Abstract
Regulatory B (Breg) cells have been demonstrated to play a suppressive role in chronic hepatitis B virus infection. However, the role of Breg in HBV-related liver cirrhosis has not been determined. Patients with chronic hepatitis B virus infection and HBV-related liver cirrhosis were enrolled. Frequencies of peripheral B cell subsets were assessed by flow cytometry. Intrahepatic B cells, IL-10 positive cells and Breg cells were determined by immunohistochemistry. We found that frequency of peripheral Breg cells was significantly higher in cirrhotic patients compared with that in healthy volunteers. Further, the amount of intrahepatic Breg cells was elevated in patients with HBV-related liver cirrhosis. We identified a positive correlation between intrahepatic Breg cells and histological fibrosis stages. Our results suggested that Breg cells may play a role in HBV-related liver cirrhosis, but the function of Breg cells in cirrhosis remains to be further elucidated.
Keywords: Regulatory B cells, IL-10, liver cirrhosis
Introduction
Liver cirrhosis is a serious global health problem, and results in 1.03 million deaths per year worldwide [1]. The most common cause of liver cirrhosis in China is chronic hepatitis B virus infection. Liver fibrosis is an immune-driving process, during which natural killer cells, T cells, dendritic cells and macrophages are involved in regulating the liver fibrosis and extracellular matrix production [2-4].
B lymphocytes can modulate immune responses by presenting antigens and secreting cytokines during infection, inflammation, and autoimmunity [5]. In recent years, B cells were thought to contribute to liver injury. In a mouse liver fibrosis model, B cells have been proven to promote liver fibrosis in an antibody-independent manner [6]. And in chronic hepatitis B (CHB) patients, intrahepatic CD20 expression was associated with stage of fibrosis [7]. In our previous research, we also found that intrahepatic B cells may play a pathologic role in inflammation in chronic liver disease patients [8].
B lymphocytes are subdivided into effector B cells (Be) and regulatory B cells (Breg) based on cytokine secretion [9]. Breg cells inhibit the function of other immune cells mainly by IL-10 secretion [10]. Some studies have suggested that Breg cells are enriched in CHB patients, which suppress HBV-specific T cell responses, enhance regulatory T cells function in an IL-10-dependent manner [11-13]. However, the role of Breg cells in HBV-related liver cirrhosis remains unclear. To explore the role of Breg cells in HBV-related liver cirrhosis, we investigated the characteristics of Breg cells in blood and liver tissues of patients with HBV-related liver cirrhosis by flow cytometry and immunohistochemistry.
Materials and methods
Patients and samples
Fresh heparinized peripheral blood samples from 32 antiviral treatment-naïve chronic hepatitis B patients, 16 HBV-related liver cirrhosis patients hospitalized to Nanjing Drum Tower Hospital during 2014-2017 and 10 healthy volunteers were collected. Patients who had received immunotherapy or combined with infection, immune diseases and cancer were excluded. Liver biopsy specimens from 16 patients with chronic hepatitis B and 8 patients with HBV-related liver cirrhosis among these patients were included. Healthy liver specimens from 10 transplantation donors were included as controls. Clinical data from the patients were acquired from their medical records at the time of blood collection or liver biopsy before therapy. The research protocol was approved by the Human Ethics Committee of the Nanjing Drum Tower Hospital, and all participants offered written informed consent in this study.
Flow cytometry
A four-parameter flow cytometric analysis of fresh whole blood cells was performed. Fifty microliters of whole blood were directly stained with combinations of the following monoclonal antibodies: anti-CD45 conjugated to PerCP-Cy5.5 (BD Biosciences), anti-CD19 conjugated to APC (BD Pharmingen, cat # 652804), anti-CD24 conjugated to PE (BD Pharmingen, cat # 555428), and anti-CD38 conjugated to FITC (BD Pharmingen, cat # 555459). After 20 min incubation, whole blood was lysed with red blood cell lysis buffer (BD Biosciences, cat # 349202), then the cells were washed twice with PBS, further resuspended in PBS and immediately analyzed.
Histopathology
Liver tissue was fixed in paraformaldehyde for 48 h, then dehydrated, embedded with paraffin and sectioned to 3 μm-thin slices. Hematoxylin-eosin staining of liver tissue specimens was performed to evaluate the inflammation grade and fibrosis stage according to the Batts-Ludwig grading and staging system [14]. The sections were judged by two pathologists separately.
