Abstract
Proliferative myositis (PM) and nodular fasciitis (NF) are two diseases easily misdiagnosed as cancer, often promoting unnecessary invasive procedures. To make accurate diagnoses of PM and NF and for the differential diagnosis between them, we performed a retrospective study to evaluate the roles of the clinical, radiologic, and pathologic characteristics of PM and NF. With an emphasis on the clinicopathologic and radiologic characteristics, we conducted a retrospective study of 8 cases of PM and 64 cases of NF that were diagnosed between 2012 and 2018. According to MRI findings, the lesions of PM and NF appeared as homogeneous masses with homogenous hypointensity or isointensity on T1-weighted images and as moderately or markedly hyperintense signals on T2-weighted images compared to skeletal muscle. In terms of histopathologic features, the differences between PM and NF mainly consisted of the presence of ganglion-like myofibroblasts with vesicular nuclei and basophilic cytoplasm in PM. The areas abundant in myxoid stroma with inflammatory infiltration that did not have abundant ganglion-like cells suggest NF. Immunohistochemically, the spindle-shaped cells of PM stained positive for smooth muscle actin (SMA), while the ganglion-like cells were negative. The spindle-shaped cells of NF showed diffuse expression of SMA, calponin, and vimentin. Our comprehensive study further demonstrated that PM and NF had a wide clinicopathologic and radiologic spectrum. Correlation with the clinical, radiologic and pathologic characteristics may help clinicians and pathologists make accurate diagnoses.
Keywords: Proliferative myositis, nodular fasciitis, clinical characteristics, radiological characteristics, pathology
Introduction
Proliferative myositis (PM) and nodular fasciitis (NF) are within a spectrum of benign myofibroblastic tumor-like lesions [1,2]. Since they appear as rapidly growing masses, PM and NF are easily and mistakenly diagnosed as each other or even as sarcoma [1,3]. The differential diagnosis depends on the consideration of magnetic resonance imaging (MRI) or computed tomography (CT) images and pathologic and clinical characteristics [4-9]. The clinical, radiologic and pathologic findings are variable, and the distinction between soft tissue masses is difficult. These challenges may lead to unnecessary radical surgery as treatment for these benign lesions.
As they are prone to misdiagnosis, we performed a retrospective study of the clinical, radiologic and pathologic characteristics of PM and NF. With this work, we hope to deepen the understanding of PM and NF and to broaden the differential diagnostic possibilities to help surgeons, radiologists, and pathologists avoid misdiagnosing soft tissue masses.
Materials and methods
Patient cohort
With the hospital information management system, a retrospective review of the medical records was performed among all patients with pathological diagnoses of PM or NF that were evaluated at the Affiliated Hospital of Guangdong Medical University between Nov 2012 and Nov 2018. Using the terms “Proliferative myositis” or “Nodular fasciitis”, eight cases of PM as well as sixty-four cases of NF were accrued from the files of the Department of Pathology. A retrospective search of the imaging data features of those patients with histologically proven PM or NF was then conducted in the files of the Department of Radiology. Data, including diagnosis, age, sex, presenting symptoms, comorbidities, diagnostic procedures and imaging, surgical management, complications, and follow-up time, were extracted by the researchers from the medical records and radiology and pathology reports and were analyzed.
Imaging
MRI was performed on a 3.0-T scanner (General Electric Company, Discovery MR750 3.0 Tesla), using spin-echo (SE) T1-weighted [repetition time (TR) 360-610/echo time (TE) 1.6-9.4] and spin-echo (SE) T2-weighted [repetition time (TR) 2300-5500/echo time (TE) 75-90], without fat saturation, acquiring imaging sequences in axial, coronal and sagittal planes using 4- to 8-mm slices.
All CT examinations were performed on spiral scanners (General Electric Company, Optima TM CT680 or Siemens, Somatomsensation64) to produce postcontrast axial images with 0.6- to 1.0-mm collimation.
The exclusion criteria include cases with no corresponding radiology since this was a correlation study. Of the 8 patients with histologically proven PM, they all had recorded examinations on radiology before excision. Three patients received MRIs. CT scans were performed for 5 patients. Of the 64 patients with histologically proven NF, 21 patients had recorded examinations on radiology before excision. Thirteen patients received MRIs. CT scans were performed for 8 patients.
Image analysis
CT and MR imaging characteristics were investigated regarding the location, size, internal content, and margins of the lesion. The degrees of the MRI and CT scans were subjectively assessed as being hypointense, isointense, and hyperintense signal compared to skeletal muscle. Imaging interpretations of CT and MRI scans were conducted by two senior radiologists. We reexamined all cases’ imaging data to investigate the imaging characteristics of PM or NF.
Histopathology
Six patients with histologically proven PM had records of immunohistochemical examinations in the files of the Department of Pathology. Smooth muscle actin (SMA), vimentin, S-100 protein, cytokeratin (CK), desmin, and myoglobin were evaluated in those patients’ specimens. Thirty-nine patients with histologically proven NF had records of immunohistochemical examinations in the files of the Department of Pathology. SMA, calponin, vimentin, desmin, CK, CD34 and S-100 protein were evaluated in those patients’ specimens.
