Abstract
The Cereblon-CRL4 complex has been studied predominantly with regards to thalidomide treatment of multiple myeloma. Nevertheless, the role of Cereblon-CRL4 in Thalidomide Embryopathy (TE) is still not understood. Not all embryos exposed to thalidomide develop TE, hence here we evaluate the role of the CRL4-Cereblon complex in TE variability and susceptibility. We sequenced CRBN, DDB1, CUL4A, IKZF1, and IKZF3 in individuals with TE. To better interpret the variants, we suggested a score and a heatmap comprising their regulatory effect. Differential gene expression after thalidomide exposure and conservation of the CRL4-Cereblon protein complex were accessed from public repositories. Results suggest a summation effect of Cereblon variants on pre-axial longitudinal limb anomalies, and heatmap scores identify the CUL4A variant rs138961957 as potentially having an effect on TE susceptibility. CRL4-Cereblon gene expression after thalidomide exposure and CLR4-Cereblon protein conservation does not explain the difference in Thalidomide sensitivity between species. In conclusion, we suggest that CRL4-Cereblon variants act through several regulatory mechanisms, which may influence CRL4-Cereblon complex assembly and its ability to bind thalidomide. Human genetic variability must be addressed not only to further understand the susceptibility to TE, but as a crucial element in therapeutics, including in the development of pharmacogenomics strategies.
Subject terms: Next-generation sequencing, Molecular biology
Introduction
Thalidomide and its derivatives, lenalidomide and pomalidomide, are potent immunomodulatory (IMiDs), antiangiogenic and anti-inflammatory drugs1. Identified in 1961, Thalidomide Embryopathy (TE) is the consequence of embryonic exposure to thalidomide and is mainly characterized as limb reduction defects, although it can affect almost every organ and system2,3.
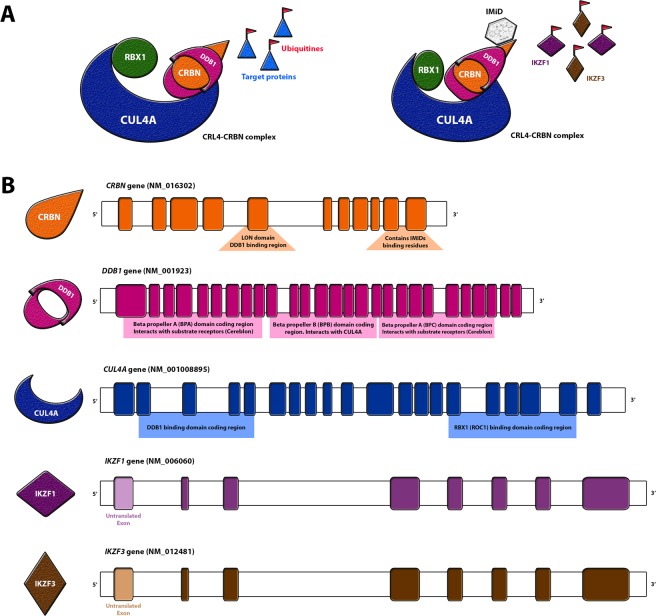
In 2010, Ito et al. demonstrated thalidomide’s ability to bind the Cereblon protein, a substrate receptor of an E3-ubiquitin-ligase complex (CRL4CRBN)4. CRL4 complexes have four subunits: the cullin protein CUL4A acts as a molecular scaffold in the assemble of the subunits; the adaptor protein DDB1 anchors the substrate receptor; the ROC1 protein, with the RING domain, catalyzes substrate ubiquitination; the substrate receptor, a DDB1 and CUL4-associated factor (DCAF), here represented by Cereblon (CRBN), targets the proteins to be ubiquitinated (Fig. 1A)5,6.
Figure 1.
CRL4 complex assembly in the presence and absence of immunomodulatory drugs, and its respective coding genes. (A) Graphic representation of the CRL4 complex in the absence (left) and presence (right) of thalidomide, where IKZF1 and IKZF3 are targeted; (B) CRL4 complex genes and encoded domains. IMiD = immunomodulatory drugs.
An early hypothesis suggested that thalidomide-CRL4CRBN binding might interfere in the ubiquitination of the target proteins by inhibiting CRL4CRBN complex formation4, affecting downstream limb development genes and resulting in TE4. However, it was later identified that the IMiDs cause an allosteric modification of the CRL4CRBN complex, resulting in ubiquitination and degradation of transcription factors (TFs) Ikaros (IKZF1) and Aiolos (IKZF3)(Fig. 1B)7,8. IKZF1 and IKZF3 are highly expressed in multiple myeloma, which lead to the discovery of the IMiDs as important therapeutic drugs in this condition7,9. Additional proteins potentially affected by the IMiDs binding CRL4CRBN have since been identified (Table 1); any effect on proteins might be IMiD-dependent or simply intensified by the drugs exposure. Nevertheless, the only identified therapeutic effect of the IMiDs remains the reduced expression of IKZF1 and IKZF3 in multiple myeloma9,10.
Table 1.
Literature main findings for CRL4CRBN-thalidomide interaction and induced protein effect.
| Pubmed ID | Protein Interaction | IMiD tested in the study | Is the mechanism IMiD dependent? | IMiD-protein interaction effect* |
|---|---|---|---|---|
| 27142104 | AGO2 | lenalidomide | yes | alters expression. |
| 26990986 | GS | none | no | |
| 27294876 | CD147 | none, Thal, Len, Pom | no | competition for CRBN binding reduces expression |
| 27294876 | MCT1 | none, Thal, Len, Pom | no | competition for CRBN binding reduces expression |
| 26021757 | CLC1 | none | no | |
| 19295130 | DDA1 | none | no | |
| 24292623 | IKZF1 | Thal, Len, Pom | yes | induced degradation |
| 24292623 | IKZF3 | Thal, Len, Pom | yes | induced degradation |
| 27468689 | TAB2 | none | no | |
| 27468689 | TRAF6 | none | no | |
| 25043012 | MEIS2 | lenalidomide | no | block MEIS2 binding to CRBN |
| 21232561 | AMPKA1 | none | no | |
| 23026050 | PSMB4 | none | no | |
| 27601648 | RABGEF1 | lenalidomide | no | prevents association to CRBN |
| 31591562 | TP63 | thalidomide | yes | neosubstrate of thalidomide-CRBN |
| 30067223 | SALL4 | Thal, Len, Pom | yes | thalidomide-CRBN induced degradation (C2H2 motif) |
Thal: thalidomide; Len: lenalidomide; Pom: pomalidomide; *differential mechanism in presence or absence of the IMiD.
Experiments by Ito et al. suggested that thalidomide-CRL4CRBN binding might be the primary mechanism of thalidomide teratogenicity4. Since then, the evaluation of thalidomide-Cereblon binding and CRL4CRBN complex has been the focus of most research efforts into potential therapeutic use, especially with regards to multiple myeloma. Our group has previously studied CRBN gene exons that encode the thalidomide-Cereblon binding region in individuals with TE to show that thalidomide-CRBN region is very conserved10. However, many gaps still remain in our understanding of the role of CRL4CRBN in TE.
Complex genetic conditions are often very challenging to evaluate because they are not a result of a single mutation, rather the aggregation of many low risk genetic variants, mainly in regulatory regions, combined with environmental influences11. In common, complex disorders, strategies including genome wide association studies (GWAS) and polygenic risk scores help in the identification and interpretation of low risk variants12,13. Rare, complex conditions cannot be evaluated by the same strategies because the relative low sample sizes impede a robust statistical approach. Low risk, regulatory variants, are mainly bypassed or poorly interpreted.
