Abstract
A flexible de novo route capable of producing libraries of 2,6-dideoxy sugars is described. We have found that Au(JackiePhos)SbF6MeCN promotes the conversion of homopropargyl orthoesters into functionalized 2,3-dihydro-4H-pyran-4-ones in good to excellent yields (71–90%). These latter compounds can be easily converted into a number of otherwise difficult to access 2,6-dideoxy sugars.
Graphical Abstract

It is estimated that nearly 20% of all natural products possess glycans, many of which contain 2,6-dideoxy sugars. The glycans can play a critical role in modulating natural product bioactivity.1 In extreme cases, deoxy sugars may comprise a significant portion of a natural product, such as in the antibiotic saccharomicin B (Figure 1).2-4 Altering the composition of these sugars through glycorandomization can dramatically affect the bioactivity of a natural product, including increasing potency or mitigating toxicity.5-9 While glycorandomization holds promise as a new tool for drug discovery,10-12 a major hurdle to its implementation is the lack of availability of deoxy sugars.
Figure 1.
Heptadecasaccharide antibiotic saccharomicin B.
Numerous approaches to the synthesis of deoxy sugars have been developed.13-15 In many cases, deoxy sugar monosaccharide construction relies on de novo synthesis.16-38 While there are a myriad of approaches to de novo synthesis, the majority of routes that have been reported are target specific. A more flexible approach relies on carrying out modifications on glycals,39-48 which permits the generation of multiple different sugars from the same starting material. However, this strategy is cost-effective only when the glycal in question can be generated from readily available sugars, which is often not the case. Furthermore, modification of glycals often involves transforming them into the corresponding 2,3-dihydro-4H-pyran-4-ones, a process that requires harsh oxidations that do not always proceed to completion.49-59 Thus, a general approach to deoxy sugar library construction remains elusive.
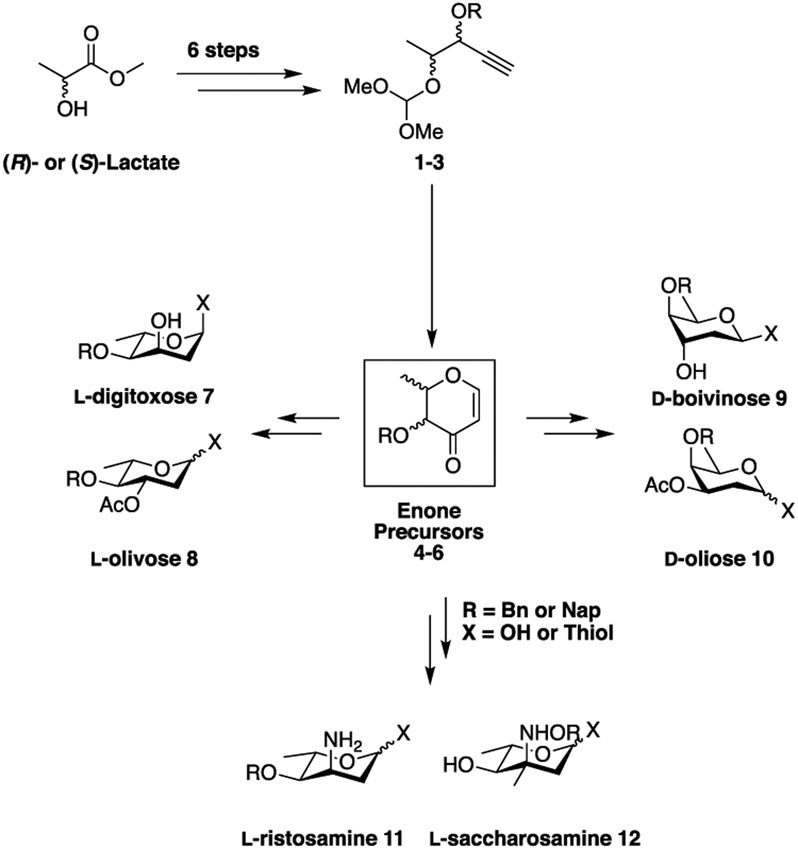
To address this issue, we sought an approach that would allow us to access all four possible diastereomers of suitably functionalized 2,3-dihydro-4H-pyran-4-ones. To this end, we drew inspiration from prior work by the Rhee lab, who demonstrated that homopropargyl acetals could be cyclized into the corresponding tetrahydro-4H-pyran-4-ones using gold(I) catalysts.60,61 We envisioned that applying this cyclization chemistry to homopropargyl orthoesters 1–3 would provide access to all four possible diastereomers of 2,3-dihydro-4H-pyran-4-one [4–6 (Scheme 1)]. These latter compounds could then be converted into an array of 2,6-dideoxy sugars, such as 7–12.
