Abstract
BACKGROUND
Bacille Calmette-Guérin (BCG) vaccine is protective against Tuberculosis (TB) in children, but its efficacy wanes with age. Consequently, determining if BCG revaccination augments anti-TB immunity in young adults in TB endemic regions is vital.
METHODS
Two hundred healthy adults, BCG vaccinated at birth, were tested for their IFN-γ release assay (IGRA) status. Of these, 28 IGRA+ and 30 IGRA– were BCG revaccinated, and 24 IGRA+ and 23 IGRA– subjects served as unvaccinated controls. T and innate cell responses to mycobacterial antigens were analyzed by 14-color flow cytometry over 34 weeks.
RESULTS
IFN-γ and/or IL-2 Ag85A- and BCG-specific CD4+ and CD8+ T cell responses were boosted by revacciantion at 4 and 34 weeks, respectively, and were > 2-fold higher in IGRA+ compared with IGRA– vaccinees. Polyfunctional Ag85A, BCG, and mycobacterium tuberculosis (Mtb) latency Ag–specific (LTAg-specific) CD4+ T cells expressing up to 8 cytokines were also significantly enhanced in both IGRA+ and IGRA– vaccinees relative to unvaccinated controls, most markedly in IGRA+ vaccinees. A focused analysis of Th17 responses revealed expansion of Ag85A-, BCG-, and LTAg-specific total IL-17A+,IL-17F+,IL-22+, and IL-10+ CD4+ T cell effectors in both IGRA+ and IGRA– subjects. Also, innate IFN-γ+ NK/γδ/NKT cell responses were higher in both IGRA+ and IGRA– vaccinees compared with controls. This is the first evidence to our knowledge that BCG revaccination significantly boosts antimycobacterial Th1/Th17 responses in IGRA+ and IGRA– subjects.
CONCLUSION
These data show that BCG revaccination is immunogenic in IGRA– and IGRA+ subjects, implying that Mtb preinfection in IGRA+ subjects does not impact immunogenicity. This has implications for public health and vaccine development strategies.
FUNDING
This work was funded principally by DBT-NIH (BT/MB/Indo-US/HIPC/2013).
Keywords: Immunology, Infectious disease
Keywords: Adaptive immunity, Innate immunity, Tuberculosis
A detailed analysis is provided of immune subsets boosted by BCG revaccination of young adults living in a tuberculosis endemic region of India.
Introduction
Tuberculosis (TB) is the leading cause of death from a single infectious agent, Mycobacterium tuberculosis (Mtb). An estimated 10.4 million new TB cases occurred in 2017 (1). Almost a quarter of the world’s population in Asia and Africa is estimated to have latent Mtb infection (1, 2), with approximately 5%–10% of these infected subjects at a higher risk of progressing toward disease during their lifetimes (3). Bacille Calmette-Guérin (BCG) — a live attenuated strain of Mycobacterium bovis, first introduced in 1921 — is the only clinically approved TB vaccine (4). Although BCG administered at birth significantly reduces the incidence of severe miliary and meningeal TB in infants and children, it is less effective against pulmonary TB in adults, the most common form of TB disease and the major source of transmission worldwide (5–8). Meta-analysis of 14 prospective BCG efficacy studies involving 3855 participants revealed an estimated protective efficacy of 19% against Mtb infection based on IFN-γ release assay (IGRA) positivity and 58% against TB disease (9). Clearly, a more effective TB vaccine strategy is needed worldwide, and efforts to understand protective immunity against Mtb infection are a major global research priority. Here, we present a study designed to investigate the effects of BCG revaccination in boosting Mtb immunity in South Indian young adults, who are highly vulnerable to TB disease, as their immunity to Mtb is likely waning since vaccination at birth.
Many factors can account for the variable efficacy of BCG in different countries, but a consistent theme is that efficacy is suboptimal and protection wanes as children reach adolescence. Thus, the protective efficacy of BCG administered at birth rarely persists beyond 15–20 years in TB endemic regions, is highly variable in adults (6, 8), and differs considerably with geographical location and prior sensitization to Mtb or even other environmental and nontuberculous mycobacteria (NTM) (10–12). Evidence from other studies shows that BCG and NTM responses can influence each other. BCG vaccination can offer protection against NTM infection in children (13, 14). BCG administration and Mtb infection in mice and humans induces NTM cross-reactive T cells (15). In a 15-year follow-up of a randomized controlled BCG trial in South India, average protection was 32% (95% CI, 3%–52%) among people who were initially nonresponsive to NTMs detected by a tuberculin skin test (TST); by contrast, no significant protection was observed in vaccine recipients previously exposed to NTMs (16). On the other hand, there is also evidence to suggest that prior exposure to NTM can affect BCG vaccine efficacy and the results of purified protein derivative (PPD) skin test (17). Some have postulated that the lack of sustained protection after BCG vaccination may relate to the failure to establish a long-term central memory response (18). Furthermore, a reduced protective effect of BCG may occur with coinfections, especially HIV-1 infection (6, 7, 19), a known major predisposing factor for TB incidence (20–22).
Recent efforts have focused on developing improved vaccines to prevent Mtb infection, since such a vaccine could have a high population-level impact for TB control and tailored preexisting immunity may be more effective during acute infection than after persistent chronic infection (23). Current approaches under evaluation in animal models and some in humans include: (a) recombinant live attenuated vaccines with improved efficacy over BCG, such as new mycobacterial vaccine designs, as well as CMV and chimp adenovirus vectors containing Mtb antigens; (b) boosting BCG with homologous BCG or subunit vaccines such as H4:IC31; and (c) delivery of TB vaccines by aerosolization rather than by systemic processes (24–27). Revaccination with BCG in adolescence has been in routine practice in many countries throughout the world with variable benefit (28). Moreover, BCG revaccination in diverse age groups and regional TB incidence has yielded inconsistent levels of protection against TB disease (29–42).
There is renewed interest in BCG revaccination of young adults, especially in countries with high TB burden (43, 44), based on recent positive results from the Aeras C-040-404 randomized, placebo-controlled, prevention of infection TB vaccine study (45). This study was conducted in the Western Cape Province of South Africa among 990 HIV-, healthy adolescents who were BCG vaccinated at birth. Mtb-uninfected subjects were randomized to receive either placebo, H4:IC31, or BCG revaccination. When compared with placebo, neither vaccine achieved statistical significance in preventing an initial QuantiFERON-TB Gold In-Tube Test (QFT-GIT) conversion. However, analysis of the secondary efficacy endpoint indicated that BCG revaccination reduced sustained Mtb infection in young adolescents by 45.4% and that H4:IC31 reduced sustained Mtb infection by 30.5% (45). A follow-up efficacy trial is planned to confirm these findings and to examine potential biomarkers of protective immunity.
Few studies have reported detailed analysis of the immune profile alterations following BCG revaccination in humans, and among these, populations evaluated have varied by age group and status of Mtb infection. Revaccination of infants induced a significant increase in IFN-γ and IL-10 concentrations in supernatants from whole blood after stimulation by PPD for 3 days (46, 47). Others have reported that BCG revaccination of TST+ subjects on isoniazid (INH) treatment elicited long-lived memory NK and NKT-like cells besides transiently boosting BCG-specific Th1 cytokines in CD4+, CD8+, and T cell receptor (TCR) γδ cells using an intracellular cytokine staining (ICS) assay (48). In an extensive immune profiling study, we recently reported a key distinguishing feature of subjects with latent TB infection (not on INH treatment) to be the presence of circulating Mtb-specific central memory polyfunctional CD4+ T cells, that coexpressed IL-17A/IL-17F/IL-22 with IL-10; this specific Th17 subset was either lacking or was significantly reduced in the bronchoalveolar lavage of subjects with pulmonary disease or in the blood of subjects with extrapulmonary TB (49). By contrast, subjects with either pulmonary or extrapulmonary TB had expanded frequencies of proinflammatory Th17 cells that coexpressed IL-17A/IL-17F/IL-22 with IFN-γ (49). These data from our laboratory on a potential role for Mtb-specific IL-17+ and IL-10+ cells in anti-TB immunity have been utilized in a recent publication using a macaque in vivo TB challenge model, which highlighted that the key cells associated with protection against live bacterial challenge following BCG vaccination were Mtb-specific CD4+ T cells that expressed IL-17 and other IL-10–producing immune cells (50). Therefore, a major aim of our current study was to determine if BCG revaccination can boost circulating frequencies of Mtb-specific Th17 CD4+ T cells in both IGRA+ and IGRA– young adults, a highly vulnerable population, living in an HIV endemic area of South India.
Detailed immune profiling of subjects following BCG revaccination has not been reported from India. We present potentially novel data to show that BCG revaccination of young adults in India can enhance a Mtb-specific CD4+ T cell immune signature potentially associated with controlled TB infection.
Results
Mtb antigen–specific CD4+ T cells are preserved in young adults receiving BCG at birth.
Two hundred volunteers who were HIV–, TB–, hepatitis B antibody–negative, and BCG vaccinated at birth — living in Madanapalle, India — were recruited (Figure 1A). Volunteers were divided into 4 groups based on screening for IGRA status and then randomization to receive BCG revaccination at T0 (Figure 1, A and B). All volunteers subsequently received 3 doses of hepatitis B virus surface antigen vaccine at weeks 4 (T2), 10 (T4), and 30 (Figure 1, A and B). The median age of the study population was 20 years (range, 18–28 years), and 58.75% were female (Table 1 and Supplemental File 1; supplemental material available online with this article; https://doi.org/10.1172/jci.insight.130540DS1). The approximate male/female ratio in different clinical groups was: Group 1, 43:57; Group 2, 39:61; Group 3, 61:39; and Group 4, 22:78. The median IGRA levels in Groups 1 and 3 were similar (median for Group 1, 3.6; for Group 3, 2; range 0.46–10). The median IGRA values for IGRA– subjects (Groups 2 and 4) were ≤ 0.1 (range 0–0.3) (Table 1 and Supplemental File 1).
Figure 1. Overall study design.
(A) CONSORT flow diagram of participant recruitment and enrolment. (B) A diagrammatic representation of study design, including schedule of vaccination and blood draw. Group 1 and Group 2 received BCG at day 0 (T0) and then 3 doses of HBV at weeks 4 (T2), 10, and 30 (T4). Group 3 and Group 4 did not receive BCG vaccine but did receive 3 doses of HBV. Immunization time points are shown by blue arrows, and the 6 blood sampling time points (T0–T5) are indicated by red arrows for all groups.
Table 1. Clinical characteristics of study recruits.
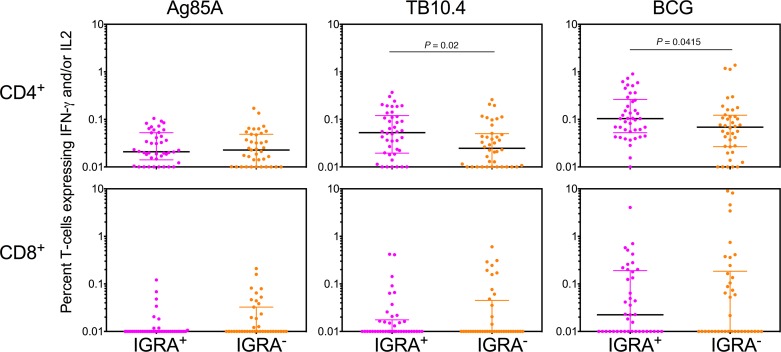
Multiparameter flow cytometry was performed on whole blood to assess the presence of T cells recognizing Mtb antigens prior to revaccination (Ag85A, TB10.4, BCG), which would reflect baseline responses maintained either by BCG vaccination at birth and/or prior exposure to cross-reactive NTM. Representative whole blood flow cytometry plots depicting the stepwise gating strategy to identify functional subsets are shown in Supplemental Figure 1 (supplemental material available online with this article; https://doi.org/10.1172/jci.insight.130540DS1). Cross-sectional analysis of baseline CD4+ T cell responses measured as frequencies of IFN-γ– and/or IL-2–expressing cells in IGRA+ versus IGRA– donors revealed that recall responses to Mtb antigens TB10.4 and Ag85A, as well as BCG (used as antigen for in vitro stimulation), were well preserved in both groups, with high basal responses, particularly to BCG antigens (Figure 2). As expected, IGRA+ subjects had significantly higher baseline CD4+ T cell responses to epitopes in BCG and TB10.4 compared with IGRA– subjects recruited to the study (Figure 2). Baseline antigen-specific CD8+ T cell frequencies were generally lower than the CD4+ T cell frequencies, and notably, the BCG-specific CD8+ T cell responses were greater than the responses to Ag85A and TB10.4 (Figure 2). No significant differences were observed between the median IGRA+ and IGRA– CD8+ T cell recall responses at baseline (Figure 2). These data provide clear evidence of baseline CD4+ T cell responses to Ag85A and TB10.4 peptides in IGRA+ and IGRA– subjects and to epitopes presented by BCG, with the magnitude of this baseline response varying significantly between donors in a group. These data indicate preservation of memory CD4+ T cells recognizing BCG and Mtb antigens in both IGRA+ and IGRA– volunteers recruited to this study, reflecting responses either induced by BCG vaccination at birth or by exposure to environmental cross-reactive mycobacteria.
