Abstract
Purpose:
To compare the efficacy of subthreshold micropulse yellow laser (SMYL) and intravitreal aflibercept injection (IAI) combination therapy with IAI monotherapy in the treatment of diabetic macular edema (DME) and to evaluate the number of injections and SMYL sessions required.
Methods:
This prospective study compared a group of 28 patients treated with a combination of SMYL and IAI with a group of 28 patients treated only with IAI. All patients initially received 3 monthly IAIs, and the monotherapy group was given additional injections as needed. The combination therapy patients additionally received SMYL after the loading phase. The primary outcome measures were the change in the best-corrected visual acuity (BCVA) and central macular thickness (CMT) from baseline to month 12; the secondary outcomes were the mean number of required injections and SMYL sessions.
Results:
In the monotherapy group, the BCVA improved from 0.38 ± 0.10 to 0.20 ± 0.10 logMAR; in the combination group, BCVA improved from 0.40 ± 0.09 to 0.17 ± 0.06 logMAR at the end of the 12th month. The CMT was reduced from 451.28 ± 44.85 to 328.8 ± 49.69 μm in the monotherapy group and from 466.07 ± 71.79 to 312.0 ± 39.29 μm in the combination group. Improvement of the mean BCVA and reduction of the mean CMT were similar in each group. The combination group required significantly fewer injections (3.21 ± 0.41 vs 5.39 ± 1.54; P < 0.001). By month 12, 75% of patients in the monotherapy group had required additional IAIs when compared with 16% in the combination group (P < 0.001).
Conclusion:
SMYL combination therapy demonstrated significant visual improvements in patients with DME. In the combination group, the retreatment rate and number of required injections were significantly lower compared with the IAI monotherapy group.
Keywords: Anti-VEGF injection, diabetic macular edema, subthreshold micropulse yellow laser
Diabetic retinopathy and its associated conditions, such as diabetic macular edema (DME), are serious diabetes mellitus (DM) complications. They are the leading causes of blindness and visual disability in working-age adults. Approximately 50% of persons with type 2 DM may develop retinopathy, while 25% of people with DM go on to exhibit DME.[1]
Although intravitreal pharmacotherapies have significantly improved the treatment of DME over the past decade, retinal nondamaging laser technologies have begun to change the management of DME. Anti-vascular endothelial growth factor (VEGF) monotherapy is the standard of care for patients with DME in high-resource settings due to its rapid and prominent effects on vision improvement; these effects have been supported in several multicentre, randomized trials.[2,3] Some trials have also documented that frequent intravitreal injections, such as 7–12 in the first year and slightly fewer in subsequent years, are needed to achieve and maintain visual improvement in these patients.[4,5]
Despite intensive anti-VEGF therapy, there is still a significant role for conventional macular thermal laser (CMTL) therapy in most studies. CMTL was applied in more than 30% of eyes with center-involving DME, receiving ranibizumab (RNB) every 4 weeks in the 24-month RISE and RIDE phase 3 trial.[6] It is known that CMLT causes visible laser scars that can lead to several complications over the long-term, such as subretinal fibrosis, choroidal neovascularization, and vision field sensitivity deterioration.[7] However, advances in laser technology have led to the development of selective treatment without chorioretinal damage. Friberg and Karatza first reported the clinical application of 810-nm diode micropulse laser therapy for DME.[8] Several clinical studies have since demonstrated the efficacy of this method using different wavelength subthreshold laser treatments in DME.[9,10] However, there are several treatment strategies concerning prompt versus deferred macular laser treatment, some of which can be combined with anti-VEGF treatment.[6,11]
As shown in DRCR.net's Protocol T, when the initial best-corrected visual acuity (BCVA) score was worse than 20/50, improvement of the BCVA in the intravitreal aflibercept injection (IAI) group was significantly higher than that seen in the ranibizumab and bevacizumab injection groups. Because of this result, we preferred aflibercept as an anti-VEGF agent for this study.[12] The 577-nm yellow laser light provides maximum absorption by both oxyhaemoglobin and melanin.[13] This leads to energy being concentrated in a smaller volume, which in turn allows for a reduction in power and a shortened pulse duration. It also has negligible xanthophyll absorption, which allows more secure treatment close to the fovea. In this study, we compared the efficacy of a combination therapy regimen [three IAIs followed by subthreshold micropulse yellow laser (SMYL)] compared with IAI monotherapy, as well as the retreatment rate, overall injection burden, and safety profile).
