Abstract
The objective of the study was to evaluate the activity of OXA-48 against different broad-spectrum cephalosporins and to identify the reaction products by MALDI-TOF MS. The action of OXA-48 on cefotaxime, ceftazidime, and ceftriaxone was assessed by this method, using an Escherichia coli J53 transconjugant carrying only the ~62 Kb IncL plasmid containing the blaOXA-48 gene, and the same strain without any plasmid was included as a negative control. In addition, a collection of 17 clinical OXA-48-producing Enterobacteriaceae, which were susceptible to broad-spectrum cephalosporins, was evaluated. MALDI-TOF MS-based analysis of the E. coli transconjugant carrying the blaOXA-48-harboring plasmid, and also the clinical isolates, showed degradation of cefotaxime into two inactive compounds—decarboxylated and deacetylated cefotaxime (~370 Da) and deacetyl cefotaxime (~414 Da), both with the hydrolyzed beta-lactam ring. Reaction products were not obtained when the experiment was performed with ceftriaxone or ceftazidime. From a clinical point of view, our study supports the idea that the efficacy of cefotaxime against OXA-48-producing Enterobacteriaceae is doubtful, in contrast to ceftazidime and ceftriaxone which could be valid choices for treating infections caused by these bacteria. However, further clinical studies confirming this hypothesis are required.
Keywords: OXA-48, MALDI-TOF, broad-spectrum cephalosporins
1. Introduction
The oxacillinases comprise a heterogeneous group of class D β-lactamases that are able to hydrolyze amino- and carboxypenicillins and, for some members of the group, also cephalosporins and carbapenems to a greater or lesser extent [1,2]. OXA-48 is a carbapenem-hydrolyzing OXA-type enzyme which was first identified in a carbapenem-resistant Klebsiella pneumoniae recovered from a patient in Turkey in 2004 and then spread worldwide to different species of Enterobacteriaceae [2,3,4]. Currently, OXA-48 is the most frequently detected carbapenemase among carbapenem-resistant-Enterobacteriaceae in many countries of the Middle East, North Africa, and Europe [5,6]. Therapeutic options against infections caused by carbapenemase-producing Enterobacteriaceae are scarce and consequently these bacteria represent a major threat for patient safety worldwide [6]. OXA-48 and its variants, with the exception of OXA-163, hydrolyze carbapenems at low level, but do not confer resistance to broad-spectrum cephalosporins in contrast to other carbapenemases whose activity against these drugs is well known [2,4]. Nonetheless, previous studies showed that the catalytic activity (Kcat/Km) of OXA-48 for cefotaxime is moderate, whereas it is practically null for ceftazidime or cefepime [3,7]. Consequently, from the point of view of clinical efficacy, in vivo experiments in mice evidenced that therapy with ceftazidime was effective against OXA-48-producing Enterobacteriaceae lacking extended-spectrum beta-lactamases (ESBLs) or AmpC-like β-lactamases, whereas the therapy with cefotaxime had little impact in reducing lethality of the rodents [8,9].
Since at least some cephalosporins may be a possible alternative for the treatment of invasive infections caused by OXA-48-producing Enterobacteriaceae, the aim of the present study was to evaluate the activity of OXA-48 against three commonly used broad-spectrum cephalosporins, for which data are still limited (cefotaxime, ceftazidime) or unavailable (ceftriaxone), and to analyze the reaction products, using matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS).
