Abstract
Calcium ion dyshomeostasis contributes to the progression of many neurodegenerative diseases and represents a target for the development of neuroprotective therapies (1–3). Dysfunctional ryanodine receptors contribute to calcium ion dyshomeostasis and potentially to the pathogenesis of neurodegenerative diseases by generating abnormal calcium ion release from the endoplasmic reticulum (4–5). Since ryanodine receptors share functional and structural similarities with potassium channels (6), and small molecules with anti-oxidant properties, such as resveratrol (3,5,4′-trihydroxy-trans-stilbene), directly control the activity of potassium channels (7–12), we hypothesized that trans-resveratrol can modulate intracellular calcium signaling through direct binding and functional regulation of ryanodine receptors. The goal of our study was to identify and measure the control of ryanodine receptor activity by trans-resveratrol. Mechanisms of calcium signaling mediated by the direct interaction between trans-resveratrol and ryanodine receptors, were identified and measured with single channel electrophysiology. Addition of trans-resveratrol to the cytoplasmic face of the ryanodine receptor increased single channel activity at physiological and elevated pathophysiological cytoplasmic calcium ion concentrations. The open probability of the channel increases after interacting with the small molecule in a dose-dependent manner, but remains also dependent on the concentration of its physiological ligand, cytoplasmic free calcium ions. This study provides the first evidence of a direct functional interaction between trans-resveratrol and ryanodine receptors. Such functional control of ryanodine receptors by trans-resveratrol as a novel mechanism of action could provide additional rationales for the development of novel therapeutic strategies to treat and prevent neurodegenerative diseases.
Keywords: 3,5,4′-trihydroxy-trans-stilbene; calcium; Ca2+; electrophysiology; neurodegeneration; neuroprotection; neuroprotective therapies; pharmacology; RyR; trans-resveratrol
Introduction
Calcium ions (Ca2+) contribute to multiple signaling pathways in neurons, functioning as a universal secondary messenger with the activation of Ca2+-controlled signaling pathways dependent on both intracellular Ca2+ stores and the rate of change in intracellular Ca2+ concentrations (13–14). Cellular Ca2+ entry is mediated through Voltage-Operated Ca2+ Channels (VOCC), Receptor-Operated Ca2+ Channels (ROCC), and Store-Operated Ca2+ Channels (SOCC) and is amplified by calcium-induced Ca2+ -release (CICR) channels in the endoplasmic reticulum (ER) and mitochondrial membranes mediating localized Ca2+ release from ER and mitochondrial Ca2+ stores (15–20). The termination of such intracellular Ca2+ transients and restoration of resting cytoplasmic Ca2+ concentrations is accomplished by the activity of transmembrane proteins of the plasma membrane (plasma membrane Ca2+ ATPase, PMCA; Na+/Ca2+ exchanger, NCX), the ER membrane (Sarco-/endoplasmic reticulum Ca2+-ATPase, SERCA), and the mitochondrial inner membrane (mitochondrial calcium uniporter, MCU) (21–22). Additionally, Ca2+ sequestration by Ca2+-binding proteins and Ca2+ buffering molecules contributes to restoring resting cytoplasmic Ca2+ concentrations (17–18, 23–29). Ryanodine receptors (RyR) and inositol-1, 4, 5-trisphosphate receptors (IP3R), ER transmembrane proteins, are clustered in micro-domains and respond to and amplify VOCC/ROCC/SOCC-mediated Ca2+ entry, thereby mediating CICR (30–31).
Ca2+ dyshomeostasis, a hallmark of several neurodegenerative diseases including Alzheimer’s disease (AD), Huntington’s disease (HD), Parkinson’s disease (PD), and amyotrophic lateral sclerosis (ALS) results in and is associated with altered Ca2+ buffering capacity, altered regulation of Ca2+ channels and pumps, and altered neuronal excitability (28, 32–33). Experimental models of neuronal aging have provided evidence for the “Calcium hypothesis of brain aging,” which describes how aging is closely linked to dysregulation of Ca2+ homeostasis resulting in a chronically elevated level of cytosolic Ca2+, which in turn contributes to age-related deficits in memory and learning through altered intracellular Ca2+ transients (1, 34). Similarly, pathogenic mechanisms disrupt Ca2+ homeostasis either in chronic (e.g. AD and ALS) or acute disease conditions (e.g. cerebral ischemia and traumatic brain injury) (32, 34–37). While the hypothesis focused initially on plasma membrane VOCC and decreased PMCA activity (40, 64–65), altered functional interactions between VOCC and RyR and abnormal Ca2+ buffering capacity of aged neurons were identified subsequently as contributing mechanisms (38–42).