Histopathologic analysis
For each case’s pathological diagnosis, the pathology specimens were reviewed by two senior pathologists. We reexamined all cases’ 4-μm-thick hematoxylin and eosin (H&E)-stained sections and immunohistochemical sections to investigate the histopathology of PM and NF.
Statistical analysis
GraphPad Prism 5 (GraphPad software, La Jolla, CA, USA) was used for unpaired t-test analyses and to analyze the median and average ages of patients with NF.
Results
Clinical findings
Clinically, all patients had symptoms of masses that grew rapidly for 1 week to 3 months. All patients were treated by surgery. In the patients with follow-up information (ranging from 3 to 65 months), no patient experienced local recurrence.
The ages at presentation for the sixty-four patients with NF ranged from 3 to 84 years. Approximately 64.06% of the patients were between 21 and 50 years old, whereas pediatric (aged <10 years, 3 cases) and elderly patients (aged >60 years, 4 cases) accounted for 4.7% and 6.3% of all patients, respectively. The mean age of the patients was 37.48 years, and the median age was 38 years. Forty-one patients in this series were male, and twenty-three patients were female, for a M:F ratio of 1.78. Clinically, most patients presented with a solitary subcutaneous nodule, which grew rapidly for 2 weeks to 3 months before surgical excision. However, patients with a preoperative duration of several months were not uncommon. One patient presented with a 14-month history. Approximately half of the patients presented with slight pain or tenderness on palpation.
With regard to the anatomic site, the lesions were in the neck, armpits, shoulders, wrists, hips and other parts of the body. Twenty-five patients (39.06%) had lesions located in the upper extremities, especially the forearm (n=9 cases, 14%). In total, 13 patients (20.3%) had lesions in the head and neck region, 18 patients (28.13%) had lesions in the trunk, 6 patients (9.38%) had lesions in the lower extremities, 1 patient (1.56%) had lesions in the groin and 1 patient (1.56%) had lesions in the vulva. Of the lesions in the trunk, 2 were located in the breast. All patients were treated by surgery. Among the patients with follow-up information (ranging from 4 to 98 months), no patient experienced local recurrence.
Imaging findings
Of the 8 patients with PM, 3 patients received MRIs. One patient’s T1-weighted image (WI) mainly presented isointense signal and a small patchy shadow of slightly hyperintense signal (Figure 1A); T2WI showed hyperintense signal, with empty blood vessels (Figure 1B), and diffusion-weighted imaging (DWI) showed hyperintense signal. The other 2 patients’ T1WIs showed slightly hypointense signal, and T2WIs and DWIs showed hyperintense signal. In the MRIs of three patients, the T1WIs with enhanced scanning were all significantly enhanced (Figure 1C). CT scans were performed for 5 patients. One patient’s mass, which was in the chest wall, presented uneven density, unclear boundaries and expansive growth (Figures 1D, S1). The mass invaded the fifth front rib, leading to significant bone destruction (Figures 1E, S1). The masses in the other 3 patients presented uneven density with blurred edges. The image of one patient’s mass in the chest wall displayed a slightly hypointense density with blurred edges.
Figure 1.
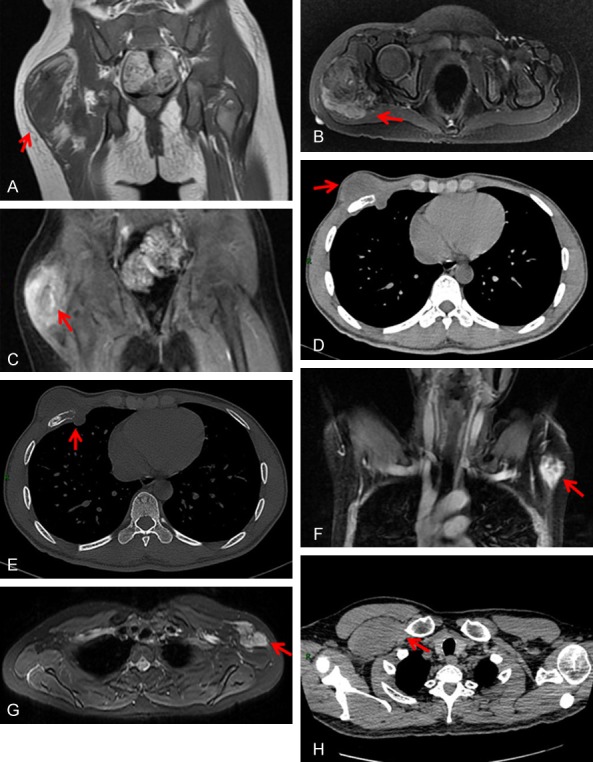
Radiologic examinations of PM and NF. A-C. MRI examinations of PM. A 4-year-old girl with an irregular soft tissue mass in the right hip muscle. A. T1WI showed mainly isointense signal and a small patch of slightly high signal. B. T2WI showed high signal, with empty blood vessels. C. Enhanced scanning showed significant enhancement and enhanced vascular shadows. D, E. CT examinations of PM. A 28-year-old man with a mass in the right front chest wall. D. A mass with a CT value of 16 Hu, uneven density, unclear boundaries, and expansive growth. E. The mass invaded the fifth front rib, with significant bone destruction. F, G. MRI examinations of NF. A 44-year-old woman with irregular soft tissue masses on the left side of the armpit. F. Enhanced scanning with significant enhancement and tortuous vascular shadows. G. T2WI showed high signal in the lesion, with empty blood vessels. H. CT examinations of NF, A 56-year-old man with a mass in the right front chest wall. A large soft tissue mass next to the right small chest muscle; the signals from the lesions were isodense compared with those from the adjacent muscles.
