Abstract
Pneumococcal Surface Protein A (PspA) has been successfully tested as vaccine candidate against Streptococcus pneumoniae infections. Vaccines able to induce PspA-specific antibodies and Th1 cytokines usually provide protection in mice. We have shown that the whole cell pertussis vaccine (wP) or components from acellular pertussis vaccines, such as Pertussis Toxin or Filamentous Hemagglutinin (FHA), are good adjuvants to PspA, suggesting that combined pertussis-PspA vaccines would be interesting strategies against the two infections. Here, we evaluated the potential of wP as a delivery vector to PspA. Bordetella pertussis strains producing a PspA from clade 4 (PspA4Pro) fused to the N-terminal region of FHA (Fha44) were constructed and inactivated with formaldehyde for the production of wPPspA4Pro. Subcutaneous immunization of mice with wPPspA4Pro induced low levels of anti-PspA4 IgG, even after 3 doses, and did not protect against a lethal pneumococcal challenge. Prime-boost strategies using wPPspA4Pro and PspA4Pro showed that there was no advantage in using the wPPspA4Pro vaccine. Immunization of mice with purified PspA4Pro induced higher levels of antibodies and protection against pneumococcal infection than the prime-boost strategies. Finally, purified Fha44:PspA4Pro induced high levels of anti-PspA4Pro IgG, but no protection, suggesting that the antibodies induced by the fusion protein were not directed to protective epitopes.
Introduction
Lower respiratory infections are among the most important causes of death globally, affecting more than two million people from all ages in 2016 [1]. Streptococcus pneumoniae (pneumococci) is the most frequent etiological agent, contributing with more than 1 million deaths in all ages and around 350 thousand in children under 5 years of age [1]. After almost 20 years of use, pneumococcal conjugate vaccines, composed of polysaccharides from prevalent pneumococcal serotypes conjugated to protein carriers, have greatly contributed to reductions in pneumococcal colonization and invasive diseases around the world [2,3]. However, increase in diseases caused by non-vaccine serotypes were observed in several countries and may affect vaccine efficacy against pneumococcal diseases in different populations [4]. Pneumococcal proteins are alternatives for the development of vaccines with broad-serotype coverage [5]. Pneumococcal Surface Protein A (PspA) is a virulence factor that helps bacteria to escape the immune system by interfering with complement deposition on its surface [6] and with the bactericidal activity of the host apolactoferrin [7].
We have previously shown that the whole cell pertussis vaccine (wP) is a potent adjuvant to PspA, able to enhance the induction of specific antibodies and protection against invasive pneumococcal infection and nasal colonization in animal models [8,9]. Besides the whole bacteria, Pertussis toxin (PT) and Filamentous hemagglutinin (FHA) can also exert adjuvant activity when combined to PspA [10] leading us to propose that combined pertussis- PspA vaccines could be interesting approaches to immunize against infections caused by both Bordetella pertussis and pneumococci.
Instituto Butantan, São Paulo, Brazil, produces the wP vaccine that is administered as DTwP (triple Diphtheria, Tetanus, Pertussis vaccine) to Brazilian children since 1980. Here, we have tested the potential of wP as a delivery system for PspA, by the construction of a recombinant B. pertussis expressing a fusion of PspA4Pro (the N-terminal region of PspA from clade 4 including a proline rich sequence) with the N-terminal fragment of FHA (Fha44:PspA4Pro), using the Brazilian vaccine strain. Mice were immunized with wPPspA4Pro and the induction of anti-PspA antibodies, as well as protection against pneumococcal infection were analyzed. The potential of purified recombinant Fha44:PspA4Pro protein as a vaccine candidate was also tested.
Materials and methods
Ethics statement
This study was performed according to the guidelines outlined by the Brazilian National Council for Control of Animal Experimentation (CONCEA). Experimental protocols were approved by the Ethic Committee on Animal Use of the Butantan Institute (CEUAIB) (protocol numbers 1363/15 and 3154200117). Six animals were housed per cage inside a ventilated cabinet under controlled temperature and light cycle (12/12 hours, light/dark cycle) with daily monitoring in a BSL2 animal facility. Food and water were given ad libitum. Monitoring and manipulation was done by trained personnel.
Bacteria and plasmids construction
B. pertussis NIH137[11] is the vaccine strain used for the production of wP at Instituto Butantan, São Paulo, Brazil and was used in this work. B. pertussis 18323 (used in the potency tests for of wP vaccines at Instituto Butantan) was used for the production of B. pertussis protein lysates. Bacteria were grown on Bordet-Gengou (BG–Difco, New Jersey, USA) agar plates, supplemented with 1% glycerol and 20% defibrinated sheep blood, at 35°C. Nalidixic acid (Nal, 30 μg/mL) or gentamycin (Gm,10 μg/mL) were added to the BG-blood agar plates when required. Escherichia coli DH5α SM-10 λ Pir or BL21 (DE3) star pLysS were grown in Luria Bertani (LB—Difco), supplemented with 10μg/mL Gm or 100 μg/mL ampicillin for the selection of plasmid-containing clones. S. pneumoniae ATCC6303 (serotype 3) was grown on Triptic soy agar containing 5% defibrinated sheep blood (Laborclin, SP, Brazil) at 37°C. Stocks for challenge were prepared in liquid Todd-Hewitt media (Difco) supplemented with 0.5% yeast extract (THY). Bacteria were grown until exponential phase (OD600nm 0.4), centrifuged, and suspended in 1:10 of the initial volume in THY containing 40% glycerol. Stocks were maintained at -80°C and quantified by plating on blood agar. The plasmid pXR1Fha44 [12], which confers resistance to Gm, was used for the expression of PspA4 in B. pertussis NIH137. The sequence coding for the N-terminal region of PspA from clade 4, containing a proline block (PspA4Pro) was amplified by PCR from the pAE-PspA4Pro plasmid [13] using the oligonucleotides 5’ ACGCGTGTAAGAGCAGAAGAAGCC 3’ and 5’ CATATGTGGTTTTGGTGCTGGAGC 3’.The fragment was inserted between the MluI and NdeI restriction sites of pXR1Fha44, yielding a plasmid that contains a hybrid gene coding for the N-terminal region of filamentous hemagglutinin (Fha44) fused to PspA4Pro. For the expression of the recombinant protein in E. coli, a PCR for the amplification of the fragment coding for the Fha44:PspA4Pro excluding the fha signal peptide was performed using the oligonucleotides 5’ GGATCCCAAGGTCTGGTTCCGCAG 3’ and 5’ AAGCTTCGAATTCCTAGGTACCTTATGGTTTTGGT 3’. The sequence was inserted between the BamHI and the HindIII restriction sites in the pAE vector [14], yielding a plasmid that codes for Fha44:PspA4Pro fused to a histidine tag.
