCs) are extended during the early stage of derivation and reach a relatively stable level. The ALT mechanism is not observed in human ESCs, unlike the situation in mouse ESCs. Similarly, telomeres of iPSCs (induced pluripotent stem cells) are remodeled to a state resembling ESCs mainly by telomerase-dependent and possible ALT-like pathways and continuously elongate during the acquisition of fully reprogrammed iPSCs.
During the process of iPSC reprogramming, telomerase activity increases gradually before the emergence of endogenous pluripotency genes [92]. Ectopic expression of factors that could protect telomeres by increasing ALT-like activity, together with Zscan4, can increase the efficiency and pluripotency of iPSCs dramatically [93]. Additionally, small molecules that promote Zscan4 expression and telomeric elongation by ALT-like pathway facilitate the generation of high-quality iPSCs [94,95]. Telomeres of iPSCs are remodeled to a state resembling ESCs mainly by telomerase-dependent and possible ALT-like pathways [92,96] and continuously elongate during the acquisition of fully reprogrammed iPSCs [95].
PSCs exhibit a more open telomeric chromatin structure, which becomes condensed during differentiation [97]. Hence, the telomeric structure of PSCs may be in a dynamic state that experiences remodeling in the process of differentiation [98]. The epigenetic state appears to play a critical role in the telomeric homeostasis of PSCs by affecting the process of ALT. Downregulation of H4K20me3 and H3K9me3 results in abnormally elongated telomeres [99,100]. Tet enzymes regulate telomere length by participating in epigenetic modification, with an essential role in pluripotency [101,102]. Moreover, ESC culture conditions can impact epigenetic modification and transcription and ultimately affect the telomere regulation mode of PSCs. For example, the 2i (MEK inhibitor and Gsk3β inhibitor)+LIF culture medium can decrease DNA methylation and H3K9me3 levels to affect telomeric homeostasis [81]. Additionally, DNA methylation and H3K9me3 prevent reprogramming in association with the negative regulation of telomere length. In contrast, DNA demethylation and increased histone acetylation can promote epigenetic reprogramming by lengthening telomeres [79]. Notably, PSCs derived from Terc-/- somatic cell nuclear transfer (SCNT) exhibit longer telomeres, higher pluripotency, and a greater self-renewal capacity than Terc-/- iPSCs, indicating that a telomerase-independent telomere elongation mechanism, possibly ALT, is more efficient in SCNT [103]. Hence, PSCs, including ESCs and iPSCs, must extend their telomeres during derivation and passaging to maintain their self-renewal ability and pluripotency [96]. A better understanding of how PSCs maintain their telomeres via telomerase and/or ALT-like activity is of significance in the fields of aging, stem cell biology, and regenerative medicine.
7. Potential Therapeutic Strategy Targeting ALT
Targeting of telomeres has received much attention regarding its potential clinical anti-cancer applications. At the molecular level, most of the cancer cells require one or the other TMM to maintain their telomeres to achieve immortalization [39,104]. Here, we list a number of potential therapeutic strategies in the treatment of tumors that rely on ALT.
Mutations in ATRX/DAXX as well as histone H3.3 are major causes of ALT activation in pediatric glioblastomas, pancreatic neuroendocrine cancers, and other cancers of the central nervous system [105,106,107]. Thus, it is likely that inactivation of ATRX/DAXX/H3.3 is an essential step in the generation of ALT in those tumors [108], which provides potential targets for anti-cancer therapies with transgenic technology. Recently, a screened mutant herpes simplex virus is shown to kill ATRX-deficient ALT tumors directly, through the recognition of ATRX [109]. Targeting ATRX/DAXX and telomerase simultaneously or separately may effectively induce tumor senescence and apoptosis in malignant gliomas.
Several drugs targeting the cell cycle and DNA damage response have entered clinical trials for ALT tumors. Cell cycle G2/M checkpoint deficiency offers a potential vulnerability specific to ALT-positive cells. Thus, G2/M checkpoint depressors have been developed for ALT-positive cancers [110]. Additionally, non-S-phase telomere synthesis is considered as a valuable therapeutic target in ALT tumors due to its difference from canonical S-phase replication [60].
Roles of apoptosis in anti-tumor effects have been investigated in many different cancers, such as cervical cancer and Epstein-Barr virus (EBV)-positive B lymphocytes [111]. Suppression of telomere-related genes through telomerase and ALT mechanisms prevents development of laryngeal squamous cell carcinoma by facilitating cell apoptosis through a mechanism involving the PI3K/Akt pathway [112], which may provide a potential therapeutic strategy via promoting apoptosis. G-quadruplex ligands have been indicated to disturb telomere replication, with an anti-proliferative result [113,114]. Several G-quadruplex ligands with high affinity for telomeric G-quadruplexes are proved to have potential anti-tumor properties [115,116]. G-quadruplex stabilization might have synergistic effects with other DNA-damaging therapies, such as ionizing radiation, in ATRX-deficient tumors [117,118].
Most tumors use just one of the TMMs to maintain homeostasis. Nevertheless, the coexistence of ALT and telomerase has been identified both in vitro and in vivo [28,119]. Interestingly, osteosarcoma and neuroblastoma are characterized by inner-tumoral heterogeneity in TMM, where ALT and telomerase activity exist in different cells of the same tumor [120,121,122]. In contrast, breast cancer samples have revealed both telomerase and ALT within the same cell [123]. Moreover, after treatment with drugs targeting telomerase, a number of cancer cells escape death, switching from telomerase activity to ALT [104]. The molecular foundation underlying this switch or coexistence of both TMMs within the same cell or cell population remains to be elucidated. A characteristic description of these distinct cancers could improve our progression towards not only understanding the exact mechanisms of the two TMMs but also discovery of effective therapeutic strategies.
