Abstract
Objective:
To investigate the frequency of hippocampal sulcus remnants (HSRs) in non-elderly adults using ultra-high resolution 7T MR images as well as their imaging features.
Methods:
33 healthy adults underwent 7T MR and multiplanar images of 66 temporal lobes were reviewed independently by 2 neuroradiologists. The detectability of the hippocampal sulcus remnant was calculated. In addition, the inter-observer agreement on the rating scale was evaluated using the κ statistic.
Results:
Both observers identified HSRs with 7T MR images in all subjects. Excellent inter-observer agreement was shown (κ = 1.0). The shape of HSRs was variable (spot-like, curvilinear, ovoid, or beaded appearance). Volumes of the HSRs were not correlated with age.
Conclusions:
HSRs are commonly seen in healthy non-elderly adults using 7T MR imaging. Accurate diagnosis of HSR based on the microanatomy of hippocampus makes it easier to differentiate them from lesions and it may help prevent unnecessary treatment.
Keywords: Hippocampus, Magnetic resonance imaging, Anatomy
Introduction
Hippocampus plays pivotal roles in memory and spatial cognition. Consequently, its detailed function as well as development has been greatly focused on many previous research articles. The hippocampal fissure is uniformly present in fetuses of 12–13.5 weeks1. The hippocampus normally unfolds along the medial surface of the temporal lobe at this age. Subsequently, gradual infolding of dentate gyrus and cornu ammonis occurs around the hippocampal sulcus and the sulcus is eventually obliterated by 18–21 weeks of fetal life2, 3. The remnant of primitive hippocampal sulcus (HSR) may persist into adult life. The hippocampus is supplied by blood vessels derived from posterior cerebral artery, its branches and also often the anterior choroidal artery. Feeding arteries enter the hippocampus by penetrating the dentate gyrus, the fimbriodentate sulcus, and the hippocampal sulcus and its remnants irrespective of their origin4. Taking the above anatomy into consideration, HSRs could appear in all age groups.
The mechanism leading to presence of HSRs is unknown. HSRs are occasionally observed in healthy subjects and are considered normal anatomic variations5, 6. Previous reports indicate that detection rate of HSRs correlates with age2, 6–10 and hypertension9, 10. Establishing the frequency of HSRs in normal adults may address the point.
Here, we hypothesized that HSRs can be commonly visualized using ultra-high resolution MR imaging. Our purpose was to investigate the frequency of HSRs in non-elderly adults using ultra-high resolution 7T MR images as well as their imaging features.
Materials and Methods
This study was approved by the local institutional review board. Informed consent for study participation was obtained from all subjects prior to 7T MR scanning.
MR imaging data of consecutively studied healthy subjects between 21–55 years of age were obtained prospectively. All subjects fulfill the following criteria: (a) No history of hypertension or diabetes mellitus (b) No history of cerebrovascular disease, and (c) able to provide consent; while exclusion criteria were: pregnancy, breast-feeding, or unable to consent/cooperate with the performance of MR imaging.
All images were obtained with a 7T MR scanner (Siemens Healthcare, Erlangen, Germany) using Nova 32-channel receive and 8-channel transmit coil.
Images were acquired using a 3D T2 weighted variable flip angle TSE sequence with the following parameters: TR/TE = 3000/328 ms, NEX = 1, 248 transverse sections, GRAPPA factor = 3, FOV = 210 × 210 × 99.2 mm3, acquisition matrix = 512 × 525 × 256, reconstruction matrix = 512 × 512 × 248, voxel resolution = 0.41 × 0.41× 0.4 mm3, slice thickness = 0.4 mm (no gap), image acquisition time = 8 min 3 sec.
First, the acquired 7T transverse T2-weighted images were displayed for analysis in coronal (slice thickness = 0.41 mm), sagittal (slice thickness = 0.41 mm) and transverse (slice thickness = 0.4 mm) views using ITK-SNAP software (http://www.itksnap.org/). Both medial temporal lobes were independently reviewed by 2 neuroradiologists. HSRs were diagnosed when T2 hyperintense structures similar to CSF were located between dentate gyrus and cornu ammonis (Figs. 1–4). Confidence level for the presence of HSR was graded on a 2-point scale: positive or negative. Frequency of HSRs was calculated. In addition, inter-observer agreement on the rating scale was evaluated using the κ statistic method11. The age of the subjects was compared between male and female using unpaired t-test.
Fig. 1.
Example of the voxel of interests (VOIs). Source image (a). The VOIs are indicated colored blocks (b).