Methods
This study was a 12-month, prospective comparison of 56 patients with center-involving DME conducted at the Departments of Ophthalmology, Fatih Sultan Mehmet Training and Research Hospital and Lutfi Kirdar Training and Research Hospital. The patients either randomly received a combination treatment consisting of IAI plus SMYL or IAI monotherapy as two coexisting standard treatments [Fig. 1]. All patients were native, and they had no medical history for treatment of DME. Consecutive patients with DME were randomly allocated to one of the two groups by a parallel assignment (1:1). The allocation codes were generated by a web-based module and were stratified according to the clinical center with a permuted block method with randomly chosen block sizes.
Figure 1.
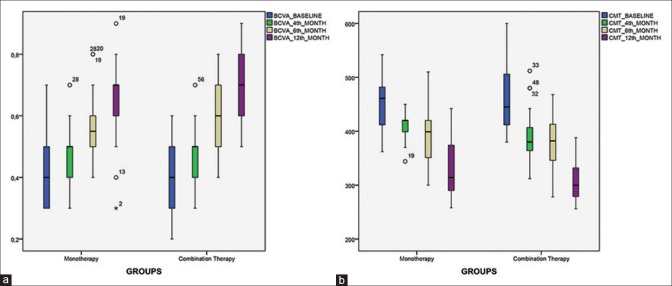
(a and b) The median, interquartile range, and variability in the data across time points
The study was conducted in accordance with the Declaration of Helsinki. Approval was obtained from the Institutional Review Board, and written informed consent was provided by each patient.
Patients affected by DME who were observed between April 2015 and November 2017 were prospectively enrolled. The inclusion criteria for all participants were as follows: (1) a minimum age of 18 years and a prior diagnosis of type 2 DM with central (foveal) involving DME; (2) BCVA ranging from 0.9 to 0.2 (Snellen); (3) central macular thickness (CMT) of at least 300 μm (with foveal involvement), measured by spectral domain optical coherence tomography (SD-OCT) (Nikon RS-3000, Japan); (4) no ischemic maculopathy seen in fluorescein angiography (FA); (5) no proliferative diabetic retinopathy or macular edema due to other underlying retinal vascular disease, vitreomacular traction, or epiretinal membranes. Subfoveal choroidal thickness (SFCT), duration time of DM (time of the diagnosis of DM), and hemoglobin A1C (HbA1c) levels were noted for all patients.
All participants underwent a baseline examination including BCVA, slit-lamp examination, dilated fundoscopy, SD-OCT, and FA. We evaluated the BCVA and SD-OCT parameters at every monthly visit. BCVA was assessed with Snellen charts and converted to logarithm of the minimum angle of resolution (logMAR) units for statistical analysis. The SFCT measurements were performed using enhanced depth imaging mode. After eligibility was confirmed, all participants then received 2-mg IAIs at 1-month intervals as loading-phase therapy.
SMYL combination therapy group
SMYL therapy was performed 1 month after the three loading-phase injections if CMT had decreased to 450 μm or below. Otherwise, eyes that did not reach less than 450 μm thickness had one or more IAI and SMYL applied 4 weeks after. All treatments were provided with the 577-nm yellow laser system (Supra 577Y Subliminal Laser System, Quantel Medical, Clermont-Ferrand, France) using an Area-Centralis lens (Volk Optical, Mentor, OH, USA), and the micropulse laser power used by the SMYL was derived for each eye based on a test burn. The test burn was performed with a 577-nm yellow laser in micropulse mode using a 160-μm spot diameter with a 5% duty cycle and a 20-ms duration outside the vascular arcade, with the power titrated from 50 mW upward until a burn became slightly visible. The SMYL was then performed contiguously on the macular area using the same spot size by reducing the laser to half the power of the test burn. SMYL was applied for each treatment to easily deliver over the area of increased retinal thickness according to SD-OCT. The number of spots varied according to the extension of DME. After the first SMYL application, the patients were followed according to the pro re nata (PRN) protocol. Patients who have increase of at least 20% CMT values and/or 1 line decreased at BCVA after the SMYL treatment, the laser procedure was repeated 2 months after any SMYL session. SMYL therapy was paused if the patients demonstrated stable BCVA and CMT on three consecutive visits or if the BCVA reached 1.0 line or the DME was resolved (CMT <300 μm). All SMYL applications were performed by two ophthalmologists (AA and HSK).
