Abstract
The present study aimed to examine the separate and combined association of self-reported sleep duration and quality with the prevalence of metabolic syndrome (MetS) in adult Chinese males. A total of 4,144 subjects were enrolled in the present crossed-sectional study. All participants were subjected to anthropometric measurements, blood tests and a survey based on a standardized questionnaire. Multivariate logistic regression was used to assess the influence of sleep duration and quality on the prevalence of MetS. The group that had 7 h of sleep had the best results as compared with those with shorter or longer sleep durations, and the prevalence of MetS was the lowest in this group. In addition, poor vs. good sleep quality was associated with an increased risk of MetS. Further analysis suggested that sleep duration and quality had an additive effect on the prevalence of MetS. In conclusion, sleep duration as well as quality should be considered when exploring the potential association between sleep and other conditions.
Keywords: sleep duration, sleep quality, metabolic syndrome
Introduction
Metabolic syndrome (MetS) is a clinical syndrome characterized by several associated risk factors for cardiovascular diseases and diabetes, including obesity, hypertension, elevated blood glucose and dyslipidemia (1). Epidemiological data suggest that in Chinese populations, the prevalence of MetS has exhibited a rapid increase in recent years (2). Preventing MetS by controlling relevant risk factors is a crucial and urgent task for the National Health Commission of the People's Republic of China.
Sleep deficiency has become prevalent in modern society, and it has been proven that chronic sleep deprivation may interrupt numerous essential homeostatic mechanisms. Various studies have demonstrated that short sleep duration is associated with the prevalence of various cardiovascular and metabolic diseases, including hypertension, coronary heart disease and diabetes (3–5). However, as to the potential association of sleep duration and the prevalence of MetS, no consistent conclusion has been reached so far. Certain previous studies have reported a U-shaped association between sleep duration and MetS (6,7). However, this conflicted with other studies, which demonstrated that a short rather than long sleep duration was associated with a higher risk of MetS (3,8). In addition, certain studies indicated that individuals with long sleep duration were predisposed to MetS (9,10). To date, the potential association of sleep duration with the prevalence of MetS has remained to be clarified.
However, the evaluation of sleep has qualitative and quantitative aspects. Perhaps it is not comprehensive to measure sleep by sleep duration only. Recently, the role of sleep quality in the development of cardiometabolic diseases has raised concern among cardiologists (11). Certain studies have demonstrated that poor sleep quality may contribute to the development of MetS (12,13). Furthermore, certain studies have attempted to explore the interactive effects of sleep duration and sleep quality on certain components of MetS, including abnormal glucose control and high blood pressure, and an additive effect of insufficient sleep duration and quality was identified (14,15). It is therefore necessary to elucidate the potential association of sleep duration with the prevalence of MetS while including the possible confounding effect of sleep quality.
The present cross-sectional survey aimed to explore the potential association of self-reported sleep duration and quality with the prevalence of MetS in adult Chinese males. In addition, the separate and combined effects of sleep duration and sleep quality on the prevalence of MetS were analyzed.
Materials and methods
Study design and population
This study was designed as a cross-sectional study. A total of 6,120 participants aged >18 years residing in the Fangezhuang, Tangshan, Lvjiatuo and Qianjiaying communities of Tangshan city located in North China were enrolled in the study between September 2013 and December 2013, and the data was collected and analyzed at Kailuan General Hospital (Tangshan, Hebei, China). Participants with obstructive sleep apnea syndrome (OSAS), restless legs syndrome (RLS) or snoring were excluded due to their complex mechanistic association with hypertension, which was difficult to measure and control. Most participants were employees of the Kailuan Group, a large-scale comprehensive enterprise that mainly manages coal products, where female employees make up a small proportion. Only 571 female participants were initially enrolled and the sample size was too small to be analyzed to accomplish the aim of the study; therefore, only the results of the association of sleep with MetS in male participants are included. All study protocols were approved by the Ethics Committee of Kailuan General Hospital [Tangshan, Hebei, China; approval reference no. 2013(5)] and written informed consent was obtained from all study participants.
Anthropometric measurements
Anthropometric measurements were performed on participants wearing light clothes but no shoes. The waist circumference was measured with participants standing up straight at the navel level as participants exhaled lightly. Body height was measured to the nearest 0.1 cm and weight was measured to the nearest 0.1 kg. Using a calibrated standard mercury sphygmomanometer (Yuyue Medical Equipment & Supply Co., Ltd.), blood pressure was measured twice in a sitting position and the mean reading was recorded. If the difference between two readings was >5 mmHg, a third reading was taken and the mean of the three readings was used.
Blood test
Participants were requested to fast overnight prior to the collection of a venous blood sample. Serum was prepared by centrifuging the blood at 500 × g for 10 min at 4°C within 4 h of collection. Serum triglyceride, high-density lipoprotein cholesterol (HDL-C) and fasting blood glucose were measured using an automatic biochemical analyzer (Hitachi 717; Hitachi).
