Abstract
Libidibia ferrea (juca) is a plant belonging to the Fabaceae (Leguminosae) family, whose antioxidant activity has been widely described in the literature. We evaluated this parameter of Aqueous ethanol extract (AE), ethyl acetate (ACO), chloroform (CLO) and hexane (HEX) extracts of L. ferrea. We then tested the most active extract for its toxicity and ability to inhibit migratory activity in the ACP02 gastric adenocarcinoma cell line in vitro. The AE and ACO extracts both had antioxidant activity, the AE extract showing greater potential. This may reflect that both extracts contained phenolic compounds. Although AE extract showed no cytotoxic, mutagenic or genotoxic effect, it altered cell morphology and migration activity. Analysis of apoptosis/necrosis indicated that this parameter does not appear to account for the apparent ability of AE to inhibit cancer cell migration. We speculate that the morphological changes in AE-treated cells could be due to cytoskeleton alterations related to the presence of myo-inositol in AE extract. Together, our results demonstrate this extract of L. ferrea can act as an exogenous antioxidant and might prove useful in efforts to fight secondary tumors.
1. Introduction
The multiple stages of cancer range from alteration of genetic material to the migration and establishment of altered cells in neighboring tissues and organs. This process of dissemination, known as metastasis, leads to aggravation of the disease and hinders conventional treatment [1]. For this reason, it is important that we identify additional safe and efficient antimetastatic substances.
The accumulation of empirical knowledge regarding medicinal plants has facilitated their increasing use as complementary and alternative therapies for numerous diseases, including cancer [2]. Libidibia ferrea, which is commonly called juca or pau-ferro, belonging to the Fabaceae (Leguminosae) family. The L. ferrea is an Angiosperm medium-size tree previously classified as Caesalpinia ferrea, Caesalpininoideae subfamily and Caesalpinieae tribe [3]. This is a monophyletic family with the following synapomorphies: leaves composite, alternate, with pulvinus; petal differentiated adaxial and monocarpellary ovary [4]. The fruits are usually from the legumen type with some variations [5]. The plants are presented as annual herbs or perennial, erect, prostrate, diffuse, vines, lianas, undergrowth, shrubs and small, medium or large trees [6]. That has been widely utilized in traditional medicine as an antipyretic, antidiabetic and wound-healing agent. Research has shown that this plant has anti-inflammatory, analgesic, anticoagulant, antiulcerogenic, antihistaminic and cancer-preventing activities [7,8]. Furthermore, it has been widely cited in the literature for its antioxidant activity [9,10].
Studies have correlated the antioxidant potential of L. ferrea extracts with the presence of phenols [11,12], which exert antioxidant activities by preventing or retarding oxidation via the blockade/capture of free radicals [13]. It has been proposed that juca can act as an exogenous antioxidant by preventing free radicals from interacting with fundamental molecules of the organism to cause cellular instability and trigger pathologies such as cancer [14]. Thus, the juca became a plant of interest because it is widely used by the population based on empirical knowledge, but without studies related to its activity in cancer cells, including its action in preventing new tumor formation and cell migration.
In vitro assays can be used to examine the safety of plant preparations and their phytochemical constituents [15], while wound-healing assays and other tests can be used to evaluate the ability of plant preparations to inhibit aspects of tumorigenesis. Indeed, many medicinal plants and their constituents have been shown to inhibit the migratory capacity of cancer cells [16,17]. The ACP02 cell line is often used for this type of research [18,19] because it shares important traits with its tumor of origin, including amplification of the MYC oncogene and deletion of the TP53 tumor suppressor gene. As most cancer are characterized by a high degree of metabolic activity, ACP02 cells displays the requirements to be used in the research and a good model for the in vitro screening of anticancer drugs [20,21].
Here, we obtained four extracts from the pods of juca and assessed them for antioxidant activity. The most active extract was tested for its toxicity and inhibition of cell migration in ACP02 cell line.
2. Materials and methods
2.1 Collection of samples
The pods of L. ferrea were collected in the city of Marabá/PA (latitude 05°22’07”S, longitude 49°07’04”W), in July 2014 (authorization number 13248). The plant was identified, by botanist Seidel Santos and a voucher sample (no002780) was deposited in the MFS herbarium of the Universidade do Estado do Pará (UEPA). JCP has a permanent field permit, number 13248 from “Instituto Chico Mendes de Conservação da Biodiversidade”. The Cytogenetics Laboratory from UFPa has permit number 19/2003 from the Ministry of Environment for sample transport and permit 52/2003 for using the samples for research. The Ethics Committee (Comitê de Ética Animal da Universidade Federal do Pará) approved this research (Permit 68/2015).
2.2 Preparation of extracts
Dried and powdered pods (300 g) were subjected to selective extractions with organic solvents in the following order of polarity: n-hexane, chloroform, ethyl acetate and alcohol 70% solution. The solvent: material ratio was 2:1 and the mixture was subjected to the extraction. Ultrasound-assisted extraction was performed in an ultrasonic cleaner bath (USC-1800) with a volume of 9 L, an input power of 155 W, 40 KHz of frequency, and at 30°C (± 3) and 30 min for hexane (HEX), chloroform (CLO) and acetate (ACO) extracts; and 45°C (± 3) and 30 min for aqueous ethanol extract (AE). The ultrasonic power inside de extract container was estimated to 70 W.cm-2. The extracts were concentrated with a Buchi R3 rotary evaporator (V 700 vacuum pump, V 850 vacuum controller) was used to remove the solvent at 45°C and 156 mbar, 207 mbar, 240 mbar, 240 mbar and 58 mbar pressure, respectively [22].
2.3 Chemical characterization of samples
2.3.1 Derivatization
Derivatization was performed as described by [23]. For the dried HA, ACO and CLO extracts, 5 mg of extract was resuspended in 100 μL of the derivatization reagent, N,O-bis(trimethylsilyl)-trifluoroacetamide (BSTFA), with stirring at 600 rpm for 15 min at 45°C. For the HEX extract, 5 mg of dried extract was resuspended in NaOH+MeOH (9:1) at 45°C for 20 min, 500 μL of hexane:ether (1:1) was added, and the mixture was stirred (45°C/5 psi/60 min). The solution was evaporated to dryness, and the lipid residue was resuspended in 100 mL of BSTFA with stirring at 600 rpm/45°C for 15 min.
