Abstract
Background
The optimal upper and lower limits of blood pressure in preterm infants are not known. Exceeding these thresholds may contribute to intraventricular hemorrhage (IVH).
Methods
Preterm infants born ≤30 weeks GA were identified. Infants had continuous measurement of mean arterial blood pressure (MABP) for 7d and cranial ultrasound imaging. IVH was classified as severe IVH (grade 3/4), no severe IVH (no IVH; grade 1/2), or no IVH.
Mean±SEM MABP values from hours 1–168 were calculated and sorted into bins 2 mmHg wide. The normalized proportion of each recording spent in each bin was then calculated. Candidate limits were identified by comparison of MABP distribution in those with severe IVH vs. those without severe IVH.
Results
85 million measurements were made from 157 infants. Mean EGA was 25.2 weeks; mean BW was 749g; 65/157 female; inotrope use in 59/157; grade 3/4 IVH in 29/157. Infants with severe IVH spent significantly more time with extreme MABP measurements (<23 mmHg or >46mmHg) compared to those without severe IVH (12% vs. 8% of recording, p=0.02).
Conclusion
Infants who developed severe IVH had substantially more unstable MABP and spent a significantly greater period of time with MABP outside of the optimal range.
INTRODUCTION
Despite advances in neonatal care, intraventricular hemorrhage (IVH) remains a common complication of prematurity, and the source of potentially severe neurodevelopmental consequences (1). Although rates of IVH have declined, owing largely to the use of antenatal steroids (2), it remains a persistent concern, affecting approximately 25% of all infants born before 30 weeks gestation (3). Despite efforts to reduce rates of IVH through “bundles” of clinical care practices, rates have remained essentially unchanged.
Our understanding of the mechanism(s) behind IVH has evolved over time, with a significant amount of recent research focused on the role of cerebrovascular autoregulation in the development of hemorrhage (4–7). In these studies, concurrent proxy measures of systemic blood flow (arterial blood pressure) and cerebral blood flow (derived by near-infrared spectroscopy, NIRS) were used to quantify the function of the cerebrovascular autoregulatory system. Although important from a mechanistic standpoint, this approach remains abstracted from actionable clinical changes and requires continuous NIRS monitoring, often a challenge in the most preterm of infants with extremely fragile skin. A more clinically accessible target is the arterial blood pressure, a value which is correlated with cerebral oxygenation (8). There has been extensive description of “normative” blood pressure values in this population, yet these studies have yielded conflicting results, largely due to varying and inconsistent methodologic approaches (9–14). The optimal blood pressure range in very-low-birth weight (VLBW) infants is unclear but might be a more easily accessed physiologic biomarker of brain injury risk.
We hypothesize that the risk of IVH is directly related to the length of time spent with blood pressure exceeding upper and lower thresholds. The objective of this study was to empirically derive these thresholds, define the optimal arterial blood pressure range, and to evaluate the relationship between the burden of blood pressure extremes and severe IVH.
METHODS
Patient selection
Patients were selected from a prospectively recorded, multi-institutional library of physiologic data collected from two level IV NICUs (St. Louis Children’s Hospital, University of Virginia Children’s Hospital) over a 5-year period (2012–2017). Both units provide care to a mix of in-born and outborn infants. Inclusion criteria were estimated gestational age (GA) ≤ 30 weeks, cranial ultrasound imaging in the first week of life, and a minimum of seven days of arterial blood pressure (BP) data (umbilical or peripheral arterial line). Infants were excluded if they did not meet the above requirements or if they were transferred or expired prior to the end of the first week of life. The Institutional Review Board at each center reviewed and approved the study protocol.
Sample characteristics
Comprehensive demographic and outcome data were collected for all included infants, including GA at birth, birthweight (BW), small for gestational age (defined as BW at < 10th centile), sex, race, highest grade of IVH, highest mode of ventilatory support, and mode of delivery. Exposure to inotropic medication (i.e. dopamine, epinephrine) and antenatal steroids was recorded. All infants had cranial ultrasound imaging performed at least once in the first week of life, and the highest noted grade of IVH was used for analysis.
