Abstract
Nasopharyngeal carcinoma (NPC) is a common disease in Southern China with high prevalence. miR-944 has been reported to play a vital role in progression of a variety of cancers. The present study aimed to investigate the potential role of miR-944 in NPC cell migration and invasion through elucidating the interaction with its target genes, MACC1. Expression of miR-944 in NPC tissues and cell lines was examined with quantitative RT-PCR. Overexpressed miR-944 and suppressed miR-944 were established with miR-944 mimics and miR-944 inhibitor, respectively. The effect of miR-944 on cell migration and invasion was determined using Transwell cell migration and Matrigel invasion assay. Luciferase assay was used to determine the target of miR-944. Knocked down of MACC1 was done by MACC1 siRNA. Expression of MET related-markers was examined using Western blot analysis. The expression level of miR-944 was downregulated in NPC tissues and cell lines. Overexpression of miR-944 significantly inhibited the cell migration and invasion in NPC 6-10B cells. In contrast, suppression of miR-944 promoted cell migration and invasion in NPC C-6661 cells. MACC1 is a direct target of miR-944. MACC1 expression was repressed in miR-944 mimic transfected cells while it was enhanced in miR-944 inhibitor transfected cells. MACC1 knock down inhibited cell migration and invasion. Either miR-944 restoration or MACC1 knockdown caused enhanced E-cadherin, reduced N-cadherin, and vimentin expression. In conclusion, miR-944 could inhibit MET and metastasis of NPC by targeting MACC1. This study suggests that miR-944 has anti-tumor and anti- metastatic properties and could thus be a novel therapeutic agent for NPC treatment.
Keywords: Nasopharyngeal carcinoma, miR-944, MACC1, cell migration, invasion
Introduction
Nasopharyngeal carcinoma (NPC) is an uncommon disease in America, but not in southern China [1]. Despite the great attempt made on reducing the NPC-related death toll, the 5-year of survival rate remains only 50-60% especially among the patients with metastasis [2,3]. NPC is a head and neck cancer, but is different from other types of head and neck cancer since NPC cells are likely to metastasize to lymph nodes or invade to surrounding tissues at early stages of tumor development [4]. This is the major cause for poor prognostic outcomes of NSP, because it is hard to detect at an early stage and 75% of patients present with late stage when they diagnosed [5]. Although a numbers of studies have been performed to elucidate the underlying mechanism of invasion and metastasis in NPC, there is still need for development of novel and efficient biomarkers.
Micro RNAs (miRNAs) are small non-coding RNAs that can regulate gene expression via binding to 3’-untranslated region of target mRNAs [6]. In recent years, a regulatory role for miR-944 has been reported in cervical cancer [7], breast cancer [8,9], lung cancer [10,11], gastric cancer [12], colorectal cancer [13], and endometrial cancer [14]. These studies have suggested that miR-944 may play a different role in different conditions of cancer targeting diverse genes. However, to the best of our knowledge, no study has reported a functional role for miR-944 in NPC.
Differentially expressed metastasis-associated in colon cancer 1 (MACC1) is initially identified in primary and metastatic colon cancer by genome-wide search [15]. MACC1 is the main regulator of epithelial-mesenchymal transition (EMT) and by doing so it promotes the proliferation, migration, and invasion of colon cancer cells in metastatic mouse model [15]. In recent years, the clinicopathological significance of MACC1 has been reported in hepatocellular carcinoma [16], breast cancer [17], tongue squamous cell carcinoma [18], glioma [19], gastric adenocarcinoma [20], lung cancer [21], and ovarian carcinoma [22]. In addition, inhibitory effect of suppressed MACC1 on cell proliferation of NPC has previously reported [23]. In present study, we attempt to investigate therapeutic role of miR-944 in NPC and its correlation with MACC1.