2. Materials and Methods
A previously characterized OXA-48-producing K. pneumoniae strain (Kp-HUCA-4; [10]) carrying the blaOXA-48 gene without any other resistant determinant in the original ~62-Kb IncL/M plasmid (later reclassified as IncL) was chosen for a preliminary experiment. A conjugation assay was performed using Kp-HUCA-48 as donor and the sodium azide-resistant Escherichia coli J53 as the recipient strain. Transconjugants were selected on eosin methylene blue agar containing sodium azide (100 mg/L) plus ertapenem (0.5 mg/L), and plasmid DNA was extracted using the Kado and Liu technique [10]. The activity of OXA-48 against broad-spectrum cephalosporins was assessed by an MALDI-TOF MS assay, using the J53 transconjugant carrying only the ~62 Kb IncL plasmid containing the blaOXA-48 gene. The J53 strain lacking the plasmid was included as a negative control. Isolates were previously grown overnight at 37 °C in trypticase soy agar with 5% sheep blood (Becton Dickinson, Heidelberg, Germany). The activity on cefotaxime (Sigma-Aldrich, Hamburg, Germany; 0.50 mg/mL), ceftazidime (Sigma-Aldrich; 0.25 mg/mL), and ceftriaxone (Sigma-Aldrich; 0.25 mg/mL) was evaluated after resuspension of the bacteria that filled a 1 μL inoculation loop in 50 μL of the antibiotic solution in buffer containing 10 mM NH4HCO3, 10 µg/mL ZnCl2, and 0.001% SDS, as previously described [11]. The suspension was incubated at 37 °C under agitation during 30 min for ceftriaxone, 60 min for cefotaxime, and 3 h for ceftazidime as previously described for the MALDI-TOF MS analysis of ESBL enzymes [11]. The tubes were then centrifuged for 2 min at 13,000× g at room temperature, and 1 μL of the supernatant was applied mixed with 1 μL of α-cyano-4-hydroxycinnamic acid (IVD Matrix HCCA-portioned, Bruker Daltonik, Bruker Daltonics GmbH, Bremen, Germany). Spectra were acquired in an MALDI Microflex LT/SH bench-top mass spectrometer (Bruker Daltonik), equipped with a 60 Hz nitrogen laser in the mass range between 100 and 1000 Da, with a mass peak resolution >300 by employing an optimized acquisition method using FlexControl v.3.4 software (Bruker Daltonics GmbH). MALDI-TOF MS analysis, calibration, and spectra processing were carried out as previously reported [11,12]. The MBT STAR-BL Software module (Bruker Daltonics GmbH) was used to evaluate the spectra. The STAR-BL module was used for automated interpretation of spectra, by calculating the logRQ value which indicates the rate of degradation for the different antibiotics by calculating the logarithmic ratio of the intensity of the mass peaks of the hydrolyzed antibiotic in the intensities of the non-hydrolyzed antibiotic. LogRQ values were normalized according to defined negative and positive control strains. Specifically, E. coli ATCC 25922 was used as negative control and a PCR-confirmed VIM-1-producing E. coli was the positive control. Normalized logRQ values below 0.2 represent negative results, while values above 0.4 indicate beta-lactamase activity. Normalized logRQ values between 0.2 and 0.4 correspond to an ambiguous result [12,13]. Next, the same protocol was applied to a collection of 17 clinical OXA-48-producing K. pneumoniae (8) and E. coli (9) isolates that did not coproduce extended-spectrum beta-lactamases (ESBL), AmpC beta-lactamases, or other carbapenemases, all of them susceptible to cefotaxime (MIC ≤ 1).