Scheme 1.
Proposed Synthesis and Derivatization of Enone Precursors into Hard To Access Deoxy Sugars
To test the key gold-catalyzed cyclization, 1,2-anti- and 1,2-syn-substituted homopropargyl orthoesters 1–3 were prepared. The synthesis of the 1,2-anti-substituted homopropargyl orthoester (−)-1 began with protecting methyl (R)-(+)-lactate as a tert-butyldimethylsilyl ether (TBS) (Scheme 2),62 followed by reduction using DIBAL-H63 to afford aldehyde (+)-14 in 78% yield over two steps. Alkynylation under conditions described by Marshall afforded (−)-15 in 82% yield as a 16:1 mixture of isomers when (S)-BINOL was used as the chiral ligand.64 After separation of the diastereomers, we were pleased to find that benzylation of the newly formed hydroxyl under standard Williamson ether conditions proceeded smoothly with concomitant deprotection of the alkyne to give (−)-16 in 84% yield. Removal of the TBS group using TBAF afforded alcohol (−)-17 in 93% yield. Finally, treating (−)-17 with trimethyl orthoformate and magnesium chloride provided the key orthoester substrate (−)-1 in 86% yield.65 To test the general applicability of the proposed cyclization, we also synthesized orthoester (+)-2 possessing the more labile naphthylmethyl (Nap) ether.66 Gratifyingly, the synthesis of this substrate proved to be uneventful, and we were able to obtain (+)-2 through a similar sequence using (R)-BINOL to set the stereochemistry of the propargylic alcohol (+)-15. Finally, the 1,2-syn diastereomers (+)-3 and (−)-3 were synthesized, albeit with a lower dr, through a similar sequence (see the Supporting Information).67
Scheme 2.
Synthesis of Orthoesters (−)-1, (+)-1, (+)-2, (−)-3, and (+)-3
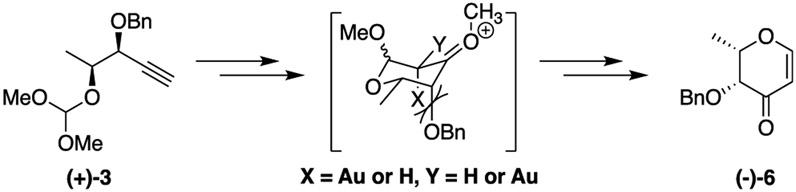
With the requisite orthoesters in hand, we turned our attention to the key gold-catalyzed cyclization reaction. Our initial attempts at cyclization used AuSbF6 with the JohnPhos ligand as described by Rhee and co-workers (Table 1, entry 1).60 Unfortunately, these conditions resulted in decomposi-tion of the orthoester to afford alcohol (−)-17 as the major product. Reasoning that the loss of the orthoester was due to trace amounts of acid, we conducted the reaction in the presence of 2,4,6-tri-tert-butylpyrimidine (TTBP). Adding the acid scavenger afforded desired ketone (+)-4 in a modest 11% yield. Increasing the catalyst load to 5% did not have a major impact on the reaction (Table 1, entry 3). We next examined electron deficient ligands to determine if they would further improve the yield. While tris(pentafluorophenyl)phosphine61 failed to improve the yield of the reaction, the use of the JackiePhos ligand68 led to the formation of the desired pyranone (+)-4 in excellent yield (Table 1, entry 5). Further optimization of the reaction revealed that the catalyst load could be reduced to 3.5% without any deleterious effect on the reaction. Smaller catalyst loads led to a corresponding decrease in the yield.
Table 1.
Optimization of Gold Cyclization
 |
|||||
|---|---|---|---|---|---|
| entry | anion | additive | ligands | catalyst (mol %) |
Yield (%)a |
| 1 | SbF6 | none | JohnPhos, MeCN | 3.0 | 0 |
| 2 | SbF6 | TTBP | JohnPhos, MeCN | 3.0 | 11 |
| 3 | SbF6 | TTBP | JohnPhos, MeCN | 5.0 | 6 |
| 4 | Cl | DTBP, 4 Å MS | (C6F5)3P | 7.0 | 5 |
| 5 | SbF6 | DTBP, 4 Å MS | JackiePhos, MeCN | 7.0 | 86 |
| 6 | SbF6 | DTBP, 4 Å MS | JackiePhos, MeCN | 3.5 | 87 |
Based on isolated weight.
