Abstract
Introduction
Cell sheets consisting of adipose-derived stem cells (ADSCs) have been reported to be effective for wound healing. We conducted this study to clarify the efficacy of ADSC sheets in wound healing at the duct-to-duct biliary anastomotic site in pigs.
Methods
Eleven female pigs (20–25 kg) were divided into two groups: biliary anastomosis with an ADSC sheet (n = 6) or without an ADSC sheet (n = 5). To follow the transplanted ADSCs, PKH26GL-labeled sheets were used in one of the ADSC pigs. Two weeks prior to laparotomy, ADSCs were isolated from the lower abdominal subcutaneous adipose tissue. After three passages, ADSCs were seeded on temperature-responsive culture dishes and collected as cell sheets. ADSC sheets were gently transplanted on the anastomotic site. We evaluated specimens by PKH26GL labeling, macroscopic changes, infiltration of inflammatory cells, and collagen content.
Results
Labeled ADSCs remained around the bile duct wall. In the no-ADSC group, more adhesion developed at the hepatic hilum as observed during relaparotomy. Histopathological examination showed that the diameter and cross-sectional area of the bile duct wall were decreased in the ADSC group. In the no-ADSC group, a large number of inflammatory cells and more collagen fibers were identified in the bile duct wall.
Conclusions
The present study demonstrated that autologous ADSC sheet transplantation reduced hypertrophic changes in the bile duct wall at the anastomotic site. A long-term follow-up is required to evaluate the efficacy of this mechanism in prevention of biliary anastomotic strictures.
Keywords: Adipose-derived stem cell, Cell sheet, Biliary anastomosis, Anastomotic healing
Abbreviations: ADSC, adipose-derived stem cell; APC, allophycocyanin; BAS, biliary anastomotic strictures; BMSC, bone marrow stem cells; CBD, common bile duct; FBS, fetal bovine serum; FGF, fibroblast growth factor; HGF, hepatocyte growth factor; MSC, mesenchymal stem cell; VEGF, vascular endothelial growth factor
Highlights
-
•
The first study to show the effect of ADSCs on healing of biliary anastomosis.
-
•
Hypertrophic change in bile duct wall was observed at biliary anastomotic site.
-
•
Autologous ADSC transplantation reduced the hypertrophic change.
-
•
The presence of transplanted ADSC was confirmed by PKH26GL-positive ADSCs.
1. Introduction
Biliary anastomotic strictures (BAS) are one of the most common complications following liver transplantation. Frequent treatment for BAS deteriorates the patient's quality of life. Furthermore, prolonged biliary congestion can lead to secondary liver failure and the need for retransplantation. BAS incidence has been reported to be from 5% to 15% in deceased donor liver transplantations [1] and from 4% to 25% in living donor liver transplantations [2,3]. To reduce these complications, surgical techniques have been developed, such as preservation of vascularization around the bile duct by minimal hilar dissection [4,5] or biliary reconstruction [6] in addition to optimal graft selection, progress in preservation, and standardization of surgical procedures. However, BAS still occur to some extent. New strategies for efficiently enhancing the healing process of the anastomotic site are anticipated to achieve optimal outcomes.
Recently, regenerative therapy using mesenchymal stem cells (MSCs), such as bone marrow stem cells (BMSCs) or adipose-derived stem cells (ADSCs), have been widely studied for their potential to accelerate wound healing [[7], [8], [9], [10], [11], [12], [13]]. Stimulation of angiogenesis, secretion of growth factors, differentiation, immunomodulatory effects, and anti-inflammatory activities are essential functions of MSCs [14,15]. Although BMSCs and ADSCs show a similar ability to differentiate and regenerate [16], ADSCs are easier to use in clinical settings because harvest and extraction of ADSCs are easier and safer compared with BMSCs [7].
Although several studies have reported a local administration technique for ADSC transplantation [12,13,17], whether injected cells remain around the intended area is unknown. In addition, direct injection into thin tissue such as the bile duct wall is not technically feasible. In this regard, cell sheet transplantation is a reasonable approach to overcome this issue [18,19]. Previous reports have indicated the effectiveness of cell sheet transplantation in cornea recreation [8], prevention of esophageal stenosis after endoscopic submucosal dissection [9], treatment of myocardial infarction [20], prevention of pancreatic fistula [10], and treatment of intestinal anastomosis [11]. However, there are no reports on the effects of cell sheet transplantation on healing of the biliary anastomotic site.
The aim of the present study was to assess the effect of ADSC sheet transplantation on end-to-end biliary anastomosis in a pig model.
2. Methods
2.1. Animals
Eleven female pigs (20–25 kg) were obtained from Miyashita pig farm (Nagasaki, Japan) and used in the present study. All animals were treated with experimental procedures approved by the Biomedical Research Center and Committee of Nagasaki University (approval number 1602031279-2). The experiments were performed in accordance with institutional and national guidelines.