As RyR and IP3R CICR channels are responsible for amplifying Ca2+ signals under normal physiological conditions, but also during periods of intracellular Ca2+ dysregulation, they represent potential targets for neuroprotective therapy development. RyR2 and RyR3 are the two prominent isotypes found in the cerebellum, cortex, and hippocampus regions of the brain (1, 6, 43–44). Brain RyR activation is highly dependent on cytosolic Ca2+ concentrations; however, the receptor also contains a secondary, low affinity Ca2+ binding site that inhibits channel activity when high levels of free cytosolic Ca2+ are present (6, 32–33, 45). In addition to Ca2+, other small molecules, both endogenous signaling substances as well as pharmacological agents, bind to the RyR and can modulate the release of Ca2+ from the ER (46–50). The goal of our study was to identify and measure the control of brain RyR activity by one such molecule with potential pharmacological benefits for the therapy of neurodegenerative processes, trans-resveratrol. As a naturally occurring stilbenoid compound with multiple potential medicinal applications in cardiovascular, neurological and inflammatory disorders (51–55), trans-resveratrol affects several cellular signaling pathways involved in disease pathogenesis, most notably through its antioxidant properties by directly scavenging reactive oxygen species and by upregulating neuronal antioxidant proteins, such as quinone reductase 2 and heme oxygenase 1 (56–60), but also by a range of pleiotropic effects such as inhibiting pro-inflammatory signaling and enhancing NF-κB signaling (54, 58, 61). While trans-resveratrol directly attenuates programmed cell death and calcium toxicity induced by oxygen-glucose deprivation in primary neurons (62), it also stimulates cell cycle arrest, caspase activation, cytochrome c release and apoptosis in a range of cancer cell lines (63–67). Some of these pharmacological properties of trans-resveratrol are likely caused by the control of cellular signaling pathways, specifically through interactions with ion channels and related signaling proteins. Trans-resveratrol regulates the biophysical properties of a variety of ion channels including the inhibition of the gamma-aminobutyric acid (GABA) receptor (68) and the activation of 5-hydroxytryptamine (5-HT) / serotonin receptors (69) and some transient receptor potential (TRP) channels, such as TRPA channels, but not TRPV1 receptors (70). Trans-resveratrol treatment reduces the activity of L- and T-type VOCCs and voltage-gated sodium channels in cardiomyocytes, neurons, and cell lines at the single channel level (7–8, 71–72). In addition, a range of studies document the control of potassium channels, including Ca2+-activated K+ (KCa), voltage-gated K+, and inwardly rectifying K+ channels by trans-resveratrol. Such pharmacological activity includes the activation of large conductance Ca2+-activated K+ channels (BKCa) (7–10) and the inhibition of ATP-sensitive K+ channels (KATP) (11–12) where specific underlying mechanisms of action, such as direct modulation of the ion channel versus effects mediated by posttranslational modifications for example, remain to be identified. While trans-resveratrol’s direct interaction with L-Type VOCC channels, PMCA, and KATP channels are well documented, studies identifying trans-resveratrol-mediated modulation of intracellular Ca2+ signaling lack a clear mechanistic focus on proteins controlling CICR. The present study tests the hypothesis that direct binding of trans-resveratrol to intracellular calcium release channels (RyR) controls channel activity based on the rationale that RyRs and K+ channels share significant structural homology in their pore forming transmembrane regions (12, 73–75).
In a potential coupling effect, trans-resveratrol directly regulates L- and T-type Ca2+ ion channels that can mediate CICR. Trans-resveratrol also attenuates expression and activity of Ca2+ handling proteins such as CaMKII and prevents downregulation of Ca2+-sequestering protein pump levels in neuronal pathophysiology (76–78).
The pharmacological control of abnormal and potentially neurotoxic Ca2+ signaling as an approach for the treatment of or the protection against neurodegenerative diseases has been established through a variety of in vitro and in vivo models, but currently available data is focused on voltage-gated Ca2+ and storage-operated Ca2+ channels of the plasma membrane (1, 8). However, while the role of intracellular Ca2+-induced Ca2+-release channels in neuronal physiology and in neurodegenerative diseases has been studied, novel research into the regulation of these channels by trans-resveratrol has significant potential to identify innovative therapeutic approaches. In the present study, it is hypothesized that trans-resveratrol directly binds neuronal RyRs and thereby controls their activity. To test this hypothesis, single channel electrophysiology was used to determine the change in biophysical properties of brain RyRs in the presence of trans-resveratrol.
Materials and Methods
Measurement of RyR single channel biophysical properties with planar lipid bilayer electrophysiology
Isolation and enrichment of mouse brain endoplasmic reticulum
Endoplasmic reticulum (ER) was isolated from brain tissue of C57BL/6 mice (7–9 weeks of age; The Jackson Laboratory, Sacramento, CA; approved by the Institutional Animal Care and Use Committee). Following dissection, tissue was washed in chilled (4°C), Ca2+- and Mg2+-free phosphate buffered saline and transferred to homogenization buffer [260mM sucrose, 5 mM HEPES, 1 mM EGTA (ethylene glycol-bis(β-aminoethyl ether)-N,N,N′,N′-tetraacetic acid), 1 mM DTT (dichlorodiphenyltrichlorethane), and 10 μg/mL protease inhibitor cocktail consisting of PMSF (phenylmethylsulfonyl fluoride), aprotinin, pepstatin, trypsin inhibitor at pH 7.35]. The tissue was homogenized with a glass Dounce homogenizer in an excess of cold homogenization buffer and the homogenate was transferred to an ultracentrifuge tube to separate cell debris and the nuclear fraction by centrifugation at 1,000 × g for 10 min in a Sorvall™ TFT-80.2 rotor and WX ultracentrifuge (ThermoFisher Scientific, Waltham, MA). Supernatant between the debris pellet and floating lipid layer was transferred to a new chilled ultracentrifuge tube and spun down at 12,000 × g for 20 minutes to separate the ER from the mitochondrial fractions. One final spin at 100,000 × g for 75 min was performed after the supernatant (between the mitochondrial fraction pellet and lipid layer) was transferred to a new chilled tube to collect the ER fraction pellet. Storage buffer, mimicking the homogenization buffer with the omission of EGTA, was used to resuspend the ER pellet. The enriched ER microsome preparations were then quickly aliquoted, snap frozen in liquid nitrogen, and stored at −80°C until use.