2.3.2 Metabolite identification (GC/MS)
Gas chromatography/mass spectrometry (GC/MS) was performed following the description of [24]. We used a Thermo Scientific Trace 1300 GC device coupled to a Thermo Scientific MS-ISQ Single Quadrupole mass spectrometer with an AI 1310 autosampler, which was equipped with a RTX-65 TG column (15 m x 0.25 mm x 0.1 μm), a DB-5 column (15 m x 0.25 mm x 0.1 μm) or a similar column. Helium gas was used as the carrier at a flow rate of 1 mL/min. The sample (1.0 μL) was injected in the Split mode at a ratio of 1:5. The injector was operated at 250°C. The oven temperature began at 40°C and then ramped up to 200°C at 6°C/min, remained there for 1 min, increased to 300°C at 15°C/min, remained there for 5 min, increased again to 340°C at 15°C/min, and remained there for 9 min. The MS-ISQ parameters were set as follows: interface, 280°C; ionization source, 280°C; mass range, 40–1000 Da; and electronic ionization, 70 eV. The substances were identified by comparing their mass spectra with those listed in commercial libraries NIST2011-WILEI2009-FAMES2011. The triterpene concentration was determined by calculating the normalized peak area.
2.4 DPPH (2,2-difenil-1-picrilhidazil) test
The antioxidant activities of the four juca extracts were assayed in triplicate using the procedure described by [25] with some modifications. Briefly, DPPH was diluted to 0.04 mg/mL in methanol, and 900 μL of DPPH solution was mixed with 100 μL of extract diluted in methanol to a concentration of 6.25 μg/mL or 400 μg/mL. Each mixture was kept for 20 min in a light-protected place. The absorbance (OD) was measured at 515 nm using a spectrophotometer (Epoch, Biotek) and the Gen5 (2003) version 2.03.01 software. The results were expressed as the percentage of DPPH radical inhibition (%Inhibition) using the following equation: %Inhibition = {[A1-{(A2+A3)]/A1]/100}, where A1 is the DPPH absorbance, A2 is the DPPH absorbance + extract, and A3 is the methanol absorbance + extract. We then calculated the concentration of extract required to capture 50% of the free radical DPPH (EC50), using linear regression analysis performed with the aid of the Graphpad Prism software, version 6.01.
2.5 ABTS [2,2’-azino-bis(3-ethylbenzothiazoline)- 6-sulfonic acid] test
The ABTS antioxidant test was performed according to literature [26] with modifications. ABTS was dissolved in water at a concentration of 7 μM and mixed with Potassium Persulphate at a concentration of 2.45 μM, in the absence of light, at room temperature 12 to 16 h before use for use of radical cation (ABTS +). For the test, an ABTS + solution diluted in water was prepared to an absorbance of 0.700 ± 0.02 at 734 nm. Once the allowed absorbance was reached, 3 ml of the solution was mixed at 100 μl of different filters from each extracted test. After 6 min of reaction the absorbance was read on the spectrophotometer. To calculate the percentage inhibition of the ABTS radical at each concentration, the following equation was used: % inhibition = [(A blank—sample) / A blank] x 100 where "A" is an absorbance of each well and "blank" or one well without medium or cells. The EC50 was estimated as a useful % inhibition at each concentration.
2.6 ORAC (Oxygen Radical Absorbance Capacity) test
The ORAC protocol was adapted from the method previously developed [27] and then modified [28,29] for microplates using fluorescein. The analysis was performed on 96-well fluorimetry microplates (Greiner-Germany) and on a Microplate Fluorescence Reader fluorimeter—Bio-Tek Instruments, Inc (USA). A 25 μL volume of the sample was mixed with 150 μL fluorescein (55.5 nM) and incubated for 15 min at 37°C in the microplate before automatically injecting 25 μL AAPH solution (153 mM). The fluorescence was followed for 50 min by readings (λexcitation = 485 nm; λemission = 520 nm). Trolox solutions were prepared for the calibration curve. All solutions were diluted in phosphate buffer (75 mM, pH 7.4).
2.7 Cell line, culture conditions and biological assays
ACP02 gastric adenocarcinoma cell line was kindly provided by the Laboratory of Human Cytogenetics and Toxicological Genetics (UFPA). The cells were cultured in RPMI supplemented with 10% fetal bovine serum (FBS), amphotericin (2.5 μg/mL), penicillin (100 IU/mL), streptomycin (100 μg/mL), tylosin (8 μg/mL), ciprofloxacin (10 μg/mL), L-glutamine (0.1 mg/L), and sodium bicarbonate (2.2 mg/L). The flasks were maintained in a 5% CO2 incubator at 37°C.
2.7.1 Evaluation of cytotoxicity
Cytotoxicity was evaluated using the MTT test (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) following the protocol described by [30] with alterations. A total of 5x104 cells were seeded to each well of a 96-well plate, which was incubated in a CO2 incubator at 37°C. After 24 h, triplicate samples were tested with seven concentrations of HA extract: 6.25 μg/mL, 12.5 μg/mL, 25 μg/mL, 50 μg/mL, 100 μg/mL, 200 μg/mL and 400 μg/mL. As a negative control (NC), the cells were exposed to the RPMI culture medium. As a positive control (PC) we used doxorubicin at the concentration of 200 μg/mL. As a vehicle control, cells were exposed to 0.1% DMSO in medium. After 24 or 48 h, the cells were exposed to 100 μL of MTT (5 mg/mL) for 3 h, MTT was removed and 100 μl of DMSO was added to each well. After 1 h, the absorbance at 570 nm was measured using a spectrophotometer (Epoch Biotek) and the Gen5 (2003) software, version 2.03.1. Cell viability (%S) was determined from the formula: %S = 100x[(Atested–Ablank)/ (Anegativecontrol-Ablank)], where “A” is the absorbance of each well and “blank” refers to a well without medium or cells.
2.7.2 Evaluation of the genotoxicity and mutagenicity of Jucá HA extract
We evaluated the DNA-level effects of juca AE extract at concentrations of 25 μg/mL, 50 μg/mL, 100 μg/mL and 200 μg/mL, according to the scheme proposed [31]. As a positive control for the micronucleus test, we used colchicine (0.02 μg/mL), as suggested [32]. As a positive control for the comet test, we used H2O2 (100 μM), as proposed [33]. As a negative control, we used RPMI alone. As a vehicle control, we used 0.1% DMSO diluted in medium.
2.7.3 Mutagenicity test: Micronucleus assay
The micronucleus test was performed according to the standards listed in [24]. Cells were seeded to a six-well plate at 2.02 x 105 cells/well, cytochalasin B was added to 6 μg/mL, and the plate was incubated for 24 h. After 24 h of exposure, the cells were trypsinized, transferred to a Falcon tube, and mixed with 5 mL of hypotonic solution (KCl). After 3 min, 2 mL of Carnoy fixative with methanol and acetic acid (3:1) was added. The mixture was centrifuged, the supernatant was mixed with 1 mL of Carnoy fixative, and the tube was stored. This analysis was performed in a blinded manner, and 1000 cells per sample were analyzed. The cytokinesis-block proliferation index (CBPI) was generated using the formula CBPI = [M1+2(M2)+3(M3)+4(M4)]/N, where M1 to M4 represent the numbers of cells with 1, 2, 3 and 4 nuclei, respectively, and N is the total number of viable cells. 500 total cells were counted and the binucleate cell count remained until 1000 cells were counted for micronucleus observation and for determination of its frequency through the formula fMN = n°MN/1000 [34].