All pigs received an acclimation period of 3 days prior to any procedures, and no premedication was administered during this time. Animals were anesthetized after an overnight fast based on their body weight using 0.2 mg/kg midazoram and 0.1 mg/kg medetomidine hydrochloride. An endotracheal tube was inserted, and general anesthesia was maintained during procedures with sevoflurane and oxygen. Benzylpenicillin (2 × 105 U) was injected intravenously 30 min before laparotomy as an antimicrobial prophylaxis.
2.2. Isolation of ADSCs
Two weeks prior to laparotomy for biliary anastomosis, ADSCs were isolated from the lower abdominal subcutaneous adipose tissue of pigs as reported elsewhere [11,21]. Briefly, 20 g of minced subcutaneous adipose tissue were enzymatically digested with 0.1% collagenase (Collagenase NB 4G Proved Grade, SERVA Electrophoresis, Heidelberg, Germany) under shaking at 37 °C for 1 h. The stromal-vascular fraction was collected after centrifugation at 400× g for 5 min at room temperature. After filtration through a 40-mm strainer (BD Biosciences, Franklin Lakes, NJ, USA) and centrifugation at 1500× g for 5 min, the cells were cultured in complete medium (low-glucose Dulbecco's modified Eagle's medium, Wako Pure chemical industries, Osaka, Japan) supplemented with 10% fetal bovine serum (FBS) (Thermo Fisher Scientific, Waltham, MA, USA) and 1% penicillin/streptomycin (Thermo Fisher Scientific) in a humidified atmosphere with 5% CO2 at 37 °C. ADSCs were passaged every 5 days by seeding onto 100-mm culture dishes at a density of 1.1 × 106 cells/dish until the third passage.
2.3. Flow cytometry
Isolated ADSCs from the third passage were suspended in 100 μL of PBS containing 10% FBS and examined for surface marker expression by flow cytometry. To detect the markers, a phycoerythrin-coupled antibody against CD44 (BioLegend, San Diego, CA, USA), fluorescein isothiocyanate-coupled antibody against CD90 (Sigma–Aldrich, St. Louis, MO, USA), allophycocyanin (APC)-coupled antibody against CD31 (Sigma–Aldrich), and APC-coupled antibody against CD45 (Sigma–Aldrich) were used. Fluorescence was measured using a flow cytometer (CytoFLEX, Beckman Coulter, Brea, CA, USA).
2.4. Preparation of ADSC sheets
ADSCs from the third passage were seeded on temperature-responsive culture dishes (UpCell, CellSeed, Tokyo, Japan) at a density of 2.6 × 106 cells/35-mm dish and cultured in complete medium under a humidified atmosphere with 5% CO2 at 37 °C. After 4 days of culture, the ADSC sheets were harvested from the dishes by lowering the temperature to 20 °C for 30 min.
2.5. PKH26GL staining
ADSCs from the third passage were stained using a PKH26GL Red Fluorescent Cell Linker Kit (Sigma–Aldrich). Isolated cells (2 × 106) were stained with 4 × 10−3 mol PKH26GL dye, in accordance with the manufacturer's protocol. Stained cells were seeded on temperature-responsive culture dishes, and then harvested as ADSC sheets after 4 days.
2.6. Surgical procedure
Pigs were divided into two groups: duct-to-duct biliary anastomosis with an ADSC sheet (n = 6) or without an ADSC sheet (n = 5). In both groups, an open laparotomy with a midline incision was performed under general anesthesia. The liver hilum was gently exposed. Then, the common bile duct (CBD) proximal to the cystic duct junction was circumferentially isolated from the surrounding fatty tissue. The CBD was then completely divided by scissors. Biliary anastomosis was performed subsequently with a 6-0 absorbable monofilament suture. The back wall was anastomosed by a continuous suture, and the front wall was anastomosed by an interrupted suture. After completion of anastomosis, an ADSC sheet was transplanted on the anastomotic site in the ADSC group. The ADSC sheet was scooped from the culture dishes using a sterilized mesh and transported to the operative field. The sheet was attached to the biliary anastomotic site without the need for sutures or clips. The sheet naturally adhered by placing a few drops of saline on the mesh. After the ADSC sheet transplantation, the anastomotic site was kept stationary for 20 min to avoid dislocation of the transplanted sheet. One of the six pigs in the ADSC group received an autologous ADSC sheet labeled with PKH26GL to follow the presence of transplanted cells. The abdomen was closed layer-to-layer. Blood samples were obtained on the day of the laparotomy and on days 7 and 14 after the operation. The pigs were sacrificed on day 14 after collection of the anastomosed CBD. All the procedures were performed by one board-certified surgeon with more than 10 years of experience in hepatobiliary surgery.
