Abstract
microRNA-1471 (miR-1471) is a newly identified miRNA that is downregulated in breast cancer. However, its biological roles in human tumors are largely unknown. This study aimed to investigate the clinical significance and functions of miR-1471 in glioma. We found miR-1471 expression was significantly reduced in glioma tissues and cell lines. Forced expression of miR-1471 remarkedly suppressed glioma cell proliferation and invasion. Notably, metadherin (MTDH) was validated as a direct target of miR-1471 and the restoration of MTDH expression reversed the inhibitory effects of miR-1471 on glioma cell proliferation and invasion. Also, low miR-1471 expression was a predictor for worse 5-year overall survival of glioma patients. Overall, these results reveal the tumor suppressive role of miR-1471 in glioma, highlighting the potential to consider miR-1471/MTDH axis as a therapeutic target for the treatment of glioma in the near future.
Keywords: miR-1471, MTDH, glioma, proliferation, invasion
Introduction
Glioma is the most aggressive and malignant brain tumor type that represents approximately 30% of all brain and central nervous system tumors [1]. The improvements in treatment measures including chemotherapy, radiotherapy and surgery have greatly increase glioma patients’ survival but it is still adverse [2-4]. The main obstacle is the molecular mechanisms behind glioma progression were not fully understood [4]. Therefore, investigations on these mechanisms will advance the development of novel therapeutic targets.
Metadherin (MTDH), also knownas the name of astrocyte elevated gene-1 (AEG-1), has been widely recognized as an important regulator for the malignancy of various human tumors [5]. It has also been reported to be overexpressed in numerous human cancers including oral squamous cell carcinoma, glioma, osteosarcoma, and non-small cell lung cancer since its first identification in 2002 [6-10]. MTDH severs as critical molecular in a series of human signal pathways [11-13]. MTDH expression can be triggered by Ha-Ras to active the Phosphoinositide 3-kinase (PI3K)/AKT serine/threonine kinase (Akt) pathway [11]. As a result, c-Myc, a transcription factor, is recruited to the E-box element in the promoter region of MTDH to regulate MTDH transcription [11]. It was found MTDH overexpression reversed the chemoresistance of trastuzumab in HER2 positive breast cancer by inhibiting the expression of phosphatase and tensin homolog deleted on chromosome ten (PTEN) through the Nuclear factor-Kappa B (NF-κB) pathway [12]. Moreover, the progression of chronic lymphocytic leukemia (CLL) is stimulated by MTDH through the Wnt/β-catenin pathway, indicating MTDH may be a treatment target for CLL [13]. Furthermore, expression of MTDH in multiple human cancers was also found negatively regulated by miRNAs [7-10]. However, the function of MTDH involved in the miRNA regulatory network is still poorly understood in glioma [8,14]. For instance, it was demonstrated that the tumor suppressive role of miR-379 was exerted through inhibiting MTDH expression by the PTEN/Akt pathway [8]. Recently, it was shown that miR-30b-5p regulates the cell behaviors of glioma through targeting MTDH and revealed the importance of miR-30b-5p in glioma [14].
miR-1471 is a newly identified miRNA that reported to function as a crucial role in the progression of breast cancer [15]. However, whether or not miR-1471 has a connection with MTDH in glioma remains unclear. In this study, we showed miR-1471 expression was significantly downregulated in glioma tissues and cell lines compared with the noncancerous tissues and normal cell line. Further in vitro functional analyses showed that miR-1471 modulates glioma cell proliferation and invasion through targeting MTDH expression. Our study suggested a novel therapeutic target for the treatment of glioma.
Materials and methods
Human glioma tissues
Tissues were collected from 30 patients who underwent treatment at Renmin Hospital of Wuhan University. These tissues were snap-frozen in liquid nitrogen and then stored at -80°C. Written informed consent was obtained from all the participants. The study protocol was approved by the ethic committee of Renmin Hospital of Wuhan University. The tumor stage of these recruited patients was diagnosed based on the World Health Organization (WHO) stage and grading system [16]. The clinicopathological parameters of these 30 patients were collected and the association with miR-1471 expression was summarized in Table 1.
Table 1.