Figure 2. Baseline Mtb antigen–specific T cell responses in whole blood of IGRA+ versus IGRA– subjects.
Scatter plots of Ag85A, TB10.4, and BCG reactive IFN-γ– and/or IL-2–expressing CD4+ (upper panels) and CD8+ (lower panels) T cells in whole blood are shown after background subtraction at baseline in IGRA+ versus IGRA– subjects. Number of subjects in each group range from 38–42. Significant differences between groups were determined using the Mann–Whitney U test (P < 0.05 shown).
Longitudinal analysis reveals BCG revaccination significantly enhanced Ag85A- and BCG-specific CD4+ and CD8+ T cell responses.
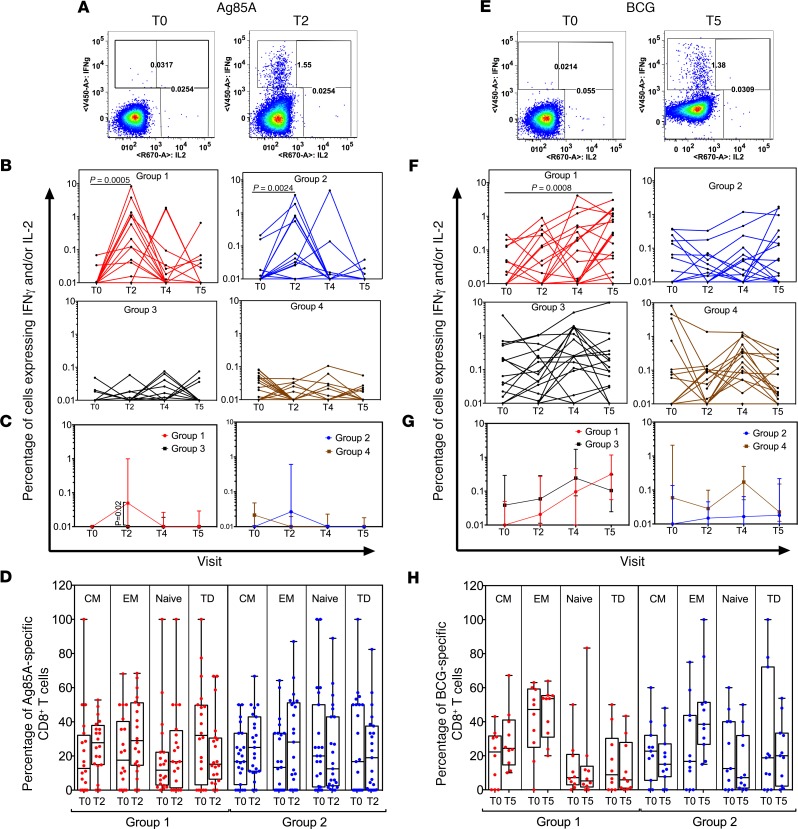
We next determined whether BCG revaccination could increase Mtb-specific CD4+ and CD8+ T cell responses in whole blood of IGRA+ and IGRA– young adults above baseline responses in a longitudinal study, with representative gating shown in Supplemental Figure 1. We demonstrate significant enhancement after vaccination, in frequencies of Ag85A-specific (Figure 3A) and BCG-specific (Figure 3E) CD4+ T cells expressing IFN-γ and/or IL-2. The peak median fold increase over baseline was 2.3-fold for Ag85A (at T2) and 5.7-fold for BCG (at T5) in Group 1 IGRA+ subjects (Figure 3, B and F). Similar enhancement was also noted for Ag85A- (Figure 3B) but not BCG-specific (Figure 3F) CD4+ T cell frequencies in Group 2 IGRA– vaccinees, possibly due to baseline responses in the IGRA– subjects being lower than the IGRA+ subjects (Figure 2). By comparison, these responses did not significantly differ over time in nonvaccinated Group 3 and Group 4 control subjects (Figure 3, B and F). Evidence of an enhanced CD4+ T cell response in vaccinees was maintained in a cross-sectional analysis of this data, despite significant intra-group variation. Thus, higher Ag85A- (Figure 3C) and BCG-specific (Figure 3G) IFN-γ+ and/or IL-2+ CD4+ T cell frequencies were noted in Group 1 compared with Group 3 subjects, with Ag85A responses peaking at T2 (Figure 3C) and BCG responses at T5 (Figure 3G).
Figure 3. Longitudinal analysis reveals that BCG revaccination significantly enhances Ag85A- and BCG-specific IFN-γ and/or IL-2 CD4+ T cell responses in whole blood.
Left panel shows ICS data for Ag85A, and right panel shows data for BCG following in vitro stimulation of whole blood from BCG-revaccinated and control subjects. Representative flow cytometry plots show IFN-γ and/or IL-2 cytokine–positive CD4+ T cells after in vitro stimulation with either Ag85A (A) or BCG (E) at T0 and T2 (A) or T0 and T5 (E). Total frequencies of Ag85A (B) and BCG-specific (F) IFN-γ and/or IL-2 cytokine–positive CD4+ T cells in all participants are shown after background subtraction. Line graphs show changes in frequencies of specific CD4+ T cells over time in BCG-revaccinated (Group 1 [IGRA+, red, n = 19] and Group 2 [IGRA–, blue, n = 18]) subjects and unvaccinated controls (Group 3 [IGRA+, black, n = 18] and Group 4 [IGRA–, brown, n = 18]). P values for longitudinal samples were calculated by comparing each time point with baseline using the Freidman test and corrected for multiple comparisons using Dunn’s test. Further, significant differences were determined using the Wilcoxon paired, 2-tailed t test before and after vaccination. Bonferroni adjusted P value threshold of 0.02 was considered statistically significant. Median frequencies of Ag85A- (C) and BCG-specific (G) IFN-γ and/or IL-2 cytokine–positive CD4+ T cells over time were compared between unvaccinated and revaccinated IGRA+ and IGRA– subjects (Group 1 versus Group 3 and Group 2 versus Group 4). Mann-Whitney U test was used to determine significant differences between groups. P < 0.05 was considered significant. Analysis of CD45RA and CD27 expression was used to determine percentage distribution of naive (CD45RA+CD27+), central memory (CM, CD45RA–CD27+), effector memory (EM, CD45RA–CD27–), and terminally differentiated T effector memory cells (TD, CD45RA+CD27–) Ag85A- (D) and BCG-specific (H) IFN-γ and/or IL-2 CD4+ T cells in BCG-revaccinated (Group 1 [IGRA+] and Group 2 [IGRA–]) subjects. Wilcoxon paired, 2-tailed t test was used to determine significant differences before and after vaccination. P < 0.05 was considered significant.
BCG revaccination substantially enhanced Ag85A- (Figure 4A) and BCG-specific (Figure 4E) IFN-γ+ and/or IL-2+ CD8+ T cell frequencies. In Group 1 IGRA+ vaccinees, median CD8+ T cell responses were enhanced above baseline (T0) by 15.1-fold for Ag85A at T2 (Figure 4B) and 14.4-fold for BCG at T5 (Figure 4F). However, in Group 2 IGRA– vaccinees, only Ag85A-specific, but not BCG-specific, CD8+ T cell responses were boosted (Figure 4, B and F). This enhancement was not observed in unvaccinated Group 3 and Group 4 control subjects (Figure 4, B and F). Cross-sectional analysis of this data showed higher CD8+ T cell responses to Ag85A at T2 but not to BCG in Group 1 versus Group 3 (Figure 4, C and G).
Figure 4. Longitudinal analysis reveals that BCG revaccination significantly enhances Ag85A- and BCG-specific IFN-γ and/or IL-2 CD8+ T cell in whole blood.
Left panel shows ICS data for Ag85A, and right panel shows data for BCG following in vitro stimulation of whole blood from BCG-revaccinated and control subjects. Representative flow cytometry plots show IFN-γ and/or IL-2 cytokine–positive CD8+ T cells after in vitro stimulation with either Ag85A (A) or BCG (E) at T0 and T2 (A) or T0 and T5 (E) Total frequencies of Ag85A- (B) and BCG-specific (F) IFN-γ and/or IL-2 cytokine–positive CD8+ T cells in all participants are shown after background subtraction. Line graphs show changes in frequencies of specific CD8+ T cells over time in BCG-revaccinated (Group 1 [IGRA+, red, n = 19] and Group 2 [IGRA–, blue, n = 18]) subjects and unvaccinated controls (Group 3 [IGRA+, black, n = 18] and Group 4 [IGRA–, brown, n = 18]). P values for longitudinal samples were calculated by comparing each time point with baseline using the Freidman test and corrected for multiple comparisons using Dunn’s test. Further, significant differences were determined using the Wilcoxon paired, 2-tailed t test before and after vaccination. Bonferroni adjusted P value threshold of 0.02 was considered statistically significant. Median frequencies of Ag85A (C) and BCG (G) specific IFN-γ and/or IL-2 cytokine–positive CD8+ T cells over time were compared between unvaccinated and revaccinated IGRA+ and IGRA– subjects (Group 1 versus Group 3 and Group 2 versus Group 4). Mann-Whitney U test was used to determine significant differences between groups. P < 0.05 was considered significant. Analysis of CD45RA and CD27 expression was used to determine percentage distribution of naive (CD45RA+CD27+), central memory (CM, CD45RA–CD27+), effector memory (EM, CD45RA–CD27–), and terminally differentiated T effector memory cells (TD, CD45RA+CD27–) Ag85A- (D) and BCG-specific (H) IFN-γ and/or IL-2 CD8+ T cells in BCG-revaccinated (Group 1 [IGRA+] and Group 2 [IGRA–]) subjects. Wilcoxon paired, 2-tailed t test was used to determine significant differences before and after vaccination. P < 0.05 was considered significant.
The memory composition of vaccine-induced T cells was analyzed using CD45RA and CD27 markers. The baseline IFN-γ– and/or IL-2–expressing CD4+ T cells specific for Ag85A and BCG comprised a mixture of naive (N), central memory (CM), effector memory (EM) and terminally differentiated EM-expressing CD45RA (TD) cells (Figure 3, D and H). BCG revaccination expanded Ag85A-specific CD4+ EM T cells in Group 1 IGRA+ subjects but not Group 2 IGRA– subjects at T2 (4 weeks) relative to T0 (Figure 3D); in contrast, there was no significant change in memory subset composition in BCG-specific cells (Figure 3H). In addition, there were no significant longitudinal changes in subset composition in the CD8+ T cell compartment (Figure 4, D and H).
The Mtb-specific vaccine–induced T cell response was probed further by analyzing CD4+ and CD8+ T cell frequencies to another immunodominant antigen — namely TB 10.4, which is a part of the well-recognized esat-6 gene family (51) — using a pool of overlapping peptides. In contrast to Ag85A- and BCG-specific responses, TB10.4 responses were not significantly induced in either IGRA+ and IGRA– vaccinees (see Supplemental Figure 2), demonstrating selectivity in the enhancement of Mtb-specific responses by revaccination.