2.7. Histopathological examination
The isolated specimens were fixed in 4% paraformaldehyde and processed into paraffin-embedded sections. The site of anastomosis was easily detected by the suture thread. Bile duct specimens were prepared at three locations: the anastomotic site, the 2-mm oral side, and the 2-mm caudal side. The sections were stained with hematoxylin and eosin, and Masson's trichrome using conventional methods. The length and area of sections were calculated using NDP.view2 Viewing software U12388-01 (Hamamatsu Photonics, Shizuoka, Japan). The specimen with the PKH26-labeled ADSC sheet was sectioned and frozen immediately in liquid nitrogen. The frozen tissue was mounted using O.C.T. compound (Sakura Tissue-Tek, Tokyo, Japan). After cutting the tissue into 4-μm-thick sections and mounting on coverslips, the sections were examined under a fluorescence microscope.
2.8. Statistical analysis
IBM SPSS Statistics 21 (SPSS, Chicago, IL, USA) was used for statistical analysis. Numerical data are presented as the mean ± SD. The Mann–Whitney U-test was used for analysis. P < 0.05 was considered statistically significant.
3. Results
3.1. Characterization of ADSCs
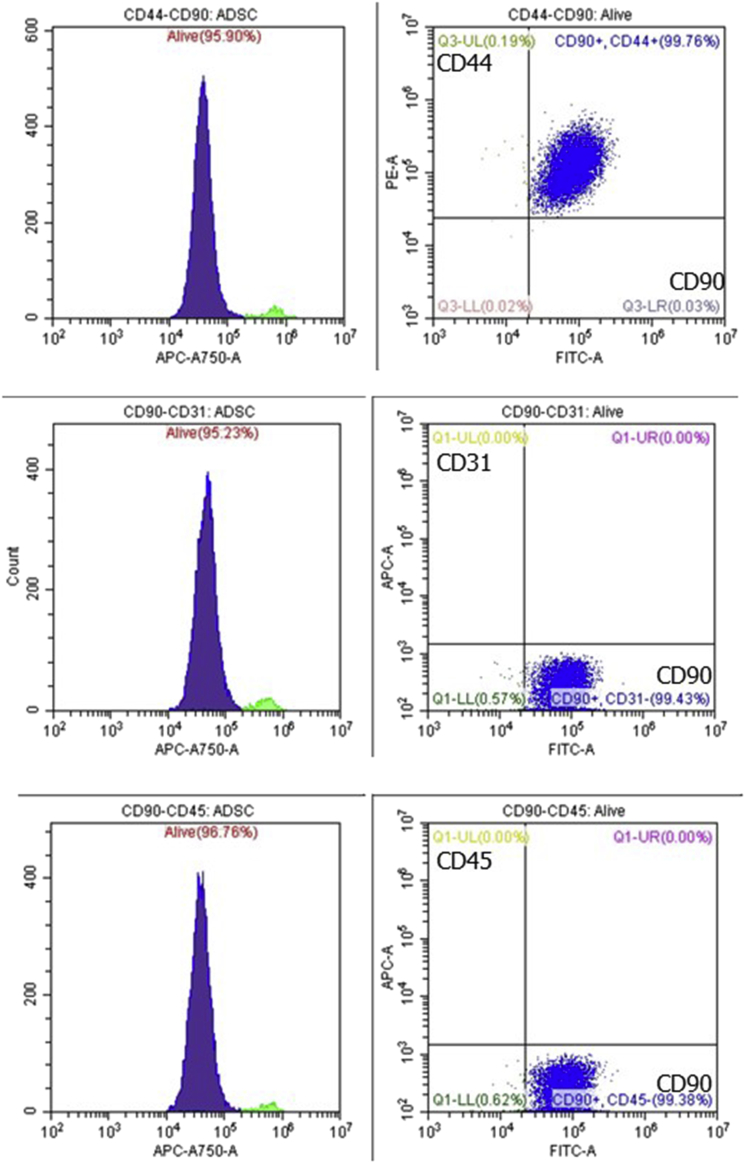
Harvested cells were a single cell population (>95%). Flow cytometry analysis of ADSC primary cultures revealed that the cells were positive for MSC-specific markers (CD90 and CD44) and negative for hematopoietic markers (CD31 and CD45; Fig. 1).
Fig. 1.
Flow cytometry analysis of ADSC primary cultures showing expression of MSC-specific markers (CD90 and CD44) and no expression of hematopoietic markers (CD31 and CD45).