Blood pressure data collection
At each center, physiologic data (including mean arterial BP, MABP) from the patient monitor was prospectively archived in a database (BedMasterEx, ExcelMedical, Jupiter, FL) with a sampling rate of 0.5 Hz. The file associated with each infant was then extracted from the database and converted to a MATLAB matrix for further processing.
Pre-processing
All data underwent a simple one-step error correction process which removed out of range values (MABP < 15 mmHg or > 80 mmHg) as occurs during blood sampling or infusion. Next, the data were aligned by postnatal age (hours since birth) and the mean MABP for each hour was calculated. All measured mean MABP values were then sorted into one of fifteen bins, each 2-mmHg wide, from 20–21 mmHg to 48–49 mmHg. To account for variation in recording length, the proportion of time spent in each bin was normalized by recording length.
Empirically defining optimal blood pressure
Current literature provides conflicting guidance as to the ideal MABP of a preterm infant and three divergent theories have emerged. In one proposal, a target MABP threshold of 30 mmHg emerged after a series of studies suggested a lower incidence of brain injury when MABP remains above this level (10,12,15). A second approach, based on normative data, calls for a target MABP threshold at or above the GA in weeks (9). A third approach, often called “permissive hypotension,” is not focused on hard thresholds, but rather is tolerant of low MABP with the hypothesis that the link between MABP and brain injury in tenuous at best (16).
Although there is disagreement over the value of these bounds, there are unequivocal blood pressure limits, beyond which there is pathologic cerebral blood flow (17–19). We hypothesized that infants with severe brain injury (IVH) will spend a greater proportion of the time with MABP values at the extremes of the distribution and that there is a direct correlation between the temporal burden of MABP extremes and the risk of severe IVH.
First, infants were divided into three groups by IVH status: severe IVH (grade 3 or 4), no severe IVH (no IVH or grades 1–2), or no detectable IVH. Next, the mean normalized proportion of the total recording length was averaged by group for each MABP bin. In order to identify candidate MABP thresholds, beyond which there is increased risk of severe IVH, we systematically examined differences in the distribution of blood pressures between the groups.
Statistical approach
Univariate analysis of clinical, demographic, and physiologic characteristics was conducted using the Fisher’s Exact test (categorical variables), the Mann-Whitney test (two-group continuous variables), or the Kruskall-Wallis test (three-group continuous variables).
To identify the optimal thresholds in an unbiased fashion, we serially examined the time spent in each MABP bin with a univariate binary logistic regression model in order to determine at which threshold there was the greatest discrimination between groups, defined by greatest area under the curve (AUC). As IVH itself may cause hemodynamic instability, this calculation was performed twice – first with a comparison of the severe IVH and not severe IVH groups followed by a comparison of severe IVH to no detectable IVH.
Although the initial thresholds were identified by comparing infants with and without severe IVH to maximize the sensitivity of the threshold, we hypothesized that there is in fact a dose-response relationship between the burden of MABP extremes and the severity of IVH. To this end, we developed a multivariate linear regression model to investigate the impact of MABP extreme burden on the severity of IVH (graded from 0 to 4) while controlling for other factors known to influence IVH (gestational age, birth weight, antenatal steroids, and inotrope exposure).
All statistical analysis was conducted in R version 3.6.0 (R Foundation for Statistical Computing, Vienna, Austria).
RESULTS
Cohort characteristics
During the study period, 1392 infants were born at or before 30 completed weeks of gestation, 157 of which met all inclusion criteria. For the included infants, the mean EGA was 25.2 ± 1.8 weeks; mean BW was 749 ± 201 g; 65/157 female; inotrope use in 59/157. IVH of any grade was present in 61/157 (38%) of the infants, grade 3/4 was present in 29/157 (18%) of the infants and was diagnosed at a median age of 47.5 hours. Inotrope exposure differed between infants with and without severe IVH (69% vs. 30%, p < 0.01) as did documented antenatal steroid administration (62% vs. 77%, p=0.02). A complete listing of infants’ characteristics is shown in Table 1.