Questionnaire survey
All participants were subjected to a structured questionnaire including age, history of diagnosed hypertension and type 2 diabetes and/or receiving of hypertension and diabetes medication, smoking status, drinking status, educational level, physical activity, sleep duration and sleep quality. Details regarding the definition as well as the classification of certain items listed above were identical to those of a previous study by our group (14). Sleep duration was evaluated based on self-reported average sleep duration in the past month, which precluded the time spent awake in bed. Sleep duration was categorized into 5 groups, ‘<6’, ‘6’, ‘7’, ‘8’ and ‘>8 h’, as stated in the questionnaire. Sleep quality was assessed using the standard Pittsburgh Sleep Quality Index (PSQI; Table SI) (16). The PSQI is a 19-item self-reporting measure of sleep quality over the past month and contains 7 component scales: Sleep quality, sleep latency, sleep duration, sleep efficiency, sleep disturbances, use of sleep medication and daytime dysfunction. The global PSQI score may vary within a range of 0–21. In the present study, sleep quality of participants was classified into four groups: ‘Very good’ (PSQI of <3), ‘good’ (PSQI of 3 to <6), ‘poor’ (PSQI of 6 to <9) and ‘very poor’ (PSQI ≥9). In addition, the General Anxiety Disorder-7 (GAD-7) and Patient Health Questionnaire-9 (PHQ-9) scales were used to evaluate the anxiety and depression status of participants, respectively. GAD-7 is a seven-question inventory for self-assessment and is one of the most common instruments for measuring severity of anxiety (17). PHQ-9 is a widely used nine-question inventory for the self-assessment of depression (18).
Definition of MetS
According to the definition of MetS issued in the Dyslipidemia Prevention and Cure Guidelines of China in 2007 (19), MetS was diagnosed when any three or more of the following five risk factors were present: i) Abdominal obesity: Waist circumference ≥90 cm for males, ≥85 cm for females; ii) elevated blood glucose: Fasting blood glucose ≥6.l mmol/l or 2-h post-prandial plasma glucose ≥7.8 mmol/l and/or with diagnosed diabetes and current use of anti-diabetic therapy; iii) hypertension: Blood pressure ≥130/85 mmHg and/or with diagnosed hypertension and iv) fasting HDL-C<l.04 mmol/l.
Statistical analysis
In the present study, continuous variables were expressed as mean ± SE and compared using one-way analysis of variance followed by Dunnett's post-hoc test. Categorical variables were expressed as n (%) and compared using the χ2 test. Cochran-Armitage tests were used to test the association between the prevalence of MetS and sleep duration/quality. The potential correlation between sleep duration and sleep quality with the prevalence of MetS was explored by logistic regression analysis with or without adjustment for plausible confounders, including age, smoking status, drinking status, physical activity, educational level, and anxiety and depression scores. Furthermore, to investigate the interactive association of sleep duration and sleep quality with the prevalence of MetS, participants were divided into groups according to different combinations of sleep duration and sleep quality. Odds ratios (ORs) and 95% CIs of each group were calculated using multiple logistic regression analysis, with the group with a sleep duration of 7 h and very good sleep quality used as the reference. For all comparisons, the level of statistical significance was set at P<0.05 (two-sided). SPSS 19.0 (IBM Corp.) was used for all statistical analyses.
Results
Basic characteristics
Of the 6,120 initially enrolled participants, 351 (5.7%) were diagnosed with OSAS, 43 (0.7%) with RLS, 1,011 (16.5%) reported snoring and the 571 females (9.3%) were excluded. The remaining 4,144 males were included into the final analysis. Participants with <6, 6, 7, 8 and >8 h of sleep accounted for 8.6% (n=356), 24.6% (n=1,021), 34.9% (n=1,446), 26.1% (n=1,082) and 5.8% (n=239) of the cohort, respectively. With regard to sleep quality, the participants with sleep quality rated as very good, good, poor and very poor accounted for 57.2% (n=2,369), 27.7% (n=1,146), 11.3% (n=468) and 4.6% (n=191) of the cohort, respectively (Tables I and II).
Table I.
Basic characteristics of adult Chinese male participants according to sleep duration.
