Abstract
Background: Currently, the accepted standard management of limited-stage small cell lung cancer (SCLC) is concurrent chemoradiotherapy (CCRT), but the frequency of radiotherapy is controversial. Therefore, this meta-analysis, which compared the efficacy and toxicity between twice-daily (BID) and once-daily (OD) CCRT, was performed to help clinicians make better decisions.
Methods: Relevant randomized controlled trials (RCTs) were collected by searching the PubMed, Ovid MEDLINE, Embase, ScienceDirect, Web of Science, the Cochrane Library, Scopus and Google Scholar databases to assess antitumor effects (overall survival, OS; progression-free survival, PFS; overall response rate, ORR) and toxicity (adverse effects, AEs).
Results: We screened 1499 articles and included 5 RCTs including 1421 patients. We found that BID CCRT improved OS (hazard ratio, HR = 0.88, 95% confidence interval, CI 0.78–0.99, p = 0.03), the 1-year OS rate (OSR-1y, risk ratio, RR = 1.07, 95%CI 1.01–1.13, p = 0.03), and OSR-4y (RR = 1.22, 95%CI 1.03–1.43, p = 0.02), with better trends in OSR-2y, OSR-3y, and OSR-5y, compared to OD CCRT. In addition, BID CCRT had a higher complete response (CR, RR = 1.31, 95%CI 1.01–1.70, p = 0.04) than OD CCRT. PFS (HR = 0.92, 95%CI 0.79–1.07, p = 0.29), annual PFS rate, ORR (RR = 0.99, 95%CI 0.93–1.05, p = 0.72), and AEs for all grades (RR = 1.00, 95%CI 0.98–1.01, p = 0.57), and grades 3–5 (RR = 1.02, 95%CI 0.95–1.09, p = 0.60) were similar between the two arms.
Conclusions: BID CCRT appears to be better than OD CCRT for limited-stage SCLC, with better antitumor effects (OS, OSR, and CR) and similar AEs. However, the high levels of AEs in both arms should be taken as a sign of caution. More large sample and high-quality RCTs need to be conducted to confirm our conclusions.
Keywords: twice-daily, once-daily, concurrent chemoradiotherapy, limited-stage small cell lung cancer, meta-analysis
Introduction
It is estimated that 228,150 new cases of lung cancer will be diagnosed in 2019, ~15–30% of which will be small cell lung cancer (SCLC) (1, 2). As reported by the Surveillance, Epidemiology, and End Results (SEER) Cancer Statistics Review, the annual incidence rates of SCLC are decreasing, but the 5-years period survival is only 6.5% (3). This deadly neuroendocrine tumor, which has the characteristics of a short doubling time and early metastasis, is difficult for oncologists to treat (4). For limited-stage SCLC, the current standard treatment is concurrent chemoradiotherapy (CCRT) (5). However, the optimal frequency of CCRT is still controversial.
In light of the National Comprehensive Cancer Network (NCCN) guidelines, twice-daily (BID), and once-daily (OD) CCRT are recommended, but it is not clear which treatment is better (6). Based on the Intergroup 0096 study, 45 Gy in 30 fractions BID CCRT was considered better than 45 Gy in 25 fractions OD CCRT (7). However, this result was not widely accepted because BID CCRT resulted in severe esophagitis and logistical problems (8). The latest large phase III randomized controlled trial (RCT) has reported that the survival outcomes and toxicity are similar between the two regimens (9). Meanwhile, some studies have even shown that OD CCRT improves survival or reduces toxicity compared with BID CCRT (10, 11). To date, the two regimens are both in clinical use with different doses.
As a result, this meta-analysis was performed to compare the efficacy and toxicity of BID CCRT with OD CCRT for limited-stage small cell lung cancer to provide information to clinicians so that they can make better decisions.
Materials and Methods
We performed this meta-analysis in accordance with the Preferred Reporting Items for Systematic review and Meta-Analysis (Table S1) (12).
Search Strategy
We searched the following electronic databases: (1) PubMed; (2) Ovid MEDLINE; (3) Embase; (4) ScienceDirect; (5) Web of Science; (6) The Cochrane Library; (7) Scopus; and Google Scholar. The last search was on July 5, 2019. The main search terms were “twice-daily,” “once-daily,” and “small cell lung cancer.” Furthermore, relevant references of the included studies were browsed and manually screened. The retrieval strategies used to search the electronic databases are presented in Table S2.
Selection Criteria
We set the inclusion criteria based on the principles of PICOS as follows: (1) patient: patients with limited-stage SCLC; according to a 2-stage (limited-stage and extensive-stage) classification scheme, limited-stage SCLC should include patients with a primary tumor and nodal involvement limited to the ipsilateral hemithorax or without distant metastasis (13); (2) intervention and comparison: BID CCRT vs. OD CCRT; (3) outcome: antitumor effect (overall survival, OS; progression-free survival, PFS; overall response rate, ORR), and toxicity (adverse effects, AEs); and (4) study design: RCTs published in English.
We excluded articles without original data, with abstracts only, or with duplicated data as well as reviews, meta-analyses, animal experiments, and unpublished conference papers and clinical trials.
Data Extraction
The data collected from each included study were general information; characteristics of the study participants; specific modalities of radiation and chemotherapy; survival outcomes (OS, PFS, ORR), including follow-up periods; and AEs, which included the total AEs, all grades and grade 3–5 AEs. The whole information extraction process was completed by two researchers independently, and any disagreements were solved through discussion.
Quality Assessment
We analyzed the methodological quality of the RCTs according to the Cochrane Handbook, which considers seven dimensions of risk of bias (14). Each dimension contains at least one item, which was used to determine “low risk,” “high risk,” and “risk unknown.” One point was given for “low risk,” and high-quality studies had a score ≥3 points.