Single channel planar lipid bilayer system configuration
The principle underlying planar lipid bilayer electrophysiology is the formation of an artificial lipid bilayer across a small aperture connecting two independent reservoirs (Suppl. Fig. 1) and the handling of two distinct buffers representing the cytosol (cis reservoir) and the ER lumen (trans reservoir). The planar lipid bilayer (Warner Instruments, Hamden, CT) chamber contains two reservoirs, cis and trans, with the trans side containing a polystyrene cuvette with an aperture size of 150–200 μm. The cis reservoir and trans-cuvette are filled with 1 mL of cis-buffer (250 mM HEPES (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid), 110 mM Tris (2-Amino-2-(hydroxymethyl) propane-1,3-diol), pH of 7.35 ± 0.05) and trans-buffer (50 mM barium hydroxide (Ba(OH)2) and 250 mM HEPES, pH of 7.5 ± 0.05), respectively. Two millimeter silver-silver chloride electrodes (Ag-AgCl) from the headstage were inserted into their respective salt buffer chambers where approximately 200 μL of a 3 M KCl solution was added in conjunction with salt bridges (5% agar in 1 M KCl) to effectively connect the cis or trans reservoirs to the electrical signal amplifier (Warner Instruments BCD525). Two lipid compositions were prepared by mixing phosphatidylcholine (PC) and phosphatidylserine (PS) (Avanti Polar Lipids, Alabaster, AL) 4:1 to a final concentration of 25 mg/mL or phosphatidylethanolamine (PE) and phosphatidylserine 3:1 to a final concentration of 40 mg/mL dissolved in decane. The aperture is initially primed with lipids using the PC:PS mixture to facilitate the collection and arrangement of the PE:PS lipid bilayer mixture. The thickness of the bilayer is visualized in a capacitance test to ensure the pore forming region of the channel can pass completely through the lipid bilayer. When needed, the bilayer thickness was manually adjusted with a small air bubble, protruding from a capillary tube, passing over the aperture to disperse the lipids. A final integrity test was performed with the trans reservoir grounded in the chamber and the cis side clamped at 0 mV. A stable and rigid seal of the aperture was monitored by the current movement between the cis and trans reservoirs with a measurement of 0 pA correlating to a closed membrane.
Measurement of brain ryanodine receptor biophysical activity, confirmation of channel orientation, and experimental protocol
Once optimal bilayer thickness was achieved (≥ 80 pF) ER microsomes containing RyRs were added to the cis reservoir, a brief vortex distributed the microsomes followed by a 20–30 min incubation. Microsome fusion occurred when microsomes move in proximity to the artificial bilayer and spontaneously fuse into the lipid bilayer. This process can also be facilitated with the addition of small 3 M KCl volumes (50 – 100 μL) to the cis reservoir, effectively forming an osmotic gradient, resulting in the swelling of the microsome and enhancing fusion. Fusion was visualized by the appearance of channel activity and confirmed with the absence of activity after the cis reservoir was washed with 10 mL of cis buffer. To ensure only cis facing RyRs (with their cytosolic portion in the cis reservoir) are measured the use of barium (Ba2+) as a current carrying ion was used. The Ba2+ ion can pass through the RyR, replicating the movement of Ca2+ through RyRs, but does not affect channel activity as it does not bind to the Ca2+ binding sites; any RyRs facing the trans reservoir cannot be activated by Ba2+. Activation of RyR currents was stimulated with increasing additions of Ca2+ and EGTA in proportions, calculated by MaxChelator software (Stanford University, Sanford, CA), to achieve a desired free cytosolic Ca2+ concentration. Ca2+ concentrations are expressed as pCa, defined as the negative decadic logarithm of the free Ca2+ concentration.
A baseline of RyR current activity was recorded for 3.5 min after observing activity at a desired Ca2+ concentration establishing the channel’s baseline activity. An increasing concentration of trans-resveratrol (dissolved in 100% DMSO and diluted with cis buffer) was added to the cis reservoir, vortexed for ~10 sec, and allowed to incubate for 30 sec before a 3.5 min recording was collected. Each recording was performed at room temperature with the traces collected and saved individually. At the conclusion of each experiment 10 μM ruthenium red (RR) was added to the cis reservoir as a selective inhibitor of RyRs as a secondary pharmacological tool to confirm the presence of RyRs.
Software configuration for data acquisition and analysis of single channels
Amplifying the analog electrophysiological signals was performed using a BCD525 amplifier with filter parameters to reduce noise amplitudes at 500 Hz. The analog to digital conversation was performed at 10 kHz within a Molecular Devices (Sunnyvale, CA) Digidata 1322A system and pClamp 10 software. Each individual single channel’s recording baseline was set to 0 pA, representing a closed channel, and a Gaussian low pass (320 Hz) filter was applied to minimize the electrical noise generated by the digitalization process. A single-channel search was performed using ClampFit 10 software over a 3 min timeline with two identified sub-conductance states, S2 and S4. Four biophysical properties were calculated by the software in each sub-conductance level including the number of channel openings (number of events), duration of channel openings (dwell time), average current transferred (average amplitude), and the probability of the channel being open over the given time (open probability, Po).
Biophysical data analysis and comparison of single channels in the absence or presence of trans-resveratrol
To limit the impact of variance in RyR channel activity, because of scaffolding proteins being removed during ER enrichment process or different levels of posttranslational modifications, normalized biophysical values were reported as a percentage of the baseline. Each initial baseline, i.e. RyR activity after Ca2+ activation, was used to normalize subsequent trans-resveratrol dosage recordings to off-set the variable activity among single channels and allow comparison of replicates. Trans-resveratrol was applied to the cis reservoir, vortexed for 10 sec, and channel activity was recorded for 3.5 min. Subsequent trans-resveratrol volumes were added to the cis reservoir up to a final 100 μM trans-resveratrol concentration. Prism GraphPad software was used to calculate and plot the normalized values and express the three levels of significance (p < 0.05, p < 0.01, and p < 0.001) using one-way ANOVA with Dunnett’s post hoc test.