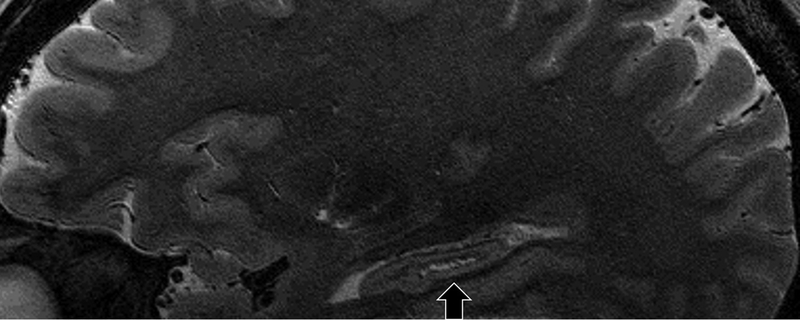
Fig. 4.
51-year-old male. Magnified coronal (a) and continuous sagittal images (b-d). The sagittal images indicate the tubular hypointense structure (arrow) within the HSR (arrowhead), which seems to be blood vessel.
Second, measurement of the HSR volumes was performed. The voxels-of-interest (VOIs) were manually placed by a neuroradiologist, followed by visual inspection by another neuroradiologist on the 7T T2-weighted images using the ITK-SNAP software. Location of HSRs was determined using coronal as well as transverse and sagittal images (Fig. 1 shows an example of the VOIs). Pearson correlation coefficients were calculated to determine if there was significant correlation between subject age and volume of HSRs. Relationship between volumes of the right-sided HSRs and those of the left-sided HSRs was also evaluated.
All statistical analyses were performed using JMP 13 software (SAS Institute, Cary, NC). A p-value less than 0.05 was considered to indicate statistical significance.
Results
33 healthy volunteers consisted of 7 males (mean age ± standard deviation [SD], 32 ± 11 years; range, 21–51 years), and 26 females (mean age ± SD, 37 ± 11 years; range, 21–55 years). No significant differences were found in ages between genders. 66 medial temporal lobes were assessed on 7T T2-weighted images. All studies obtained at 7T were judged to be of excellent quality.
HSRs were found in the hippocampal body and head and also identified 36.4 % (24/66 temporal lobes) in the hippocampal tail (Fig. 2). Their shape was variable (spot-like [59/66], curvilinear [31/66], ovoid [25/66], or beaded [4/66]) (Fig. 3 and Table 1). A tubular hypointense structure was identified within the HSR in 25.8 % (17/66 temporal lobes), and believed to represent a blood vessel (Fig. 4).
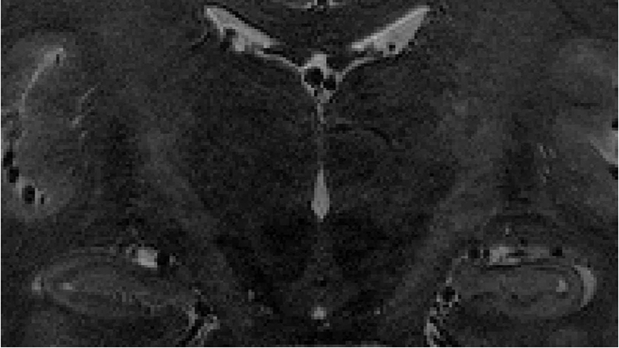
Fig. 2.
21-year-old male. Sagittal (a), coronal (b), and transverse planes (c). The hippocampal sulcus remnants (HSRs) are identified in the hippocampal tail.
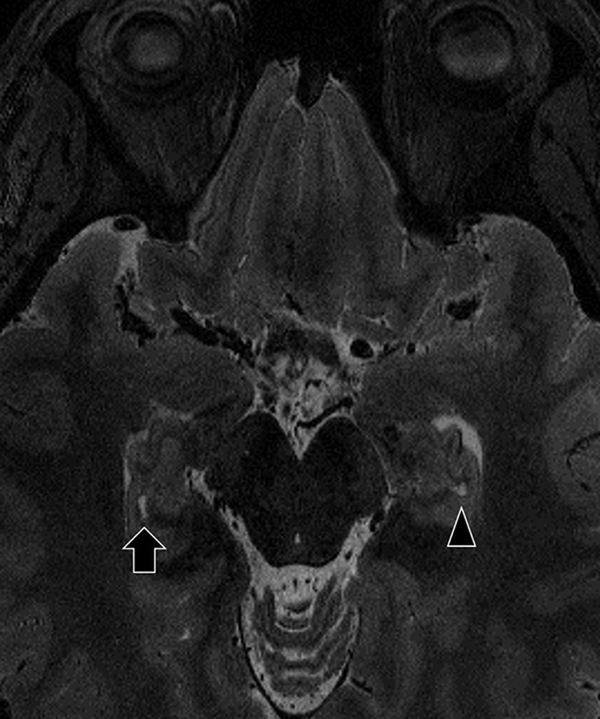
Fig. 3.