We rated the evidence quality for each outcome according to the GRADE (Grading of Recommendations Assessment, Development, and Evaluation) system, as follows: (1) High; (2) Moderate; (3) Low; and (4) Very low (15). The included RCTs may have been degraded by the following five factors: risk of bias, inconsistency, indirectness, imprecision, and publication bias.
Statistical Analysis
We used Review Manager 5.3 and STATA 12.0 for data analysis. For the time-to-event data, such as OS and PFS, the hazard ratio (HR) with the 95% confidence interval (CI) was used and pooled as an effective indicator (HR > 1 favors the OD arm). If the HR did not appear in the literature, we obtained it from Kaplan–Meier curves through the method described by Tierney et al. (16). In addition, for dichotomous outcomes, such as ORR and AEs, we calculated and pooled risk ratios (RR) with the 95% CI (ORR: RR > 1 favors the BID arm; AEs: RR > 1 favors the OD arm). Subgroup analyses of OS and PFS were carried out for the continent, models of CCRT regarding the radiotherapy dose (fractional and full) and timing, and chemotherapy cycle. The fixed effect model was initially used. χ2 tests and the I2 index were used to investigate heterogeneity among trials. When substantial heterogeneity appeared (I2 > 50% or p < 0.1 for the χ2 test), the random effect model was used and sensitivity analysis was conducted (14). Publication bias detection was performed for OS and PFS. P < 0.05 indicated a significant difference.
Results
Search Results and Study Quality Assessment
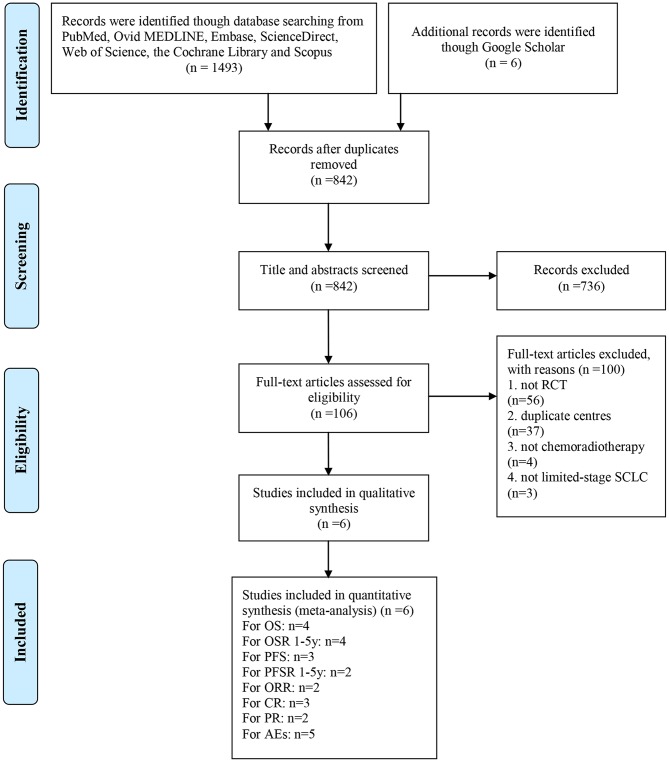
A total of 1499 studies were identified from the search of the electronic databases, and six related articles based on five RCTs including 1,421 patients were eligible for inclusion (7, 9, 17–20). Bonner et al. (17) and Schild et al. (18) described the same study and different results. The former described AEs and the response rate, and the latter described survival outcomes. The literature selection is shown in Figure 1. All of the studies were of high quality. The detailed quality assessment of each included study is shown in Figure S1. The evidence quality of 24/46 outcomes was “High,” and the rest was “Moderate” (Table S3).
Figure 1.
Flow chart of literature selection.
Basic Characteristics of the Studies
Of the five studies, two were conducted in America (7, 17, 18), one in Norway (20), one in England (19), and one with patients from multiple centers from eight countries (9). Eligible patients included in these trials were required to have a good performance status (the performance score was recorded as 0–2) and a limited-stage disease that met the above definition. The mode of CCRT was not identical in each experiment. Physical dose was converted into equivalent dose in 2 Gy/f (EQD2) linear quadratic model, with alpha/beta = 10 for early reaction tissues (tumor tissue) and alpha/beta = 3 for late reaction tissues (normal tissue), according to the method provided by Fowler (21). Patients who achieved complete response (CR), which referred to the complete disappearance of the tumor in this meta-analysis, received prophylactic cranial irradiation (PCI). The specific basic characteristics of the studies are presented in Table 1.
Table 1.
Basic characteristics of included literatures.
| Study | Country | Patient (n) | Median age (year) | Gender M/F (n) | Radiotherapy dose | Radiotherapy timing | Chemotherapy scheme | Median follow-up (months) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BID | OD | BID | OD | BID | OD | Physical dose | (Gy) | (Gy) | |||||||
| NCCTG | 1999 | Bonner et al. (17) | America | 130 | 132 | - | - | 74/56 | 77/55 | 48Gy/1.5Gy/32F/3W; | 46.0; | 43.2; | W13 | EP: W1,5,9,13,17,21 | 39 |
| 50.4Gy/1.8Gy/28F/6W | 49.6 | 48.4 | |||||||||||||
| NCCTG | 2004 | Schild et al. (18) | America | 130 | 131 | 62.5 | 63 | 74/56 | 76/55 | Same as above | 88.8 | ||||
| ECOG | 1999 | Turrisi et al. (7) | America | 211 | 206 | 61 | 63 | 58/42 | 59/41 | 45Gy/1.5Gy/30F/3W; | 43.1; | 40.5; | W1 | EP: W1,4,7,10 | 96 |
| 45Gy/1.8Gy/25F/5W | 44.2 | 43.2 | |||||||||||||
| CNFT | 2012 | Colaco et al. (19) | UK | 12 | 26 | 61 | –/– | –/– | 45Gy/1.5Gy/30F/3W; | 43.1; | 40.5; | W4; | EP: W1,4,7,10 | 16.9 | |
| 66Gy/2.0Gy/33F/6W | 66 | 66 | |||||||||||||
| RHA | 2016 | Grønberg et al. (20) | Norway | 73 | 84 | 63 | 63 | 36/37 | 45/39 | 45Gy/1.5Gy/30F/3W; | 43.1; | 40.5; | W4 | EP: W1,4,7,10 | 81 |
| 42Gy/2.8Gy/15F/3W | 44.8 | 48.7 | |||||||||||||
| CONVERT | 2017 | Faivre-Finn et al. (9) | Multiple | 274 | 273 | 62 | 63 | 147/127 | 150/123 | 45Gy/1.5Gy/30F/3W; | 43.1; | 40.5; | W4; | EP: W1,4,7,10,13,16 | 45 |
| 66Gy/2.0Gy/33F/7W | 66 | 66 | |||||||||||||
BID, twice-daily; OD, once-daily; M, male; F, female; D, days; W, week(s); OS, overall survival; PFS, progression-free survival; Gy, grays; F(f), fraction(s); EP, etoposide/cisplatin.