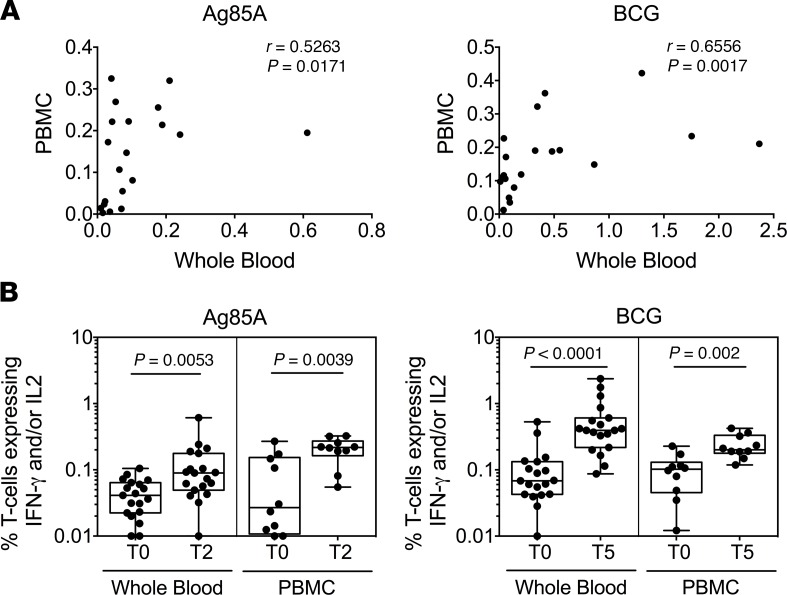
Finally, a comparison of immunostaining of matched whole blood and peripheral blood mononuclear cell (PBMC) samples in 10 subjects showed significant correlation (Figure 5A). Representative PBMC flow cytometry plots depicting gating strategy are shown in Supplemental Figure 3. Ag85A- and BCG-specific IFN-γ+ and/or IL-2+ CD4+ T cell frequencies show significantly higher frequencies over baseline T0 responses, with Ag85A at T2 and BCG at T5 in Group 1 IGRA+ in whole blood and PBMC (Figure 5B). Taken together, these data provide strong evidence of the capacity of BCG revaccination to significantly boost IFN-γ+ and/or IL-2+ CD4+ and CD8+ T cell responses in IGRA+ subjects over and above the inherently variable baseline responses. Also, the data show that BCG revaccination induced a transient increase of Ag85A responses in Group 2 IGRA– participants, but no change in BCG responses (Figure 3 and 4).
Figure 5. Comparative analysis of whole blood and PBMC ICS assay reveals similar pattern of increase in CD4+ T cell responses after BCG revaccination.
(A) IFN-γ and/or IL-2 frequencies in response to Ag85A (T2) and BCG (T5) stimulation were compared in matched whole blood and PBMC samples from IGRA+ Group 1 subjects. Spearman’s correlation coefficient (r) and significance values (P) are indicated. (B) Box-and-whisker plots show changes in IFN-γ and/or IL-2 frequencies of CD4+ T cells over time in BCG-revaccinated IGRA+ (Group 1) in response to in vitro Ag85A (left panel) or BCG (right panel) restimulation in whole blood (n = 19) and PBMC (n = 10). Significant differences before and after vaccination were determined using the Wilcoxon paired, 2-tailed t test. P < 0.05 was considered significant.
Longitudinal analysis reveals BCG revaccination to significantly enhance Ag85A- and BCG-specific CD4+ and CD8+ T cell polyfunctional response in both IGRA+ and IGRA– subjects.
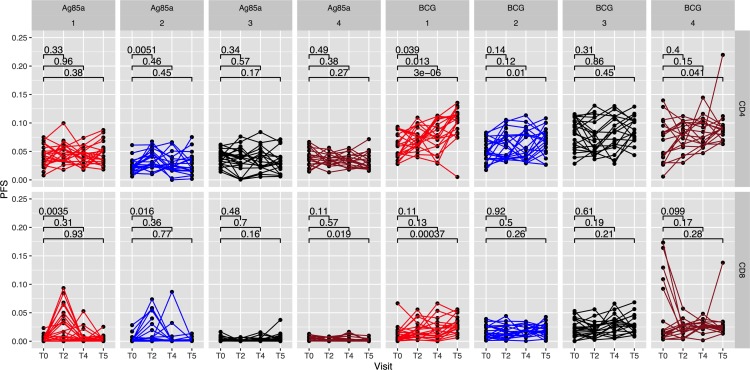
To examine coexpression of effector cytokines, we used COMbinatorial Polyfunctionality Analysis of Antigen-Specific T cell Subsets (COMPASS), as in our previous study (49), to enumerate antigen-specific polyfunctional responses of total CD4+ and CD8+ T cells in whole blood samples by analyzing all 64 possible combinations of IFN-γ, IL-2, TNF-α, IL-17A, and MIP-1β. Polyfunctional responses to Ag85A and BCG stimulation were determined over time and summarized through the COMPASS polyfunctionality score (PFS) (Figure 6). Differences in PFS between groups were estimated through a linear model fit to the PFS for each antigen (see Methods). After multiple testing adjustments, significant increase in PFS compared with baseline were observed in Group 1 IGRA+ and Group 2 IGRA– CD4+ T cell responses to BCG epitopes at T5 and in Group 2 Ag85A-specific CD4+ T cells at T2. Polyfunctional CD8+ T cell responses were significantly enhanced over baseline in Group 1 in response to both Ag85A at T2 and BCG epitopes at T5, and in Group 2 in response to Ag85A at T2. However, no increase in CD4+ and CD8+ T cell PFS was noted in subjects from unvaccinated controls in Group 3 (IGRA+) and Group 4 (IGRA–), relative to baseline responses at T0. The CD4+ T cell polyfunctionality response to BCG was more prominent than the CD8+ T cell response, whereas the Ag85A-specific CD8+ polyfunctional response was the dominant one compared with its CD4+ response, as reflected in the P values between T0 versus T2 for Ag85A and T0 versus T5 for BCG in both Groups 1 and 2 (Figure 6).
Figure 6. COMPASS analysis reveals enhanced polyfunctionality scores in CD4+ and CD8+ T cell responses in whole blood to Ag85A and BCG stimulation following BCG revaccination.
COMPASS polyfunctionality scores (PFS) for CD4+ and CD8+ T cell subsets were determined for each group and for each antigen stimulation at T0 (baseline), T2 (4 weeks), T4 (10 weeks), and T5 (34 weeks) after BCG revaccination. Pairwise differences between time points were based on a group-wise linear model fit to the PFS (null: β[T2/T5]-βT0 = 0, 2-sided test). Polyfunctionality scores were calculated for (Group 1, n = 21; Group 2, n = 20; Group 3, n = 18; Group 4, n = 18). Statistical analysis was performed using paired Wilcoxon test compared with baseline.
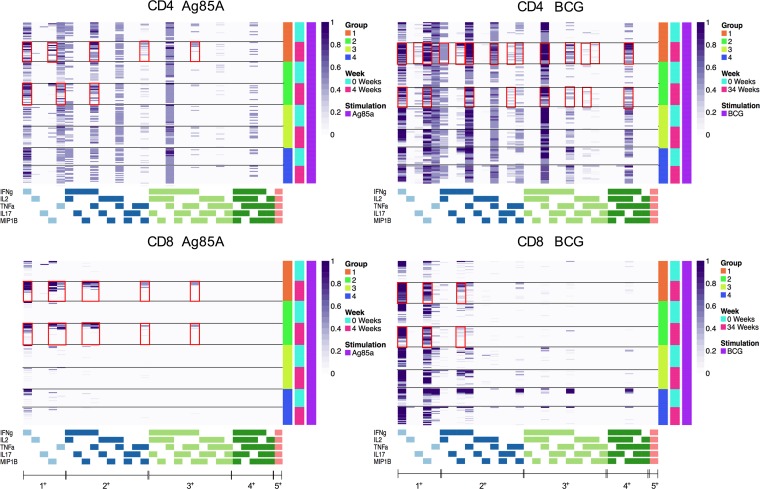
Heatmaps were constructed to identify the specific combinations of immune subsets that were expanded by BCG revaccination in Groups 1 and 2. Data from all subjects in the 4 clinical groups were analyzed for CD4+ (Figure 7, upper panel) and CD8+ (Figure 7, lower panel) polyfunctional T cell responses at baseline and at T2 and T5 after BCG vaccination following Ag85A and BCG stimulation, respectively. The Ag85A-specific CD4+ T cell response was more marked in IGRA+ Group 1 vaccinees, with enhanced expression of 1+ (IFN-γ, MIP-1β), 2+ (TNF-α/MIP-1β, IFN-γ/MIP-1β), and 3+ (IFN-γ/TNF-α/MIP-1β) subsets compared with baseline or Group 3 unvaccinated controls. The data also highlight that the CD4+ T cell responses induced by BCG antigens were more polyfunctional compared with those induced by Ag85A, with higher frequencies of 3+ and 4+ subsets producing different combinations of IFN-γ, IL-2, IL-17, TNF-α, and MIP-1β, in addition to 1+ and 2+ subsets. Longitudinal analysis of polyfunctional BCG-specific CD4+ T cells shows Group 1 and Group 2 vaccinees to have higher probabilities of the 4+ (IFN-γ/IL-2/TNF-α/MIP-1β), 3+ (IFN-γ/IL-2/TNF-α, IFN-γ/IL-2/MIP-1β), and 2+ (IFN-γ/IL-2) subsets compared with baseline (Figure 7, upper panel). Among CD8+ T cells, BCG revaccination induced a robust increase in 3+ subset–expressing 3 cytokines (IFN-γ/TNF-α/MIP-1β), as well as 2+ (combinations of IFN-γ, TNF-α, MIP-1β) and 1+ (IFN-γ, TNF-α, MIP-1β) subsets in IGRA+ Group 1 and IGRA– Group 2 subjects versus unvaccinated controls (Group 3 and Group 4) in response to Ag85A stimulation. BCG-specific CD8+ responses (1+ and 2+ subsets) were high for Group 1 and Group 2 vaccinees compared with baseline (Figure 7, lower panel).
Figure 7. COMPASS heatmaps showing different polyfunctional CD4+ and CD8+ T cell subsets after BCG vaccination.
Stacked COMPASS heatmaps displaying CD4+ (upper panel) and CD8+ (lower panel) whole blood T cell responses to Ag85A (left panel) and BCG (right panel) in BCG revaccinees (Group 1 [IGRA+, orange] and Group 2 [IGRA–, bright green]) versus unvaccinated controls (Group 3 [IGRA+, light green] and Group 4 [IGRA–,blue]). In the heatmap, columns correspond to the different disjoint cell subsets in which responses were detected and are color-coded in the x-axis legend by the cytokines they express (white, “off”; shaded, “on”; grouped by color, “degree of functionality”) and are displayed in order of increasing functionality from left to right (sky blue to peach). For example, the first column represents CD4+ T cells that produce IFN-γ but none of the other functions. Rows represent study subjects (Group 1, n = 21; Group 2, n = 20; Group 3, n = 18; Group 4 n = 18), which are ordered by the group they belong to and the time point (0 [turquoise] and 4 [pink] weeks for Ag85A and 0 [turquoise] and 34 [pink] weeks for BCG) as shown in the legend at the right. Each cell of the heatmap shows the probability estimated by COMPASS that the observed response is antigen specific in the corresponding subject (row) and cell subset (column), where the probability is color-coded from white (probability, 0) to purple (probability, 1). A probability of 0 indicates certainty that the observed response is background, while a probability of 1 indicates certainty that the observed response is antigen specific. Horizontal lines were added to separate the time points, and red boxes were inserted to highlight the subsets of interest.