The aim of this study is to assess the sequence of CRBN, CUL4A, DDB1, IKZF1, and IKZF3 genes in Brazilian individuals with TE, in order to establish a molecular profile for each individual. We devised a unique score and a heatmap results system for robust interpretation of regulatory variants, and conducted methylation analyses of the CRBN gene to search for possible epigenetic marks caused by thalidomide. We also conducted a secondary data approach, investigating the differential gene expression (DGE) of the listed targets in embryonic cells and tissues after exposure to thalidomide. We then performed protein conservation analyses across different species that have well characterized TE in order to assess the different sensitivity to thalidomide in these organisms. Finally, through systems biology, we evaluated Gene Ontologies (GO) to understand the role of CRL4CRBN genes during embryonic development.
Results
Thalidomide Embryopathy sample represents generations of survivors
Thirty-five individuals with TE, born between 1959 and 2010 were included in this study. Individuals presented upper and/or lower limbs anomalies. Pre-axial longitudinal defects, mainly characterized by malformations in the first digit or intercalary transverse anomalies (phocomelia) were identified; none of the subjects were diagnosed with amelia. Additional external anomalies included eye, ear and other craniofacial malformations. Internal anomalies were mainly cardiovascular and genitourinary. A complete description of individuals clinical characteristics is available in previous research from our group3 and in Table S1.
Gene panel sequencing reveals extremely conserved coding regions
In order to evaluate variants of CRL4 genes, next-generation sequencing was performed using Ion PGM technology (ThermoFisher, USA). Briefly, a gene panel was designed to comprise of all exons, 50 bp of adjacent introns and untranslated regions (5’UTR and 3’UTR). The average depth of sequencing coverage was 491.5X, with at least 100X in 92.44% of the bases, and at least 20X in 97.73% of the regions sequenced.
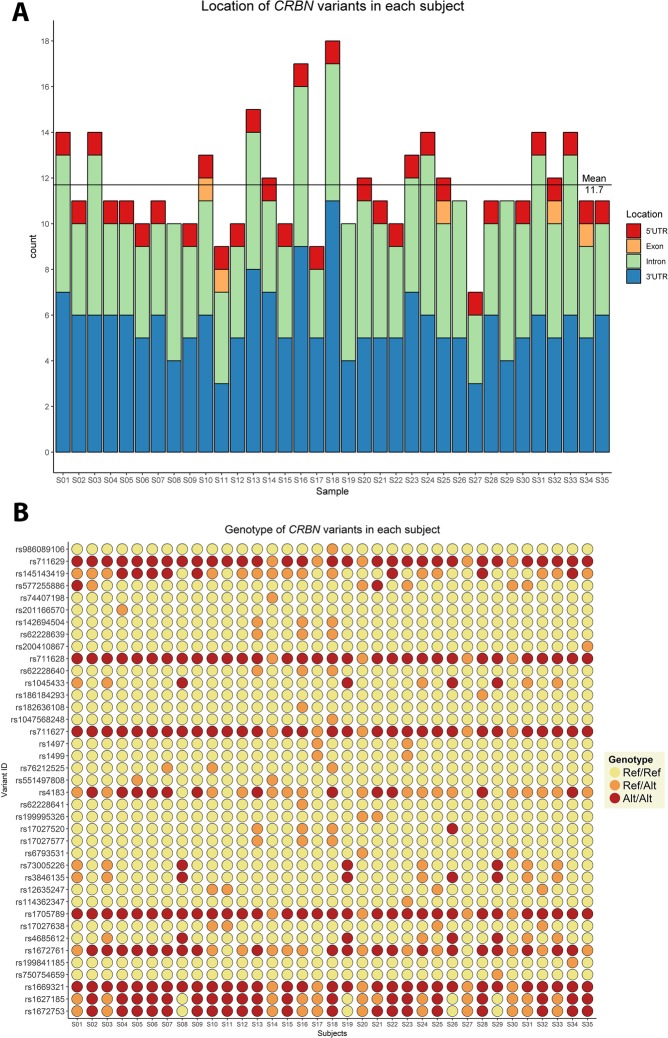
A total of 145 variants were identified across the five genes (Table S2). CUL4A presented three exonic variants, whilst the other four genes, CRBN, DDB1, IKZF1, and IKZF3 presented only two coding variants; 7.5% of variants are detected in coding regions. CRBN and IKZF1 had the highest number of variants, most located in the 3’UTR region. CRBN presented the highest mean of variants per individual (Fig. 2A); the genotype for each subject is represented in Fig. 2B (for DDB1, CUL4A, IKZF1, and IKZF3 genotypes, see Figs. S1–S4). Three new variants were identified, two in IKZF1 and one in DDB1. The low number of exonic variants in all genes, and the amount of 3’UTR alterations in CRBN and IKZF1 in the sample is remarkable. Moreover, according to the population database, DDB1 presented the highest number of variants; in our TE sample this was the second most conserved gene. A description of the number of variants identified for each gene and their location can be in Fig. 3.
Figure 2.
Distribution of CRBN variants in subjects with Thalidomide Embryopathy, by gene position and genotypes. (A) Number of variants in the CRBN gene in each subject with TE, and their respective gene location; (B) Genotypes of the CRBN variants in each subject with TE. Yellow: Ref/Ref – individual does not present the variant; Orange: Ref/Alt – individual is heterozygous for the variant; Red: Alt/Alt – individual is homozygous for the variant.
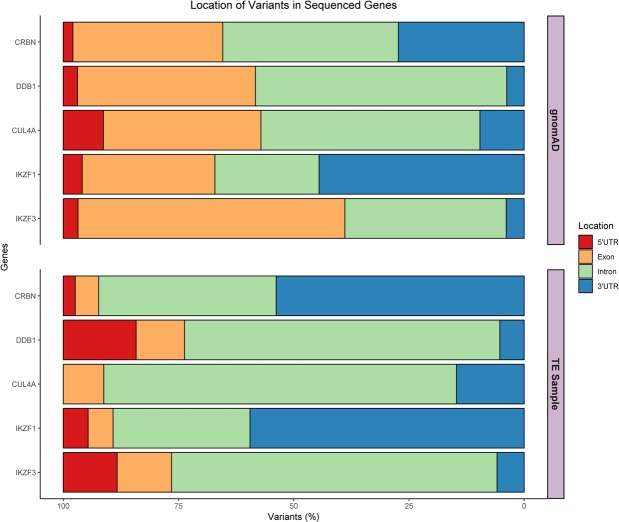
Figure 3.
Variants in the Non-Finnish European individuals of the gnomAD database compared to those in the TE sample. Comparison of the variant location distributions between gnomAD and TE, and the percentage of total gene variants at each location.
To verify whether the distribution of identified variants across the genes was similar to a comparison sample, we assessed the variants in the same genomic regions of a gnomAD non-Finnish European sample (subjects without congenital anomalies). This analysis suggested the occurrence of variants between the two samples was statistically different (p < 0.001).
Extensive in silico functional predictions suggest some variants may affect gene regulatory mechanisms
Missense variants rs2302757 (CUL4A, NM_001008895) and rs112301322 (IKZF3, NM_012481) were evaluated in SIFT and PolyPhen-2 tools and were predicted to be “tolerated”. Analysis of all nine synonymous variants in SILVA algorithm categorized them as “likely benign”. The effect of all the polymorphisms was evaluated in MutationTaster, PredictSNP-2 and DANN. Analyses also included alteration of splicing and polyA motifs, TFs and microRNA binding sites, CpG islands sizes and mRNA conformation and stability. Characteristics and details of the predictors used are available in Table S3. The profile of each polymorphism regarding population frequency, linkage disequilibrium and previously associated phenotypes were also evaluated.