2.7.4 Genotoxicity test: Comet assay
The comet assay was performed according to the methodology proposed by [35], with adaptations. A total of 5x105 cells were seeded in 25 cm2 bottles and incubated in CO2 incubators for 24 h. The cells were then exposed to the juca AE extract for 3 h, trypsinized and mixed with 0.5% low-melting-point agarose (20 μL cell suspension in agarose). The mixture was placed on slides pre-coated with normal 1.5% agarose, and coverslips were used to cover the samples. The slides were incubated for 15 min at 4°C and then exposed to lysis solution (2.5 M NaCl, 100 mM EDTA and 10 mM Tris, pH 10.0–10.5) containing 1% Triton X-100 and 10% DMSO. After 24 h, the slides were transferred to a horizontal electrophoresis cube, covered with alkaline buffer (300 mM NaOH and 1 mM EDTA, pH>13) for 30 min, and subjected to electrophoresis for 30 min at 0.8 V/cm. The slides were neutralized by three 5-min washes with deionized water, fixed for 5 min in ethylic alcohol P.A., dried, and stored in a refrigerator until they were subjected to ethidium bromide staining and analyzed under fluorescence microscopy (Nikon H550S with a 510 to 560-nm filter, a 590-nm filter barrier and a zoom of 400x). Comets were categorized based on their tail sizes [36] and 300 cells were analyzed from each group. The damage index (DI) was calculated using the formula: DI = [(1×n1)+(2×n2)+(3×n3)+(4×n4)/n]x100, where n is the total number of analyzed cells and n1 to n4 indicate the numbers of cells with damage levels from 0 to 4.
2.8 Cell migration assay
Cell migration was tested using a modification of the previously described wound-healing assay [37]. Cells were seeded to six-well plates at 2.02 x105 cells/well and incubated for 24 h to allow monolayers to form, and a slit was made in each monolayer. The wounded monolayers were exposed to the AE extract at concentrations of 25 μg/mL, 50 μg/mL, 100 μg/mL and 200 μg/mL. RPMI alone was used as the negative control and 0.1% DMSO in medium was used as the vehicle control. The plates were placed in a CO2 incubator and photographed after 12, 24 and 48 h, and cellular migration was analyzed using the ToupView software, version 3.5. For our statistical analysis, the wounded areas were measured using the ImageJ software. To calculate the slit-opening (SO) percentage, the values measured at the time of wounding (T0) were taken as 100%, while the other percentages were calculated using the formula %SO = CTx * 100%/ CT0, where C represents the length in pixels and Tx is the analyzed time.
2.9 Apoptosis and necrosis test (ao/eb)
The cells were plated and treated with extract as described in section 2.3. RPMI alone was used as a negative control, DMSO diluted in medium was used as the vehicle control, and doxorubicin (100 μg/mL) was used as the positive control. We followed the protocol described by [38] with adaptations. Briefly, at 24 and 48 h post-exposure, the cells were trypsinized and transferred to a Falcon tube. For analysis, acridine orange (100 μg/mL) and ethidium bromide (100 μg/mL) were mixed at a ratio of 1:1, and 2 μL of the mixture was combined with 20 μL of the cell solution on a clean slide. The sample was covered with a coverslip and analyzed under florescence microscopy (Nikon H550S). The test was performed in duplicate and 300 cells were analyzed per sample. The percentage of viable, necrosis and apoptosis cells was calculated from the formula %Cells = (number of cells of interest/total cells analyzed) *100.
2.10 Statistical analysis
Statistical analysis was performed using the Bioestat 5.0 software, with p≤0.05 considered statistically significant for all analyzed parameters. Normality was validated using the Kolmogorov-Smirnov test, and multiple comparisons were performed using the Tukey test followed by ANOVA [39].
3. Results
3.1 Chemical characterization of samples
The four juca extracts were chemically characterized, as presented in Table 1. We found the following: the AE extract contained phenolic compounds and carbohydrates; the ACO extract contained lipids and had a predominance of organic acids; the CLO extract contained organic acids and had a predominance of lipids; and the HEX extract contained some alcohols and had a predominance of lipids.
Table 1. Analysis of the global profile of the extracts composition of the Libidibia ferrea fruit (juca).
| RT | SUBSTANCE | CLASS |
|---|---|---|
| AQUEOUS ETHANOL EXTRACT | ||
| 10.14 | GLYCEROL 3TMS | Alcohol |
| 18.14 | D-FRUTOSE 5TMS | Carbohydrate |
| 18.27 | MIO-INOSITOL 6TMS | Carbohydrate |
| 18.84 | CHEMICAL ACID TMS | Phenolic |
| 19.21 | GLUCOPYRANOSIS 5TMS | Carbohydrate |
| 20.31 | GLICOSE 5TMS | Carbohydrate |
