Abstract
Lung cancer, which is a leading cause of cancer-related deaths, is diagnosed at a male to female ratio of 2.1:1. Serine-threonine kinase 31 (STK31) is a novel cancer/testis (CT)-related gene that is highly expressed in several types of cancers, such as lung and colorectal cancer, and plays crucial roles in cancer. In the present study, increased expression of STK31 and β-catenin was observed in lung cancer tissues and cell lines. Downregulation of STK31 expression in lung cancer cells significantly inhibited their proliferation by arresting the cell cycle in the G1 phase concurrent with decreased β-catenin, c-myc and cyclin D1 protein levels, while upregulation of STK31 had the opposite effects. In addition, STK31-induced lung cancer cell viability, proliferation, cell cycle progression, and expression of related genes were completely attenuated by a Wnt/β-catenin inhibitor (XAV939). Similar to XAV939, a c-myc inhibitor (10058-F4) also significantly attenuated STK31-induced proliferation and cell cycle progression in lung cancer cells. Inhibiting c-myc and TRRAP significantly decreased the expression of STK31, and a chromatin immunoprecipitation (ChIP) assay confirmed that c-myc directly bound to the STK31 promoter. These results indicated that STK31 may act as an oncogene in lung cancer and that c-myc may be the transcription factor that promotes STK31 expression. Moreover, the results suggested that c-myc can also regulate STK31 expression in a positive feedback loop, and the downregulation of STK31 in lung cancer cells had an inhibitory effect on cell viability, cell proliferation and cell cycle progression, likely by inactivating the Wnt/β-catenin pathway and positive feedback regulation by c-myc.
Keywords: serine-threonine kinase 31, lung cancer, proliferation, cell cycle, Wnt/β-catenin pathway
Introduction
Lung cancer is a leading cause of cancer-related deaths worldwide, accounting for 18% of cancer-related deaths in 2008 (1); The incidence ratio between men and women is 2.1:1 (2,3). Typically, lung cancer is classified into two main types, namely, small-cell and non-small-cell cancers (4), and tobacco smoke has been revealed to be the primary cause of lung cancer, causing ~85% of all cases of lung cancer (5). Due to the lack of observable symptoms at the early stages, the long-term prognosis of lung cancer is poor, with a low 5-year relative survival of 6–14% for men and 7–18% for women (6).
The Wnt/β-catenin pathway is normally inactive in many tissues in adults (7), and inappropriate activation is thought to be the initiating event in intestinal epithelial cell transformation (8). Intracellularly, Wnt signaling is transduced by disheveled (Dsh) proteins, leading to the accumulation of β-catenin in the cytosol, which then translocates to the nucleus to form complexes with transcription factors, such as the T-cell factor family proteins (TCFs). These transcription factors transactivate many target genes, such as the oncogenes c-myc and cyclin D1, which regulate cell proliferation, development and genes involved in tumorigenesis (8–10). A previous study revealed that the Wnt/β-catenin signaling cascade plays a key role in cancer (11), and Wnt family genes have been shown to be upregulated in many cancers, including lung cancer (12,13). In addition, it has been revealed that the metastasis of lung tumor cell lines was enhanced by increased Wnt/β-catenin signaling (14).
Serine-threonine kinases (STKs) comprise a primary family of kinases in the human kinase group, and their expression has frequently been revealed to be altered in human cancers, suggesting a key role for the STK family in cancer development (15,16). STK31, which is a member of the STK family, is a novel cancer/testis (CT)-related gene that is critical in human cancers. It has been revealed that STK31 regulates the cell cycle phases and is highly expressed in several types of cancers, including lung and colorectal cancers (17). The dysregulated expression of cell cycle kinases has been revealed to lead to uncontrolled cell proliferation and genomic instability, both of which are hallmarks of carcinogenesis (18). As a cell cycle-regulated protein, STK31 has been reported to contribute to the tumorigenicity of epithelial cancer, and overexpression of STK31 promoted cell migration and invasion, whereas STK31 knockdown induced apoptosis (17). In addition, STK31 has been revealed to be a novel biomarker for the risk of colorectal cancer metastasis (19,20). However, the roles and underlying mechanisms of STK31 in lung cancer cells remain unclear.
In the present study, analysis of the lung cancer The Cancer Genome Atlas (TCGA) dataset revealed that STK31 was highly expressed in lung cancer, and the Wnt/β-catenin pathway was positively correlated with STK31 expression, which is consistent with the high expression of STK31 and β-catenin that is typically observed in lung cancer patients. Downregulation of STK31 in lung cancer cells significantly suppressed cell proliferation by blocking cell cycle progression, whereas upregulation of STK31 resulted in the opposite effect. In addition, a Wnt/β-catenin inhibitor (XAV939) completely attenuated the effects of STK31 on the lung cancer cells, and a c-myc inhibitor had an effect similar to that of XAV939. A chromatin immunoprecipitation (ChIP) assay confirmed that c-myc directly bound to the STK31 promoter. These findings indicated that STK31 may be an oncogene in lung cancer and involved in the Wnt/β-catenin pathway and c-myc expression.
Materials and methods
Tissue samples of lung cancer and normal lung
After written informed consent was obtained, 30 patients with lung cancer who were treated at Linyi Central Hospital (Shandong, China) aged 55–80 years old were enrolled in the study. Tissue samples of lung cancer and normal lung were collected and frozen in liquid nitrogen, followed by RNA and protein extraction. The expression of STK31 and β-catenin in tissue samples was determined by real-time PCR and western blot analysis. All experiments performed in the present study were approved by the Research Ethics Committee of Linyi Central Hospital of Shandong (Linyi, China).
Cell culture
Seven human lung cancer cell lines (A549, NCIH1299, CALU3, NCIH23, NCIH358, NCIH1650 and NCIH1975) and 16HBE, a normal lung cell line, were purchased from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). All cell lines were cultured in an incubator at 37°C with 5% CO2 (Thermo Forma cat. no. 3111; Thermo Fisher Scientific, Inc., Waltham, MA, USA) in RPMI-1640 medium (cat. no. SH30809.01B; HyClone, Logan, UT, USA) containing 10% fetal bovine serum (FBS; cat. no. 16000-044; Gibco; Thermo Fisher Scientific, Inc.) and 1% antibiotic (penicillin and streptomycin; cat. no. P1400-100; Beijing Solarbio Science & Technology Co., Ltd., Shanghai, China). The medium was refreshed every two days during culturing.