In a gnomAD non-Finnish European sample, almost 30% (43/145) of the variants encountered were considered rare (Minor Allelic Frequency, MAF < 0.01). Of the 145 variants encountered, 24 are inserted in CpG island regions. Functional prediction demonstrated that ten of these, of which four are found in the CUL4A gene, might cause a creation, disruption or size alteration to the CpG islands. microRNA analysis revealed ten variants altered the binding site for miRNAs expressed in human embryonic stem cells (hESC); none of these variants was located in IKZF3.
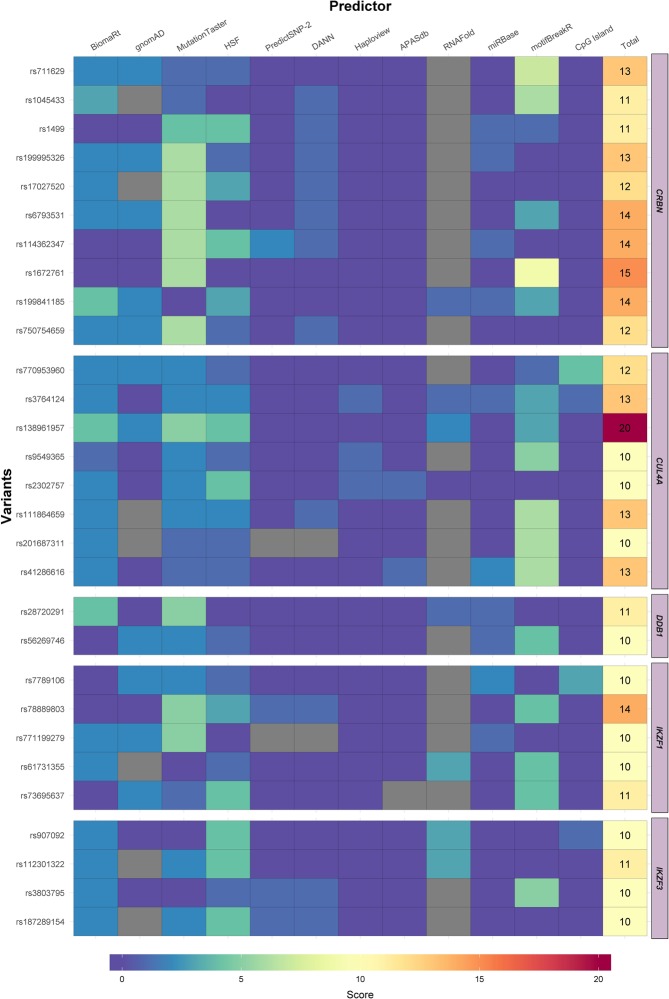
In order to better comprehend the results of functional predictions and the consequences of the regulatory variants, a score was applied based on the severity of the predicted effect (Table S4). A heatmap then comprised all the functional predictions and all polymorphisms in each gene to give a final score (Figs. S5–S9), ranging from 1 to 20. All variants that presented a score of at least 10 points are comprised in Fig. 4. Each of the three new variants identified, #0000439088 (DDB1 gene), #0000439108, and #0000439110 (IKZF1 gene), were scored over 10. The heatmap reveals rs138961957 of CUL4A gene as the variant possessing the highest score (20 points), hence the most deleterious to the regulatory mechanisms of all genes examined. rs138961957 is a synonymous c.1392 G > T polymorphism in exon 13 (NM_001008895). This is a very rare variant that was present in two individuals in the TE sample (MAF = 1.499e-05 in European Non-Finnish, according to ExAc database). According to the functional predictions of motifBreakR and MutationTaster c.1392 G > T could interfere with splicing and IKZF1 binding site consensus sequence. It may also affect messenger RNA (mRNA) conformation, resulting in reduced mRNA stability (Fig. S10). Variants rs61731355 of IKZF1 (Fig. S10), and rs907092 and rs112301322 of IKZF3, also synonymous variants, may too alter mRNA conformation. Codon usage in the translation process was evaluated in these three variants, along withCUL4A rs138961957. CUL4A rs138961957 and IKZF1 rs61731355 variants lead to incorporation of an alternative codon in comparison to the codon most frequently incorporated, demonstrated both when evaluating gene translation and when accessing the whole human genome (File S2). The other synonymous variation, IKZF3 rs907092, results in incorporation of the most frequent codon upon evaluation of IKZF3 gene translation, but incorporation of a less frequent one on evaluation of the whole genome (File S2).
Figure 4.
Most relevant variants of CRBN, CUL4A, DDB1, IKZF1 and IKZF3 genes according to the heatmap score. Heatmaps for the genes studied, comprised of variants achieving a score of ≥10 in the in silico functional prediction.
The CRBN gene had ten variants with a score of at least ten points. The majority of these polymorphisms scored highly because any splicing site alteration before exon 10 might lead to the loss of the C-terminal region and thalidomide binding site. Moreover, CRBN rs711629 also affected IKZF1 binding site consensus sequence.
IMiDs cause a conformational change in the CRL4CRBN complex, hence it is important to verify the position of variants across the protein domains (Fig. S11). Variants with higher score (black flags) are usually in proximity to exons that do not encode the domains for assembling the CRL4CRBN complex, the exception being the CUL4A region that encodes the ROC1 binding site. Additionally, no variant was identified around the IMiDs binding residues.
In summary, a low number of exonic variants were identified in individuals with TE, and most variants are synonymous. Many of the non-coding variants encountered are rare and impact regulatory mechanisms that may influence gene expression and protein function. CUL4A variant rs138961957 was considered the most deleterious.
A high number of variants in individuals with TE are related to pre-axial longitudinal upper limb anomalies
To determine whether the variants identified could have a clinical effect we performed a statistical analysis correlating the variants with the spectrum of congenital anomalies of the individuals with TE. Evaluation was performed (1) considering the stochastic effect of variants in each individual (absolute number of variants); and (2) considering the sum of the scores for each variant.
Results analyzing absolute number of variants were not statistically significant, although the sum of all the variants scores is statistically associated to one group of congenital anomalies in the upper limbs: pre-axial longitudinal defects (p = 0.048), which mainly affects the thumbs. Individuals with these patterns of malformations (n = 10) had a mean score of 228.9 points, whilst subjects with intercalary transverse defects (n = 14), known as phocomelia, had an average score of 195 points (Table S5).The effect is more significant if we look at the score of CRBN variants separately: pre-axial longitudinal defects were associated to a score of 94.9 vs. 78 points of the intercalary transverse anomalies (p = 0.021) (Table S5).
To verify whether this effect of stochastic variants is inherent to the studied genes or if it is associated to TE, the rate of incidence of the variants in our TE sample was compared to that of 99 individuals of the 1000Genomes sample (Europeans, CEU group).Individuals with TE present a statistically significant summation effect of variants in the five genes when compared with the control sample. This association is valid both when evaluating the number of variants and when analyzing heatmap scores (Table S6). This effect was also significant when evaluating CRBN and DDB1 separately for total numbers of variants and heatmap scores (Table S6). For IKZF1, this association was only statistically significant when evaluating the heatmap scores. The presence CUL4A and IKZF3 variants was not statistically different between the groups (Table S6).
To further explore this association, we divided the TE sample into two groups; individuals with pre-axial longitudinal defects (less severe anomalies, mostly affecting thumbs); individuals with intercalary transverse defects, more severe malformations affecting the long bones and leading to phocomelia. An ANOVA-test was performed between the two TE groups and the 99 individuals of the CEU sample from 1000Genomes. Statistical analysis suggested the statistical association previously identified is probably due to the high score of the individuals with pre-axial upper limb anomalies when compared to the other two groups (Table S7).