Of the 21 patients with NF, 13 patients had MRIs before excision. CT data were available in 8 cases. The MRI scans usually presented slightly hypointense or isointense signals on T1WIs, and the enhanced scans were significantly unevenly enhanced (Figure 1F); T2WIs (Figure 1G) and DWIs usually showed hyperintense signals. On CT, the signals from the lesions were hypointense or isodense compared with those from the adjacent muscles (Figure 1H), and the enhanced scanning did not show significant enhancement.
Pathological findings
All lesions of PM were described as well circumscribed, soft, slightly nodular, masses with pale yellow fat-like tissue or gray translucent tissue in the foci on cut sections. The excised specimens measured from 1.0 to 6.0 cm in diameter, with mean and median sizes of 2.5 and 1.83 cm. All lesions of NF were described as well circumscribed, soft, rubbery-to-firm nodules or masses with glistening appearance on cut sections. The excised specimens measured from 0.3 to 6.0 cm in diameter, with mean and median sizes of 2.35 and 2.18 cm, respectively. Of note, the lesions were smaller than 3.95 cm in 64 cases (87.5%). Those with a greatest dimension larger than 4 cm were mostly deeply seated or intramuscular lesions.
In pathology samples from the 8 patients with PM, fibroblasts were widely proliferated in the interstitium, and the local muscle tissue was atrophic or completely replaced. Fibroblast proliferation separated the muscle bundle. Samples of the eight patients were observed under a low-power microscope, and the transverse section of the lesion had a checkerboard pattern (Figure 2A, 2B). In all samples, large-sized spindle cells were observed and were polygonal or irregular, with a clear nuclear membrane and visible nucleoli (Figure 2C). Furthermore, there were ganglion-like basophilic giant cells or ganglion-like myofibroblasts in the area of mucoid degeneration in one sample (Figure 2D).
Figure 2.

Pathologic examinations of PM and NF. A-D. A 57-year-old woman with a mass in the left thigh. Microscopic examination revealed proliferating spindle-shaped cells, checkerboard-like pattern, and larger basophilic cells that resemble ganglion cells, which are diagnostic features for PM. A. Low-power microscopy view of proliferative myositis. The muscle bundles are separated by endomysial, perimysial, and epimysial proliferation of fibrous connective tissue in a checkerboard-like pattern (hematoxylin-eosin, original magnifications × 100). B. Medium-power microscopy view of proliferating spindle-shaped cells and skeletal muscle fibers with secondary atrophy (hematoxylin-eosin, original magnifications × 200). C. High-power microscopy view of basophilic ganglion-like cells (hematoxylin-eosin, original magnifications × 400). D. Ganglion-like basophilic giant cells or ganglion-like myofibroblasts in the area of mucoid degeneration (hematoxylin-eosin, original magnifications × 200). E-G. A 40-year-old woman with a mass in the chest wall. Microscopic examination revealed areas of uniform bland spindle cells that are arranged in short intersecting fascicles within a loose stroma accompanied by extravasated erythrocytes and scattered lymphocytes, which are diagnostic features for NF. E. The lesion was composed of spindle-shaped cells with abundant elastic fibers. The spindle-shaped cells grew in a mucus-like matrix and had irregular infiltrative margins partially extending into the adipose tissue (hematoxylin-eosin, original magnifications × 100). F. Proliferating spindle-shaped cells are arranged in bundles and interlaced bands (hematoxylin-eosin, original magnifications × 100). G. Spindle-shaped cells indicate the proliferation of fibroblasts or myofibroblasts without hyperchromasia or pleomorphism. Extravasated red blood cells are also present (hematoxylin-eosin, original magnifications × 400). H-K. Immunohistochemical results of PM. A 57-year-old woman with a mass in the left thigh. H. The spindle-shaped cells stained diffuse positive for SMA (original magnifications × 200). I. The ganglion-like cells were negative for SMA (original magnifications × 400). J. PM was negative for myoglobin (original magnifications × 200). K. PM was negative for myoD1 (original magnifications × 200). L-O. Immunohistochemical results of NF. A 31-year-old man with a mass in the right thigh. L. The spindle-shaped cells were diffuse positive for SMA (original magnifications × 200). M. The spindle-shaped cells were positive for calponin (original magnifications × 200). N. The spindle-shaped cells were negative for desmin (original magnifications × 200). O. The spindle-shaped cells were negative for CD34 (original magnifications × 200).