Construction of the lentivirus
Specific shRNAs targeting various sites of the STK31 gene (cat. no. AF285599.1; as shown in Table I) were synthesized, and then inserted into the pLKO.1-puro vector using the restriction sites AgelI and EcolI followed by transformation and plasmid extraction. Meanwhile, through Genewiz, Inc. (South Plainfield, NJ, USA), the coding DNA sequence (CDS) region of STK31 (containing EcoRI/BamHI sites, full-length 3,060 bp) was synthesized, and then was inserted into the pLVX-Puro vector using the EcoRI/BamHI restriction sites. After the constructs were sequenced (Shanghai Majorbio Pharmaceutical Technology Co., Ltd., Shanghai, China), pLKO.1-shSTK31 or pLVX-Puro-STK31 was co-transfected into 293T cells with the viral packaging plasmids psPAX2 and pMD2G (Addgene, Inc., Cambridge, MA, USA) using Lipofectamine 2000 (Invitrogen; Thermo Fisher Scientific, Inc.). The viral particles in the supernatant of the medium were collected 48 h post-transfection.
Table I.
STK31 interference sequences.
| Name | Sequences |
|---|---|
| STK31 target site 1 | GCTGCTGTGGATTTGACTA |
| (1045–1063) | |
| STK31 target site 2 | GGAGATAGCTCTGGTTGAT |
| (1713–1731) | |
| STK31 target site 3 | GCTGCGCAATAATGTCTTT |
| (2034–2052) |
STK31, serine-threonine kinase 31.
Experimental grouping
In vitro, the expression of STK31 in lung cancer cells was regulated by lentivirus-mediated RNA interference or overexpression. NCIH1299 cells were infected with lentiviruses carrying the STK31 overexpression vector (STK31), whereas NCIH358 and NCIH1975 cells were infected with lentiviruses carrying the negative control (shNC)/STK31 interference (shSTK31-1, shSTK31-2 and shSTK31-3). Cells treated with RPMI-1640 medium alone served as an additional control. After 48 h of infection, the efficiency of the STK31/shSTK31 lentivirus infection was determined by real-time PCR and western blot analysis.
Following infection with shNC, shSTK31-1 and shSTK31-2, including medium-treated cells as a control, the cell viability and proliferation of NCIH358 or NCIH1975 cells at 0, 24, 48 and 72 h were evaluated. The cell cycle and expression levels of several related genes were also analyzed. In addition, NCIH1299 cells were treated with vector + dimethyl sulfoxide (DMSO), vector + 10 µmol/l XAV939 (a Wnt/β-catenin inhibitor; cat. no. S1180; Selleck Chemicals, Houston, TX, USA), or STK31 + DMSO, STK31 + 10 µmol/l XAV939, and NCIH358 cells were treated with vector + DMSO, vector + 100 µmol/l 10058-F4 (a c-myc inhibitor; cat. no. S7153; Selleck Chemicals), STK31 + DMSO, or STK31 + 100 µmol/l 10058-F4. After each treatment, the cell viability, proliferation, cell cycle and expression levels of several related genes of the NCIH1299 or NCIH358 cells were evaluated.
Real-time polymerase chain reaction (RT-PCR) analysis
Using TRIzol reagent (1596–026; Invitrogen; Thermo Fisher Scientific, Inc.), total RNA was extracted from tissues or cells, and then quantified. Following the confirmation of the RNA quality, reverse transcription was performed using a reverse transcription kit (cat. no. K1622; Fermentas; Thermo Fisher Scientific, Inc.) to obtain complementary DNA (cDNA). Using the cDNA as a template, RT-PCR reactions were performed in triplicate with a SYBR-Green PCR kit (cat. no. K0223; Thermo Fisher Scientific, Inc.) on an ABI-7300 Real-Time PCR system (Applied Biosystems; Thermo Fisher Scientific, Inc.). The thermocycling program for the RT-PCR reactions was as follows: 95°C, 10 min (95°C, 15 sec; 60°C, 45 sec) ×40 cycles; 95°C, 15 sec; 60°C, 1 min; 95°C, 15 sec; 60°C, 15 sec (21). Subsequently, the mRNA expression of STK31 and β-catenin in tissues and cells was calculated using the 2−ΔΔCq method (22). Gene expression was normalized to that of the internal reference, GAPDH. The primers used were as follows: STK31 forward (F), 5′-TGAACTCTGGTGGTCTCCTTAC-3′ and reverse (R), 5′-CTTGGCTTCTGTGTCAACATCC-3′; β-catenin F, 5′-CGACACCAAGAAGCAGAGATG-3′ and R, 5′-GGGACAAAGGGCAAGATTTCG-3′; transformation/transcription domain-associated protein (TRRAP) forward, 5′-GCCCGACTTCCTCTACGACC-3′ and reverse, 5′-GCGACTCCTTCAGCATCTTCC-3′; and GAPDH forward, 5′-AATCCCATCACCATCTTC-3′ and reverse, 5′-AGGCTGTGTCATACTTC-3′.
Western blot analysis
Total protein was extracted from tissues or cells using RIPA buffer (cat. no. R0010; Beijing Solarbio Science & Technology Co., Ltd.) supplemented with protease and phosphatase inhibitors. Following protein quantification using the BCA quantification kit (cat. no. PICPI23223; Thermo Fisher Scientific, Inc.), 25 µg protein was separated by 10 or 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), and then semi-transferred onto polyvinylidene fluoride (PVDF) membranes (cat. no. HATF00010; EMD Millipore, Billerica, MA, USA). Subsequently, the membranes were blocked at room temperature for 1 h in 5% skim milk (BYL40422; BD Biosciences, Franklin Lakes, NJ, USA) followed by overnight incubation with primary antibodies against STK31 (dilution 1:1,000; cat. no. ab69678; Abcam, Cambridge, MA, USA), β-catenin (dilution 1:5,000; cat. no. ab32572; Abcam), c-myc (dilution 1:1,000; cat. no. ab32072; Abcam), cyclin D1 (dilution 1:10,000; cat. no. ab134175; Abcam) or GAPDH (dilution 1:1,000; cat. no. 5174; Cell Signaling Technology Inc., Danvers, MA, USA) at 4°C with gentle shaking. Next, after 5–6 washes with Tris-buffered saline with Tween-20 (TBST), the membranes were incubated with HRP-labeled goat anti-rabbit (cat. no. A0208) and goat anti-mouse (cat. no. A0216) secondary antibodies (dilution 1:1,000; Beyotime Institute of Biotechnology, Haimen, China) for 2 h at room temperature. Finally, the blots were developed with the chemiluminescent reagent (cat. no. WBKLS0100; EMD Millipore) for 5 min after being washed with TBST, and the blots were exposed on an ECL imaging system (Tanon 5200; Tanon Science and Technology Co., Ltd., Shanghai, China). Protein expression relative to GAPDH was analyzed and calculated using ImageJ software version 1.47v (NIH; National Institutes of Health, Bethesda, MD, USA).