26-year-old female. Sagittal (a) and transverse planes (b). The shape of the right HSRs (arrow) appear to be beaded structure on the sagittal image. The left HSR (arrowhead) is also identified on the transverse image.
Table 1:
Shape of the hippocampal sulcus remnants
| Spot-like | 89.4 % (59/66) |
| Curvilinear | 47.0% (31/66) |
| Ovoid | 37.9% (25/66) |
| Beaded | 6.1% (4/66) |
Note: The hippocampal sulcus remnants are composed of more than one types of shape features in several subject
Both observers identified HSRs with 7T MR images in all subjects (66/66). Excellent inter-observer agreement was shown (κ = 1.0). HSRs were located between the dentate gyrus and the cornu ammonis.
Volume of HSRs was variable ranging from 0.34 mm3 to 23.8 mm3 (mean ± SD = 5.02 ± 4.09 mm3). Their volumes did not correlate with age (r = 0.02, p = 0.88; Fig. 5). On the other hand, there was a significant correlation between the volumes of the HSRs bilaterally (r = 0.62, p = 0.0001; Fig. 6). The volume of the right HSRs (mean ± SD = 6.10 ± 4.98 mm3) showed a trend toward being larger than those of the left HSRs (mean ± SD = 3.94 ± 2.62 mm3) although no significant differences were found using Wilcoxon’s test (p = 0.07).
Fig. 5.
Scatter plots of the age and the volume of the HSR. Note that the volume of the HSR is not correlated with age (r = 0.02, p = 0.88)
Fig. 6.
Scatter plots of the right and the left HSR volumes. Solid and dashed lines represent linear regression of the plots and 95% confidence intervals, respectively. There is the significant correlation between the volume of the HSR bilaterally (r = 0.62, p = 0.0001).
Discussion
HSRs were observed in all healthy adult subjects using high resolution 7T MR imaging. It is known that 7T imaging provides better visualization of small structures such perivascular spaces12, 13 and multiple sclerosis lesions14 than lower magnetic fields probably due to increased signal-to-noise ratio, spatial resolution and improved grey/white matter contrast. In in vivo 7T studies, tiny Virchow-Robin spaces have been identifiable using sub-millimeter spatial resolution13, 15. Therefore, high magnetic field strength and high resolution contribute to identification of HSRs.
Unlike other studies5, 6, 9, 10, we were able to confidently identify HSRs in several locations and evaluate additional their imaging features such as shape and volume. HSRs with ultra-high resolution 7T MR imaging varied in shape, and these results might shed light on new perspective although validation studies are needed. In our study, a blood vessel-like tubular hypointense structure was visualized within the HSR. Since blood vessels are known to traverse it, identifying them allows differentiation of this normal structure from lesions.
Volume of HSRs did not correlate with age in our healthy subjects but we found a significant correlation between volumes of HSRs bilaterally. It has been reported that the detection rate of HSRs in elderly subjects correlates with age2, 6–10 and hypertension9, 10, 16. Because of our relatively small sample it is unclear if brain atrophy and disease plays a role in identification of HSRs as it does with perivascular spaces. Barborial et al. suggested that hippocampus sulcus cavity scores were greater in normal elderly subjects with apolipoprotein E (APOE) ε3/4, ε4/4, and ε2/3 when compared with the subjects carrying the APOE ε3/3 genotype2. Batres-Faz et al. demonstrated a correlation between high hippocampal sulcus cavity scores and APOC1 genetic polymorphism in elderly patients with memory impairment7.
We also documented a trend toward a larger volume of the right HSRs although no significant differences were seen. A right-larger-than-left hippocampal asymmetry has been documented in prior studies17–19. Handedness17, environmental factors20, and different developmental process of visuospatial versus linguistic ability19 are presumed causes of asymmetrical growth of the hippocampi. These hippocampal asymmetries may be related to asymmetry of the HSRs.
One of limitations in this study is that HSRs were not confirmed pathologically. Therefore, there was no independent gold standard to obtain true sensitivity. Another limitation is that MR imaging sequences other than T2-weighted images are not available.
Conclusion
HSRs are commonly seen in healthy non-elderly adults using ultra-high resolution 7T MR imaging. Accurate diagnosis of HSR based on the microanatomy of hippocampus makes it easier to differentiate them from lesions and it may help prevent unnecessary treatment.
Abbreviations:
- HSR
hippocampal sulcus remnant
- SD
standard deviation
- VOI
voxel-of-interest
Footnotes
Disclosure of interest
The authors declare that there is no relationships/conditions/circumstances that present a potential conflict of interest in this manuscript.