8. Perspectives
Based on the current understanding of the features of ALT, some of therapeutic strategies targeting this pathway have entered phase I/II clinical trials, such as the use of Trabectedin in treating soft tissue sarcoma [124]. Nevertheless, numerous puzzling problems remain regarding this specific mechanism: Why do differences in ALT or ALT-like activities exist in tumors, especially in cancer stem cells and PSCs? What are the potential differences in the mechanisms of ALT in these different cell types? Simultaneous single-cell measurements of telomeres and analyses of transcriptome, epigenome, or proteome in the same cell may resolve these issues in the future. Further studies are necessary to assess the unique biological characteristics and possible prognosis of ALT-positive cancers and will be significant in designing novel anti-cancer therapeutics targeting the ALT pathway.
Acknowledgments
We indeed are grateful to the three reviewers for critical comments and advice on improving this review manuscript.
Funding
National Nature Science Foundation of China (31430052, 31571546); China Ministry of Science and Technology China National Key R&D Program (2018YFA0107000, 2018YFA1003004).
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Makarov V.L., Hirose Y., Langmore J.P. Long G tails at both ends of human chromosomes suggest a C strand degradation mechanism for telomere shortening. Cell. 1997;88:657–666. doi: 10.1016/S0092-8674(00)81908-X. [DOI] [PubMed] [Google Scholar]
- 2.Wright W.E., Tesmer V.M., Huffman K.E., Levene S.D., Shay J.W. Normal human chromosomes have long G-rich telomeric overhangs at one end. Genes Dev. 1997;11:2801–2809. doi: 10.1101/gad.11.21.2801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sfeir A., de Lange T. Removal of shelterin reveals the telomere end-protection problem. Science. 2012;336:593–597. doi: 10.1126/science.1218498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Griffith J.D., Comeau L., Rosenfield S., Stansel R.M., Bianchi A., Moss H., de Lange T. Mammalian telomeres end in a large duplex loop. Cell. 1999;97:503–514. doi: 10.1016/S0092-8674(00)80760-6. [DOI] [PubMed] [Google Scholar]
- 5.De Lange T. Shelterin-Mediated Telomere Protection. Annu. Rev. Genet. 2018;52:223–247. doi: 10.1146/annurev-genet-032918-021921. [DOI] [PubMed] [Google Scholar]
- 6.Hayashi M.T. Telomere biology in aging and cancer: Early history and perspectives. Genes Genet. Syst. 2018;92:107–118. doi: 10.1266/ggs.17-00010. [DOI] [PubMed] [Google Scholar]
- 7.Jafri M.A., Ansari S.A., Alqahtani M.H., Shay J.W. Roles of telomeres and telomerase in cancer, and advances in telomerase-targeted therapies. Genome Med. 2016;8:69. doi: 10.1186/s13073-016-0324-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Olovnikov A.M. A theory of marginotomy. The incomplete copying of template margin in enzymic synthesis of polynucleotides and biological significance of the phenomenon. J. Theor. Biol. 1973;41:181–190. doi: 10.1016/0022-5193(73)90198-7. [DOI] [PubMed] [Google Scholar]
- 9.Levy M.Z., Allsopp R.C., Futcher A.B., Greider C.W., Harley C.B. Telomere end-replication problem and cell aging. J. Mol. Biol. 1992;225:951–960. doi: 10.1016/0022-2836(92)90096-3. [DOI] [PubMed] [Google Scholar]
- 10.Hewitt G., Jurk D., Marques F.D., Correia-Melo C., Hardy T., Gackowska A., Anderson R., Taschuk M., Mann J., Passos J.F. Telomeres are favoured targets of a persistent DNA damage response in ageing and stress-induced senescence. Nat. Commun. 2012;3:708. doi: 10.1038/ncomms1708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Anderson B.H., Kasher P.R., Mayer J., Szynkiewicz M., Jenkinson E.M., Bhaskar S.S., Urquhart J.E., Daly S.B., Dickerson J.E., O’Sullivan J., et al. Mutations in CTC1, encoding conserved telomere maintenance component 1, cause Coats plus. Nat. Genet. 2012;44:338–342. doi: 10.1038/ng.1084. [DOI] [PubMed] [Google Scholar]
- 12.Simon A.J., Lev A., Zhang Y., Weiss B., Rylova A., Eyal E., Kol N., Barel O., Cesarkas K., Soudack M., et al. Mutations in STN1 cause Coats plus syndrome and are associated with genomic and telomere defects. J. Exp. Med. 2016;213:1429–1440. doi: 10.1084/jem.20151618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Larizza L., Magnani I., Roversi G. Rothmund-Thomson syndrome and RECQL4 defect: Splitting and lumping. Cancer Lett. 2006;232:107–120. doi: 10.1016/j.canlet.2005.07.042. [DOI] [PubMed] [Google Scholar]