Expression of FHA44:PspA4Pro in B. pertussis and E. coli
Conjugation of B. pertussis NIH137 and E. coli SM-10 λ pir was performed for the generation of B. pertussis clones producing Fha44:PspA4Pro (BpPspA4Pro). Briefly, B. pertussis grown on BG-blood agar was harvested and plated onto a fresh BG-blood agar plate. One colony of SM-10 λ pir transformed with the pXR1Fha44:PspA4Pro plasmid was mixed with B. pertussis on the plate and incubated at 35°C for 6h. Bacteria were then collected and plated onto BG-blood agar containing Gm and Nal for the selection of recombinant B. pertussis and elimination of E. coli. Clones able to grow on Gm were selected once again on Nal to confirm proper selection. Stocks of the recombinant clones were maintained at -80°C and expression was evaluated by western-blot of protein extracts using anti-PspA4 or anti-FHA polyclonal antisera (produced at the laboratory) and Amersham ECL Prime detection reagent (GE Healthcare).
Purified recombinant Fha44:PspA4Pro proteins were obtained from E. coli BL21 Star (DE3) pLysS transformed with the pAE-Fha44:PspA4Pro plasmid as follows. Recombinant bacteria were grown in LB-amp until OD600nm = 0.5 and expression of the protein was induced by the addition of 1.2mM IPTG for 3h. Protein expression was confirmed by SDS-PAGE. Bacteria were harvested by centrifugation and lysates were prepared in a PANDA homogenizer (GEA Niro Soavi, Parma, Italy). The Fha44:PspA4Pro protein was detected in inclusion bodies that were dissolved in 50mM Tris (pH 8.0), 150mM NaCl, 5 mM imidazole, 8M urea. Purification of the recombinant protein was performed through affinity chromatography using 5mL His Trap columns (GE Healthcare, Illinois, USA) in the AKTA Prime equipment (GE Healthcare). After the adsorption of the protein to the column, washing steps with increasing concentrations of imidazole were performed and protein was eluted in 250mM imidazole. 8 M urea was maintained during the entire process and the purified Fha44:PspA4Pro was also stored in 8 M urea, since efforts to remove it by dilution or dialysis resulted in the precipitation of the protein. Purified PspA4Pro [15] was kindly provided by Dr. Viviane Maimoni Gonçalves, Instituto Butantan, São Paulo, Brazil. SDS-PAGE and Western blots images were produced using the KodaK Gel Logic 200 equipment with the Carestream software except for S2 Fig. that was produced using the Amersham Image 680 equipment.
Production of wPPspAPro vaccines and immunization of mice
B. pertussis producing Fha44:PspA4Pro was grown on BG-blood agar at 35°C. Bacteria were suspended in non-pyrogenic PBS (Gibco) to an OD600nm = 1.8 and washed once in PBS. Pellets were suspended in 1/10 of the initial volume of PBS and aliquots were used for quantification by plating on BG-agar plates containing Gm. Samples were centrifuged, suspended in the same volume of PBS containing 0.2% formaldehyde and incubated at 35°C for 24h under agitation. Bacteria were then washed once in PBS and suspended in the same volume of PBS. This protocol was based on that used for the production of wP at Instituto Butantan. Non-recombinant B. pertussis NIH137 was subjected to the same protocol as a control. wP and wPPspA4Pro vaccines were maintained at 4°C until use.
Female SPF BALB/c mice were produced by the animal facility from the Medical School, University of São Paulo. Animals (6 per group) were supplied with food and water ad libitum. Mice were immunized through the subcutaneous route with the equivalent of 1x109 CFU of B. pertussis, in a total volume of 100μL, with booster doses given at 15-days intervals, up to three doses. In prime-boost protocols, mice received the same dose of bacteria,1μg of PspA4Pro or saline through the subcutaneous route, followed by two boosters with saline or PspA4Pro. For the experiments with purified recombinant Fha44:PspA4Pro, mice were immunized twice subcutaneously at a 15-day interval with 1μg of protein. FHA (Sigma-Aldrich, Missouri, USA) and PspA4Pro were used as controls. Blood was collected 14 days after each immunization through the retroorbital plexus, under local anesthesia with 5% proxymetacaine chloride eye drops (Alcon, Texas, USA). Blood was incubated at 37°C for 30 min, following by incubation at 4°C for 10 min. Samples were centrifuged at 800 x g for 10 min for the collection of sera. Induction of IgG, IgG1 and IgG2a was evaluated by ELISA using plates coated with 1μg/mL of PspA4Pro or protein lysates from B. pertussis 18323. Sera collected from immunized mice were tested in serial dilutions and Horseradish Peroxidase anti-mice IgG, IgG1 or IgG2a (Southern Biotech, Alabama, USA) were used as secondary antibodies. Reactions were developed using o-Phenylenediamine dihydrochloride as substrate and the absorbances were measured at 492nm in a spectrophotometer multiskan EX (Thermo Fisher Scientific, Massachusetts, USA). Antibody titers were defined as the reciprocal of the dilution that produced an absorbance of 0.1.
Antibody binding and complement deposition to pneumococcal surface
S. pneumoniae ATCC6303 was grown on blood agar plates for 18h. Bacteria were diluted in THY, grown until OD600 nm = 0.4–0.5 (~108 CFU/mL) and harvested by centrifugation at 3,200 x g for 10 min. Bacteria were then washed, suspended in PBS and incubated with 5% (V/V) of sera from immunized mice (pooled sera from each experimental group) during 30 min on ice. Samples were then washed once with PBS before incubation for 30 min with fluorescein isothiocyanate (FITC)-conjugated goat anti-mouse IgG (MP Biomedicals, California, USA), diluted 1:100 in PBS. For complement deposition assays, sera were previously inactivated at 56°C for 30 min and incubated with bacteria at a concentration of 10 or 20%, for 30 min on ice. Samples were washed once with PBS and incubated with 10% of normal mouse serum as source of complement, in Gelatin Veronal buffer (Sigma Aldrich), at 37 ºC for 30 min. After washing, samples were incubated with FITC-conjugated anti-mouse C3 IgG (MP Biomedicals) in PBS, for 30 min on ice. In both experiments, samples were fixed with 200 μL of cytofix (BD Biosciences, California, USA) after two washing steps. Flow cytometry analysis was conducted using FACS Canto II (BD Biosciences), and 15,000 gated events were recorded. Fluorescence was analyzed by histograms using the Flow Jo 10.1 software and medians of the curves were used to compare the groups.