Table 1.
Sample and Recording Characteristics
| No severe IVH (n=128) | Severe IVH (n=29) | P value | |
|---|---|---|---|
| GA, mean (SD), weeks | 25.2 (1.9) | 25.1 (1.5) | 0.92 |
| Birth weight, mean (SD), grams | 741 (203) | 786 (195) | 0.21 |
| Male sex, n (%) | 72 (56) | 20 (69) | 0.30 |
| Inotrope exposure in first 7d, n (%) | 39 (30) | 20 (69) | <0.01* |
| Antenatal steroids, n (%) | 99 (77) | 18 (62) | 0.02* |
| Vaginal delivery, n (%) | 52 (41) | 13 (45) | 0.68 |
| Race/Ethnicity | |||
| Asian, n (%) | 1 (1) | 0 (0) | 0.29 |
| Black, n (%) | 32 (25) | 12 (41) | |
| Hispanic, n (%) | 7 (5) | 1 (4) | |
| White, n (%) | 79 (62) | 13 (45) | |
| Not listed, n (%) | 9 (7) | 3 (10) | |
| Highest mode of ventilatory support, n (%) | |||
| CPAP | 20 (15) | 4 (14) | 0.09 |
| Conventional mechanical ventilation | 75 (59) | 11 (38) | |
| High-frequency oscillatory ventilation | 33 (26) | 14 (48) | |
| Mean MABP, mean (SD), mmHg | 34.0 (4.6) | 34.2 (4.4) | 0.87 |
| Recording with MABP < GA, mean (SD), % | 14 (13) | 26 (29) | 0.48 |
| Recording with MABP < 30 mmHg, mean (SD), % | 58 (30) | 67 (21) | 0.29 |
| Raw recording length, median (IQR), hours | 153 (151–166) | 143 (146–167) | 0.35 |
| Recording rejected for errors, mean (SD), % | 9 (25) | 15 (24) | 0.35 |
Footnote:
denotes significance at p < 0.05.
Data quality
A total of 85 million measurements from the 157 infants were included in the final analysis; a mean of 10% of the data were rejected for errors. The overall median recording length was 163 hours and was not different between groups (153 hours for no severe IVH, 143 hours for severe IVH groups, p=0.35).
MABP distribution
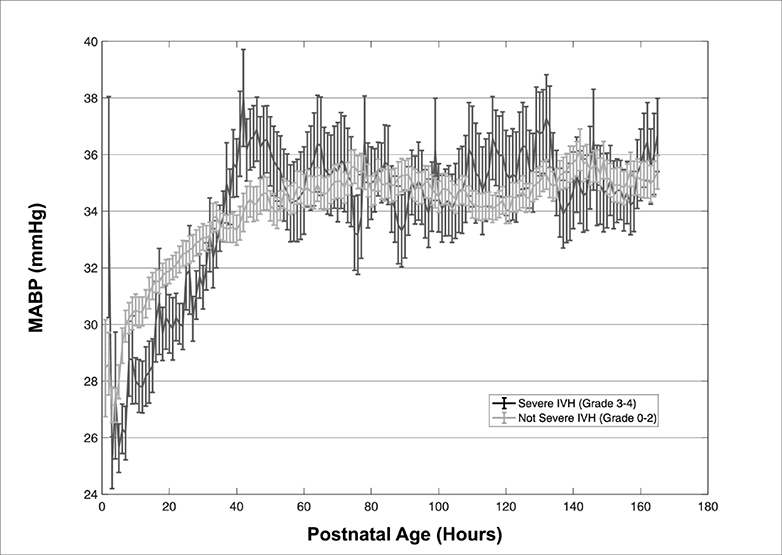
The mean MABP over the 7-day period was not different between infants with severe IVH, without severe IVH, and no detectable IVH (34.1 vs. 33.8 vs. 34.2 mmHg, p=0.58), but visual comparison of the hourly mean MABP shows markedly different patterns. Infants without severe IVH had a gradual increase in MABP while those with severe IVH had a markedly lower MABP for the first 40h followed by significant high-amplitude fluctuations over the next 48–72h. (Figure 1). When using the Zubrow definition of hypotension (MABP < GA in weeks), infants with severe IVH spent a greater proportion of time with hypotension than those without severe IVH and no detectable IVH (29% vs. 23% vs. 12%), although this difference was not statistically significant (p=0.78). When using the Mial-Allen definition of hypotension (MABP < 30 mmHg, regardless of GA), infants with severe IVH spent slightly less time with hypotension than infants without severe IVH and no IVH (21% vs. 30% vs 28%), although this difference again was not statistically significant (p=0.54).