- 14.Benarroch-Popivker D., Pisano S., Mendez-Bermudez A., Lototska L., Kaur P., Bauwens S., Djerbi N., Latrick C.M., Fraisier V., Pei B., et al. TRF2-Mediated Control of Telomere DNA Topology as a Mechanism for Chromosome-End Protection. Mol. Cell. 2016;61:274–286. doi: 10.1016/j.molcel.2015.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gong Y., de Lange T. A Shld1-controlled POT1a provides support for repression of ATR signaling at telomeres through RPA exclusion. Mol. Cell. 2010;40:377–387. doi: 10.1016/j.molcel.2010.10.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Munoz-Lorente M.A., Cano-Martin A.C., Blasco M.A. Mice with hyper-long telomeres show less metabolic aging and longer lifespans. Nat. Commun. 2019;10:4723. doi: 10.1038/s41467-019-12664-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Perez-Romero C.A., Lalonde M., Chartrand P., Cusanelli E. Induction and relocalization of telomeric repeat-containing RNAs during diauxic shift in budding yeast. Curr. Genet. 2018;64:1117–1127. doi: 10.1007/s00294-018-0829-5. [DOI] [PubMed] [Google Scholar]
- 18.Deng Z., Norseen J., Wiedmer A., Riethman H., Lieberman P.M. TERRA RNA binding to TRF2 facilitates heterochromatin formation and ORC recruitment at telomeres. Mol. Cell. 2009;35:403–415. doi: 10.1016/j.molcel.2009.06.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sarek G., Vannier J.B., Panier S., Petrini J.H.J., Boulton S.J. TRF2 recruits RTEL1 to telomeres in S phase to promote t-loop unwinding. Mol. Cell. 2015;57:622–635. doi: 10.1016/j.molcel.2014.12.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.O’Sullivan R.J., Karlseder J. Telomeres: Protecting chromosomes against genome instability. Nat. Rev. Mol. Cell Biol. 2010;11:171–181. doi: 10.1038/nrm2848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Schaffitzel C., Berger I., Postberg J., Hanes J., Lipps H.J., Pluckthun A. In vitro generated antibodies specific for telomeric guanine-quadruplex DNA react with Stylonychia lemnae macronuclei. Proc. Natl. Acad. Sci. USA. 2001;98:8572–8577. doi: 10.1073/pnas.141229498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Paeschke K., Simonsson T., Postberg J., Rhodes D., Lipps H.J. Telomere end-binding proteins control the formation of G-quadruplex DNA structures in vivo. Nat. Struct. Mol. Biol. 2005;12:847–854. doi: 10.1038/nsmb982. [DOI] [PubMed] [Google Scholar]
- 23.Ding H., Schertzer M., Wu X., Gertsenstein M., Selig S., Kammori M., Pourvali R., Poon S., Vulto I., Chavez E., et al. Regulation of murine telomere length by Rtel: An essential gene encoding a helicase-like protein. Cell. 2004;117:873–886. doi: 10.1016/j.cell.2004.05.026. [DOI] [PubMed] [Google Scholar]
- 24.Lin W., Sampathi S., Dai H., Liu C., Zhou M., Hu J., Huang Q., Campbell J., Shin-Ya K., Zheng L., et al. Mammalian DNA2 helicase/nuclease cleaves G-quadruplex DNA and is required for telomere integrity. EMBO J. 2013;32:1425–1439. doi: 10.1038/emboj.2013.88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Paeschke K., Bochman M.L., Garcia P.D., Cejka P., Friedman K.L., Kowalczykowski S.C., Zakian V.A. Pif1 family helicases suppress genome instability at G-quadruplex motifs. Nature. 2013;497:458–462. doi: 10.1038/nature12149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bodnar A.G., Ouellette M., Frolkis M., Holt S.E., Chiu C.P., Morin G.B., Harley C.B., Shay J.W., Lichtsteiner S., Wright W.E. Extension of life-span by introduction of telomerase into normal human cells. Science. 1998;279:349–352. doi: 10.1126/science.279.5349.349. [DOI] [PubMed] [Google Scholar]
- 27.Heaphy C.M., de Wilde R.F., Jiao Y., Klein A.P., Edil B.H., Shi C., Bettegowda C., Rodriguez F.J., Eberhart C.G., Hebbar S., et al. Altered telomeres in tumors with ATRX and DAXX mutations. Science. 2011;333:425. doi: 10.1126/science.1207313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cerone M.A., Londono-Vallejo J.A., Bacchetti S. Telomere maintenance by telomerase and by recombination can coexist in human cells. Hum. Mol. Genet. 2001;10:1945–1952. doi: 10.1093/hmg/10.18.1945. [DOI] [PubMed] [Google Scholar]
- 29.Xin H., Liu D., Wan M., Safari A., Kim H., Sun W., O’Connor M.S., Songyang Z. TPP1 is a homologue of ciliate TEBP-beta and interacts with POT1 to recruit telomerase. Nature. 2007;445:559–562. doi: 10.1038/nature05469. [DOI] [PubMed] [Google Scholar]
- 30.Gavory G., Farrow M., Balasubramanian S. Minimum length requirement of the alignment domain of human telomerase RNA to sustain catalytic activity in vitro. Nucleic Acids Res. 2002;30:4470–4480. doi: 10.1093/nar/gkf575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Greider C.W., Blackburn E.H. A telomeric sequence in the RNA of Tetrahymena telomerase required for telomere repeat synthesis. Nature. 1989;337:331–337. doi: 10.1038/337331a0. [DOI] [PubMed] [Google Scholar]