Cell viability and proliferation assay
A Cell Counting Kit-8 (CCK-8; CP002; SAB Biotherapeutics, Inc., Sioux Falls, SD, USA) assay was used to analyze the viability of the lung cancer cells. NCIH358, NCIH1975 and NCIH1299 cells were seeded at a density of 3,000 cells/well into 96-well plates (TR4001; Trueline Inc., Romeoville, IL, USA) and cultured overnight. The following day, the three types of cells were treated as described above in the ‘experimental grouping’ section. After 0, 24, 48 and 72 h of treatment, 100 µl of CCK-8 solution (CCK-8: serum-free medium = 1:10) was added followed by 1 h of incubation at 37°C. Subsequently, using a microplate reader, the absorbance value (OD) of the lung cancer cells at 450 nm (CCK-8) was measured (DNM-9602; Beijing Perlong Medical Instrument Ltd., Beijing, China). In addition, the proliferation of the lung cancer cells was assessed by bromodeoxyuridine (BrdU)-enzyme-linked immunosorbent assay (ELISA) using a BrdU Cell Proliferation ELISA kit (cat. no. Ab126556; Abcam) according to the manufacturer's protocol.
Cell cycle detection
NCIH358, NCIH1975 and NCIH1299 cells in the logarithmic growth phase were trypsinized, resuspended at a density of 500,000 cells/well and then inoculated in 6-well plates. Following overnight incubation in a 5% CO2 incubator at 37°C, the three types of cells were divided and treated as described above in the ‘Experimental grouping’ section. After treatment, all the cells were collected and centrifuged for 5 min at 1,000 × g, followed by resuspension in 300 µl phosphate-buffered saline (PBS) containing 10% FBS (16000-044; Gibco; Thermo Fisher Scientific, Inc.). Next, the cells were fixed in 700 µl absolute ethanol (precooled to 20°C) at 4°C for 24 h, and then centrifuged at 1,000 × g for 5 min. After being washed with 1 ml PBS (precooled), the fixed cells were slowly and fully resuspended in 100 µl RNase A solution (1 mg/ml; R8020-25; Beijing Solarbio Science & Technology Co., Ltd.) and incubated at 37°C for 30 min in the dark. Following a 10 min incubation in 400 µl propidium iodide (PI) solution (50 µg/ml; C001-200; 7Sea Biotech, Shanghai, China) in the dark, red fluorescence was detected at 488 nm (excitation wavelength) by flow cytometry (Accuri C6; BD Biosciences), and then cell cycle progression was analyzed using FlowJo software (version 7.6.1; Tree Star, Inc., Ashland, OR, USA).
Online dataset analysis
The mRNA expression levels of STK31 in The Cancer Genome Atlas (TCGA) Project for Lung Cancer (https://tcga-data.nci.nih.gov/tcga/; 488 lung cancer tissues and 58 normal lung tissues) were analyzed.
To probe for STK31-associated pathways in lung cancer, the STK31 expression data from the lung cancer TCGA dataset were subjected to Gene Set Enrichment Analysis (GSEA) as previously described (23).
ChIP assay
Cells (~10,000,000) were lysed in sodium dodecyl sulfate (SDS containing lysis buffer) following a 10-min treatment with 1% formaldehyde at room temperature. After sonification, the cell lysates were centrifuged for 10 min at 8,000 × g, and then the supernatant was collected and incubated with the anti-c-myc antibody (1 µg/µl; cat. no. Ab56; Abcam) or the control IgG overnight. Using protein A/G beads, the immune complex was captured, followed by DNA purification. The binding of c-myc to SKT31 was detected using SYBR-Green Real-Time PCR with the following STK31 promoter primers: STK31 promoter forward (F), 5′-AGATTAGGAAAGACCACGAA-3′ and reverse (R), 5′-GACCCTGGACCCACATAC-3′.
Statistical analysis
All statistical analyses were performed with GraphPad Prism 7.0 software (GraphPad Software, Inc., San Diego, CA, USA). The data were presented as the mean ± standard deviation (SD), and the experiments were performed in triplicate. A two-tailed Student's t-test was used to examine the differences between groups, and comparisons between multiple groups were determined by one-way analysis of variance (ANOVA) followed by Tukey's multiple comparison. Differences were considered statistically significant at P<0.05.
Results
Analysis of the available STK31 expression data from the lung cancer TCGA dataset
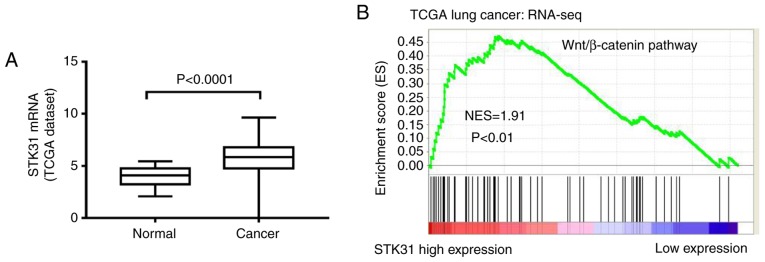
The analysis of the available expression data from 488 cancer tissues and 58 normal samples in the lung cancer TCGA dataset revealed that the expression of STK31 was significantly increased in lung cancer tissues compared to normal tissues (Fig. 1A), suggesting that STK31 may function as an oncogene in lung cancer. Furthermore, to probe the STK31-associated pathways in lung cancer, we performed GSEA using lung cancer samples from the TCGA dataset. As revealed in Fig. 1B, the Wnt/β-catenin pathway was identified as being positively correlated with STK31 expression.
Figure 1.
Analysis of the available STK31 expression data from the lung cancer TCGA dataset. (A) Analysis of the mRNA expression of STK31 in 488 cancer tissues and 58 normal tissues from the lung cancer TCGA dataset (P<0.0001). (B) GSEA analysis identified a positive correlation between STK31 expression and the Wnt/β-catenin pathway in the TCGA lung cancer dataset. NES, normalized enrichment score (P<0.01); STK31, serine-threonine kinase 31; TCGA, The Cancer Genome Atlas; GSEA, Gene Set Enrichment Analysis.