Intranasal Pneumococcal challenge
Immunized mice were challenged with S. pneumoniae ATCC6303 by the intranasal inoculation of 3X105 bacteria in 50μL saline, 21 days after the last immunization. Challenge was performed under anesthesia, using 100 mg/Kg of ketamine chloride (Ceva, São Paulo, Brazil) and 20mg/Kg of xylazine chloride (Ceva) intraperitoneally and survival was followed for up to 15 days. Animals were monitored twice daily after challenge and lethargic animals with reduced ability to move were euthanized immediately through the ip route with a lethal dose of a xylazine/ketamine solution (60 mg/Kg of xylazine and 300 mg/Kg of ketamine). No animals died before meeting criteria for euthanasia.
Statistical analyses
Differences in antibody concentrations were analyzed by One-way ANOVA followed by Tukey´s post-test for the comparisons between two groups. Survival was analyzed by the Log-rank survival curve using the Mantel-Cox test for the comparison between groups. Statistical analyses were performed using Prism 6.0 software and P≤0.05 was considered significantly different.
Results
Recombinant B. pertussis produces Fha44:PspA4Pro
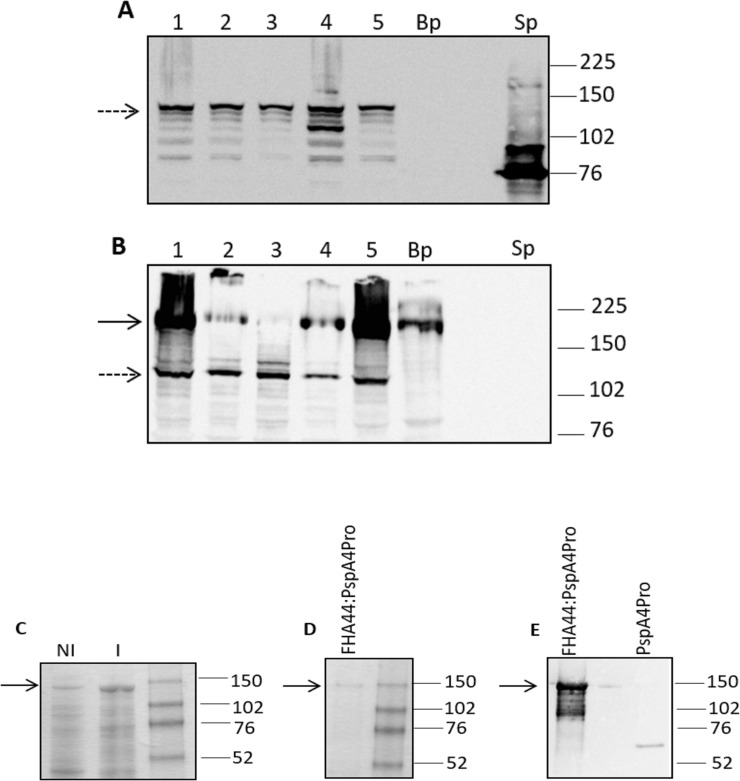
B. pertussis NIH137 was conjugated with E. coli SM-10 λ Pir (pXR1Fha44:PspA4Pro) to generate clones producing Fha44:PspA4Pro (BpPspA4Pro). In three independent experiments, several clones were selected with expression of the protein of interest. Western blot analysis using anti-PspA4 antisera revealed a band of 130kDa, the expected size for Fha44:PspA4Pro, in 5 representative clones of BpPspA4Pro and no reactivity with extracts from non-recombinant B. pertussis NIH137 (Bp) (Fig 1A). In addition, lower-Mr bands reacting with the anti-PspA4 antisera were observed, suggesting partial degradation. The same clones were also examined using anti-FHA antisera. As shown in Fig 1B, the anti-FHA antisera revealed a band of around 220 kDa corresponding to full-length FHA produced by BPPspA4Pro as well as by Bp, albeit with variable intensity. The 130kDa band, corresponding to Fha44:PspA4Pro, was only observed on BpPspA4Pro. In order to evaluate whether the fusion with Fha44 had driven PspA4Pro to the surface of the bacteria, flow cytometry on two BpPspA4Pro clones and Bp NIH137 was performed, using anti-FHA and anti-PspA4 antisera. S1 Fig shows positive reactivity of all bacteria with anti-FHA antibodies and low but positive reactivity of the BpPspA4Pro clones with the anti-PspA4 antisera, indicating that at least part of the Fha44:PspA4Pro protein is exposed on the surface of the recombinant bacteria.
Fig 1. Expression of FHA44:PspA4Pro.
(A and B) Lysates of 5 recombinant B. pertussis clones or non-recombinant B. pertussis (Bp) were evaluated by western-blot using anti-PspA (A) or anti-FHA antisera (B). The 255/00 pneumococcal strain lysate was used as a PspA4 control (Sp). (C and D) FHA44:PspA4Pro was also expressed in E. coli, NI, lysate from a representative clone in non-induced condition; I, lysate from the same clone after induction with IPTG (C) and purified by affinity chromatography (D). Identification of the purified recombinant protein was performed by western-blot using anti-PspA4 antisera (E). In (C), 15 μg of total protein were applied to the gel; In D and E, 0.5 μg of purified protein was applied to the gel. In all figures the GE Healthcare rainbow full range molecular weight marker was used.