Finally, based on heatmap results we evaluated any correlation between the highest ranked variants of our study and genotype and TE endophenotypes. Variant rs1045433, a SNP (NM_016302, c.*1123 A > G) in the 3’UTR with a score of 11 points in our heatmap, was statistically significant, demonstrating a high frequency in individuals with pre-axial limb anomalies. Six individuals with the G allele (two in heterozygosis and four in homozygosis) present pre-axial limb anomalies, only one individual heterozygous for the G SNP presents with an intercalary transverse anomaly (p = 0.004) (Table S8).
CRL4CRBN complex genes are ubiquitously expressed in developing human limbs
To explain the statistical association between genotypes and pre-axial limb anomalies, we performed a DGE analysis from secondary data obtained from the Gene Expression Omnibus (GEO) database. Samples of the first vs. second-to-fifth digits from the upper limbs of human embryos at embryonic day 44 were obtained from GSE4241314. None of the genes evaluated were differentially expressed in the first digit compared to the second-to-fifth digits samples (Table S9).
Despite the lack of association between DGE analyses and the variants identified, we aimed to evaluate whether these genes were highly expressed in the developing human upper limbs. Fig. S12 demonstrates DDB1 and RBX1 are highly expressed (>200 transcripts per million, TPM) and that IKZF3 expression was below the minimal levels of detection (<0.5 TPM). To act as controls, we assessed the expression profiles of genes crucial during limb development such as SALL4, BMP4, FGF8, FGF10, and SHH; the latter especially highly expression in early stages of limb development. Gene expression levels for CDH5 (encodes ubiquitously expressed cadherin cell-adhesion protein 5), PAX6 (paired-box development gene 6, expression restricted to brain, pancreas and eyes), and TLR4 (toll-like receptor 4, not expressed in this stage of development) were analyzed as negative controls.
From the gene expression profile, it is not possible to rule out a role of the CRL4CRBN complex during upper limb development, indeed it is possible DDB1 and RBX1 play important roles in this tissue and stage of development. The role of the CRL4-complex might be independent of CRBN, acting with other substrate receptors, for example DCAF proteins.
CRBN is not methylated in individuals with Thalidomide Embryopathy
It is not known whether thalidomide-Cereblon binding results in epigenetic alterations of the human genome. Since gene panel analysis demonstrated some of the identified gene variants could impact CpG islands size, we evaluated 17 CpG dinucleotides located in the promoter region of CRBN by bisulfite sequencing (Fig. S13), including cg20912439 and cg15874300. We did not identify any methylation at these sites in the sample of 35 individuals with TE (Fig. S13). Additionally we assessed a methylation array (GSE48472)15 from the GEO database and did not identify methylation at the CRBN promoter sites when evaluating human saliva samples.
Purifying selection explains conservation of the proteins across species
To verify whether the observed gene conservation is present across species differently affected by thalidomide exposure, we performed a comparative analysis of ROC1 and the five target genes in eight different species: human, rhesus monkey, crab-eating macaque, rabbit, chicken and zebrafish (present typical TE) versus mouse and rat (less sensitive to thalidomide, do not present typical TE).
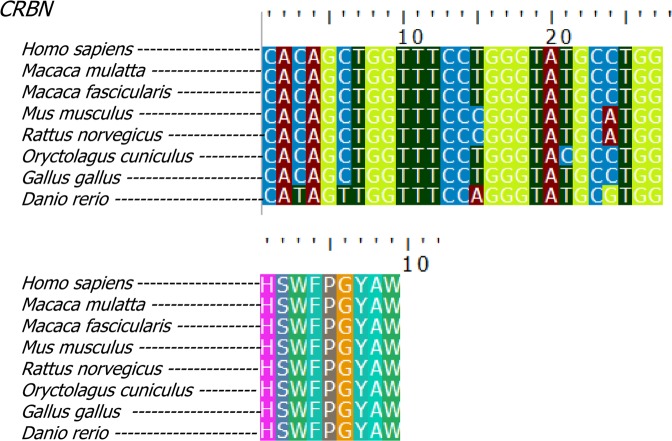
On initial NsSites analysis, the P-Values obtained reflect a high state of conservation: ω = 0.0306 at 89% of CRBN sites (see Table S10). Across all eight species, CRBN complex genes are conserved with the exception of IKZF3, which is absent in zebrafish but well conserved among remaining species. Comparison of TE-sensitive species to that of mouse and rat revealed equal conservation between the two groups (Fig. 5, File S1). Purifying selection is the largest force acting on these genes to remove deleterious alleles (P-Value < 1), and according to these results, it is not possible to explain resistance of mouse and rat to TE.
Figure 5.
Cereblon gene and protein conservation across different vertebrate species (top) alignment of CRBN CULT-domain coding region and translated protein (bottom). Species affected by typical TE are Homo sapiens (human), Macaca mulatta (Rhesus monkey), Macaca fascicularis (crab-eating monkey), Oryctolagus cuniculus (rabbit), Gallus gallus (chicken) and Danio rerio (zebrafish). The rodents Rattus norvegicus (rat) and Mus musculus (mouse) do not present typical TE, after exposure to thalidomide during embryonic development. Nucleotides (top) and amino acids (bottom) alignment between the eight species studied. Only synonymous mutations are identified in the region. Source: UNIPROT.
The effect of Thalidomide on target gene expression is different between mice and humans
In order to determine whether thalidomide affects CRBN, DDB1, CUL4A, IKZF1, and IKZF3 expression in different species, we assessed three studies from the GEO database: mouse (GSE61306)16, crab-eating monkey (GSE15542)17, and human (GSE63935)18. Exposing mouse embryonic stem cells (mESC) to thalidomide or saline solution demonstrates that thalidomide reduces Cul4a relative expression for 48 hours post-exposure (p = 0.0068 at 24 hours, p = 0.0195 at 48 hours). Crbn, Ddb1 and Rbx1 (which encodes Roc1) relative expression is likewise reduced (p = 0.0244, p = 0.0365 and p = 0.0117, respectively). 72 hours after thalidomide exposure none of the genes differs statistically in expression levels compared to control samples (Table S11). In crab-eating monkey secondary data analysis, only Cul4a was reduced 6 hours post-thalidomide when compared to the controls(p = 0.0121) (Table S12). Two days and six days after exposure of thalidomide to human pluripotent stem cells (hPSC), the DGE was not statistically different for any of the genes (Table S13).
Network analysis of the CRL4CRBN demonstrates its role in repair and cancer pathways
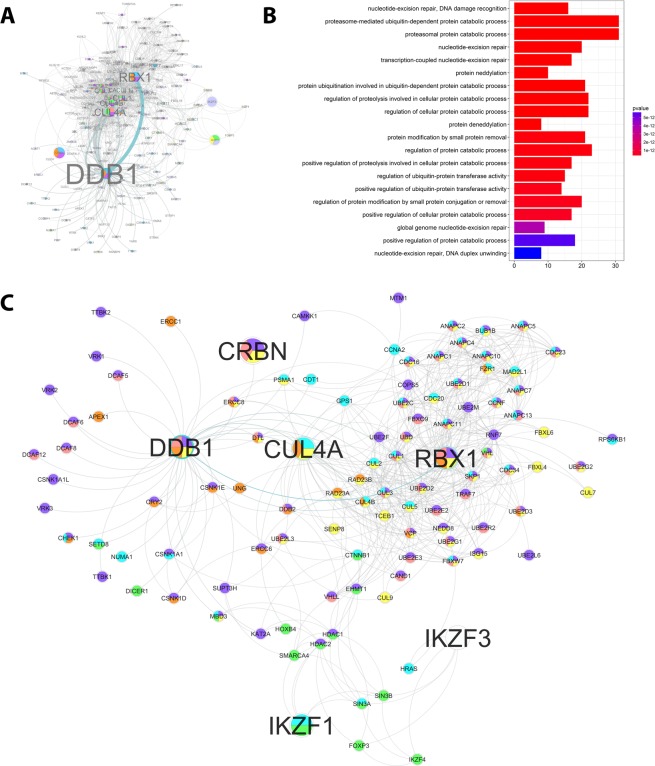
To help determine the roles of the CRL4CRBN complex during embryonic development, a systems biology network was established. Analysis of protein-protein interactions (PPI) revealed 199 targets of CRBN, CUL4A, DDB1, ROC1, IKZF1, and IKZF3; DDB1 appears to be the main hub of the network (Fig. 6A).