aDE (Equivalent dose in 2 Gy/f of tumor tissue) = full dose*(fractional dose+10)/(2+10).
bDL (Equivalent dose in 2 Gy/f of normal tissue) = full dose*(fractional dose+3)/(2+3).
Survival Outcomes
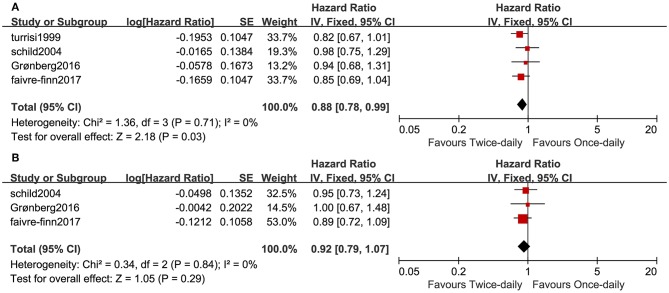
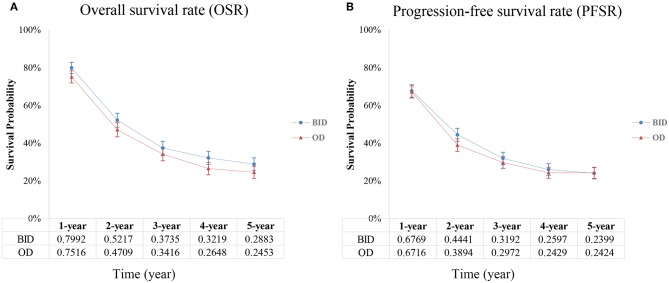
Four studies compared OS in the two arms and included 1,378 patients (687 for the twice-daily group and 691 for the once-daily group). We found that BID CCRT led to better OS (HR = 0.88, 95%CI 0.78–0.99, p = 0.03, I2 = 0%; Figure 2A) and increased median OS by ~4 months (25.6 months vs. 21.6 months) vs. OD CCRT. The BID arm had a higher 1-year overall survival rate (OSR-1y, RR = 1.07, 95%CI 1.01–1.13, p = 0.03, I2 = 3%) and OSR-4y (RR = 1.22, 95%CI 1.03–1.43, p = 0.02, I2 = 0%), and OSR-2y, OSR-3y and OSR-5y tended to favor the BID arm compared with the OD arm (Figure 3A and Figure S2).
Figure 2.
Forest plots of OS (A) and PFS (B).
Figure 3.
Annual OSR (A) and annual PFSR (B).
Three studies analyzed PFS. We found that PFS was similar (HR = 1.08, 95%CI 0.93–1.25, p = 0.30, I2 = 0%; Figure 2B) between the two arms. The annual progression-free survival rate (PFSR) within 5 years tended to favor the BID arm but without a significant difference (Figure 3B and Figure S3).
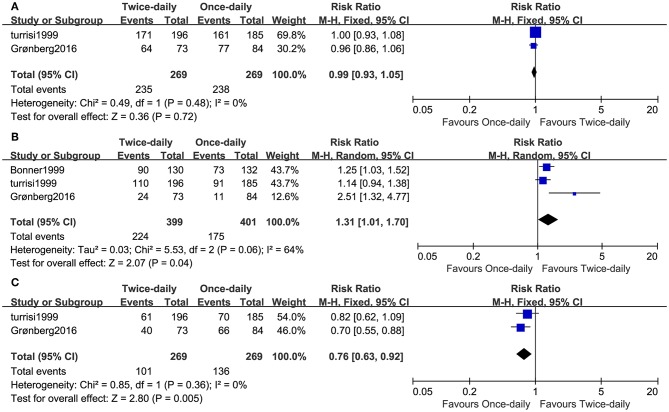
Two studies including 538 patients reported the ORR. We found that the ORR was similar (RR = 0.99, 95%CI 0.93–1.05, p = 0.72, I2 = 0%; Figure 4A) and high (BID: 87% vs. OD: 88%) between the two arms. Three studies compared CR, while two studies compared partial response (PR). The BID arm had higher CR (RR = 1.31, 95%CI 1.01–1.70, p = 0.04, I2 = 64%; Figure 4B) and lower PR (RR = 0.76, 95%CI 0.63–0.92, p = 0.005, I2 = 0%; Figure 4C) compared with the OD arm.
Figure 4.
Forest plots of ORR (A), CR (B), and PR (C).
Toxicity
Two studies compared all grades of AEs. We found that the total AEs (RR = 1.00, 95%CI 0.98–1.01, p = 0.57) and the ten most reported AEs (myelotoxicity, leukopenia, fatigue, neutropenia, anemia, esophagitis, nausea, thrombocytopenia, weight loss, and anorexia) were similar between the two arms (Table 2).
Table 2.