Immunohistochemistry
Immunohistochemical staining of liver tissue sections was processed by EnVision two-step method. Monoclonal antibodies against CD20 (DAKO, cat # M0755, 1:100, USA), IL-10 (Abcam, cat # ab134742, 1:100, USA), PAX5 [15] (Abcam, cat # ab109433, 1:100, USA) and IL-10 double-labeled were used to represent for B cells, IL-10 positive cells and IL-10 producing regulatory B cells, respectively. The dewaxing, antigen retrieval, antibody fixation, and DAB/Fast Red staining were performed according to the standard protocol. The evaluation was carried by two pathologists. Five high-power visual fields per slide were analyzed. The mean number of staining positive cells per high-power field (HPF) was counted for each slide.
Statistical analysis
All statistical analyses were used with the statistical software SPSS 22.0 (Chicago, USA) and P value <0.05 was considered statistically significant. ANOVA or Mann Whitney U test was used to compare data among groups. Correlation analysis was performed by Pearson or Spearman correlation coefficient.
Results
General characteristics of the study population
There were normal or mild to moderately elevated transaminase levels among 48 patients, and the range of alanine aminotransferase level was 10-235 U/L, and that of aspartate transaminase level was 13-105 U/L. The cirrhotic patients were all at decompensation stage, but they didn’t have spontaneous peritonitis, variceal bleeding, or encephalopathy at enrollment time. The characteristics of patients were listed on Table 1.
Table 1.
Clinical profile of enrolled subjects
| Group | HC (n=10) | CHB (n=32) | HBV-LC (n=16) |
|---|---|---|---|
| Age (years) | 31.25 ± 8.54 | 39.09 ± 9.48 | 48.66 ± 10.49 |
| Gender (M/F) | 5/5 | 18/14 | 11/5 |
| ALT (U/L) | 17.62 ± 8.30 | 52.82 ± 51.19 | 36.93 ± 32.71 |
| AST (U/L) | 16.76 ± 5.15 | 34.12 ± 19.44 | 36.00 ± 23.26 |
| ALB (g/L) | 44.35 ± 2.52 | 41.87 ± 3.81 | 36.49 ± 6.87 |
| GLB (g/L) | 20.51 ± 2.77 | 22.59 ± 3.22 | 25.43 ± 5.56 |
| A/G | 2.71 ± 0.18 | 1.88 ± 0.26 | 1.50 ± 0.47 |
| TBIL (μmol/L) | 7.93 ± 1.85 | 14.22 ± 5.85 | 19.33 ± 11.58 |
| WBC (×109/L) | 7.43 ± 0.86 | 5.66 ± 1.68 | 3.88 ± 2.20 |
| PLT (×109/L) | 181.66 ± 18.53 | 160.80 ± 55.37 | 102.20 ± 91.87 |
| CRP (mg/L) | 1.66 ± 0.72 | 2.26 ± 1.97 | 2.55 ± 1.91 |
| Child-Pugh score | NA | NA | 5.91 ± 1.56 |
Data presentation: M ± SEM; NA: not applicable.
Peripheral regulatory B cells are enriched in patients with HBV-related liver cirrhosis
To determine the role of Breg cells in HBV-related liver cirrhosis, we measured the frequency of peripheral Breg cells in patients with CHB and HBV-related liver cirrhosis (HBV-LC) by flow cytometry, respectively. There was no difference in the percentage of CD19+B cells between CHB, HBV-LC and controls (Figure 1A). A significantly higher percentage of CD19+CD24hiCD38hi Breg cells was observed in both HBV-LC patients (6.46 ± 1.16%) and CHB (4.47 ± 0.52%) patients compared with controls (2.13 ± 0.28%) (P<0.01, P<0.05, respectively) (Figure 1B).
Figure 1.