| Sleep duration (h) | |||||||
|---|---|---|---|---|---|---|---|
| Item | <6 (n=356) | 6 (n=1,021) | 7 (n=1,446) | 8 (n=1,082) | >8 (n=239) | Total (n=4,144) | P-value |
| Age (years) | 47.91±0.37 | 46.84±0.23 | 46.92±0.18 | 46.98±0.41 | 47.47±0.42 | 47.04±0.14 | 0.33 |
| WC (cm) | 91.73±6.02 | 88.71±4.11 | 88.78±4.10 | 89.03±4.37 | 91.27±5.68 | 88.94±4.21) | <0.01 |
| BMI (kg/m2) | 25.45±0.19 | 25.17±0.11 | 25.44±0.10 | 25.19±0.10 | 25.05±0.19 | 25.28±0.06 | 0.16 |
| SBP (mmHg) | 130.61±0.71 | 129.79±0.47 | 129.96±0.38 | 129.62±0.44 | 131.84±0.81 | 130.02±0.22 | 0.18 |
| DBP (mmHg) | 85.87±0.47 | 85.9±0.31 | 85.80±0.25 | 85.71±0.30 | 86.86±0.55 | 85.88±0.15 | 0.50 |
| TG (mmol/l) | 2.03±0.12 | 1.95±0.03 | 1.84±0.06 | 1.89±0.07 | 1.95±0.12 | 1.90±0.04 | 0.07 |
| HDL-C (mmol/l) | 1.33±0.02 | 1.34±0.02 | 1.34±0.01 | 1.35±0.01 | 1.37±0.02 | 1.33±0.01 | 0.53 |
| FBG (mmol/l) | 5.57±0.08 | 5.41±0.05 | 5.43±0.0 | 5.5±0.05 | 5.29±0.07 | 5.45±0.02 | 0.08 |
| Previous hypertension | 72 (20.2) | 157 (15.4) | 201 (13.9) | 120 (11.1) | 34 (14.3) | 584 (14.1) | <0.01 |
| Previous diabetes | 52 (14.6) | 85 (8.3) | 103 (7.1) | 80 (7.4) | 24 (10.2) | 344 (8.3) | <0.01 |
| GAD-7 score | 4.40±0.28 | 3.35±0.15 | 1.63±0.08 | 1.51±0.09 | 1.93±0.30 | 2.27±0.06 | <0.01 |
| PHQ-9 score | 5.06±0.32 | 3.29±0.16 | 2.02±0.10 | 1.63±0.10 | 2.25±0.32 | 2.48±0.07 | <0.01 |
| Current smoking | 215 (60.5) | 611 (59.8) | 781 (54.0) | 576 (53.2) | 133 (55.8) | 2316 (55.9) | <0.01 |
| Current drinking | 115 (32.2) | 289 (28.3) | 320 (22.1) | 285 (26.2) | 62 (25.9) | 1071 (25.8) | <0.01 |
| Active exercise habit | 109 (30.6) | 311 (30.5) | 508 (35.1) | 392 (36.2) | 59 (24.6) | 1379 (33.3) | <0.01 |
| High level of education | 140 (39.4) | 415 (40.6) | 450 (31.1) | 413 (38.2) | 92 (38.7) | 1510 (36.4) | <0.01 |
Values are expressed as mean ± standard error or n (%). WC, waist circumference; BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; TG, serum triglyceride; HDL-C, high-density lipoprotein cholesterol; FBG, fasting blood glucose; GAD-7, General Anxiety Disorder-7; PHQ-9, Patient Health Questionnaire-9.
Table II.
Basic characteristics of adult Chinese male participants according to sleep quality.
| Sleep quality | ||||||
|---|---|---|---|---|---|---|
| Item | Very good (n=2,369) | Good (n=1,146) | Poor (n=468) | Very poor (n=191) | Total (n=4,144) | P-value |
| Age (years) | 47.20±0.14 | 46.38±0.40 | 47.48±0.36 | 47.34±0.52 | 47.01±0.14 | 0.06 |
| WC (cm) | 88.72±4.19 | 89.21±4.11 | 90.41±5.13 | 92.01±5.73 | 88.94±4.21 | <0.01 |
| BMI (kg/m2) | 25.26±0.08 | 25.33±0.11 | 25.41±0.18 | 25.01±0.23 | 25.29±0.06 | 0.58 |
| SBP (mmHg) | 130.53±0.30 | 129.16±0.42 | 130.16±0.69 | 128.32±1.04 | 130.01±0.23 | 0.02 |
| DBP (mmHg) | 86.24±0.20 | 85.32±0.28 | 85.69±0.48 | 85.38±0.7 | 85.89±0.15 | 0.05 |
| TC (mmol/l) | 2.02±0.03 | 1.94±0.09 | 1.89±0.12 | 2.01±0.12 | 1.90±0.04 | 0.09 |
| HDL-C (mmol/l) | 1.36±0.03 | 1.29±0.02 | 1.26±0.02 | 1.27±0.03 | 1.34±0.02 | 0.19 |
| FBG (mmol/l) | 5.47±0.03 | 5.44±0.05 | 5.38±0.06 | 5.42±0.10 | 5.45±0.02 | 0.68 |
| Previous hypertension | 296 (12.5) | 170 (14.8) | 74 (15.8) | 44 (23.0) | 584 (14.1) | <0.01 |
| Previous diabetes | 173 (7.3) | 93 (8.1) | 48 (10.3) | 30 (15.7) | 344 (8.3) | <0.01 |
| GAD-7 score | 1.15±0.06 | 3.06±0.12 | 4.54±0.26 | 6.4±0.43 | 2.27±0.06 | <0.01 |
| PHQ-9 score | 1.21±0.06 | 3.54±0.13 | 4.60±0.26 | 7.9±0.53 | 2.48±0.07 | <0.01 |
| Current smoking | 1,279 (54.0) | 662 (57.8) | 256 (54.7) | 119 (62.2) | 2,316 (55.9) | 0.04 |
| Current drinking | 572 (24.1) | 289 (25.2) | 132 (28.3) | 78 (40.8) | 1,071 (25.8) | <0.01 |
| Active exercise habit | 785 (33.1) | 416 (36.3) | 126 (26.9) | 52 (27.2) | 1,379 (33.3) | <0.01 |
| High level of education | 811 (34.2) | 452 (39.4) | 162 (34.7) | 85 (44.4) | 1,510 (36.4) | <0.01 |
Values are expressed as mean ± standard error or n (%). WC, waist circumference; BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; TG, serum triglyceride; HDL-C, high-density lipoprotein cholesterol; FBG, fasting blood glucose; GAD-7, General AnxietyDisorder-7; PHQ-9, Patient Health Questionnaire-9.