Figure 1–
Hourly plot of mean ± SEM of MABP for infants with severe IVH and without severe IVH. Note the significantly greater fluctuations of the severe IVH group.
Threshold identification and model development
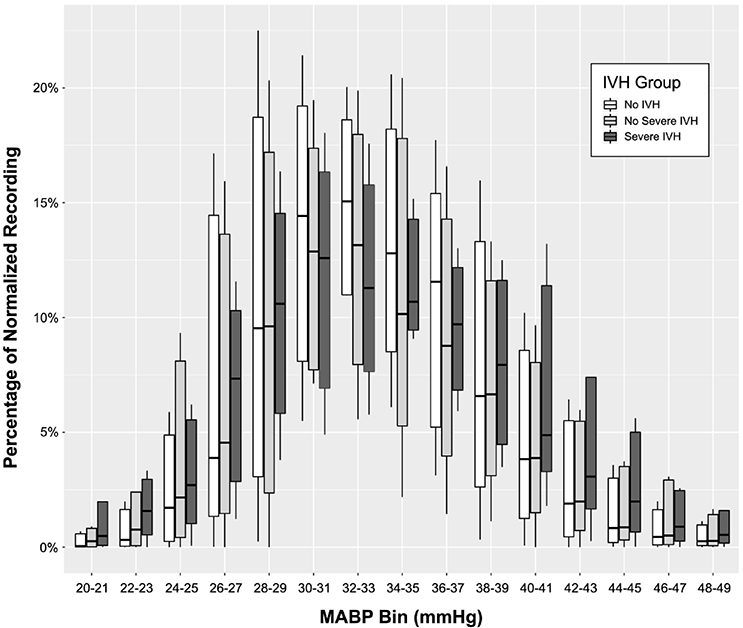
A visual inspection of grouped box and whisker plots (Figure 2) reveals obvious qualitative differences; the infants with severe IVH spend a proportionally greater amount of time at the extreme ends of the distribution, particularly below 26 and above 39 mmHg. This finding is in contrast to the infants without IVH or with mild IVH who spend a proportionally greater amount of time in the middle range of blood pressures.
Figure 2–
Grouped boxplots demonstrating the burden of MABP at each bin by group. Of note, infants with severe IVH spent a significantly greater proportion of time with MABP at the extremes of the distribution, both high and low.
The mean percentage of the recording at or beyond each bin was systematically evaluated in order to identify candidate thresholds for the final model, defined by the MABP bin with the greatest AUC in univariate analysis. When comparing infants with severe IVH to those without severe IVH, these MABP thresholds are noted at ≤ 23 mmHg (AUC = 0.612) and ≥ 46 mmHg (AUC = 0.624) (Table 2a). When comparing infants with severe IVH to those without detectable IVH, the maximal AUC occurs at the same BP thresholds, ≤ 23 mmHg (AUC = 0.664) and ≥ 46 mmHg (AUC = 0.637) (Table 2b). When the burden of MABP values above and below these thresholds are combined, the discriminatory power of the models become even stronger (AUC = 0.650 and 0.698, respectively.
Table 2a.