Correlations of miR-1471 and clinicopathologic features in glioma patients
| Clinicopathologic features | No. | Low miR-1471 (n = 17) | High miR-1471 (n = 13) | P value |
|---|---|---|---|---|
| Age | ||||
| ≥ 60 | 17 | 10 | 7 | 0.228 |
| < 60 | 13 | 7 | 6 | |
| Gender | ||||
| Male | 16 | 8 | 8 | 0.167 |
| Female | 14 | 9 | 5 | |
| KPS | ||||
| ≥ 90 | 10 | 5 | 5 | 0.022 |
| < 90 | 20 | 12 | 8 | |
| WHO grade | ||||
| I-II | 10 | 6 | 4 | 0.025 |
| III | 20 | 11 | 9 | |
miR-1471: microRNA-1471; KPS: Karnofsky performance scale; WHO: World Health Organization.
Cell culture
Glioma cell lines U251, U87, SNB19, A172 and normal human astrocytes (NHAs) were purchased from the Cell Bank of Shanghai Life Academy of Science (Shanghai, China). These cells were maintained in Dulbecco’s Modified Eagle Medium (DMEM, Thermo Fisher Scientific, Inc., Waltham, MA, USA) supplemented with 100 U/ml penicillin, 100 μg/ml streptomycin (Thermo Fisher Scientific, Inc.) and 10% fetal bovine serum (FBS, Invitrogen, Thermo Fisher Scientific, Inc.) at a humidified incubator containing 5% of CO2 at 37°C.
Cell transfection was conducted using Lipofectamine 2000 (Invitrogen, Thermo Fisher Scientific, Inc.) in follow with the manufacturer’s instructions. The synthetic miRNAs including miR-1471 mimic, miR-1471 inhibitor, and scramble miRNA (miR-NC) purchased from RiboBio Inc. (Guangzhou, China) were used to manipulate the expression levels of miR-1471. pcDNA3.1-MTDH construct purchased from GenScript (Nanjing, China) was used to overexpress MTDH. Cells were incubated to 70% confluence and mixed with miRNAs (100 nM) or MTDH overexpression construct (100 nM) for 48 h. Transfection efficiency was measured by quantitative real-time PCR (qRT-PCR) or western blot.
RNA isolation and qRT-PCR
RNA samples from the tissue samples and cell lines were extracted using Trizol reagent (Invitrogen, Thermo Fisher Scientific, Inc.) in line with the standard protocols. cDNA was synthesized using BeyoRTTM cDNA First-strand synthesis kit (Beyotime, Haimen, Jiangsu, China). qRT-PCR was conducted in an ABI 7500 Real-Time PCR system (Applied Biosystems, Foster City, CA, USA) using BeyoFastTM SYBR Green qPCR Mix (Beyotime). U6 small nuclear RNA (U6 snRNA) was used as internal control. Relative miR-1471 levels were calculated using the 2-ΔΔCt method. The sequence of primers used in this study was as follows: miR-1471: F-5’-TGGACCCTGGTCTACTCCTG-3’, R-5’-CAAGAGCCCCTGTACAGCAT-3’; U6 snRNA: F-5’-CTCGCTTCGGCAGCACA-3’, R-5’-AACGCTTCACGAATTTGCGT-3’.
Western blot
Protein samples from the tissue samples and cell lines were extracted using RIPA lysis buffer supplemented with phenylmethylsulfonyl fluoride (Beyotime) according to the manufacturer’s instructions. Protein concentration was analyzed using bicinchoninic acid (BCA) kit (Beyotime). These samples were separated using 10% SDS-PAGE and transferred onto nitrocellulose membranes. Then, the membranes were blocked with 5% fat-free milk and incubated with primary antibodies of MTDH (ab227981, Abcam, Cambridge, MA, USA) and GAPDH (ab181602, Abcam). After washing with TBST, the membranes were incubated with secondary antibody (ab205718, Abcam). Protein bands were visualized using BeyoECL Plus Kit (Beyotime) and quantified by Quantity One v4.62 software (Bio-Rad, Hercules, CA, USA).