Prevalence of MetS
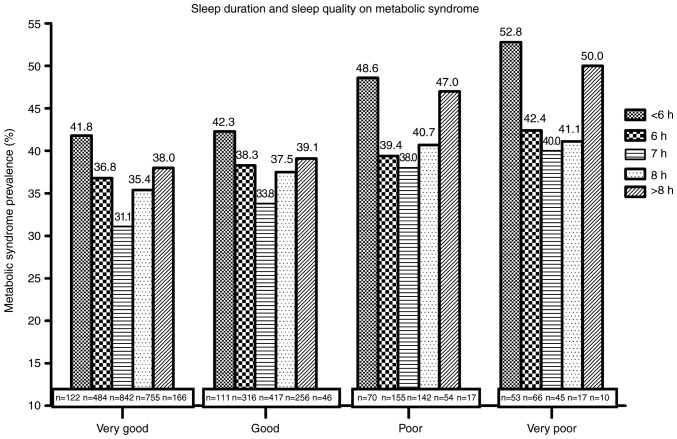
The prevalence of MetS in participants with different sleep duration or quality is provided in Fig. 1. The participants with a sleep duration of 7 h and sleep quality rated as very good had the lowest prevalence of MetS. Compared to participants with a sleep duration of 7 h, those with a shorter and longer sleep duration were indicated to have a higher prevalence of MetS, and a U-shaped association between sleep duration and the prevalence of MetS was observed. This trend was statistically significant (P<0.05). As for sleep quality, participants with poorer sleep quality were observed to have a higher prevalence of MetS as compared with those with very good sleep quality (P<0.05).
Figure 1.
Prevalence of metabolic syndrome in participants with different combinations of sleep duration and sleep quality. The number at the bottom of each column indicates the sample size for the respective column.
Association of sleep duration or sleep quality with the prevalence of MetS
Multivariate logistic regression analysis was used to explore the potential association of sleep duration or sleep quality with the prevalence of MetS. After adjustment for age, smoking, drinking, exercise, education level, anxiety scores and depression scores, compared with participants with 7 h of sleep, the ORs and 95% Cis for MetS of those with <6, 6, 8 and >8 h of sleep were 1.57 (95% CI, 1.26–1.95), 1.26 (95% CI, 1.14–1.39), 1.21 (95% CI, 1.11–1.32) and 1.32 (95% CI, 1.06–1.65), respectively. Furthermore, compared with the group with very good sleep quality, the ORs and 95% CIs for MetS in patients with good, poor and very poor sleep quality were 1.12 (95% CI, 1.01–1.23), 1.20 (95% CI, 1.02–1.40) and 1.51 (95% CI, 1.21–1.88), respectively (Table III).
Table III.
Logistic regression analysis of the influence of sleep duration and sleep quality on the prevalence of metabolic syndrome in adult Chinese males.
| A, Influence of sleep duration | |||||
|---|---|---|---|---|---|
| Sleep duration (h) | n | Unadjusted OR (95%CI) | P-value | Adjusted OR (95%CI)a | P-value |
| <6 | 356 | 1.55 (1.23–1.92) | – | 1.57 (1.26–1.95) | – |
| 6 | 1,021 | 1.23 (1.12–1.36) | <0.00 | 1.26 (1.14–1.39) | <0.00 |
| 7 | 1,446 | Reference | – | Reference | – |
| 8 | 1,082 | 1.24 (1.13–1.35) | <0.00 | 1.21 (1.11–1.32) | <0.00 |
| >8 | 239 | 1.29 (1.03–1.60) | P-value | 1.32 (1.06–1.65) | P-value |
| B, Influence of sleep quality | |||||
| Sleep duration (h) | n | Unadjusted OR (95%CI) | P-value | Adjusted OR (95%CI)a | P-value |
| Very good | 2,369 | Reference | – | Reference | – |
| Good | 1,146 | 1.15 (1.02–1.30) | <0.00 | 1.12 (1.01–1.23) | <0.00 |
| Poor | 438 | 1.21 (1.03–1.42) | <0.00 | 1.20 (1.02–1.40) | <0.00 |
| Very poor | 191 | 1.53 (1.23–1.92) | <0.00 | 1.51 (1.21–1.88) | <0.00 |
Adjusted for age, body mass index, status of smoking and drinking, exercise habit, educational level, and score of anxiety and depression. Sleep quality was evaluated by the standard Pittsburgh Sleep Quality Index and is categorized based on total score. CI, confidence interval; OR, odds ratio.