Top 10 adverse effects (all grades).
| All grades adverse effects |
Twice-daily arm (event/total) | Once-daily arm (event/total) | The incidence of adverse effects (%)a | RR (95% CI) | P-value | Heterogeneity | |
|---|---|---|---|---|---|---|---|
| I2 (%) | P-value | ||||||
| Total | 204/206 | 202/203 | 99.27 | 1.00 [0.98, 1.01] | 0.57 | - | - |
| Myelotoxicityb | 199/206 | 201/203 | 97.80 | 0.98 [0.95, 1.00] | 0.10 | - | - |
| Leukopenia | 197/206 | 190/203 | 94.62 | 1.02 [0.98, 1.07] | 0.36 | - | - |
| Fatigue | 243/266 | 247/263 | 92.63 | 0.97 [0.93, 1.02] | 0.26 | - | - |
| Neutropenia | 421/472 | 396/466 | 87.10 | 1.05[1.00, 1.10] | 0.05 | 0 | 0.36 |
| Anemia | 401/472 | 390/466 | 84.33 | 1.02 [0.96, 1.07] | 0.59 | 0 | 0.70 |
| Esophagitis | 365/460 | 315/499 | 74.81 | 1.19 [0.75, 1.90] | 0.46 | 96 | <0.00001 |
| Nausea | 195/266 | 197/263 | 74.10 | 0.98 [0.88, 1.08] | 0.68 | - | - |
| Thrombocytopenia | 134/206 | 125/203 | 63.33 | 1.06 [0.91, 1.22] | 0.47 | - | - |
| Weight loss | 136/206 | 118/203 | 62.10 | 1.14 [0.98, 1.32] | 0.10 | - | - |
| Anorexia | 153/266 | 150/263 | 57.28 | 1.01 [0.87, 1.17] | 0.91 | - | - |
RR, risk ratio; CI, confidence interval.
Event (twice-daily arm + once-daily arm)/Total (twice-daily arm + once-daily arm).
Myelotoxicity was defined as any decrease in marrow-derived cells in the peripheral-blood counts.
Five studies including 1,366 patients analyzed grade 3–5 AEs. We found that the total AEs (RR = 1.17, 95%CI 0.80–1.72, p = 0.42) and the 10 most reported AEs (myelotoxicity, leukopenia, neutropenia, thrombocytopenia, esophagitis, anemia, infection, fatigue, and vomiting) were similar between the two arms (Table 3).
Table 3.
Top 10 adverse effects (grade 3–5).
| Grade 3–5 adverse effects | Twice-daily arm (event/total) | Once-daily arm (event/total) | The incidence of adverse effects (%)a | RR (95% CI) | P-value | Heterogeneity | |
|---|---|---|---|---|---|---|---|
| I2 (%) | P-value | ||||||
| Total | 255/336 | 230/335 | 72.28 | 1.17 [0.80, 1.72] | 0.42 | 87 | 0.005 |
| Myelotoxicityb | 179/206 | 173/203 | 86.06 | 1.02 [0.94, 1.10] | 0.63 | - | - |
| Leukopenia | 340/409 | 334/419 | 81.40 | 1.04 [0.98, 1.11] | 0.23 | 0 | 0.61 |
| Neutropenia | 422/545 | 395/550 | 74.61 | 1.06 [0.95, 1.17] | 0.30 | 57 | 0.1 |
| Thrombocytopenia | 129/409 | 155/419 | 34.30 | 0.86 [0.72, 1.03] | 0.10 | 24 | 0.27 |
| Esophagitis | 157/675 | 121/691 | 20.35 | 1.39 [0.91, 2.11] | 0.13 | 67 | 0.002 |
| Anemia | 109/675 | 100/682 | 15.40 | 1.10[0.86, 1.40] | 0.46 | 11 | 0.34 |
| Infection | 91/675 | 97/682 | 13.71 | 0.99[0.77, 1.26] | 0.93 | 0 | 0.72 |
| Nausea | 45/396 | 48/395 | 11.76 | 0.94[0.60, 1.37] | 0.74 | 0 | 0.70 |
| Fatigue | 31/266 | 31/263 | 11.72 | 0.99 [0.62, 1.58] | 0.96 | - | - |
| Vomiting | 52/602 | 50/598 | 8.50 | 1.04[0.72, 1.50] | 0.85 | 0 | 0.84 |
RR, risk ratio; CI, confidence interval.
Event (twice-daily arm + once-daily arm)/Total (twice-daily arm + once-daily arm).
Myelotoxicity was defined as any decrease in marrow-derived cells in the peripheral-blood counts.
Four studies compared the treatment-related mortality (TRM). We found that the TRM (RR = 0.96, 95%CI 0.50–1.87, p = 0.92, I2 = 33%; Figure S4A) was similar between the two arms. The top three causes of treatment-related deaths were pulmonary effects, infection and neutropenic sepsis, and no significant difference was found between the two arms (Table S4).
Subgroup Analysis
To explore whether the survival outcomes changed, we established subgroups for OS and PFS according to continent, radiotherapy dose (fractional and full) and timing, and chemotherapy cycle. We found that BID CCRT led to better OS vs. OD CCRT in standard fractionation (1.8 or 2 Gy/f, HR = 0.87, 95%CI 0.76–0.98, p = 0.03, I2 = 0%). The results of other subgroups tended to support BID CCRT compared with OD CCRT although no significant differences were found (Table 4).
Table 4.