Materials and methods
Tissue specimens and cell culture
Total of 20 pairs of tissue samples, including 20 NPC and 20 NPC adjacent tissues, were acquired from the First Affiliated Hospital of Kunming Medical University. Each patient provided written informed consent. No patients subjected the radiotherapy or chemotherapy before biopsy. Clinicopathological characteristics of NPC patients were shown in Table 1. Human NPC cell lines (CNE2, CNE1, HNE1 and C666-1) and a nasopharyngeal epithelium cell, NP69, were provided by ATCC. Cells were maintained in RPMI 1640 medium supplemented with fetal bovine serum (FBS, 10%), penicillin (100 units/mL), and streptomycin (0.1 mg/mL), which were purchased from Sigma-Aldrich (Sigma, St. Luis, MO). All the cells were grown in a humidified atmosphere at 37°C with 5% CO2.
Table 1.
Clinicopathologic characteristics of NPC patient samples (n = 20)
| Characteristics | Number of cases |
|---|---|
| Age (years) | |
| < 50 | 15 |
| ≥ 50 | 5 |
| Gender | |
| Male | 12 |
| Female | 8 |
| TNM staging | |
| I | 12 |
| II | 6 |
| III | 3 |
| Distant metastasis | |
| M0 | 18 |
| M1 | 2 |
NPC: nasopharyngeal carcinoma.
Cell transfection
The miR-944-mimic, miR-944-inhibitor, and their corresponding negative controls were synthesized by RiboBio (Guangzhou, China). MACC1 siRNA and a scrambled control siRNA were designed and synthesized by Biogot Biotechnology (Nanjing, China). Cells were seeded onto six-well plates (2×105 cells/well) and incubated for 24 h. The miR-944-mimic, miR-944-inhibitor, and their corresponding negative controls were transfected to cells in compliance with the manufacturer’s guidance using Lipofactamine 2000 (Thermo Fisher Scientific).
Wound healing assay
After being seeded onto six-well plates (2×105 cells/well), cells were scratched with 100 μL tip. The cell migration was observed under microscope after incubation.
Transwell migration and invasion assay
Cell migration was evaluated using Transwell Inserts (Corning, NY, USA). After transfection, cells were seeded into upper chamber of inserts (8 μm pore size), whereas the lower chamber contained medium (0.5 mL) with 10% FBS and incubated for 36 h. The non-migrated cells in the upper chamber were removed with cotton. After being washed several times with PBS, cells were stained with 0.1% crystal violet. The number of migrated cells were on the bottom of chamber was counted under microscope.
For cell invasion assessment the Matrigel invasion chamber assay (BD bioscience, San Jose, CA, USA) was used. Cells (1×105 cells per Transwell) were placed in upper chamber and incubated for 12 h in serum-free medium. The lower chamber was placed with medium containing 10% FBS and incubated for 24 h. The invaded cells were fixed with methanol and stained with crystal violet. The number of invaded cells was counted under light microscope.
Dual-luciferase reporter assay
The pGL3-MACC1 3’UTR-wild type (WT) plasmid or pGL3-MACC1 3’UTR-mutant (Mut) plasmid (Invitrogen) were co-transfected with control vector pRL-TK (Promega Corporation, Fitchburg, WI, USA), miR-944-mimic, and negative control mimics using Lipofectamine 2000 (Thermo Fisher Scientific). Cells were incubated for 48 h and then harvested. Dual-Luciferase® Reporter 1000 Assay System (Promega Corporation, Fitchburg, WI, USA) was performed to assess Firefly and Renilla luciferase activity. Renilla luciferase activity was used for normalization.
Quantitative RT-PCR analysis
Total RNA was extracted from cells and tissues samples using Trizol reagent (Invitrogen, Carlsbad, CA, USA). Single stranded RNA was reverse transcribed into cDNA using PrimeScript RT Reagent Kit (Takara, Shiga, Japan). Gene amplification was done using SYBR Green Tag II kit (Takara, Shiga, Japan) according the manufacturer’s (Bio-Rad Laboratories, Hercules, CA, USA) instructions. The gene expression level was normalized to β-actin and U6 was used as endogenous control of miR-944. Independent experiment was conducted in triplicate and repeated three times.
Western blot analysis
After total protein was extracted and quantified, 30 μg of protein was separated in 10% of SDS-PAGE gel and transferred into polyvinylidine difluoride membranes (Millipore, Bedford, MA). The membranes were incubated with antibody against MACC1 (1:200, Cell Signaling, Danvers, MA) followed by HRP-labeled secondary antibodies. The intensity was detected by the enhanced chemiluminescence detection system (Amersham Life Science, Piscataway, NJ). The MACC1 protein level was normalized to β-acting level.