- 32.Lue N.F., Chan J., Wright W.E., Hurwitz J. The CDC13-STN1-TEN1 complex stimulates Pol alpha activity by promoting RNA priming and primase-to-polymerase switch. Nat. Commun. 2014;5:5762. doi: 10.1038/ncomms6762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wang F., Stewart J.A., Kasbek C., Zhao Y., Wright W.E., Price C.M. Human CST has independent functions during telomere duplex replication and C-strand fill-in. Cell Rep. 2012;2:1096–1103. doi: 10.1016/j.celrep.2012.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lawlor R.T., Veronese N., Pea A., Nottegar A., Smith L., Pilati C., Demurtas J., Fassan M., Cheng L., Luchini C. Alternative lengthening of telomeres (ALT) influences survival in soft tissue sarcomas: A systematic review with meta-analysis. BMC Cancer. 2019;19:232–238. doi: 10.1186/s12885-019-5424-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Henson J.D., Hannay J.A., McCarthy S.W., Royds J.A., Yeager T.R., Robinson R.A., Wharton S.B., Jellinek D.A., Arbuckle S.M., Yoo J., et al. A robust assay for alternative lengthening of telomeres in tumors shows the significance of alternative lengthening of telomeres in sarcomas and astrocytomas. Clin. Cancer Res. 2005;11:217–225. [PubMed] [Google Scholar]
- 36.Zhou J., Reddy M.V. MR Imaging Characteristics Associate with Tumor-Associated Macrophages in Glioblastoma and Provide an Improved Signature for Survival Prognostication. AJNR. Am. J. Neuroradiol. 2018;39:252–259. doi: 10.3174/ajnr.A5441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pompili L., Leonetti C., Biroccio A., Salvati E. Diagnosis and treatment of ALT tumors: Is Trabectedin a new therapeutic option? J. Exp. Clin. Cancer Res. 2017;36:189–198. doi: 10.1186/s13046-017-0657-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Henson J.D., Reddel R.R. Assaying and investigating alternative lengthening of telomeres activity in human cells and cancers. FEBS Lett. 2010;584:3800–3811. doi: 10.1016/j.febslet.2010.06.009. [DOI] [PubMed] [Google Scholar]
- 39.Heaphy C.M., Subhawong A.P., Hong S.M., Goggins M.G., Montgomery E.A., Gabrielson E., Netto G.J., Epstein J.I., Lotan T.L., Westra W.H., et al. Prevalence of the alternative lengthening of telomeres telomere maintenance mechanism in human cancer subtypes. Am. J. Pathol. 2011;179:1608–1615. doi: 10.1016/j.ajpath.2011.06.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Xue Y., Li L., Zhang D., Wu K., Chen Y., Zeng J., Wang X., He D. Twisted epithelial-to-mesenchymal transition promotes progression of surviving bladder cancer T24 cells with hTERT-dysfunction. PLoS ONE. 2011;6:e27748. doi: 10.1371/journal.pone.0027748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Perrem K., Bryan T.M., Englezou A., Hackl T., Moy E.L., Reddel R.R. Repression of an alternative mechanism for lengthening of telomeres in somatic cell hybrids. Oncogene. 1999;18:3383–3390. doi: 10.1038/sj.onc.1202752. [DOI] [PubMed] [Google Scholar]
- 42.Hu J., Hwang S.S., Liesa M., Gan B., Sahin E., Jaskelioff M., Ding Z., Ying H., Boutin A.T., Zhang H., et al. Antitelomerase therapy provokes ALT and mitochondrial adaptive mechanisms in cancer. Cell. 2012;148:651–663. doi: 10.1016/j.cell.2011.12.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chang S., Khoo C.M., Naylor M.L., Maser R.S., DePinho R.A. Telomere-based crisis: Functional differences between telomerase activation and ALT in tumor progression. Genes Dev. 2003;17:88–100. doi: 10.1101/gad.1029903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pickett H.A., Reddel R.R. Molecular mechanisms of activity and derepression of alternative lengthening of telomeres. Nat. Struct. Mol. Biol. 2015;22:875–880. doi: 10.1038/nsmb.3106. [DOI] [PubMed] [Google Scholar]
- 45.Episkopou H., Draskovic I., Van Beneden A., Tilman G., Mattiussi M., Gobin M., Arnoult N., Londono-Vallejo A., Decottignies A. Alternative Lengthening of Telomeres is characterized by reduced compaction of telomeric chromatin. Nucleic Acids Res. 2014;42:4391–4405. doi: 10.1093/nar/gku114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gibbons R.J., McDowell T.L., Raman S., O’Rourke D.M., Garrick D., Ayyub H., Higgs D.R. Mutations in ATRX, encoding a SWI/SNF-like protein, cause diverse changes in the pattern of DNA methylation. Nat. Genet. 2000;24:368–371. doi: 10.1038/74191. [DOI] [PubMed] [Google Scholar]
- 47.Nan X., Hou J., Maclean A., Nasir J., Lafuente M.J., Shu X., Kriaucionis S., Bird A. Interaction between chromatin proteins MECP2 and ATRX is disrupted by mutations that cause inherited mental retardation. Proc. Natl. Acad. Sci. USA. 2007;104:2709–2714. doi: 10.1073/pnas.0608056104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Watson L.A., Solomon L.A., Li J.R., Jiang Y., Edwards M., Shin-ya K., Beier F., Berube N.G. Atrx deficiency induces telomere dysfunction, endocrine defects, and reduced life span. J. Clin. Investig. 2013;123:2049–2063. doi: 10.1172/JCI65634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Li F., Deng Z., Zhang L., Wu C., Jin Y., Hwang I., Vladimirova O., Xu L., Yang L., Lu B., et al. ATRX loss induces telomere dysfunction and necessitates induction of alternative lengthening of telomeres during human cell immortalization. EMBO J. 2019;38:e96659. doi: 10.15252/embj.201796659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Cesare A.J., Reddel R.R. Alternative lengthening of telomeres: Models, mechanisms and implications. Nat. Rev. Genet. 2010;11:319–330. doi: 10.1038/nrg2763. [DOI] [PubMed] [Google Scholar]