3.2. Effect of the ADSC sheet on biliary anastomosis
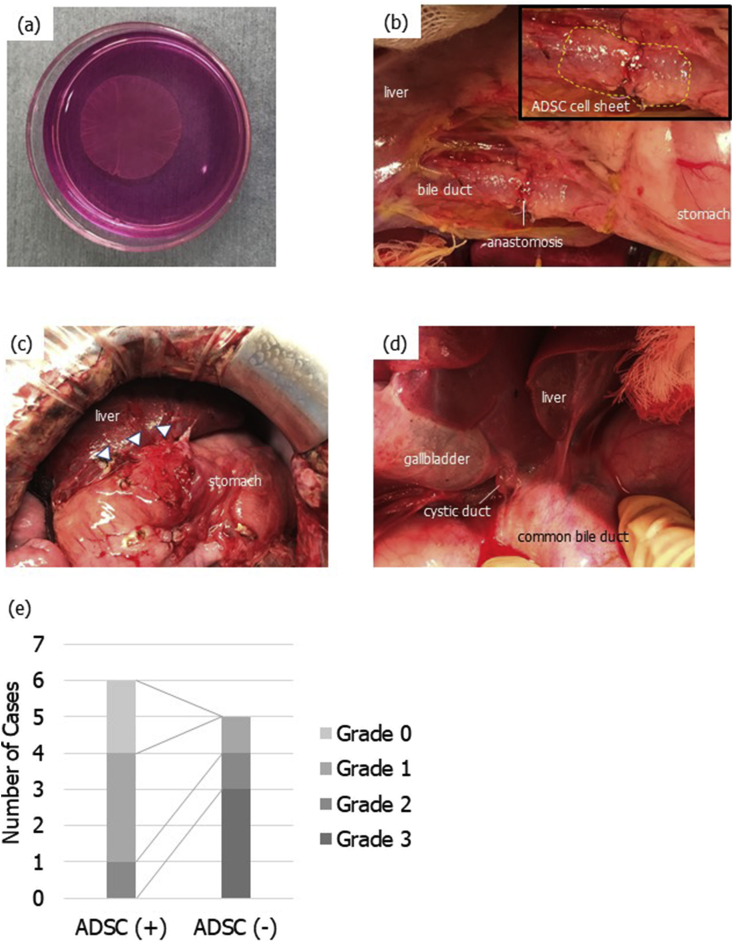
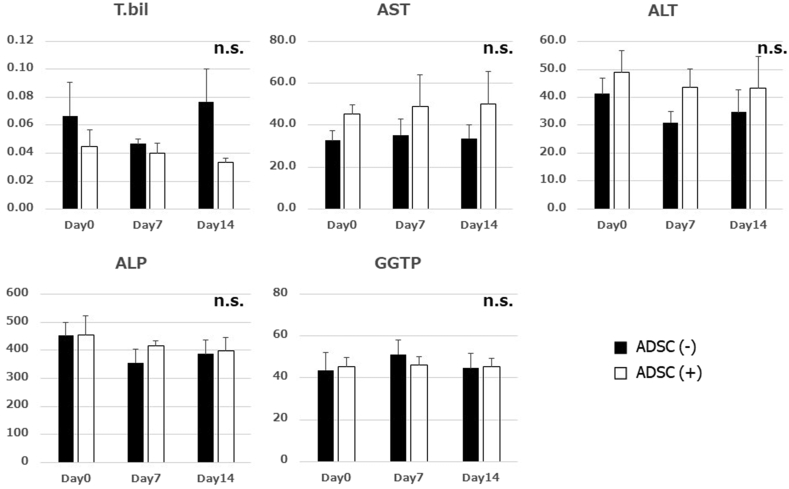
The cultured ADSCs were harvested as cell sheets by reducing the temperature of the temperature-sensitive dishes (Fig. 2a). An ADSC sheet was transplanted on the biliary anastomotic site (Fig. 2b). Two weeks later, inspection of the abdominal cavity and collection of the anastomosed bile duct were performed. The adhesion around the liver hilum was more severe in the no-ADSC group, which required sharp dissection (Fig. 2c and d). Evaluating the adhesion severity using a previously published classification (grade 0: none, grade 1: film-like with no neovascularization, grade 2: moderately thick with partial neovascularization, grade 3: thick, solid adhesion with neovascularization) [22], we noted grade 2 or higher in 17% (1/6) of the ADSC group and in 80% (4/5) of the no ADSC group, though the difference was not statistically significant (P = 0.07; Fig. 2e). There were no signs of bile leakage or intra-abdominal abscesses in all pigs. Dilatation of the distal bile duct due to severe anastomotic stricture was not clearly observed. Serum bilirubin, aspartate transaminase, alanine transaminase, alkaline phosphatase, and γ-glutamyl transpeptidase were measured before surgery and on postoperative days 7 and 14. Their levels were not significantly different throughout the experiment (Fig. 3). Thus, there were no signs of cholestasis.
Fig. 2.
Preparation and transplantation of autologous ADSC sheets. ADSCs were harvested as cell sheets using temperature-responsive culture dishes (a). End-to-end biliary anastomosis was performed, and then an ADSC sheet was transplanted (the area indicated by a dotted line) (b). Two weeks after the surgery, severe adhesion around the liver hilum developed in the no-ADSC group, which required sharp dissection (arrowhead) (c). In contrast, adhesiolysis was not required in the ADSC group (d). Evaluating the severity of adhesion using a previously published classification (grade 0: none, grade 1: film-like with no neovascularization, grade 2: moderately thick with partial neovascularization, grade 3: thick, solid adhesion with neovascularization), we noted grade 2 or higher in 17% (1/6) of the ADSC group and in 80% (4/5) of the no-ADSC group, though the difference was not statistically significant (P = 0.07) (e).