Association of combined sleep duration and sleep quality with the prevalence of MetS
The association of the combined effects of sleep duration and quality on MetS was analyzed by univariate and multivariate logistic regression analysis (Table IV). Compared with participants with 7 h sleep duration and very good sleep quality (used as the reference), those in all other groups were observed to have a higher prevalence of MetS after adjusting for potential confounding factors. The ORs as well as their 95% CIs for each group are stated below, indicating an obvious additive effect of inadequate sleep duration and quality on the prevalence of MetS: Sleep duration of <6 h combined with any sleep quality [1.52 (1.23–1.88) for very good; 1.64 (1.30–1.86) for good; 2.25 (1.60–3.03) for poor and very poor], sleep duration of 6 h combined with any sleep quality [1.24 (1.14–1.36) for very good; 1.33 (1.13–1.52) for good; 1.49 (1.21–1.67) for poor and very poor], 7 h sleep duration combined with any sleep quality [1.17 (1.05–1.31) for good; 1.34 (1.09–1.63) for poor and very poor], sleep duration of 8 h combined with any sleep quality [1.25 (1.12–1.39) for very good; 1.30 (1.15–1.46) for good; 1.52 (1.22–1.93) for poor and very poor], and sleep duration of >8 h combined with any sleep quality [1.37 (1.11–1.67) for very good; 1.39 (1.13–1.73) for good; 2.03 (1.45–2.86) for poor and very poor].
Table IV.
Logistic regression analysis of the influence of different combinations of sleep duration and quality on the prevalence of metabolic syndrome in adult Chinese males.
| A, Unadjusted analysis | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Sleep duration (h) | ||||||||||
| <6 | 6 | 7 | 8 | >8 | ||||||
| Sleep quality | n | OR (95% CI) | n | OR (95% CI) | n | OR (95% CI) | n | OR (95% CI) | n | OR (95% CI) |
| Very good | 122 | 1.54 (1.25–1.90)b | 484 | 1.27 (1.16–1.39)b | 842 | Reference | 755 | 1.23 (0.11–1.38)d | 166 | 1.34 (1.10–1.63)c |
| Good | 111 | 1.62 (1.28–1.84)b | 316 | 1.36 (1.15–1.55)b | 417 | 1.16 (1.05–1.30)c | 256 | 1.32 (1.17–1.50)b | 46 | 1.41 (1.14–1.75)b |
| Poor and very poor | 123 | 2.21 (1.62–3.06)b | 221 | 1.47 (1.20–1.65)b | 187 | 1.38 (1.13–1.68)b | 71 | 1.50 (1.21–1.90)b | 27 | 1.98 (1.40–2.78)b |
| B, Adjusted analysisa | ||||||||||
| Sleep quality | n | OR (95% CI) | n | OR (95% CI) | n | OR (95% CI) | n | OR (95% CI) | n | OR (95% CI) |
| Very good | 122 | 1.52 (1.23–1.88)b | 484 | 1.24 (1.14–1.36)b | 842 | Reference | 755 | 1.25 (1.12–1.39)b | 166 | 1.37 (1.11–1.67)c |
| Good | 111 | 1.64 (1.30–1.86)b | 316 | 1.33 (1.13–1.52)c | 417 | 1.17 (1.05–1.31)c | 256 | 1.30 (1.15–1.46)b | 46 | 1.39 (1.13–1.73)c |
| Poor and very poor | 123 | 2.25 (1.60–3.03)b | 221 | 1.49 (1.21–1.67)b | 187 | 1.34 (1.09–1.63)c | 71 | 1.52 (1.22–1.93)b | 27 | 2.03 (1.45–2.86)b |
Adjusted for age, body mass index, status of smoking and drinking, exercise habit, educational level, and score of anxiety and depression.
P<0.01
P<0.05
P>0.05. Sleep quality was evaluated by the standard Pittsburgh Sleep Quality Index and is categorized based on total score. CI, confidence interval; OR, odds ratio.
Discussion
In the present study on adult Chinese males, a U-shaped association between sleep duration and the prevalence of MetS was observed and it was also noted that poor sleep quality was associated with a high prevalence of MetS. Furthermore, the combined effects of sleep duration and sleep quality on the prevalence of MetS in the study population were explored and the results indicated an additive effect of inadequate sleep duration and sleep quality on the prevalence of MetS.
A U-shaped association between sleep duration and MetS was observed in the present study. This result was consistent with those of previous studies (6,7). However, the studies of Wu et al (3) and Najafian et al (20), which indicated that a short rather than long sleep duration increased the risk of MetS, were not in accordance with the present results. Apart from differences in age, ethnicity and basic characteristics, negligence of the confounding effect of sleep quality is one of the important potential reasons for the conflicting results mentioned above. Sleep has quantitative as well as qualitative aspects. Sleep duration is not the only criterion for determining the presence of sleep deficiency. Therefore, it may not be sufficient to evaluate sleep only based on sleep duration, which was performed in most previous studies on the potential association between sleep and the prevalence of MetS (3,6–10). Failure to assess sleep comprehensively may be the most important reason for the conflicting results among those previous studies.