- 51.Bryan T.M., Englezou A., Gupta J., Bacchetti S., Reddel R.R. Telomere elongation in immortal human cells without detectable telomerase activity. EMBO J. 1995;14:4240–4248. doi: 10.1002/j.1460-2075.1995.tb00098.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Yu J., Lan J., Wang C., Wu Q., Zhu Y., Lai X., Sun J., Jin C., Huang H. PML3 interacts with TRF1 and is essential for ALT-associated PML bodies assembly in U2OS cells. Cancer Lett. 2010;291:177–186. doi: 10.1016/j.canlet.2009.10.009. [DOI] [PubMed] [Google Scholar]
- 53.Amorim J.P., Santos G., Vinagre J., Soares P. The Role of ATRX in the Alternative Lengthening of Telomeres (ALT) Phenotype. Genes. 2016;7:66. doi: 10.3390/genes7090066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Yeager T.R., Neumann A.A., Englezou A., Huschtscha L.I., Noble J.R., Reddel R.R. Telomerase-negative immortalized human cells contain a novel type of promyelocytic leukemia (PML) body. Cancer Res. 1999;59:4175–4179. [PubMed] [Google Scholar]
- 55.Osterwald S., Deeg K.I., Chung I., Parisotto D., Worz S., Rohr K., Erfle H., Rippe K. PML induces compaction, TRF2 depletion and DNA damage signaling at telomeres and promotes their alternative lengthening. J. Cell Sci. 2015;128:1887–1900. doi: 10.1242/jcs.148296. [DOI] [PubMed] [Google Scholar]
- 56.Henson J.D., Cao Y., Huschtscha L.I., Chang A.C., Au A.Y., Pickett H.A., Reddel R.R. DNA C-circles are specific and quantifiable markers of alternative-lengthening-of-telomeres activity. Nat. Biotechnol. 2009;27:1181–1185. doi: 10.1038/nbt.1587. [DOI] [PubMed] [Google Scholar]
- 57.Cesare A.J., Griffith J.D. Telomeric DNA in ALT cells is characterized by free telomeric circles and heterogeneous t-loops. Mol. Cell Biol. 2004;24:9948–9957. doi: 10.1128/MCB.24.22.9948-9957.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cesare A.J., Kaul Z., Cohen S.B., Napier C.E., Pickett H.A., Neumann A.A., Reddel R.R. Spontaneous occurrence of telomeric DNA damage response in the absence of chromosome fusions. Nat. Struct. Mol. Biol. 2009;16:1244–1251. doi: 10.1038/nsmb.1725. [DOI] [PubMed] [Google Scholar]
- 59.Mender I., Shay J.W. Telomere Dysfunction Induced Foci (TIF) Analysis. Bio-Protocol. 2015;5:e1656. doi: 10.21769/BioProtoc.1656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Dilley R.L., Verma P., Cho N.W., Winters H.D., Wondisford A.R., Greenberg R.A. Break-induced telomere synthesis underlies alternative telomere maintenance. Nature. 2016;539:54–58. doi: 10.1038/nature20099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Henson J.D., Neumann A.A., Yeager T.R., Reddel R.R. Alternative lengthening of telomeres in mammalian cells. Oncogene. 2002;21:598–610. doi: 10.1038/sj.onc.1205058. [DOI] [PubMed] [Google Scholar]
- 62.Cesare A.J., Heaphy C.M., O’Sullivan R.J. Visualization of Telomere Integrity and Function In Vitro and In Vivo Using Immunofluorescence Techniques. Curr. Protoc. Cytom. 2015;73:1–31. doi: 10.1002/0471142956.cy1240s73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Lau L.M., Dagg R.A., Henson J.D., Au A.Y., Royds J.A., Reddel R.R. Detection of alternative lengthening of telomeres by telomere quantitative PCR. Nucleic Acids Res. 2013;41:34–42. doi: 10.1093/nar/gks781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Roumelioti F.M., Sotiriou S.K., Katsini V., Chiourea M., Halazonetis T.D. Alternative lengthening of human telomeres is a conservative DNA replication process with features of break-induced replication. EMBO Rep. 2016;17:1731–1737. doi: 10.15252/embr.201643169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Cox K.E., Marechal A., Flynn R.L. SMARCAL1 Resolves Replication Stress at ALT Telomeres. Cell Rep. 2016;14:1032–1040. doi: 10.1016/j.celrep.2016.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Agathangelou K., Apostolou Z., Garinis G.A. Nuclear DNA Damage and Ageing. Subcell Biochem. 2018;90:309–322. doi: 10.1007/978-981-13-2835-0_10. [DOI] [PubMed] [Google Scholar]
- 67.Fallet E., Jolivet P., Soudet J., Lisby M., Gilson E., Teixeira M.T. Length-dependent processing of telomeres in the absence of telomerase. Nucleic Acids Res. 2014;42:3648–3665. doi: 10.1093/nar/gkt1328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Tacconi E.M., Tarsounas M. How homologous recombination maintains telomere integrity. Chromosoma. 2015;124:119–130. doi: 10.1007/s00412-014-0497-2. [DOI] [PubMed] [Google Scholar]
- 69.Liu H., Xie Y., Zhang Z., Mao P., Liu J., Ma W., Zhao Y. Telomeric Recombination Induced by DNA Damage Results in Telomere Extension and Length Heterogeneity. Neoplasia. 2018;20:905–916. doi: 10.1016/j.neo.2018.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Zhang J.M., Yadav T., Ouyang J., Lan L., Zou L. Alternative Lengthening of Telomeres through Two Distinct Break-Induced Replication Pathways. Cell Rep. 2019;26:955–968. doi: 10.1016/j.celrep.2018.12.102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Verma P., Dilley R.L., Zhang T., Gyparaki M.T., Li Y., Greenberg R.A. RAD52 and SLX4 act nonepistatically to ensure telomere stability during alternative telomere lengthening. Genes Dev. 2019;33:221–235. doi: 10.1101/gad.319723.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Benson F.E., Baumann P., West S.C. Synergistic actions of Rad51 and Rad52 in recombination and DNA repair. Nature. 1998;391:401–404. doi: 10.1038/34937. [DOI] [PubMed] [Google Scholar]