| 25.15 | 1,2-BENZENODYCARBOXYLIC ACID | Phenolic |
| ACETATE EXTRACT | ||
| 7.71 | OXALIC ACID 2TMS | Organic acid |
| 10.14 | GLYCEROL 3TMS | Alcohol |
| 10.69 | BUTANEDIOIC ACID 2TMS | Organic acid |
| 10.87 | PYROTARTARIC ACID 2TMS | Organic acid |
| 12.06 | PENTANOIC ACID 2TMS | Lipid |
| 13.57 | MALIC ACID 3TMS | Organic acid |
| 14.86 | PENTANODIOIC ACID 3TMS | Lipid |
| 15.66 | ARABINOIC ACID 3TMS | Organic acid |
| 16.43 | OCTANEDIOIC ACID 2TMS | Lipid |
| 17.74 | AZELAIC ACID 2TMS | Organic acid |
| 18.12 | D-GALACTOPIRANOSIL | Carbohydrate |
| 18.29 | MIO-INOSITOL 6TMS | Carbohydrate |
| 18.86 | CHEMICAL ACID 5TMS | Phenolic |
| 19.23 | GLICOSE 5TMS | Carbohydrate |
| 19.84 | BENZOIC ACID 4TMS | Phenolic |
| 20.31 | ALPHA-D-GLYCOPYRANOSIS | Carbohydrate |
| 20.6 | PALMITIC ACID TMS | Lipid |
| 22.56 | STEARIC ACID | Lipid |
| 23.96 | ACID 2-BROMOSBACICO 2TMS | Lipid-halogenated |
| 25.12 | 1,2-BENZENODYCARBOXYLIC ACID | Phenolic |
| 27.24 | TETRACOSANOIC ACID TMS | Lipid |
| 28.13 | CHEMICAL ACID 5TMS | Phenolic |
| 30.74 | NO IDENTIFIED | ----- |
| 32.19 | NO IDENTIFIED | ----- |
| CHLOROFORM EXTRACT | ||
| 5.3 | N-VALERIC ACID TMS | Lipid |
| 6.56 | ALPHA-HYDROXY ACID ISOBUTIRICACY 2TMS | Lipid |
| 6.66 | ACIDO CAPROICO TMS | Lipid |
| 7.71 | OXALIC ACID 2TMS | Organic acid |
| 8.21 | HEPTANOIC ACID TMS | Lipid |
| 9.8 | OCTANOIC ACID TMS | Lipid |
| 10.14 | GLYCEROL 3TMS | Alcohol |
| 10.56 | MALEIC ACID 2TMS | Lipid |
| 10.67 | PYROTARTARIC ACID 2TMS | Organic acid |
| 11.36 | PELARGONIC ACID TMS | Organic acid |
| 12.93 | NO IDENTIFIED | ----- |
| 13.57 | MALIC ACID 3TMS | ----- |
| 14.54 | TETRADECANOIC ACID TMS | Lipid |
| 15.11 | PIMELIC ACID 2TMS | Organic acid |
| 16.41 | SUBERICO ACID 2TMS | Organic acid |
| 17.7 | AZELAIC ACID 2TMS | Organic acid |
| 18.3 | MIRISTIC ACID TMS | Lipid |
| 19.22 | D-MANOSE 5TMS | Carbohydrate |
| 19.51 | N-PENTADECANOIC ACID TMS | Lipid |
| 20.63 | PALMITIC ACID TMS | Lipid |
| 21.62 | NO IDENTIFIED | ----- |
| 22.37 | TES CHOLESTEROL | Steroid |
| 22.55 | STEAM ACID TMS | Lipid |
| 23.96 | ACID 2-BROMOSBACICO 2TMS | Lipid-halogenated |
| 25.12 | 1,2-BENZENODYCARBOXYLIC ACID | Phenolic |
| 25.53 | MONOPALMITINE 2TMS | Lipid |
| 25.82 | DOCOSANOIC ACID TMS | Lipid |
| 27.24 | TETRACOSANOIC ACID TMS | Lipid |
| 29.9 | NO IDENTIFIED | ----- |
| 30.74 | NO IDENTIFIED | ----- |
| 32.19 | N-VALERIC ACID TMS | ----- |
| HEXANE EXTRACT | ||
| 9.77 | GLYCEROL 3TMS | Alcohol |
| 14.14 | N-DODECANOL 1TMS | Alcohol |
| 17.89 | MIRISTIC ACID 1TMS | Lipid |
| 18.82 | METILA PALMITATO | Lipid |
| 20.26 | PALMITIC ACID 1TMS | Lipid |
| 20.75 | METILA OIL | Lipid |
| 21.03 | METILA ESTEARATE | Lipid |
| 22.2 | STEAM ACID 1TMS | Lipid |
| 22.91 | METILA ARAQUIDOATE | Lipid |
| 23.61 | NO IDENTIFIED | ----- |
| 23.89 | ARAQUID ACID 1TMS | Lipid |
| 24.46 | NO IDENTIFIED | ----- |
| 24.58 | METILA BEENOATE | ----- |
| 26.09 | METILA LIGNOCERATE | Lipid |
| 26.57 | NO IDENTIFIED | ----- |
| 26.86 | TETRACOSANOIC ACID 1TMS | Lipid |
| 27.27 | NONACOSAN | Lipid |
| 28.86 | OCTACOSANOL 1TMS | Alcohol |
| 29.7 | CAMPESTEROL 1TMS | Steroid |
RT: Retention time (min)
3.2 Antioxidant evaluation
3.2.1 DPPH test
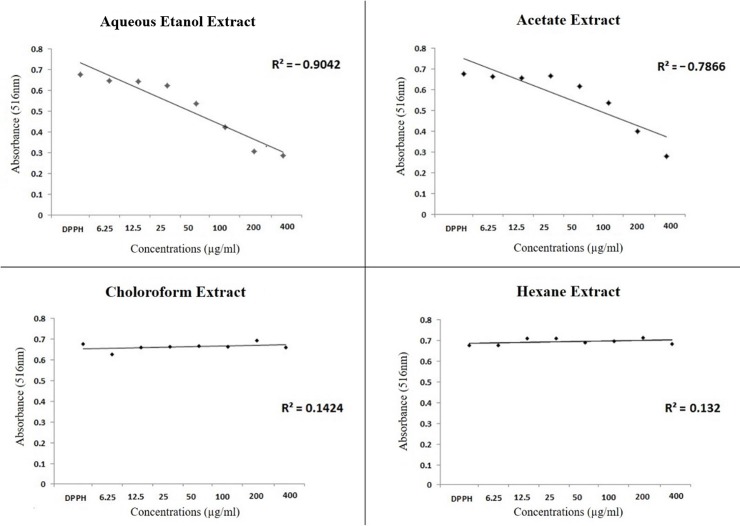
The DPPH free radical-capture test showed that the AE and ACO extracts exhibited dose-dependent antioxidant activity, whereas the CLO and HEX extracts showed no such activity, even at the highest tested concentration (Fig 1). The HA extract showed the highest antioxidant potential among the tested extracts, with an EC50 of 74.36 μg/mL (Table 2).
Fig 1. Correlation between the absorbance values according with the concentrations of the different extracts after reaction with the DPPH reagent.
Linear regression test.
Table 2. Effective concentration capable of capturing 50% (EC50) of free radicals DPPH and ABTS after exposure to four different juca extracts.
| Extract | EC50 (μg/mL) | |
|---|---|---|
| DPPH | ABTS | |
| Aqueous etanol extract | 74.36 | 9,76 |
| Acetate extract | 116.10 | 29,13 |
| Chloroform extract | >400* | >60* |
| Hexane extract | >400* | >60* |
* There was no inhibition until the maximum concentration tested.
3.2.2 ABTS test
The results obtained in the ABTS test corroborate those observed in the DPPH test. The EC50 of the four juca extracts are expressed in Table 2 and show the highest antioxidant activity of the AE extract with EC50 of only 9.76 μg/mL.
3.2.3 ORAC test
The ORAC test confirmed the antioxidant capacity of the juca AE extract with an average of 314.29 μmol Trolox Equivalent / 100 g of extract. Thus, due to the results obtained in antioxidant assays, we focus on AE extract for the following experiments.