Using the pAE vector, expression of Fha44:PspA4Pro was also observed in recombinant E. coli after induction with IPTG (Fig 1C). The purified protein obtained by Ni2+- affinity chromatography (Fig 1D) was also detected by the anti-PspA4 antisera (Fig 1E).
wPPspA4Pro vaccines induce low levels of anti-PspA4 antibodies and are not protective against pneumococcal infection
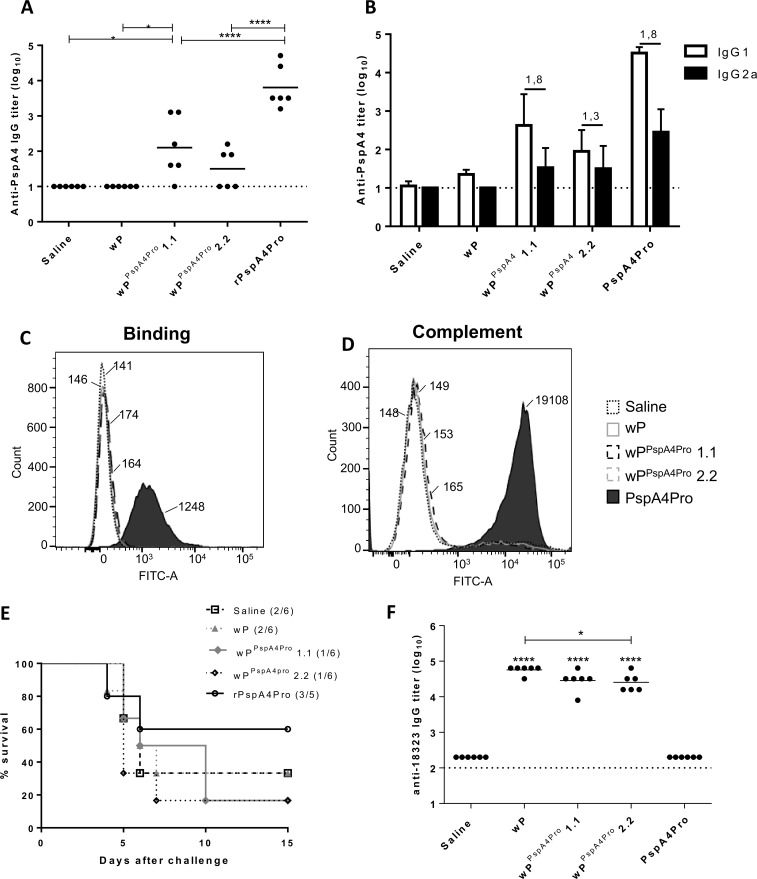
Selected clones were used for the production of inactivated wPPspA4Pro vaccines. Mice received three doses of the inactivated vaccines wpPspA4Pro or wP (equivalent of 1x109 CFU of B. pertussis per dose). An estimative made by comparative western-blot analysis, based on a curve with purified Fha44:PspA4Pro, indicated that around 10ng of the protein was present in each dose (S2 Fig). Purified PspA4Pro was used as a positive control in the immunization experiment. Even after three doses, only low levels of anti-PspA4Pro IgG and highly variable responses among the animals were observed in the groups of mice immunized with wPPspA4Pro, produced from two different clones (Fig 2A). In contrast, high levels of anti-PspA4Pro IgG were observed in mice immunized with three doses of the recombinant protein. Characterization of the IgG subtypes revealed balanced IgG1:IgG2a ratios in the immunized groups, with no significant differences observed between animal vaccinated with wPPspA4Pro or PspA4Pro (Fig 2B).
Fig 2. wPPspA4Pro induces low levels of anti-PspA4 IgG.
BALB/c mice were immunized with three doses of wPPspA4Pro, wP or PspA4Pro. (A, B and F) Anti-PspA4Pro IgG (A), IgG1 and IgG2a (B) or anti-B. pertussis 18323 protein lysates (F) were measured by ELISA. (A and E), circles represent individual data and lines represent the means for each group. (B), bars represent the means for each group with the standard deviations; numbers above the bars indicate the IgG1:IgG2a ratios. Titers in control groups were below the limit of detection and, therefore IgG1:IgG2a ratios were not calculated for them. (C and D) Pools of sera from each group of mice were evaluated for the capacity to bind (C) or to induce complement deposition (D) on the surface of S. pneumoniae ATCC6303 by flow cytometry. Numbers indicate the medians of fluorescence intensity for each curve. (E) Intranasal challenge with S. pneumoniae ATCC6303 was performed in immunized mice and survival was followed for 15 days. *P<0.05; ****P<0.0001, One-way ANOVA with Tukey post-test or Log-Rank survival curve with Mantel-Cox test (E).
Reactivity of the antisera with native PspA was tested in vitro by binding assays to the surface of S. pneumoniae ATCC6303. Only the sera from mice immunized with PspA4Pro were able to recognize the protein on the pneumococcal surface (median of fluorescence intensity of 1248, Fig 2C). Sera from mice immunized with wPPspA4Pro vaccines produced from two different clones, showed reactivity at the same levels of the sera obtained from mice immunized with saline or the wP vaccine (with medians of fluorescence intensity below 200), probably reflecting the low levels of anti-PspA4Pro IgG induced by these vaccines. In addition, only the sera from mice immunized with PspA4Pro induced significant deposition of complement C3 on pneumococcal surface (median of fluorescence intensity of 19108, Fig 2D).
Immunized mice were challenged with S. pneumoniae ATCC6303 by the intranasal route and only PspA4Pro was able to induce 60% survival of mice, a level usually observed by our group when the recombinant protein is administered to mice without any adjuvant [8]. No improvement in survival of mice immunized with the wPPspA4Pro vaccines was observed when compared with the controls immunized with wP or saline (Fig 2E).
We then analyzed the induction of antibodies against B. pertussis antigens by the wPPspA4Pro vaccine. Sera collected from mice after the second dose showed high levels of IgG reacting with total protein extracts from B. pertussis 18323. No differences in the levels of antibodies were observed when comparing wP with wPPspA4Pro vaccines (Fig 2F). As expected, purified recombinant PspA4Pro did not induce any anti-B. pertussis antibodies. Thus, expression of PspA4Pro did not affect the response to B. pertussis antigens and the vaccine formulations were immunogenic in mice.