Statistical analysis
Data are presented as the mean ± SEM and analyzed by PRISM 5 software (GraphPad Software, Inc., San Diego, CA, USA). Comparison between groups was analyzed using Student’s t test or ANOVA. Correlation analysis was performed by Spearman’s rank correlation test. P < 0.05 was considered statistically significant.
Results
Relative miR-944 expression is decreased in tumor tissues and nasopharyngeal carcinoma cell lines
In comparison of tumor and tumor adjacent tissues based on 20 pairs of cases, relative miR-944 expression was significantly lower than its control tissues (P < 0.05) (Figure 1A). In addition, compared to a control cell line (NP69), relative miR-944 expression level was significantly lower in C666-1, 5-8F, SUNE1, and 6-10B cell lines (P < 0.05) (Figure 1B). Since miR-944 expression level was most suppressed in 6-10B, we selected this cell line for following functional experiments.
Figure 1.

Downregulation of miR-944 in NPC tissues and cell lines. A: Expression of miR-944 in 20 pair of clinical tissue specimens (NPC tissues Kunming 650500, Yunnan Province, China 20, tumor adjacent tissues = 20). B: Expression of miR-944 in human NPC cell lines (CNE2, CNE1, HNE1 and C666-1) and a nasopharyngeal epithelium cell, NP69. ***P < 0.001 vs normal control.
Overexpressed miR-944 inhibits migration and invasion of 6-10B cell lines
In order to explore exact function of miR-944 towards cell migration and invasion, we transfected 6-10B cells with miR-944 mimics and negative control (NC) mimics. Overexpressed miR-944 cells were successfully established (Figure 2A). After incubation for 48 h, the percentage of wound area was significantly higher in miR-944 mimics transfected cells compared to negative control cells (P < 0.001) (Figure 2B). This phenomenon was further confirmed by cell migration and invasion assay. The number of migratory and invasive cells was remarkably lower in cells transfected with miR-944 mimics compared to cells transfected with NC mimics (P < 0.001) (Figure 2C, 2D).
Figure 2.

Effects of miR-944 overexpression on 6-10B cell migration and invasion. (A) Relative expression of miR-944 after mimic transfection. (B) Cell migration assessed by wound healing assay. Cell migration (C) and invasion (D) confirmed by Transwell assay in 6-10B cells. ***P < 0.001 vs negative control.
Inhibition of miR-944 expression promotes migration and invasion of C666-1 cells
Since miR-944 expression was relatively high in C666-1 cell lines (Figure 1B), we selected this cell line to assess the impact of miR-944 loss on cell migration and invasion. The cell model in which miR-944 expression was suppressed was established with miR-944 inhibitor transfection (Figure 3A). Wound healing analysis showed that the percentage of wound area was significantly reduced in cells with miR-944 inhibitor transfection compared to cells transfected with negative control after 48 h of culturing (P < 0.001) (Figure 3B). This result was further confirmed by cell migration and invasion assay. With miR-944 inhibitor transfection, the number of migrated and invaded cells was remarkably increased compared to negative control inhibitor (P < 0.05) (Figure 3C, 3D).
Figure 3.

Effects of miR-944 suppression on C666-1 cell migration and invasion. (A) Relative expression of miR-944 after inhibitor transfection. (B) Cell migration assessed by wound healing assay. Cell migration (C) and invasion (D) confirmed by Transwell assay in C666-1 cells. *P < 0.05 vs negative control, ***P < 0.001 vs negative control.
MACC1 is a direct target of miR-944
The sequence of a miR-944 predicted binding site within the 3’-UTR region of MACC1 mRNA was acquired using TargetScan program (Figure 4A). Luciferase assay was performed to assess whether MACC1 is a direct target of miR-944. The result exhibited that luciferase activity was significantly inhibited in wild type (WT) cells with transfection of miR-944 mimics compared to NC mimics in both 6-10B and C666-1 cell lines (P < 0.001) (Figure 4B, 4C). This phenomenon was not observed in cells with a mutant binding site. In addition, we conducted Western blot analysis to further validate MACC1 protein expression in transfected cells. MACC1 expression was significantly suppressed with miR-944 mimics while MACC1 expression was enhanced with miR-944 inhibitor compared to their negative controls in both 6-10B and C666-1 cell lines (Figure 4D, 4E).