3.3 In vitro evaluation
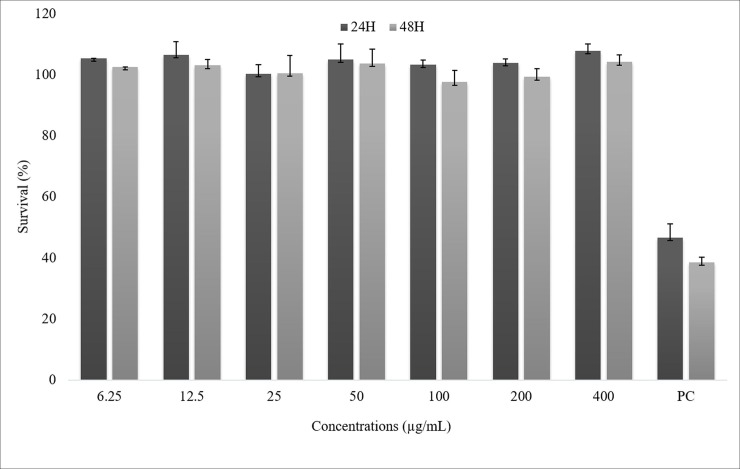
On the cytotoxicity evaluation, the MTT assay demonstrated that the AE extract did not alter cell survival at any tested concentration (Fig 2). The Ao/Eb also did not reveal any significant increase of apoptotic or necrotic cells among the juca AE extract-treated cultures. At 24 hours, all treatment groups differed from the positive control in terms of both necrotic and apoptotic cells. At 48 hours, the treatment groups differed from the positive control group only in terms apoptosis (Table 4).
Fig 2. Percentage of average of survival ACP02 cells by exposure to the aqueous ethanol extract of Libidibia ferrea at 24 and 48 hours.
ANOVA parametric test; Tukey-Kramer Multiple comparisons (p <0.05). In wich NC means negative control and PC means positive control.
Table 4. Average and Standard Deviation (%) of viable, apoptosis and necrosis ACP02 cells at 24 and 48 hours after treatment exposure.
| 24 Hour | 48 Hour | |||||
|---|---|---|---|---|---|---|
| Treatments | %Viable | %Apoptosis | %Necrosis | %Viable | %Apoptosis | %Necrosis |
| NC | 85 (±15) | 6 (±7)a | 9 (±8)a | 98 (±2) | 0 (±0)a | 2 (±2) |
| DMSO (0,1%) | 80 (±16) | 10 (±6)a | 10 (±10)a | 96 (±3) | 0.3 (0.5)a | 4 (±4) |
| PC | 8 (±5) | 50 (±5) | 8 (±10) | 31 (±39) | 56 (±33) | 13 (±5) |
| 25μg/ml | 77(±18) | 10 (±9)a | 15 (±8) | 67 (±2) | 13 (±2) | 6 (±4) |
| 50 μg/ml | 73 (±9) | 27 (±9) | 0 (±0)a | 86 (±6) | 6 (±3) | 8 (±3) |
| 100 μg/ml | 90 (±2) | 3 (±1)a | 7 (±3)a | 81 (±10) | 2 (±0.2)a | 17 (±11) |
| 200 μg/ml | 93 (±2) | 2 (±0.1)a,b | 5 (±3)a | 93(±0.7) | 1 (±0)a | 2 (±0.7) |
ANOVA parametric test; Tukey-Kramer Multiple comparisons (p <0.05); NC means negative control. PC means positive control with doxorubicin.
aDiffers from other treatments.
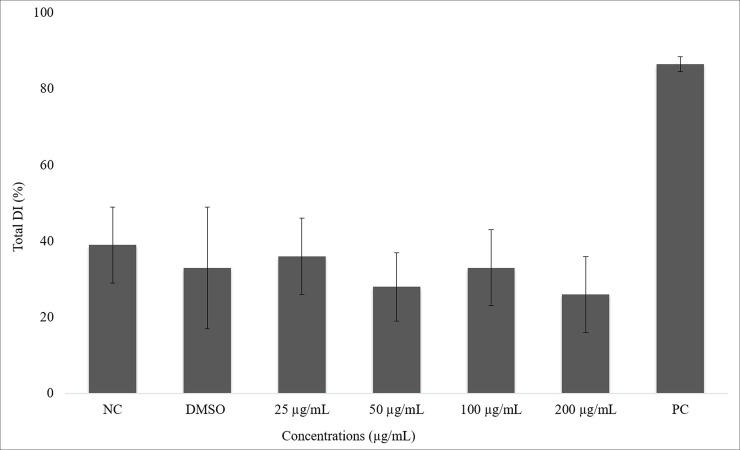
On the genotoxicity evaluation, the micronucleus test reveals that CBPI values did not differ for any of the treatments tested. Regarding the frequency of micronuclei, only the positive control (colchicine-treated) group differed from the other groups (Table 3). The comet assay showed no statistical difference among the AE extract-treated and negative control groups. There was an increase in the damage index of cells treated with H2O2 (positive control group) (Fig 3).
Table 3. Average and standard deviation of CBPI (cytokinesis blocking proliferation index) and micronucleus frequency in ACP02 cells after 24 hours exposure to juca aqueous ethanol extract and its controls.
| Treatment | CBPI | MNf |
|---|---|---|
| NC | 1.4(±0.09) | 0.011(±0.006) |
| DMSO (0.1%) | 1.3(±0.13) | 0.005(±0.002) |
| 25 μg/ml | 1.4(±0.09) | 0.005(±0.003) |
| 50 μg/ml | 1.3(±0.14) | 0.006(±0.001) |
| 100 μg/ml | 1.2(±0.16) | 0.005(±0.002) |
| 200 μg/ml | 1.4(±0.02) | 0.003(±0.004) |
| Colchicine (0.02 μg/ml) | 1.3(±0.1) | 0.04(±0.04)* |
ANOVA parametric test; Tukey-Kramer Multiple comparisons (p <0.05); NC means negative control and
* Differs from other treatments.
Fig 3. Percentage of average of total damage index in ACP02 cells after 23 hours of exposure to juca aqueous ethanol extract and it controls.
ANOVA parametric test; Tukey-Kramer Multiple comparisons (p <0.05); NC means negative control, PC means positive control and * Differs from other treatments.