Figure 6.
CRL4 complex protein-protein interactions and gene ontologies. (A) CRL4 protein-protein interactions (PPI), with letter size corresponding to the number of interactions; (B) Gene ontologies (GO) enrichments for the proteins that interact with the CRL4 complex; (C) CRL4 complex PPI network representing GO more relevant in terms of TE. Blue: cell cycle; purple: proteolysis; pink: post-translational modification; yellow: ubiquitination; orange: DNA repair; green: gene expression negative regulation.
Enrichment analysis of Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) Pathways identified biological processes associated to DNA repair and ubiquitination as the most represented for the CRL4CRBN network (Figs. 6B and S14). Additional associated biological processes identified in analysis of GOs that may aid understanding of the effect of thalidomide on embryonic development are: cell cycle; proteolysis; post-translational protein modification; negative regulation of gene expression (Fig. S14). There was no above-average representation of processes associated with “embryogenesis” or “development” (Fig. 6C).
Discussion
Genes of the Ubiquitin E3 ligase complex CUL4A, DDB1, their receptor CRBN, and transcription factors IKZF1, and IKZF3, are well-conserved, especially in the regions that encode main protein domains. It is estimated that around 40% of the children affected by TE died in infancy due to severe internal malformations19, and perinatal complications of TE may have caused many more stillbirths. It cannot be determined whether pregnancy loss due to TE complications was associated with deleterious CRL4CRBN complex variants.
Two studies recently demonstrated that Cereblon targets protein SALL4 for degradation in a thalidomide-dependent manner20,21, a process which appears to be species-specific when evaluating mice, rabbits and humans, due to missense mutations in the SALL4 coding sequence, but not in CRBN20,21. Our results support these studies, demonstrating genetic variation of CRBN genetic variation is not sufficient to explain occurrence of TE. SALL4 mutations in humans cause Duane-radial-ray syndrome22,23, a phenocopy of deformities caused by TE. In a previous study from our group, we performed an analysis of SALL4 in TE individuals and identified 15 variants. Of these 15, ten were located in exons, seven synonymous and three missense24. The lack of exonic variants identified in our study prevented the analysis of the effects of combined SALL4 and CRBN variants.
In this study 92.5% of identified variants were located in regulatory regions. The proportion of exonic variants present in individuals with TE was significantly different compared to the gnomAD database sample; TE individuals have much less coding variants than general sample from gnomAD database. This underrepresentation of coding variants might be an effect of the small sample size or might be inherent of the characteristics of these subjects. GWAS reveal, however, almost 90% of disease-associated variants are located outside protein coding regions11, and this combined with advancing NGS technologies are promoting studies into regulatory variants and their impact on regulatory elements25. The few germinative CRBN variants that have been linked to thalidomide responsiveness in treatment of MM are found in regulatory regions26–28. Indeed, in a microarray of more than 300,000 exonic SNPs performed in MM patients treated with a chemotherapy scheme comprising thalidomide and bortezomib, no significance was found in the presence of any variant29. The continued study of regulatory variants is of great importance towards determining the cause and predictability of thalidomide pharmacogenomic and toxicogenomic effects.
We integrated all generated functional prediction data in a heatmap produced a score for each variant. This methodical approach highlighted those variants more likely to impact TE development, and is useful for further studies of complex disorders with small sample sizes.
In accordance with previous findings10, individuals with TE presented a high number of rare variants (MAF < 0.01). Rare variants have been widely studied in complex traits including diabetes30, Alzheimer31 and cancer32, reflecting a shift in human genetic research of multifactorial diseases33.Comparison with a European sample of the 1000Genomes project demonstrated individuals with TE present a summation effect of identified variants. The results were similar when evaluating the total number of variants in TE individuals and comparing scores determined in the heatmap. Heatmap scores also suggest synonymous variants may have effects such as altered mRNA conformation and stability and codon usage bias, which influences a series of molecular aspects such as RNA stability, protein folding and gene expression34. Indeed, a number of studies have demonstrated the effect of synonymous mutations on efficiency of translation speed and protein folding and a summation effect of synonymous variants may increase the probability of reduced translation efficiency35,36. Synonymous mutations have been linked to over 50 genetic diseases37, with at least 4% deleterious to splicing motifs38. This highlights the importance of further investigation into variants identified in this study, in particular CUL4A rs138961957. Statistical analysis revealed no one variant is a determinant for TE incidence, the causative effect linked only to thalidomide. CRBN 3’UTR variant rs1045433 is associated with upper limb pre-axial longitudinal anomalies, but not associated with a response to thalidomide treatment26. Upper limb pre-axial longitudinal anomalies in upper limbs were associated with higher heatmap scores for multiple genes when evaluating TE individuals only or in comparison with the 1000Genomes European sample. There was no differential expression of analyzed genes in the first digit (pre-axial) compared to second through to fifth, however we cannot rule out differential expression of CRL4CRBN downstream targets. It can be hypothesized that the summation of regulatory variants might influence expression and consequent effect of CRBN, impacting the response of exposure to thalidomide. Studying genome and transcriptome analyses after thalidomide exposure may help confirm or disprove this hypothesis.
We highlight the regulatory effect, leading to alterations in mRNA conformation, of many evaluated variants on the binding site of IKZF1, a TF involved in treatment of MM with thalidomide7,9. Although predictions pointed to thalidomide affecting CpG island properties, in the experiments to evaluate epigenetic features of TE individuals we found no methylation in the CRBN promoter region.
Reduced expression of evaluated genes in a mESC assay was the only association that suggests an impact of thalidomide on transcription rates. Although mice and rats do not present typical TE, a justification for the use of mESCs is the crucial cross regulation between E3-ligases and deubiquitinating enzymes during pluripotency and differentiation of the stem cells39. Additional functions of the CRL4 complex during development include regulation of cell-cycle progression40 and genome reprogramming at preimplantation41. However, the role of the CRL4CRBN during embryogenesis is not well understood. CRBN, CUL4A, DDB1 and RBX1 are expressed in digits at human embryonic day 44, therefore we cannot rule out a role for CRL4CRBN in normal limb development. It is possible that inhibition of CRL4CRBN complex formation by thalidomide results in severe downregulation of the molecular mechanisms that are CRBN-independent during normal embryogenesis.
CRBN is a well conserved protein across different vertebrate species4. Our detection of purifying selection and the high level of gene expression suggests CRBN plays a role in embryogenesis, particularly during brain development. Indeed, CRBN is linked to mild mental retardation syndrome42; since thalidomide is a synthetic molecule, purifying selection pressures acting on CRBN cannot be explained by its ability to bind thalidomide.
The resulting proteins of the genes included in this study have well characterized roles during ubiquitination and DNA repair processes but no overrepresented GOs during development. A few known targets may be associated with embryogenesis, and a systems biology approach would be a preferred to identify new targets.
This is the first comprehensive study evaluating the CRL4CRBN complex in TE, despite a small sample size. Future studies would ideally include a control group of Brazilians exposed to thalidomide that did not develop TE.