Figure 4.

MACC1 is a direct target of miR-944. (A) miR-944 binding site within 3’-UTR of MACC1 mRNA predicted by TargetScan. Dual luciferase activity in wild type and Mut type of 3’-UTR of MACC1 mRNA in cells transfected with miR-944 mimics (B) and miR-944 inhibitor (C). Relative MACC1 expression in cells transfected with miR-944 mimics (D) and miR-944 inhibitor (E) measured by Western blot analysis.
MACC1 knockdown suppresses cell migration and invasion
To determine the functional activity of MACC1 towards cell migration and invasion, we knocked down MACC1. Successful MACC1 knockdown cells were established in 6-10B cell lines as it shown in Figure 5A. In accordance with cell migration and invasion analysis, the number of migrated and invaded cells was reduced in MACC1 knocked down cells compared to its negative controls (P < 0.001) (Figure 5B, 5C).
Figure 5.

Effects of MACC1 knock down on 6-10B cell migration and invasion. A: Knock down efficiency of siMACC1 measured by Western blot analysis. B: Observation of migratory and invasive cells under light microscope. C: Histogram of number of cells that migrated and invaded after transfected with either siNC or siMACC1. ***P < 0.001 vs negative control.
Suppressed MACC1 and overexpressed miR-944 might own similar function towards cell migration and invasion. Thus, we conducted Western blot analysis to further confirm this result by examining the epithelial-mesenchymal transition (EMT)-related molecules, E-cadherin, N-cadherin, and vimentin. The result show that in cells transfected with miR-944 mimics E-cadherin expression was enhanced while N-cadherin and vimentin expression was suppressed compared to cells transfected with NC mimics (Figure 6A). The same trend of expression pattern was observed in MACC1 knockdown cells (Figure 6B).
Figure 6.
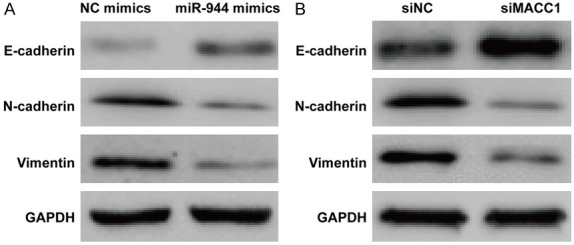
Expression of ETM related markers measured by Western blot. A: Expression of E-cadherin, N-cadherin, and Vimentin in cells transfected with miR-944 mimics. B: Expression of E-cadherin, N-cadherin, and Vimentin in cells in which siMACC1 is knocked down.
Discussion
Abnormal expression of miRNAs has been implicated in regulation of diverse biological activities of NPC [24]. As either tumor suppressors or oncogenes, miRNAs are involved in initiation or progression of tumor by negatively regulating downstream targets [25-27]. A previous study has reported that MACC1 expression is higher in NPC cells and tissues [23]. In present study we used gain- and loss-of-function methods to determine the potential role of miR-944 in pathogenesis of NPC by elucidating its correlation with MACC1. Our results revealed that relative miR-944 expression is significantly lower in NPC cells and tissue specimens than normal controls. Overexpression of miR-944 inhibits cell migration and invasion and inhibition of miR-944 restores the ability of cell migration and invasion. We also found that MACC1 is a direct target of miR-944 and silencing of MACC1 resulted in reduced cell migration and invasion. In addition, our result also exhibited upregulation of E-cadherin, downregulation of N-cadherin and vimentin by overexpressed miR-944 and suppressed MACC1. These results suggest that miR-944 serves as tumor suppressor inhibiting tumor migration and invasion targeting MACC1 in NPC.