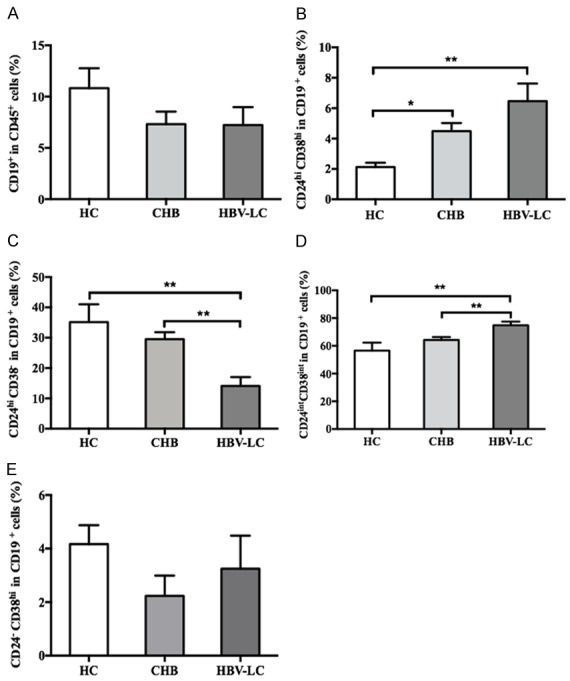
Frequencies of peripheral B cells and B cells subsets in patients with CHB and HBV-related liver cirrhosis. A. Total B cells; B. Breg cells; C. Memory B cells; D. Mature B cells; E. Plasmablasts. **; P<0.01, *; P<0.05.
In addition, we determined the frequencies of other B cell subsets: CD19+CD24hiCD38- memory B cells, CD19+CD24intCD38int mature B cells, and CD19+CD24-CD38hi plasmablasts. The memory B cells were significantly lower in patients with HBV-LC (14.10 ± 2.93%), compared to CHB patients (29.50 ± 2.33%) and healthy controls (35.14 ± 5.89%) (P<0.01, both) (Figure 1C). The mature B cells were significantly higher in patients with HBV-LC (74.80 ± 2.62%), compared to CHB (64.23 ± 2.02%) and controls (56.54 ± 5.77%) (P<0.01, both) (Figure 1D). There was no difference in the frequency of plasmablasts among three groups (Figure 1E).
Intrahepatic regulatory B cells increased in patients with HBV-related liver cirrhosis
We further investigated the intrahepatic B cells, IL-10 secreting cells and Breg cells in patients by immunohistochemistry. The intrahepatic accumulation of CD20 positive B cells in HBV-LC (44.07 ± 18.55/HPF) and CHB patients (36.49 ± 6.45/HPF) was significantly higher compared to controls (3.10 ± 0.90/HPF) (P<0.01, both) (Figure 2A, 2B). The intrahepatic CD20 positive B cells in patients with G2 (43.10 ± 9.58/HPF) and G3-4 (44.90 ± 16.73/HPF) were significantly increased compared to controls (3.10 ± 0.90/HPF) (P<0.01, both) (Figure 2C). The intrahepatic accumulation of CD20 positive B cells in patients with S1-S4 (S1: 28.86 ± 17.75/HPF, S2: 29.31 ± 9.00/HPF, S3: 47.48 ± 10.33/HPF, S4: 44.07 ± 18.55/HPF) was significantly higher compared to controls (3.10 ± 0.90/HPF) (all P<0.01) (Figure 2D).
Figure 2.

Intrahepatic B cells were increased in patients with HBV-related liver cirrhosis. (A) Immunohistochemical staining for CD20; (B) Comparison of intrahepatic CD20 positive B cells between healthy controls, CHB, and HBV-LC patients; Comparison of intrahepatic CD20 positive B cells in patients with different inflammatory grades (C) and fibrotic stages (D). G, inflammatory grades; S, fibrotic stages. **; P<0.01.
The intrahepatic accumulation of IL-10 positive cells in HBV-LC patients (3.75 ± 0.16/HPF) was significantly higher compared to CHB (1.12 ± 0.20/HPF) and controls (0.05 ± 0.05/HPF) (P<0.05, P<0.01, respectively) (Figure 3A, 3B). The intrahepatic accumulation of IL-10 positive cells in patients with G3-4 (3.50 ± 0.28/HPF), G2 (1.70 ± 0.36/HPF) and G1 (1.66 ± 0.33/HPF) was significantly increased compared to controls (0.05 ± 0.05/HPF) (P<0.01, P<0.01, P<0.05, respectively) (Figure 3C). There was no difference in the intrahepatic accumulation of IL-10 positive cells among other groups. The intrahepatic accumulation of IL-10 positive cells in patients with S4 (3.75 ± 0.16/HPF) was significantly higher compared to controls (0.05 ± 0.05/HPF), S1 (0.75 ± 0.47/HPF) and S2 (0.33 ± 0.33/HPF) (P<0.01, P<0.05, P<0.01, respectively) (Figure 3D).
Figure 3.