Having established the optimal conditions for cyclization, we turned our attention to examining the scope of the reaction. We found that the 1,2-anti orthoesters (−)-1, (+)-1, and (+)-2 consistently gave excellent yields of the desired ulose (Table 2, entries 1–3, respectively). On the other hand, 1,2-syn orthoesters (−)-3 and (+)-3 reacted to afford the desired products in slightly attenuated yields (Table 2, entries 4 and 5, respectively). The lower yields with the syn isomers may be due to an unfavorable steric interaction between the axial benzyl ether and the substituents on C-2 of the pyranose ring initially formed in the reaction (Figure 2).60,69 Importantly, the gold-catalyzed cyclizations could be run on multigram scale, providing access to significant quantities of all of the 2,3-dihydro-4H-pyran-4-one diastereomers needed for deoxy sugar construction.
Table 2.
Cyclization of Orthoester Substrates into Glycals
| Entry | Substrate | Mol % Cat. |
Product | Yield (%)a |
|---|---|---|---|---|
| 1 |  |
3.5 |  |
87 |
| 2 |  |
3.5 |  |
86 |
| 3 |  |
3.5 |  |
90 |
| 4 |  |
3.5 |  |
71 |
| 5 |  |
3.5 |  |
74 |
Based on isolated weight.
Figure 2.
Possible steric interaction arising during cyclization of syn-homopropargyl orthoesters.
We next turned our attention to transforming the cyclization products into various d- or l-deoxy sugars. Our initial targets were l-digitoxose and l-olivose, which could be readily obtained from (−)-5 (Scheme 3). To this end, treating (−)-5 with cesium carbonate and ethanethiol afforded ketone (−)-22 as a 12.5:1 mixture of isomers.70 Reduction of (−)-22 using sodium borohydride stereoselectively generated l-digitoxose (−)-7 in 80% yield.71 To access l-olivose (−)-8, the enone was selectively reduced under Luche conditions to provide allylic alcohol (−)-23.72 Acetate protection and hydration of the glycal with triphenylphosphine hydrogen bromide afforded l-olivose hemiacetal (−)-8 in 88% yield over two steps.
Scheme 3.
Derivatization of Enone (−)-5 into l-Digitoxose 7 and l-Olivose 8
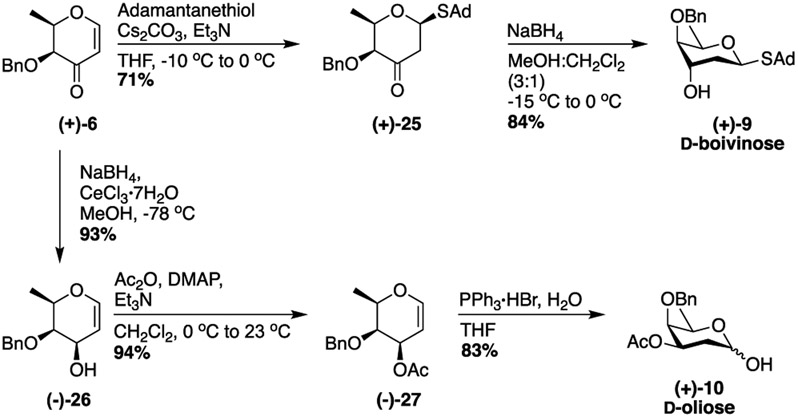
A similar approach was used to obtain d-boivinose (+)-9 and d-oliose (+)-10 (Scheme 4). Treating enone (+)-6 with adamantanethiol, cesium carbonate, and triethyl amine cleanly afforded ketone (+)-25 as a single anomer.70 The ketone was then reduced using sodium borohydride to afford d-boivinose 9.73 To prepare d-oliose (+)-10, enone (+)-6 was subjected to a Luche reduction to stereoselectively generate alcohol (−)-26.45 Acetate protection followed by hydration afforded d-oliose (+)-10 in 78% yield over two steps.
Scheme 4.
Derivatization of Enone (+)-6 into D-Boivinose 9 and d-Oliose 10
Having established scalable routes to the four diastereomeric 2,6-dideoxy sugars, we turned our attention to deoxy amino sugar synthesis. To install amino functionality, 1,4-conjugate addition of thiophenol to enone (−)-5 was followed by addition of O-benzylhydroxylamine to generate oxime (−)-28 (Scheme 5).39,74,75 Refluxing (−)-28 in THF in the presence of 3 equiv of lithium aluminum hydride reduced the oxime to the corresponding amine to afford l-ristosamine (−)-11.76,31
Scheme 5.