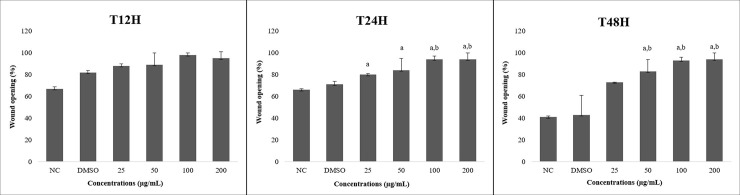
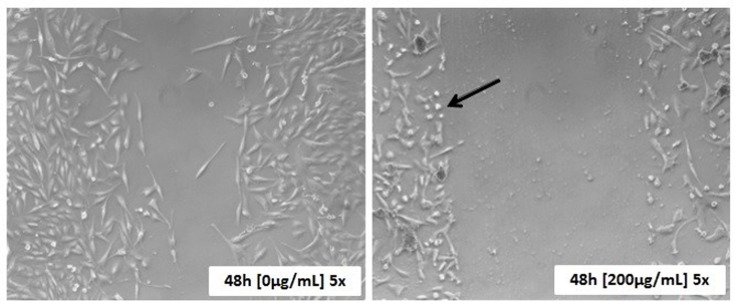
The wound-healing test showed that there was no significant between-group difference at 12 hours post-exposure. However, at 24 and 48 hours, ACP02 cells treated with juca AE extract exhibited decreased migratory activity, with a dose-response effect that increased at concentrations of 50 μg/mL and beyond (Fig 4). We also observed a change in the morphology of AE extract-treated cells, which exhibited decreased sizes at 24- and 48-hours post-exposure (Fig 5).
Fig 4. Percentage of wound opening made for the wound healing test after exposure to treatments and their respective control at times of 12, 24 and 48 hours using the ACP02 cell line.
ANOVA parametric test; Tukey-Kramer Multiple comparisons (p <0.05); NC means negative control. aDiffers from NC. bDiffers from DMSO.
Fig 5. Human gastric adenocarcinoma cells (ACP02) untreated and treated with juca aqueous ethanol extract after 48 hours.
48h: 48 hours after the wound is made. 0 μg / mL and 200 μg / mL: concentrations of the juca extract. 5x: 5-fold increase.
4. Discussion
When studying phytochemicals for their biotechnological value, it can be useful to find a product of natural origin that has an antioxidant effect. Here, we report that AE and ACO extracts of jucá exhibit antioxidant activity. Consistent with this, both extracts contained phenolic compounds, such as quinic acid, 1,2-benzenedicarboxylic acid and benzoic acid, which have been shown to block and capture free radicals [40,41]. The juca AE extract exhibited higher antioxidant activity, which was demonstrated by the three antioxidants assays performed and was therefore used for the subsequent experiments. However, we do not necessarily refer to anticarcinogenic activity. We only consider that if reactive oxygen species formation is related to various diseases (including cancer), then the ability to sequester free radicals may prevent DNA damage. For this reason, the extract with the highest antioxidant activity was chosen for toxicity and migration assays. Furthermore research can be done evaluating the effects of the other extracts.
The AE extract did not appear to have any cytotoxic, genotoxic or mutagenic potential in ACP02 cells. It did, however, appear to inhibit cell migration in this cancer cell line. [42] noted that analyses of in vitro migration provide good evidence for similar effects on in vivo migration ability, which is critical to metastasis. Thus, our results indicate that the juca AE extract may have promise as an antimetastatic agent.
Next, we examined some possible explanations for the apparent inhibition of migration in juca AE-treated ACP02 cells. Ao/Eb staining showed that this treatment did not induce apoptosis, ruling out the possibility that the observed inhibition of migration could reflect increased cell death. In vertebrates, cell movement can reflect reorganization of the cytoskeleton [43,44], which is a fiber- and protein tubule-based structure responsible for maintaining the cell shape and assisting with cell movement [45]. Indeed, we observed that the cell size was decreased in ACP02 cells treated with juca AE extract for 24 or 48 hours. We thus speculate that the extract could trigger cytoskeletal disorganization, and thereby inhibit cell migration. A similar situation was observed in a study analyzing the anti-metastatic potential of biflorin [16].
Notably, the extracellular microenvironment has been shown to modulate the organization and expression of the filaments that form the cytoskeleton [43]. The juca AE extract was found to contain myo-inositol, which is involved in cytoskeletal assembly/disassembly and the cell adhesion that is important for cell locomotion [46]. We therefore speculate that the addition of other compounds, constituent of juca AE extract, such as myo-inositol (unrelated to its antioxidant potential) to the extracellular medium can alter the organization and/or polymerization/depolymerization of the cytoskeleton, and thus prevent cell migration.
The ability of juca AE extract to inhibit cell migration suggest that this natural product might help prevent the formation of secondary tumors in an organism. The absence of cytotoxicity indicates that the extract will not act to eliminate cancerous cells. However, it might be co-treated with anticancer drugs as a means to decrease cancer progression. Our present findings therefore indicate that juca AE extract is worthy of further study as a natural product that may help prevent the progression of cancer metastasis.
Together, our results show for the first time that the aqueous ethanol extract of Libidibia ferrea can function as an exogenous antioxidant in vitro, and thus could potentially act against oxidative stress-related diseases and/or strengthen the health and well-being of an organism. Our findings suggest that this extract may counter the formation of secondary tumors (metastasis), and thus might prove useful when administered alongside treatments that eliminate cancer cells. More studies aimed at elucidating the action mechanism of the L. ferrea HA extract are needed to enable the effective use of its biological activities.
5. Conclusion
Together, our results show for the first time that the aqueous ethanol extract of Libidibia ferrea can function as an exogenous antioxidant in vitro, and thus could potentially act against oxidative stress-related diseases and/or strengthen the health and well-being of an organism. Our findings suggest that this extract may counter the formation of secondary tumors (metastasis), and thus might prove useful when administered alongside treatments that eliminate cancer cells. More studies aimed at elucidating the action mechanism of the L. ferrea AE extract are needed to enable the effective use of its biological activities.
Acknowledgments
This study was funded by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES; Edital Pro-Amazônia Biodiversidade e Sustentabilidade 047/2012) and Banco Nacional de Desenvolvimento Econômico e Social–BNDES (Operation 2.318.697.0001) in projects coordinated by Julio Cesar Pieczarka. This study is part of the mastership dissertation of Luana França Calandrini de Azevedo in the post-graduation program in Genetics and Molecular Biology of the Universidade Federal do Pará (scholarship granted by CAPES).
Data Availability
All relevant data are within the paper.