The expression of STK31 and β-catenin is significantly increased in lung cancer tissues
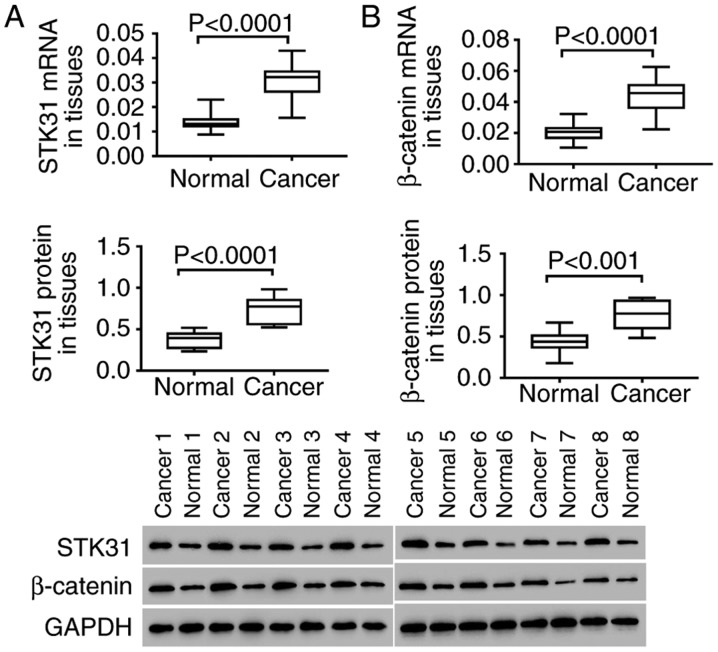
In vitro, the expression of STK31 and β-catenin was analyzed in lung cancer and normal tissues. As revealed in Fig. 2, both the mRNA (upper) and protein (lower) expression of STK31 were significantly and highly expressed in the tumors of 30 lung cancer patients compared to normal lung tissues (Fig. 2A). In addition, high expression of β-catenin was also observed in lung cancer tissues (Fig. 2B). These findings were consistent with the results of the analysis of lung cancer from the TCGA dataset, further suggesting the critical role of STK31 in lung cancer, which may involve the Wnt/β-catenin pathway.
Figure 2.
The expression of STK31 and β-catenin is significantly increased in lung cancer tissues. Following the extraction of mRNA and proteins from cancer and normal tissues (30 pairs) (A) the mRNA and protein expression of STK31 was analyzed (P<0.0001); and (B) the mRNA and protein levels of β-catenin were determined (P<0.001). STK31, serine-threonine kinase 31.
STK31 is increased in lung cancer cells, and the expression of STK31 is regulated by lentiviral infection
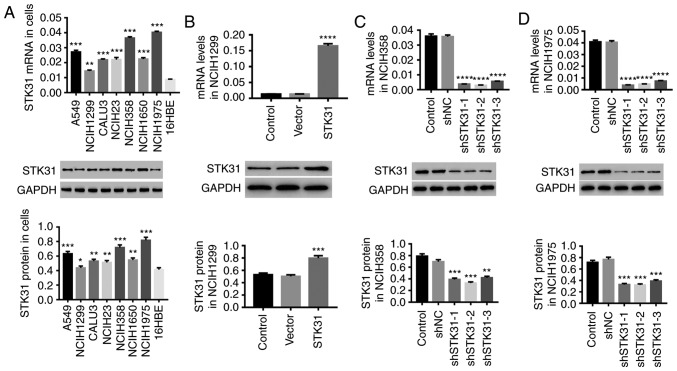
We examined the expression of STK31 in seven lung cancer cell lines (A549, NCIH1299, CALU3, NCIH23, NCIH358, NCIH1650 and NCIH1975) and one lung epithelial cell line, 16HBE. The results presented in Fig. 3A revealed a high expression of STK31 in lung cancer cell lines, further suggesting the important role of STK31 in the progression of lung cancer. Compared with the other lung cancer cell lines, STK31 expression was much higher in NCIH358 and NCIH1975 cells but lower in NCIH1299 cells. Therefore, in NCIH358, NCIH1975 and NCIH1299 cells, lentivirus-mediated STK31 interference or overexpression was utilized to regulate STK31 expression. As revealed in Fig. 3B-D, STK31 expression in NCIH1299 cells was significantly upregulated by STK31 lentivirus infection (Fig. 3B) at both the mRNA and protein levels, whereas infection with shSTK31 lentivirus (shSTK31-1, shSTK31-2 and shSTK31-3) significantly downregulated STK31 expression in NCIH358 cells (Fig. 3C) and NCIH1975 cells (Fig. 3D). The vectors shSTK31-1 and shSTK31-2 resulted in better downregulation efficiency and were therefore selected for the following experiments.
Figure 3.
STK31 is increased in lung cancer cells, and STK31 expression is regulated by lentivirus infection. (A) The mRNA (upper) and protein (lower) expression levels of STK31 in A549, NCIH1299, CALU3, NCIH23, NCIH358, NCIH1650, NCIH1975 and 16HBE cells were detected by RT-PCR and western blotting, respectively. Among these lung cancer cell lines, NCIH358, NCIH1299 and NCIH1975 were infected with lentiviruses, and medium-treated cells served as the control. (B) The upregulation efficiency of STK31 lentivirus was analyzed NCIH1299 cells. (C and D) The downregulation efficiency of shSTK31 lentivirus (shSTK31-1, shSTK31-2 and shSTK31-3) was analyzed in NCIH358 and NCIH1975 cells. Data are presented as the mean ± SD, *P<0.05, **P<0.01, ***P<0.001 and ****P<0.0001 compared to 16HBE, vector or shNC. STK31, serine-threonine kinase 31.