In pathology samples from the 64 patients with NF, abundant spindle-shaped cells proliferated in the loose mucus-like matrix and infiltrated around the lesion boundary (Figure 2E). The proliferating spindle-shaped cells were arranged in bundles and intertwined bands (Figure 2F). Vascular hyperplasia, lymphocytic infiltration and red blood cell extravasation were also observed. In some cases, large spindle cells with mitotic figures were found, but there were no abnormal mitotic figures (Figure 2G).
In the immunohistochemical sections of PM, the spindle-shaped cells stained diffuse positive for vimentin and SMA (Figure 2H), whereas the ganglion-like cells stained strong diffuse positive for vimentin. Meanwhile, the ganglion-like cells were negative for SMA (Figure 2I). Both cell styles were negative for S-100 protein, desmin, CK, myoglobin (Figure 2J), and myoD1 (Figure 2K).
In the immunohistochemical sections of NF, the spindle-shaped cells showed diffuse expression of SMA (Figure 2L), calponin (Figure 2M) and vimentin. Other markers, including desmin (Figure 2N), CK, CD34 (Figure 2O), and S-100 protein, were all negative.
Discussion
A previous study found that most patients with PM were over 45 years old, but there were also pediatric cases [1,9-14]. In the 8 cases of PM, other than one case of a 4-year-old child and one case of a 28-year-old man, the other 6 patients were all over 45 years old. Prior to this description, only 11 cases of PM in children had been reported in the literature [11-13,15-17].
There are three patients who had lesions in the skeletal muscles of the chest, and one patient’s lesion was in the skeletal muscle of the thigh, which are the two most common lesion locations for PM. However, in our study, we found a 4-year-old female child with a lesion on the buttock. To the best of our knowledge, no similar case has been reported in previous studies [1,11-13,15-17]. The clinical progress of the masses seemed to be rapid, with growth periods ranging from 1 week to 3 months. Such a rapid growth rate was similar to that observed in previous reports [1,9,12,18].
The ages of the patients and the locations of the lesions in our study were all typical for NF. Lesions of NF can occur at any age, but presentation between the ages of 21 and 50 years is the most common [4,5,19]. A retrospective study reviewed 272 cases of NF and showed that the mean and median ages were both 36 years (range, newborn-84 years). Approximately 65% of the patients were between 21 and 50 years old, whereas pediatric (aged <10 years, 9 cases) and elderly patients (aged >60 years, 16 cases) accounted for 3.3% and 5.9% of the patients, respectively. Our study was mostly consistent with previous studies on the age of onset for NF [6]. In accordance with previous reports, lesions usually occurred in the upper limbs, particularly the forearms, followed by the head and neck region and the trunk. Less frequently, lesions occurred in the lower extremities, especially on the thigh [6,14,20,21].
There were occasional cases of lesions that developed in the breast, mesentery, groin, and vulva. In these series, there were 11 lesions in the breast, 8 lesions in the groin, and 7 lesions in the vulva [22-28]. In these unusual sites, NF might not be considered for the diagnosis, particularly for patients with unusually long clinical histories [6]. The importance of recognizing NF in the breast, vulva, and groin is to prevent its misdiagnosis as a malignancy [6,22,29,30]. In our study, approximately half of the patients presented with slight pain or tenderness on palpation. The clinical duration for most patients who presented with a rapidly growing solitary subcutaneous nodule ranged between 2 weeks and 3 months before surgical excision. This time period is generally similar to that of a previously reported series [6,19,20,28,31]. However, there was one patient with a 14-month clinical duration in our study.
PM and NF are rare in children, both in our study and in the reviewed literature. In our study and previous reviews, most cases of PM and NF presented with a solitary subcutaneous nodule that grew rapidly for 1 week to 3 months before surgical excision. Both entities can display rapid growth. PM and NF lesions can easily be mistaken for malignancy due to their rapid growth [4]. The most common locations for PM lesions were the chest in our study. However, the shoulder was also a common PM lesion location in our review [14,32,33]. These locations are quite different from those of NF lesions. The most common locations for NF lesions in our study were the upper extremities, especially the forearm, followed by the trunk, then the head and neck region, and finally the lower extremities.
MRIs of PM have been reported in several cases. Compared with skeletal muscle, the lesions appear as hypointense or isointense T1 signal, with almost homogeneous enhancement in contrast-enhanced T1WIs. T2WIs typically demonstrate a moderately or markedly hyperintense soft-tissue mass [8,9,11,12,34]. After an intravenous injection of contrast agent, the lesion demonstrated marked enhancement [1,8,12]. However, although MRI findings may be suggestive of an inflammatory process, such as PM, a definite diagnosis of PM is not always possible with MRI findings alone, and histologic confirmation is required [7,12,13].
No characteristic CT findings have been described for the diagnosis of PM. Some cases, including ours, have reported ill-defined, expansile lesions that appear iso- or hypointense compared to the surrounding muscle with unenhanced CT scanning [12,17]. Although the lesion may show homogenous or heterogeneous enhancement after contrast material administration, no enhancement may be detected in some cases [7,9,12,18]. CT may not play an unimportant role in the characterization of PM [7].