High and low MABP burdens, no severe IVH vs. severe IVH
| No severe IVH | Severe IVH | ||
|---|---|---|---|
| Threshold (mmHg) | Mean normalized percentage of recording beyond threshold | AUC | |
| ≤ 21 | 0.01 | 0.02 | 0.634 |
| ≤ 23 | 0.03 | 0.04 | 0.637 |
| ≤ 25 | 0.07 | 0.09 | 0.612 |
| ≤ 27 | 0.14 | 0.17 | 0.582 |
| ≥ 40 | 0.16 | 0.20 | 0.606 |
| ≥ 42 | 0.10 | 0.13 | 0.616 |
| ≥ 44 | 0.06 | 0.08 | 0.614 |
| ≥ 46 | 0.03 | 0.04 | 0.624 |
| ≥ 48 | 0.01 | 0.02 | 0.619 |
| ≤ 23 and ≥ 46 | 0.08 | 0.12 | 0.659 |
Table 2b.
High and low MABP burdens, no IVH vs. severe IVH
| No IVH | Severe IVH | ||
|---|---|---|---|
| Threshold (mmHg) | Mean normalized percentage of recording beyond threshold | AUC | |
| ≤ 21 | 0.01 | 0.02 | 0.650 |
| ≤ 23 | 0.02 | 0.04 | 0.664 |
| ≤ 25 | 0.06 | 0.09 | 0.638 |
| ≤ 27 | 0.14 | 0.16 | 0.606 |
| ≥ 40 | 0.15 | 0.20 | 0.615 |
| ≥ 42 | 0.09 | 0.13 | 0.629 |
| ≥ 44 | 0.05 | 0.08 | 0.628 |
| ≥ 46 | 0.03 | 0.04 | 0.637 |
| ≥ 48 | 0.01 | 0.02 | 0.631 |
| ≤ 23 and ≥ 46 | 0.06 | 0.12 | 0.698 |
On average infants without IVH spent 2% of the recording (202 minutes) with MABP ≤ 23 mmHg while infants with severe IVH spent twice as much time (4% of the recording, 404 minutes) below the same threshold. Similarly, the infants without IVH spent an average of 3% of the recording (303 minutes) with MABP ≥ 46 mmHg while infants with severe injury spent 4% of the recording (404 minutes) above the same level.
In the linear regression model, inotrope exposure (OR 1.73, p=0.02), antenatal steroid exposure (OR 0.47, p <0.01), and the burden of MABP extremes (OR 19.15, p=0.01) were independently predictive of IVH risk and severity, when controlling for gestational age at birth, and birth weight. See Table 3 for complete regression model results.
Table 3.
Regression model output
| Variable | Adjusted OR (95% CI) | P value |
|---|---|---|
| Gestational age at birth | 0.92 (0.79–1.10) | 0.29 |
| Birth weight | 1.00 (0.99–1.01) | 0.15 |
| Antenatal steroids | 0.47 (0.27–0.82) | < 0.01* |
| Inotrope exposure | 1.73 (1.08–2.78) | 0.02* |
| Percent time spent with MABP beyond thresholds | 19.15 (1.75–88.96) | 0.01* |
| Model: IVH Grade ∼ GA + BW + antenatal steroids + inotrope exposure + % outside threshold Model multiple R-squared = 0.164, Model F=5.42, p < 0.01 | ||
Footnote:
denotes significance at p < 0.05.
As the distribution of blood pressures in Figure 1 demonstrates, there is a distinct MABP pattern for infants who are later found to have severe IVH—lower blood pressure in the first 36 hours (the highest risk period of IVH) followed by higher, more variable blood pressures thereafter. When the “low” and “high” burdens are added separately to the model, neither main effect is statistically significant, but their interaction is strongly significant (p< 0.01) suggesting that this particular pattern is uniquely associated with IVH (Table 4).
Table 4.