IAI monotherapy group
The patients were initially treated with three IAIs at 1-month intervals. After the “loading phase,” injections were delivered PRN. According to the PRN protocol, injection therapy was paused after the loading phase for patients who demonstrated the same characteristics as those in the combination therapy group. IAIs were resumed if there was a reduction of more than one line in the BCVA compared with the baseline BCVA or if a CMT increase of at least 20% was observed. Retreatment was continued until the BCVA and CMT were again stable for at least three consecutive visits.
Statistical analyses
All analyses were performed using SPSS software version 22 (Chicago, IL, USA). In evaluations of BCVA and CMT, the changes from baseline and differences between groups during the study period were analyzed using two-way repeated-measures analysis of variance and post hoc Dunnett's correction. Paired t-tests or Mann–Whitney U-test was used for continuous variables. For categorical analysis, Chi-square or Fisher's exact tests were adopted. P value <0.05 was judged as statistically significant.
Results
In total, 56 patients were included in this prospective comparison study; 28 patients received IAI monotherapy, whereas the other 28 received SMYL combination therapy. The demographic data and baseline characteristics for the study patients are shown in Table 1. Among all subjects, diabetic retinopathy was classified as mild or moderate nonproliferative retinopathy in 40 out of 56 eyes (71.42%) and as severe nonproliferative retinopathy in 16 out of 56 eyes (28.58%). No significant differences between groups were found for the mean age, gender, preoperative HbA1c level, or diabetic retinopathy severity at baseline.
Table 1.
Baseline characteristics of the study groups
| IAI monotheraphy | IAI + SMYL combination | P | |
|---|---|---|---|
| Mean age±SD (years) | 62.64±9.03 | 63.42±10.14 | 0.26 |
| Gender | |||
| Female | 43% | 46% | 0.94 |
| Male | 57% | 54% | 0.85 |
| Mean DM duration time±SD (years) | 18.28±2.24 | 18.76±2.08 | 0.67 |
| HbA1c level | 8.02±2.43 | 7.97±2.47 | 0.66 |
IAI=Intravitreal aflibercept injection; SMYL=Subthreshold micropulse yellow laser; SD=Standard deviation; DM=Diabetes mellitus
Anatomic and functional changes
The mean baseline CMT values were 451.28 ± 44.85 and 466.07 ± 71.79 μm for the monotherapy and combination groups, respectively (P = 0.35). The mean baseline BCVA values were 0.41 ± 0.11 and 0.39 ± 0.09 respectively (P = 0.45).
One month after the loading phase, the BCVA had improved to 0.48 ± 0.08 in the monotherapy group, while it was 0.46 ± 0.09 in the combination group. Both groups reached commensurate and significant BCVA improvements (P = 0.47). The mean CMT decreased to 411.21 ± 24.46 μm in the monotherapy group and 408.34 ± 20.56 μm in the combination group (P = 0.77). Three patients required one more injection after the three loading injections in the combination group.
At the sixth-month visits, the BCVA improvements were sustained; the BCVA was 0.56 ± 0.12 in the monotherapy group and 0.58 ± 0.11 in the combination group. These differences were not statistically significant (P = 0.41). By month 12, the BCVA in the monotherapy group had improved to 0.64 ± 0.13, and in the combination group it improved to 0.68 ± 0.10. While there was a trend toward a better mean BCVA outcome in the combination therapy group, this difference did not reach statistical significance (P = 0.22).
The changes in the CMT were similar to the noted BCVA improvements. The mean CMT was reduced to 387.92 ± 47.71 μm in the monotherapy group and 377.3 ± 45.61 μm in the combination group at the sixth month (P = 0.4). By month 12, the mean CMT was 328.8 ± 49.69 μm in the monotherapy group and 312.0 ± 39.29 μm in the combination group, and the differences between the two groups were not statistically significant (P = 0.16). Fig. 1a and b shows the median, interquartile range, and variability in the data across time points.
The mean baseline SFCT was 256.7 ± 63.59 μm in the monotherapy group and 262.75 ± 61.17 μm in the combination group (P = 0.55). No significant change in the SFCT was observed in either group at 1 month after loading phase, 6, and 12 months of follow-up. Table 2 shows the BCVA, CMT, and SFCT changes for both groups from the initial assessment to the end of the 12th month.
Table 2.