The present study indicated that poor sleep quality was associated with MetS, which was consistent with the results of previous studies (12,13). Kazman et al (21) also assessed the sleep quality by using the PSQI, but failed to observe an association between poor sleep quality and the prevalence of MetS among African Americans. Sociodemographic factors, health behaviors and socioeconomic status have already been proven to be linked to MetS and sleep (3,10,22,23). However, health-associated behaviors (e.g., smoking, drinking and exercise) and socioeconomic status were not considered and adjusted for in the study by Kazman et al (21), which may explain the discrepancies between the previous study by Kazman et al and the present study. It has been reported that poor mental health status, including depression or anxiety, may increase the risk of MetS (24,25), and sleep disorders are also frequently concomitant in patients with depression and anxiety (26). Therefore, whether poor mental and emotional health are independent risk factors of MetS or whether sleep disorders mediate the association of mental/emotional health with the development of MetS has not been confirmed. However, this was neglected in most previous studies. In the present study, the depression and anxiety status of all participants was evaluated by using the PHQ-9 and GAD-7 scales and adjustment for these parameters was included in the logistic regression analysis for the influence of sleep on MetS.
As mentioned above, it is preferable to consider and measure sleep duration as well as quality when exploring the potential association of sleep with MetS. To elucidate the exact effect of combined sleep duration and quality on MetS, the influence of different combinations of sleep duration and sleep quality on the prevalence of MetS was investigated in the present study. The results suggest that in spite of long sleep duration, such as 8 h or more, there was still a high prevalence of MetS in those participants with poor sleep quality, suggesting that sleep duration and sleep quality had a combined effect on the prevalence of MetS. Evaluation of sleep based on either sleep quality or quantity on its own may inevitably lead to bias of the results, which should be considered in future studies on the link between sleep and diseases.
The mechanisms underlying the association between sleep and MetS remain to be fully elucidated. It has been indicated that short sleep duration affects the levels of endocrine hormones, including leptin and ghrelin, finally resulting in increased appetite and obesity (27). A recent study performed on Chinese children suggested a strong link between short sleep duration and adverse adipokine secretion patterns (28). Lusardi et al (29) reported that sleep deprivation was associated with sympathetic hyperactivity and elevated blood pressure. Insulin resistance, which is a key factor in the development of MetS, was also suggested to be associated with short sleep duration (30). Furthermore, long sleep duration was observed to increase the risk of MetS components. Long sleep duration may be associated with obesity due to a decline in activity levels and energy consumption (7). It was also reported that people getting a sufficient sleep duration had lower levels of inflammation and oxidative stress and higher levels of anti-oxidants compared with short or long sleepers (31). From the above, it is obvious that further studies are required to elucidate the potential link between sleep disorders and MetS.
The present study has several limitations. First of all, it was difficult to assess the causal association between sleep status and the prevalence of MetS due to the cross-sectional design of the study. It is likely that sleep disorders are associated with an increased risk of MetS, considering the fact that certain cohort studies have confirmed that sleep disorders area major cause of hypertension and diabetes, which are components of MetS (5,32). Furthermore, self-reported sleep duration may not represent real sleep duration. However, Signal et al (33) indicated that self-reported sleep duration was close to the polysomnography result. In addition, the 2-h post-prandial blood glucose of the enrolled participants was not measured in the present study due to limited time and funding. This may have led to underestimation of the number of participants with impaired glucose metabolism, considering the fact that a sizeable proportion of the Chinese diabetic population presents with normal fasting blood glucose (34).
Despite the limitations mentioned above, the present study indicated a U-shaped association between sleep duration and MetS and indicated that poor sleep quality was associated with an elevated prevalence of MetS in male Chinese adults. Furthermore, the study also revealed that inadequate sleep duration and sleep quality have an additive effect on the prevalence of MetS, which was the most important result. This indicated that sleep duration as well as quality should be measured to avoid a biased result when exploring the potential association of sleep with cardiometabolic diseases.
Supplementary Material
Acknowledgements
The authors thank the staff of Kailuan Study for their efforts in data collection. Additionally, the authors express their gratitude to Mrs. Mele Vaikoloa Fonua, Graduate College of Chongqing Medical University, China, for her work to improve the language and style of the manuscript.
Funding
This study was funded by grants from the 12th Five-Year Science and Technology Support Program of the Ministry of Science and Technology of China (grant no. 2013BAI06B02).
Availability of data and materials
The datasets generated and/or analyzed during the current study are not publicly available due to the present study being one part of the 12th Five-Year Science and Technology Support Program of the Ministry of Science and Technology of China (grant no. 2013BAI06B02) which has not finished officially. As such, the relevant raw data can not be made freely available to any researchers at this current time. However, any researchers who wish to use them for non-commercial purposes whilst preserving the necessary confidentiality can contact the corresponding author and the data are available from the corresponding author on reasonable request.
Authors' contributions
DH and HX developed the idea for the study and carried out the design with all co-authors. KL is responsible for the recruitment and follow-up of study participants as well as data collection. JC carried out the analysis. YZ participated in the analysis and drafted the manuscript which was revised by all authors. All authors read and approved the final manuscript.