Interaction model investigation
| Variable | T statistic | P value |
|---|---|---|
| Gestational age at birth | −0.89 | 0.37 |
| Birth weight | 1.60 | 0.11 |
| Antenatal steroids | −2.84 | < 0.01* |
| Inotrope exposure | 1.71 | 0.09 |
| Percent time spent with MABP ≤ 23 mmHg | 1.74 | 0.09 |
| Percent time spent with MABP ≥ 46 mmHg | 0.76 | 0.44 |
| MABP Interaction | 1.98 | 0.04* |
Footnote:
denotes significance at p < 0.05.
DISCUSSION
Defining the “normal” blood pressure of a preterm infant remains one of the most challenging conundrums in Neonatology. Although the blood pressure itself is a dynamic factor, it is only one piece of a complex system of cardiac output, vascular autoregulation, and metabolic demand (20). Previous normative studies have helped to outline what the blood pressure of infants with good outcomes is, but not necessarily what it needs to be. IVH represents the most consequential outcome of a failure to keep the blood pressure within these boundaries. The fragile germinal matrix of the preterm brain is poorly suited to handle such fluctuation. Furthermore, clinical interventions such as the administration of intravenous fluids, inotropic agents, and postnatal corticosteroids introduces additional instability to the blood pressure, creating the potential for therapeutic overshoot and possibly contributing to IVH risk.
We have hypothesized that IVH results from an excessive burden of blood pressure values outside of a narrow envelope. The MABP is an ideal metric for this investigation, as it is a single measure reflective of the composite influence of cardiac output, volume status, pH, amongst others. When examined over time, the value only increases; infants with severe IVH have lower MABP values as well as substantially more unstable values, as clearly demonstrated in Figure 1. When examined as a cumulative burden in this data-rich cohort, a clear pattern emerges; not only do infants with severe IVH spend more time at the extremes of MABP, but clear transition points can be identified where the severe IVH group makes up the majority of observations beyond those points. Furthermore, there is a very specific pattern of early low blood pressure followed by widely variable high blood pressure. Given the statistically significant interaction, both must occur for severe IVH to result.
These results offer a hint towards possible “guard rails” for blood pressure management, namely that maintenance of MABP within the 23–46 mmHg window may be the ideal target range in preterm infants <30 weeks gestation. It is crucial to emphasize that infants with good outcomes do spend time outside of this narrow window, nearly 8% of the recording (approximately 14 hours in the first week) on average. In a secondary analysis of this cohort, infants without exposure to inotropes spent an average of 4% of the recording (nearly 7 hours) with a MABP ≥ 46h mmHg, suggesting that MABP values this high routinely occur as a part of the natural progression of the blood pressure rather than from over-treatment. However, persistent and prolonged hypo- or hypertension should be a warning sign prompting further investigation and possibly intervention.
We have also demonstrated that the burden of hypotension by traditional BP definitions (MABP < GA in weeks, MABP < 30 mmHg) is not associated with severe IVH. This is not surprising. The Zubrow study (9) utilized oscilliometric measurements of blood pressure three times daily. In addition to significant issues with test-retest reliability for non-invasive BP measurement, the sampling rate is wholly insufficient to capture the widely-varying BP fluctuations characteristic of the first week of a preterm infant’s life. The Miall-Allen study (10) does not suffer from the same issues as invasive arterial measurements were used. The authors noted that sixty-minutes of exposure to a MABP under 30 mmHg was associated with an increased risk of severe hemorrhage. The present study demonstrates otherwise; infants without severe hemorrhage spent slightly more time with MABP < 30 mmHg (30% of recording) compared to those with severe hemorrhage (21%). One limitation of the Miall-Allen study is that it was performed more than 30 years ago, in a very different era of neonatology before the widespread use of antenatal steroids and surfactant replacement therapy. It is possible that changes in perinatal management permit wider latitude in acceptable blood pressures than was previously possible. It is also possible that the 30 infants in that study were not fully representative of the premature population as a whole.