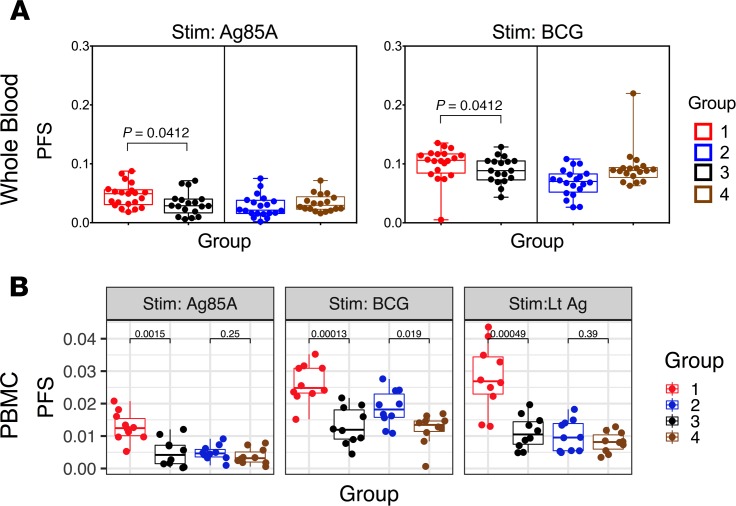
The above whole blood data were extended using PBMC staining with a panel measuring 8 effector cytokines (IFN-γ, IL-2, TNF-α, MIP-1β, IL-17A, IL-17F, IL-22, and IL-10; Supplemental Table 1). COMPASS analysis of PBMC data confirms the enhanced Ag85A and BCG CD4+ PFS noted in whole blood and extends the data to include latency antigens (Figure 8). A cross-sectional analysis of whole blood data shows significantly higher Ag85A and BCG PFS in Group 1 versus Group 3 (Figure 8A). At the PBMC level, these differences are confirmed (Figure 8B), but in addition, we note higher PFS to BCG but not Ag85A in Group 2 versus Group 4 subjects. We further demonstrate, through analysis of PBMC, that Group 1 IGRA+ vaccinees had significantly higher latency Ag (LTAg) PFS compared with unvaccinated controls (Figure 8B). These data collectively confirm that BCG revaccination has the potential to boost Mtb-specific polyfunctional CD4+ and CD8+ T cells, in both IGRA+ and IGRA– young adults, with the boosting effect being more consistent across all antigens tested in IGRA+ subjects.
Figure 8. Polyfunctional CD4+ T cell responses induced upon BCG revaccination are comparable in whole blood and PBMC of BCG-revaccinated IGRA+ subjects.
(A) Polyfunctionality scores at T5 were calculated for CD4+ T cells after restimulation of whole blood (Group 1, n = 21; Group 2, n = 20; Group 3, n = 18; Group 4 n = 18) with Ag85A or BCG, taking into account T cells positive for IL-2, IFN-γ, TNF-α, IL-17A, or MIP-1β. (B) Similarly, polyfunctionality scores at T5 were calculated for CD4+ T cells after restimulation of PBMCs (n = 10) with Ag85A, BCG, or LTAg, taking into account T cells positive for IFN-γ, IL-2, TNF-α, IL-17A, IL-17F, IL-22, IL-10, or MIP-1β. Box-and-whisker plots show comparison of COMPASS polyfunctionality scores (PFS) between revaccinated subjects (Groups 1 and 2) and unvaccinated subjects (Groups 3 and 4) in both whole blood and PBMCs. Statistical significance of differences between groups was determined by Mann-Whitney U test. P < 0.05 was considered significant.
BCG revaccination enhances Mtb-specific Th17 responses.
Our previous studies highlight a key potential role of CD4+ Th17 cells in Mtb infection, with functional differences noted between subjects with latent TB and TB disease (49). Thus, Mtb latency antigen-specific IL-10+ regulatory Th17 cells expressing IL-17A, IL-17F, or IL-22 were significantly higher in subjects with latent TB compared with those with active TB disease. By contrast, subjects with TB disease had significantly higher numbers of Mtb-specific IL-17A, IL-17F, or IL-22 cells that coexpressed IFN-γ but not IL-10, indicating a shift to Mtb-specific Th17 proinflammatory CD4+ T cells during disease, with this shift being best revealed by CD4+ T cells specific for the DosR latency antigens, rather than the commonly studied secretory mycobacterial antigens (49). Therefore, we sought to determine if BCG revaccination could boost circulating frequencies of Mtb-specific IL17+ and IL-10+ total and double-positive Th17 cells in both IGRA+ and IGRA– vaccinees, comparing PBMC responses specific for Ag85A, BCG, and latency antigens.
First, we show, in Figure 9 (upper panels), representative dot plots of CD4+ T cells expressing Th17 cytokines at 34 weeks (T5) after vaccination in BCG-stimulated PBMCs from a Group 1 and Group 3 subject (see Supplemental Figure 4 for immunostaining of unstimulated control sample). Figure 9 (lower panels) summarizes total frequencies of effector CD4+ T cells that express IL-17A, IL-17F, IL-22, IFN-γ, and IL-10 and highlights all these cytokine-positive cells to be significantly higher in IGRA+ vaccinees (Group 1) compared with unvaccinated IGRA+ controls (Group 3), especially to Ag85A and BCG stimulation. LTAg responses were similar but not significantly different for IL-17A (Figure 9, lower panels). We also record significantly higher frequencies of total CD4+ T cells expressing IL-17A, IL-17F, IL-22, and IL-10 in IGRA– Group 2 vaccinees compared with Group 4 unvaccinated IGRA– controls (Figure 9, lower panels). These data provide strong evidence that BCG revaccination has the potential to boost Mtb-specific Th17 responses in both IGRA+ and IGRA– subjects.
Figure 9. BCG revaccination significantly induces Mtb-specific cytokines in PBMC.
Representative flow cytometry plots (upper panel) show total IFN-γ, IL-17A, IL-17F, IL-22,IL-2, and IL-10 cytokine–positive CD4+ T cells after in vitro stimulation with BCG at T5 in Group 1 versus Group 3 subjects. The flow cytometry plots for the same donors from Groups 1 and 3 for BCG stimulation are shown again in Supplemental Figures 4 and 5. Mtb-specific T cell responses in PBMCs from BCG-revaccinated IGRA+ and IGRA– subjects (Groups 1 and 2, n = 10) were compared with unvaccinated control IGRA+ and IGRA– subjects (Groups 3 and 4, n = 10) in a standard ICS assay (lower panels). PBMCs were stimulated with Ag85A or BCG or a pool of LTAg (Rv1733c, Rv1737c, Rv2029, and Rv2628). CD3+CD4+ T cells were analyzed for intracellular expression of indicated cytokines. Scatter plots show median (range) percentages of total IFN-γ+, IL-17A+, IL-17F+, IL-22+, and IL-10+ CD4+ T cells. Unadjusted P values were calculated with the Mann-Whitney U test, comparing frequencies of cytokine-positive cells between the 2 groups. To correct for multiple testing (Bonferroni method), P values below 0.025 were considered statistically significant.
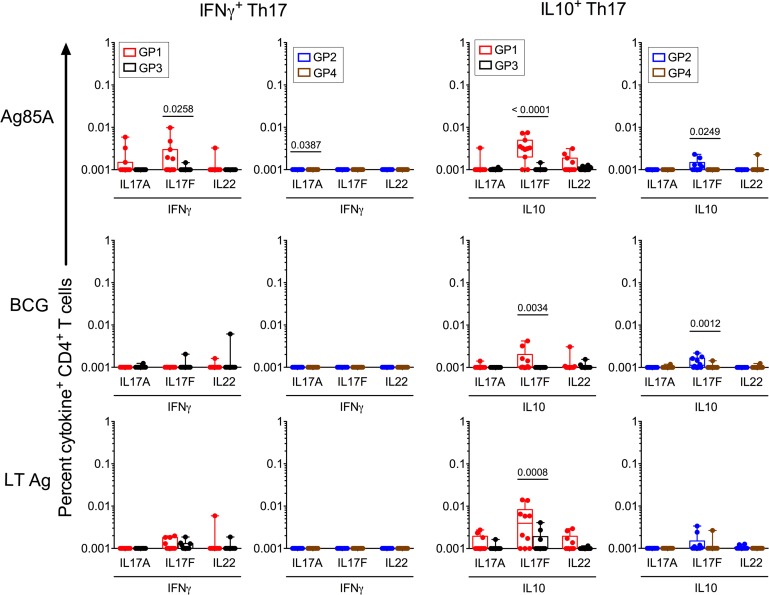
We next determined the frequencies of double-positive–specific CD4+ T cells that coexpressed IL-17A, IL-17F, or IL-22 with either IFN-γ (proinflammatory Th17) or IL-10 (regulatory Th17); representative gating data shown in Supplemental Figures 5 and 6 highlight these double-positive cells to be a minor subset, confirmed by backgating to be viable cells. Figure 10 shows significantly higher frequencies of IL-17F+IL-10+ cells specific for Ag85A and BCG in both Group 1 IGRA+ and Group 2 IGRA– vaccinees relative to unvaccinated controls, with a higher proportion of responders in Group 1 (50%–80%) and Group 2 (20%–40%) compared with unvaccinated Group 3 (10%) and Group 4 (10%), respectively. LTAg-specific IL-17F/IL-10 was also higher than unvaccinated controls but only in Group 1 IGRA+ vaccinees. With the exception of Ag85A in Group 1 vaccinees, revaccination did not enhance Th17 proinflammatory cells that coexpressed IL-17A, IL-17F, or IL-22 with IFN-γ (Figure 10). Thus, the extent to which specific cells were upregulated differed by antigen specificity and was evident in some but not all vaccinees. These data demonstrate that BCG revaccination has the potential to enhance a minor Mtb-specific antiinflammatory, regulatory Th17 subset in both IGRA+ and IGRA– subjects.
Figure 10. BCG revaccination significantly induces Mtb-specific regulatory IL-10+ Th17 responses.
CD4+ T cell subsets expressing IL-17A, IL-17F, or IL-22 (Th17) — either in combination with IFN-γ or IL-10 from BCG-revaccinated IGRA+ and IGRA– subjects (Groups 1 and 2, n = 10) — were compared with unvaccinated control IGRA+ and IGRA– subjects (Groups 3 and 4, n = 10) at T5 to Ag85A, BCG, and LTAg stimulation. For this analysis, we calculated cytokine-positive cells based on the gating shown in Supplemental Figure 6. Statistical analysis was performed using a 1-way ANOVA with Bonferroni post hoc test defining differences as significant (*P < 0.05).
BCG revaccination induces innate specific cells.
A previous study (48) had reported that BCG revaccination in adults transiently induces BCG-reactive γδ+ and CD56+ NKT-like, as well as CD56dim and CD56hi NK cells, that express IFN-γ in IGRA+ subjects. We therefore evaluated whether BCG revaccination modulated the frequencies and function of innate lymphocyte populations. A representative example of the flow cytometric gating strategy to analyze these cells is shown in Supplemental Figure 7. Revaccination induced significant expansion of Ag85A reactive innate effectors (γδ, CD56hi NK cells) above baseline T0 responses in both Group 1 IGRA+ and Group 2 IGRA– vaccinees and NKT cells in Group 1 only but not Group 3 and Group 4 unvaccinated controls (Figure 11); the boosting effect ranged from 2- to 10-fold, depending on the innate effector subset, and was more consistent (in terms of proportion of vaccinees responding) in Group 1 IGRA+ subjects. BCG reactive innate effectors (γδ, CD56dim NK, CD56hi NK, and NKT cells) were significantly boosted at T5 (2- to 10-fold) above baseline T0 only in Group 1 IGRA+ and not in Group 2 IGRA– vaccinees. Interestingly, the pattern of induction of these innate subsets was similar to the induced adaptive response (Figures 3 and 4). Thus, IFN-γ+ innate lymphocytes induced by Ag85A restimulation peaked at T2 (4 weeks) after revaccination, whereas those specific to BCG epitopes were observed later at T5 (34 weeks) after revaccination. These data highlight the potential of BCG to boost innate Mtb-reactive cells in both Group 1 and Group 2 IGRA+ and IGRA– subjects.
Figure 11. BCG revaccination induces innate effector response in whole blood.