The downregulation of STK31 in lung cancer cells suppresses proliferation by blocking cell cycle progression
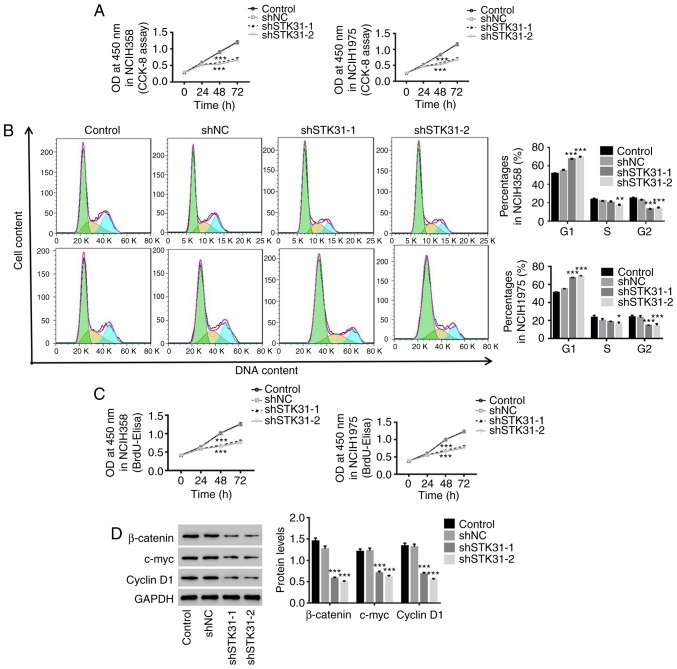
To investigate the effect of STK31 on cell viability, proliferation and cell cycle progression in lung cancer cells, NCIH358 and NCIH1975 cells were infected with the shSTK31 lentivirus. As revealed in Fig. 4, the downregulation of STK31 significantly inhibited the cell viability and proliferation of NCIH358 and NCIH1975 cells (Fig. 4A and C) and arrested the cell cycle in the G1 phase, which reduced the proportion of cells in the S/G2 phase (Fig. 4B). Moreover, there was a decrease in β-catenin, c-myc and cyclin D1 in STK31-silenced lung cancer cells (Fig. 4D). These results indicated that the downregulation of STK31 had a strong inhibitory effect on the proliferation of lung cancer cells by arresting the cell cycle. These findings may contribute to lung cancer treatment.
Figure 4.
The downregulation of STK31 expression in lung cancer cells suppresses proliferation by blocking the cell cycle progression. NCIH358 and NCIH1975 cells were infected with shNC, shSTK31-1 and shSTK31-2, and medium-treated cells served as the control. (A) Cell viability was assessed by CCK-8 assay at 0, 24, 48 and 72 h. (B) The proportion of cells in each phase of the cell cycle was evaluated by flow cytometry after 48 h of infection. (C) Cell proliferation was assessed at 0, 24, 48 and 72 h by BrdU-ELISA. (D) The protein expression of β-catenin, c-myc and cyclin D1 was analyzed by western blotting. All data are presented as the mean ± SD, *P<0.05, **P<0.01 and ***P<0.001 compared to shNC. STK31, serine-threonine kinase 31; CCK-8, Cell Counting Kit-8; BrdU-ELISA, bromodeoxyuridine-enzyme-linked immunosorbent assay.
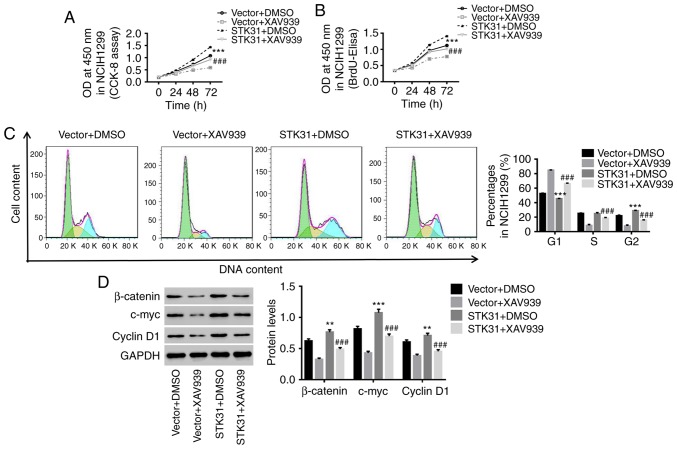
STK31 regulates proliferation and the cell cycle in lung cancer cells via activation of the Wnt/β-catenin pathway
It has been reported that activation of the Wnt/β-catenin pathway is a critical oncogenic event during the initiation and progression of tumors, and the oncogenes c-myc and cyclin D1 are two downstream targets of the Wnt/β-catenin pathway (24). In the present study, the mechanism underlying the STK31-mediated regulation of lung cancer cells was investigated. As revealed in Fig. 5, the upregulation of STK31 significantly increased the viability and proliferation of lung cancer cells and promoted lung cancer cell entry into the S phase from the G1 phase (Fig. 5A-C) accompanied by an increase in the protein expression of β-catenin, c-myc and cyclin D1 (Fig. 5D). In contrast, treatment with XAV939, a Wnt/β-catenin inhibitor, potently attenuated the effects of STK31 in lung cancer cells. These findings were agreement with prior reports that the activation of the Wnt/β-catenin pathway is frequently observed in lung cancer and promotes lung cancer cell proliferation (14,25). Therefore, it can be concluded that c-myc is a downstream target of STK31/β-catenin and that STK31 likely regulates the proliferation and cell cycle progression of lung cancer cells by activating the Wnt/β-catenin pathway.
Figure 5.
STK31 regulates proliferation and the cell cycle in lung cancer cells via the activation of the Wnt/β-catenin pathway. NCIH1299 cells were treated with vector + DMSO, vector + 10 µmol/l XAV939 (a Wnt/β-catenin inhibitor), STK31 + DMSO, or STK31 + 10 µmol/l XAV939. (A and B) Cell viability and proliferation were assessed by CCK-8 and BrdU-ELISA assays, respectively, at 0, 24, 48 and 72 h. (C) The proportion of cells in each phase of the cell cycle was evaluated by flow cytometry after 48 h of treatment. (D) Proteins related to β-catenin, c-myc and cyclin D1 protein levels were detected by western blotting. Data are presented as the mean ± SD, **P<0.01 and ***P<0.001 compared to vector, ###P<0.001 compared to STK31. STK31, serine-threonine kinase 31; DMSO, dimethyl sulfoxide; CCK-8, Cell Counting Kit-8; BrdU-ELISA, bromodeoxyuridine-enzyme-linked immunosorbent assay.