On the MRIs of NF, the lesion often appears as homogenous T1 signal that is iso- or hypointense to the adjacent skeletal muscle and as heterogeneous intermediate to hyperintense signal on T2WIs with enhancement after gadolinium administration [4-6,20]. This is consistent with the MRI features of our series. Furthermore, NF lesions also show heterogeneous intermediate-to-high signals with different enhancement patterns on T2- or STIR-weighted sequences, which are attributable to the variable components and cellular histology. Some cases may demonstrate distinct and discrete internal and central high T2W signal areas with low T2W signal intensity in the peripheral rim, which represents reticulated perilesional soft tissue edema on fluid-sensitive sequences [5,6]. However, one study reported that all of the lesions in three patients with NF appeared slightly hyperintense compared to skeletal muscle on T1WIs, although T2WIs appeared hyperintense and enhanced during scanning with heterogeneous contrast enhancement [21]. On CT, the image features of NF were usually fascia-based, oval in shape, and homogeneous and low or isodense compared with the adjacent muscles. In patients with cystic and mucoid degeneration, the contrast-enhanced CT scan showed a heterogeneous appearance, with prominent peripheral rim-like enhancement in some cases [5,6]. However, no distinctive enhancement patterns have been reported for CT or MRI [3].
Regardless of MRI or CT, there was no definitive image pattern for the diagnosis of PM or NF in either our series or previous studies. However, in our series, we unexpectedly found a case of PM, with a mass in the chest wall. On the CT scan, the mass had uneven density, unclear boundaries, and expansive growth; the mass invaded the fifth front rib, leading to significant bone destruction (Figure 1D, 1E). There have been no similar reports in previous studies [1,7-18,34]. Although bone involvement in NF is very rare and is easily visualized on CT [4,35,36], we found a study that reported a case of NF with osseous involvement of the hallux [37]. Therefore, these findings remind us that although there are no unique radiologic findings for PM and NF, these entities must be included in the preoperative differential diagnosis of soft-tissue masses that demonstrate rapid growth.
The characteristics of PM on histology include a checkerboard pattern of myofibroblasts that infiltrate the surrounding healthy muscle, ganglion-like basophilic giant cells, and no sign of atypical mitosis [1,12,14,18,32,38]. A previous report focused on the presence of ganglion-like cells and the possibility of these cells leading to a mistaken diagnosis of sarcoma due to their resemblance to rhabdomyoblasts [33]. In another study, the key distinguishing features between the two entities were the absence of cytoplasmic cross-striations in the ganglion-like cells and the presence of fibroblasts, both of which were useful for excluding rhabdomyosarcoma [38]. Although numbers of factors, including the increase in nuclear size, prominent nucleoli and high mitotic rate, can lead to concern, the smooth nuclear contour and finely distributed chromatin are indicative of the cell’s benign nature [39].
There are various histological characteristics of NF, including plump immature fibroblast-like spindle cells, chronic inflammatory cells, capillary proliferation, vascular channels with extravasated red blood cells and myxoid material [5,23]. Due to its rapid growth, rich cellularity and increased mitotic activity, NF may be confused with sarcoma [40], but NF can also be differentiated by a number of features, including: (a) the nuclei are not hyperchromatic; (b) the lesion does not extend into the skin; (c) there are a limited number of mitoses (not greater than 1 per high-power field) that are not atypical; and (d) there is a smaller amount of collagen in the stroma than in fibrosarcoma [23].
NF closely resembles PM. The difference between these two entities mainly consists of the presence of ganglion-like myofibroblasts with vesicular nuclei and basophilic cytoplasm in PM. These ganglion-like cells are absent in NF [1]. Furthermore, areas abundant in myxoid stroma with inflammatory infiltration that do not have abundant ganglion-like cells suggest NF [1]. Similar to a previously reported study [1], our series had one case of PM with mucoid degeneration accompanied by many ganglion-like myofibroblasts in the stroma (Figure 2D). However, areas in which the mucoid stroma with inflammatory infiltration is prominent but does not have abundant ganglion-like cells suggest NF (Figure 2E).
Previous studies showed the immunohistochemical profile of PM, which differed between the spindle-shaped cells and the ganglion-like cells. The spindle-shaped cells stained positive for vimentin and SMA, whereas the ganglion-like cells stained positive for vimentin. Both cell types were negative for S-100 protein, CK, desmin, and myoglobin [32,38,41]. Our study was mostly consistent with previous studies on the immunohistochemical features of PM (Figure 2H-K). The other features of PM, such as sudden onset, rapid growth rate, tenderness and inconclusive radiological findings, and cytomorphological features such as enlarged nuclei, prominent nucleoli, and high mitotic rate, can easily lead to a mistaken diagnosis of sarcoma, such as rhabdomyosarcoma [32,38]. However, the immunohistochemical profile of rhabdomyosarcoma is positive for myoglobin, desmin, and myoD1 [42-44]. Myogenin and myoD1 are myogenic nuclear transcription factors present early in skeletal muscle differentiation and are currently used as a standard approach to diagnosis, with a sensitivity exceeding 95% and a specificity of virtually 100% [44]. These markers are considered more sensitive and specific than desmin, with myogenin showing no reactivity in other spindle cell proliferations, such as NF, malignant peripheral nerve sheath tumor, leiomyosarcoma, or myofibrosarcoma, which may be included in the differential diagnosis [44]. Our study demonstrated that desmin, and myoglobin and myoD1 in particular, contribute greatly to the differential diagnosis between PM and NF.