Teratogenesis is a systemic event initiated an exogenous, teratogenic agent such as thalidomide43. Independent of affected molecular mechanisms, for example CRBN-binding, angiogenesis and oxidative stress, gene expression is, most likely deregulated upon teratogenic exposure resulting in a cascade of cellular events ultimately impacting signaling pathways44. Outcomes of teratogenic exposure might include lethality, occurrence of malformations such as TE, or normal development45. One of the predictions for individual response to exposure may be genomic variability. The use of animal models and cell lines are invaluable in determining the molecular mechanisms of a teratogen, but they lack intraspecific genomic variability. The approach in this study is similar to well stablished pharmacogenomics and toxicogenomic methods, although from a teratogenic perspective.
This study aimed to provide insight into the effect of thalidomide on CRL4CRBN complex formation and consequent development of TE. We discovered that coding regions of the genes in this complex are largely comparable to controls in TE affected individuals, and further studies into the effects of the many regulatory variants identified are important in further understanding the development of TE and predicting individual response to thalidomide exposure.
Materials and Methods
Ethical issues
These projects are approved by the Committee of Ethics in Research of Hospital of Clinics of Porto Alegre, Brazil (#10-0244, #10-0410, and #17-0248). All methods were carried out in accordance with relevant guidelines and regulations. Informed consent was obtained from all subjects or from a parent and/or legal guardian (subjects under 18).
Sample collection and DNA extraction
Subjects included in the study were assessed through the Brazilian Association of People with Thalidomide Syndrome (ABPST). Informed consent was obtained from all subjects or from a parent/legal guardian for the subjects under 18 years. Saliva samples were collected with Oragene OG-500 kit (Genotek, USA). DNA extraction was performed according to the manufacturer’s instructions.
Gene panel analysis
A customized gene panel was designed with Ion Ampliseq (ThermoFisher, USA). All the exons and untranslated regions (5’UTR and 3’UTR) of the genes CRBN (NM_016302), DDB1 (NM_001923), CUL4A (NM_001008895), IKZF1 (NM_006060), and IKZF3 (NM_012481) were included, as well as 50 bp of adjacent introns. Next generation sequencing was performed with Ion PGM (ThermoFisher, USA) technology with a 316Chip. Non-covered regions were repeated by Sanger sequencing using an ABI 3130XL genetic analyzer. IonReporter software v.5.2 was used for obtaining the variant call file (vcf), evaluating all the alterations encountered in the analyzed sample.
Variants interpretation and in silico functional prediction
Variants were catalogued through Ion Reporter software v.5.2 (ThermoFisher, USA). Functional prediction was performed using MutationTaster46, SIFT47, PolyPhen-248, SILVA49, RNAFold50, miRbase51, Human Splicing Finder 352, PredictSNP-253, ApasDB54, EnhancerAtlas55, MethPrimer56, and Haploview v.4.257, according to the developers instructions. Packages motifbreakR58 and biomaRt59 were integrated in RStudio v.3.3 for the functional predictions.
Statistical analysis and figures
Chi-square and t-tests were performed in SPSS v.18 software. Other statistical analyses were performed in R environment (v.1.0.6). Figures were assembled in RStudio v.3 through package ggplot260.
Methylation analysis
Bisulfite conversion in genomic DNA was performed with Cell-to-CpG bisulfite kit (ThermoFisher, USA). Promoter regions were sequenced from genomic DNA. Primers for methylation analyses were designed with MethPrimer56. Original and converted DNA was evaluated through Sanger sequencing in an ABI 3130XL genetic analyzer.
Protein conservation analysis
The sequences were aligned using MUSCLE algorithm in MEGA 7 software. To verify positive selection, the NsSites test, included in PAML 4.961, was applied.
Differential gene expression analysis
Expression studies were downloaded from Gene Expression Omnibus (GEO) database (NCBI, USA)62,63. Secondary data analysis was performed in RStudio v.3.3. Packages oligo64 and affy65 were used for microarray analysis, whilst RNAseq data was evaluated through edgeR66 package. Microarray data was normalized through robust multi-array average (RMA) and RNA seq data by trimmed mean of M-values (TMM). Differential gene expression (DGE) was defined as adjusted P-value < 0.05.
Systems biology analyses
Protein-protein interaction networks were obtained from STRING database v.10.5 and transferred to Cytoscape software v.3.6. Cytoscape apps BINGO and GOlorize were used for Gene Ontology visual representation. Gene ontology and KEGG pathway enrichment analyses were performed in clusterProfileR67 package, RStudio v.3.3.
Supplementary information
Acknowledgements
We are indebted to the Brazilian Association of People with Thalidomide Syndrome (ABPST) for the support in the identification and monitoring of the TE patients. We deeply thank A. Dupont, M. Furtado, D. Leao, B. Rengel, M. Siebert and Professor Ursula Matte for the technical assistance, and B. Alemar, I. Sartor, and I. Vieira for the scientific support. We also thank Dr Neil Vargesson of the University of Aberdeen for all the helpful discussions and critical comments. The authors would like to acknowledge the financial support: INAGEMP (National Institute of Population Medical Genetics; Grant CNPq 573993/2008-4, 465549/2014-4, FAPERGS 17/2551.0000521-0 and CAPES); FIPE/HCPA (GPPG #10-0244, #10-0410 and #17-0248); CAPES (Coordination of Improvement of Higher Education Personnel, Grant 88881.132344/2016-01) and CNPq (National Council of Scientific and Technologic Development, Grant 423249/2016-9 (Universal Project), 312993/2017-0, and 156158/2018-3).
Author contributions
T.W.K. contributed devising the concept, designing and conducting the experiments, performing the in silico and statistical analyses, and writing the manuscript. J.A.G. contributed devising the concept, designing the experiment and performing in silico analyses. G.B.C.G. contributed performing conservation analyses and writing the manuscript. L.R.F. contributed designing the experiment and correcting the manuscript. V.R.P.C. and M.R.M. contributed devising and supervising the analyses. M.T.V.S. contributed in the clinical evaluation and supervising the analyses. L.S.F. contributed in the clinical evaluation, supervising the execution and analyses. F.S.L.V. contributed devising the concept, designing the experiments, supervising the analyses and correcting the manuscript. All authors discussed the results and contributed scientifically to the manuscript.
Data availability
The datasets generated and analyzed during the current study are not publicly available to maintain patient confidentiality. Moreover, this type of request has not been previously approved by participants nor the Human Research Committee. This data could, however, be available (anonymously) from the corresponding author on reasonable request. Gene panel sequencing files have been deposited in the repository Sequence Read Archive (SRA) of the National Center for Biotechnology Information (NCBI), under accession number SRP160424. The new variants identified in the present study were submitted to the Leiden Open Variation Database (LOVD3), under numbers #0000439088 (DDB1), #0000439108 and #0000439110 (IKZF1). Gene expression analyses were performed with data from public repository Gene Expression Omnibus (GEO) and are available under the following numbers: GSE42413, GSE61306, GSE15542, GSE63935, and GSE48472.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Thayne Woycinck Kowalski, Email: thaynewk@gmail.com.
Fernanda Sales Luiz Vianna, Email: fslvianna@gmail.com.
Supplementary information
is available for this paper at 10.1038/s41598-020-57512-x.