Funding Statement
JCP received the grant 047/2012 from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (https://www.capes.gov.br) and 2.318.697.0001 from Banco Nacional de Desenvolvimento Econômico e Social (http://www.bndes.gov.br). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Alizadeh A.M.; Shiri S.; Farsinejad S. Metastasis review: from bench to bedside. Tumor Biol 2014; 35: 8483–8523. 10.1007/s13277-014-2421-z [DOI] [PubMed] [Google Scholar]
- 2.Yunes R.A.; Pedrosa R.C.; Cechinel Filho V. Fármacos e fitoterápicos: a necessidade do desenvolvimento da indústria de fitoterápicos e fitofármacos no Brasil. Quím. Nova 2011; 24(1): 147–152. 10.1590/s0100-40422001000100025 [DOI] [Google Scholar]
- 3.GRIN—USDA, ARS, National Genetic Resources Program. Germplasm Resources Information Network—(GRIN) [Online Database]. 2017. National Germplasm Resources Laboratory, Beltsville, Maryland: Disponível em: <https://npgsweb.ars-grin.gov/gringlobal/taxonomydetail.aspx?id=465144 >. Acess in December 2019. [Google Scholar]
- 4.Chappill J. A. Cladistic analysis of the Leguminosae: the development of na explicit phylogenetic hypothesis In: Crisp M. & Doyle J. J. (eds.), 7th edition, Advances in Legume Systematic 7: Phylogeny. Royal Botanic Gardens, London: Royal Botanic Gardens, 1995. [Google Scholar]
- 5.Barroso G. M.; Morim M. P.; Peixoto A. L.; Ichaso C. L. F. Frutos e sementes: morfologia aplicada à sistemática de dicotiledôneas. Imprensa Universitária, Viçosa: 1999; 443pp. [Google Scholar]
- 6.Lewis G. Legumes of Bahia. Kew: Royal Botanic Gardens 1987;369pp. [Google Scholar]
- 7.Sampaio F.C.; Pereira M.S.V.; Dias C.S.; Costa V.C.O.; Conde N.C.O.; Buzalaf M.A.R. In vitro antimicrobial activity of Caesalpinia ferrea Martius fruits against oral pathogens. J. Ethnopharmacol 2009; 124: 289–294. 10.1016/j.jep.2009.04.034 [DOI] [PubMed] [Google Scholar]
- 8.Oliveira A.F.; Batista J.S.; Paiva E.S.; Silva A.E.; Farias Y.J.M.D. Avaliação da atividade cicatrizante do jucá (Caesalpinia ferrea Mart. ex Tul. var.ferrea) em lesões cutâneas de caprinos. Revista Bras. Plantas Med 2010; 12(3): 302–310. 10.1590/s1516-05722010000300007 [DOI] [Google Scholar]
- 9.Albuquerque U.P.; Medeiros P.M.; Almeida A.L.S., Lins Neto E.M.F.; Melo J.G.; Santos J.P. Medicinal Plants of Caatinga (semi arid) vegetation of NE Brasil: A quantitative approach. J. Ethnopharmacol 2007; 114 (3): 325–354. 10.1016/j.jep.2007.08.017 [DOI] [PubMed] [Google Scholar]
- 10.Guerra, A.C.V.A. Avaliação das atividades antitumoral e antioxidante in vitro em extratos de Libidibia ferrea em células de cancer colorretal. [Mastership dissertation]. Natal: Programa de Pós Graduação em Ciências da Saúde, UFRN, 2017.
- 11.Silva L.C.N.; da Silva C.A. Jr; Souza R.M.; Macedo A. J.; Silva M. V.; Correia M.T.S. Comparative analysis of the antioxidant and DNA protection capacities of Anadenanthera colubrina, Libidibia ferrea and Pityrocarpa moniliformis fruits. Food. Chem. Toxicol 2011; 49: 2222–2228. 10.1016/j.fct.2011.06.019 [DOI] [PubMed] [Google Scholar]
- 12.Port’s P.S.; Chisté R.C.; Godoy H.T.; Prado M.A. The phenolic compounds and the antioxidant potential of infusion of herbs from the Brazilian Amazonian region. Food. Res. Int 2012; 53: 875–881. 10.1016/j.foodres.2013.02.010 [DOI] [Google Scholar]
- 13.Cunha A. P.; Silva A.P.; Roque O.R. Plantas e produtos vegetais em fitoterapia, 4ª edição, Brasil: Lisboa: Fundação Calouste Gulbenkian; 2012. [Google Scholar]
- 14.Sousa T.M.; Severi J.A.; Silva V.Y.A.; Santos E.; Pietro L.C.L.R. Bioprospecção de atividade antioxidante e antimicrobiana da casca de Strtphnodendron adstringens (Mart.) Coville (Leguminosae-Mimosoidae. Rev. Ciênc. Farm. Básica Apl 2007; 28(2): 221–226. [Google Scholar]
- 15.Harvey A.L.; Bradley K.N.; Cochran S.A.; Rowan E.G.; Pratt J.A.; Quillfeldt J. A.; et al. What can toxins tell us for drug discovery?. Toxicon 1998; 36 (11): 1635–1640. 10.1016/s0041-0101(98)00156-1 [DOI] [PubMed] [Google Scholar]
- 16.Carvalho, A.A. Estudo do potencial antimetastático da bioflorina. [Doctoral thesis]. Ceará: Programa de Pós Graduação em Farmacologia, UFC, 2011.
- 17.Elias, S.T. Avaliação in vitro do potencial antineoplásico de plantas do cerrado em carcinoma de cabeça e pescoço. [Doctoral thesis]. Brasília: Programa de Pós-Graduação em Ciências da Saúde, UNB, 2014.
- 18.Ramos, J.A.B. Avaliação in vitro do potencial citotóxico e genotóxico do e-2-benzo[d]thiazol em células normais e tumorais. [Bachelor Degree]. Pará: Faculdade de Biomedicina. UFPA, 2012.
- 19.Valente, T.O.N. Análise das proteínas relacionadas a formação de metástase em linhagens de adenocarcinoma gástrico. [Mastership dissertation]. Pará: Programa de Pós Graduação em Oncologia e Ciências Médicas. UFPA, 2014.
- 20.OECD. Test No. 487: In Vitro Mammalian Cell Micronucleus Test, OECD Guidelines for the Testing of Chemicals, Section 4, OECD Publishing, Paris, 10.1787/9789264264861-en. 2016. Access in July 2018. [DOI]
- 21.Leal M.F.; Calcagno D.Q.; Costa J.F.F.B.; Silva T.C.R.; Khayat A.S.; Chen E.S., et al. MYC, TP53, and Chromosome 17 Copy-Number Alterations in Multiple Gastric Cancer Cell Lines and in Their Parental Primary Tumors. J. Biomed. Biotechnol 2011; 2011: 8 8 10.1155/2011/631268 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Reis, A. Aplicação de técnicas de lipidômica na investigação do perfil fitosteroides e tetranorterpenoides das sementes de carapa guianensis aubl obtidos por extração assistida em ultrassom. [Mastership dissertation]. Pará: Programa de Pós-Graduação em Biotecnologia, UFPA, 2017.