Fig. 3.
The levels of serum bilirubin, aspartate transaminase, alanine transaminase, alkaline phosphatase, and γ-glutamyltranspeptidase were not significantly different between the groups.
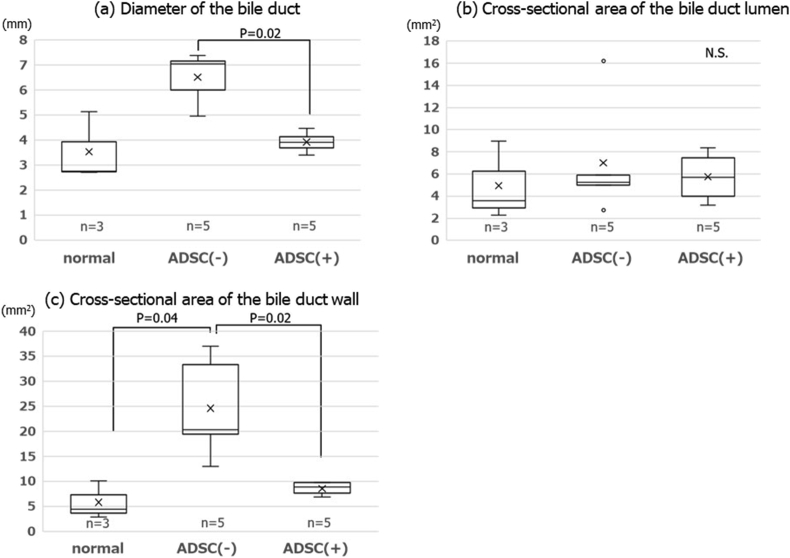
The macroscopic findings of the anastomotic site on postoperative day 14 are summarized in Fig. 4. In the collected specimens, the diameter of the bile duct increased after biliary anastomosis compared with the normal bile duct without any manipulation. The diameter in the no-ADSC group was significantly larger than that in the ADSC group (P = 0.02). Observation of the cross section of the anastomotic site revealed that although the luminal area was not different between the groups, the area of the bile duct wall was significantly larger in the no-ADSC group than in the normal bile duct (P = 0.04) and ADSC group (P = 0.02).
Fig. 4.
Macroscopic findings of the anastomotic site on postoperative day 14. The diameter of the bile duct in the no-ADSC group was significantly larger than in the ADSC group (a). The cross-section of the luminal area was not different between the groups (b). The area of the bile duct wall was significantly larger in the no-ADSC group than in the normal bile duct and ADSC group (c).
3.3. Histological examination of the anastomotic site
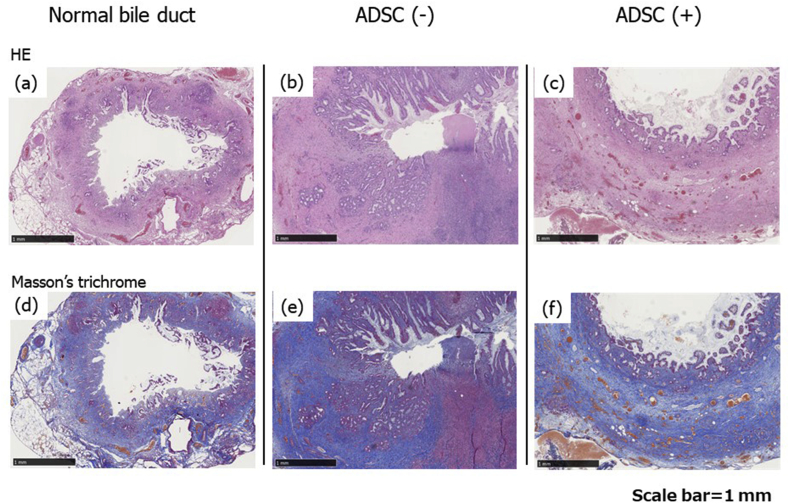
Anastomotic sites on postoperative day 14 were evaluated histologically. In the no-ADSC group, a large number of inflammatory cells were identified in the bile duct wall. Masson's trichrome staining showed that the bile duct wall was thickened by an increase in collagen fibers. In contrast, fewer inflammatory cells were observed in the ADSC group. Additionally, many small vessels were confirmed in the wall compared with the no-ADSC group (Fig. 5).
Fig. 5.
Microscopy findings of the normal bile duct without manipulation, anastomotic site without an ADSC sheet on postoperative day 14, and anastomotic site with an ADSC sheet on postoperative day 14. H&E (a–c) and Masson's trichrome (d–f). A large number of inflammatory cells and increased collagen content were identified in the no-ADSC group (b, e). In contrast, fewer inflammatory cells and many small vessels were observed in the ADSC group (c, f).