Prime-boost protocols using wPPspAPro and PspA4Pro do not protect mice against pneumococcal respiratory infection
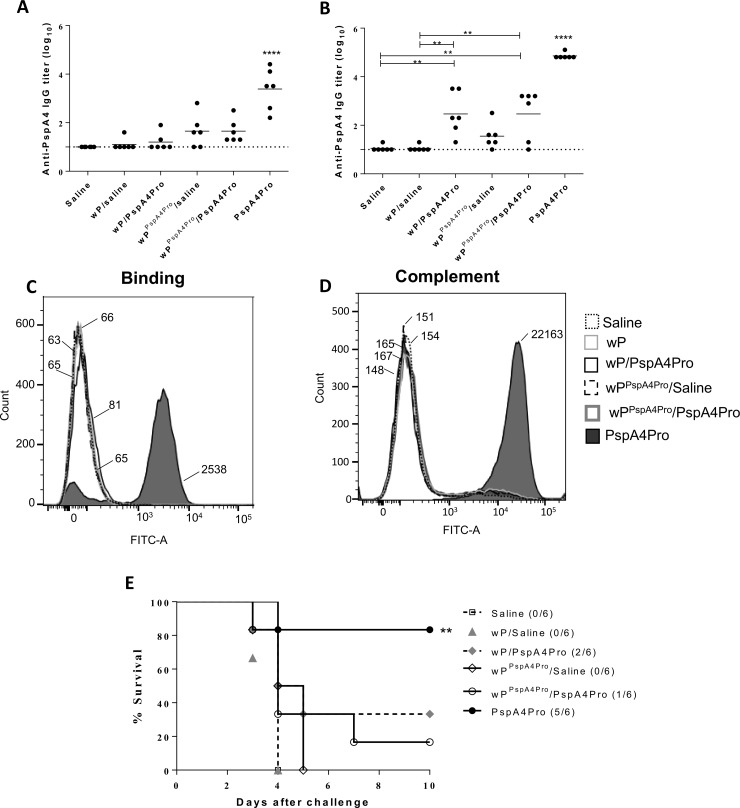
Since immunization with wPPspA4Pro vaccines did not induce high levels of antibodies nor did they protect mice against invasive pneumococcal infection, we tested prime-boost protocols using recombinant PspA4Pro as boost, to evaluate possible benefits on the modulation of the immune system by the inactivated vaccine. Mice received wPPspA4Pro as the priming vaccine and saline or PspA4Pro in the booster vaccine. As controls, mice were primed with wP or PspA4Pro. As shown in Fig 3A, the prime-boost regimen using wPPspA4Pro followed by one dose of PspA4Pro induced low levels of anti-PspA4Pro antibodies, that were comparable to the levels observed in mice immunized with wPPspA4Pro followed by saline or wP followed by PspA4Pro. In contrast, high levels of anti-PspA4Pro IgG were observed in mice immunized with two doses of the recombinant protein. A third dose was given to the animals and, the highest levels of anti-PspA4Pro IgG were again induced in animals immunized with three doses of the recombinant protein (Fig 3B). No differences between the groups of mice primed with wP or wPPspA4Pro were observed, indicating no benefits for the use of wPPspA4Pro. Sera collected after the third dose were tested for binding to the surface of S. pneumoniae ATCC6303. Binding of IgG was only detected in sera from mice immunized with three doses of recombinant PspA4Pro (Fig 3C) and this was the only sera that induced the deposition of complement C3 on bacterial surface (Fig 3D). In addition, this was the only group of mice that were significantly protected against the invasive challenge with S. pneumoniae ATCC6303 (Fig 3E).
Fig 3. Prime-boost protocol with wPPspA4Pro and PspA4Pro does not improve protection against pneumococcal infection.
(A and B) BALB/c mice were immunized with one dose of wP or wPPspA4Pro followed by one (A) or two doses (B) of saline or PspA4Pro. Control mice received only PspA4Pro. Anti-PspA4Pro IgG were measured by ELISA. Circles represent individual data and lines represent the means for each group. (C and D) Pools of sera from each group of mice were evaluated for the capacity to bind (C) or to induce complement deposition (D) on the surface of S. pneumoniae ATCC6303 by flow cytometry. Numbers indicate the medians of fluorescence intensity for each curve. (E) Intranasal challenge with S. pneumoniae ATCC6303 was performed in immunized mice and survival was followed for 15 days. **P<0.01; ****P<0.0001, One-way ANOVA with Tukey post-test (A and B) or Log-Rank survival curve with Mantel-Cox test (E).
Fha44:PspA4Pro induces similar levels of anti-PspA4Pro IgG but no protection against pneumococcal infection
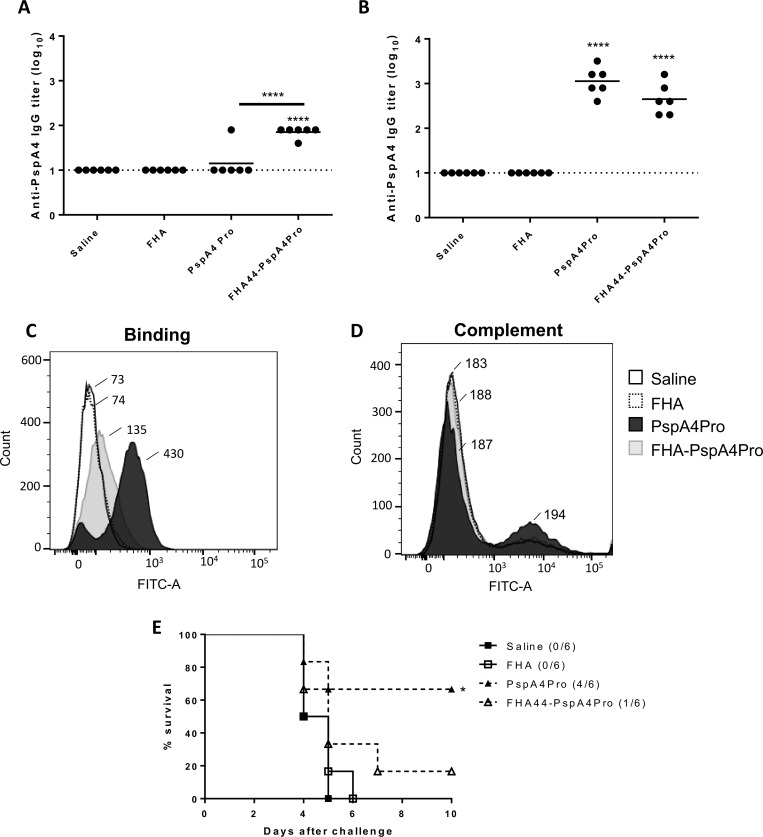
Adjuvant properties have been described for different B. pertussis antigens, including PT and FHA. We therefore tested purified Fha44:PspA4Pro as a vaccine candidate against pneumococcal infection. A single dose of Fha44:PspA4Pro indeed induced higher levels of anti-PspA4Pro IgG compared with PspA4Pro (Fig 4A). However, after one dose neither protein induced antibodies at levels that usually afford protection against pneumococcal invasive infection in mice. After the second dose, the superior response of Fha44:PspA4Pro over PspA4Pro was no longer observed (Fig 4B). Both proteins induced high levels of anti-PspA4Pro IgG, with no significant differences between them. However, IgG induced by PspA4Pro was able to bind more effectively to the S. pneumoniae ATCC6303 surface, compared to the IgG induced by Fha44:PspA4Pro (Fig 4C), although low levels of complement deposition was observed (Fig 4D), probably due to the relative low binding capacity of the sera after only two doses of PspA4Pro. This activity reflected in lower protective capacity of Fha44:PspA4Pro against the pneumococcal infection when compared with PspA4Pro (Fig 4E).