Line graphs show the frequencies of IFN-γ–expressing γδ T, NKT, CD56hi NK, and CD56dim NK cells in BCG-revaccinated IGRA+ and IGRA– subjects (Group 1, n = 20; Group 2, n = 18) versus unvaccinated control IGRA+ and IGRA– subjects (Group 3, n = 19; Group 4, n = 18) in response to Ag85A and BCG stimulation at baseline and at T2 and T5 post-BCG revaccination respectively. Significance between longitudinal samples in a group was calculated using Wilcoxon matched pairs test. P < 0.05 was considered significant.
Discussion
We provide the first detailed evidence to our knowledge of the impact of BCG revaccination on Mtb-specific T cell immunity in young adults from a TB endemic region of India. Our work confirms — and extends, in particular — the report of Suliman et al. on immune stimulating effects of BCG revaccination of young adults with latent TB living in South Africa (48). Potentially novel aspects of our work include: (a) detailed investigation of polyfunctional Mtb-specific T cells including up to 256 immune subsets; (b) focused analysis of Mtb-specific proinflammatory as well as regulatory Th17 responses, the latter of which are highlighted to be of increasing importance in anti-TB immunity, based on our work and that of others (49, 50); and (c) comparative longitudinal analysis of vaccine-induced responses in both IGRA+ versus IGRA– subjects, in the absence of anti-TB treatment.
The strong evidence we provide that BCG revaccination can boost polyfunctional Th1/Th17 and innate effectors in subjects living with latent TB in the absence of INH treatment distinguishes our study from that of the Suliman et al. study (48), where TST+ subjects were randomized into 2 arms: 1 receiving BCG revaccination after INH and the other receiving BCG revaccination first and then INH. In the Suliman et al. study, immune responses did not differ between these treatment groups, implying that pretreatment with INH does not interfere with BCG revaccination potency. In this paper, we asked a different question: can BCG revaccination boost anti-TB immunity in the absence of INH treatment altogether? The fact that we demonstrate this to be the case is important, as current thinking proposes that underlying infection with Mtb or cross-reactive NTM responses (10–13, 17) can potentially blunt responses to BCG revaccination. Hence, most BCG revaccination studies have focused on subjects without Mtb infection (IGRA–) or latent TB subjects (IGRA+) after INH. IGRA+ individuals live with a considerably higher risk of succumbing to active TB (52). The results from our study show that Ag85A-, BCG-, and LTAg-specific CD4+ and CD8+ T cell responses are boosted to a greater extent in IGRA+ rather than IGRA– individuals. Thus, in India BCG revaccination might be more beneficial to the more susceptible IGRA+ population. This has implications for future BCG revaccination/other TB vaccination strategies.
All the antigens tested in this study (Ag85A, TB10.4, and DosR regulon encoded latency antigens) are encoded by BCG (53–56). Our own and previous studies have shown that BCG vaccination does not lead to generation of potent immune responses against DosR-encoded latency antigens (49, 55, 56). However, we show in this paper that DosR-specific T cell responses can be boosted by BCG revaccination of young Indian adults. Ag85A and TB10.4 are both vaccine candidates and have shown protection against Mtb infection in animal models (57–60). In addition, Ag85A was found to be well tolerated in BCG vaccinated infants and significantly boosted Ag85A-specific Th1 and Th17 responses (61). H4:IC31, another candidate vaccine comprising Ag85B and TB10.4, was tested in adolescents and found to be immunogenic and capable of reducing the rate of sustained QuantiFERON (QFT) conversion (45). DosR-encoded latency antigens, which we and others show to distinguish subjects with latent and active TB individuals (49, 62), were also shown to have protective efficacy in NHP, most likely via induction of CD4+ as well as CD8+ T cells. In this context, whether BCG revaccination of young IGRA+ adults can induce protective immunity and reversion of IGRA+ untreated subjects to an IGRA– state remains to be tested.
The importance of probing adaptive T cell responses to Mtb antigens is based on significant data from mouse KO models demonstrating the dominant role of CD4+, and to a lesser extent CD8+ Mtb-specific T cells in protection against Mtb infection following immunization with BCG (63, 64). While Suliman et al. (48) reported peak responses at 3 weeks after revaccination, we report peak BCG responses to occur later, at 34 weeks after revaccination. This disparity may reflect differences in the BCG strains used. Further, the TST+ subjects recruited to the Suliman et al. study received INH, whereas IGRA+ subjects recruited to our study remained treatment naive. We also show significantly enhanced Ag85A responses in both IGRA+ and IGRA– revaccinated subjects that transiently peaked at T2 (4 weeks) after revaccination, consistent with the observation that Ag85A responses following BCG vaccination in infants (61) and in murine models are transient based on low antigen load (65). Comparative analysis of the boosting effect in IGRA+ versus IGRA– subjects revealed a more marked effect in IGRA+ subjects, who had a higher baseline-specific T cell response; consequently, the more efficacious boosting effect of BCG revaccination may be due to expansion of preexisting memory T cells. Beyond analysis of T cell responses and in keeping with the findings of Suliman et al. (48), our study also shows that NK, NKT, and γδ T cell BCG–specific responses are boosted in IGRA+ vaccinees. These data provide strong evidence that BCG revaccination has the potential to boost both the innate and adaptive arms of the immune system in adults in the time frame of 9 months/34 weeks after vaccination.
Beyond IFN-γ– and/or IL-2–specific T cell responses, we used the statistically robust COMPASS analysis to probe polyfunctional T cell responses encompassing 8 effectors representing a total of 256 immune subsets (66). Polyfunctional Mtb-specific CD4+ T cells simultaneously expressing IFN-γ, IL-2, and TNF-α in blood are implicated in anti-TB immunity (67, 68) and are shown to be critical in modulating vaccine-mediated immunity in murine rechallenge studies (69–72). Further, we previously showed Mtb-specific polyfunctional cells expressing up to 4 cytokines to be associated with controlled latent infection (49). Herein, we report that BCG revaccination significantly boosts circulating frequencies of CD4+ and CD8+ polyfunctional T cells expressing up to 4 effectors (IFN-γ, IL-2, TNF-α, IL-17A) specific to Ag85A and BCG, in both IGRA+ and IGRA– subjects. In addition, we conducted a more focused analysis of Th17 cells, which have emerged as potential correlates of protective immunity in human studies (49) and in vaccine-induced protection against TB (50, 73–76). Th17 subsets that express proinflammatory cytokines (IL-17A, IL-17F, IL-22 in combination with IFN-γ) are associated with pathogenesis of several chronic disease conditions; in contrast, subsets that express Th17 cytokines in combination with IL-10 can protect against disease pathology and are therefore implicated to be protective in chronic conditions (77). In this manuscript, we demonstrate that BCG revaccination significantly boosted a minor population of antiinflammatory CD4+ T cells specific for Ag85A, BCG, and LTAg that coexpressed IL-17F and IL-10 in a proportion of IGRA+ and IGRA– vaccinees, while the boosting effect of proinflammatory cells expressing IFN-γ and IL-17 was weaker and only noted to Ag85A stimulation. These data call for further studies to probe the potential efficacy of BCG revaccination in regulating Th17 responses.
There is evidence for at least 3 potential mechanisms for the immunogenic function of BCG. First, BCG revaccination may expand preexisting memory T cells. Continuous T cell stimulation by underlying infection is known to maintain a higher basal Mtb-specific adaptive T cell response in IGRA+ compared with IGRA– subjects, and BCG revaccination may induce the proliferation of these preexisting cells. Secondly, BCG may boost T cell responses through immunomodulatory antiinflammatory effects, especially through IL-10 upregulation (78). This is consistent with our observation that BCG revaccination can induce total IL-10 Mtb-specific CD4+ T cells and is supported by data on mucosal BCG vaccination in mice, which induces airway CD103−CD69+CD4+ memory T cells, likely to be tissue resident memory (TRM) cells, that represent a heterogeneous population composed of Foxp3+ or T-bet+ T cell subsets, producing IL-10 in addition to Th1 response (79). Thirdly, BCG may regulate innate immune cells in a pathogen-specific manner and thereby provide resistance to secondary infections through “trained immunity” through potential epigenetic changes (80–82). Suliman et al. reported that BCG-reactive CD3+CD56+ NKT-like cells, as well as CD56hiCD16lo and CD56dimCD16+ NK cells, persist for up to 1 year after BCG revaccination, suggesting memory function akin to conventional T cells (48). We, too, report an increase of CD3+CD56+ NKT and CD56hi and CD56hi NK cells in BCG-revaccinated IGRA+ subjects in response to Ag85A and BCG stimulation at 4 and 34 weeks after revaccination, confirming a further mechanism of the beneficial effects of BCG revaccination through potential regulation of innate immunity.
The importance of the immunostimulatory effects of BCG revaccination has to be placed in the broader context of immune components that may protect against Mtb infection. This has fundamental challenges because dominant immune responses elicited by vaccines in blood may not predict necessary effector responses in lung tissue following Mtb exposure by inhalation. Murine studies demonstrate that Mtb infects phagocytes with diverse phenotypes and that infected myeloid DCs migrate from lungs to local lymph nodes early in infection, where T cell activation can occur (83). Thus, a desirable vaccine might elicit innate immune activation of local myeloid cells that can kill the bacteria in the lung, as well as T cells that can rapidly enter the lung parenchyma, recognize infected cells, and destroy them (84, 85). This calls for further studies on key immune innate and adaptive phenotypes after immunization, including specific T cells that are imprinted to traffic to lung tissue. Our data on the boosting effects of BCG in a TB endemic region of South India, together with the reported efficacy of BCG revaccination in prevention of infection in South Africa (45), provide a rational basis for further mechanistic analysis of the immunostimulatory effects of BCG in divergent populations based on geography and NTM exposure.
Methods
Study population
A total of 200 young adults (age 18–28 years) — mostly nursing college students from Madanapalle, India, a TB endemic area — were prospectively recruited between January 2014 and December 2015 for the vaccination study, primarily by college-wide informational seminars. All participants were confirmed as HIV-1 seronegative and BCG vaccinated at birth and had a visible BCG scar. Relevant clinical information of study participants was documented in a proforma and is summarized in Table 1 and Supplemental File 1. Blood from study participants was screened for Mtb infection by the standard QFT TB Gold In-tube test (Qiagen) performed at the blood collection center in Madanapalle, India, and was classified as either IGRA+ or IGRA– (86). IGRA+ subjects were enrolled only if they had no known TB disease and had not received preventive/curative therapy for TB in the past. Two hundred subjects were enrolled and then divided into 4 clinical groups based on their IGRA result and randomization to receive BCG (Figure 1A).
Vaccination
BCG vaccine (TUBERVAC, Russian BCG strain manufactured at Serum Institute of India, Pune, India), used widely in the Indian national immunization program, was administered intradermally at day 0 at an adult dose of 2 × 105 to 8 × 105 CFU in participants from group 1 (IGRA+, n = 28) and group 2 (IGRA–, n = 30). Group 3 (IGRA+ n = 24) and Group 4 (IGRA– n = 23) subjects were not BCG revaccinated and served as controls for Groups 1 and 2, respectively. As greater than 90% of the volunteers recruited were nursing students, hepatitis B vaccination (HBV) was given to fulfill their immunization requirements as health care workers, as they are at high risk for Hepatitis B; additionally, this vaccination enhanced compliance to our longitudinal BCG revaccination study protocol. All participants (irrespective of whether they received BCG or not) therefore received HBV at 4 (T2), 10 (T4), and 30 weeks after BCG vaccination. Blood was collected from participants at days 0 (T0) and 3, and at weeks 4 (T2), 5 (T3), 10 (T4) and 34 (T5) after BCG vaccination (Figure 1, A and B). Some vaccinees, after BCG vaccination, reported minor side effects, which included itching, rash, or pain at the site of vaccination; mild fever; cough; and headache (Supplemental File 1). No serious side effects were reported and none of the participants become active TB+ during the entire duration of study.