References
- 1.Vargesson N. Thalidomide-induced teratogenesis: history and mechanisms. Birth Defects Res. C. Embryo Today. 2015;105:140–156. doi: 10.1002/bdrc.21096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lenz W. A short history of thalidomide embryopathy. Teratology. 1988;38:203–215. doi: 10.1002/tera.1420380303. [DOI] [PubMed] [Google Scholar]
- 3.Kowalski Thayne Woycinck, Sanseverino Maria Teresa Vieira, Schuler-Faccini Lavinia, Vianna Fernanda Sales Luiz. Thalidomide embryopathy: Follow-up of cases born between 1959 and 2010. Birth Defects Research Part A: Clinical and Molecular Teratology. 2015;103(9):794–803. doi: 10.1002/bdra.23376. [DOI] [PubMed] [Google Scholar]
- 4.Ito T, et al. Identification of a primary target of thalidomide teratogenicity. Science. 2010;327:1345–1350. doi: 10.1126/science.1177319. [DOI] [PubMed] [Google Scholar]
- 5.Sarikas A, Hartmann T, Pan ZQ. The cullin protein family. Genome Biol. 2011;12:220. doi: 10.1186/gb-2011-12-4-220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Collins I, Wang H, Caldwell JJ, Chopra R. Chemical approaches to targeted protein degradation through modulation of the ubiquitin-proteasome pathway. Biochem. J. 2017;474:1127–1147. doi: 10.1042/BCJ20160762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lu G, et al. The myeloma drug lenalidomide promotes the cereblon-dependent destruction of Ikaros proteins. Science. 2014;343:305–309. doi: 10.1126/science.1244917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Liu Y, et al. A novel effect of thalidomide and its analogs: suppression of cereblon ubiquitination enhances ubiquitin ligase function. FASEB J. 2015 doi: 10.1096/fj.15-274050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lindner S, Krönke J. The molecular mechanism of thalidomide analogs in hematologic malignancies. J. Mol. Med. (Berl.) 2016;94:1327–1334. doi: 10.1007/s00109-016-1450-z. [DOI] [PubMed] [Google Scholar]
- 10.Vianna FS, et al. Genomic and in silico analyses of CRBN gene and thalidomide embryopathy in humans. Reprod. Toxicol. 2016;66:99–106. doi: 10.1016/j.reprotox.2016.10.003. [DOI] [PubMed] [Google Scholar]
- 11.Edwards SL, Beesley J, French JD, Dunning AM. Beyond GWASs: illuminating the dark road from association to function. Am. J. Hum. Genet. 2013;93:779–797. doi: 10.1016/j.ajhg.2013.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lambert Samuel A, Abraham Gad, Inouye Michael. Towards clinical utility of polygenic risk scores. Human Molecular Genetics. 2019;28(R2):R133–R142. doi: 10.1093/hmg/ddz187. [DOI] [PubMed] [Google Scholar]
- 13.Jansen, A. G. et al. Psychiatric Polygenic Risk Scores as Predictor for Attention Deficit/Hyperactivity Disorder and Autism Spectrum Disorder in a Clinical Child and Adolescent Sample. Behav Genet, 10.1007/s10519-019-09965-8 (2019). [DOI] [PMC free article] [PubMed]
- 14.Cotney J, et al. The evolution of lineage-specific regulatory activities in the human embryonic limb. Cell. 2013;154:185–196. doi: 10.1016/j.cell.2013.05.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Slieker RC, et al. Identification and systematic annotation of tissue-specific differentially methylated regions using the Illumina 450k array. Epigenetics Chromatin. 2013;6:26. doi: 10.1186/1756-8935-6-26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gao X, Sprando RL, Yourick JJ. Transcriptomic changes in mouse embryonic stem cells exposed to thalidomide during spontaneous differentiation. Data Brief. 2015;4:199–202. doi: 10.1016/j.dib.2015.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ema M, et al. Fetal malformations and early embryonic gene expression response in cynomolgus monkeys maternally exposed to thalidomide. Reprod. Toxicol. 2010;29:49–56. doi: 10.1016/j.reprotox.2009.09.003. [DOI] [PubMed] [Google Scholar]
- 18.Schwartz MP, et al. Human pluripotent stem cell-derived neural constructs for predicting neural toxicity. Proc. Natl Acad. Sci. USA. 2015;112:12516–12521. doi: 10.1073/pnas.1516645112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Smithells RW, Newman CG. Recognition of thalidomide defects. J. Med. Genet. 1992;29:716–723. doi: 10.1136/jmg.29.10.716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Donovan, K. A. et al. Thalidomide promotes degradation of SALL4, a transcription factor implicated in Duane Radial Ray syndrome. Elife710.7554/eLife.38430 (2018). [DOI] [PMC free article] [PubMed]
- 21.Matyskiela ME, et al. SALL4 mediates teratogenicity as a thalidomide-dependent cereblon substrate. Nat. Chem. Biol. 2018;14:981–987. doi: 10.1038/s41589-018-0129-x. [DOI] [PubMed] [Google Scholar]
- 22.Kohlhase J, et al. Okihiro syndrome is caused by SALL4 mutations. Hum. Mol. Genet. 2002;11:2979–2987. doi: 10.1093/hmg/11.23.2979. [DOI] [PubMed] [Google Scholar]
- 23.Borozdin W, et al. SALL4 deletions are a common cause of Okihiro and acro-renal-ocular syndromes and confirm haploinsufficiency as the pathogenic mechanism. J. Med. Genet. 2004;41:e113. doi: 10.1136/jmg.2004.019901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gomes JDA, et al. The role of ESCO2, SALL4 and TBX5 genes in the susceptibility to thalidomide teratogenesis. Sci. Rep. 2019;9:11413. doi: 10.1038/s41598-019-47739-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Rojano Elena, Seoane Pedro, Ranea Juan A G, Perkins James R. Regulatory variants: from detection to predicting impact. Briefings in Bioinformatics. 2018;20(5):1639–1654. doi: 10.1093/bib/bby039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Butrym A, et al. Cereblon and IRF4 Variants Affect Risk and Response to Treatment in Multiple Myeloma. Arch. Immunol. Ther. Exp. (Warsz.) 2016;64:151–156. doi: 10.1007/s00005-016-0442-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Szudy-Szczyrek A, et al. Polymorphisms in the promoter region of the. Oncotarget. 2018;9:24054–24068. doi: 10.18632/oncotarget.25307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mlak Radosław, Szudy‐Szczyrek Aneta, Mazurek Marcin, Szczyrek Michał, Homa‐Mlak Iwona, Mielnik Michał, Chocholska Sylwia, Jankowska‐Łęcka Olga, Małecka‐Massalska Teresa, Hus Marek. Polymorphisms in the promotor region of theCRBNgene as a predictive factor for peripheral neuropathy in the course of thalidomide‐based chemotherapy in multiple myeloma patients. British Journal of Haematology. 2019;186(5):695–705. doi: 10.1111/bjh.15972. [DOI] [PubMed] [Google Scholar]
- 29.García-Sanz R, et al. Prediction of peripheral neuropathy in multiple myeloma patients receiving bortezomib and thalidomide: a genetic study based on a single nucleotide polymorphism array. Hematol. Oncol. 2017;35:746–751. doi: 10.1002/hon.2337. [DOI] [PubMed] [Google Scholar]
- 30.Jun G, et al. Evaluating the contribution of rare variants to type 2 diabetes and related traits using pedigrees. Proc. Natl Acad. Sci. USA. 2018;115:379–384. doi: 10.1073/pnas.1705859115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nho K, et al. Association analysis of rare variants near the APOE region with CSF and neuroimaging biomarkers of Alzheimer’s disease. BMC Med. Genomics. 2017;10:29. doi: 10.1186/s12920-017-0267-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lu C, et al. Patterns and functional implications of rare germline variants across 12 cancer types. Nat. Commun. 2015;6:10086. doi: 10.1038/ncomms10086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Johnston HR, Hu Y, Cutler DJ. Population genetics identifies challenges in analyzing rare variants. Genet. Epidemiol. 2015;39:145–148. doi: 10.1002/gepi.21881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Im EH, Choi SS. Synonymous Codon Usage Controls Various Molecular Aspects. Genomics Inf. 2017;15:123–127. doi: 10.5808/GI.2017.15.4.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gingold H, Pilpel Y. Determinants of translation efficiency and accuracy. Mol. Syst. Biol. 2011;7:481. doi: 10.1038/msb.2011.14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Mittal P, Brindle J, Stephen J, Plotkin JB, Kudla G. Codon usage influences fitness through RNA toxicity. Proc. Natl Acad. Sci. USA. 2018;115:8639–8644. doi: 10.1073/pnas.1810022115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sauna ZE, Kimchi-Sarfaty C. Understanding the contribution of synonymous mutations to human disease. Nat. Rev. Genet. 2011;12:683–691. doi: 10.1038/nrg3051. [DOI] [PubMed] [Google Scholar]
- 38.Livingstone M, et al. Investigating DNA-, RNA-, and protein-based features as a means to discriminate pathogenic synonymous variants. Hum. Mutat. 2017;38:1336–1347. doi: 10.1002/humu.23283. [DOI] [PubMed] [Google Scholar]
- 39.Choi J, Baek KH. Cellular functions of stem cell factors mediated by the ubiquitin-proteasome system. Cell Mol. Life Sci. 2018;75:1947–1957. doi: 10.1007/s00018-018-2770-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Li B, Ruiz JC, Chun KT. CUL-4A is critical for early embryonic development. Mol. Cell. Biol. 2002;22:4997–5005. doi: 10.1128/mcb.22.14.4997-5005.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zhang, Y.-L. et al.