Ectopic expression of miR-944 has been reported to play an essential role in cell migration and invasion of diverse cancers. The expression level of miR-944 is notably lower in colorectal cancer patients and overexpression of miR-944 inhibits cell migration and invasion via targeting downstream regulator MACC1, suggesting that miR-944 may play an anti-metastatic role in colorectal cancer [13]. In addition, miR-944 was also reported to be upstream negative regulator of SIAH1 and PTP4A1 and suppression of miR-944 is of metastatic potential in breast cancer [9]. Since loss of miR-944 could promote the EMT of gastric cancer, it has been suggested that miR-944 could be an inhibitor of EMT and metastasis of gastric cancer by targeting MACC1 [12]. Although this literature demonstrated the functional role of miR-944 as tumor suppressor, it may vary depending on the different condition of cancers. It was reported that miR-944 was frequently overexpressed in cervical carcinomas and promoted cells proliferation, migration and invasion through targeting HECW2 and S100PBP. However, the result of present study exhibit that miR-944 is downregulated in NPC patients and overexpression of miR-944 leads to reduced cell migration and invasion.
Expression of MACC1 has been reported to be significantly elevated in metastatic tumor tissues [28]. MACC1 induces cell dissemination, migration and invasion in colorectal cancer cell cultures [28]. Transplantation of MACC1-expressing tumor cells in mice promoted tumor growth and liver metastasis were found [15]. MACC1 knockdown inhibited cell proliferation, migration and invasion of NPC CNE2 cells [23], and ovarian carcinoma OVCAR-3 cells [29]. One of the factors responsible for overexpressed MACC1 in tumors may be its upstream transcriptional regulator, miRNA. Concomitant upregulation of MACC1 and downregulation of miR-1 functionally synergize in promoting MET overexpression, consequently contributing to tumor progression and metastasis [30]. Direct interaction of miR-143 and MACC1 in human colorectal cancer was reported using in silico analysis [31]. They found that combination of miR-143 restoration and MACC1 knockdown resulted in significant inhibition of cell proliferation, migration, and invasion. The results of our work show that MACC1 is a direct target of miR-944. Either miR-944 restoration or MACC1 knockdown led to significant reduction in number of migratory and invasive NPC cells, as well as inhibition of EMT. Therefore, introduction of miR-944 inhibits EMT and tumor metastasis by targeting MACC1 and could be a novel anti-tumor and anti-metastatic agent for NPC.
Acknowledgements
This study was supported by Yunnan Provincial Science and Technology Department- Kunming Medical University United Fund (Grant No. 2015FB077).
Disclosure of conflict of interest
None.
References
- 1.Kamran SC, Riaz N, Lee N. Nasopharyngeal carcinoma. Surg Oncol Clin N Am. 2015;24:547–561. doi: 10.1016/j.soc.2015.03.008. [DOI] [PubMed] [Google Scholar]
- 2.El-Sherbieny E, Rashwan H, Lubis SH, Choi VJ. Prognostic factors in patients with nasopharyngeal carcinoma treated in Hospital Kuala Lumpur. Asian Pac J Cancer Prev. 2011;12:1739–1743. [PubMed] [Google Scholar]
- 3.O’Sullivan B. Nasopharynx cancer: therapeutic value of chemoradiotherapy. Int J Radiat Oncol Biol Phys. 2007;69:S118–121. doi: 10.1016/j.ijrobp.2007.04.085. [DOI] [PubMed] [Google Scholar]