Intrahepatic IL10 secreting cells were increased in patients with HBV-related liver cirrhosis. (A) Immunohistochemical staining of IL-10; (B) Comparison of intrahepatic IL-10 positive cells between healthy controls, CHB, and HBV-LC patients; Comparison of intrahepatic IL-10 positive cells in patients with different inflammatory grades (C) and fibrotic stages (D). G, inflammatory grades; S, fibrotic stages. *; P<0.05, **; P<0.01.
The intrahepatic accumulation of PAX5/IL-10 double positive Breg cells in HBV-LC patients (0.87 ± 0.08/HPF) was significantly higher compared to CHB (0.11 ± 0.04/HPF) and controls (0) (P<0.01, both) (Figure 4A and 4B). Intrahepatic accumulation of PAX5/IL-10 double positive Breg cells in patients with G3-4 (0.52 ± 0.20/HPF) and G2 (0.38 ± 0.20/HPF) were significantly increased compared to controls (0) (P<0.05, both) (Figure 4C).
Figure 4.

Intrahepatic Breg cells were increased in patients with HBV-related liver cirrhosis. (A) Immunohistochemical double-staining of PAX5/IL-10; (B) Comparison of intrahepatic PAX5/IL-10 double-positive cells between healthy controls, CHB, and HBV-LC patients; Comparison of intrahepatic PAX5/IL-10 double-positive cells in patients with different inflammatory grades (C) and fibrotic stages (D). G, inflammatory grades; S, fibrotic stages. *; P<0.05, **; P<0.01.
Intrahepatic accumulation of PAX5/IL-10 double positive Breg cells in patients with S4 (0.87 ± 0.08/HPF) was elevated compared to controls (0), S1 (0) and S2 (0.041 ± 0.041/HPF) (P<0.01, all) (Figure 4D).
We analyzed the correlation between intrahepatic B cells, IL-10 positive cells, or Breg cells and liver inflammatory grade or fibrotic stage and clinical parameters. The expression of intrahepatic B cells was positively correlated with liver inflammatory grade (G). Both amounts of intrahepatic IL-10 positive cells and Breg cells were positively correlated with liver fibrotic stage (S). There was no significant correlation between the level of intrahepatic regulatory B cells and serum alanine aminotransferase, aspartate transaminase, albumin, globulin, and Child-Pugh scores.
Discussion
Regulatory B cells (Breg) have been found to suppress immune responses in autoimmunity, infection, and cancer mainly through the production of IL-10 [10]. IL-10 is an important anti-inflammatory and anti-fibrosis cytokine which can balance Th1/Th2 responses, enhance regulatory T cell function, and promote IL-4 secretion. IL-10 also can inhibit the activation, and promote the apoptosis of hepatic stellate cells, and regulate the production and degradation of extracellular matrix [16,17]. The source of IL-10 is multicellular, as T cells, monocytes, macrophages, and hepatic stellate cells et al. are known to produce IL-10. It has been proven that B lymphocytes can produce IL-10 [18]; IL-10-producing Breg cells were enriched in CHB patients and able to suppress HBV-specific T cell responses in an IL-10-dependent manner [11]. Breg cells could also accelerate hepatocellular carcinoma progression [19]. The role of Breg cells in fibrosis development remains elusive. An animal model had shown that adoptive transfer of Breg cells could reduce skin and pulmonary fibrosis [20], indicating that Breg cells might have a beneficial role against fibrosis.
In the present study, we determined the frequency of Breg cells in peripheral blood of HBV-related cirrhosis patients by cytometry. We found that the frequency of peripheral Breg cells significantly increased in liver cirrhosis patients without remarkable liver inflammation. Consistent with our results, Liu et al. [13] also showed that the proportion of peripheral Breg cells was positively correlated with the progression of liver fibrosis. Further, it is also shown that the dynamics of Breg cells in patients with pulmonary fibrosis. Specifically, peripheral Breg cells in patients with systemic sclerosis are reduced and functionally impaired, especially in patients with pulmonary fibrosis [21]. These studies suggested that Breg cells might play a role in the pathogenesis of fibrosis.
Previous data had indicated that the frequency of intrahepatic Breg cells was correlated with the level of peripheral Breg cells in CHB patients [13]. We also investigated the regulatory B cells in liver tissue of HBV-related cirrhosis patients by immunohistochemistry. As there is no identified surface marker for intrahepatic Breg cells, we detected the distribution of IL-10-produing B cells by IL-10 and PAX5 double labelling using immunohistochemistry. Breg cells were not detected in normal liver, but significantly increased in patients’ liver even though the amount of Breg cells was still relatively low. The accumulation of intrahepatic Breg cells in patients with the G3-4 and S4 was higher than that in the other groups, and there was a positive correlation between the accumulation of intrahepatic Breg cells and fibrosis score. These results indicated that Breg cells may play a role in the pathogenesis of liver fibrosis.