3.4. Confirmation of transplanted ADSCs
To confirm the presence of the ADSC sheet after transplantation, one of the six pigs in the ADSC group received autologous ADSCs labeled with the fluorescent dye, PKH26GL (Fig. 6a). Biliary anastomosis and transplantation of the ADSC sheet were performed in the same manner as the other pigs. Fluorescence microscopy of frozen sections of a resected specimen revealed the presence of PKH26GL-positive ADSCs around the bile duct wall (Fig. 6b).
Fig. 6.
To follow the presence of transplanted ADSCs, cells were labeled with PKH26GL (a). Fluorescence microscopy of frozen sections revealed the presence of PKH26GL-positive ADSCs around the bile duct wall on postoperative day 7 (arrow) (b).
4. Discussion
The present study showed that ADSCs reduced the hypertrophic change in the bile duct wall at the anastomotic site. The transplanted ADSC sheet remained on the anastomotic site without signs of intra-abdominal abscesses.
The ADSCs in the present study were characterized by flow cytometry. Harvested cells from adipose tissue expressed CD90 and CD44, which are MSC-specific surface markers. In contrast, these cells did not express CD31 and CD45, which are hematopoietic markers. Additionally, differentiation into adipogenic and osteogenic lineages was confirmed in a previous study that used the same protocol as we did [11]. These results indicate that the ADSCs used in our study possessed MSC-like properties.
Laursen et al. have previously observed healing of anastomoses of the extrahepatic bile duct in a pig model. They reported that the collagen content in the anastomotic site increased postoperatively from day 0–1, reached steady state by day 3, increased gradually until day 6, and then remained stable until day 14 [23]. According to these findings, we set the postoperative following period to 14 days in the present study. Histological examination of biliary anastomosis in the no-ADSC group revealed an increase in inflammatory cells and a thickened bile duct wall by an increase in collagen fibers. In contrast, these findings were not observed in the ADSC group. These results indicate that the hypertrophic changes in the bile duct wall were suppressed by the introduction of ADSCs into the anastomotic site. Hypertrophic changes in the bile duct wall are frequently caused by inflammation such as cholangitis, minor bile leakage, or stent placement in routine clinical practice. Because the ADSC sheet is fragile and thin, a prevention effect on minor biliary leakage was not expected in the present study, hence the alleviation of the hypertrophic change in the bile duct wall was likely due to other mechanisms. Moreover, the anti-inflammatory effect and immunomodulatory capacity possessed by ADSCs have been reported previously [14,15]. Another important mechanism is the secretion of trophic factors such as hepatocyte growth factor (HGF) and vascular endothelial growth factor (VEGF). Nie et al. have reported that the angiogenic effect of these factors accelerated wound healing [13]. In fact, many small vessels were confirmed in the bile duct wall of the ADSC group. These newly formed vessels may have affected the difference in healing between the ADSC and no-ADSC groups in the present study. Additionally, an antifibrogenic effect of HGF has been reported in various organs. Suga et al. have revealed that ADSCs promoted secretion of HGF by fibroblast growth factor (FGF)-2 stimulation [24]. They also found an increase in fibrogenesis by suppression of HGF through inhibition of FGF-2 signaling. These results suggest that HGF secreted by ADSCs also had a role in the antifibrogenic effect in our pig biliary anastomotic model. On the whole, an anti-inflammatory effect and immunomodulatory capacity, an angiogenic effect, and an antifibrogenic effect by secretion of trophic factors were considered to be among the reasons for the alleviation of the hypertrophic change in the ADSC group.
Interestingly, adhesion around the hepatoduodenal ligament during the second laparotomy was mild in the ADSC group. We propose two possible mechanisms that would explain the reduced adhesion around the liver hilum in the ADSC group: (1) inflammation around the damaged and anastomosed bile duct was reduced by ADSCs as we already described, and (2) the ADSC sheet acted as a physical barrier on the damaged bile duct and suppressed adhesion. A similar mechanism using a sodium hyaluronate-based bioresorbable membrane has been reported [25] and already adopted in a clinical setting.
To the best of our knowledge, this is the first study to show the effect of ADSCs on healing of biliary anastomosis. Although there were advantages in our study, such as an autologous cell source, large animal experiment, and secured biliary anastomosis without surgical complications, there were also several limitations. First, our animal model was the duct-to-duct biliary reconstruction model and not the biliary stricture model. This was because BAS did not occur during the follow-up period of 14 days. Long-term follow-up may be required to evaluate the occurrence of BAS. Thus, the efficacy of ADSCs in preventing anastomotic strictures still remains unclear. In a future study, we plan to investigate the long-term effect of ADSC sheets beyond 14 days after surgery. Second, further studies are needed to establish the required number of ADSCs to constantly improve the hypertrophic change at the biliary anastomotic site.
In conclusion, we showed that autologous ADSC transplantation reduced the hypertrophic change at the biliary anastomotic site, and that cell sheet technology is a reasonable approach for administering ADSCs to a biliary anastomotic site. Further studies are necessary to reveal the molecular mechanisms involved.
Funding
This work was not funded by any educational or commercial organization.