Luciferase assay
The miR-1471 binding site located at 4019-4025 of MTDH 3’-UTR as predicted by TargetScan. The wild-type (wt) or mutant (mut) of MTDH 3’-UTR was cloned into a psiCHECK-2 vector (Promega, Madison, WI, USA) to generate MTDH-wt or MTDH-mut construct. For luciferase reporter assay, cells were co-transfected with MTDH-wt or MTDH-mut and miR-1471 mimic or miR-NC using Lipofectamine 2000. After 48 h of transfection, luciferase activity was measured using the dual luciferase reporter assay kit (Promega) according to the manufacturer’s protocol.
Proliferation assay
Cell proliferation assay was conducted using Cell counting kit-8 (CCK-8, Beyotime) according to manufacturer’s instruction. Briefly, cells were incubated in the 96-well plate at the density of 1 × 104 cells/well. Then, 10 μl CCK-8 reagent was added to the media at 0, 24, 48, and 72 h after transfection and further incubated at 37°C for 2 h. The optical density was measured at 450 nm using a micro-plate analyzer (Bio-Rad).
Invasion assay
Cell invasion assay was performed using Transwell invasion assay. The cells (5 × 104 cells) to be investigated were added to the upper chamber in FBS-free DMEM, while the DMEM supplemented with FBS was added to the lower chamber. After incubation for 48 h, the noninvasive cells were scraped and washed with PBS for three times. Then, the membranes were fixed with 4% paraformaldehyde and stained with 0.1% crystal violet (Beyotime). The numbers of invasive cells were counted from 5 independent fields.
Statistical analysis
Data were presented as mean ± SD and analyzed using GraphPad Prism 6 (GraphPad Software Inc., San Diego, CA, USA). Student’s t-test or one-way ANOVA and Tukey post-hoc test was used to analyze difference in two or above groups. The association between miR-1471 and clinicopathological parameters was analyzed by chi-square test. Correlations between miR-1471 and MTDH levels were analyzed by Pearson’s correlation coefficient. Statistical difference was considered as significant when P < 0.05.
Results
miR-1471 was downregulated in glioma
We first examined the levels of miR-1471 in four glioma cell lines (U251, U87, SNB19, A172) and NHAs cell line. These glioma cell lines displayed dramatically decreased levels of miR-1471 compared with NHAs cell line (Figure 1A). Levels of miR-1471 in four glioma cell lines in descending order were: SNB19, A172, U87, and U251 (Figure 1A). Therefore, the U87 and U251 cell lines were used for the following functional studies. Moreover, downregulation of miR-1471 was also found in clinical tumor samples (Figure 1B). We also found low miR-1471 expression was closely associated with WHO grade (P = 0.025) and Karnofsky performance scale (KPS, P = 0.022) but not associated with age and gender (P > 0.05, Table 1). These results demonstrated that miR-1471 was downregulated in glioma tissues and cell lines and low miR-1471 expression was correlated with the grade of glioma.
Figure 1.

Downregulation of miR-1471 in glioma cell lines and tissues. A. qRT-PCR analysis of miR-1471 expression in glioma cell lines and NHAs cell line. B. qRT-PCR analysis of miR-1471 expression in glioma tissues and normal tissues. (**P < 0.01, ***P < 0.001) miR-1471: microRNA-1471; qRT-PCR: quantitative real-time PCR.
Overexpression of miR-1471 inhibits glioma cell proliferation and invasion
We wanted to investigate the biological roles of miR-1471 in glioma since it was found downregulated in glioma cell lines. The synthetic miRNAs were transfected into U87 and U251 cell lines and the qRT-PCR analysis results showed that miR-1471 levels were significantly increased by miR-1471 mimic but decreased by miR-1471 inhibition compared with the miR-NC (Figure 2A). CCK-8 assay showed that cell proliferation in U87 and U251 cell lines was enhanced by miR-1471 inhibitor but decreased by miR-1471 mimic (Figure 2B). Moreover, ectopic expression of miR-1471 could inhibit the invasion of U87 and U251 cell lines (Figure 2C). These results showed that miR-1471 overexpression inhibits glioma cell proliferation and invasion.
Figure 2.