Derivatization of Enone (−)-5 into l-Ristosamine 11 and l-Saccharosamine 12
To synthesize l-saccharosamine (−)-12, we first unmasked the C-4 hydroxyl on (−)-5 because the Nap group can interfere with installation of the C-3 methyl group (Scheme 5).39,77 The resultant enone was subjected to a 1,4-conjugate addition/oxime formation sequence to afford (−)-29 in 76% yield over three steps. Treatment of the oxime with methyl cerium under conditions previously described by Scharf afforded l-saccharosamine (−)-12 as a single isomer in 64% yield.39
In summary, we have developed a flexible de novo route for preparing an array of difficult to access 2,6-dideoxy sugars. This process provides access to all four diastereomers of protected 2,3-dihydro-4H-pyran-4-ones starting from inexpensive d- or l-methyl lactate. The resulting pyranones can be readily transformed into a variety of orthogonally protected 2,6-dideoxy- and 2,3,6-trideoxy-3-amino sugars. We anticipate this approach will both greatly accelerate deoxy sugar oligosaccharide synthesis and be an invaluable tool for glycodiversification studies of bioactive natural products.
Supplementary Material
ACKNOWLEDGMENTS
The authors thank the National Science Foundation (CHE-1566233) and the National Institutes of Health (R01-GM115779 and U01-GM120414) for generous financial support.
Footnotes
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.orglett.9b03812.
Experimental details and 1H NMR and 13C NMR spectra (PDF)
The authors declare no competing financial interest.
REFERENCES
- (1).Elshahawi SI; Shaaban KA; Kharel MK; Thorson JS A Comprehensive Review of Glycosylated Bacterial Natural Products. Chem. Soc. Rev 2015, 44, 7591–7697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (2).Kong F; Zhao N; Siegel MM; Janota K; Ashcroft JS; Koehn FE; Borders DB; Carter GT Saccharomicins, Novel Heptadecaglycoside Antibiotics Effective against Multidrug-Resistant Bacteria. J. Am. Chem. Soc 1998, 120, 13301–13311. [Google Scholar]
- (3).Shi SD-H; Hendrickson CL; Marshall AG; Siegel MM; Kong F; Carter GT Structural validation of saccharomicins by high resolution and high mass accuracy fourier transform-ion cyclotron resonance-mass spectrometry and infrared multiphoton dissociation tandem mass spectrometry. J. Am. Soc. Mass Spectrom 1999, 10, 1285–1290. [Google Scholar]
- (4).Singh MPS; Petersen PJ; Weiss WJ; Kong F; Greenstein M Saccharomicins, Novel Heptadecaglycoside Antibiotics Produced by Saccharothrix espanaensis: Antibacterial and Mechanistic Activities. Antimicrob. Agents Chemother 2000, 44, 2154–2159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (5).Williams GJ; Zhang C; Thorson JS Expanding the Promiscuity of a Natural-Product Glycosyltransferase by Directed Evolution. Nat. Chem. Biol 2007, 3, 657–662. [DOI] [PubMed] [Google Scholar]
- (6).Langenhan JM; Peters NR; Guzei IA; Hoffmann FM; Thorson JS Enhancing the anticancer properties of cardiac glycosides by neoglycorandomization. Proc. Natl. Acad. Sci. U. S. A 2005, 102, 12305–12310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (7).Langenhan JM; Griffith BR; Thorson JS No Title. J. Nat. Prod 2005, 68, 1696–1711. [DOI] [PubMed] [Google Scholar]
- (8).Daniel PT; Koert U; Schuppan J Apoptolidine: induction of apoptosis by a natural product. Angew. Chem 2006, 118, 886–908. [DOI] [PubMed] [Google Scholar]