In our study, elevated Breg cells were observed in the blood and liver of cirrhosis patients, suggesting they might exert regulatory effects both in the circulation and the liver. However, the mechanism and function of elevated Breg cells in liver fibrosis needs further investigation. It was thought that Breg cells could migrate from spleen to lesion location such as joints, lymph nodes, and nervous system [22,23]. A previous study had suggested that the activation of iNKT cells in the liver could cause liver damage and raise Breg cells to enter to inhibit inflammation of the liver [24]. It was necessary to further clarify whether the proliferation of Breg cells in the liver was due to the migration of splenic Breg cells or the differentiation of liver B cells in situ under a fibrotic microenvironment. In a mouse model, B cells played a positive role in liver fibrosis through secreting cytokines such as IL-6 to promote fibrogenesis and stimulate HSC proliferation [6,25]. But the contribution of Breg cells to liver fibrogenesis and HSC proliferation needs further investigation.
Acknowledgements
The study was supported from the National Natural Science Foundation of China (81672025 and 81702011), Medical Science and Technology Development Foundation of Nanjing (ZDX16004 and YKK16118) and Natural Science Foundation of Jiangsu Province for Young Scholar (BK20160121), Jiangsu Science and Technology Development Plan (BE2017605), Jiangsu Provincial Medical Innovation Team (CXTDA2017005) and Nanjing Medical Science and Technique Development Foundation (QRX17121).
Disclosure of conflict of interest
None.
References
- 1.Tsochatzis EA, Bosch J, Burroughs AK. Liver cirrhosis. Lancet. 2014;383:1749–61. doi: 10.1016/S0140-6736(14)60121-5. [DOI] [PubMed] [Google Scholar]
- 2.Szabo G, Mandrekar P, Dolganiuc A. Innate immune response and hepatic inflammation. Semin Liver Dis. 2007;27:339–50. doi: 10.1055/s-2007-991511. [DOI] [PubMed] [Google Scholar]
- 3.Muhanna N, Abu Tair L, Doron S, Amer J, Azzeh M, Mahamid M, Friedman S, Safadi R. Amelioration of hepatic fibrosis by NK cell activation. Gut. 2011;60:90–8. doi: 10.1136/gut.2010.211136. [DOI] [PubMed] [Google Scholar]
- 4.Wynn TA, Barron L. Macrophages: master regulators of inflammation and fibrosis. Semin Liver Dis. 2010;30:245–57. doi: 10.1055/s-0030-1255354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Carter RH. B cells in health and disease. Mayo Clin Proc. 2006;81:377–84. doi: 10.4065/81.3.377. [DOI] [PubMed] [Google Scholar]
- 6.Novobrantseva TI, Majeau GR, Amatucci A, Kogan S, Brenner I, Casola S, Shlomchik MJ, Koteliansky V, Hochman PS, Ibraghimov A. Attenuated liver fibrosis in the absence of B cells. J Clin Invest. 2005;115:3072–82. doi: 10.1172/JCI24798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mohamadkhani A, Naderi E, Sotoudeh M, Katoonizadeh A, Montazeri G, Poustchi H. Clinical feature of intrahepatic B-lymphocytes in chronic hepatitis B. Int J Inflam. 2014;2014:896864. doi: 10.1155/2014/896864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Huang R WH, Liu Y, et al. B cells may play a pathological role in hepatic in ammation in chronic liver diseases. Int J Clin Exp Pathol. 2016;9:6. [Google Scholar]
- 9.Hamze M, Desmetz C, Guglielmi P. B cell-derived cytokines in disease. Eur Cytokine Netw. 2013;24:20–6. doi: 10.1684/ecn.2013.0327. [DOI] [PubMed] [Google Scholar]
- 10.Rosser EC, Mauri C. Regulatory B cells: origin, phenotype, and function. Immunity. 2015;42:607–12. doi: 10.1016/j.immuni.2015.04.005. [DOI] [PubMed] [Google Scholar]