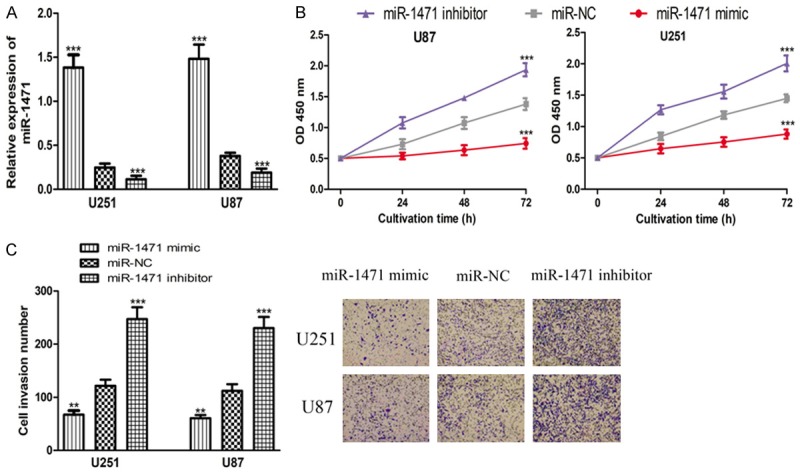
Effects of miR-1471 on glioma cell proliferation and invasion. (A) qRT-PCR analysis of miR-1471 expression, (B) Cell proliferation and (C) Cell invasion in U251 and U87 cells transfected with miR-1471 mimic, inhibitor, or miR-NC. (**P < 0.01, ***P < 0.001) miR-1471: microRNA-1471; qRT-PCR: quantitative real-time PCR; miR-NC: negative control miRNA.
MTDH was a direct target of miR-1471
We investigated the potential target of miR-1471 by TargetScan. MTDH attracted our attention as it contains a putative binding site for miR-1471 (Figure 3A). To investigate whether miR-1471 could bind to the 3’-UTR of MTDH, a luciferase reporter assay was conducted. It was found miR-1471 mimic could impair the luciferase activity of cells transfected with MTDH-wt but not MTDH-mut (Figure 3B). The western blot analysis results revealed that MTDH protein levels were significantly reduced by miR-1471 mimic in U87 and U251 cell lines (Figure 3C). Correlation analysis showed that miR-1471 and MTDH expression levels were inversely correlated (Figure 3D). These results showed that MTDH was a direct target of miR-1471.
Figure 3.

MTDH is a direct target of miR-1471. A. Schematic representation of MTDH 3’-UTRs showing the putative miR-1471 target site. B. Analysis of the relative luciferase activities of MTDH-wt and MTDH-mut. C. Western blot analysis of MTDH expression in U251 and U87 cells transfected with miR-1471 mimic or miR-NC. D. Pearson’s correlation analysis of miR-1471 and MTDH in glioma. (ns not significant, **P < 0.01, ***P < 0.001) miR-1471: microRNA-1471; miR-NC: negative control miRNA; UTR: untranslated region; wt: wild-type; mut: mutant; MTDH: metadherin.
MTDH reversed the effects of miR-1471 on glioma cell proliferation and invasion
In light of the above finding between miR-1471 and MTDH, we examined whether MTDH was a downstream effector for miR-1471 in glioma. Rescue experiments were performed on the U251 cell line. It was found that MTDH construct significantly enhanced the levels of MTDH in U251 cell line and rescued the inhibitory effect of miR-1471 mimic on MTDH (Figure 4A). Then, we performed CCK-8 and transwell invasion assays to evaluate the effects of MTDH overexpression on cell proliferation and invasion. Overexpression of MTDH enhanced glioma cell proliferation and invasion and attenuated the inhibitory effects of miR-1471 mimic (Figure 4B and 4C). These results showed that MTDH was a functional target of miR-1471 in glioma.
Figure 4.

Overexpression of MTDH partially reversed the effects of miR-1471-overexpression on glioma cells. (A) Western blot analysis of MTDH expression, (B) Cell proliferation and (C) Cell invasion in U251 cells transfected with miR-1471 mimic or MTDH construct. (**P < 0.01, ***P < 0.001) miR-1471: microRNA-1471; MTDH: metadherin.