The differential diagnosis of NF includes lesions mainly characterized by proliferation of spindle cells; spindle cell carcinoma and sarcoma among malignant lesions and fibromatosis and myofibroblastoma among benign lesions morphologically resemble NF and need to be distinguished [28]. There is no doubt of the importance of morphology for the confirmation of diagnosis, but immunohistochemistry is also a useful auxiliary means of identification. The characteristic immunostaining patterns of NF are positive for SMA, calponin, and vimentin, and negative for CD34, S-100, CK, and β-catenin [6,24,28]. Positive staining of SMA and calponin suggests myofibroblastic differentiation and less possibility of a sarcoma [24,45]. In our series, NF was diagnosed based on the immunohistochemical results (positive for SMA and calponin, negative for CD34, CK, and S-100) in addition to the morphologic findings.
The diagnosis of PM and NF relies on the clinical, radiologic, and pathologic characteristics. Considering these, PM and NF can be distinguished from each other and from proliferative lesions of soft tissue or even from sarcoma. For awareness of the rare occurrence of PM and NF in unusual locations and at uncommon age of onset, image features and clinical settings is critical.
Acknowledgements
This study was funded by Guangdong Medical University Research Fund (grant no. GDMUM 201818, GDMUM 201922 and GDMUM 201936), National Natural Science Foundation of China (grant no. 81702285), Natural Science Foundation of Guangdong Province (grant no. 2016A030313822) and Funds for the Construction of Basic Medical Disciplines in Guangdong Medical University (grant no. 4SG19047G). All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by the Institutional Ethical Review Board of the affiliated hospital of Guangdong Medical University. Due to the retrospective design of the current study and patient anonymization, the review board determined that informed consent was not required.
Disclosure of conflict of interest
None.
Supporting Information
References
- 1.Jarraya M, Parva P, Stone M, Klein MJ, Guermazi A. Atypical proliferative myositis: original MR description with pathologic correlation: case report. Skeletal Radiol. 2014;43:1155–1159. doi: 10.1007/s00256-014-1849-y. [DOI] [PubMed] [Google Scholar]
- 2.de Schepper AM, Vandevenne JE. Tumors of connective tissue. Imaging of Soft Tissue Tumors. 2006:167–202. [Google Scholar]
- 3.Hseu A, Watters K, Perez-Atayde A, Silvera VM, Rahbar R. Pediatric nodular fasciitis in the head and neck: evaluation and management. JAMA Otolaryngol Head Neck Surg. 2015;141:54–59. doi: 10.1001/jamaoto.2014.2797. [DOI] [PubMed] [Google Scholar]
- 4.Wang XL, De Schepper AM, Vanhoenacker F, De Raeve H, Gielen J, Aparisi F, Rausin L, Somville J. Nodular fasciitis: correlation of MRI findings and histopathology. Skeletal Radiol. 2002;31:155–161. doi: 10.1007/s00256-001-0462-z. [DOI] [PubMed] [Google Scholar]
- 5.Dinauer PA, Brixey CJ, Moncur JT, Fanburgsmith JC, Murphey MD. Pathologic and MR imaging features of benign fibrous soft-tissue tumors in adults. Radiographics. 2007;27:173–187. doi: 10.1148/rg.271065065. [DOI] [PubMed] [Google Scholar]
- 6.Lu L, Lao IW, Liu X, Yu L, Wang J. Nodular fasciitis: a retrospective study of 272 cases from China with clinicopathologic and radiologic correlation. Ann Diagn Pathol. 2015;19:180–185. doi: 10.1016/j.anndiagpath.2015.03.013. [DOI] [PubMed] [Google Scholar]
- 7.Pagonidis K, Raissaki M, Gourtsoyiannis N. Proliferative myositis: value of imaging. J Comput Assist Tomogr. 2005;29:108–111. doi: 10.1097/01.rct.0000150142.14113.70. [DOI] [PubMed] [Google Scholar]
- 8.Yiğit H, Turgut AT, Koşar P, Astarci HM, Koşar U. Proliferative myositis presenting with a checkerboard-like pattern on CT. Diagn Interv Radiol. 2009;15:139–142. [PubMed] [Google Scholar]
- 9.Mustafa Kemal D, Mahmut B, Okan A. Case 118: proliferative myositis. Radiology. 2007;244:613. doi: 10.1148/radiol.2442041504. [DOI] [PubMed] [Google Scholar]