- (9).Iyer AKV; Zhou M; Azad N; Elbaz H; Wang L; Rogalsky DK; Rojanasakul Y; O’Doherty GA; Langenhan JM A Direct Comparison of the Anticancer Activities of Digitoxin MeON-Neoglycosides and O-Glycosides. ACS Med. Chem. Lett 2010, 1, 326–330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (10).Weymouth-Wilson AC The role of carbohydrates in biologically active natural products. Nat.Prod. Rep 1997, 14, 99–110. [DOI] [PubMed] [Google Scholar]
- (11).Rohr J; Thiericke R Angucycline group antibiotics. Nat. Prod. Rep 1992, 9, 103–135. [DOI] [PubMed] [Google Scholar]
- (12).McCranie EK; Bachmann BO Bioactive oligosaccharide natural products. Nat. Prod. Rep 2014, 31, 1026–1042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (13).Bennett CS; Galan MC Methods for 2-Deoxyglycoside Synthesis. Chem. Rev 2018, 118, 7931–7985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (14).Hou D; Lowary TL Recent Advances in the Synthesis of 2-Deoxy-Glycosides. Carbohydr. Res 2009, 344, 1911–1940. [DOI] [PubMed] [Google Scholar]
- (15).Ding F; Cai S; William R; Liu XW Pathways Leading to 3-Amino- and 3-Nitro-2,3-Dideoxy Sugars: Strategies and Synthesis. RSC Adv. 2013, 3, 13594–13621. [Google Scholar]
- (16).Ko SY; Lee AWM; Masamune S; Reed LA; Sharpless KB; Walker FJ Total Synthesis of the L-Hexoses. Science 1983, 220, 949–951. [DOI] [PubMed] [Google Scholar]
- (17).Ko SY; Lee AWM; Masamune S; Reed LA; Barry Sharpless K; Walker FJ Total Synthesis of the L-Hexoses. Tetrahedron 1990, 46, 245–264. [Google Scholar]
- (18).Zhang Y; O’Doherty GA Remote Steric Effect on the Regioselectivity of Sharpless Asymmetric Dihydroxylation. Tetrahedron 2005, 61, 6337–6351. [Google Scholar]
- (19).Ahmed MM; Berry BP; Hunter TJ; Tomcik DJ; O’Doherty GA De Novo Enantioselective Syntheses of Galacto-Sugars and Deoxy Sugars via the Iterative Dihydroxylation of Dienoate. Org. Lett 2005, 7, 745–748. [DOI] [PubMed] [Google Scholar]
- (20).Ahmed MM; O’Doherty GA De Novo Synthesis of Galacto-Sugar δ-Lactones via a Catalytic Osmium/Palladium/Osmium Reaction Sequence. Tetrahedron Lett. 2005, 46, 3015–3019. [Google Scholar]
- (21).Babu RS; Zhou M; O’Doherty GA De Novo Synthesis of Oligosaccharides Using a Palladium-Catalyzed Glycosylation Reaction. J. Am. Chem. Soc 2004, 126, 3428–3429. [DOI] [PubMed] [Google Scholar]
- (22).Babu RS; Chen Q; Kang S-W; Zhou M; O’Doherty GA De Novo Asymmetric Synthesis of All-d-, All-l-, and d-/l-Oligosaccharides Using Atom-Less Protecting Groups. J. Am. Chem. Soc 2012, 134, 11952–11955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (23).Wang H-YL; O’Doherty GA De Novo Synthesis of Deoxy Sugarvia a Wharton Rearrangement. Chem. Commun 2011, 47, 10251–10253. [DOI] [PubMed] [Google Scholar]
- (24).Shan M; Xing Y; O’Doherty GA De Novo Asymmetric Synthesis of an α-6-Deoxyaltropyranoside as Well as Its 2-/3-Deoxy and 2,3-Dideoxy Congeners. J. Org. Chem 2009, 74, 5961–5966. [DOI] [PubMed] [Google Scholar]
- (25).Haukaas MH; O’Doherty GA Enantioselective Synthesis of 2-Deoxy- and 2,3-Dideoxyhexoses. Org. Lett 2002, 4, 1771–1774. [DOI] [PubMed] [Google Scholar]
- (26).Haukaas MH; O’Doherty GA Synthesis of D- and l-Deoxymannojirimycin via an Asymmetric Aminohydroxylation of Vinylfuran. Org. Lett 2001, 3, 401–404. [DOI] [PubMed] [Google Scholar]
- (27).Cutchins WW; McDonald FE Stereoselective Synthesis of Vancosamine and Saccharosamine Glycals via Tungsten-Catalyzed Alkynol Cycloisomerization. Org. Lett 2002, 4, 749–752. [DOI] [PubMed] [Google Scholar]