- 11.Das A, Ellis G, Pallant C, Lopes AR, Khanna P, Peppa D, Chen A, Blair P, Dusheiko G, Gill U, Kennedy PT, Brunetto M, Lampertico P, Mauri C, Maini MK. IL-10-producing regulatory B cells in the pathogenesis of chronic hepatitis B virus infection. J Immunol. 2012;189:3925–35. doi: 10.4049/jimmunol.1103139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gong Y, Zhao C, Zhao P, Wang M, Zhou G, Han F, Cui Y, Qian J, Zhang H, Xiong H, Sheng J, Jiang T. Role of IL-10-producing regulatory B cells in chronic hepatitis B virus infection. Dig Dis Sci. 2015;60:1308–14. doi: 10.1007/s10620-014-3358-1. [DOI] [PubMed] [Google Scholar]
- 13.Liu Y, Cheng LS, Wu SD, Wang SQ, Li L, She WM, Li J, Wang JY, Jiang W. IL-10-producing regulatory B-cells suppressed effector T-cells but enhanced regulatory T-cells in chronic HBV infection. Clin Sci (Lond) 2016;130:907–19. doi: 10.1042/CS20160069. [DOI] [PubMed] [Google Scholar]
- 14.Batts KP, Ludwig J. An update on terminology and reporting. Am J Surg Pathol. 1995;19:1409–17. doi: 10.1097/00000478-199512000-00007. [DOI] [PubMed] [Google Scholar]
- 15.Cobaleda C, Schebesta A, Delogu A, Busslinger M. Pax5: the guardian of B cell identity and function. Nat Immunol. 2007;8:463–70. doi: 10.1038/ni1454. [DOI] [PubMed] [Google Scholar]
- 16.Tsukamoto H. Is interleukin-10 antifibrogenic in chronic liver injury? Hepatology. 1998;28:1707–9. doi: 10.1002/hep.510280635. [DOI] [PubMed] [Google Scholar]
- 17.Zhang LJ, Wang XZ. Interleukin-10 and chronic liver disease. World J Gastroenterol. 2006;12:1681–5. doi: 10.3748/wjg.v12.i11.1681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mauri C. Regulation of immunity and autoimmunity by B cells. Curr Opin Immunol. 2010;22:761–7. doi: 10.1016/j.coi.2010.10.009. [DOI] [PubMed] [Google Scholar]
- 19.Shao Y, Lo CM, Ling CC, Liu XB, Ng KT, Chu AC, Ma YY, Li CX, Fan ST, Man K. Regulatory B cells accelerate hepatocellular carcinoma progression via CD40/CD154 signaling pathway. Cancer Lett. 2014;355:264–72. doi: 10.1016/j.canlet.2014.09.026. [DOI] [PubMed] [Google Scholar]
- 20.Yoshizaki A, Fukasawa T, Ebata S, Sato S. The ex vivo-induced regulatory B cells ameliorate tissue fibrosis and autoimmunity via an antioxidative effect in systemic sclerosis. J Dermatol Sci. 2017;86:e11. [Google Scholar]
- 21.Mavropoulos A, Simopoulou T, Varna A, Liaskos C, Katsiari CG, Bogdanos DP, Sakkas LI. Breg cells are numerically decreased and functionally impaired in patients with systemic sclerosis. Arthritis Rheumatol. 2016;68:494–504. doi: 10.1002/art.39437. [DOI] [PubMed] [Google Scholar]
- 22.Yanaba K, Kamata M, Ishiura N, Shibata S, Asano Y, Tada Y, Sugaya M, Kadono T, Tedder TF, Sato S. Regulatory B cells suppress imiquimod-induced, psoriasis-like skin inflammation. J Leukoc Biol. 2013;94:563–73. doi: 10.1189/jlb.1112562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Maseda D, Candando KM, Smith SH, Kalampokis I, Weaver CT, Plevy SE, Poe JC, Tedder TF. Peritoneal cavity regulatory B cells (B10 cells) modulate IFN-gamma+CD4+ T cell numbers during colitis development in mice. J Immunol. 2013;191:2780–95. doi: 10.4049/jimmunol.1300649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Almishri W, Deans J, Swain MG. Rapid activation and hepatic recruitment of innate-like regulatory B cells after invariant NKT cell stimulation in mice. J Hepatol. 2015;63:943–51. doi: 10.1016/j.jhep.2015.06.007. [DOI] [PubMed] [Google Scholar]
- 25.Hasegawa M, Fujimoto M, Takehara K, Sato S. Pathogenesis of systemic sclerosis: altered B cell function is the key linking systemic autoimmunity and tissue fibrosis. J Dermatol Sci. 2005;39:1–7. doi: 10.1016/j.jdermsci.2005.03.013. [DOI] [PubMed] [Google Scholar]