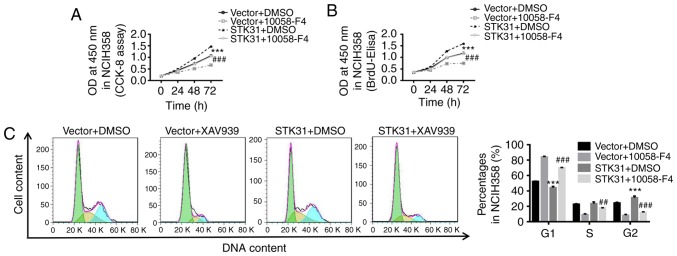
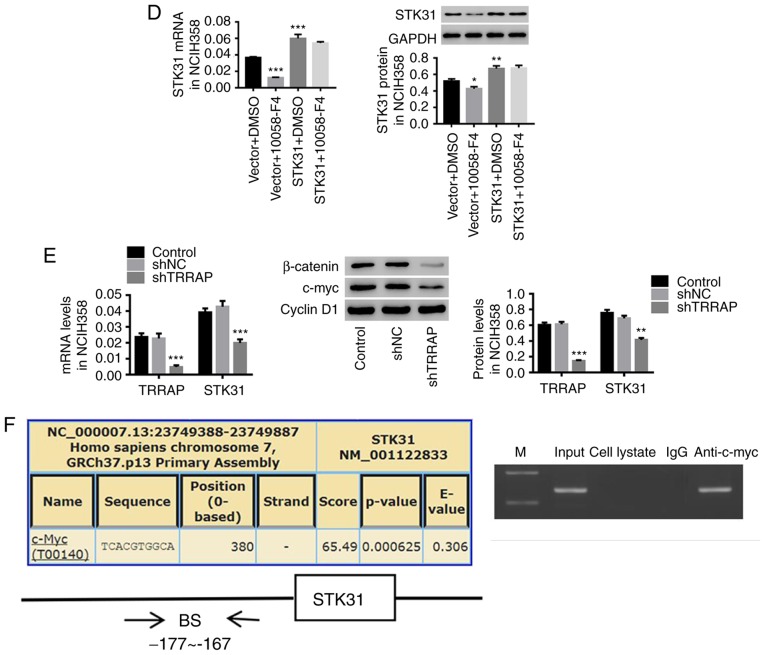
STK31-mediated regulation of proliferation and the cell cycle in lung cancer cells may be achieved through positive feedback regulation by c-myc
Using LASAGNA-Search 2.0 (http://biogrid-lasagna.engr.uconn.edu/lasagna_search/), a c-myc binding site was predicted in the STK31 promoter (−177 to −167) (Fig. 6F). In the present study, we used 10058-F4, a c-myc inhibitor, combined with the STK31 lentivirus to explore the role of c-myc in the regulation of STK31 in lung cancer cells. As revealed in Fig. 6, STK31-induced cell viability, proliferation and cell cycle progression were significantly counteracted by a c-myc inhibitor (Fig. 6A-C). Moreover, the mRNA and protein expression of STK31 were markedly decreased by the c-myc inhibitor (Fig. 6D), indicating feedback regulation of STK31 expression by c-myc. It is known that another protein, TRRAP, interacts with myc and is required for the mitotic checkpoint and normal cell cycle progression (26,27). We found that the knockdown of TRRAP significantly inhibited STK31 expression (Fig. 6E). As shown in Fig. 6F, a ChIP assay confirmed that c-myc directly binds to the STK31 promoter. It could be inferred that c-myc may also act upstream of STK31 and that STK31 potentially regulates proliferation and the cell cycle progression in lung cancer cells through positive feedback regulation by c-myc.
Figure 6.
The STK31-mediated regulation of proliferation and the cell cycle in lung cancer cells may act through the positive feedback regulation by c-myc. NCIH358 cells were treated with vector + DMSO, vector + 100 µmol/l 10058-F4 (a c-myc inhibitor), STK31 + DMSO, or STK31 +100 µmol/l 10058-F4. (A and B) Cell viability and proliferation were assessed by CCK-8 and BrdU-ELISA assays, respectively, at 0, 24, 48 and 72 h. (C) The proportion of cells in each phase of the cell cycle was evaluated by flow cytometry after 48 h of treatment. (D) The levels of STK31 mRNA (left) and protein (right) were determined. (E) The expression levels of TRRAP and STK31 were detected by RT-PCR and western blotting. (F) The direct binding of c-myc to the STK31 promoter was confirmed by a ChIP assay. The data are expressed as the mean ± SD, *P<0.05, **P<0.01 and ***P<0.001, compared to vector or shNC, ##P<0.01, ###P<0.001 compared to STK31. STK31, serine-threonine kinase 31; DMSO, dimethyl sulfoxide; CCK-8, Cell Counting Kit-8; TRRAP, transformation/transcription domain-associated protein; ChIP, chromatin immunoprecipitation; BrdU-ELISA, bromodeoxyuridine-enzyme-linked immunosorbent assay.
Discussion
Emerging research indicates that the STK family of proteins plays a crucial role in the development of lung cancer. STK15 is involved in the initiation and prognosis of lung cancer, and its polymorphisms are reported to be closely associated with the risk of lung cancer (28). STK1, a cell cycle-dependent marker, has prognostic value and is a monitoring factor for NSCLC (29). STK11 is a tumor suppressor gene in lung cancer (30,31), and STK39 acts as a tumor oncogene in NSCLC, promoting cell growth, migration and invasion (32). The results of the present study demonstrated high expression of STK31 and β-catenin in lung cancer tissues and cell lines, suggesting that STK31 may function as an oncogene in lung cancer and may contribute to the carcinogenesis of lung cancer. The downregulation of STK31 in lung cancer cells exerted an inhibitory effect on cell viability, cell proliferation and cell cycle progression. This finding indicated that the downregulation of STK31 could suppress the proliferation of lung cancer cells by blocking cell cycle progression, which may be beneficial for lung cancer treatment.