- (28).Danishefsky SJ; DeNinno MP Totally Synthetic Routes to the Higher Monosaccharides. Angew. Chem., Int. Ed. Engl 1987, 26, 15–23. [Google Scholar]
- (29).Parker KA; Chang W Regioselectivity of Rhodium Nitrene Insertion. Syntheses of Protected Glycals of l-Daunosamine, d- Saccharosamine, and l-Ristosamine. Org. Lett 2005, 7, 1785–1788. [DOI] [PubMed] [Google Scholar]
- (30).Hiramatsu N; Takahashi N; Noyori R; Mori Y A Stereoselective Route to Multi-Substituted Tetrahydropyrans by Vinyl Radical Cyclization. Tetrahedron 2005, 61, 8589–8597. [Google Scholar]
- (31).Zeng J; Sun G; Yao W; Zhu Y; Wang R; Cai L; Liu K; Zhang Q; Liu X-W; Wan Q 3-Aminodeoxypyranoses in Glycosylation: Diversity-Oriented Synthesis and Assembly in Oligosaccharides. Angew. Chem., Int. Ed 2017, 56, 5227–5231. [DOI] [PubMed] [Google Scholar]
- (32).Danishefsky SJ; Pearson WH; Segmuller BE Total Synthesis of (±)-3-Deoxy-D-Manno-2-Octulopyranosate (KDO). J. Am. Chem. Soc 1985, 107, 1280–1285. [Google Scholar]
- (33).Danishefsky S; Phillips G; Ciufolini M A Fully Synthetic Route to the Papulacandins. Stereospecific Spiroacetalization of a C-1-Arylated Methyl Glycoside. Carbohydr. Res 1987, 171, 317–327. [DOI] [PubMed] [Google Scholar]
- (34).McDonald FE; Wu M Stereoselective Synthesis of L-Oliose Trisaccharide via Iterative Alkynol Cycloisomerization and Acid-Catalyzed Glycosylation. Org. Lett 2002, 4, 3979–3981. [DOI] [PubMed] [Google Scholar]
- (35).McDonald FE; Reddy KS; Díaz Y Stereoselective Glycosylations of a Family of 6-Deoxy-1,2-Glycals Generated by Catalytic Alkynol Cycloisomerization. J. Am. Chem. Soc 2000, 122, 4304–4309. [Google Scholar]
- (36).Ahmed MM; O’Doherty GA De Novo Asymmetric Syntheses of D- and l-Talose via an Iterative Dihydroxylation of Dienoates. J. Org. Chem 2005, 70, 10576–10578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (37).Ahmed MM; O’Doherty GA De Novo Synthesis of a Galacto-Papulacandin Moiety via an Iterative Dihydroxylation Strategy. Tetrahedron Lett. 2005, 46, 4151–4155. [Google Scholar]
- (38).Gao D; O’Doherty GA Enantioselective Synthesis of 10-Epi-Anamarine via an Iterative Dihydroxylation Sequence. Org. Lett 2005, 7, 1069–1072. [DOI] [PubMed] [Google Scholar]
- (39).Greven R; Jütten P; Scharf HD A New Stereoselective Route to Branched-Chain Nitro and Amino Sugars: Synthesis of Both Enantiomers of Decilonitrose and Avidinosamine. J. Org. Chem 1993, 58, 3742–3747. [Google Scholar]
- (40).Pelyvás I; Sztaricskai F; Bognár R Synthetic Aspects of Aminodeoxy Sugars of Antibiotics. Carbohydr. Res 1979, 76, 257–260. [Google Scholar]
- (41).Jung G; Klemer A Reaktionen von Acetalen der L-Rhamnose mit Lithiumorganylen Neue Synthesen von Mycaral, Olivomycal und Methyl-2,3,6-tridesoxy-α-L-erythro-hex-2-enopyranosid. Chem. Ber 1981, 114, 740–745. [Google Scholar]
- (42).Pelyvás I; Hasegawa A; Whistler R L. Synthesis of N-trifluoroacetyl-l-acosamine, N-trifluoroacetyl-l-daunosamine, and their 1-thio analogs. Carbohydr. Res 1986, 146, 193–203. [DOI] [PubMed] [Google Scholar]
- (43).Kimura Y; Matsumoto T; Suzuki M; Terashima S No Title. Bull. Chem. Soc. Jpn 1986, 59, 663–664. [Google Scholar]
- (44).Noecker L; Duarte F; Bolton SA; McMahon WG; Diaz MT; Giuliano RM Glycosylation of Branched Amino and Nitro Sugars. 2. Synthesis of the Cororubicin Trisaccharide. J. Org. Chem 1999, 64, 6275–6282. [Google Scholar]
- (45).Timmons SC; Jakeman DL On the synthesis of the 2,6-dideoxysugar l-digitoxose. Carbohydr. Res 2007, 342, 2695–2704. [DOI] [PubMed] [Google Scholar]