3. Results and Discussion
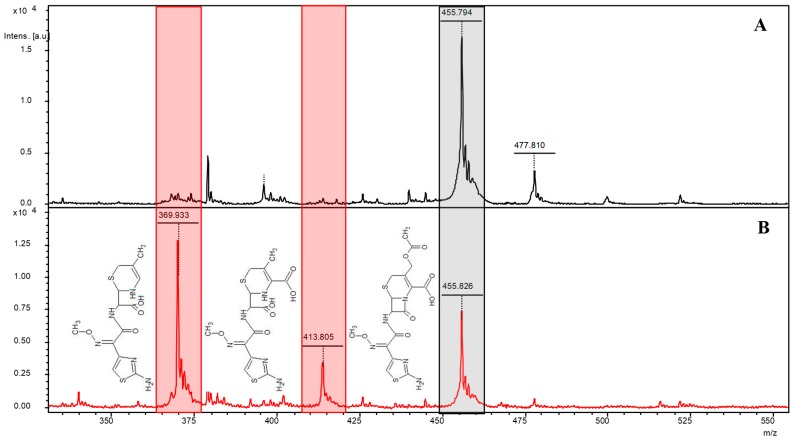
MALDI-TOF MS-based analysis of the transconjugant carrying the blaOXA-48-harboring plasmid showed degradation of cefotaxime into two products of ~414 and ~370 Da. However, these products did not appear in the experiment performed with the same strain lacking the plasmid, used as negative control (Figure 1). Furthermore, no reaction products were obtained when the experiment was performed with ceftriaxone or ceftazidime (not shown). Mass peaks of ~414 and ~370 Da have previously been reported as a consequence of cefotaxime degradation by ESBL-producing Enterobacteriaceae [11,13]. The ~370 Da peak corresponds to a decarboxylated and deacetylated derivative of cefotaxime in the hydrolyzed form, while the ~414 Da peak is deacetyl cefotaxime also hydrolyzed [14].
Figure 1.
Mass spectra of cefotaxime exposed to lysates of Escherichia coli strains producing and not producing OXA-48. (A) Mass spectra of cefotaxime 60 min after exposure to the bacterial lysate of the E. coli J53 control strain. (B) Mass spectra of cefotaxime 60 min after exposure to the bacterial lysate of the E. coli J53 transconjugant carrying the ~62-Kb IncL plasmid with the blaOXA-48 gene. In both images, the black shading corresponds to the mass peak of cefotaxime (~455 Da), whereas the red shading corresponds to the mass peaks of the two degradation products, namely decarboxyl and deacetyl cefotaxime (~370 Da) and deacetyl cefotaxime (~414 Da), both with the hydrolyzed beta-lactam ring. The chemical structure of the compounds is shown next to each peak.
Both compounds are microbiologically inactive. However, the fact that hydrolysis of cefotaxime is not complete can explain why OXA-48-producing enterobacterial isolates remain phenotypically susceptible to cefotaxime in vitro. Activity on cefotaxime can also explain the eventual positive results obtained by colorimetric assays based on pH reduction, aimed at ESBL detection [15,16], which could be due to carboxyl-acid formation after hydrolysis of the beta-lactam ring. From a clinical point of view, the efficacy of cefotaxime against OXA-48-producing Enterobacteriaceae could be impaired by the aforementioned hydrolysis and this is consistent with the previously reported lower efficacy of this drug in vivo with respect to ceftazidime or cefepime [8,9].
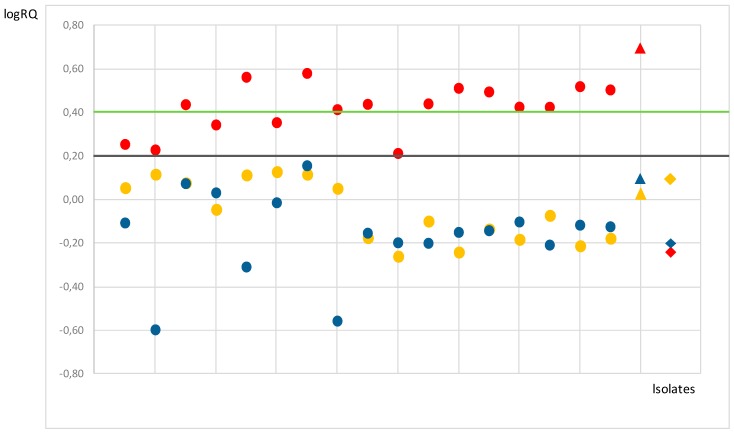
When the MALDI-TOF MS-based experiment was carried out with 17 OXA-48-producing K. pneumoniae and E. coli clinical isolates, similar results were observed. As shown in Figure 2, the logRQ cut-off value established by the STAR-BL module demonstrated a clear degradation of cefotaxime (logRQ > 0.4) by the transconjugant carrying the blaOXA-48-harboring plasmid and most of the OXA-48-producing clinical strains (12/17). The remaining isolates (5/17; 2 of K. pneumoniae and 3 of E. coli) showed ambiguous patterns (logRQ between 2 and 4) for cefotaxime. Consistent with the aforementioned results, activity against ceftazidime and ceftriaxone (logRQ < 2) was not evidenced for any of the tested strains (not shown).