- 4.Farias TP, Dias FL, Lima RA, Kligerman J, de Sa GM, Barbosa MM, Goncalves FB Jr. Prognostic factors and outcome for nasopharyngeal carcinoma. Arch Otolaryngol Head Neck Surg. 2003;129:794–799. doi: 10.1001/archotol.129.7.794. [DOI] [PubMed] [Google Scholar]
- 5.Pua KC, Khoo AS, Yap YY, Subramaniam SK, Ong CA, Gopala Krishnan G, Shahid H. Nasopharyngeal carcinoma database. Med J Malaysia. 2008;63(Suppl C):59–62. [PubMed] [Google Scholar]
- 6.Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116:281–297. doi: 10.1016/s0092-8674(04)00045-5. [DOI] [PubMed] [Google Scholar]
- 7.Xie H, Lee L, Scicluna P, Kavak E, Larsson C, Sandberg R, Lui WO. Novel functions and targets of miR-944 in human cervical cancer cells. Int J Cancer. 2015;136:E230–241. doi: 10.1002/ijc.29160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.He H, Tian W, Chen H, Jiang K. miR-944 functions as a novel oncogene and regulates the chemoresistance in breast cancer. Tumour Biol. 2016;37:1599–1607. doi: 10.1007/s13277-015-3844-x. [DOI] [PubMed] [Google Scholar]
- 9.Flores-Perez A, Marchat LA, Rodriguez-Cuevas S, Bautista VP, Fuentes-Mera L, Romero-Zamora D, Maciel-Dominguez A, de la Cruz OH, Fonseca-Sanchez M, Ruiz-Garcia E, la Vega HA, Lopez-Camarillo C. Suppression of cell migration is promoted by miR-944 through targeting of SIAH1 and PTP4A1 in breast cancer cells. BMC Cancer. 2016;16:379. doi: 10.1186/s12885-016-2470-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Liu M, Zhou K, Cao Y. MicroRNA-944 affects cell growth by targeting EPHA7 in non-small cell lung cancer. Int J Mol Sci. 2016;17 doi: 10.3390/ijms17101493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Powrozek T, Krawczyk P, Kowalski DM, Winiarczyk K, Olszyna-Serementa M, Milanowski J. Plasma circulating microRNA-944 and microRNA-3662 as potential histologic type-specific early lung cancer biomarkers. Transl Res. 2015;166:315–323. doi: 10.1016/j.trsl.2015.05.009. [DOI] [PubMed] [Google Scholar]
- 12.Pan T, Chen W, Yuan X, Shen J, Qin C, Wang L. miR-944 inhibits metastasis of gastric cancer by preventing the epithelial-mesenchymal transition via MACC1/Met/AKT signaling. FEBS Open Bio. 2017;7:905–914. doi: 10.1002/2211-5463.12215. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 13.Wen L, Li Y, Jiang Z, Zhang Y, Yang B, Han F. miR-944 inhibits cell migration and invasion by targeting MACC1 in colorectal cancer. Oncol Rep. 2017;37:3415–3422. doi: 10.3892/or.2017.5611. [DOI] [PubMed] [Google Scholar]
- 14.He Z, Xu H, Meng Y, Kuang Y. miR-944 acts as a prognostic marker and promotes the tumor progression in endometrial cancer. Biomed Pharmacother. 2017;88:902–910. doi: 10.1016/j.biopha.2017.01.117. [DOI] [PubMed] [Google Scholar]
- 15.Stein U, Walther W, Arlt F, Schwabe H, Smith J, Fichtner I, Birchmeier W, Schlag PM. MACC1, a newly identified key regulator of HGF-MET signaling, predicts colon cancer metastasis. Nat Med. 2009;15:59–67. doi: 10.1038/nm.1889. [DOI] [PubMed] [Google Scholar]
- 16.Sun DW, Zhang YY, Qi Y, Liu GQ, Chen YG, Ma J, Lv GY. Prognostic and clinicopathological significance of MACC1 expression in hepatocellular carcinoma patients: a meta-analysis. Int J Clin Exp Med. 2015;8:4769–4777. [PMC free article] [PubMed] [Google Scholar]
- 17.Sueta A, Yamamoto Y, Yamamoto-Ibusuki M, Hayashi M, Takeshita T, Yamamoto S, Omoto Y, Iwase H. Differential role of MACC1 expression and its regulation of the HGF/cMet pathway between breast and colorectal cancer. Int J Oncol. 2015;46:2143–2153. doi: 10.3892/ijo.2015.2907. [DOI] [PubMed] [Google Scholar]