Declaration of Competing Interest
The authors declare no conflict of interest.
Acknowledgment
We thank Mitchell Arico from Edanz Group (www.edanzediting.com/ac) for editing a draft of this manuscript.
Footnotes
Peer review under responsibility of the Japanese Society for Regenerative Medicine.
Contributor Information
Takanobu Hara, Email: harataka66@gmail.com.
Akihiko Soyama, Email: soyapop@hotmail.com.
Toshiyuki Adachi, Email: toshi.number9@gmail.com.
Shinichiro Kobayashi, Email: skobayashi1980@gmail.com.
Yusuke Sakai, Email: y.sakai.bioeng@gmail.com.
Yasuhiro Maruya, Email: maruyayasuhiro0626@hotmail.co.jp.
Tota Kugiyama, Email: tota0427@yahoo.co.jp.
Masaaki Hidaka, Email: mahidaka@nagasaki-u.ac.jp.
Satomi Okada, Email: okada_s_1107@yahoo.co.jp.
Takashi Hamada, Email: taka.hama84@gmail.com.
Kyoichiro Maekawa, Email: minomushi0418@yahoo.co.jp.
Shinichiro Ono, Email: shinichi0320_com@yahoo.co.jp.
Tomohiko Adachi, Email: adatomo@nagasaki-u.ac.jp.
Mitsuhisa Takatsuki, Email: takapon@nagasaki-u.ac.jp.
Susumu Eguchi, Email: sueguchi@nagasaki-u.ac.jp.
References
- 1.Takahashi K., Nagai S., Putchakayala K.G., Safwan M., Gosho M., Li A.Y. Prediction of biliary anastomotic stricture after deceased donor liver transplantation: the impact of platelet counts - a retrospective study. Transpl Int – Off J Eur Soc Organ Transpl. 2017;30(10):1032–1040. doi: 10.1111/tri.12996. [DOI] [PubMed] [Google Scholar]
- 2.Akamatsu N., Sugawara Y., Hashimoto D. Biliary reconstruction, its complications and management of biliary complications after adult liver transplantation: a systematic review of the incidence, risk factors and outcome. Transpl Int – Off J Eur Soc Organ Transpl. 2011;24(4):379–392. doi: 10.1111/j.1432-2277.2010.01202.x. [DOI] [PubMed] [Google Scholar]
- 3.Wang S.F., Huang Z.Y., Chen X.P. Biliary complications after living donor liver transplantation. Liver Transplant. 2011;17(10):1127–1136. doi: 10.1002/lt.22381. [DOI] [PubMed] [Google Scholar]
- 4.Ikegami T., Shirabe K., Morita K., Soejima Y., Taketomi A., Yoshizumi T. Minimal hilar dissection prevents biliary anastomotic stricture after living donor liver transplantation. Transplantation. 2011;92(10):1147–1151. doi: 10.1097/TP.0b013e3182336073. [DOI] [PubMed] [Google Scholar]
- 5.Takatsuki M., Eguchi S., Yamanouchi K., Hidaka M., Soyama A., Kanematsu T. Technical refinements of bile duct division in living donor liver surgery. J Hepatobiliary Pancreat Sci. 2011;18(2):170–175. doi: 10.1007/s00534-010-0322-0. [DOI] [PubMed] [Google Scholar]
- 6.Kim S.H., Lee K.W., Kim Y.K., Cho S.Y., Han S.S., Park S.J. Tailored telescopic reconstruction of the bile duct in living donor liver transplantation. Liver Transplant. 2010;16(9):1069–1074. doi: 10.1002/lt.22116. [DOI] [PubMed] [Google Scholar]
- 7.Kern S., Eichler H., Stoeve J., Kluter H., Bieback K. Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue. Stem Cells. 2006;24(5):1294–1301. doi: 10.1634/stemcells.2005-0342. [DOI] [PubMed] [Google Scholar]
- 8.Nishida K., Yamato M., Hayashida Y., Watanabe K., Yamamoto K., Adachi E. Corneal reconstruction with tissue-engineered cell sheets composed of autologous oral mucosal epithelium. N Engl J Med. 2004;351(12):1187–1196. doi: 10.1056/NEJMoa040455. [DOI] [PubMed] [Google Scholar]
- 9.Ohki T., Yamato M., Ota M., Takagi R., Murakami D., Kondo M. Prevention of esophageal stricture after endoscopic submucosal dissection using tissue-engineered cell sheets. Gastroenterology. 2012;143(3):582–588. doi: 10.1053/j.gastro.2012.04.050. e2. [DOI] [PubMed] [Google Scholar]
- 10.Tanaka T., Kuroki T., Adachi T., Ono S., Kitasato A., Hirabaru M. Development of a novel rat model with pancreatic fistula and the prevention of this complication using tissue-engineered myoblast sheets. J Gastroenterol. 2013;48(9):1081–1089. doi: 10.1007/s00535-012-0706-9. [DOI] [PubMed] [Google Scholar]