Subgroup analysis for overall survival and progression-free survival.
| Subgroup | OS | PFS | ||||||
|---|---|---|---|---|---|---|---|---|
| No.of studies | HR (95% CI) | P | I2 (%) | No.of studies | HR (95% CI) | P | I2 (%) | |
| Total | 4 | 0.88 [0.78, 0.99] | 0.03 | 0 | 3 | 0.92 [0.79, 1.07] | 0.29 | 0 |
| Continent | ||||||||
| North America | 2 | 0.88 [0.75, 1.03] | 0.12 | 6 | 1 | 0.95 [0.73, 1.24] | 0.71 | - |
| Europe | 1 | 0.94 [0.68, 1.31] | 0.73 | - | 1 | 1.00 [0.67, 1.48] | 0.98 | - |
| Radiotherapy fractional dose | ||||||||
| 1.5Gy (BID) vs. 1.8Gy/2.0Gy (OD) | 3 | 0.87 [0.76, 0.98] | 0.03 | 0 | 2 | 0.91 [0.77, 1.07] | 0.26 | 0 |
| 1.5Gy (BID) vs. 2.8Gy (OD) | 1 | 0.94 [0.68, 1.31] | 0.73 | - | 1 | 1.00 [0.67, 1.48] | 0.98 | - |
| Equivalent dose in 2 Gy/f | ||||||||
| < 60 Gy | 3 | 0.89 [0.77, 1.03] | 0.55 | 0 | 2 | 0.96 [0.77, 1.20] | 0.75 | 0 |
| ≥60 Gy | 1 | 0.85 [0.69, 1.04] | 0.11 | - | 1 | 0.89 [0.72, 1.09] | 0.25 | - |
| Radiotherapy fractional dose | ||||||||
| Radiotherapy timing | ||||||||
| The first cycle of chemotherapy | 1 | 0.82 [0.67, 1.01] | 0.06 | - | 1 | 0.89 [0.72, 1.09] | 0.25 | - |
| The second cycle of chemotherapy | 2 | 0.87 [0.73, 1.04] | 0.13 | 0 | 2 | 0.91 [0.76, 1.09] | 0.31 | 0 |
| The fourth cycle of chemotherapy | 1 | 0.98 [0.75, 1.29] | 0.91 | - | 1 | 0.95 [0.73, 1.24] | 0.71 | 0 |
| Chemotherapy cycle | ||||||||
| Four cycles | 2 | 0.86 [0.72, 1.02] | 0.08 | 0 | 1 | 1.00 [0.67, 1.48] | 0.98 | - |
| Six cycles | 2 | 0.89 [0.76, 1.05] | 0.18 | 0 | 2 | 0.91 [0.77, 1.07] | 0.26 | 0 |
OS, overall survival; PFS, progression-free survival; HR, hazard ratio; CI, confidence interval; Gy, grays; BID, twice-daily; OD, once-daily; f, fraction.
Sensitivity Analysis
To evaluate sensitivity and stability, we conducted a sensitivity analysis for CR with significant heterogeneity. We found that the outcome of CR was stable after omitting one study (Figure S5).
Publication Bias
We found no publication bias for OS (Begg's test p = 0.734; Egger's test p = 0.160) and PFS (Begg's test p = 0.296; Egger's test p = 0.205) in this meta-analysis (Figure S6).
Discussion
The median survival of limited-stage SCLC has been reported to be 15–20 months, indicating that this cancer extremely malignant (22). For limited-stage SCLC that is not suitable for surgery, the accepted standard treatment is CCRT (23). However, there is much debate about whether radiotherapy should be BID or OD (24). This is the first meta-analysis to determine the optimal scheme between the two regimens; this meta-analysis includes five RCTs including 1,421 patients. The results of this meta-analysis indicated that BID CCRT improved OS, OSR-1y, and OSR-4y, with better trends in OSR-2y, OSR-3y, and OSR-5y, compared to OD CCRT. Subgroup analyses of OS in standard fractionation also supported this result. In addition, BID CCRT had a higher complete response than OD CCRT. No significant differences were found for PFS, annual PFSR, ORR, or AEs of grade 3–5 or all grades between the two arms. Other subgroup analyses of OS and PFS regarding continent, radiotherapy dose (fractional and full) and timing, and chemotherapy cycle tended to favor BID CCRT but without significant differences between the two arms.
In fact, Turrisi et al. reported that the OS of BID CCRT was better than that of OD CCRT (p = 0.04), which agreed with the results of this meta-analysis (median OS: 25.6 months vs. 21.6 months) (7). The annual OSR suggested that there was a potential survival advantage in BID CCRT compared to OD CCRT. Schreiber et al. also found that compared with OD CCRT with different doses, the median survival time of 45 Gy BID CCRT was longer (BID: 22.1 months vs. OD: 45 Gy, 17.2 months, 46–59.4 Gy, 18.3 months, 60–61.2 Gy, 19.2 months, and 62–72 Gy, 19.5 months) (25). However, a cohort study in Asia found that OD CCRT had a significantly longer median OS than BID CCRT (47.2 months vs. 32.8 months) (10). The studies we included in this meta-analysis mainly focused on Europe and the Americas, and the results of this meta-analysis may only apply in these areas. Subgroup analyses of OS in standard fractionation indicated BID CCRT was superior to OD CCRT but no significant difference was found in hypofractionation (>2 Gy/f) and other subgroup analysis results, which might be caused by an insufficient sample size. Xia et al. study also shown that 2.5 Gy/f OD CCRT had favorable survival (26). The ongoing CALGB 30610/RTOG 0538 study (NCT00632853) is further exploring the effects of high dose fractions (27). According to the NCCN guidelines, which recommend the first or second cycle of chemotherapy be combined with radiotherapy, we performed a set of subgroup analyses and found that OS was improved in the BID arm when radiotherapy was initiated from the first or second cycle of chemotherapy vs. the OD arm (HR = 0.85, 95%CI 0.75–0.97, p = 0.02). One possible reason for this improvement is that repopulation of tumor cells is accelerated during chemotherapy cycles and proliferating cells may be more sensitive to BID irradiation (28). Another possible reason for the improvement is that lower overall doses lead to improved treatment delivery of BID CCRT compared with OD CCRT; thus, more patients receive full-dose irradiation. The PFS and PFSR results were consistent with the included studies, which tended to favor BID CCRT but without statistically significant differences compared with OD CCRT. The subgroup analysis reported similar results. However, a previous cohort study reported that the median PFS was longer in the OD arm vs. the BID arm (20.1 months vs. 18.8 months), although there was no significant difference. We recommend that high-quality and well-designed RCTs should take the Asian region into account.