Antigen and antibody reagents
Peptide pools that spanned the amino acid sequence of mycobacterial proteins Ag85A, TB10.4, and hepatitis B surface antigen were constructed. Each peptide was 15 amino acids and overlapped by 12 amino acids with the next sequential peptide (BIoSynthesis) and was 1 μg/mL as previously described (87). Four DosR regulon–encoded latency antigens (Rv1733c, Rv1737c, Rv2029, and Rv2628) were synthesized as recombinant proteins at the Department of Infectious Diseases, Leiden University Medical Center, The Netherlands. BCG Vaccine (TUBERVAC) was reconstituted in RPMI 1640 (final concentration 1.2–33.0 x 106 CFU/mL blood). All ICS assay stimulations were performed in the presence of anti-CD28/CD49d costimulatory antibodies (at 1 μg/mL, BD Biosciences). Fluorochrome conjugated monoclonal antibodies to cell surface, and intracellular markers used in flow cytometry assays are listed in Supplemental Table 1. Antibodies were sourced from BD Biosciences, BioLegend, and eBioscience (Supplemental Table 1, A and B).
PBMC isolation.
Anticoagulated blood (16 mL) was collected in ACD tubes (BD Biosciences), and PBMCs were isolated using ACCUSPIN (Sigma-Aldrich) tubes by density centrifugation following manufacturer’s instructions. Blood was diluted 2-fold with PBS (Thermo Fisher Scientific) + 2% FBS (Thermo Fisher Scientific), pipetted into ACCUSPIN tubes prefilled with Histopaque 1077, and centrifuged at 1000 g for 15 minutes at room temperature without deceleration. PBMCs from the buffy coat were washed twice with PBS + 2% FBS, then resuspended at 5 × 106 cells/mL in cryopreservation medium (90% FBS and 10% DMSO) and incubated overnight at –80°C (in Mr. Frosty freezing container; Nalgene) and were stored in liquid nitrogen until further analyses.
ICS assay and multiparameter flow cytometry
Whole blood.
Heparinized whole blood was collected from participants and processed within 30–45 minutes of phlebotomy, as previously described (88). Briefly, 400 μL of blood was pipetted into Sarstedt tubes and stimulated with peptide pools (1 μg/mL per peptide) or with BCG (2.4 – 60 × 104 CFU/mL) together with anti-CD28/CD49d costimulatory mAbs at 0.5 μg/mL. Phytohemagglutinin (PHA) (Remel, Thermo Fisher Scientific) was used as a positive mitogen control and culture medium, with anti-CD28/CD49d used as unstimulated negative control. Blood was incubated at 37°C for a total of 12 hours, and Brefeldin A (Sigma-Aldrich) — at a concentration of 10 μg/mL — was added in the final 5 hours of stimulation. After stimulation, blood was treated with 2 μM EDTA (MilliporeSigma), RBCs were lysed with 4.5 mL 1× FACS Lysing solution (BD Biosciences), and fixed cells were transferred to liquid nitrogen in freezing medium containing 10% DMSO (MilliporeSigma), 40% FCS (Thermo Fisher Scientific), and 50% RPMI 1640 (Thermo Fisher Scientific). For staining, cryopreserved whole-blood samples were thawed in a water bath at 37°C for 2 minutes. Thawed cells were transferred to labeled tubes containing 2 mL of PBS and were centrifuged at 800 g for 5 minutes. Cells were then stained with a 50 μL cocktail containing cell surface antibodies for 30 minutes at room temperature in the dark. Next, cells were washed with PBS, permeabilized with 200 μL 1× Perm/Wash solution (BD Biosciences), and incubated at room temperature for 20 minutes. Pelleted cells were immediately stained with a 50 μL cocktail containing antibodies against intracellular markers for 30 minutes at room temperature. Cells were washed and resuspended in 100 μL of 1% paraformaldehyde (Electron Microscopy Sciences) for flow cytometry analysis.
PBMC.
ICS assay with cryopreserved, antigen-stimulated PBMCs was performed as previously described (89). Cryopreserved PBMC samples were rapidly thawed in a 37°C water bath, transferred to 15-mL tubes containing about 3 mL PBS and centrifuged at 800 g for 5 minutes at room temperature. One million cells resuspended in 200 μL culture medium (RPMI-1640 [Invitrogen] supplemented with 10% FCS [Thermo Fisher Scientific], 100 U/mL penicillin, and 100 μg/mL streptomycin, [both MilliporeSigma]) were seeded per well in 96-well round-bottom plates (Costar) and stimulated with either Ag85A peptide pools (1 μg/mL) or recombinant latency proteins (10 μg/mL), or BCG (2.4 × 104 to 60 × 104 CFU/mL) or PHA (1 μg/mL) at 37°C and 5% CO2. After 4 hours of stimulation, brefeldin A (10 μg/mL) was added. On the next day, PBMC were washed after incubation with EDTA and first stained with 5 μL Live/Dead Aqua (Invitrogen) followed by a 50 μL cell surface staining cocktail for 30 minutes at room temperature in the dark. Cells were then fixed for 20 minutes with 100 μL 1× FACS lysis buffer and permeabilized with 200 μL 1× BD Perm/Wash buffer for 20 minutes. PBMCs were washed and incubated for 30 minutes at room temperature in the dark with a 50 μL intracellular staining cocktail. Finally, cells were washed with 150 μL Perm/Wash buffer and resuspended in about 100μL of 1% paraformaldehyde for flow cytometry analysis.
Flow cytometry analysis.
Samples were acquired on BD FACSAria Fusion flow cytometer and BD FACSDiva version 8.0.1 software (BD Biosciences). Cytometer Setting and Tracking (CST) beads (BD Biosciences) were acquired before each experiment to ensure consistency across all experiments. Stained samples were acquired with a standard stopping gate set at 1 × 105 CD3+ lymphocytes. Unstained cells and single-stained beads (eBioscience) were used for calculating the compensation matrix. Data was analyzed and Boolean cytokine combinations were generated using FlowJo version 9.9.4 software (Treestar Inc.). Background subtractions were performed in Pestle version 1.8.
COMPASS application of flow cytometry data.
Cell counts were analyzed using the COMPASS algorithm as described (66). Briefly, COMPASS is a statistical model developed for high-dimensional flow cytometry data analysis that can detect antigen-specific changes across all observable functional T cell subsets, without the need to limit the analysis to very specific subsets based on expected biological significance. COMPASS was applied to the 3 antigens in 2 (CD4+ and CD8+) T cell subsets, leading to 20 analyses. Each analysis was unbiased and considered all of the 31 possible cytokine functions (defined as Boolean combination). To evaluate differences in polyfunctionality between groups, a linear model estimating the group-wise mean PFS was fit to each antigen, and the difference between IGRA+ and other groups was tested (Wald test, null: β̂ group– β̂ IGRA= 0, 2-sided test). Resulting P values were adjusted for multiple testing (across all 60 tests and models) to control the FDR using the method of Benjamini and Hochberg (90). Significant differences were designated at the 5% FDR level. Magnitudes of T cell responses were calculated independent of COMPASS as the maximum of zero or the proportion of gated events in the stimulated condition minus the proportion of gated events in the unstimulated condition.
Hepatitis B ELISA.
Antibodies to hepatitis B surface antigen were measured in plasma at 1:2 dilution using the anti-HepB surface antigen ELISA kit from XpressBio Life Science Products (catalog WB2896). All study participants received 3 doses of HBV (Figure 1). However, we did not notice any benefits of BCG vaccination on hepatitis B responses in our study, as all subjects across all 4 groups in our study had comparable peak hepatitis B antibody titers at T5 (week 35) after the third hepatitis B vaccination with no significant differences of the peak response over time points tested (Supplemental Figure 8).
Statistics.
All antigen-stimulated wells were adjusted for nonspecific responses by background subtraction (media alone). Statistical analyses were performed, and graphs were created using GraphPad Prism software version 6.0.7 (GraphPad). Paired longitudinal comparisons within the same group were done using the 2-sided Wilcoxon matched pairs signed rank test. Comparisons across 2 treatment groups were done using unpaired Mann-Whitney U test. One-way ANOVA, followed by Bonferroni post hoc tests for multiple comparisons, was used to compare 3 cytokines between 2 groups. Differences were considered statistically significant for P < 0.05. Box-and-whisker plots depict the interquartile range with median bars and with whiskers depicting minimum and maximum values. Scatter plots depict median values with range. In Figure 3, C and G, and Figure 4, C and G, mean ± SD was plotted. For COMPASS, resulting P values were adjusted for multiple testing (across all 60 tests and models) to control the FDR using the method of Benjamini and Hochberg (90).
Study approval.
This study was performed in accordance with the relevant guidelines and regulations stated in the Declaration of Helsinki and approved by the Ethical Review Committee of Arogyavaram Medical Centre (Madanapalle, Andhra Pradesh, India, IEC - AMC 2014/5). All study volunteers provided written consent prior to enrolment. Study protocols were approved by IRBs of Arogyavaram Medical Centre.
Author contributions
MJM and AV conceived the project. SR, AA, SCDR, and AV designed the experiments. SR and AA performed the immunology experiments and analyzed the data. SCDR and VA helped with flow cytometer instrument set up, acquisition, and data analysis. BKS and PNS helped with processing of blood from clinical cohorts. GF performed the main bioinformatic and immunoinformatic analyses. GD and JK were clinical investigators. WB and CJ collected clinical samples, provided the patient details, and wrote the clinical methodology for the manuscript. THMO supplied the latency antigen recombinant proteins. SR, AA, and AV wrote the manuscript. KLMCF synthesized LTAg and ESAT-6/CFP-10 antigens. RG helped with COMPASS analysis. KDS helped with original grant submission and editing the manuscript. SCDR, THMO, MJM, and AV edited the manuscript.
Supplementary Material
Acknowledgments
The authors thank the volunteers for participating in this study and acknowledge the contributions of clinical research workers at Arogyavaram Medical Centre and St. John’s Research Institute. This work was funded principally by DBT-NIH (BT/MB/Indo-US/HIPC/2013) to AV and JM and also supported by the National Institute of Allergy and Infectious Diseases of the NIH (UM1 AI 068618). We acknowledge additional funding for antigen synthesis by EC HORIZON2020 TBVAC2020 and EC FP7 EURIPRED (FP7-INFRA-2012 grant agreement no. 312661) to AV and THMO.
Version 1. 11/19/2019
In-Press Preview
Version 2. 12/19/2019
Electronic publication
Funding Statement
This is a joint funding award by the NIH and the DBT India
Footnotes
Conflict of interest: The authors have declared that no conflict of interest exists.
Copyright: © 2019, American Society for Clinical Investigation.
Reference information: JCI Insight. 2019;4(24):e130540.https://doi.org/10.1172/jci.insight.130540.
Contributor Information
Srabanti Rakshit, Email: srabanti28@yahoo.com.
Asma Ahmed, Email: asma@iisc.ac.in.
Vasista Adiga, Email: vasistaadiga@gmail.com.
Bharath K. Sundararaj, Email: bharathrajks@gmail.com.
Pravat Nalini Sahoo, Email: prabha.nalini@gmail.com.
John Kenneth, Email: johnkennet@gmail.com.
Wesley Bonam, Email: wesleywesleyamc@yahoo.co.in.
Christina Johnson, Email: kittu16in@yahoo.co.in.
Kees L.M.C. Franken, Email: C.L.M.C.Franken@lumc.nl.
Tom H.M. Ottenhoff, Email: t.h.m.ottenhoff@lumc.nl.
Greg Finak, Email: gfinak@fredhutch.org.
Raphael Gottardo, Email: rgottard@fhcrc.org.
Kenneth D. Stuart, Email: ken.stuart@cidresearch.org.
Stephen C. De Rosa, Email: sderosa@fredhutch.org.
M. Juliana McElrath, Email: jmcelrat@fredhutch.org.
Annapurna Vyakarnam, Email: anna.vyakarnam@kcl.ac.uk.