BCVA, CMT, and SFCT changes for both groups from the initial to the end of the 12th month
| Initial | 1 Month after loading | 6 Months | 12 Months | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mono. | Comb. | P | Mono. | Comb. | P | Mono. | Comb. | P | Mono. | Comb. | P | |
| BCVA | 0.41±0.11 | 0.39±0.09 | 0.45 | 0.48±0.08 | 0.46±0.09 | 0.47 | 0.56±0.12 | 0.58±0.11 | 0.41 | 0.64±0.13 | 0.68±0.10 | 0.22 |
| BCVA (LogMar) | 0.38±0.10 | 0.40±0.09 | 0.57 | 0.32±0.08 | 0.33±0.08 | 0.44 | 0.26±0.09 | 0.23±0.10 | 0.4 | 0.20±0.10 | 0.17±0.06 | 0.19 |
| CMT (μm) | 451.28±44.85 | 466.07±71.79 | 0.35 | 411.21±24.46 | 408.34±20.56 | 0.77 | 387.92±47.71 | 377.3±45.61 | 0.4 | 328.8±49.69 | 312.0±39.29 | 0.16 |
| SFCT (μm) | 256.7±63.59 | 262.75±61.17 | 0.75 | 252.8±58.44 | 258.6±52.48 | 0.68 | 250.4±62.78 | 260.8±66.44 | 0.45 | 248.9±64.32 | 264.6±58.8 | 0.24 |
BCVA=Best-corrected visual acuity; CMT=Central macular thickness; SFCT=Subfoveal choroidal thickness
Requirement of retreatment rates
By the end of the 12th month, 21 of 28 patients (75%) in the monotherapy group required additional IAI. The mean number of injections (including three loading-phase injections) was 5.39 ± 1.54. Of 28 eyes treated with SMYL, 13 eyes (46.4%) were treated twice, 4 eyes (14.2%) were treated three times, and 11 (39.6%) eyes were treated once. Only three patients in the combination group required additional IAI during the follow-up period.
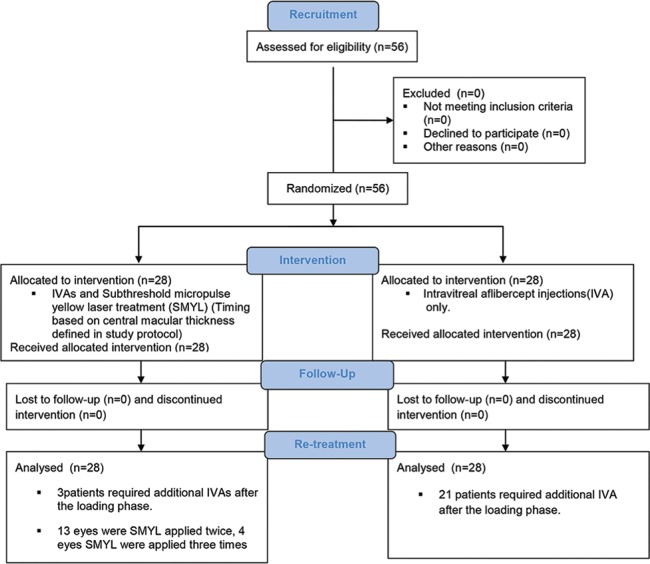
Overall, the subjects received an average of 4.30 ± 1.57 IAIs over 12 months. The mean number of injections required was 3.21 ± 0.41 in the combination group and 5.39 ± 1.54 in the monotherapy group (P < 0.001). At the end of the study, the combination group required significantly fewer IAIs than the monotherapy group. Specifically, 84% of the combination group needed no further IAIs, while 75% of the monotherapy group required additional IAIs (P < 0.001). Fig. 2, which is a CONSORT diagram, shows the workflow used during the study.
Figure 2.
CONSORT diagram: recruitment, intervention, follow-up, and retreatment of the patients who were included in this prospective comparison of combined SMYL treatment and IVA monotherapy
Discussion
The prevalence of DME and visual losses due to DME are expected to increase substantially.[14] Untreated, clinically significant DME can lead to a 3-year, 32% risk of moderate visual loss, resulting in both individual disability and socioeconomic costs.[15] Intravitreal anti-VEGF therapies have evolved substantially over the past decade to become the standard of care in DME.[16] Many studies have pointed out that the number of intravitreal injections required in a year can range from 7 to 12.[11,17,18] This frequently used procedure has disadvantages as well as anatomical and functional advantages.