Results
Trans-resveratrol potentiates brain ryanodine receptor activity at elevated cytosolic calcium concentrations
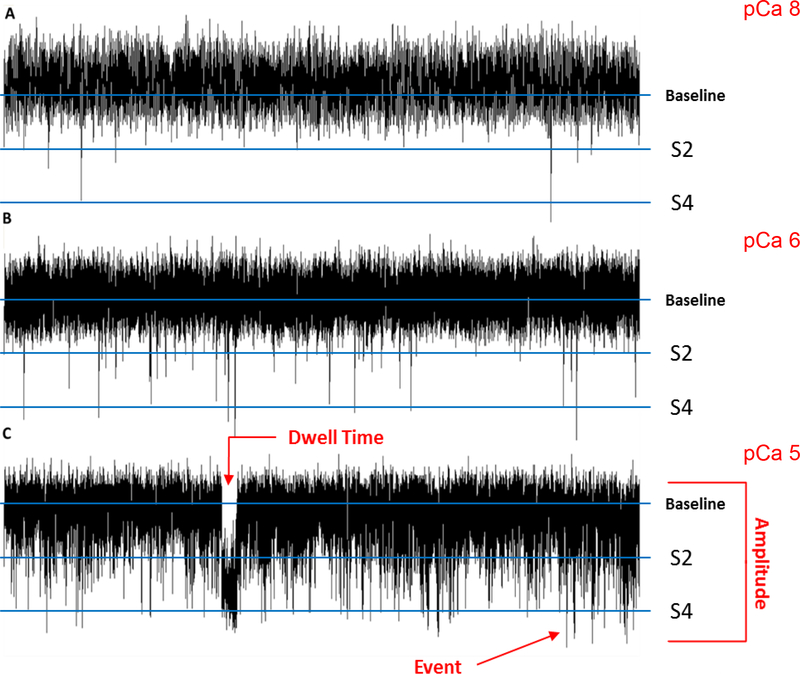
Figure 1 shows brain RyR single channel activity at three different cytosolic Ca2+ concentrations. The representative traces demonstrate more frequent channel openings with cytosolic Ca2+ concentrations increasing from 10 nM to 10 μM (pCa, defined as the negative decadic logarithm of the free Ca2+ concentration, 8 to 5; Fig. 1) without changes in single channel amplitude (Fig. 2). Three unique RyR sub-conductance states with distinct single channel amplitudes are indicated in Fig. 1, closed, partially open (S2), fully open (S4) subconductance states.
Figure 1. Representative traces of mouse brain ryanodine receptor activity.
Sample 30 s current traces at resting (A, 10 nM, pCa 8) and elevated cytosolic Ca2+ concentrations (B, 1 μM, pCa 6; C, 10 μM, pCa 5). Channel openings are represented by downward deflections from the closed state at zero current baseline to a 2 pA subconductance state (S2) and to the fully open state (S4) at 4 pA.
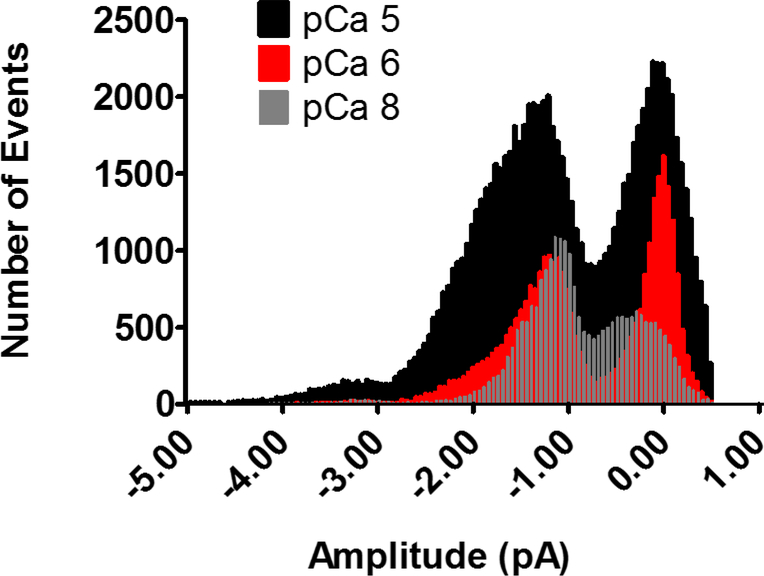
Figure 2. Channel amplitude histograms from mouse brain ryanodine receptor single channel activity recorded at varying cytosolic Ca2+ concentrations.
Channels were activated by 10 μM (pCa 5; black), 1 μM (pCa 6; red), and 10 nM (pCa 8; grey) free intracellular calcium concentrations. In response to increased cytosolic Ca2+ concentrations, the frequency of events at both the 2 pA subconductance state (S2) and the 4 pA fully open state (S4) is increased.