Figure 2.
Comparison of normalized cefotaxime (red), ceftriaxone (yellow), and ceftazidime (blue) logRQ values for 17 OXA-48 producers (clinical isolates of Klebsiella pneumoniae and Escherichia coli) represented by dots. LogRQ values above the cut-off are considered positive (0.4, green line) and values below the cut-off (0.2, black line) are considered negative. Intermediate values (between the green and black lines) are ambiguous. The E. coli J53 control strain and the transconjugant of the latter carrying the ~62-Kb IncL plasmid with the blaOXA-48 gene are represented by diamonds and triangles, respectively, using the same color code as in the experiment.
The activity of different ESBLs against broad-spectrum cephalosporins has been previously assessed by MALDI-TOF MS [11,12,13,14]. However, as far as we know, this is the first study in which hydrolysis of these drugs by the OXA-48 carbapenemase has been evaluated by this approach. Moreover, the action of OXA-48 on ceftriaxone, a drug widely used in hospitals to fight different infections, has been here investigated for the first time. According to our results, ceftriaxone, like ceftazidime, could be a valid choice for the treatment of infections caused by OXA-48-producing Enterobacteriaceae that do not coproduce ESBLs or AmpC-like β-lactamases. Although some studies in animal models have been published for ceftazidime and cefepime [8,9], further in vivo studies and clinical experience are required for all broad-spectrum cephalosporins. Unfortunately, cefepime degradation could not be evaluated due to the poor specificity of the MALDI-TOF MS-based assay previously reported for this drug [12].
4. Conclusions
The present study confirmed the degradation of cefotaxime by the OXA-48 enzyme, providing further evidence to rule out the use of this antibiotic for the treatment of OXA-48-producing Enterobacteriaceae. Our results also support the conclusion that ceftazidime and ceftriaxone could be valid choices for treating infections caused by these bacteria, if they do not have other mechanisms conferring resistance to broad-spectrum cephalosporins. The use of these drugs could be of great interest for hospital antimicrobial stewardship initiatives, leading to a reduction in the usage of carbapenems against OXA-48-producing Enterobacteriaceae infections.
Author Contributions
J.F., M.R.R. and G.B. designed the study. M.O. and J.F. defined the methodology. M.O., J.J.H. and R.R. performed the experiments. J.F. wrote the first draft of the manuscript. All the authors reviewed and edited the manuscript and approved the final version.
Funding
This work was supported by project FIS PI17/00728 (Fondo de Investigación Sanitaria, Instituto de Salud Carlos III, Ministerio de Economía y Competitividad, Spain, cofunded by the European Regional Development Fund of the European Union).