We compared the efficacy of IAI monotherapy with a combination therapy regimen (three IAIs followed by SMYL). Both groups achieved significant visual gains attributable to the PRN therapy. The final BCVA values in the combination group were trending higher (0.68 ± 0.10) and were noninferior to the monotherapy gains (0.64 ± 0.13). The combination therapy patients had a significantly lower additional IAI rate with 75% of patients needing no further IAI after the loading phase, compared with just 16% in the monotherapy group. Therefore, adding SMYL treatment after IAI loading appeared to reduce the injection burden considerably without compromising anti-VEGF visual gains. We found that the efficacy of treatment modalities as assessed at the 12-month follow-up were not significantly different between groups.
A few similar studies have used SMYL or navigated laser photocoagulation to compare anti-VEGF monotherapy and combination therapy. Liegl et al. compared combination therapy with navigated laser photocoagulation and monotherapy with ranibizumab after a PRN regimen, and they found similar visual gains but a lower retreatment rate and a smaller number of injections in the combination group.[19] Moisseiev et al. revealed that SMYL treatments were safe and effective for DME and reduced the burden of ranibizumab injections, especially in patients with CMT under 400 μm.[20] They suggested their own treatment approach that should ideally be used in the early stages of DME. However, in their study, there were significant differences between the baseline BCVA and CMT in the laser and control groups. Another investigation found that combining an intravitreal dexamethasone implant with SMYL was safe and effective for anti-VEGF-resistant DME; however, that study had no control group for the group with a dexamethasone implant without SMYL treatment, which would be necessary to show an additive effect of the micropulse treatment to the dexamethasone implant.[21] A recent study indicated that SMYL provides a statistically significant improvement in BCVA and a reduction in CMT in mild DME with low central foveal thickness.[22] According to some investigations, SMYL might be an option for patients who do not respond sufficiently to, or who are not able to follow, anti-VEGF therapy due to its high costs or who experience compliance problems because of the frequent visits required for the injections and ophthalmological controls.[23]
There are also some completed large-population and recruited studies that compare the efficacy of a combination of anti-VEGF agents and CMTL therapy for DME. VIVID and VISTA studies demonstrated sustained anatomical and functional benefits with IAIs compared with the CMTL control arm over 2 years.[11] There are few studies combining subthreshold laser stimulation such as ReCall study (ClinicalTrials.gov Identifier: NCT02059772). Unlike CMTL, subthreshold micropulse laser treatment is a nondamaging procedure, and this modality divides a continuous stream of laser into a number of short bursts separated by pauses (off time).[24,25] According to the selected duty cycle, the laser stays on only 5%–15% of the time, thus generating less heat with subsequent less damage to the retina than continuous-wave photocoagulation.[26] The 577 nm SMYL has the advantage that xanthophyll, the pigment located in the inner and outer plexiform layers of the macula, absorbs the yellow light poorly, so treatment near the fovea is relatively safe.[27] SMYL is designed to target the retina pigment epithelium while minimizing the negative thermal effects on the neural retina and deeper structures. One of the recent study supported that SMYL did not result in measurable structural damage to the underlying photoreceptor layer.[28] This modality is termed “photostimulation,” as opposed to “photocoagulation.”[29] Studies conducted to understand cellular responses to subtreshold, sublethal hypertermia showed a significant increase in Heat Shock Protein (HSP) transcription. HSPs are a group of ubiquitous, well-described proteins that are expressed in response to cellular stress.[30] Acting as chaperone proteins, HSPs can assist in the refolding of denaturated proteins and inhibit inappropriate protein aggregation. In addition, HSPs interact with and stabilize the cytoskeleton and help maintain the cellular structure. The HSP70 in particular is known to be expressed in conditions of thermal, ischemic, and oxidative stress. Moreover, HSPs have activity against apoptotic pathways and inflammation.[31] There are concerns that treatment with anti-VEGF agents could potentially increase the risk of serious systemic and ocular adverse events.[32] Endophthalmitis, tractional-rhegmatogenous retinal detachment, ocular inflammation, and thromboembolic side effects sometimes occur after the intravitreal anti-VEGF injections.[33,34] A recent study indicated that patients with diabetes may be at higher risk of adverse events compared with nondiabetic individuals, given frequent systemic comorbities, such as cardiovascular, renal disease, and increased susceptibility to infection.[32] Systemically delivered anti-VEGF drugs are recognized to reduce vascular hyperpermeability, raise systemic arterial blood pressure, and promote tromboembolic events.[35] Furthermore, preclinical findings demonstrated that a decoy VEGF receptor promotes left ventricular dilatation and contractile dysfunction by means of transver aortic constraction.[36] Our combination group required few anti-VEGF injections which was statistically significant. Therefore, during the long-term follow-up of chronic entity, repeateble nondamaging lasers could be an effective and a safe application to reduce possible systemic side effects.