In all of the included studies, the ORRs to CCRT of the two arms were both nearly 90 percent, and no significant difference was reported, which formed the basis of our results. Kubota et al. also found that 95 percent of limited-stage SCLC patients receiving BID CCRT combined with etoposide and cisplatin had an objective response in a large sample RCT (29). In general, for treatment of a tumor with poor prognosis, the CR and PR results should support the same arm (30). However, in this meta-analysis, the higher CR and lower PR in the BID regime only demonstrated an advantage due to the high ORR. These results are in line with the RHA study and NCCTG study but not the ECOG study, in which the difference between the two arms was not significant. Salama et al. found low CR (41%) and high PR (47%) in connected Cancer and Leukemia Group B limited-stage SCLC trials (39808, 30002, and 30206) using high dose OD CCRT, which was similar to our results (OD: 44% and 51%) (31). It was possible that BID CCRT provided a higher biological effective dose for a shorter time of radiation compared with the OD arm, thus preventing further tumor replication.
Regarding AEs, although no significant difference was reported between the two arms in most of the included studies, the NCCTG study found that BID CCRT had a higher incidence of grade 3–5 AEs (54 vs. 39%) and thrombocytopenia (45 vs. 59%) and the ECOG study found that BID CCRT had a higher incidence of esophagitis (63 vs. 44%) vs. OD CCRT. Thus, we conducted a subgroup analysis of grade 3–5 esophagitis and found similar results in the subgroup from North America, where the included studies were published in 1999 (RR = 2.06, 95%CI 1.46–2.89, p < 0.0001; Figure S4B). However, a recent study reported that the incidence of severe esophagitis after BID CCRT was only 2% (29). A possible reason for this result is that previous studies used large and 2-dimensional fields with elective nodal irradiation, which increased radiation exposure to normal tissues (9). At present, 3D- or intensity-modulated radiotherapy provides fewer, higher dose fractions and achieves similar results as the hyper-fractionated regimen (32). We suggest that future studies should use advanced radiotherapy techniques to determine the optimal dose and fractionation for limited-stage SCLC therapy.
There were several limitations of this meta-analysis: (1) Only five RCTs were included, which impacted the representativeness of the results; (2) substantial heterogeneity appeared in the analysis which might make the results unstable: most of the participants were from Europe and North America, which affected the representativeness of the results; only the CONVERT study reported similar improvements in OS in elderly patients for both regimes, which might not allow the elimination of the interference of age (33); PCI might be unbalanced between the two arms (RR = 1.07, 95%CI 1.00–1.14, p = 0.06, I2 = 0%; Figure S4C), which had been reported to improve OS (34); at baseline, all patients received CT scans, and only some patients from CONVERT received PET/CT imaging. In fact, pretreatment with PET scans had been reported to improve OS (35); only the RHA study had a definite staging on CT scans before admission, and the rest were performed later, which might lead to stage migration; (3) some of the time-to-event data were obtained from Kaplan–Meier curves, which were unstable; factors such as necrosis and hypoxia could affect the alpha/beta value, which made the inaccuracy of the formula to estimate EQD2 increase; and (4) there is an economic argument for BID CCRT as compared to OD CCRT because of the shortened treatment time, but no formal economic analysis had been done on both regimens. These findings suggest that further well-designed prospective studies need to be carried out.
Conclusion
BID CCRT appears to be better than OD CCRT, with better antitumor effects (OS, OSR, and CR) and similar AEs for limited-stage SCLC patients. However, the high levels of AEs in both regimens should be a sign of caution. Subgroup analysis of OS and PFS tended to favor BID CCRT, especially comparing OD CCRT in standard fractionation. Moreover, more large sample and well-designed RCTs need to be conducted to confirm our conclusions.
Data Availability Statement
All datasets generated for this study are included in the article/Supplementary Material.
Author Contributions
QW: full access to all of the data in the manuscript and takes responsibility for the integrity of the data, the accuracy of the data analysis, and statistical analysis. QW and WZ: drafting of the manuscript. QW, WZ, YX, SZ, XC, and FY: critical revision of the manuscript for important intellectual content. YW and WZ: supervision. All authors: concept and design and acquisition, analysis, or interpretation of data.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors thank professor Bentong Yu, MD (The first affiliated hospital of Nanchang University) for her advice and professor Xiaoshu Cheng, MD, Ph.D. (The second affiliated hospital of Nanchang University) for his data collection.
Glossary
Abbreviations
- SCLC
small cell lung cancer
- SEER
Surveillance, Epidemiology, and End Results
- CCRT
concurrent chemoradiotherapy
- NCCN
National Comprehensive Cancer Network
- BID
twice-daily
- OD
once-daily
- Gy
gray
- RCT
randomized controlled trial
- PRISMA-P
Preferred Reporting Items for Systematic review and Meta-Analysis Protocols
- OS
overall survival
- PFS
progression-free survival
- ORR
objective response rate
- AEs
adverse effects
- GRADE
Grading of Recommendations Assessment, Development, and Evaluation
- HR
hazard ratio
- CI
confidence interval
- RR
risk ratio
- f
fraction
- EQD2
equivalent dose in 2 Gy/f
- CR
complete response
- PCI
prophylactic cranial irradiation
- OSR
overall survival rate
- y
year
- PFSR
progression-free survival rate
- PR
partial response
- TRM
treatment-related mortality.
Footnotes
Funding. This study was supported by National Natural Science Foundation of China (NSFC), number of grants (81560345), Natural Science Foundation of Jiangxi Province (Grant number: 20181BAB215027). The funding had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2019.01460/full#supplementary-material
Risk of bias of included literatures.
Forest plot of annual OSR.
Forest plot of annual PFSR.
Forest plots of TRM (A), grade 3–5 esophagitis (B), and PCI (C).
Sensitivity analysis about CR.