References
- 1. WHO. Global Tuberculosis Report 2018. Geneva, Switzerland: WHO Press; 2018. [Google Scholar]
- 2.Floyd K, Glaziou P, Zumla A, Raviglione M. The global tuberculosis epidemic and progress in care, prevention, and research: an overview in year 3 of the End TB era. Lancet Respir Med. 2018;6(4):299–314. doi: 10.1016/S2213-2600(18)30057-2. [DOI] [PubMed] [Google Scholar]
- 3. WHO. Latent TB Infection: Updated and consolidated guidelines for programmatic management. Geneva, Switzerland: WHO Press; 2018. [PubMed] [Google Scholar]
- 4.WHO BCG vaccine: WHO position paper, February 2018 - Recommendations. Vaccine. 2018;36(24):3408–3410. doi: 10.1016/j.vaccine.2018.03.009. [DOI] [PubMed] [Google Scholar]
- 5.Andersen P, Doherty TM. The success and failure of BCG - implications for a novel tuberculosis vaccine. Nat Rev Microbiol. 2005;3(8):656–662. doi: 10.1038/nrmicro1211. [DOI] [PubMed] [Google Scholar]
- 6.Mangtani P, et al. Protection by BCG vaccine against tuberculosis: a systematic review of randomized controlled trials. Clin Infect Dis. 2014;58(4):470–480. doi: 10.1093/cid/cit790. [DOI] [PubMed] [Google Scholar]
- 7.Moliva JI, Turner J, Torrelles JB. Immune Responses to Bacillus Calmette-Guérin Vaccination: Why Do They Fail to Protect against Mycobacterium tuberculosis? Front Immunol. 2017;8:407. doi: 10.3389/fimmu.2017.00407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dockrell HM, Smith SG. What Have We Learnt about BCG Vaccination in the Last 20 Years? Front Immunol. 2017;8:1134. doi: 10.3389/fimmu.2017.01134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Roy A, et al. Effect of BCG vaccination against Mycobacterium tuberculosis infection in children: systematic review and meta-analysis. BMJ. 2014;349:g4643. doi: 10.1136/bmj.g4643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zodpey SP, Shrikhande SN. The geographic location (latitude) of studies evaluating protective effect of BCG vaccine and it’s efficacy/effectiveness against tuberculosis. Indian J Public Health. 2007;51(4):205–210. [PubMed] [Google Scholar]
- 11.Brandt L, et al. Failure of the Mycobacterium bovis BCG vaccine: some species of environmental mycobacteria block multiplication of BCG and induction of protective immunity to tuberculosis. Infect Immun. 2002;70(2):672–678. doi: 10.1128/IAI.70.2.672-678.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Poyntz HC, Stylianou E, Griffiths KL, Marsay L, Checkley AM, McShane H. Non-tuberculous mycobacteria have diverse effects on BCG efficacy against Mycobacterium tuberculosis. Tuberculosis (Edinb) 2014;94(3):226–237. doi: 10.1016/j.tube.2013.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zimmermann P, Finn A, Curtis N. Does BCG Vaccination Protect Against Nontuberculous Mycobacterial Infection? A Systematic Review and Meta-Analysis. J Infect Dis. 2018;218(5):679–687. doi: 10.1093/infdis/jiy207. [DOI] [PubMed] [Google Scholar]
- 14.Kontturi A, Soini H, Ollgren J, Salo E. Increase in Childhood Nontuberculous Mycobacterial Infections After Bacille Calmette-Guérin Coverage Drop: A Nationwide, Population-Based Retrospective Study, Finland, 1995-2016. Clin Infect Dis. 2018;67(8):1256–1261. doi: 10.1093/cid/ciy241. [DOI] [PubMed] [Google Scholar]
- 15.Abate G, Hamzabegovic F, Eickhoff CS, Hoft DF. BCG Vaccination Induces M. avium and M. abscessus Cross-Protective Immunity. Front Immunol. 2019;10:234. doi: 10.3389/fimmu.2019.00234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Narayanan PR. Influence of sex, age & nontuberculous infection at intake on the efficacy of BCG: re-analysis of 15-year data from a double-blind randomized control trial in South India. Indian J Med Res. 2006;123(2):119–124. [PubMed] [Google Scholar]
- 17.Valadas E. Nontuberculous mycobacteria: clinical importance and relevance to bacille Calmette-Guérin vaccination. Clin Infect Dis. 2004;39(4):457–458. doi: 10.1086/422326. [DOI] [PubMed] [Google Scholar]
- 18.Orme IM. The Achilles heel of BCG. Tuberculosis (Edinb) 2010;90(6):329–332. doi: 10.1016/j.tube.2010.06.002. [DOI] [PubMed] [Google Scholar]
- 19.Hesseling AC, Jaspan HB, Black GF, Nene N, Walzl G. Immunogenicity of BCG in HIV-exposed and non-exposed infants following routine birth or delayed vaccination. Int J Tuberc Lung Dis. 2015;19(4):454–462. doi: 10.5588/ijtld.14.0608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Geldmacher C, Zumla A, Hoelscher M. Interaction between HIV and Mycobacterium tuberculosis: HIV-1-induced CD4 T-cell depletion and the development of active tuberculosis. Curr Opin HIV AIDS. 2012;7(3):268–275. doi: 10.1097/COH.0b013e3283524e32. [DOI] [PubMed] [Google Scholar]
- 21.Pawlowski A, Jansson M, Sköld M, Rottenberg ME, Källenius G. Tuberculosis and HIV co-infection. PLoS Pathog. 2012;8(2):e1002464. doi: 10.1371/journal.ppat.1002464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ahmed A, Rakshit S, Vyakarnam A. HIV-TB co-infection: mechanisms that drive reactivation of Mycobacterium tuberculosis in HIV infection. Oral Dis. 2016;22 Suppl 1:53–60. doi: 10.1111/odi.12390. [DOI] [PubMed] [Google Scholar]
- 23.Hawn TR, et al. Tuberculosis vaccines and prevention of infection. Microbiol Mol Biol Rev. 2014;78(4):650–671. doi: 10.1128/MMBR.00021-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Aerosol Vaccines for Tuberculosis Workshop Summary Group. Developing aerosol vaccines for Mycobacterium tuberculosis: Workshop proceedings: National Institute of Allergy and Infectious Diseases, Bethesda, Maryland, USA, April 9, 2014. Vaccine. 2015;33(26):3038–3046. doi: 10.1016/j.vaccine.2015.03.060. [DOI] [PubMed] [Google Scholar]
- 25.Hansen SG, et al. Prevention of tuberculosis in rhesus macaques by a cytomegalovirus-based vaccine. Nat Med. 2018;24(2):130–143. doi: 10.1038/nm.4473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kaufmann SH, Weiner J, von Reyn CF. Novel approaches to tuberculosis vaccine development. Int J Infect Dis. 2017;56:263–267. doi: 10.1016/j.ijid.2016.10.018. [DOI] [PubMed] [Google Scholar]
- 27.Satti I, et al. Safety and immunogenicity of a candidate tuberculosis vaccine MVA85A delivered by aerosol in BCG-vaccinated healthy adults: a phase 1, double-blind, randomised controlled trial. Lancet Infect Dis. 2014;14(10):939–946. doi: 10.1016/S1473-3099(14)70845-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Barreto ML, Pereira SM, Ferreira AA. BCG vaccine: efficacy and indications for vaccination and revaccination. J Pediatr (Rio J) 2006;82(3 Suppl):S45–S54. doi: 10.2223/JPED.1499. [DOI] [PubMed] [Google Scholar]
- 29.Karonga Prevention Trial Group. Randomised controlled trial of single BCG, repeated BCG, or combined BCG and killed Mycobacterium leprae vaccine for prevention of leprosy and tuberculosis in Malawi. Lancet. 1996;348(9019):17–24. doi: 10.1016/S0140-6736(96)02166-6. [DOI] [PubMed] [Google Scholar]
- 30.Leung CC, Tam CM, Chan SL, Chan-Yeung M, Chan CK, Chang KC. Efficacy of the BCG revaccination programme in a cohort given BCG vaccination at birth in Hong Kong. Int J Tuberc Lung Dis. 2001;5(8):717–723. [PubMed] [Google Scholar]
- 31.Roth AE, et al. Effect of revaccination with BCG in early childhood on mortality: randomised trial in Guinea-Bissau. BMJ. 2010;340:c671. doi: 10.1136/bmj.c671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tala-Heikkilä MM, Tuominen JE, Tala EO. Bacillus Calmette-Guérin revaccination questionable with low tuberculosis incidence. Am J Respir Crit Care Med. 1998;157(4 Pt 1):1324–1327. doi: 10.1164/ajrccm.157.4.9706037. [DOI] [PubMed] [Google Scholar]
- 33.Rahman M, Sekimoto M, Hira K, Koyama H, Imanaka Y, Fukui T. Is BCG revaccination necessary for Japanese children? Prev Med. 2002;35(1):70–77. doi: 10.1006/pmed.2002.1043. [DOI] [PubMed] [Google Scholar]
- 34.Kubit S, Czajka S, Olakowski T, Piasecki Z. [Effectiveness of BCG vaccination] Pediatr Pol. 1983;58(10):775–781. [PubMed] [Google Scholar]
- 35.Fjällbrant H, Ridell M, Larsson LO. Primary vaccination and revaccination of young adults with BCG: a study using immunological markers. Scand J Infect Dis. 2007;39(9):792–798. doi: 10.1080/00365540701367777. [DOI] [PubMed] [Google Scholar]
- 36.Whelan KT, et al. Safety and immunogenicity of boosting BCG vaccinated subjects with BCG: comparison with boosting with a new TB vaccine, MVA85A. PLoS ONE. 2009;4(6):e5934. doi: 10.1371/journal.pone.0005934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Dourado I, et al. Rates of adverse reactions to first and second doses of BCG vaccination: results of a large community trial in Brazilian schoolchildren. Int J Tuberc Lung Dis. 2003;7(4):399–402. [PubMed] [Google Scholar]
- 38.Dantas OM, et al. A case-control study of protection against tuberculosis by BCG revaccination in Recife, Brazil. Int J Tuberc Lung Dis. 2006;10(5):536–541. [PubMed] [Google Scholar]
- 39.Rodrigues LC, et al. Effect of BCG revaccination on incidence of tuberculosis in school-aged children in Brazil: the BCG-REVAC cluster-randomised trial. Lancet. 2005;366(9493):1290–1295. doi: 10.1016/S0140-6736(05)67145-0. [DOI] [PubMed] [Google Scholar]
- 40.Barreto ML, et al. Evidence of an effect of BCG revaccination on incidence of tuberculosis in school-aged children in Brazil: second report of the BCG-REVAC cluster-randomised trial. Vaccine. 2011;29(31):4875–4877. doi: 10.1016/j.vaccine.2011.05.023. [DOI] [PubMed] [Google Scholar]
- 41.Barreto ML, et al. Causes of variation in BCG vaccine efficacy: examining evidence from the BCG REVAC cluster randomized trial to explore the masking and the blocking hypotheses. Vaccine. 2014;32(30):3759–3764. doi: 10.1016/j.vaccine.2014.05.042. [DOI] [PubMed] [Google Scholar]
- 42.Hatherill M, et al. Safety and reactogenicity of BCG revaccination with isoniazid pretreatment in TST positive adults. Vaccine. 2014;32(31):3982–3988. doi: 10.1016/j.vaccine.2014.04.084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ahmad NA, Hamid HAA, Sahril N, Yusoff MFM, Naidu BM, Aris T. Bacille Calmette-Guerin (BCG) Revaccination: Is it Beneficial for Tuberculosis Control? Sci Rep. 2013;2:656 [Google Scholar]
- 44.Dye C. Making wider use of the world’s most widely used vaccine: Bacille Calmette-Guerin revaccination reconsidered. J R Soc Interface. 2013;10(87):20130365. doi: 10.1098/rsif.2013.0365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Nemes E, et al. Prevention of M. tuberculosis Infection with H4:IC31 Vaccine or BCG Revaccination. N Engl J Med. 2018;379(2):138–149. doi: 10.1056/NEJMoa1714021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Oliveira ES, Marinho JM, Barbosa T, study group Interferon-gamma production by mononuclear cells in Bacille Calmette-Guérin-revaccinated healthy volunteers predicted long-term antimycobacterial responses in a randomized controlled trial. Vaccine. 2013;31(37):3778–3782. doi: 10.1016/j.vaccine.2013.04.079. [DOI] [PubMed] [Google Scholar]
- 47.Andersen A, et al. The immunological effect of revaccination with Bacille Calmette-Guérin vaccine at 19 months of age. Vaccine. 2013;31(17):2137–2144. doi: 10.1016/j.vaccine.2013.02.050. [DOI] [PubMed] [Google Scholar]