The major limitations of this study were that there was no SMYL treatment group that did not undergo the IAI loading phase, and all SMYL treatment applied after the CMT was below 450 μm. Our personal experience data correlated with the literature's stance that micropulse laser treatment is more significantly effective in patients whose initial CMT is under 450 μm.[37,38,39] Another restriction of our study was that it did not reveal a pure SMYL effect without any anti-VEGF treatment. However, this study did suggest a compelling benefit of adding SMYL to anti-VEGF therapy, and this finding must be confirmed by larger multicenter, randomized controlled trials.
Conclusion
In conclusion, the combination therapy of IAI in a PRN regimen with SMYL was effective in the treatment of DME, and this combination may significantly reduce the number of IAI injections required to sustain the resolution of foveal edema in DME.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- 1.Lee R, Wong TY, Sabanayagam C. Epidemiology of diabetic retinopathy, diabetic macular edema and related vision loss. Eye Vis. 2015;2:17. doi: 10.1186/s40662-015-0026-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Do DV, Nguyen QD, Khwaja AA, Channa R, Sepah YJ, Sophie R, et al. Ranibizumab for edema of the macula in diabetes study: 3-year outcomes and the need for prolonged frequent treatment. JAMA Ophthalmology. 2013;131:139–45. doi: 10.1001/2013.jamaophthalmol.91. [DOI] [PubMed] [Google Scholar]
- 3.Virgili G, Parravano M, Evans JR, Gordon I, Lucenteforte E. Anti-vascular endothelial growth factor for diabetic macular oedema: A network meta-analysis. Cochrane Database Syst Rev. 2018;10:CD007419. doi: 10.1002/14651858.CD007419.pub6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Massin P, Bandello F, Garweg JG, Hansen LL, Harding SP, Larsen M. Safety and efficacy of ranibizumab in diabetic macular edema (RESOLVE Study). A 12-month, randomized, controlled, double-masked, multicenter phase II study. Diabetes Care. 2010;33:2399–405. doi: 10.2337/dc10-0493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nguyen QD, Brown DM, Marcus DM, Boyer DS, Patel S, Feiner L, et al. Ranibizumab for diabetic macular edema: Results from 2 phase III randomized trials. RİSE and RİDE. Ophthalmology. 2012;119:789–801. doi: 10.1016/j.ophtha.2011.12.039. [DOI] [PubMed] [Google Scholar]
- 6.Elman MJ, Ayala A, Bressler NM, Browning D, Flaxel CJ, Glassman AR, et al. Intravitreal ranibizumab for diabetic macular edema with prompt versus deferred laser treatment: 5-year randomized trial results. Ophthalmology. 2015;122:375–81. doi: 10.1016/j.ophtha.2014.08.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ulbig MRW, Arden GB, Hamilton AMP. Color contrast sensitivity and pattern electroretinographic findings after diode and argon laser photocoagulation in diabetic retinopathy. Am J Ophthalmol. 1994;117:583–8. doi: 10.1016/s0002-9394(14)70063-7. [DOI] [PubMed] [Google Scholar]
- 8.Friberg TR, Karatza EC. The treatment of macular disease using a micropulsed and continuous wave 810-nm diode laser. Ophthalmology. 1997;104:2030–8. doi: 10.1016/s0161-6420(97)30061-x. [DOI] [PubMed] [Google Scholar]
- 9.Kwon YH, Lee DK, Kwon OW. The short-term efficacy of subthreshold micropulse yellow (577-nm) laser photocoagulation for diabetic macular edema. Korean J Ophthalmol. 2014;28:379–85. doi: 10.3341/kjo.2014.28.5.379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Luttrull JK, Sinclair SH. Safety of transfoveal subthreshold diode micropulse laser for fovea-involving diabetic macular edema in eyes with good vısual acuıty. Retina. 2014;34:2010–20. doi: 10.1097/IAE.0000000000000177. [DOI] [PubMed] [Google Scholar]