- 10.Meis JM, Enzinger FM. Proliferative fasciitis and myositis of childhood. Am J Surg Pathol. 1992;16:364–372. doi: 10.1097/00000478-199204000-00005. [DOI] [PubMed] [Google Scholar]
- 11.Talbert RJ, Laor T, Yin H. Proliferative myositis: expanding the differential diagnosis of a soft tissue mass in infancy. Skeletal Radiol. 2011;40:1623–1627. doi: 10.1007/s00256-011-1274-4. [DOI] [PubMed] [Google Scholar]
- 12.Mulier S, Stas M, Delabie J, Lateur L, Gysen M, Dal Cin P, Robberecht C, De Wever I. Proliferative myositis in a child. Skeletal Radiol. 1999;28:703–709. doi: 10.1007/s002560050577. [DOI] [PubMed] [Google Scholar]
- 13.Pollock L, Fullilove S, Shaw DG, Malone M, Hill RA. Proliferative myositis in a child. A case report. J Bone Joint Surg Am. 1995;77:132–135. doi: 10.2106/00004623-199501000-00017. [DOI] [PubMed] [Google Scholar]
- 14.Rosai J. In: Rosai and Ackerman’s Surgical Pathology. Rosai J, editor. Singapore, Winsland House I, Killiney Road: 2012. pp. 201–201. [Google Scholar]
- 15.Ricón Recarey FJ, Cano Luis P, Martinez Guerrero RB. Proliferative myositis of the gastrocnemius muscle: a case report and review of the literature. European Journal of Orthopaedic Surgery & Traumatology. 2008;18:479–482. [Google Scholar]
- 16.Choi SS, Myer CM 3rd. Proliferative myositis of the mylohyoid muscle. Am J Otolaryngol. 1990;11:198–202. doi: 10.1016/0196-0709(90)90038-w. [DOI] [PubMed] [Google Scholar]
- 17.Kleinman GM, Zelem JD, Sanders FJ. Proliferative myositis in a two-year-old child. Pediatr Pathol. 1987;7:71–75. doi: 10.1080/15513818709177116. [DOI] [PubMed] [Google Scholar]
- 18.Colombo JR, Dagher W, Wein RO. Benign proliferative myositis of the sternohyoid muscle: review and case report. Am J Otolaryngol. 2015;36:87–89. doi: 10.1016/j.amjoto.2014.08.013. [DOI] [PubMed] [Google Scholar]
- 19.Yeomans D, Qureishi A, Shah K, Winter S. Nodular fasciitis of soft tissue over the mandible. BMJ Case Rep. 2017:2017. doi: 10.1136/bcr-2017-220611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kim ST, Kim HJ, Park SW, Baek CH, Byun HS, Kim YM. Nodular fasciitis in the head and neck: CT and MR imaging findings. AJNR Am J Neuroradiol. 2005;26:2617–2623. [PMC free article] [PubMed] [Google Scholar]
- 21.Leung LY, Shu SJ, Chan AC, Chan MK, Chan CH. Nodular fasciitis: MRI appearance and literature review. Skeletal Radiol. 2002;31:9–13. doi: 10.1007/s002560100411. [DOI] [PubMed] [Google Scholar]
- 22.Yamamoto S, Chishima T, Adachi S. Nodular fasciitis of the breast mimicking breast cancer. Case Rep Surg. 2014;2014:747951. doi: 10.1155/2014/747951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Brown V, Carty NJ. A case of nodular fascitis of the breast and review of the literature. Breast. 2005;14:384–387. doi: 10.1016/j.breast.2004.09.014. [DOI] [PubMed] [Google Scholar]
- 24.Shiga M, Okamoto K, Matsumoto M, Maeda H, Dabanaka K, Namikawa T, Uemura S, Munekage M, Kobayashi M, Hanazaki K. Nodular fasciitis in the mesentery, a differential diagnosis of peritoneal carcinomatosis. World J Gastroenterol. 2014;20:1361–1364. doi: 10.3748/wjg.v20.i5.1361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.O’Connell JX, Young RH, Nielsen GP, Rosenberg AE, Bainbridge TC, Clement PB. Nodular fasciitis of the vulva: a study of six cases and literature review. Int J Gynecol Pathol. 1997;16:117–123. doi: 10.1097/00004347-199704000-00006. [DOI] [PubMed] [Google Scholar]
- 26.Gaffney EF, Majmudar B, Bryan JA. Nodular fasciitis (pseudosarcomatous fasciitis) of the vulva. Int J Gynecol Pathol. 1982;1:307–312. doi: 10.1097/00004347-198203000-00008. [DOI] [PubMed] [Google Scholar]
- 27.Ignacioaranda FI, Laforga JB. Nodular fasditis of the vulva. Report of a case with lmmunohistochemical study. Pathol Res Pract. 1998;194:805–807. doi: 10.1016/s0344-0338(98)80071-x. [DOI] [PubMed] [Google Scholar]
- 28.Hayashi S, Yasuda S, Takahashi N, Okazaki S, Ishibashi K, Kitada M, Miyokawa N. Nodular fasciitis of the breast clinically resembling breast cancer in an elderly woman: a case report. J Med Case Rep. 2017;11:57. doi: 10.1186/s13256-017-1219-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Biedrzycki OJ, Singh N, Faruqi A. Nodular fasciitis of the vulva with an unusually long clinical history; the importance of making this unexpected diagnosis in such an unusual site. Histopathology. 2007;51:547–550. doi: 10.1111/j.1365-2559.2007.02769.x. [DOI] [PubMed] [Google Scholar]