- 73.Bhowmick R., Minocherhomji S., Hickson I.D. RAD52 Facilitates Mitotic DNA Synthesis Following Replication Stress. Mol. Cell. 2016;64:1117–1126. doi: 10.1016/j.molcel.2016.10.037. [DOI] [PubMed] [Google Scholar]
- 74.Makarova A.V., Stodola J.L., Burgers P.M. A four-subunit DNA polymerase ζ complex containing Pol δ accessory subunits is essential for PCNA-mediated mutagenesis. Nucleic Acids Res. 2012;40:11618–11626. doi: 10.1093/nar/gks948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Sobinoff A.P., Allen J.A., Neumann A.A., Yang S.F., Walsh M.E., Henson J.D., Reddel R.R. BLM and SLX4 play opposing roles in recombination-dependent replication at human telomeres. EMBO J. 2017;36:2907–2919. doi: 10.15252/embj.201796889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Teichroeb J.H., Kim J., Betts D.H. The role of telomeres and telomerase reverse transcriptase isoforms in pluripotency induction and maintenance. RNA Biol. 2016;13:707–719. doi: 10.1080/15476286.2015.1134413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Huang Y., Liang P., Liu D., Huang J., Songyang Z. Telomere regulation in pluripotent stem cells. Protein Cell. 2014;5:194–202. doi: 10.1007/s13238-014-0028-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Flores I., Canela A., Vera E., Tejera A., Cotsarelis G., Blasco M.A. The longest telomeres: A general signature of adult stem cell compartments. Genes Dev. 2008;22:654–667. doi: 10.1101/gad.451008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Liu L. Linking Telomere Regulation to Stem Cell Pluripotency. Trends Genet. 2017;33:16–33. doi: 10.1016/j.tig.2016.10.007. [DOI] [PubMed] [Google Scholar]
- 80.Pickett H.A., Cesare A.J., Johnston R.L., Neumann A.A., Reddel R.R. Control of telomere length by a trimming mechanism that involves generation of t-circles. EMBO J. 2009;28:799–809. doi: 10.1038/emboj.2009.42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Guo R., Ye X., Yang J., Zhou Z., Tian C., Wang H., Wang H., Fu H., Liu C., Zeng M., et al. Feeders facilitate telomere maintenance and chromosomal stability of embryonic stem cells. Nat. Commun. 2018;9:2620–2635. doi: 10.1038/s41467-018-05038-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Zalzman M., Falco G., Sharova L.V., Nishiyama A., Thomas M., Lee S.L., Stagg C.A., Hoang H.G., Yang H.T., Indig F.E., et al. Zscan4 regulates telomere elongation and genomic stability in ES cells. Nature. 2010;464:858–863. doi: 10.1038/nature08882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Nakai-Futatsugi Y., Niwa H. Zscan4 Is Activated after Telomere Shortening in Mouse Embryonic Stem Cells. Stem Cell Rep. 2016;6:483–495. doi: 10.1016/j.stemcr.2016.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Dan J., Liu Y., Liu N., Chiourea M., Okuka M., Wu T., Ye X., Mou C., Wang L., Wang L., et al. Rif1 maintains telomere length homeostasis of ESCs by mediating heterochromatin silencing. Dev. Cell. 2014;29:7–19. doi: 10.1016/j.devcel.2014.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Shi T., Bunker R.D., Mattarocci S., Ribeyre C., Faty M., Gut H., Scrima A., Rass U., Rubin S.M., Shore D., et al. Rif1 and Rif2 shape telomere function and architecture through multivalent Rap1 interactions. Cell. 2013;153:1340–1353. doi: 10.1016/j.cell.2013.05.007. [DOI] [PubMed] [Google Scholar]
- 86.Buonomo S.B., Wu Y., Ferguson D., de Lange T. Mammalian Rif1 contributes to replication stress survival and homology-directed repair. J. Cell Biol. 2009;187:385–398. doi: 10.1083/jcb.200902039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Chapman J.R., Barral P., Vannier J.B., Borel V., Steger M., Tomas-Loba A., Sartori A.A., Adams I.R., Batista F.D., Boulton S.J. RIF1 is essential for 53BP1-dependent nonhomologous end joining and suppression of DNA double-strand break resection. Mol. Cell. 2013;49:858–871. doi: 10.1016/j.molcel.2013.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zeng S., Liu L., Sun Y., Xie P., Hu L., Yuan D., Chen D., Ouyang Q., Lin G., Lu G. Telomerase-mediated telomere elongation from human blastocysts to embryonic stem cells. J. Cell Sci. 2014;127:752–762. doi: 10.1242/jcs.131433. [DOI] [PubMed] [Google Scholar]