- 11.Brown DM, Schmidt-Erfurth U, Do DV, Holz FG, Boyer DS, Midena E, et al. Intravitreal aflibercept for diabetic macular edema. Ophthalmology. 2015;122:2044–52. doi: 10.1016/j.ophtha.2015.06.017. [DOI] [PubMed] [Google Scholar]
- 12.Cai S, Bressler NM. Aflibercept, bevacizumab or ranibizumab for diabetic macular oedema: Recent clinically relevant findings from DRCR.net Protocol T. Curr Opin Ophthalmol. 2017;28:636–43. doi: 10.1097/ICU.0000000000000424. [DOI] [PubMed] [Google Scholar]
- 13.Mainster MA. Wavelength selection in macular photocoagulation. Ophthalmology. 1986;93:952–8. doi: 10.1016/s0161-6420(86)33637-6. [DOI] [PubMed] [Google Scholar]
- 14.Wang LZ, Cheung CY, Tapp RJ, Hamzah H, Tan G, Ting D, et al. Availability and variability in guidelines on diabetic retinopathy screening in Asian countries. Br J Ophthalmol. 2017;101:1352–60. doi: 10.1136/bjophthalmol-2016-310002. [DOI] [PubMed] [Google Scholar]
- 15.Wu Y, Ai P, Ai Z, Xu G. Subthreshold diode micropulse laser versus conventional laser photocoagulation monotherapy or combined with anti-VEGF therapy for diabetic macular edema: A Bayesian network meta-analysis. Biomed Pharmacother. 2018;97:293–9. doi: 10.1016/j.biopha.2017.10.078. [DOI] [PubMed] [Google Scholar]
- 16.Ajlan RS, Silva PS, Sun JK. Vascular endothelial growth factor and diabetic retinal disease. Semin Ophthalmol. 2016;31:40–8. doi: 10.3109/08820538.2015.1114833. [DOI] [PubMed] [Google Scholar]
- 17.Ishibashi T, Li X, Koh A, Lai TY, Lee FL, Lee WK, et al. The REVEAL Study. Ranibizumab monotherapy or combined with laser versus laser monotherapy in Asian patients with diabetic macular edema. Ophthalmology. 2015;122:1402–15. doi: 10.1016/j.ophtha.2015.02.006. [DOI] [PubMed] [Google Scholar]
- 18.Patrao NV, Antao S, Egan C, Omar A, Hamilton R, Hykin PG, et al. Real-world outcomes of ranibizumab treatment for diabetic macular edema in a United Kingdom National Health Service Setting. Am J Ophthalmol. 2016;172:51–7. doi: 10.1016/j.ajo.2016.09.002. [DOI] [PubMed] [Google Scholar]
- 19.Liegl R, Langer J, Seidensticker F, Reznicek L, Haritoglou C, Ulbig MW, et al. Comparative evaluation of combined navigated laser photocoagulation and ıntravitreal ranibizumab in the treatment of diabetic macular edema. PLoS ONE. 2014;9:e113981. doi: 10.1371/journal.pone.0113981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Moisseiev E, Abbassi S, Thinda S, Yoon J, Yiu G, Morse LS. Subthreshold micropulse laser reduces anti-VEGF injection burden in patients with diabetic macular edema. Eur J Ophthalmol. 2018;1:68–73. doi: 10.5301/ejo.5001000. [DOI] [PubMed] [Google Scholar]
- 21.Elhamid AHA. Combined ıntravitreal dexamethasone ımplant and micropulse yellow laser for treatment of anti-VEGF resistant diabetic macular edema. Open Ophthalmol J. 2017;11:164–72. doi: 10.2174/1874364101711010164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Citirik M. The impact of central foveal thickness on the efficacy of subthreshold micropulse yellow laser photocoagulation in diabetic macular edema. Lasers Med Sci. 2019;34:907–12. doi: 10.1007/s10103-018-2672-9. [DOI] [PubMed] [Google Scholar]
- 23.Scholz P, Altay L, Fauser S. A review of subthreshold micropulse laser for treatment of macular disorders. Adv Ther. 2017;34:1528–55. doi: 10.1007/s12325-017-0559-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dorin G. Subthreshold and micropulse diode laser photocoagulation. Semin Ophthalmol. 2003;18:147–53. doi: 10.1076/soph.18.3.147.29812. [DOI] [PubMed] [Google Scholar]
- 25.Brader HS, Young LHY. Subthreshold diode micropulse laser: A review. Semin Ophthalmol. 2016;31:30–9. doi: 10.3109/08820538.2015.1114837. [DOI] [PubMed] [Google Scholar]