- 42.Higgins JJ, Pucilowska J, Lombardi RQ, Rooney JP. A mutation in a novel ATP-dependent Lon protease gene in a kindred with mild mental retardation. Neurology. 2004;63:1927–1931. doi: 10.1212/01.WNL.0000146196.01316.A2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kowalski TW, et al. Assembling systems biology, embryo development and teratogenesis: What do we know so far and where to go next? Reprod. Toxicol. 2019;88:67–75. doi: 10.1016/j.reprotox.2019.07.015. [DOI] [PubMed] [Google Scholar]
- 44.Jambhekar A, Dhall A, Shi Y. Roles and regulation of histone methylation in animal development. Nat. Rev. Mol. Cell Biol. 2019;20:625–641. doi: 10.1038/s41580-019-0151-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cassina M, Salviati L, Di Gianantonio E, Clementi M. Genetic susceptibility to teratogens: state of the art. Reprod. Toxicol. 2012;34:186–191. doi: 10.1016/j.reprotox.2012.05.004. [DOI] [PubMed] [Google Scholar]
- 46.Schwarz JM, Rödelsperger C, Schuelke M, Seelow D. MutationTaster evaluates disease-causing potential of sequence alterations. Nat. Methods. 2010;7:575–576. doi: 10.1038/nmeth0810-575. [DOI] [PubMed] [Google Scholar]
- 47.Choi Y, Sims GE, Murphy S, Miller JR, Chan AP. Predicting the functional effect of amino acid substitutions and indels. PLoS One. 2012;7:e46688. doi: 10.1371/journal.pone.0046688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Adzhubei Ivan, Jordan Daniel M., Sunyaev Shamil R. Predicting Functional Effect of Human Missense Mutations Using PolyPhen-2. Current Protocols in Human Genetics. 2013;76(1):7.20.1-7.20.41. doi: 10.1002/0471142905.hg0720s76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Buske OJ, Manickaraj A, Mital S, Ray PN, Brudno M. Identification of deleterious synonymous variants in human genomes. Bioinformatics. 2013;29:1843–1850. doi: 10.1093/bioinformatics/btt308. [DOI] [PubMed] [Google Scholar]
- 50.Hofacker IL. Vienna RNA secondary structure server. Nucleic Acids Res. 2003;31:3429–3431. doi: 10.1093/nar/gkg599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kozomara A, Griffiths-Jones S. miRBase: annotating high confidence microRNAs using deep sequencing data. Nucleic Acids Res. 2014;42:D68–73. doi: 10.1093/nar/gkt1181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Desmet FO, et al. Human Splicing Finder: an online bioinformatics tool to predict splicing signals. Nucleic Acids Res. 2009;37:e67. doi: 10.1093/nar/gkp215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Bendl J, et al. PredictSNP2: A Unified Platform for Accurately Evaluating SNP Effects by Exploiting the Different Characteristics of Variants in Distinct Genomic Regions. PLoS Comput. Biol. 2016;12:e1004962. doi: 10.1371/journal.pcbi.1004962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.You L, et al. APASdb: a database describing alternative poly(A) sites and selection of heterogeneous cleavage sites downstream of poly(A) signals. Nucleic Acids Res. 2015;43:D59–67. doi: 10.1093/nar/gku1076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Gao T, et al. EnhancerAtlas: a resource for enhancer annotation and analysis in 105 human cell/tissue types. Bioinformatics. 2016;32:3543–3551. doi: 10.1093/bioinformatics/btw495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Li LC, Dahiya R. MethPrimer: designing primers for methylation PCRs. Bioinformatics. 2002;18:1427–1431. doi: 10.1093/bioinformatics/18.11.1427. [DOI] [PubMed] [Google Scholar]
- 57.Barrett JC, Fry B, Maller J, Daly MJ. Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics. 2005;21:263–265. doi: 10.1093/bioinformatics/bth457. [DOI] [PubMed] [Google Scholar]
- 58.Coetzee SG, Coetzee GA, Hazelett DJ. motifbreakR: an R/Bioconductor package for predicting variant effects at transcription factor binding sites. Bioinformatics. 2015;31:3847–3849. doi: 10.1093/bioinformatics/btv470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Durinck S, Spellman PT, Birney E, Huber W. Mapping identifiers for the integration of genomic datasets with the R/Bioconductor package biomaRt. Nat. Protoc. 2009;4:1184–1191. doi: 10.1038/nprot.2009.97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wickham, H. (ed Springer-Verlag) (New York, 2016).
- 61.Yang Z. PAML 4: phylogenetic analysis by maximum likelihood. Mol. Biol. Evol. 2007;24:1586–1591. doi: 10.1093/molbev/msm088. [DOI] [PubMed] [Google Scholar]
- 62.Edgar R, Domrachev M, Lash AE. Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res. 2002;30:207–210. doi: 10.1093/nar/30.1.207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Barrett T, et al. NCBI GEO: archive for functional genomics data sets–update. Nucleic Acids Res. 2013;41:D991–995. doi: 10.1093/nar/gks1193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Carvalho BS, Irizarry RA. A framework for oligonucleotide microarray preprocessing. Bioinformatics. 2010;26:2363–2367. doi: 10.1093/bioinformatics/btq431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Gautier L, Cope L, Bolstad BM, Irizarry R. A. affy–analysis of Affymetrix GeneChip data at the probe level. Bioinformatics. 2004;20:307–315. doi: 10.1093/bioinformatics/btg405. [DOI] [PubMed] [Google Scholar]
- 66.Robinson MD, McCarthy DJ, Smyth GK. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics. 2010;26:139–140. doi: 10.1093/bioinformatics/btp616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Yu G, Wang LG, Han Y, He QY. clusterProfiler: an R package for comparing biological themes among gene clusters. Omics. 2012;16:284–287. doi: 10.1089/omi.2011.0118. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated and analyzed during the current study are not publicly available to maintain patient confidentiality. Moreover, this type of request has not been previously approved by participants nor the Human Research Committee. This data could, however, be available (anonymously) from the corresponding author on reasonable request. Gene panel sequencing files have been deposited in the repository Sequence Read Archive (SRA) of the National Center for Biotechnology Information (NCBI), under accession number SRP160424. The new variants identified in the present study were submitted to the Leiden Open Variation Database (LOVD3), under numbers #0000439088 (DDB1), #0000439108 and #0000439110 (IKZF1). Gene expression analyses were performed with data from public repository Gene Expression Omnibus (GEO) and are available under the following numbers: GSE42413, GSE61306, GSE15542, GSE63935, and GSE48472.