Discussion
The progression of human cancers is characterized as abnormal status of cell proliferation, migration, invasion, apoptosis leading to the disruption of normal cell function [17]. Emerging evidence suggested miRNAs had crucial roles in the progression of human cancers including glioma [7-10,18-20]. Ding et al. examined miR-122 expression in glioma tissues and normal brain tissues and found miR-122 expression was downregulated in glioma tissues compared with the normal brain tissues [18]. They also found miR-122 overexpression could inhibit cell growth and induce apoptosis through regulating the expression of runt-related transcription factors (RUNX2), implicating miR-122/RUNX2 signal might be used as therapeutic targets for glioma [18]. Recently, Gu et al. conducted another set of in vitro experiments showed that miR-384 could inhibit glioma cell malignancy through targeting cell division cycle 42 [19]. miR-1471 was shown to regulate breast cancer cell progression [15]. However, the expression pattern and molecular function of miR-1471 was unclear in glioma.
In this study, we demonstrated the expression of miR-1471 was significantly downregulated in glioma tissues and cell lines. Moreover, our results demonstrated that reduced miR-1471 expression was correlated with advanced WHO grade and low KPS score. However, the effects of miR-1471 on the overall survival of glioma patients warrants further study.
The abnormal expression of miRNAs often resulted in malignant cell behaviors [7-10,18-20]. It was found that miR-1471 levels in glioma cell lines were significantly lower than in normal human astrocytes. To study the effects of miR-1471 expression on glioma cell proliferation and invasion, we upregulated and downregulated miR-1471 expression in glioma cells by miR-1471 mimic and miR-1471 inhibitor. We showed that miR-1471 overexpression suppressed glioma cell proliferation and invasion in vitro. It has been widely recognized that the biologic functions of miRNAs are exerted through regulating the expression of tumor-related genes [7-10,18-20]. Here, we demonstrated MTDH contains a putative binding site for miR-1471 in its 3’-UTR. We further validated MTDH as a direct target gene of miR-1471 using luciferase activity report assay and western blot assay. Additionally, MTDH expression was found negatively correlated with miR-1471 expression in glioma tissues. Finally, overexpression of MTDH stimulates glioma cell proliferation and invasion, which is similar to the effects of miR-1471 inhibitor. Surprisingly, we found MTDH overexpression could partially reversed the effects of miR-1471 mimic on glioma cell behaviors.
In summary, these results demonstrated that miR-1471 functions as a tumor suppressor in glioma through targeting MTDH. Our results provided novel insight into the progression of glioma and proved that the miR-1471/MTDH axis has the potential to be used as effective therapeutic targets for glioma therapy.
Disclosure of conflict of interest
None.
References
- 1.Butowski NA, Sneed PK, Chang SM. Diagnosis and treatment of recurrent high-grade gliomas. J. Clin. Oncol. 2006;24:1273–1280. doi: 10.1200/JCO.2005.04.7522. [DOI] [PubMed] [Google Scholar]
- 2.Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO European Organisation for Research and Treatment of Cancer Brain Tumor and Radiotherapy Groups; National Cancer Institute of Canada Clinical Trials Group. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352:987–996. doi: 10.1056/NEJMoa043330. [DOI] [PubMed] [Google Scholar]
- 3.Liang Y, Diehn M, Watson N, Bollen AW, Aldape KD, Nicholas MK, Lamborn KR, Berger MS, Botstein D, Brown PO, Israel MA. Gene expression profiling reveals molecularly and clinically distinct subtypes of glioblastoma multiforme. Proc Natl Acad Sci U S A. 2005;102:5814–5819. doi: 10.1073/pnas.0402870102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rao JS. Molecular mechanisms of glioma invasiveness: the role of proteases. Nat Rev Cancer. 2003;3:489–501. doi: 10.1038/nrc1121. [DOI] [PubMed] [Google Scholar]