- 26.Venkatesh P, Ramanjulu R, Azad R, Vohra R, Garg S. Subthreshold micropulse diode laser and double frequency Neodymium: Yag laser in treatment of diabetic macular edema: A prospective, randomized study using multifocal electroretinography. Photomed Laser Surg. 2011;29:727–33. doi: 10.1089/pho.2010.2830. [DOI] [PubMed] [Google Scholar]
- 27.Mainster MA. Decreasing retinal photocoagulation damage: Principles and techniques. Semin Ophthalmol. 1999;14:200–9. doi: 10.3109/08820539909069538. [DOI] [PubMed] [Google Scholar]
- 28.Wells-Gray EM, Doble N, Ohr MP, Choi SS. Structural ıntegrity of ındividual cone photoreceptors after short-wavelength subthreshold micropulse laser therapy for diabetic macular edema. Ophthal Surg Lasers Imag Retina. 2018;49:946–54. doi: 10.3928/23258160-20181203-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Koss MJ, Beger I, Koch FH. Subthreshold diode laser micropulse photocoagulation versus intravitreal injections of bevacizumab in the treatment of central serous chorioretinopathy. Eye. 2012;26:307–14. doi: 10.1038/eye.2011.282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Furukawa A, Koriyama Y. A role of Heat Shock Protein 70 in photoreceptor cell death: potential as a novel therapeutic target in retinal degeneration. CNS Neurosci Ther. 2016;22:7–14. doi: 10.1111/cns.12471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sramek C, Mackanos M, Spitler R, Leung LS, Nomoto H, Contag CH, et al. Non-damaging retinal phototherapy: Dynamic range of heat shock protein expression. Investig Ophthalmol Vis Sci. 2011;52:1780–7. doi: 10.1167/iovs.10-5917. [DOI] [PubMed] [Google Scholar]
- 32.Gupta A, Sun JK, Silva PS. Complications of ıntravitreous ınjections in patients with diabetes. Sem Ophthalmol. 2018;33:42–50. doi: 10.1080/08820538.2017.1353811. [DOI] [PubMed] [Google Scholar]
- 33.Dossarps D, Bron AM, Koehrer P, Aho-Glélé LS, Creuzot-Garcher C FRCR net (FRenCh Retina specialists net) Endophthalmitis after ıntravitreal ınjections: Incidence, presentation, management, and visual outcome. Am J Ophthalmol. 2015;160:17–25. doi: 10.1016/j.ajo.2015.04.013. [DOI] [PubMed] [Google Scholar]
- 34.Jonas JB, Schmidbauer M, Rensch F. Progression of tractional retinal detachment following intravitreal bevacizumab. Acta Ophthalmol. 2009;87:571–2. doi: 10.1111/j.1755-3768.2008.01225.x. [DOI] [PubMed] [Google Scholar]
- 35.Chen ZI, Ai DI. Cardiotoxicity associated with targeted cancer therapies. Mol Clin Oncol. 2016;4:675–81. doi: 10.3892/mco.2016.800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Izumiya Y, Shiojima I, Sato K, Sawyer DB, Colucci WS, Walsh K. Vascular endothelial growth factor blockade promotes the transition from compensatory cardiac hypertrophy to failure in response to pressure overload. Hypertension. 2006;47:887–93. doi: 10.1161/01.HYP.0000215207.54689.31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lavinsky D, Cardillo JA, Melo LA, Jr, Dare A, Farah ME, Belfort R., Jr Randomized clinical trial evaluating mETDRS versus normal or high-density micropulse photocoagulation for diabetic macular edema. Investig Ophthalmol Vis Sci. 2011;52:4314–23. doi: 10.1167/iovs.10-6828. [DOI] [PubMed] [Google Scholar]
- 38.Mansouri A, Sampat KM, Malik KJSteiner JN, Glaser BM. Efficacy of subthreshold micropulse laser in the treatment of diabetic macular edema is influenced by pre-treatment central foveal thickness. Eye. 2014;28:1418–24. doi: 10.1038/eye.2014.264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Vujosevic S, Martini F, Longhin E, Convento E, Cavarzeran F, Midena E. Subthreshold micropulse yellow laser versus subthreshold micropulse infrared laser in center-involving diabetic macular edema: Morphologic and functional safety. Retina. 2015;35:1594–603. doi: 10.1097/IAE.0000000000000521. [DOI] [PubMed] [Google Scholar]