- 18.Li HF, Liu YQ, Shen ZJ, Gan XF, Han JJ, Liu YY, Li HG, Huang ZQ. Downregulation of MACC1 inhibits invasion, migration and proliferation, attenuates cisplatin resistance and induces apoptosis in tongue squamous cell carcinoma. Oncol Rep. 2015;33:651–660. doi: 10.3892/or.2014.3612. [DOI] [PubMed] [Google Scholar]
- 19.Yang T, Kong B, Kuang YQ, Cheng L, Gu JW, Zhang JH, Shu HF, Yu SX, He WQ, Xing XM, Huang HD. Overexpression of MACC1 protein and its clinical implications in patients with glioma. Tumour Biol. 2014;35:815–819. doi: 10.1007/s13277-013-1112-5. [DOI] [PubMed] [Google Scholar]
- 20.Koh YW, Hur H, Lee D. Increased MACC1 expression indicates a poor prognosis independent of MET expression in gastric adenocarcinoma. Pathol Res Pract. 2016;212:93–100. doi: 10.1016/j.prp.2015.11.018. [DOI] [PubMed] [Google Scholar]
- 21.Wang Z, Li Z, Wu C, Wang Y, Xia Y, Chen L, Zhu Q, Chen Y. MACC1 overexpression predicts a poor prognosis for non-small cell lung cancer. Med Oncol. 2014;31:790. doi: 10.1007/s12032-013-0790-6. [DOI] [PubMed] [Google Scholar]
- 22.Sheng XJ, Li Z, Sun M, Wang ZH, Zhou DM, Li JQ, Zhao Q, Sun XF, Liu QC. MACC1 induces metastasis in ovarian carcinoma by upregulating hepatocyte growth factor receptor c-MET. Oncol Lett. 2014;8:891–897. doi: 10.3892/ol.2014.2184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Meng F, Li H, Shi H, Yang Q, Zhang F, Yang Y, Kang L, Zhen T, Dai S, Dong Y, Han A. MACC1 down-regulation inhibits proliferation and tumourigenicity of nasopharyngeal carcinoma cells through Akt/beta-catenin signaling pathway. PLoS One. 2013;8:e60821. doi: 10.1371/journal.pone.0060821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bruce JP, Liu FF. MicroRNAs in nasopharyngeal carcinoma. Chin J Cancer. 2014;33:539–544. doi: 10.5732/cjc.014.10175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhu Z, Zhang X, Wang G, Zheng H. Role of MicroRNAs in hepatocellular carcinoma. Hepat Mon. 2014;14:e18672. doi: 10.5812/hepatmon.18672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yao Y, Suo AL, Li ZF, Liu LY, Tian T, Ni L, Zhang WG, Nan KJ, Song TS, Huang C. MicroRNA profiling of human gastric cancer. Mol Med Rep. 2009;2:963–970. doi: 10.3892/mmr_00000199. [DOI] [PubMed] [Google Scholar]
- 27.Jansson MD, Lund AH. MicroRNA and cancer. Mol Oncol. 2012;6:590–610. doi: 10.1016/j.molonc.2012.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Stein U. MACC1-a novel target for solid cancers. Expert Opin Ther Targets. 2013;17:1039–1052. doi: 10.1517/14728222.2013.815727. [DOI] [PubMed] [Google Scholar]
- 29.Zhang R, Shi H, Chen Z, Wu Q, Ren F, Huang H. Effects of metastasis-associated in colon cancer 1 inhibition by small hairpin RNA on ovarian carcinoma OVCAR-3 cells. J Exp Clin Cancer Res. 2011;30:83. doi: 10.1186/1756-9966-30-83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Migliore C, Martin V, Leoni VP, Restivo A, Atzori L, Petrelli A, Isella C, Zorcolo L, Sarotto I, Casula G, Comoglio PM, Columbano A, Giordano S. MiR-1 downregulation cooperates with MACC1 in promoting MET overexpression in human colon cancer. Clin Cancer Res. 2012;18:737–747. doi: 10.1158/1078-0432.CCR-11-1699. [DOI] [PubMed] [Google Scholar]
- 31.Zhang Y, Wang Z, Chen M, Peng L, Wang X, Ma Q, Ma F, Jiang B. MicroRNA-143 targets MACC1 to inhibit cell invasion and migration in colorectal cancer. Mol Cancer. 2012;11:23. doi: 10.1186/1476-4598-11-23. [DOI] [PMC free article] [PubMed] [Google Scholar]