Egger tests about OS (A) and PFS (B).
PRISMA checklist.
Search strategies of electronic databases.
GRADE quality assessment by therapeutic strategy and study design for the outcomes of survival and toxicity.
Ranking of the cause of treatment-related mortality.
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA. (2019) 69:7–34. 10.3322/caac.21551 [DOI] [PubMed] [Google Scholar]
- 2.Pezzi TA, Schwartz DL, Mohamed AS, Welsh JW, Komaki RU, Hahn SM, et al. Barriers to combined-modality therapy for limited-stage small cell lung cancer. JAMA Oncol. (2018) 4:e174504. 10.1001/jamaoncol.2017.4504 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Howlader N, Noone A, Krapcho M, Miller D, Brest A, Yu M, et al. SEER Cancer Statistics Review, 1975–2016. Bethesda, MD: National Cancer Institute; Available online at: https://seer.cancer.gov/csr/1975_2016/, based on November 2018 SEER data submission, posted to the SEER web site, April 2019. [Google Scholar]
- 4.Gazdar AF, Bunn PA, Minna JD. Small-cell lung cancer: what we know, what we need to know and the path forward. Nat Rev Cancer. (2017) 17:725–37. 10.1038/nrc.2017.87 [DOI] [PubMed] [Google Scholar]
- 5.van Loon J, Dingemans A-MC. Is concurrent chemoradiotherapy also the best treatment for elderly patients with limited-stage small cell lung cancer?–What the CONVERT data can tell us. J Thor Oncol. (2019) 14:13–15. 10.1016/j.jtho.2018.11.005 [DOI] [PubMed] [Google Scholar]
- 6.National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology. Small Cell Lung Cancer, Version 1 (2019). Available online at: https://www.nccn.org/professionals/physician_gls/default.aspx (accessed July 30, 2019).
- 7.Turrisi AT, Kim K, Blum R, Sause WT, Livingston RB, Komaki R, et al. Twice-daily compared with once-daily thoracic radiotherapy in limited small-cell lung cancer treated concurrently with cisplatin and etoposide. N Engl J Med. (1999) 340:265–71. 10.1056/NEJM199901283400403 [DOI] [PubMed] [Google Scholar]
- 8.Movsas B, Moughan J, Komaki R, Choy H, Byhardt R, Langer C, et al. Radiotherapy patterns of care study in lung carcinoma. J Clin Oncol. (2003) 21:4553–9. 10.1200/JCO.2003.04.018 [DOI] [PubMed] [Google Scholar]
- 9.Faivre-Finn C, Snee M, Ashcroft L, Appel W, Barlesi F, Bhatnagar A, et al. Concurrent once-daily versus twice-daily chemoradiotherapy in patients with limited-stage small-cell lung cancer (CONVERT): an open-label, phase 3, randomised, superiority trial. Lancet Oncol. (2017) 18:1116–25. 10.1016/S1470-2045(17)30318-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zeng H, Li R, Hu C, Qiu G, Ge H, Yu H, et al. Association of twice-daily radiotherapy with subsequent brain metastases in adults with small cell lung cancer. JAMA Netw Open. (2019) 2:e190103. 10.1001/jamanetworkopen.2019.0103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Boyer MJ, Willaims CD, Kelley MJ, Salama JK. Once versus twice daily fractionation for limited stage SCLC. Int J Rad Oncol Biol Phys. (2017) 99:e442 10.1016/j.ijrobp.2017.06.1660 [DOI] [Google Scholar]
- 12.Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting items for systematic reviews and Meta-analysis: the PRISMA statement. Br Med J. (2009) 339:2535 10.1136/bmj.b2535 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Micke P, Faldum A, Metz T, Beeh K-M, Bittinger F, Hengstler J-G, et al. Staging small cell lung cancer: veterans administration lung study group versus international association for the study of lung cancer-what limits limited disease? Lung Cancer. (2002) 37:271–6. 10.1016/S0169-5002(02)00072-7 [DOI] [PubMed] [Google Scholar]
- 14.Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al. Cochrane Handbook for Systematic Reviews of Interventions Version 6.0. Cochrane (2019). Available online at: www.training.cochrane.org/handbook (accessed July, 2019).