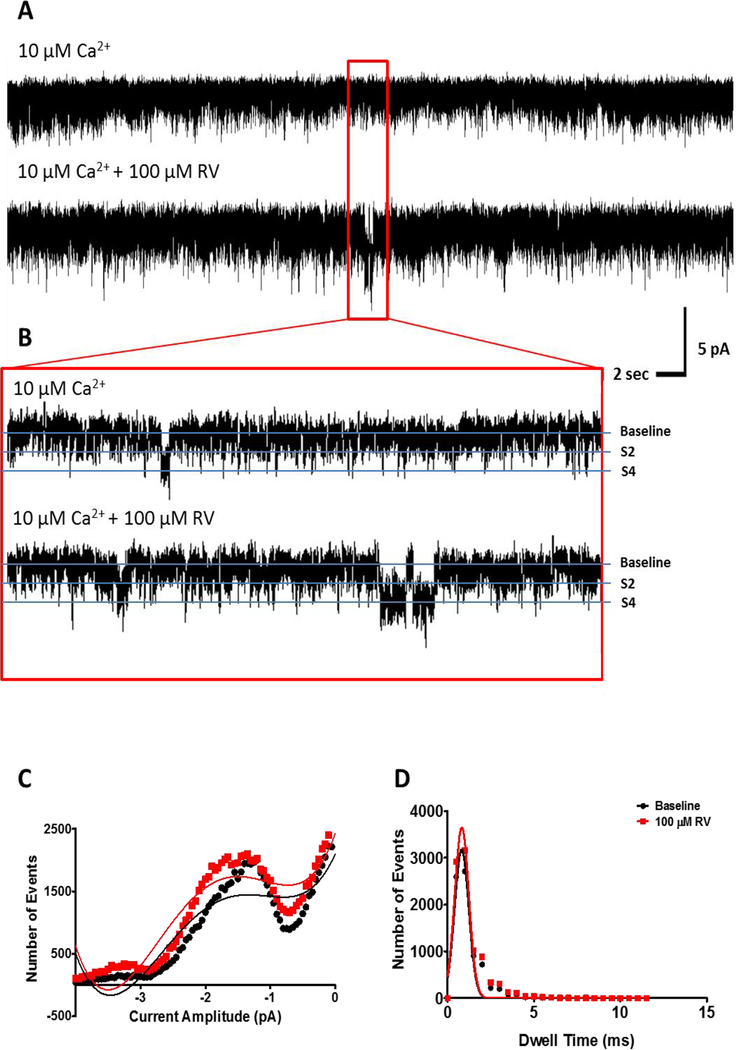
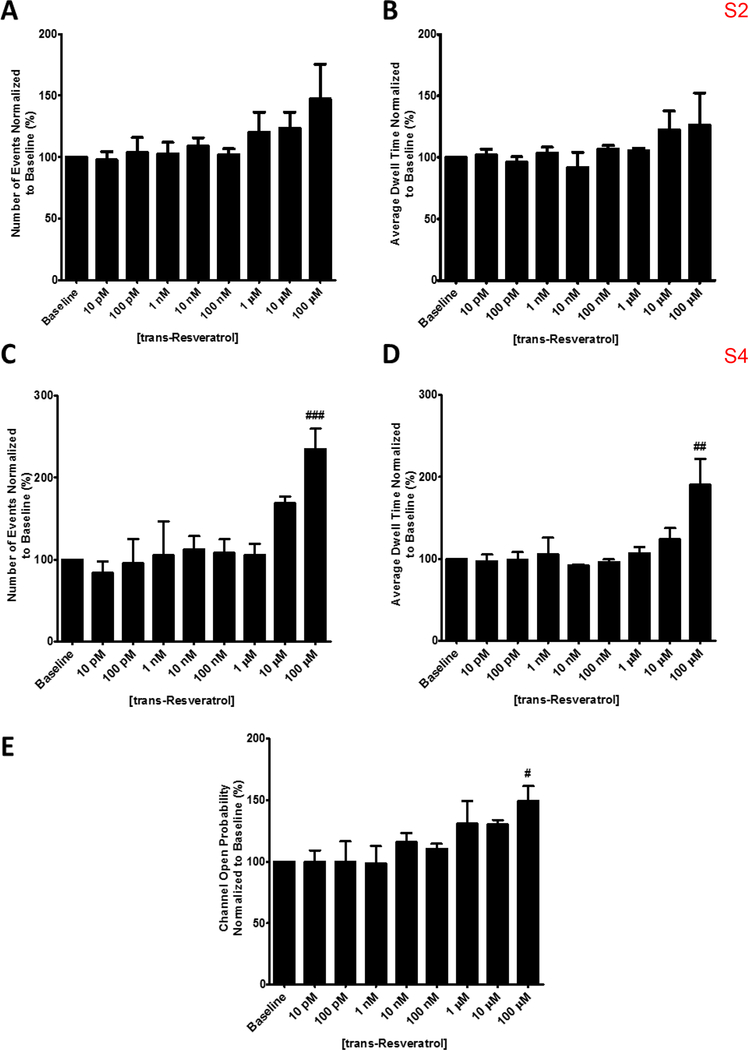
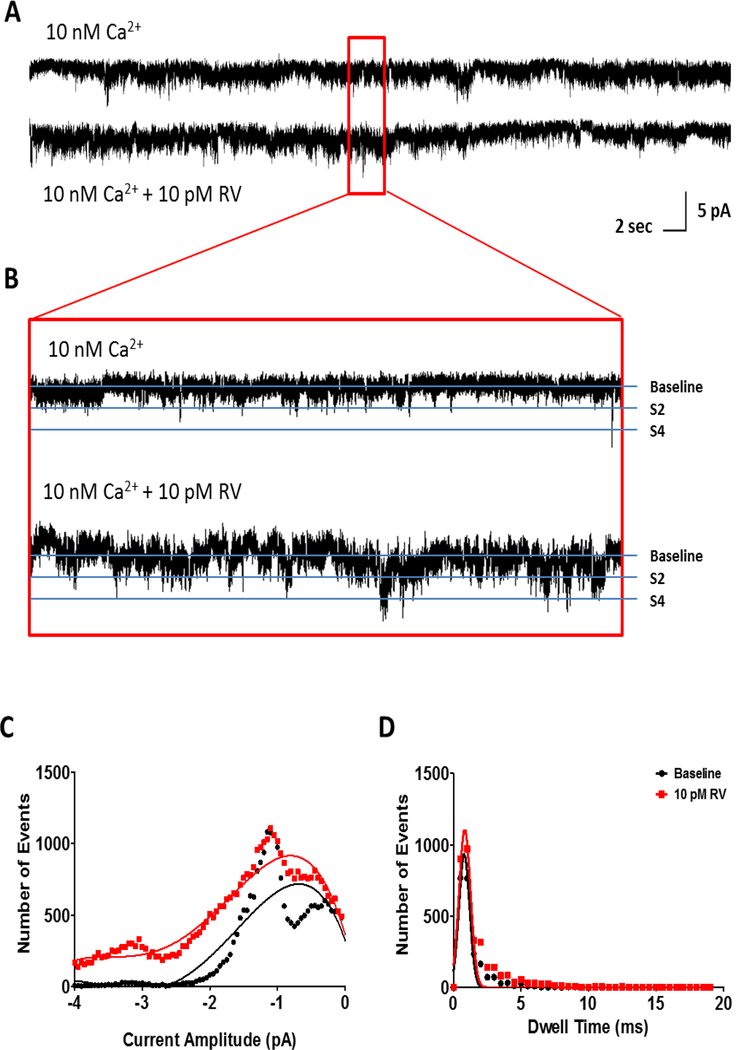
At high cytosolic Ca2+ concentrations (10 μM, pCa 5), 100 μM trans-resveratrol potentiates brain RyR activity by increasing channel open probability as indicated by the increased frequency of fully open channel events (bottom traces in Fig. 3A–B) when compared to baseline activity (top traces in Fig. 3A–B). The 2 pA subconductance state (S2) of the receptor is not significantly affected by addition of trans-resveratrol as both the number and dwell time of S2 opening events do not change significantly at any of the tested trans-resveratrol concentrations (Figs. 3 and 4A–B). However, 100 μM trans-resveratrol significantly increases the number of events and the dwell time of the fully open state (S4; Figs. 3 and 4C–D). The increase in the number of S4 events (Fig. 4C) and in the S4 dwell time (Fig. 4D) appears sufficient to increase brain RyR open probability (Fig. 4E) in the absence of significant effects of trans-resveratrol on the S2 subconductance state (Fig. 4A–B).
Figure 3. Channel open probability of mouse brain ryanodine receptors increases in the presence of trans-resveratrol at elevated cytosolic Ca2+ concentrations.