Fig 4. FHA44:PspA4Pro induces anti-PspA4Pro IgG but no protection against pneumococcal infection.
(A and B) BALB/c mice were immunized with one (A) or two (B) doses of FHA44:PspA4. FHA or PspA4Pro were used as negative or positive controls, respectively. Anti-PspA4Pro IgG were measured by ELISA. Circles represent individual data and lines represent the means for each group. Pools of sera from each group of mice were evaluated for the capacity to bind (C) or to induce complement deposition (D) on the surface of S. pneumoniae ATCC6303 by flow cytometry. Numbers indicate the medians of fluorescence intensity for each curve. (E) Intranasal challenge with S. pneumoniae ATCC6303 was performed in immunized mice and survival was followed for 15 days. *P<0.05; ****P<0.0001, One-way ANOVA with Tukey post-test (A and B) or Log-Rank survival curve with Mantel-Cox test (E).
Discussion
Different studies have shown that PspA can induce protective immunity to pneumococcal infection in animal models. The presence of high levels of anti-PspA antibodies allows efficient deposition of complement onto the pneumococcal surface, leading to opsonophagocytosis and clearance of bacteria [6]. Antibodies elicited by the immunization of humans with PspA were able to protect mice against invasive pneumococcal infection in passive immunization experiments [16]. Moreover, the induction of Th1 responses, with IgG2a against PspA, was related to optimal immunity against lethal infection and positively correlated with protection against pneumococcal nasal colonization in mice, with higher levels of anti-PspA IgG2a present in mice with lower bacterial numbers [17,18]. Such responses can be induced by bacterial delivery systems, such as live attenuated Salmonella [19] and lactic acid bacteria [20,21].
B. pertussis has also proved to be an effective live delivery vector for heterologous antigens. Virulent or attenuated B. pertussis strains expressing different antigens have been tested as vaccines against different diseases. Nasal immunization of mice with B. pertussis expressing the Schistosoma mansoni glutathione S-transferase (Sm28GST) fused to FHA induced anti-Sm28GST mucosal and systemic antibodies and protection against parasitic infection [22,23]. Similarly, the Neisseria meningitidis Transferin-Binding Protein B (TbpB) was expressed in B. pertussis fused to the N-terminal domain of FHA. Anti-TbpB antibodies, able to induce complement-mediated killing of N. menigitidis, were induced after single nasal immunization of mice [24]. Live vaccines based on the attenuated B. pertussis strain BPZE1 were also developed against influenza A virus, by the expression of the M2e epitope fused to FHA. Induction of immune responses against M2e and reduction in viral load after a challenge with influenza virus H1N1/PR8 were observed in mice immunized through the nasal route [12,25]. However, vaccination of mice with BPZE1 was shown to confer non-specific protection against influenza A virus and the expression of M2e in the recombinant strain did not improve this effect [25].
Our group has been working on the use of the adjuvant properties of wP for the formulation of PspA-based vaccines. Such combined formulations (wP plus recombinant purified PspA) have proved to be effective when inoculated through the nasal and through the subcutaneous routes in mice [8,9].
In the present work, we examined whether a recombinant inactivated pertussis vaccine would also be an effective system to induce immune responses against PspA. Our choice of this approach was due to the fact that inactivated wP vaccines are used in many countries, including Brazil. If effective, this could be a new proposal for DTwP vaccines, with additional protection against pneumococci.
The recombinant NIH137 vaccine strain was able to express PspA4Pro fused to Fha44, as detected by western-blot. Anti-PspA4 antisera also reacted with intact BpPspA4Pro, indicating that the fusion with Fha44 directed PspA4Pro to the surface of B. pertussis. However, immunization of mice with wPPspA4Pro, produced from various clones, did not induce high levels of anti-PspA4Pro antibodies, nor did it confer protection against lethal pneumococcal infection. Several factors may have contributed to the failure to induce robust immune responses against PspA4Pro. The amount of wP and wPPapA4Pro given to mice was based on the potency tests performed for the approval of wP batches for the formulation of DTwP at Butantan Institute. However, our estimates indicated that the amount of Fha44:PspA4Pro in this formulation was very low. Nevertheless, even after three doses, the induction of protective immunity against pneumococcal infection was not observed. This was an important limitation for the use of this system.
The use of formaldehyde to produce the inactivated vaccines was based on the protocol for the wP production by Butantan Institute. This may have affected the production of antibodies against PspA4 epitopes that are important for protection. The failure to observe antibody binding to pneumococcal surface in the sera from mice immunized with wPPspA4Pro also supports the hypothesis that reactivity with the native protein was low.
The combination of FHA with heterologous antigens for the immunization of mice has been shown to increase the induction of antibodies, supporting its adjuvant properties through the nasal, oral or subcutaneous routes [10,26]. The Fha44 fragment, which contains the heparin-binding domain of FHA was proposed as an alternative to the full-length protein in pertussis vaccines [27]. Immunization of mice with Fha44 induces anti-FHA antibodies and protective immunity against B. pertussis infection in mice. Since it is a smaller and more soluble protein compared to FHA, its purification from a recombinant B. pertussis strain would have advantages over the full-length antigen. Considering these characteristics and the wPPspA4Pro vaccines evaluated in this work, which contain PspA4Pro fused to Fha44, we tested the purified recombinant fusion protein produced in an E. coli expression system. However, fusion of PspA4Pro with Fha44 did not improve the induction of anti-PspA antibodies able to protect against pneumococcal infection. Despite the detection of anti-PspA4Pro IgG in sera from mice immunized with Fha44:PspA4Pro by ELISA, binding to native PspA on the pneumococcal surface was less effective compared with sera from mice immunized with PspA4Pro. Moreover, protection of mice immunized with Fha44:PspA4Pro was not observed. These results suggest that important epitopes were not being recognized by sera from mice immunized with Fha44:PspA4Pro. The failure to produce soluble Fha44:PspA4Pro protein in our expression system may have contributed to these results.