Ethics approval and consent to participate
All study protocols were approved by the Ethics Committee of Kailuan General Hospital (Tangshan, Hebei, China). Written informed consent for publication was obtained from all study participants.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Mamcarz A, Podolec P, Kopeć G, Czarnecka D, Rynkiewicz A, Stańczyk J, Undas A, Godycki-Cwirko M, Kozek E, Pajak A, et al. Polish forum for prevention guidelines on metabolic syndrome. Kardiol Pol. 2010;68:121–124. [PubMed] [Google Scholar]
- 2.Shen J, Goyal A, Sperling L. The emerging epidemic of obesity, diabetes, and the metabolic syndrome in china. Cardiol Res Pract. 2012;2012:178675. doi: 10.1155/2012/178675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wu MC, Yang YC, Wu JS, Wang RH, Lu FH, Chang CJ. Short sleep duration associated with a higher prevalence of metabolic syndrome in an apparently healthy population. Prev Med. 2012;55:305–309. doi: 10.1016/j.ypmed.2012.07.013. [DOI] [PubMed] [Google Scholar]
- 4.Katano S, Nakamura Y, Nakamura A, Murakami Y, Tanaka T, Takebayashi T, Okayama A, Miura K, Okamura T, Ueshima H, HIPOP-OHP Research Group Relationship between sleep duration and clustering of metabolic syndrome diagnostic components. Diabetes Metab Syndr Obes. 2011;4:119–125. doi: 10.2147/DMSO.S16147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gangwisch JE, Heymsfield SB, Boden-Albala B, Buijs RM, Kreier F, Pickering TG, Rundle AG, Zammit GK, Malaspina D. Short sleep duration as a risk factor for hypertension: Analyses of the first National Health and Nutrition Examination Survey. Hypertension. 2006;47:833–839. doi: 10.1161/01.HYP.0000217362.34748.e0. [DOI] [PubMed] [Google Scholar]
- 6.Ju SY, Choi WS. Sleep duration and metabolic syndrome in adult populations: A meta-analysis of observational studies. Nutr Diabetes. 2013;3:e65. doi: 10.1038/nutd.2013.8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Choi KM, Lee JS, Park HS, Baik SH, Choi DS, Kim SM. Relationship between sleep duration and the metabolic syndrome: Korean National Health and Nutrition Survey 2001. Int J Obes (Lond) 2008;32:1091–1097. doi: 10.1038/ijo.2008.62. [DOI] [PubMed] [Google Scholar]
- 8.Xi B, He D, Zhang M, Xue J, Zhou D. Short sleep duration predicts risk of metabolic syndrome: A systematic review and meta-analysis. Sleep Med Rev. 2014;18:293–297. doi: 10.1016/j.smrv.2013.06.001. [DOI] [PubMed] [Google Scholar]
- 9.Arora T, Jiang CQ, Thomas GN, Lam KB, Zhang WS, Cheng KK, Lam TH, Taheri S. Self-reported long total sleep duration is associated with metabolic syndrome: The Guangzhou Biobank Cohort Study. Diabetes Care. 2011;34:2317–2319. doi: 10.2337/dc11-0647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Santos AC, Ebrahim S, Barros H. Alcohol intake, smoking, sleeping hours, physical activity and the metabolic syndrome. Prev Med. 2007;44:328–334. doi: 10.1016/j.ypmed.2006.11.016. [DOI] [PubMed] [Google Scholar]
- 11.Basnet S, Merikanto I, Lahti T, Männistö S, Laatikainen T, Vartiainen E, Partonen T. Associations of common chronic non-communicable diseases and medical conditions with sleep-related problems in a population-based health examination study. Sleep Sci. 2016;9:249–254. doi: 10.1016/j.slsci.2016.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Okubo N, Matsuzaka M, Takahashi I, Sawada K, Sato S, Akimoto N, Umeda T, Nakaji S, Hirosaki University Graduate School of Medicine Relationship between self-reported sleep quality and metabolic syndrome in general population. BMC Public Health. 2014;14:562. doi: 10.1186/1471-2458-14-562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hung HC, Yang YC, Ou HY, Wu JS, Lu FH, Chang CJ. The association between self-reported sleep quality and metabolic syndrome. PLoS One. 2013;8:e54304. doi: 10.1371/journal.pone.0054304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lu K, Chen J, Wu S, Chen J, Hu D. Interaction of sleep duration and sleep quality on hypertension prevalence in adult Chinese males. J Epidemiol. 2015;25:415–422. doi: 10.2188/jea.JE20140139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lou P, Chen P, Zhang L, Zhang P, Chang G, Zhang N, Li T, Qiao C. Interaction of sleep quality and sleep duration on impaired fasting glucose: A population-based cross-sectional survey in China. BMJ Open. 2014;4:e004436. doi: 10.1136/bmjopen-2013-004436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Buysse DJ, Reynolds CF, III, Monk TH, Berman SR, Kupfer DJ. The pittsburgh sleep quality index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989;28:193–213. doi: 10.1016/0165-1781(89)90047-4. [DOI] [PubMed] [Google Scholar]
- 17.Donker T, van Straten A, Marks I, Cuijpers P. Quick and easy self-rating of Generalized Anxiety Disorder: Validity of the Dutch web-based GAD-7, GAD-2 and GAD-SI. Psychiatry Res. 2011;188:58–64. doi: 10.1016/j.psychres.2011.01.016. [DOI] [PubMed] [Google Scholar]