References
- 1.Humphrey T The development of the human hippocampal fissure. J Anat 1967;101:655–676 [PMC free article] [PubMed] [Google Scholar]
- 2.Barboriak DP, Doraiswamy PM, Krishnan KR, et al. Hippocampal sulcal cavities on MRI: relationship to age and apolipoprotein E genotype. Neurology 2000;54:2150–2153 [DOI] [PubMed] [Google Scholar]
- 3.Dekeyzer S, De Kock I, Nikoubashman O, et al. “Unforgettable” - a pictorial essay on anatomy and pathology of the hippocampus. Insights Imaging 2017;8:199–212 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Erdem A, Yasargil G, Roth P. Microsurgical anatomy of the hippocampal arteries. Journal of neurosurgery 1993;79:256–265 [DOI] [PubMed] [Google Scholar]
- 5.Sasaki M, Sone M, Ehara S, et al. Hippocampal sulcus remnant: potential cause of change in signal intensity in the hippocampus. Radiology 1993;188:743–746 [DOI] [PubMed] [Google Scholar]
- 6.Suzuki M, Matsui O, Ueda F, et al. MR Imaging of Hippocampal Sulcus Remnant: Age-Related Differences. Neuroradiol J 2007;20:611–616 [DOI] [PubMed] [Google Scholar]
- 7.Bartres-Faz D, Junque C, Clemente IC, et al. MRI and genetic correlates of cognitive function in elders with memory impairment. Neurobiology of aging 2001;22:449–459 [DOI] [PubMed] [Google Scholar]
- 8.Yoneoka Y, Kwee IL, Fujii Y, et al. Criteria for normalcy of cavities observed within the adult hippocampus: high-resolution magnetic resonance imaging study on a 3.0-T system. Journal of neuroimaging : official journal of the American Society of Neuroimaging 2002;12:231–235 [PubMed] [Google Scholar]
- 9.Yao M, Zhu YC, Soumare A, et al. Hippocampal perivascular spaces are related to aging and blood pressure but not to cognition. Neurobiology of aging 2014;35:2118–2125 [DOI] [PubMed] [Google Scholar]
- 10.Jimenez-Balado J, Riba-Llena I, Garde E, et al. Prevalence of hippocampal enlarged perivascular spaces in a sample of patients with hypertension and their relation with vascular risk factors and cognitive function. J Neurol Neurosurg Psychiatry 2018;89:651–656 [DOI] [PubMed] [Google Scholar]
- 11.Cohen J Weighted kappa: nominal scale agreement with provision for scaled disagreement or partial credit. Psychol Bull 1968;70:213–220 [DOI] [PubMed] [Google Scholar]
- 12.Maclullich AM, Wardlaw JM, Ferguson KJ, et al. Enlarged perivascular spaces are associated with cognitive function in healthy elderly men. J Neurol Neurosurg Psychiatry 2004;75:1519–1523 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zong X, Park SH, Shen D, et al. Visualization of perivascular spaces in the human brain at 7T: sequence optimization and morphology characterization. NeuroImage 2016;125:895–902 [DOI] [PubMed] [Google Scholar]
- 14.Kollia K, Maderwald S, Putzki N, et al. First clinical study on ultra-high-field MR imaging in patients with multiple sclerosis: comparison of 1.5T and 7T. AJNR American journal of neuroradiology 2009;30:699–702 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cai K, Tain R, Das S, et al. The feasibility of quantitative MRI of perivascular spaces at 7T. J Neurosci Methods 2015;256:151–156 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Li Y, Li J, Segal S, et al. Hippocampal cerebrospinal fluid spaces on MR imaging: Relationship to aging and Alzheimer disease. AJNR American journal of neuroradiology 2006;27:912–918 [PMC free article] [PubMed] [Google Scholar]
- 17.Szabo CA, Xiong J, Lancaster JL, et al. Amygdalar and hippocampal volumetry in control participants: differences regarding handedness. AJNR American journal of neuroradiology 2001;22:1342–1345 [PMC free article] [PubMed] [Google Scholar]
- 18.Utsunomiya H, Takano K, Okazaki M, et al. Development of the temporal lobe in infants and children: analysis by MR-based volumetry. AJNR American journal of neuroradiology 1999;20:717–723 [PMC free article] [PubMed] [Google Scholar]
- 19.Thompson DK, Wood SJ, Doyle LW, et al. MR-determined hippocampal asymmetry in full-term and preterm neonates. Hippocampus 2009;19:118–123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bootsman F, Kemner SM, Hillegers MH, et al. The association between hippocampal volume and life events in healthy twins. Hippocampus 2016;26:1088–1095 [DOI] [PubMed] [Google Scholar]