We also investigated the potential molecular mechanisms through which STK31 regulates lung cancer cell proliferation. The Wnt/β-catenin pathway was identified as being positively correlated with STK31 expression by GSEA using lung cancer samples from the TCGA dataset. This finding was in agreement with previous studies which revealed that lung cancer cell proliferation was related to the activation of the Wnt/β-catenin pathway (33,34). In lung cancer tissues and cells, STK31 and β-catenin were significantly increased, and downregulation of STK31 strongly suppressed the proliferation of lung cancer cells by arresting the cell cycle in the G1 phase concurrent with a decrease in the protein levels of β-catenin, c-myc and cyclin D1, whereas upregulation of STK31 had the opposite effects. c-myc and cyclin D1 are downstream of Wnt/β-catenin and are reported to play crucial roles in cancer (35,36). Overexpression of cyclin D1 was revealed to promote cell cycle progression by inducing cells to enter the S phase (36,37). Our findings demonstrated that STK31 may regulate lung cancer cell proliferation via activation of the Wnt/β-catenin pathway, and c-myc and cyclin D1 may be downstream of STK31/β-catenin. Furthermore, we determined that STK31-induced proliferation, cell cycle progression, and increases in β-catenin, c-myc and cyclin D1 levels in lung cancer cells were potently attenuated by a Wnt/β-catenin inhibitor. These findings further confirmed that the Wnt/β-catenin pathway is involved in the STK3-mediated regulation of lung cancer cell proliferation. STK31 is a serine-threonine kinase that can activate Wnt signaling by phosphorylation and prevent GSK-3β activation, leading to the accumulation of stable β-catenin, which regulates the expression of downstream genes, such as c-myc and cyclin D1, thereby regulating cell proliferation and cell cycle progression. In addition, the inhibition of c-myc strongly suppressed the STK-induced proliferation of lung cancer cells by promoting progression from the G to the S phase and by significantly decreasing the expression of STK31. Likewise, knockdown of TRRAP, a protein that interacts with myc, also significantly decreased STK31 expression. In agreement with the prediction of the software analysis, the ChIP assay confirmed that c-myc directly binds to the STK31 promoter. These findings indicated that c-myc may be an upstream transcription factor of STK31. We inferred that positive feedback regulation of c-myc may also be involved in STK31-mediated effects on the proliferation and cell cycle progression of lung cancer cells, which was consistent with a previous study that revealed that Wnt signaling-mediated oncogenesis was associated with the induction of c-myc in cancer (38). Collectively, these results demonstrated that STK31 may act as an oncogene in lung cancer and that downregulation of STK31 in lung cancer cells could suppress cell proliferation by arresting the cell cycle in the G1 phase, possibly by inactivating the Wnt/β-catenin pathway and by positive feedback regulation by c-myc.
In conclusion, the present study demonstrated that STK31 may be an oncogene in lung cancer. Downregulation of STK31 could potentially inhibit the proliferation of lung cancer cells by blocking the cell cycle progression, possibly through inactivation of the Wnt/β-catenin pathway and through the positive feedback regulation by c-myc. Therefore, the expression of STK31 was revealed to be closely associated with lung cancer risk and targeting STK31 may likely provide a potential therapeutic option for treating lung cancer.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during the present study are included in this published article.
Authors' contributions
PY and JX conceived and designed the study. JX, SX, ZD, LN, QX and PL performed the experiments. PY and JX wrote the manuscript. All authors read and approved the final manuscript and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ethics approval and consent to participate
All experiments conducted in the present study were approved by the Ethics Committee of Linyi Central Hospital (Shandong, China), and written informed consent was obtained.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90. doi: 10.3322/caac.20107. [DOI] [PubMed] [Google Scholar]
- 2.Ferlay J, Shin H, Bray F, Forman D. Cancer incidence and mortality worldwide: IARC cancer base no. 10. Int J Cancer J Int Du Cancer. 2015;136:E359–E386. doi: 10.1002/ijc.29210. [DOI] [PubMed] [Google Scholar]
- 3.Bray F, Jemal A, Grey N, Ferlay J, Forman D. Global cancer transitions according to the human development index (2008–2030): A population-based study. Lancet Oncol. 2012;13:790–801. doi: 10.1016/S1470-2045(12)70211-5. [DOI] [PubMed] [Google Scholar]
- 4.Hoffman PC, Mauer AM, Vokes EE. Lung cancer. Lancet. 2000;355:479–485. doi: 10.1016/S0140-6736(00)82038-3. [DOI] [PubMed] [Google Scholar]
- 5.Fucito LM, Czabafy S, Hendricks PS, Kotsen C, Richardson D, Toll BA, Association for the Treatment of Tobacco Use and Dependence/Society for Research on Nicotine and Tobacco Synergy Committee Pairing smoking-cessation services with lung cancer screening: A clinical guideline from the association for the treatment of tobacco use and dependence and the society for research on nicotine and tobacco. Cancer. 2016;122:1150–1159. doi: 10.1002/cncr.29926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Youlden DR, Cramb SM, Baade PD. The international epidemiology of lung cancer: Geographical distribution and secular trends. J Thorac Oncol. 2008;3:819–831. doi: 10.1097/JTO.0b013e31818020eb. [DOI] [PubMed] [Google Scholar]
- 7.Peifer M, Polakis P. Wnt signaling in oncogenesis and embryogenesis-A look outside the nucleus. Science. 2000;287:1606–1609. doi: 10.1126/science.287.5458.1606. [DOI] [PubMed] [Google Scholar]
- 8.Dihlmann S, von Knebel Doeberitz M. Wnt/β-catenin-pathway as a molecular target for future anti-cancer therapeutics. Int J Cancer. 2005;113:515–524. doi: 10.1002/ijc.20609. [DOI] [PubMed] [Google Scholar]
- 9.He TC, Sparks AB, Rago C, Hermeking H, Zawel L, da Costa LTD, Morin PJ, Vogelstein B, Kinzler KW. Identification of c-MYC as a target of the APC pathway. Science. 1998;281:1509–1512. doi: 10.1126/science.281.5382.1509. [DOI] [PubMed] [Google Scholar]
- 10.Tetsu O, Mccormick F. Beta-catenin regulates expression of cyclin D1 in colon carcinoma cells. Nature. 1999;398:422–426. doi: 10.1038/18884. [DOI] [PubMed] [Google Scholar]
- 11.Polakis P. Wnt signaling and cancer. Genes Dev. 2000;14:1837–1851. [PubMed] [Google Scholar]