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Evans B.A., Amyes S.G. OXA β-Lactamases. Clin. Microbiol. Rev. 2014;27:241–263. doi: 10.1128/CMR.00117-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Patel G., Bonomo R.A. “Stormy waters ahead”: Global emergence of carbapenemases. Front. Microb. 2013;4:48. doi: 10.3389/fmicb.2013.00048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Poirel L., Héritier C., Tölun V., Nordmann P. Emergence of oxacillinase-mediated resistance to imipenem in Klebsiella pneumoniae. Antimicrob. Agents Chemother. 2004;48:15–22. doi: 10.1128/AAC.48.1.15-22.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Poirel L., Potron A., Nordmann P. OXA-48-like carbapenemases: The phantom menace. J. Antimicrob. Chemother. 2012;67:1597–1606. doi: 10.1093/jac/dks121. [DOI] [PubMed] [Google Scholar]
- 5.Van Duin D., Doi Y. The global epidemiology of carbapenemase-producing Enterobacteriaceae. Virulence. 2017;8:460–469. doi: 10.1080/21505594.2016.1222343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Logan L.K., Weinstein R.A. The epidemiology of carbapenem-resistant Enterobacteriaceae: The impact and evolution of a global menace. J. Infect. Dis. 2017;215:S28–S36. doi: 10.1093/infdis/jiw282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Doquier J.D., Calderone V., De Luca F., Benvenuti M., Giuliani F., Bellucci L., Tafi A., Nordmann P., Botta M., Rossolini G.M., et al. Crystal structure of the OXA-48 beta-lactamase reveals mechanistic diversity among class D carbapenemases. Chem. Biol. 2009;16:540–547. doi: 10.1016/j.chembiol.2009.04.010. [DOI] [PubMed] [Google Scholar]
- 8.Mimoz O., Grégoire N., Poirel L., Marliat M., Couet W., Nordmann P. Broad-spectrum β-lactam antibiotics for treating experimental peritonitis in mice due to Klebsiella pneumoniae producing the carbapenemase OXA-48. Antimicrob. Agents Chemother. 2012;56:2759–2760. doi: 10.1128/AAC.06069-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wiskirchen D.E., Nordmann P., Crandon J.L., Nicolau D.P. Efficacy of humanized carbapenem and ceftazidime regimens against Enterobacteriaceae producing OXA-48 carbapenemase in a murine infection model. Antimicrob. Agents Chemother. 2014;58:1678–1683. doi: 10.1128/AAC.01947-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fernández J., Poirel L., Rodicio M.R., Nordmann P. Concomitant and multiclonal dissemination of OXA-48-producing Klebsiella pneumoniae in a Spanish hospital. J. Antimicrob. Chemother. 2016;71:1734–1736. doi: 10.1093/jac/dkv505. [DOI] [PubMed] [Google Scholar]
- 11.Oviaño M., Fernández B., Fernández A., Barba M.J., Mouriño C., Bou G. Rapid detection of Enterobacteriaceae producing extended spectrum beta-lactamases directly from positive blood cultures by matrix-assisted laser desorption ionization-time of flight mass spectrometry. Clin. Microbiol. Infect. 2014;20:1146–1157. doi: 10.1111/1469-0691.12729. [DOI] [PubMed] [Google Scholar]
- 12.Oviaño M., Gómara M., Barba M.J., Revillo M.J., Barbeyto L.P., Bou G. Towards the early detection of β-lactamase-producing Enterobacteriaceae by MALDI-TOF MS analysis. J. Antimicrob. Chemother. 2017;72:2259–2262. doi: 10.1093/jac/dkx127. [DOI] [PubMed] [Google Scholar]
- 13.Oviaño M., Bou G. Matrix-assisted laser desorption ionization-time of flight mass spectrometry for the rapid detection of antimicrobial resistance mechanisms and beyond. Clin. Microbiol. Rev. 2018;28:32. doi: 10.1128/CMR.00037-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sparbier K., Schubert S., Weller U., Boogen C., Kostrzewa M. Matrix-assisted laser desorption ionization-time of flight mass spectrometry-based functional assay for rapid detection of resistance against β-lactam antibiotics. J. Clin. Microbiol. 2012;50:927–937. doi: 10.1128/JCM.05737-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Poirel L., Nordmann P. Comparison of three biochemical tests for rapid detection of extended-spectrum-β-lactamase-producing Enterobacteriaceae. J. Clin. Microbiol. 2016;54:423–427. doi: 10.1128/JCM.01840-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Morosini M.I., García-Castillo M., Tato M., Gijón D., Valverde A., Ruiz-Garbajosa P., Cantón R. Rapid detection of β-lactamase-hydrolyzing extended-spectrum cephalosporins in Enterobacteriaceae by use of the new chromogenic βLacta test. J. Clin. Microbiol. 2014;52:1741–1744. doi: 10.1128/JCM.03614-13. [DOI] [PMC free article] [PubMed] [Google Scholar]