- (46).Tanaka H; Yamaguchi S; Yoshizawa A; Takagi M; Shin-ya K; Takahashi T Combinatorial Synthesis of Deoxyhexasaccharides Related to the Landomycin A Sugar Moiety, Based on an Orthogonal Deprotection Strategy. Chem. - Asian J 2010, 5, 1407–1424. [DOI] [PubMed] [Google Scholar]
- (47).Srinivasarao M; Park T; Chen Y; Fuchs PL Noteworthy observations accompanying synthesis of the apoptolidin disaccharide. Chem. Commun 2011, 47, 5858–5860. [DOI] [PubMed] [Google Scholar]
- (48).Kauffmann T; Klaffke W; Philipp C; Thiem J Stereospezifische Darstellung C-methylverzweigter Kohlenhydrate mit Tetramethylzirconium(IV). Carbohydr. Res 1990, 207, 33–38. [Google Scholar]
- (49).Hayashi M; Yamada K; Nakayama S.-z.; Hayashi H; Yamazaki S Environmentally Benign Oxidation Using a Palladium Catalyst System Green Context. Green Chem. 2000, 2, 257–260. [Google Scholar]
- (50).Czernecki S; Vijayakumaran K; Ville G Convenient Synthesis of Hex-1-Enopyran-3-Uloses: Selective Oxidation of Allylic Alcohols Using Pyridinium Dichromate. J. Org. Chem 1986, 51, 5472–5475. [Google Scholar]
- (51).Weinig H-G; Passacantilli P; Colapietro M; Piancatelli G Glycal-mediated synthesis of enantiomerically pure 5-substituted isoxazoles containing a differentially O-benzylated glycerol moiety. Tetrahedron Lett. 2002, 43, 4613–4615. [Google Scholar]
- (52).Chennaiah A; Verma AK; Vankar YD TEMPO-Catalyzed Oxidation of 3-O-Benzylated/Silylated Glycals to the Corresponding Enones Using a PIFA–Water Reagent System. J. Org. Chem 2018, 83, 10535–10540. [DOI] [PubMed] [Google Scholar]
- (53).Thiem J; Elvers J Synthesen und Reaktionen 3-C-methylverzweigter Glycale der D-Reihe. Darstellungen von Isomeren der endstandigen Disaccharide aus Olivomycin A und Mithramycin. Chem. Ber 1981, 114, 1442–1454. [Google Scholar]
- (54).Thiem J; Elvers J Synthesen und Reaktionen von L-Olivomycal und L-Mycaral. Chem. Ber 1979, 112, 818–822. [Google Scholar]
- (55).Bellosta V; Benhaddou R; Czernecki S Selective Homogeneous Oxidation of Allylic Alcohols with Palladium(II) Salts. Synlett 1993, 1993, 861–863. [Google Scholar]
- (56).Hayashi M; Yamada K; Nakayama S Dehydrogenation of D-glycals by palladium supported on activated charcoal under ethylene atmosphere: synthesis of 1, 5-anhydrohex-1-en-3-uloses. Synthesis 1999, 1999, 1869–1871. [Google Scholar]
- (57).Hill CK; Hartwig JF Site-selective oxidation, amination and epimerization reactions of complex polyols enabled by transfer hydrogenation. Nat. Chem 2017, 9, 1213–1221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (58).Fetizon M; Khac D. Do; Tho ND An approach to the synthesis of optically active trichothecenes from tri-O-acetyl-D-glucal. Tetrahedron Lett. 1986, 27, 1777–1780. [Google Scholar]
- (59).Kirschning A Oxidation of fully protected glycals by hypervalent iodine reagents. J. Org. Chem 1995, 60, 1228–1232. [Google Scholar]
- (60).Bae HJ; Jeong W; Lee JH; Rhee YH Gold(I)-Catalyzed Access to Tetrahydropyran-4-Ones from 4-(Alkoxyalkyl)Oxy-1-Butynes: Formal Catalytic Petasis-Ferrier Rearrangement. Chem. - Eur. J 2011, 17, 1433–1436. [DOI] [PubMed] [Google Scholar]
- (61).Kim J; Jeong W; Rhee YH Flexible Tetrahydropyran Synthesis from Homopropargylic Alcohols Using Sequential Pd-Au Catalysis. Org. Lett 2017, 19, 242–245. [DOI] [PubMed] [Google Scholar]