- 12.Chen G, Shukeir N, Potti A, Sircar K, Aprikian A, Goltzman D, Rabbani SA. Up-regulation of Wnt-1 and beta-catenin production in patients with advanced metastatic prostate carcinoma: Potential pathogenetic and prognostic implications. Cancer. 2004;101:1345–1356. doi: 10.1002/cncr.20518. [DOI] [PubMed] [Google Scholar]
- 13.Zhang WM, Lo Muzio L, Rubini C, Yan G. Effect of WNT-1 on β-catenin expression and its relation to Ki-67 and tumor differentiation in oral squamous cell carcinoma. Oncol Rep. 2005;13:1095–1099. [PubMed] [Google Scholar]
- 14.Nguyen DX, Chiang AC, Zhang XHF, Kim JY, Kris MG, Ladanyi M, Gerald WL, Massagu J. WNT/TCF signaling through LEF1 and HOXB9 mediates lung adenocarcinoma metastasis. Cell. 2009;138:51–62. doi: 10.1016/j.cell.2009.04.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Baselga J. Targeting tyrosine kinases in cancer: The second wave. Science. 2006;312:1175–1178. doi: 10.1126/science.1125951. [DOI] [PubMed] [Google Scholar]
- 16.Krause DS, Van Etten RA. Tyrosine kinases as targets for cancer therapy. N Engl J Med. 2005;353:172–187. doi: 10.1056/NEJMra044389. [DOI] [PubMed] [Google Scholar]
- 17.Kuo PL, Huang YL, Hsieh CC, Lee JC, Lin BW, Hung LY. STK31 is a cell cycle regulated protein that contributes to the tumorigenicity of epithelial cancer cells. PLoS One. 2014;9:e93303. doi: 10.1371/journal.pone.0093303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hanahan D, Weinberg RA. Hallmarks of cancer: The next generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 19.Zhong L, Liu J, Hu Y, Wang W, Xu F, Xu W, Han J, Biskup E. STK31 as novel biomarker of metastatic potential and tumorigenicity of colorectal cancer. Oncotarget. 2017;8:24354–24361. doi: 10.18632/oncotarget.15396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Watany MM, Elmashad NM, Badawi R, Hawash N. Serum FBLN1 and STK31 as biomarkers of colorectal cancer and their ability to noninvasively differentiate colorectal cancer from benign polyps. Clin Chim Acta. 2018;483:151–155. doi: 10.1016/j.cca.2018.04.038. [DOI] [PubMed] [Google Scholar]
- 21.Hong JY, Kang B, Kim A, Hwang S, Ahn J, Lee S, Kim J, Park JH, Cheon DS. Development of a highly sensitive real-time one step RT-PCR combined complementary locked primer technology and conjugated minor groove binder probe. Virol J. 2011;8:330–330. doi: 10.1186/1743-422X-8-330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 23.Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, Paulovich A, Pomeroy SL, Golub TR, Lander ES, Mesirov JP. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA. 2005;102:15545–15550. doi: 10.1073/pnas.0506580102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Vogelstein B, Kinzler KW. Cancer genes and the pathways they control. Nature Med. 2004;10:789–799. doi: 10.1038/nm1087. [DOI] [PubMed] [Google Scholar]
- 25.Lim JH, Park JW, Chun YS. Human arrest defective 1 acetylates and activates β-Catenin, promoting lung cancer cell proliferation. Cancer Res. 2006;66:10677. doi: 10.1158/0008-5472.CAN-06-3171. [DOI] [PubMed] [Google Scholar]
- 26.Bouchard C, Dittrich O, Kiermaier A, Dohmann K, Menkel A, Eilers M, Lüscher B. Regulation of cyclin D2 gene expression by the Myc/Max/Mad network: Myc-dependent TRRAP recruitment and histone acetylation at the cyclin D2 promoter. Genes Dev. 2001;15:2042–2047. doi: 10.1101/gad.907901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bhattacharya S, Ghosh MK. HAUSP regulates c-MYC expression via de-ubiquitination of TRRAP. Cell Oncol (Dordr) 2015;38:265–277. doi: 10.1007/s13402-015-0228-6. [DOI] [PubMed] [Google Scholar]
- 28.Gu J, Gong Y, Huang M, Lu C, Spitz MR, Wu X. Polymorphisms of STK15 (Aurora-A) gene and lung cancer risk in Caucasians. Carcinogenesis. 2007;28:350–355. doi: 10.1093/carcin/bgl149. [DOI] [PubMed] [Google Scholar]
- 29.Li H, Lei DS, Wang XQ, Skog S, He Q. Serum thymidine kinase 1 (STK1) is a prognostic and monitoring factor in patients with non-small-cell lung cancer. Oncol Rep. 2005;13:145–149. [PubMed] [Google Scholar]
- 30.Carretero J, Medina PP, Pio R, Montuenga LM, Sanchez-Cespedes M. Novel and natural knockout lung cancer cell lines for the LKB1/STK11 tumor suppressor gene. Oncogene. 2004;23:4037–4040. doi: 10.1038/sj.onc.1207502. [DOI] [PubMed] [Google Scholar]
- 31.Gill RK, Yang SH, Meerzaman D, Mechanic LE, Bowman ED, Jeon HS, Roy Chowdhuri S, Shakoori A, Dracheva T, Hong KM, et al. Frequent homozygous deletion of the LKB1/STK11 gene in non-small cell lung cancer. Oncogene. 2011;30:3784–3791. doi: 10.1038/onc.2011.98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Li Z, Zhu W, Xiong L, Yu X, Chen X, Lin Q. Role of high expression levels of STK39 in the growth, migration and invasion of non-small cell type lung cancer cells. Oncotarget. 2016;7:61366–61377. doi: 10.18632/oncotarget.11351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gao Y, Song C, Hui L, Li CY, Wang J, Tian Y, Han X, Chen Y, Tian DL, Qiu X, Wang E. Overexpression of RNF146 in non-small cell lung cancer enhances proliferation and invasion of tumors through the Wnt/β-catenin signaling pathway. PLoS One. 2014;9:e85377. doi: 10.1371/journal.pone.0085377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wan L, Zhang L, Fan K, Cheng ZX, Sun QC, Wang JJ. Circular RNA-ITCH suppresses lung cancer proliferation via inhibiting the Wnt/β-catenin pathway. Biomed Res Int. 2016;2016:1579490. doi: 10.1155/2016/1579490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Miller DM, Thomas SD, Islam A, Muench D, Sedoris K. c-Myc and cancer metabolism. Clin Cancer Res. 2012;18:5546–5553. doi: 10.1158/1078-0432.CCR-12-0977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hall M, Peters G. Genetic alterations of cyclins, cyclin-dependent kinases, and Cdk inhibitors in human cancer. Adv Cancer Res. 1996;68:67–108. doi: 10.1016/S0065-230X(08)60352-8. [DOI] [PubMed] [Google Scholar]
- 37.Lukas J, Bartkova J, Rohde M, Strauss M, Bartek J. Cyclin D1 is dispensable for G1 control in retinoblastoma gene-deficient cells independently of cdk4 activity. Mol Cell Biol. 1995;15:2600–2611. doi: 10.1128/MCB.15.5.2600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.You Z, Saims D, Chen S, Zhang Z, Guttridge DC, Guan KL, Macdougald OA, Brown AM, Evan G, Kitajewski J, Wang CY. Wnt signaling promotes oncogenic transformation by inhibiting c-Myc-induced apoptosis. J Cell Biol. 2002;157:429–440. doi: 10.1083/jcb.200201110. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during the present study are included in this published article.