- 23.Schummer C.; Delhomme O.; Appenzeller B.M.R.; Wennig R.; Millet M. Comparison of MTBSTFA and BSTFA in derivatization reactions of polar compounds prior to GC/MS analysis. Talanta 2009; 77: 1473–1482. 10.1016/j.talanta.2008.09.043 [DOI] [PubMed] [Google Scholar]
- 24.Moco S.; Bino R.J.; De Vos R.C.H.; Vervoort J. Metabolomics technologies and metabolite identification. Trends. Analyt. Chem 2007; 26(9): 855–866. 10.1016/j.trac.2007.08.003 [DOI] [Google Scholar]
- 25.Brand-Wiliams W.; Cuvelier M.E.; Berset C. Use of a free radical method to evaluate antioxidant activity. Food. Sci. Technol 1995; (28): 25–30. 10.1016/s0023-6438(95)80008-5 [DOI] [Google Scholar]
- 26.Re R.; Pellegrini N.; Proteggente A.; Pannala A.; Yang M.; Rice-Evans C. Antioxidant activity applying na improved ABTS radical cátion decolorization assay. Free Radical Bio. Med 1999; 26(9/10): 1231–1237. 10.1016/s0891-5849(98)00315-3 [DOI] [PubMed] [Google Scholar]
- 27.Ou B.; Hapsch-Woodill M.; Prior R. L. Development and Validation of na Improved Oxigen Radical Absorbance Capacity Assay using Fluorescein as Fluorescent Probe. J. Agric. Food Chem 2001; 49: 4619–4626. 10.1021/jf010586o [DOI] [PubMed] [Google Scholar]
- 28.Huang D.; Ou B.; Hapsch-Woodill M.; Flanagan J. A.; Prior R. L. High-Throughput Assay of Oxygen Radical Absorbance Capacity (ORAC) Using a Multichannel Liquid Handling System Coupled with a Microplate Fluorescence Reader in 96-Well Format. J. Agric. Food Chem 2002; 50: 4437–4444. 10.1021/jf0201529 [DOI] [PubMed] [Google Scholar]
- 29.Silva E.M.; Souza J.N.S.; Rogez H.; Rees J.F.; LArondelle Y. Antioxidant activities and polyphenolic contents of fifteen selected plant species from the Amazonian region. Food Chem 2007; 101: 1012–1018. 10.1016/j.foodchem.2006.02.055 [DOI] [Google Scholar]
- 30.Mosmann T. Rapid Colorimetric Assay for Cellular Growth and Survival: Application to Proliferation and Cytotoxicity Assays. J. lmmunol. Methods 1983; 65: 55–63. 10.1016/0022-1759(83)90303-4 [DOI] [PubMed] [Google Scholar]
- 31.Sperotto R.A. Protocolos e métodos de Análise em laboratórios de biotecnologia agroalimentar e de saúde humana, 1ª edição, Lajeado: Editora Univates, 2014. [Google Scholar]
- 32.Machado-Santelli G.M.; Lotfi C.F.P. Comparative analysis of colchicine induced micronuclei in different cell types in vitro. Mutat. Res 1996; 349: 77–83. 10.1016/0027-5107(95)00160-3 [DOI] [PubMed] [Google Scholar]
- 33.Andrighetti-Frohner C.R.; Kratz J.M.; Antonio R.V.; Creczynski-Pasa T.B.; Barardi C.R.M.; Simões C.M.O. In vitro testing for genotoxicity of violacein assessed by Comet and Micronucleus assays. Mutat. Res 2006; 603: 97–103. 10.1016/j.mrgentox.2005.11.001 [DOI] [PubMed] [Google Scholar]
- 34.Eastmond D.A.; Tucker J.D. Identification of aneuploidy-inducing agents using cytokinesis-blocked human lymphocytes and an antikinetochore antibody. Environ. Mol. Mutagen 1989; 13 (1): 34–43. 10.1002/em.2850130104 [DOI] [PubMed] [Google Scholar]
- 35.Singh N.P.; McCoy M.T.; Tice R.R.; Schneider E.L. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp. Cell Res 1988; 75 (1): 184–191. 10.1016/0014-4827(88)90265-0 [DOI] [PubMed] [Google Scholar]
- 36.Collins R.A.; Guo M.A.; Duthie S.J. The kinetics of repair and oxidative DNA damage (Strand breaks and oxidized pyrimidines) in human cells. Mutat. Res 1995; 336: 69–77. 10.1016/0921-8777(94)00043-6 [DOI] [PubMed] [Google Scholar]
- 37.Bürk R.R. A factor from a transformed cell line that affects cell migration. Proc. Nat. Acad 1973; 70 (2): 369–372. 10.1073/pnas.70.2.369 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.McGahon A.J.; Martin S.M.; Bissonnete R.P.; Mahboubi A.; Shi Y.; Mogil R.J., et al. The end of the (cell) line: methods for the study of apoptosis in vitro. Methods Cell. Biol 1995; 46: 153–185. 10.1016/s0091-679x(08)61929-9 [DOI] [PubMed] [Google Scholar]
- 39.Ayres M.; Ayres M. Jr; Ayres D.L.; Dos Santos A.S. Bio Estat 5.0 Aplicações Estatísticas das Ciências Biológicas e Médicas, 5ª edição, Pará: Instituto de Desenvolvimento Sustentável Mamirauá –IDSM/ MCT/ CNPq, 2007. [Google Scholar]
- 40.Degáspari C.H.; Waszczynskyj N. Antioxidants properties of phenolic compounds. Visão Acadêmica 2004; 5 (1): 33–40. 10.5380/acd.v5i1.540 [DOI] [Google Scholar]
- 41.Broinizi P.R.B.; Andrade-Wartha E.R.S.; Silva A.M.O.; Novoa A.J.V.; Torres R.P.; Azeredo H.M.C., et al. Evaluation of the antioxidant activity of phenolic compounds naturally contained in by-products of the cashew apple (Anacardium occidentale L.). Ciênc. Tecnol. Aliment 2007; 27(4): 902–908. 10.1590/s0101-20612007000400035 [DOI] [Google Scholar]
- 42.Liang C.C.; Park A.Y.; Guan J.L. In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro. Nature protocols 2007; 5 pp. Michigan, USA. 10.1038/nprot.2007.30 [DOI] [PubMed] [Google Scholar]
- 43.Krug, M.S. Análise da difusão celular em linhagens com potencial metastático. [Bachelor Degree]. Porto Alegre: Faculdade de Biotecnologia. UFRS, 2013.
- 44.Maryas J.; Faktor J.; Dvorakova M.; Struharova I.; Grell P.; Bouchal P. Proteomics in investigation of cancer metastasis: Functional and clinical consequences and methodological challenges. Proteomics 2014; 14: 426–440. 10.1002/pmic.201300264 [DOI] [PubMed] [Google Scholar]
- 45.Alberts B.; Johnson A.; Lewis J.; Morgan D.; Raff M.; Roberts K., et al. Molecular Biology of the Cell, 6th edition, New York: Garland Science, a Taylor & Francis Group, 2014. [Google Scholar]
- 46.Almeida M.V.; Da Silva A.D.; De Souza M.V.N.; Benício A.A.A. A cascata dos fosfoinositídeos. Quim. Nova 2003; 26 (1): 105–111. 10.1590/s0100-40422003000100018 [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All relevant data are within the paper.