- 11.Maruya Y., Kanai N., Kobayashi S., Koshino K., Okano T., Eguchi S. Autologous adipose-derived stem cell sheets enhance the strength of intestinal anastomosis. Regen Ther. 2017;7:24–33. doi: 10.1016/j.reth.2017.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Liu L., Chiu P.W., Lam P.K., Poon C.C., Lam C.C., Ng E.K. Effect of local injection of mesenchymal stem cells on healing of sutured gastric perforation in an experimental model. Br J Surg. 2015;102(2):158–168. doi: 10.1002/bjs.9724. [DOI] [PubMed] [Google Scholar]
- 13.Nie C., Yang D., Xu J., Si Z., Jin X., Zhang J. Locally administered adipose-derived stem cells accelerate wound healing through differentiation and vasculogenesis. Cell Transplant. 2011;20(2):205–216. doi: 10.3727/096368910X520065. [DOI] [PubMed] [Google Scholar]
- 14.Caplan A.I., Correa D. The MSC: an injury drugstore. Cell Stem Cell. 2011;9(1):11–15. doi: 10.1016/j.stem.2011.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Le Blanc K., Frassoni F., Ball L., Locatelli F., Roelofs H., Lewis I. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet. 2008;371(9624):1579–1586. doi: 10.1016/S0140-6736(08)60690-X. [DOI] [PubMed] [Google Scholar]
- 16.Uzbas F., May I.D., Parisi A.M., Thompson S.K., Kaya A., Perkins A.D. Molecular physiognomies and applications of adipose-derived stem cells. Stem Cell Rev. 2015;11(2):298–308. doi: 10.1007/s12015-014-9578-0. [DOI] [PubMed] [Google Scholar]
- 17.Joo H.H., Jo H.J., Jung T.D., Ahn M.S., Bae K.B., Hong K.H. Adipose-derived stem cells on the healing of ischemic colitis: a therapeutic effect by angiogenesis. Int J Colorectal Dis. 2012;27(11):1437–1443. doi: 10.1007/s00384-012-1470-2. [DOI] [PubMed] [Google Scholar]
- 18.Yang J., Yamato M., Kohno C., Nishimoto A., Sekine H., Fukai F. Cell sheet engineering: recreating tissues without biodegradable scaffolds. Biomaterials. 2005;26(33):6415–6422. doi: 10.1016/j.biomaterials.2005.04.061. [DOI] [PubMed] [Google Scholar]
- 19.Yamato M., Utsumi M., Kushida A., Konno C., Kikuchi A., Okano T. Thermo-responsive culture dishes allow the intact harvest of multilayered keratinocyte sheets without dispase by reducing temperature. Tissue Eng. 2001;7(4):473–480. doi: 10.1089/10763270152436517. [DOI] [PubMed] [Google Scholar]
- 20.Miyahara Y., Nagaya N., Kataoka M., Yanagawa B., Tanaka K., Hao H. Monolayered mesenchymal stem cells repair scarred myocardium after myocardial infarction. Nat Med. 2006;12(4):459–465. doi: 10.1038/nm1391. [DOI] [PubMed] [Google Scholar]
- 21.Kaibuchi N., Iwata T., Onizuka S., Yano K., Yamato M., Okano T. Cytological character of mini pig mesenchymal stromal cells from various tissues and the attempt of cell sheet formation. Regen Ther. 2017;6:83–89. doi: 10.1016/j.reth.2017.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Suto T., Watanabe M., Endo T., Komori K., Ohue M., Kanemitsu Y. The primary result of prospective randomized multicenter trial of new spray-tyoe bio-absorbable adhesion barrier system (TCD-11091) against prospective adhesion formation. J Gastrointest Surg. 2017;21:1683–1691. doi: 10.1007/s11605-017-3503-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Laursen H.B., Thorsoe H.J., Oxlund H., Yasuda Y., Funch-Jensen P., Rokkjaer M. Choledocho-choledochostomy: the natural history of healing in pigs. J Hepatobiliary Pancreat Surg. 2007;14(5):498–502. doi: 10.1007/s00534-006-1212-3. [DOI] [PubMed] [Google Scholar]
- 24.Suga H., Eto H., Shigeura T., Inoue K., Aoi N., Kato H. IFATS collection: fibroblast growth factor-2-induced hepatocyte growth factor secretion by adipose-derived stromal cells inhibits postinjury fibrogenesis through a c-Jun N-terminal kinase-dependent mechanism. Stem Cells. 2009;27(1):238–249. doi: 10.1634/stemcells.2008-0261. [DOI] [PubMed] [Google Scholar]
- 25.Becker J.M., Dayton M.T., Fazio V.W., Beck D.E., Stryker S.J., Wexner S.D. Prevention of postoperative abdominal adhesions by a sodium hyaluronate-based bioresorbable membrane: a prospective, randomized, double-blind multicenter study. J Am Coll Surg. 1996;183(4):297–306. [PubMed] [Google Scholar]