- 18.Zhang Y, Ting R, Lam M, Lam J, Nan H, Yeung R, Yang W, Ji L, Weng J, Wing YK, et al. Measuring depressive symptoms using the Patient Health Questionnaire-9 in Hong Kong Chinese subjects with type 2 diabetes. J Affect Disord. 2013;151:660–666. doi: 10.1016/j.jad.2013.07.014. [DOI] [PubMed] [Google Scholar]
- 19.Weng J, Ji L, Jia W, Lu J, Zhou Z, Zou D, Zhu D, Chen L, Chen L, Guo L, et al. Standards of care for type 2 diabetes in China. Diabetes Metab Res Rev. 2016;32:442–458. doi: 10.1002/dmrr.2827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Najafian J, Toghianifar N, Mohammadifard N, Nouri F. Association between sleep duration and metabolic syndrome in a population-based study: Isfahan healthy heart program. J Res Med Sci. 2011;16:801–806. [PMC free article] [PubMed] [Google Scholar]
- 21.Kazman JB, Abraham PA, Zeno SA, Poth M, Deuster PA. Self-reported sleep impairment and the metabolic syndrome among African Americans. Ethn Dis. 2012;22:410–415. [PubMed] [Google Scholar]
- 22.Zuo HJ, Yao CH, Hu YS, Kong LZ. Relations between smoking, alcohol intake, physical activity, sleeping hours and the metabolic syndrome in Chinese male aged 18–45 years old. Zhonghua Liu Xing Bing Xue Za Zhi. 2011;32:235–238. (In Chinese) [PubMed] [Google Scholar]
- 23.Bidulescu A, Din-Dzietham R, Coverson DL, Chen Z, Meng YX, Buxbaum SG, Gibbons GH, Welch VL. Interaction of sleep quality and psychosocial stress on obesity in African Americans: The Cardiovascular Health Epidemiology Study (CHES) BMC Public Health. 2010;10:581. doi: 10.1186/1471-2458-10-581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bonnet F. Associations between anxiety, depression, and the metabolic syndrome. Biol Psychiatry. 2007;62:1251–1257. doi: 10.1016/j.biopsych.2007.01.012. [DOI] [PubMed] [Google Scholar]
- 25.Kahl KG, Schweiger U, Correll C, Müller C, Busch ML, Bauer M, Schwarz P. Depression, anxiety disorders, and metabolic syndrome in a population at risk for type 2 diabetes mellitus. Brain Behav. 2015;5:e00306. doi: 10.1002/brb3.306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nyer M, Farabaugh A, Fehling K, Soskin D, Holt D, Papakostas GI, Pedrelli P, Fava M, Pisoni A, Vitolo O, Mischoulon D. Relationship between sleep disturbance and depression, anxiety, and functioning in college students. Depress Anxiety. 2013;30:873–880. doi: 10.1002/da.22064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Taheri S, Lin L, Austin D, Young T, Mignot E. Short sleep duration is associated with reduced leptin, elevated ghrelin, and increased body mass index. PLoS Med. 2004;1:210–217. doi: 10.1371/journal.pmed.0010062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Li L, Fu J, Yu XT, Li G, Xu L, Yin J, Cheng H, Hou D, Zhao X, Gao S, et al. Sleep duration and cardiometabolic risk among chinese school-aged children: Do adipokines play a mediating role? Sleep. 2017;40 doi: 10.1093/sleep/zsx042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lusardi P, Zoppi A, Preti P, Pesce RM, Piazza E, Fogari R. Effects of insufficient sleep on blood pressure in hypertensive patients: A 24-h study. Am J Hypertens. 1999;12:63–68. doi: 10.1016/S0895-7061(98)00200-3. [DOI] [PubMed] [Google Scholar]
- 30.Stamatakis KA, Punjabi NM. Effects of sleep fragmentation on glucose metabolism in normal subjects. Chest. 2010;137:95–101. doi: 10.1378/chest.09-0791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kanagasabai T, Ardern CI. Contribution of inflammation, oxidative stress, and antioxidants to the relationship between sleep duration and cardiometabolic health. Sleep. 2015;38:1905–1912. doi: 10.5665/sleep.5238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Knutson KL, Wu D, Patel SR, Loredo JS, Redline S, Cai J, Gallo LC, Mossavar-Rahmani Y, Ramos AR, Teng Y, et al. Association between sleep timing, obesity, diabetes: The Hispanic Community Health Study/Study of Latinos (HCHS/SOL) cohort study: Sleep timing, obesity, and diabetes. Sleep. 2017;40 doi: 10.1093/sleep/zsx014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Signal TL, Gale J, Gander PH. Sleep measurement in flight crew: Comparing actigraphic and subjective estimates to polysomnography. Aviat Space Environ Med. 2005;76:1058–1063. [PubMed] [Google Scholar]
- 34.Yang W, Lu J, Weng J, Jia W, Ji L, Xiao J, Shan Z, Liu J, Tian H, Ji Q, et al. Prevalence of diabetes among men and women in China. N Engl J Med. 2010;362:1090–1101. doi: 10.1056/NEJMoa0908292. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated and/or analyzed during the current study are not publicly available due to the present study being one part of the 12th Five-Year Science and Technology Support Program of the Ministry of Science and Technology of China (grant no. 2013BAI06B02) which has not finished officially. As such, the relevant raw data can not be made freely available to any researchers at this current time. However, any researchers who wish to use them for non-commercial purposes whilst preserving the necessary confidentiality can contact the corresponding author and the data are available from the corresponding author on reasonable request.