- 30.Paliogiannis P, Cossu A, Palmieri G, Scognamillo F, Pala C, Nonnis R, Sotgiu G, Fois A, Palomba G, Attene F. Breast nodular fasciitis: a comprehensive review. Breast Care (Basel) 2016;11:270–274. doi: 10.1159/000448185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pandian TK, Zeidan MM, Ibrahim KA, Moir CR, Ishitani MB, Zarroug AE. Nodular fasciitis in the pediatric population: a single center experience. J Pediatr Surg. 2013;48:1486–1489. doi: 10.1016/j.jpedsurg.2012.12.041. [DOI] [PubMed] [Google Scholar]
- 32.Brooks JK, Scheper MA, Kramer RE, Papadimitriou JC, Sauk JJ, Nikitakis NG. Intraoral proliferative myositis: case report and literature review. Head Neck. 2007;29:416–420. doi: 10.1002/hed.20530. [DOI] [PubMed] [Google Scholar]
- 33.Orlowski W, Freedman PD, Lumerman H. Proliferative myositis of the masseter muscle. A case report and a review of the literature. Cancer. 2015;52:904–908. doi: 10.1002/1097-0142(19830901)52:5<904::aid-cncr2820520527>3.0.co;2-h. [DOI] [PubMed] [Google Scholar]
- 34.Kent MS, Flieder DB, Port JL, Altorki NK. Proliferative myositis: a rare pseudosarcoma of the chest wall. Ann Thorac Surg. 2002;73:1296–1298. doi: 10.1016/s0003-4975(01)03266-0. [DOI] [PubMed] [Google Scholar]
- 35.Meyer CA, Kransdorf MJ, Jelinek JS, Moser RP Jr. MR and CT appearance of nodular fasciitis. J Comput Assist Tomogr. 1991;15:276–279. doi: 10.1097/00004728-199103000-00017. [DOI] [PubMed] [Google Scholar]
- 36.Koenigsberg RA, Faro S, Chen X, Marlowe F. Nodular fasciitis as a vascular neck mass. AJNR Am J Neuroradiol. 1996;17:567–569. [PMC free article] [PubMed] [Google Scholar]
- 37.Spagnolli G, Pasquali MP, Rosati M, Lisanti M. Nodular fasciitis of the hallux: difficulties in diagnosis. Acta Orthop Belg. 1997;63:46–49. [PubMed] [Google Scholar]
- 38.Klapsinou E, Despoina P, Dimitra D. Cytologic findings and potential pitfalls in proliferative myositis and myositis ossificans diagnosed by fine needle aspiration cytology: report of four cases and review of the literature. Diagn Cytopathol. 2012;40:239–244. doi: 10.1002/dc.21549. [DOI] [PubMed] [Google Scholar]
- 39.Abdul-Karim FW, Rader AE. Fine needle aspiration of soft-tissue lesions. Clin Lab Med. 1998;18:507–540. vi. [PubMed] [Google Scholar]
- 40.Stanley MW, Skoog L, Tani EM, Horwitz CA. Nodular fasciitis: spontaneous resolution following diagnosis by fine-needle aspiration. Diagn Cytopathol. 2010;9:322–324. doi: 10.1002/dc.2840090316. [DOI] [PubMed] [Google Scholar]
- 41.el-Jabbour JN, Bennett MH, Burke MM, Lessells A, O’Halloran A. Proliferative myositis. An immunohistochemical and ultrastructural study. Am J Surg Pathol. 1991;15:654–659. doi: 10.1097/00000478-199107000-00006. [DOI] [PubMed] [Google Scholar]
- 42.Pontes FS, de Oliveira JI, de Souza LL, de Almeida OP, Fregnani ER, Vilela RS, Silva WM, Fonseca FP, Pontes HA. Linicopathological analysis of head and neck rhabdomyosarcoma: a series of 10 cases and literature review head and neck rhabdomyosarcoma. Med Oral Patol Oral Cir Bucal. 2018;23:e188–e197. doi: 10.4317/medoral.22106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Machado I, Mayordomo-Aranda E, Giner F, Llombart-Bosch A. The role of immunohistochemistry in rhabdomyosarcoma diagnosis using tissue microarray technology and a xenograft model. Fetal Pediatr Pathol. 2015;34:271–281. doi: 10.3109/15513815.2015.1042604. [DOI] [PubMed] [Google Scholar]
- 44.Carroll SJ, Nodit L. Spindle cell rhabdomyosarcoma: a brief diagnostic review and differential diagnosis. Arch Pathol Lab Med. 2013;137:1155–1158. doi: 10.5858/arpa.2012-0465-RS. [DOI] [PubMed] [Google Scholar]
- 45.Morgen EK, Carter P, Weinreb I, Al-Habeeb A, Ghazarian DM. Immunohistochemistry in nodular fasciitis of the head and neck. Pathology. 2013;45:432–433. doi: 10.1097/PAT.0b013e328360f06a. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