- 89.Huang J., Wang F., Okuka M., Liu N., Ji G., Ye X., Zuo B., Li M., Liang P., Ge W.W., et al. Association of telomere length with authentic pluripotency of ES/iPS cells. Cell Res. 2011;21:779–792. doi: 10.1038/cr.2011.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Liu L., Bailey S.M., Okuka M., Munoz P., Li C., Zhou L., Wu C., Czerwiec E., Sandler L., Seyfang A., et al. Telomere lengthening early in development. Nat. Cell Biol. 2007;9:1436–1441. doi: 10.1038/ncb1664. [DOI] [PubMed] [Google Scholar]
- 91.Varela E., Schneider R.P., Ortega S., Blasco M.A. Different telomere-length dynamics at the inner cell mass versus established embryonic stem (ES) cells. Proc. Natl. Acad. Sci. USA. 2011;108:15207–15212. doi: 10.1073/pnas.1105414108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Wang F., Yin Y., Ye X., Liu K., Zhu H., Wang L., Chiourea M., Okuka M., Ji G., Dan J., et al. Molecular insights into the heterogeneity of telomere reprogramming in induced pluripotent stem cells. Cell Res. 2012;22:757–768. doi: 10.1038/cr.2011.201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Jiang J., Lv W., Ye X., Wang L., Zhang M., Yang H., Okuka M., Zhou C., Zhang X., Liu L., et al. Zscan4 promotes genomic stability during reprogramming and dramatically improves the quality of iPS cells as demonstrated by tetraploid complementation. Cell Res. 2013;23:92–106. doi: 10.1038/cr.2012.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Park H.S., Hwang I., Choi K.A., Jeong H., Lee J.Y., Hong S. Generation of induced pluripotent stem cells without genetic defects by small molecules. Biomaterials. 2015;39:47–58. doi: 10.1016/j.biomaterials.2014.10.055. [DOI] [PubMed] [Google Scholar]
- 95.Fu H., Tian C.-L., Ye X., Sheng X., Wang H., Liu Y., Liu L. Dynamics of Telomere Rejuvenation during Chemical Induction to Pluripotent Stem Cells. Stem Cell Rep. 2018;11:70–87. doi: 10.1016/j.stemcr.2018.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Marion R.M., Strati K., Li H., Tejera A., Schoeftner S., Ortega S., Serrano M., Blasco M.A. Telomeres acquire embryonic stem cell characteristics in induced pluripotent stem cells. Cell Stem Cell. 2009;4:141–154. doi: 10.1016/j.stem.2008.12.010. [DOI] [PubMed] [Google Scholar]
- 97.Wong L.H. Epigenetic regulation of telomere chromatin integrity in pluripotent embryonic stem cells. Epigenomics. 2010;2:639–655. doi: 10.2217/epi.10.49. [DOI] [PubMed] [Google Scholar]
- 98.Meshorer E., Yellajoshula D., George E., Scambler P.J., Brown D.T., Misteli T. Hyperdynamic plasticity of chromatin proteins in pluripotent embryonic stem cells. Dev. Cell. 2006;10:105–116. doi: 10.1016/j.devcel.2005.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Garcia-Cao M., O’Sullivan R., Peters A.H., Jenuwein T., Blasco M.A. Epigenetic regulation of telomere length in mammalian cells by the Suv39h1 and Suv39h2 histone methyltransferases. Nat. Genet. 2004;36:94–99. doi: 10.1038/ng1278. [DOI] [PubMed] [Google Scholar]
- 100.Benetti R., Gonzalo S., Jaco I., Schotta G., Klatt P., Jenuwein T., Blasco M.A. Suv4-20h deficiency results in telomere elongation and derepression of telomere recombination. J. Cell Biol. 2007;178:925–960. doi: 10.1083/jcb.200703081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Lu F., Liu Y., Jiang L., Yamaguchi S., Zhang Y. Role of Tet proteins in enhancer activity and telomere elongation. Genes Dev. 2014;28:2103–2119. doi: 10.1101/gad.248005.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Yang J., Guo R., Wang H., Ye X., Zhou Z., Dan J., Wang H., Gong P., Deng W., Yin Y., et al. Tet Enzymes Regulate Telomere Maintenance and Chromosomal Stability of Mouse ESCs. Cell Rep. 2016;15:1809–1821. doi: 10.1016/j.celrep.2016.04.058. [DOI] [PubMed] [Google Scholar]
- 103.Le R., Kou Z., Jiang Y., Li M., Huang B., Liu W., Li H., Kou X., He W., Rudolph K.L., et al. Enhanced telomere rejuvenation in pluripotent cells reprogrammed via nuclear transfer relative to induced pluripotent stem cells. Cell Stem Cell. 2014;14:27–39. doi: 10.1016/j.stem.2013.11.005. [DOI] [PubMed] [Google Scholar]
- 104.De Vitis M., Berardinelli F., Sgura A. Telomere Length Maintenance in Cancer: At the Crossroad between Telomerase and Alternative Lengthening of Telomeres (ALT) Int. J. Mol. Sci. 2018;19:606. doi: 10.3390/ijms19020606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Dyer M.A., Qadeer Z.A., Valle-Garcia D., Bernstein E. Atrx and daxx: Mechanisms and mutations. Cold Spring Harbor Perspect. Med. 2017;7:a026567. doi: 10.1101/cshperspect.a026567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Schwartzentruber J., Korshunov A., Liu X.Y., Jones D.T., Pfaff E., Jacob K., Sturm D., Fontebasso A.M., Quang D.A., Tonjes M., et al. Driver mutations in histone H3.3 and chromatin remodelling genes in paediatric glioblastoma. Nature. 2012;482:226–231. doi: 10.1038/nature10833. [DOI] [PubMed] [Google Scholar]