- 5.Emdad L, Das SK, Dasgupta S, Hu B, Sarkar D, Fisher PB. AEG-1/MTDH/LYRIC: signaling pathways, downstream genes, interacting proteins, and regulation of tumor angiogenesis. Adv Cancer Res. 2013;120:75–111. doi: 10.1016/B978-0-12-401676-7.00003-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Su ZZ, Kang DC, Chen Y, Pekarskaya O, Chao W, Volsky DJ, Fisher PB. Identification and cloning of human astrocyte genes displaying elevated expression after infection with HIV-1 or exposure to HIV-1 envelope glycoprotein by rapid subtraction hybridization, RaSH. Oncogene. 2002;21:3592–3602. doi: 10.1038/sj.onc.1205445. [DOI] [PubMed] [Google Scholar]
- 7.Wang Q, Lv L, Li Y, Ji H. MicroRNA-655 suppresses cell proliferation and invasion in oral squamous cell carcinoma by directly targeting metadherin and regulating the PTEN/AKT pathway. Mol Med Rep. 2018;18:3106–3114. doi: 10.3892/mmr.2018.9292. [DOI] [PubMed] [Google Scholar]
- 8.Li L, Zhang H. MicroRNA-379 inhibits cell proliferation and invasion in glioma via targeting metadherin and regulating PTEN/AKT pathway. Mol Med Rep. 2018;17:4049–4056. doi: 10.3892/mmr.2017.8361. [DOI] [PubMed] [Google Scholar]
- 9.Guo T, Pan G. MicroRNA-136 functions as a tumor suppressor in osteosarcoma via regulating metadherin. Cancer Biomark. 2018;22:79–87. doi: 10.3233/CBM-170970. [DOI] [PubMed] [Google Scholar]
- 10.Zhang Y, Wang Y, Wang J. MicroRNA-584 inhibits cell proliferation and invasion in non-small cell lung cancer by directly targeting MTDH. Exp Ther Med. 2018;15:2203–2211. doi: 10.3892/etm.2017.5624. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 11.Lee SG, Su ZZ, Emdad L, Sarkar D, Fisher PB. Astrocyte elevated gene-1 (AEG-1) is a target gene of oncogenic ha-ras requiring phosphatidylinositol 3-kinase and c-Myc. Proc Natl Acad Sci U S A. 2006;103:17390–17395. doi: 10.1073/pnas.0608386103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Du C, Yi X, Liu W, Han T, Liu Z, Ding Z, Zheng Z, Piao Y, Yuan J, Han Y, Xie M, Xie X. MTDH mediates trastuzumab resistance in HER2 positive breast cancer by decreasing PTEN expression through an NFκB-dependent pathway. BMC Cancer. 2014;14:869. doi: 10.1186/1471-2407-14-869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Li PP, Feng LL, Chen N, Ge XL, Lv X, Lu K, Ding M, Yuan D, Wang X. Metadherin contributes to the pathogenesis of chronic lymphocytic leukemia partially through wnt/β-catenin pathway. Med Oncol. 2015;32:479. doi: 10.1007/s12032-014-0479-5. [DOI] [PubMed] [Google Scholar]
- 14.Zhang D, Liu Z, Zheng N, Wu H, Zhang Z, Xu J. MiR-30b-5p modulates glioma cell proliferation by direct targeting MTDH. Saudi J Biol Sci. 2018;25:947–952. doi: 10.1016/j.sjbs.2018.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Liu X, Zhao T, Bai X, Li M, Ren J, Wang M, Xu R, Zhang S, Li H, Hu Y, Xie L, Zhang Y, Yang L, Yan C, Zhang Y. LOC101930370/MiR-1471 axis modulates the hedgehog signaling pathway in breast cancer. Cell Physiol Biochem. 2018;48:1139–1150. doi: 10.1159/000491980. [DOI] [PubMed] [Google Scholar]
- 16.Komori T. The 2016 WHO classification of tumours of the central nervous system: the major points of revision. Neurol Med Chir (Tokyo) 2017;57:301–311. doi: 10.2176/nmc.ra.2017-0010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 18.Ding CQ, Deng WS, Yin XF, Ding XD. MiR-122 inhibits cell proliferation and induces apoptosis by targeting runt-related transcription factors 2 in human glioma. Eur Rev Med Pharmacol Sci. 2018;22:4925–4933. doi: 10.26355/eurrev_201808_15631. [DOI] [PubMed] [Google Scholar]
- 19.Gu G, Wang L, Zhang J, Wang H, Tan T, Zhang G. MicroRNA-384 inhibits proliferation migration and invasion of glioma by targeting at CDC42. Onco Targets Ther. 2018;11:4075–4085. doi: 10.2147/OTT.S166747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lu K, Shen H, Zhu S, Bi S, Wu S. Effects of miRNA-130a on the proliferation and apoptosis of glioma cell lines. Oncol Lett. 2018;16:2478–2482. doi: 10.3892/ol.2018.8878. [DOI] [PMC free article] [PubMed] [Google Scholar]