(A) Representative current traces (30 s) illustrating channel openings before (top trace) and after (bottom trace) the addition of 100 μM trans-resveratrol. (B) Short (2 sec) current traces illustrate channel dwell time at baseline (top) and in the presence of 100 μM trans-resveratrol (bottom). (C) Representative amplitude histogram showing a broadening of the S2 subconductance level and an increase in fully open S4 events, indicating an increase in the number of ryanodine receptor openings in the presence of 100 μM trans-resveratrol (red) compared to baseline (black) at 10 μM Ca2+ / pCa 5. A fifth order polynomial distribution curve was fitted for baseline conditions (R2 = 0.899; pCa 5) and for channels recorded in the presence of 100 μM trans-resveratrol (R2 = 0.952; pCa 5) showing an upward shift indicative of elevated channel opening frequency. (D) Gaussian fitting (baseline, R2 = 0.937; 100 μM trans-resveratrol, R2 = 0.928) of the channel dwell time data from the same representative data set as in C indicates that average channel dwell time remains unchanged in the presence of trans-resveratrol (red).
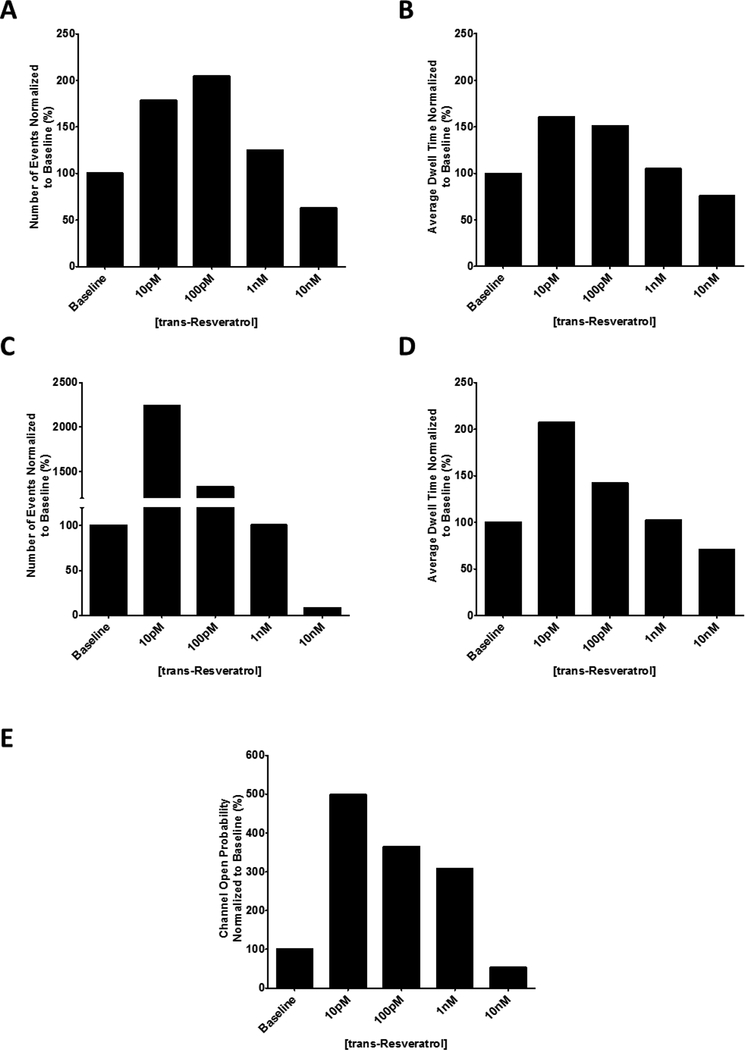
Figure 4. Quantification of brain ryanodine receptor channel open frequency, dwell time and open probability in the presence of varying concentrations of trans-resveratrol at pCa 5 / 10 μM Ca2+.