- 48.Suliman S, et al. Bacillus Calmette-Guérin (BCG) Revaccination of Adults with Latent Mycobacterium tuberculosis Infection Induces Long-Lived BCG-Reactive NK Cell Responses. J Immunol. 2016;197(4):1100–1110. doi: 10.4049/jimmunol.1501996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Rakshit S, et al. Circulating Mycobacterium tuberculosis DosR latency antigen-specific, polyfunctional, regulatory IL10+ Th17 CD4 T-cells differentiate latent from active tuberculosis. Sci Rep. 2017;7(1):11948. doi: 10.1038/s41598-017-10773-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Dijkman K, et al. Prevention of tuberculosis infection and disease by local BCG in repeatedly exposed rhesus macaques. Nat Med. 2019;25(2):255–262. doi: 10.1038/s41591-018-0319-9. [DOI] [PubMed] [Google Scholar]
- 51.Skjøt RL, et al. Epitope mapping of the immunodominant antigen TB10.4 and the two homologous proteins TB10.3 and TB12.9, which constitute a subfamily of the esat-6 gene family. Infect Immun. 2002;70(10):5446–5453. doi: 10.1128/IAI.70.10.5446-5453.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hatherill M, et al. BCG and New Preventive Tuberculosis Vaccines: Implications for Healthcare Workers. Clin Infect Dis. 2016;62 Suppl 3:S262–S267. doi: 10.1093/cid/ciw025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wiker HG, Harboe M. The antigen 85 complex: a major secretion product of Mycobacterium tuberculosis. Microbiol Rev. 1992;56(4):648–661. doi: 10.1128/mr.56.4.648-661.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Belisle JT, Vissa VD, Sievert T, Takayama K, Brennan PJ, Besra GS. Role of the major antigen of Mycobacterium tuberculosis in cell wall biogenesis. Science. 1997;276(5317):1420–1422. doi: 10.1126/science.276.5317.1420. [DOI] [PubMed] [Google Scholar]
- 55.Coppola M, et al. New Genome-Wide Algorithm Identifies Novel In-Vivo Expressed Mycobacterium Tuberculosis Antigens Inducing Human T-Cell Responses with Classical and Unconventional Cytokine Profiles. Sci Rep. 2016;6:37793. doi: 10.1038/srep37793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lin MY, et al. Lack of immune responses to Mycobacterium tuberculosis DosR regulon proteins following Mycobacterium bovis BCG vaccination. Infect Immun. 2007;75(7):3523–3530. doi: 10.1128/IAI.01999-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Billeskov R, Elvang TT, Andersen PL, Dietrich J. The HyVac4 subunit vaccine efficiently boosts BCG-primed anti-mycobacterial protective immunity. PLoS ONE. 2012;7(6):e39909. doi: 10.1371/journal.pone.0039909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Elvang T, et al. CD4 and CD8 T cell responses to the M. tuberculosis Ag85B-TB10.4 promoted by adjuvanted subunit, adenovector or heterologous prime boost vaccination. PLoS ONE. 2009;4(4):e5139. doi: 10.1371/journal.pone.0005139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Verreck FA, et al. MVA.85A boosting of BCG and an attenuated, phoP deficient M. tuberculosis vaccine both show protective efficacy against tuberculosis in rhesus macaques. PLoS ONE. 2009;4(4):e5264. doi: 10.1371/journal.pone.0005264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Goonetilleke NP, McShane H, Hannan CM, Anderson RJ, Brookes RH, Hill AV. Enhanced immunogenicity and protective efficacy against Mycobacterium tuberculosis of bacille Calmette-Guérin vaccine using mucosal administration and boosting with a recombinant modified vaccinia virus Ankara. J Immunol. 2003;171(3):1602–1609. doi: 10.4049/jimmunol.171.3.1602. [DOI] [PubMed] [Google Scholar]
- 61.Tameris MD, et al. Safety and efficacy of MVA85A, a new tuberculosis vaccine, in infants previously vaccinated with BCG: a randomised, placebo-controlled phase 2b trial. Lancet. 2013;381(9871):1021–1028. doi: 10.1016/S0140-6736(13)60177-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Arroyo L, Marín D, Franken KLMC, Ottenhoff THM, Barrera LF. Potential of DosR and Rpf antigens from Mycobacterium tuberculosis to discriminate between latent and active tuberculosis in a tuberculosis endemic population of Medellin Colombia. BMC Infect Dis. 2018;18(1):26. doi: 10.1186/s12879-017-2929-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Feng CG, Britton WJ. CD4+ and CD8+ T cells mediate adoptive immunity to aerosol infection of Mycobacterium bovis bacillus Calmette-Guérin. J Infect Dis. 2000;181(5):1846–1849. doi: 10.1086/315466. [DOI] [PubMed] [Google Scholar]
- 64.Moliva JI, Turner J, Torrelles JB. Immune Responses to Bacillus Calmette-Guérin Vaccination: Why Do They Fail to Protect against Mycobacterium tuberculosis? Front Immunol. 2017;8:407. doi: 10.3389/fimmu.2017.00407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Moguche AO, et al. Antigen Availability Shapes T Cell Differentiation and Function during Tuberculosis. Cell Host Microbe. 2017;21(6):695–706.e5. doi: 10.1016/j.chom.2017.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lin L, et al. COMPASS identifies T-cell subsets correlated with clinical outcomes. Nat Biotechnol. 2015;33(6):610–616. doi: 10.1038/nbt.3187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Marín ND, París SC, Rojas M, García LF. Functional profile of CD4+ and CD8+ T cells in latently infected individuals and patients with active TB. Tuberculosis (Edinb) 2013;93(2):155–166. doi: 10.1016/j.tube.2012.12.002. [DOI] [PubMed] [Google Scholar]
- 68.Bhatt K, Verma S, Ellner JJ, Salgame P. Quest for correlates of protection against tuberculosis. Clin Vaccine Immunol. 2015;22(3):258–266. doi: 10.1128/CVI.00721-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Yuan X, et al. A live attenuated BCG vaccine overexpressing multistage antigens Ag85B and HspX provides superior protection against Mycobacterium tuberculosis infection. Appl Microbiol Biotechnol. 2015;99(24):10587–10595. doi: 10.1007/s00253-015-6962-x. [DOI] [PubMed] [Google Scholar]
- 70.Maggioli MF, et al. Increased TNF-α/IFN-γ/IL-2 and Decreased TNF-α/IFN-γ Production by Central Memory T Cells Are Associated with Protective Responses against Bovine Tuberculosis Following BCG Vaccination. Front Immunol. 2016;7:421. doi: 10.3389/fimmu.2016.00421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.White AD, et al. Evaluation of the Immunogenicity of Mycobacterium bovis BCG Delivered by Aerosol to the Lungs of Macaques. Clin Vaccine Immunol. 2015;22(9):992–1003. doi: 10.1128/CVI.00289-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Smith SG, Zelmer A, Blitz R, Fletcher HA, Dockrell HM. Polyfunctional CD4 T-cells correlate with in vitro mycobacterial growth inhibition following Mycobacterium bovis BCG-vaccination of infants. Vaccine. 2016;34(44):5298–5305. doi: 10.1016/j.vaccine.2016.09.002. [DOI] [PubMed] [Google Scholar]
- 73.Shen H, Chen ZW. The crucial roles of Th17-related cytokines/signal pathways in M. tuberculosis infection. Cell Mol Immunol. 2018;15(3):216–225. doi: 10.1038/cmi.2017.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Khader SA, et al. IL-23 and IL-17 in the establishment of protective pulmonary CD4+ T cell responses after vaccination and during Mycobacterium tuberculosis challenge. Nat Immunol. 2007;8(4):369–377. doi: 10.1038/ni1449. [DOI] [PubMed] [Google Scholar]
- 75.Wareham AS, Tree JA, Marsh PD, Butcher PD, Dennis M, Sharpe SA. Evidence for a role for interleukin-17, Th17 cells and iron homeostasis in protective immunity against tuberculosis in cynomolgus macaques. PLoS ONE. 2014;9(2):e88149. doi: 10.1371/journal.pone.0088149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Cruz A, et al. BCG vaccination-induced long-lasting control of Mycobacterium tuberculosis correlates with the accumulation of a novel population of CD4+IL-17+TNF+IL-2+ T cells. Vaccine. 2015;33(1):85–91. doi: 10.1016/j.vaccine.2014.11.013. [DOI] [PubMed] [Google Scholar]
- 77.Alber G, Kamradt T. Regulation of Protective and Pathogenic Th17 Responses. Curr Immunol Rev. 2007;3(1):3–16. [Google Scholar]
- 78.Cavalcanti YV, Brelaz MC, Neves JK, Ferraz JC, Pereira VR. Role of TNF-Alpha, IFN-Gamma, and IL-10 in the Development of Pulmonary Tuberculosis. Pulm Med. 2012;2012:745483. doi: 10.1155/2012/745483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Perdomo C, et al. Mucosal BCG Vaccination Induces Protective Lung-Resident Memory T Cell Populations against Tuberculosis. MBio. 2016;7(6):e01686-16. doi: 10.1128/mBio.01686-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Kleinnijenhuis J, et al. Long-lasting effects of BCG vaccination on both heterologous Th1/Th17 responses and innate trained immunity. J Innate Immun. 2014;6(2):152–158. doi: 10.1159/000355628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Kleinnijenhuis J, van Crevel R, Netea MG. Trained immunity: consequences for the heterologous effects of BCG vaccination. Trans R Soc Trop Med Hyg. 2015;109(1):29–35. doi: 10.1093/trstmh/tru168. [DOI] [PubMed] [Google Scholar]
- 82.Sia JK, Georgieva M, Rengarajan J. Innate Immune Defenses in Human Tuberculosis: An Overview of the Interactions between Mycobacterium tuberculosis and Innate Immune Cells. J Immunol Res. 2015;2015:747543. doi: 10.1155/2015/747543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Sada-Ovalle I, Chiba A, Gonzales A, Brenner MB, Behar SM. Innate invariant NKT cells recognize Mycobacterium tuberculosis-infected macrophages, produce interferon-gamma, and kill intracellular bacteria. PLoS Pathog. 2008;4(12):e1000239. doi: 10.1371/journal.ppat.1000239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Rahman S, et al. Compartmentalization of immune responses in human tuberculosis: few CD8+ effector T cells but elevated levels of FoxP3+ regulatory t cells in the granulomatous lesions. Am J Pathol. 2009;174(6):2211–2224. doi: 10.2353/ajpath.2009.080941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Sakai S, et al. Cutting edge: control of Mycobacterium tuberculosis infection by a subset of lung parenchyma-homing CD4 T cells. J Immunol. 2014;192(7):2965–2969. doi: 10.4049/jimmunol.1400019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Pattnaik S, John KR, Shalini E, Michael JS. Agreement between skin testing and QuantiFERON-TB Gold In-Tube assay (QFT-TB) in detecting latent tuberculosis infection among household contacts in India. Indian J Tuberc. 2012;59(4):214–218. [PubMed] [Google Scholar]
- 87.Horton H, et al. Optimization and validation of an 8-color intracellular cytokine staining (ICS) assay to quantify antigen-specific T cells induced by vaccination. J Immunol Methods. 2007;323(1):39–54. doi: 10.1016/j.jim.2007.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kagina BM, et al. Qualification of a whole blood intracellular cytokine staining assay to measure mycobacteria-specific CD4 and CD8 T cell immunity by flow cytometry. J Immunol Methods. 2015;417:22–33. doi: 10.1016/j.jim.2014.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Smith SG, et al. Intracellular Cytokine Staining and Flow Cytometry: Considerations for Application in Clinical Trials of Novel Tuberculosis Vaccines. PLoS ONE. 2015;10(9):e0138042. doi: 10.1371/journal.pone.0138042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Green GH, Diggle PJ. On the operational characteristics of the Benjamini and Hochberg False Discovery Rate procedure. Stat Appl Genet Mol Biol. 2007;6:Article27. doi: 10.2202/1544-6115.1302. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.