- (62).Lagisetti C; Yermolina MV; Sharma LK; Palacios G; Prigaro BJ; Webb TR Pre-MRNA Splicing-Modulatory Pharmacophores: The Total Synthesis of Herboxidiene, a Pladienolide-Herboxidiene Hybrid Analog and Related Derivatives. ACS Chem. Biol 2014, 9, 643–648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (63).Ito Y; Kobayashi Y; Kawabata T; Takase M; Terashima S Novel Syntheses of the Carbapenem Key Intermediates, (3R,4R)-4-Acetoxy-3-[(R)-1-(t-Butyldimethylsilyloxy)Ethyl]-2-Azetidinone and (3S,4R)-3-[(R)-1-(t-Butyldimethylsilyloxy)Ethyl]-4-Carboxymethyl-2-Azetidinone, from (S)-Ethyl Lactate. Tetrahedron 1989, 45, 5767–5790. [Google Scholar]
- (64).Marshall JA; Bourbeau MP Synthesis of Enantioenriched Propargylic Alcohols Related to Polyketide Natural Products. A Comparison of Methodologies. Org. Lett 2003, 5, 3197–3199. [DOI] [PubMed] [Google Scholar]
- (65).Perron F; Gahman TC; Albizati KF A Mild and General Synthesis of Mixed Orthoesters. Tetrahedron Lett. 1988, 29, 2023–2026. [Google Scholar]
- (66).Gaunt MJ; Yu J; Spencer JB Rational Design of Benzyl-Type Protecting Groups Allows Sequential Deprotection of Hydroxyl Groups by Catalytic Hydrogenolysis. J. Org. Chem 1998, 63, 4172–4173. [Google Scholar]
- (67).Gao Y; Klunder JM; Hanson RM; Masamune H; Ko SY; Sharpless KB Catalytic Asymmetric Epoxidation and Kinetic Resolution: Modified Procedures Including in Situ Derivatization. J. Am. Chem. Soc 1987, 109, 5765–5780. [Google Scholar]
- (68).Hicks JD; Hyde AM; Cuezva AM; Buchwald SL No Title. J. Am. Chem. Soc 2009, 131, 16720–16734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (69).Smith AB; Hogan A-ML; Liu Z; Razler TM; Meis RM; Morinaka BI; Molinski TF Phorboxazole Synthetic Studies: Design, Synthesis and Biological Evaluation of Phorboxazole A and Hemi-Phorboxazole A Related Analogues. Tetrahedron 2011, 67, 5069–5078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (70).Ganguly D; Tang H; Rodriguez MJ Cesium Carbonate-Promoted Michael-Type Addition of Thiols to Hex-1-En-3-Ulose: A Practical Synthesis of 2-Deoxy-1-Thio-α-Hexopyranosid- 3-Ulose Template. Synth. Commun 2007, 37, 4219–4226. [Google Scholar]
- (71).Soliman SE; Bennett CS Reagent-Controlled Synthesis of the Branched Trisaccharide Fragment of the Antibiotic Saccharomicin B. Org. Lett 2018, 20, 3413–3417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (72).Sridhar PR; Venukumar P A Ring Expansion-Glycosylation Strategy toward the Synthesis of Septano-Oligosaccharides. Org. Lett 2012, 14, 5558–5561. [DOI] [PubMed] [Google Scholar]
- (73).Perry MB; Daoust V A Synthesis of 2-Acetamido-2,6-Dideoxy-D-Gulose, 2-Acetamido-2,6-Dideoxy-D-Idose, and 2,6-Di-deoxy-D-Xylo-Hexose (Boivinose). Can. J. Chem 1973, 51, 3039–3043. [Google Scholar]
- (74).Noecker L; Duarte F; Bolton SA; McMahon WG; Diaz MT; Giuliano RM Glycosylation of Branched Amino and Nitro Sugars. 2. Synthesis of the Cororubicin Trisaccharide. J. Org. Chem 1999, 64, 6275–6282. [Google Scholar]
- (75).Bylsma M; Bennett CS Stereospecific Synthesis of the Saccharosamine-Rhamnose-Fucose Fragment Present in Saccharomicin B. Org. Lett 2018, 20, 4695–4698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (76).Walter CR Preparation of Primary Amines by Reduction of Oximes with Lithium Aluminum Hydride and by the Leuckart Reaction. J. Am. Chem. Soc 1952, 74, 5185–5187. [Google Scholar]
- (77).Kim HM; Kim IJ; Danishefsky SJ Total Syntheses of Tumor-Related Antigens N3: Probing the Feasibility Limits of the Glycal Assembly Method. J. Am. Chem. Soc 2001, 123, 35–48. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.