Normalized channel open frequency (A) and normalized average dwell time (B) for subconductance level S2 indicating no change over baseline at all concentrations of trans-resveratrol tested. 100 μM trans-resveratrol increases the channel open frequency (C) and average dwell time (D) of the fully open conductance state S4 by 134.9% and 90.2% respectively. (E) The open probability of the brain ryanodine receptor increases by 49.3% in the presence of 100 μM trans-resveratrol. All values were normalized to baseline and are averages ± standard error of the mean. One-way ANOVA with Dunnett’s post hoc test; #, p < 0.05; ##, p < 0.01; ###, p < 0.001; n=3.
Open probability of brain ryanodine receptors is elevated in the presence of trans-resveratrol at resting baseline cytosolic Ca2+ levels
At low intracellular Ca2+ concentrations (10 nM; pCa 8), trans-resveratrol potentiated RyR activity by increasing channel open probability. Representative 30 s (Fig. 5A) and 2 s (Fig. 5B) current traces show an increase in channel open frequency for both the S2 and S4 states in the presence of 10 pM trans-resveratrol as the underlying mechanism of action. The upward shift of the amplitude histogram curve (Fig. 5C) of 78% for the S2 and approximately 2100% for the S4 peaks, when compared to baseline (Figs. 5C, 6A and C), indicates that channel open frequency drives the trans-resveratrol-mediated increase in channel open probability. The rightward shift of the dwell time histogram curve (Fig. 5D) is indicative of an increase in channel open time. The S2 channel openings exhibit a 60% increase in duration (Fig. 6B), whereas the open dwell time of the fully open state S4 doubles when compared to baseline conditions (Fig. 6D). The combined increase in S2 and S4 opening frequency and dwell time results in an approximately fourfold increase in open probability of the channel in the presence of 10 pM trans-resveratrol (Fig. 6E).
Figure 5. Trans-resveratrol potentiates brain ryanodine receptor activity through increased channel opening frequency and dwell time at low cytosolic Ca2+ levels.
Representative 30 s (A) and 2 s (B) ryanodine receptor channel recordings at baseline conditions (10 nM Ca2+, pCa 8, top) and after addition of 10 pM trans-resveratrol (bottom). (C) Representative amplitude histograms at pCa 8 (black) and pCa 8 in the presence of 10 pM trans-resveratrol (red) indicating that trans-resveratrol increases the number of fully open S4 events and 2 pA subconductance state S2 events. A fourth order polynomial distribution was fitted for baseline conditions (pCa 8; R2 = 0.782) and for channels recorded in the presence of 10 pM trans-resveratrol (pCa 8; R2 = 10.922) displaying an upward shift for both S2 and S4 states indicative of elevated channel opening frequency. (D) Gaussian fitting (baseline, R2 = 0.946; 10 pM trans-resveratrol, R2 = 0.895) of the channel dwell time data from the same representative data set as in C shows that average channel dwell time increases significantly after trans-resveratrol administration as indicated by a rightward shift of the curve.
Figure 6. Quantification of brain ryanodine receptor channel open frequency, dwell time and open probability in the presence of varying concentrations of trans-resveratrol at pCa 8 / 10 nM Ca2+.
Normalized channel open frequency (A) and normalized average dwell time (B) for subconductance level S2 indicating potentiation by 10 pM trans-resveratrol (78% and 60% respectively) of a representative response. 10 pM trans-resveratrol significantly increases the channel open frequency (C) and average dwell time (D) of the fully open conductance state, S4, of a representative channel by approximately 2100% and 1200% respectively. (E) The open probability of a representative brain ryanodine receptor increases by approximately 400% in the presence of 10 pM trans-resveratrol.
Discussion
Nerve cells affected by neurodegenerative diseases display a decreased capacity to handle elevated ROS generation and Ca2+ signaling dyshomeostasis (28, 32–33, 79). The discovery of novel therapeutically active, pleiotropic agents, such as small molecules or biologics that have a dual function by regulating Ca2+ signaling and at the same time increasing neuronal resistance to oxidative stress, could lead to novel therapeutic approaches for neurodegenerative diseases. Here, we identified such a novel mechanism of action controlling neuronal Ca2+ signaling for the well-characterized antioxidant polyphenolic phytostilbene, trans-resveratrol.