Based on the results presented here and the previous results from our group, combination of wP with PspA is a better strategy for a combined pertussis-pneumococcal vaccine than inactivated vaccines produced from recombinant BpPspA clones. However, the use of live attenuated B. pertussis expressing PspA against pneumococcal infections remains to be tested. An important difference between the use of recombinant live B. pertussis vaccines and wPPspA4Pro vaccines, is the fact that the former are able to colonize the respiratory tract of mice for several days after intranasal inoculation, and, therefore, the availability of the heterologous antigens may be prolonged when compared to vaccination with inactivated vaccines.
Supporting information
Expression of FHA and FHA44:PspA4Pro on the surface of Bp137 and two clones of BpPspA4Pro (1.1 and 2.2) were tested using anti-FHA (A) or anti-PspA4 (B) antisera, by flow cytometry. BpPspA4Pro1.1 without primary sera was used as control. Numbers indicate the medians of fluorescence intensity for each curve.
(TIF)
Lysates from Bp137 (Bp) and two clones of BpPspA4Pro (1.1 and 2.2) corresponding to one dose of wP or wPpspAPro vaccines were compared to a curve established with recombinant Fha44:PspA4. Samples were evaluated by western-blot using anti-PspA4 and bands were analyzed using the Amersham Imager 680 analysis software. MW: GE Healthcare rainbow full range molecular weight marker.
(TIF)
Images were captured with the Kodak GelLogic 200 and the Carestream software. MW, GE Healthcare rainbow full range molecular weight marker.
(PDF)
Images were captured with the Kodak GelLogic 200 and the Carestream software. MW, GE Healthcare rainbow full range molecular weight marker.
(PDF)
Image was captured with the Amersham Imager 680 MW, GE Healthcare rainbow full range molecular weight marker.
(PDF)
Acknowledgments
We are grateful to Dominique Raze for the help with the construction of the pXR1Fha44:PspA4Pro plasmid. We also thank Aline Marques Cavalher, Vera Lúcia de Almeida and Ricardo Alexandre de Souza Silva for the excellent technical support. This work was supported by FAPESP (grant 2016/13134-3), CNPq (301856/2016-8) and Fundação Butantan. Castro JT received a PhD fellowship and Nishigasako MA received an undergraduate fellowship (FAPESP 2016/17258-9 and 2017/01992-8); Oliveira GS received a Msc fellowship from CAPES, Brasil).
Data Availability
All relevant data are within the manuscript and its Supporting Information files.
Funding Statement
This work was supported by FAPESP (grant 2016/13134-3), CNPq (301856/2016-8) and Fundação Butantan. Castro JT received a PhD fellowship and Nishigasako MA received an undergraduate fellowship (FAPESP 2016/17258-9 and 2017/01992-8); Oliveira GS received a Msc fellowship from CAPES, Brasil. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.GBD (2018) Estimates of the global, regional, and national morbidity, mortality, and aetiologies of lower respiratory infections in 195 countries, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Infect Dis 18: 1191–1210. 10.1016/S1473-3099(18)30310-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Yildirim I, Shea KM, Pelton SI (2015) Pneumococcal Disease in the Era of Pneumococcal Conjugate Vaccine. Infect Dis Clin North Am 29: 679–697. 10.1016/j.idc.2015.07.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Davis SM, Deloria-Knoll M, Kassa HT, O'Brien KL (2013) Impact of pneumococcal conjugate vaccines on nasopharyngeal carriage and invasive disease among unvaccinated people: review of evidence on indirect effects. Vaccine 32: 133–145. 10.1016/j.vaccine.2013.05.005 [DOI] [PubMed] [Google Scholar]
- 4.Weinberger DM, Malley R, Lipsitch M (2011) Serotype replacement in disease after pneumococcal vaccination. Lancet 378: 1962–1973. 10.1016/S0140-6736(10)62225-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Miyaji EN, Oliveira ML, Carvalho E, Ho PL (2013) Serotype-independent pneumococcal vaccines. Cell Mol Life Sci 70: 3303–3326. 10.1007/s00018-012-1234-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ren B, Li J, Genschmer K, Hollingshead SK, Briles DE (2012) The absence of PspA or presence of antibody to PspA facilitates the complement-dependent phagocytosis of pneumococci in vitro. Clin Vaccine Immunol 19: 1574–1582. 10.1128/CVI.00393-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Shaper M, Hollingshead SK, Benjamin WH Jr., Briles DE (2004) PspA protects Streptococcus pneumoniae from killing by apolactoferrin, and antibody to PspA enhances killing of pneumococci by apolactoferrin [corrected]. Infect Immun 72: 5031–5040. 10.1128/IAI.72.9.5031-5040.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Oliveira ML, Miyaji EN, Ferreira DM, Moreno AT, Ferreira PC, Lima FA et al. (2010) Combination of pneumococcal surface protein A (PspA) with whole cell pertussis vaccine increases protection against pneumococcal challenge in mice. PLoS One 5: e10863 10.1371/journal.pone.0010863 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tostes RO, Rodrigues TC, da Silva JB, Schanoski AS, Oliveira ML,Miyaji EN. (2017) Protection Elicited by Nasal Immunization with Recombinant Pneumococcal Surface Protein A (rPspA) Adjuvanted with Whole-Cell Pertussis Vaccine (wP) against Co-Colonization of Mice with Streptococcus pneumoniae. PLoS One 12: e0170157 10.1371/journal.pone.0170157 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Salcedo-Rivillas C, Debrie AS, Miyaji EN, Ferreira JM Jr., Raw I, Locht C, et al. (2014) Pertussis toxin improves immune responses to a combined pneumococcal antigen and leads to enhanced protection against Streptococcus pneumoniae. Clin Vaccine Immunol 21: 972–981. 10.1128/CVI.00134-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Akamatsu MA, Nishiyama MY Jr., Morone M, Oliveira UC, Bezerra MF, Sakauchi MA, et al. (2015) Whole-Genome Sequence of a Bordetella pertussis Brazilian Vaccine Strain. Genome Announc 3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kammoun H, Roux X, Raze D, Debrie AS, De Filette M, Ysenbaert T, et al. (2013) Immunogenicity of live attenuated B. pertussis BPZE1 producing the universal influenza vaccine candidate M2e. PLoS One 8: e59198 10.1371/journal.pone.0059198 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Moreno AT, Oliveira ML, Ferreira DM, Ho PL, Darrieux M, Leite LC, et al. (2010) Immunization of mice with single PspA fragments induces antibodies capable of mediating complement deposition on different pneumococcal strains and cross-protection. Clin Vaccine Immunol 17: 439–446. 10.1128/CVI.00430-09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ramos CR, Abreu PA, Nascimento AL, Ho PL (2004) A high-copy T7 Escherichia coli expression vector for the production of recombinant proteins with a minimal N-terminal His-tagged fusion peptide. Braz J Med Biol Res 37: 1103–1109. 10.1590/s0100-879x2004000800001 [DOI] [PubMed] [Google Scholar]