- 15.Guyatt G, Oxman AD, Akl EA, Kunz R, Vist G, Brozek J, et al. GRADE guidelines: 1. Introduction—GRADE evidence profiles and summary of findings tables. J Clin Epidemiol. (2011) 64:383–94. 10.1016/j.jclinepi.2010.04.026 [DOI] [PubMed] [Google Scholar]
- 16.Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials. (2007) 8:16. 10.1186/1745-6215-8-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bonner JA, Sloan JA, Shanahan TG, Brooks BJ, Marks RS, Krook JE, et al. Phase III comparison of twice-daily split-course irradiation versus once-daily irradiation for patients with limited stage small-cell lung carcinoma. J Clin Oncol. (1999) 17:2681. 10.1200/JCO.1999.17.9.2681 [DOI] [PubMed] [Google Scholar]
- 18.Schild SE, Bonner JA, Shanahan TG, Brooks BJ, Marks RS, Geyer SM, et al. Long-term results of a phase III trial comparing once-daily radiotherapy with twice-daily radiotherapy in limited-stage small-cell lung cancer. Int J Rad Oncol Biol Phys. (2004) 59:943–51. 10.1016/j.ijrobp.2004.01.055 [DOI] [PubMed] [Google Scholar]
- 19.Colaco R, Sheikh H, Lorigan P, Blackhall F, Hulse P, Califano R, et al. Omitting elective nodal irradiation during thoracic irradiation in limited-stage small cell lung cancer–evidence from a phase II trial. Lung Cancer. (2012) 76:72–7. 10.1016/j.lungcan.2011.09.015 [DOI] [PubMed] [Google Scholar]
- 20.Grønberg BH, Halvorsen TO, Fløtten Ø, Brustugun OT, Brunsvig PF, Aasebø U, et al. Randomized phase II trial comparing twice daily hyperfractionated with once daily hypofractionated thoracic radiotherapy in limited disease small cell lung cancer. Acta Oncol. (2016) 55:591–7. 10.3109/0284186X.2015.1092584 [DOI] [PubMed] [Google Scholar]
- 21.Fowler JF. The linear-quadratic formula and progress in fractionated radiotherapy. Br J Radiol. (1989) 62:679–94. 10.1259/0007-1285-62-740-679 [DOI] [PubMed] [Google Scholar]
- 22.Sun A, Durocher-Allen LD, Ellis PM, Ung YC, Goffin JR, Ramchandar K, et al. Guideline for the initial management of small cell lung cancer (limited and extensive stage) and the role of thoracic radiotherapy and first-line chemotherapy. Clin Oncol (R Coll Radiol). (2018) 30:658–666. 10.1016/j.clon.2018.06.008 [DOI] [PubMed] [Google Scholar]
- 23.Blackhall F, Frese KK, Simpson K, Kilgour E, Brady G, Dive C. Will liquid biopsies improve outcomes for patients with small-cell lung cancer? Lancet Oncol. (2018) 19:e470–81. 10.1016/S1470-2045(18)30455-8 [DOI] [PubMed] [Google Scholar]
- 24.Glatzer M, Schmid S, Radovic M, Früh M, Putora PM. The role of radiation therapy in the management of small cell lung cancer. Breathe. (2017) 13:e87–94. 10.1183/20734735.009617 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Schreiber D, Wong AT, Schwartz D, Rineer J. Utilization of hyperfractionated radiation in small-cell lung cancer and its impact on survival. J Thor Oncol. (2015) 10:1770–5. 10.1097/JTO.0000000000000672 [DOI] [PubMed] [Google Scholar]
- 26.Xia B, Hong L-Z, Cai X-W, Zhu Z-F, Liu Q, Zhao K-L, et al. Phase 2 study of accelerated hypofractionated thoracic radiation therapy and concurrent chemotherapy in patients with limited-stage small-cell lung cancer. Int J Rad Oncol Biol Phys. (2015) 91:517–23. 10.1016/j.ijrobp.2014.09.042 [DOI] [PubMed] [Google Scholar]
- 27.Manoharan P, Salem A, Mistry H, Gornall M, Harden S, Julyan P, et al. 18F-Fludeoxyglucose PET/CT in SCLC: analysis of the CONVERT randomized controlled trial. J Thor Oncol. (2019) 14:1296–305. 10.1016/j.jtho.2019.03.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Davis AJ, Tannock IF. Repopulation of tumour cells between cycles of chemotherapy: a neglected factor. Lancet Oncol. (2000) 1:86–93. 10.1016/S1470-2045(00)00019-X [DOI] [PubMed] [Google Scholar]
- 29.Kubota K, Hida T, Ishikura S, Mizusawa J, Nishio M, Kawahara M, et al. Etoposide and cisplatin versus irinotecan and cisplatin in patients with limited-stage small-cell lung cancer treated with etoposide and cisplatin plus concurrent accelerated hyperfractionated thoracic radiotherapy (JCOG0202): a randomised phase 3 study. Lancet Oncol. (2014) 15:106–13. 10.1016/S1470-2045(13)70511-4 [DOI] [PubMed] [Google Scholar]
- 30.Andtbacka R, Kaufman HL, Collichio F, Amatruda T, Senzer N, Chesney J, et al. Talimogene laherparepvec improves durable response rate in patients with advanced melanoma. J Clin Oncol. (2015) 33:2780–8. 10.1200/JCO.2014.58.3377 [DOI] [PubMed] [Google Scholar]
- 31.Salama JK, Hodgson L, Pang H, Urbanic JJ, Blackstock AW, Schild SE, et al. A pooled analysis of limited-stage small-cell lung cancer patients treated with induction chemotherapy followed by concurrent platinum-based chemotherapy and 70 Gy daily radiotherapy: CALGB 30904. J Thor Oncol. (2013) 8:1043–9. 10.1097/JTO.0b013e318293d8a4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rwigema J-C, Verma V, Lin L, Berman A, Md M, Levin W, et al. Prospective study of proton-beam radiation therapy for limited-stage small cell lung cancer. Cancer. (2017) 123:4244–51. 10.1002/cncr.30870 [DOI] [PubMed] [Google Scholar]
- 33.Christodoulou M, Blackhall F, Mistry H, Leylek A, Knegjens J, Remouchamps V, et al. Compliance and outcome of elderly patients treated in the concurrent once-daily versus twice-daily radiotherapy (CONVERT) trial. J Thor Oncol. (2019) 14:63–71. 10.1016/j.jtho.2018.09.027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Salem A, Mistry H, Hatton M, Locke I, Monnet I, Blackhall F, et al. Association of chemoradiotherapy with outcomes among patients with stage I to II vs. stage III small cell lung cancer: secondary analysis of a randomized clinical trial. JAMA Oncol. (2019) 5:e185335. 10.1001/jamaoncol.2018.5335 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Xanthopoulos EP, Corradetti MN, Mitra N, Fernandes AT, Kim M, Grover S, et al. Impact of PET staging in limited-stage small-cell lung cancer. J Thor Oncol. (2013) 8:899–905. 10.1097/JTO.0b013e31828e8996 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Risk of bias of included literatures.
Forest plot of annual OSR.
Forest plot of annual PFSR.
Forest plots of TRM (A), grade 3–5 esophagitis (B), and PCI (C).
Sensitivity analysis about CR.
Egger tests about OS (A) and PFS (B).
PRISMA checklist.
Search strategies of electronic databases.
GRADE quality assessment by therapeutic strategy and study design for the outcomes of survival and toxicity.
Ranking of the cause of treatment-related mortality.
Data Availability Statement
All datasets generated for this study are included in the article/Supplementary Material.