- 107.Jiao Y., Shi C., Edil B.H., de Wilde R.F., Klimstra D.S., Maitra A., Schulick R.D., Tang L.H., Wolfgang C.L., Choti M.A., et al. DAXX/ATRX, MEN1, and mTOR pathway genes are frequently altered in pancreatic neuroendocrine tumors. Science. 2011;331:1199–1203. doi: 10.1126/science.1200609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Lovejoy C.A., Li W., Reisenweber S., Thongthip S., Bruno J., de Lange T., De S., Petrini J.H., Sung P.A., Jasin M., et al. Loss of ATRX, genome instability, and an altered DNA damage response are hallmarks of the alternative lengthening of telomeres pathway. PLoS Genet. 2012;8:1002772. doi: 10.1371/journal.pgen.1002772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Han M., Napier C.E. Synthetic lethality of cytolytic HSV-1 in cancer cells with ATRX and PML deficiency. J. Cell Sci. 2019;132:222349–222358. doi: 10.1242/jcs.222349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Chen Y.J., Hakin-Smith V., Teo M., Xinarianos G.E., Jellinek D.A., Carroll T., McDowell D., MacFarlane M.R., Boet R., Baguley B.C., et al. Association of mutant TP53 with alternative lengthening of telomeres and favorable prognosis in glioma. Cancer Res. 2006;66:6473–6476. doi: 10.1158/0008-5472.CAN-06-0910. [DOI] [PubMed] [Google Scholar]
- 111.Celeghin A., Giunco S., Freguja R., Zangrossi M., Nalio S., Dolcetti R., De Rossi A. Short-term inhibition of TERT induces telomere length-independent cell cycle arrest and apoptotic response in EBV-immortalized and transformed B cells. Cell Death Dis. 2016;7:2562–2566. doi: 10.1038/cddis.2016.425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Wang F., Sheng J.F., Cai L., Xu Y., Liao H., Tao Z.Z. The Telomerase and Alternative Lengthening of Telomeres Mechanisms Regulate Laryngeal Cancer Cell Apoptosis via the PI3K/Akt Pathway. ORL. 2018;80:227–237. doi: 10.1159/000489461. [DOI] [PubMed] [Google Scholar]
- 113.Salvati E., Leonetti C., Rizzo A., Scarsella M., Mottolese M., Galati R., Sperduti I., Stevens M.F., D’Incalci M., Blasco M., et al. Telomere damage induced by the G-quadruplex ligand RHPS4 has an antitumor effect. J. Clin. Investig. 2007;117:3236–3247. doi: 10.1172/JCI32461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Rizzo A., Salvati E., Porru M., D’Angelo C., Stevens M.F., D’Incalci M., Leonetti C., Gilson E., Zupi G., Biroccio A. Stabilization of quadruplex DNA perturbs telomere replication leading to the activation of an ATR-dependent ATM signaling pathway. Nucleic Acids Res. 2009;37:5353–5364. doi: 10.1093/nar/gkp582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Porru M., Zizza P., Franceschin M., Leonetti C., Biroccio A. EMICORON: A multi-targeting G4 ligand with a promising preclinical profile. Biochim. Biophys. Acta (BBA) 2017;1861:1362–1370. doi: 10.1016/j.bbagen.2016.11.010. [DOI] [PubMed] [Google Scholar]
- 116.Zheng X.H., Nie X., Fang Y., Zhang Z., Xiao Y., Mao Z., Liu H., Ren J., Wang F., Xia L., et al. A Cisplatin Derivative Tetra-Pt(bpy) as an Oncotherapeutic Agent for Targeting ALT Cancer. J. Natl. Cancer Inst. 2017;109 doi: 10.1093/jnci/djx061. [DOI] [PubMed] [Google Scholar]
- 117.Wang Y., Yang J., Wild A.T., Wu W.H., Shah R., Danussi C., Riggins G.J., Kannan K., Sulman E.P., Chan T.A., et al. G-quadruplex DNA drives genomic instability and represents a targetable molecular abnormality in ATRX-deficient malignant glioma. Nat. Commun. 2019;10:943. doi: 10.1038/s41467-019-08905-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Neidle S. Quadruplex Nucleic Acids as Novel Therapeutic Targets. J. Med. Chem. 2016;59:5987–6011. doi: 10.1021/acs.jmedchem.5b01835. [DOI] [PubMed] [Google Scholar]
- 119.Bojovic B., Booth R.E., Jin Y., Zhou X., Crowe D.L. Alternative lengthening of telomeres in cancer stem cells in vivo. Oncogene. 2015;34:611–630. doi: 10.1038/onc.2013.603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Hu Y., Shi G., Zhang L., Li F., Jiang Y., Jiang S., Ma W., Zhao Y., Songyang Z., Huang J. Switch telomerase to ALT mechanism by inducing telomeric DNA damages and dysfunction of ATRX and DAXX. Sci. Rep. 2016;6:32280–32289. doi: 10.1038/srep32280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Gocha A.R., Nuovo G., Iwenofu O.H., Groden J. Human sarcomas are mosaic for telomerase-dependent and telomerase-independent telomere maintenance mechanisms: Implications for telomere-based therapies. Am. J. Pathol. 2013;182:41–48. doi: 10.1016/j.ajpath.2012.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Pezzolo A., Pistorio A., Gambini C., Haupt R., Ferraro M., Erminio G., De Bernardi B., Garaventa A., Pistoia V. Intratumoral diversity of telomere length in individual neuroblastoma tumors. Oncotarget. 2014;6:7493–7503. doi: 10.18632/oncotarget.2115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Xu B., Peng M., Song Q. The co-expression of telomerase and ALT pathway in human breast cancer tissues. Tumor Biol. 2014;35:4087–4093. doi: 10.1007/s13277-013-1534-0. [DOI] [PubMed] [Google Scholar]
- 124.Nakamura T., Matsumine A., Sudo A. The value of trabectedin in the treatment of soft tissue sarcoma. Ther. Clin. Risk Manag. 2016;12:73–79. doi: 10.2147/TCRM.S84789. [DOI] [PMC free article] [PubMed] [Google Scholar]