- 15.Figueiredo DB, Carvalho E, Santos MP, Kraschowetz S, Zanardo RT, Campani G Jr et al. (2017) Production and purification of an untagged recombinant pneumococcal surface protein A (PspA4Pro) with high-purity and low endotoxin content. Appl Microbiol Biotechnol 101: 2305–2317. 10.1007/s00253-016-7983-9 [DOI] [PubMed] [Google Scholar]
- 16.Briles DE, Hollingshead SK, King J, Swift A, Braun PA, Park, MK et al. (2000) Immunization of humans with recombinant pneumococcal surface protein A (rPspA) elicits antibodies that passively protect mice from fatal infection with Streptococcus pneumoniae bearing heterologous PspA. J Infect Dis 182: 1694–1701. 10.1086/317602 [DOI] [PubMed] [Google Scholar]
- 17.Ferreira DM, Darrieux M, Oliveira ML, Leite LC, Miyaji EN (2008) Optimized immune response elicited by a DNA vaccine expressing pneumococcal surface protein a is characterized by a balanced immunoglobulin G1 (IgG1)/IgG2a ratio and proinflammatory cytokine production. Clin Vaccine Immunol 15: 499–505. 10.1128/CVI.00400-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ferreira DM, Oliveira ML, Moreno AT, Ho PL, Briles DE, Miyaji EN. (2010) Protection against nasal colonization with Streptococcus pneumoniae by parenteral immunization with a DNA vaccine encoding PspA (Pneumococcal surface protein A). Microb Pathog 48: 205–213. 10.1016/j.micpath.2010.02.009 [DOI] [PubMed] [Google Scholar]
- 19.Wang S, Li Y, Shi H, Scarpellini G, Torres-Escobar A, Roland KL, et al. (2010) Immune responses to recombinant pneumococcal PsaA antigen delivered by a live attenuated Salmonella vaccine. Infect Immun 78: 3258–3271. 10.1128/IAI.00176-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Campos IB, Darrieux M, Ferreira DM, Miyaji EN, Silva DA, Areas AP, et al. (2008) Nasal immunization of mice with Lactobacillus casei expressing the Pneumococcal Surface Protein A: induction of antibodies, complement deposition and partial protection against Streptococcus pneumoniae challenge. Microbes Infect 10: 481–488. 10.1016/j.micinf.2008.01.007 [DOI] [PubMed] [Google Scholar]
- 21.Hanniffy SB, Carter AT, Hitchin E, Wells JM (2007) Mucosal delivery of a pneumococcal vaccine using Lactococcus lactis affords protection against respiratory infection. J Infect Dis 195: 185–193. 10.1086/509807 [DOI] [PubMed] [Google Scholar]
- 22.Renauld-Mongenie G, Mielcarek N, Cornette J, Schacht AM, Capron A, Riveau G, et al. (1996) Induction of mucosal immune responses against a heterologous antigen fused to filamentous hemagglutinin after intranasal immunization with recombinant Bordetella pertussis. Proc Natl Acad Sci U S A 93: 7944–7949. 10.1073/pnas.93.15.7944 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mielcarek N, Riveau G, Remoue F, Antoine R, Capron A, Locht C. (1998) Homologous and heterologous protection after single intranasal administration of live attenuated recombinant Bordetella pertussis. Nat Biotechnol 16: 454–457. 10.1038/nbt0598-454 [DOI] [PubMed] [Google Scholar]
- 24.Coppens I, Alonso S, Antoine R, Jacob-Dubuisson F, Renauld-Mongenie G, Jacobs E, et al. (2001) Production of Neisseria meningitidis transferrin-binding protein B by recombinant Bordetella pertussis. Infect Immun 69: 5440–5446. 10.1128/IAI.69.9.5440-5446.2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Li R, Lim A, Ow ST, Phoon MC, Locht C, Chow VT, et al. (2011) Development of live attenuated Bordetella pertussis strains expressing the universal influenza vaccine candidate M2e. Vaccine 29: 5502–5511. 10.1016/j.vaccine.2011.05.052 [DOI] [PubMed] [Google Scholar]
- 26.Poulain-Godefroy O, Menozzi FD, Alonso S, Vendeville C, Capron A, Locht C, et al. (2003) Adjuvant activity of free Bordetella pertussis filamentous haemagglutinin delivered by mucosal routes. Scand J Immunol 58: 503–510. 10.1046/j.1365-3083.2003.01336.x [DOI] [PubMed] [Google Scholar]
- 27.Alonso S, Reveneau N, Pethe K, Locht C (2002) Eighty-kilodalton N-terminal moiety of Bordetella pertussis filamentous hemagglutinin: adherence, immunogenicity, and protective role. Infect Immun 70: 4142–4147. 10.1128/IAI.70.8.4142-4147.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Expression of FHA and FHA44:PspA4Pro on the surface of Bp137 and two clones of BpPspA4Pro (1.1 and 2.2) were tested using anti-FHA (A) or anti-PspA4 (B) antisera, by flow cytometry. BpPspA4Pro1.1 without primary sera was used as control. Numbers indicate the medians of fluorescence intensity for each curve.
(TIF)
Lysates from Bp137 (Bp) and two clones of BpPspA4Pro (1.1 and 2.2) corresponding to one dose of wP or wPpspAPro vaccines were compared to a curve established with recombinant Fha44:PspA4. Samples were evaluated by western-blot using anti-PspA4 and bands were analyzed using the Amersham Imager 680 analysis software. MW: GE Healthcare rainbow full range molecular weight marker.
(TIF)
Images were captured with the Kodak GelLogic 200 and the Carestream software. MW, GE Healthcare rainbow full range molecular weight marker.
(PDF)
Images were captured with the Kodak GelLogic 200 and the Carestream software. MW, GE Healthcare rainbow full range molecular weight marker.
(PDF)
Image was captured with the Amersham Imager 680 MW, GE Healthcare rainbow full range molecular weight marker.
(PDF)
Data Availability Statement
All relevant data are within the manuscript and its Supporting Information files.