There are important limitations to consider in the present study. One potential concern is selection bias introduced by the inclusion criteria, namely that infants in modern neonatal care who have arterial lines in place for at least 7 days are likely to be more immature and more sick than infants with fewer line days (or no arterial line at all), indeed only about 10% of the total infants admitted to the two NICUs met this stringent criteria. Although this was an intentional study design decision, to include the entire risk period for IVH, future studies should confirm that similar upper and lower boundaries are found for infants with shorter duration recordings. Second, these findings are based on a single metric (MABP), by design. It is possible, and indeed likely, that the inclusion of other physiologic measures will add additional value but will be more difficult to obtain and translate into clinical practice. Third, the timing of cranial ultrasounds was entirely at the discretion of the clinical team and was highly variable. As a result, discrete timing of the hemorrhage and clear identification of pre- and post-hemorrhage periods was not possible. Future studies should focus on consistent and frequent imaging (ideally once per day) to more clearly delineate these periods. Finally, a note of caution should be urged against over-interpretation of the presented data. These results represent the findings of infants at two institutions over a short interval of time. These results should be evaluated at other centers, with different demographic characteristics and clinical practices to ensure their robustness and before these thresholds are implemented in clinical practice.
Given the infrequent nature of cranial ultrasounds, this study design does not permit investigation into the burdens of hypo- and hypertension before and after IVH. The hypothesis of this study, supported by previous reports (1), is that hypotension is a causative factor for IVH. However, it is possible that the hemorrhage could itself cause hemodynamic instability, manifested as hypotension. Additionally, the elevated blood pressure values may be deleterious or may reflect the need for higher cerebral blood flow in the post-hemorrhage state, driven by elevated intracranial pressure, as is seen in adult stroke (21,22). Future investigation, with more frequent ultrasounds and more precisely timed hemorrhages will be better suited to answer these questions.
Conclusions
In this study we have demonstrated that infants who develop severe IVH have substantially more unstable blood pressures in the first week of life. Evaluating the distribution of blood pressure measurements, candidate thresholds of optimal blood pressure were derived. Infants with severe IVH have a consistent pattern of low blood pressure initially followed by an “overshoot” and instability. It remains unclear as to where in this pattern severe IVH occurs. Follow-up studies with close attention to the discrete timing of IVH may offer the possibility of a therapeutic BP target
Acknowledgments
Statement of financial support:
This work was supported by the following grants:
1. Washington University Institute of Clinical and Translational Sciences KL2 Training Program (NIH/NCATS KL2 TR000450)
2. The Barnes-Jewish Hospital Foundation and the Washington University Institute of Clinical and Translational Sciences Clinical and Translational Funding Program (NIH/NCATS UL1 TR000448)
3. National Institutes of Health grant numbers K23 NS111086, R01 HD072071, and R01 HL133708
Footnotes
Disclosure: The authors have no financial ties or potential/perceived conflicts of interest with any of the products or manufacturers described in this manuscript.
REFERENCES
- 1.Volpe JJ. Neurology of the newborn. 5th ed. Philadelphia: Saunders/Elsevier; 2008. [Google Scholar]
- 2.Ment LR, Oh W, Ehrenkranz RA, Philip AG, Duncan CC, Makuch RW. Antenatal steroids, delivery mode, and intraventricular hemorrhage in preterm infants. Am J Obstet Gynecol 1995;172:795–800. [DOI] [PubMed] [Google Scholar]
- 3.Stoll BJ, Hansen NI, Bell EF, et al. Neonatal outcomes of extremely preterm infants from the NICHD Neonatal Research Network. Pediatrics 2010;126:443–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Vesoulis ZA, Liao SM, Trivedi SB, Ters NE, Mathur AM. A novel method for assessing cerebral autoregulation in preterm infants using transfer function analysis. Pediatr Res 2016;79:453–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Soul JS, Hammer PE, Tsuji M, et al. Fluctuating pressure-passivity is common in the cerebral circulation of sick premature infants. Pediatr Res 2007;61:467–73. [DOI] [PubMed] [Google Scholar]
- 6.Chock VY, Ramamoorthy C, Van Meurs KP. Cerebral autoregulation in neonates with a hemodynamically significant patent ductus arteriosus. J Pediatr 2012;160:936–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Alderliesten T, Lemmers PMA, Smarius JJM, van de Vosse RE, Baerts W, van Bel F. Cerebral oxygenation, extraction, and autoregulation in very preterm infants who develop peri-intraventricular hemorrhage. J Pediatr 2013;162:698–704.e2. [DOI] [PubMed] [Google Scholar]
- 8.Tsuji M, Saul JP, du Plessis A, et al. Cerebral intravascular oxygenation correlates with mean arterial pressure in critically ill premature infants. Pediatrics 2000;106:625–32. [DOI] [PubMed] [Google Scholar]
- 9.Zubrow AB, Hulman S, Kushner H, Falkner B. Determinants of blood pressure in infants admitted to neonatal intensive care units: a prospective multicenter study. Philadelphia Neonatal Blood Pressure Study Group. J Perinatol 1995;15:470–9. [PubMed] [Google Scholar]
- 10.Miall-Allen VM, de Vries LS, Whitelaw AG. Mean arterial blood pressure and neonatal cerebral lesions. Arch Dis Child 1987;62:1068–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bada HS, Korones SB, Perry EH, et al. Mean arterial blood pressure changes in premature infants and those at risk for intraventricular hemorrhage. J Pediatr 1990;117:607–14. [DOI] [PubMed] [Google Scholar]
- 12.Vesoulis ZA, El Ters NM, Wallendorf M, Mathur AM. Empirical estimation of the normative blood pressure in infants <28 weeks gestation using a massive data approach. J Perinatol 2016;36:291–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lee J, Rajadurai VS, Tan KW. Blood pressure standards for very low birthweight infants during the first day of life. Archives of Disease in Childhood - Fetal and Neonatal Edition 1999;81:F168–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hegyi T, Carbone MT, Anwar M, et al. Blood pressure ranges in premature infants. I. The first hours of life. J Pediatr 1994;124:627–33. [DOI] [PubMed] [Google Scholar]
- 15.St. Peter D, Gandy C, Hoffman SB. Hypotension and Adverse Outcomes in Prematurity: Comparing Definitions. Neonatology 2017;228–33. [DOI] [PubMed] [Google Scholar]
- 16.Alderliesten T, Lemmers PMA, van Haastert IC, et al. Hypotension in Preterm Neonates: Low Blood Pressure Alone Does Not Affect Neurodevelopmental Outcome. J Pediatr 2014; [DOI] [PubMed] [Google Scholar]
- 17.Lou HC, Skov H, Pedersen H. Low cerebral blood flow: a risk factor in the neonate. J Pediatr 1979;95:606–9. [DOI] [PubMed] [Google Scholar]
- 18.Ment LR, Ehrenkranz RA, Lange RC, Rothstein PT, Duncan CC. Alterations in cerebral blood flow in preterm infants with intraventricular hemorrhage. Pediatrics 1981;68:763–9. [PubMed] [Google Scholar]
- 19.Van Bel F, Van de Bor M, Stijnen T, Baan J, Ruys JH. Aetiological rôle of cerebral bloodflow alterations in development and extension of peri-intraventricular haemorrhage. Dev Med Child Neurol 1987;29:601–14. [DOI] [PubMed] [Google Scholar]
- 20.Noori S, McCoy M, Anderson MP, Ramji F, Seri I. Changes in Cardiac Function and Cerebral Blood Flow in Relation to Peri/Intraventricular Hemorrhage in Extremely Preterm Infants. The Journal of Pediatrics 2014;164:264–270.e3. [DOI] [PubMed] [Google Scholar]
- 21.Wallace JD. Blood Pressure After Stroke. JAMA: The Journal of the American Medical Association 1981;246:2177. [PubMed] [Google Scholar]
- 22.Leonardi-Bee J, Bath PMW, Phillips SJ, Sandercock PAG. Blood Pressure and Clinical Outcomes in the International Stroke Trial. Stroke 2002;33:1315–20. [DOI] [PubMed] [Google Scholar]