Trans-resveratrol directly controls intracellular calcium signaling by modulating ryanodine receptor function
Trans-resveratrol directly interacts with K+ channels as determined previously in single channel electrophysiology planar lipid bilayer and patch-clamp studies (7–12). A trans-resveratrol binding site has been identified on the cytosolic sulfonylurea receptor subunit of KATP channels, while another binding site has been postulated to exist at or near the ion pore forming region of K+ channels (11–12). Structurally, K+ channels share direct homology with RyRs in the ion pore region (73–75). Taken together, these structural and sequence homologies among RyRs and K+ channels provide a rationale for trans-resveratrol to directly bind to brain RyRs and subsequently, to alter their activity. In the present study, we determined, using single channel electrophysiology, that trans-resveratrol increases the open probability of brain RyR channels in a dose- and Ca2+ dependent manner (Figs. 4 and 6). At elevated intracellular Ca2+ concentrations (pCa 5) and high, micromolar concentrations of trans-resveratrol, the open probability of RyRs increases due to a higher opening frequency and increased dwell time of the channel in the fully open S4 subconductance state (Figs. 3–4). However, a higher open probability was already observed in the presence of 10 nM trans-resveratrol reaching significance (p < 0.05) at 100 μM (Fig. 4E). This direct interaction implies that trans-resveratrol binding to brain RyRs increases the channel’s open probability by favoring its fully open S4 subconductance state thus increasing the amount of Ca2+ released from the ER into the cytosol. At resting cytosolic Ca2+ concentrations (pCa 8), trans-resveratrol significantly potentiates RyR channel opening frequency (Figs. 5C, 6A and C) and dwell times (Figs. 5D, 6B and D) for both S2 and S4 subconductance states, unlike the pharmacological control of predominantly only the fully open S4 subconductance state at higher cytosolic Ca2+ concentrations (pCa 5). Consequently, a significantly increased open probability was already seen at low trans-resveratrol concentrations (10 pM), an effect that is attenuated as trans-resveratrol concentrations increase with intermediate trans-resveratrol concentrations (10 nM) being similar to activity measured at pCa 5 (Fig. 6E).
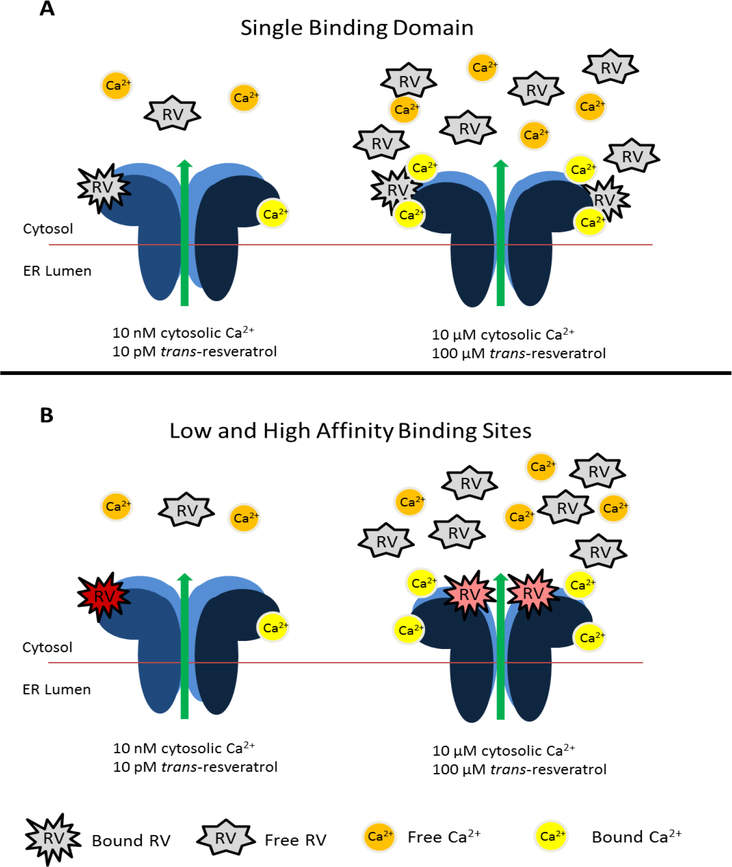
Our data provide evidence for two potential mechanisms of action of trans-resveratrol summarized and illustrated in figure 7. Assuming a single binding domain for trans-resveratrol on the RyR, such a site is accessible under low cytosolic Ca2+ concentrations when conformational changes of the ryanodine receptor due to Ca2+ binding on the cytosolic face of the channel are minimal (Fig. 7A, left panel). However, as cytosolic Ca2+ concentrations increase conformational changes of the ryanodine receptor become more extensive resulting in either reduced accessibility or reduced affinity of the trans-resveratrol binding domain for trans-resveratrol, thereby requiring higher trans-resveratrol concentrations to potentiate RyR activity (Fig. 7A, right panel).
Figure 7. Potential mechanisms of action underlying trans-resveratrol binding to and control of brain ryanodine receptors.
(A) A single trans-resveratrol (RV) binding domain is accessible at low cytosolic Ca2+concentrations (left) resulting in minimal RyR conformational change (dark blue subunit) allowing low concentrations of trans-resveratrol to potentiate RyR activity. Higher concentrations of trans-resveratrol are needed to potentiate RyR activity when significant conformational changes due to Ca2+ binding at high cytosolic Ca2+concentrations (right). (B) The bottom panel illustrates regulation of the RyR by two separate binding domains for trans-resveratrol. The high affinity site (red, left) is accessible at resting Ca2+ concentrations, but is blocked when Ca2+ binds the receptor and induces a conformational change. The conformational change of the RyR at high cytosolic Ca2+concentrations uncovers a low affinity binding site for trans-resveratrol (pink, right), requiring higher concentrations of trans-resveratrol for binding and subsequent potentiation of RyR activity. The locations of each site are hypothetical and do not imply identified structural domains.
A second potential mechanism of action of trans-resveratrol assumes two binding sites for trans-resveratrol on the RyR (Fig. 7B): At 10 pM trans-resveratrol concentrations, direct binding of trans-resveratrol to the RyR results in potentiation of channel activity through a single high affinity binding site that is accessible at resting, low cytosolic Ca2+ concentrations (pCa 8; Fig. 7B, left panel). At higher intracellular Ca2+ concentrations typically seen during high neuronal activity (pCa 5; Fig. 7B, right panel), the RyR undergoes conformational changes induced by Ca2+ binding (6, 80–83), one or more of these conformational changes thereby allowing trans-resveratrol to access a now exposed second low affinity binding site, but no longer the high affinity trans-resveratrol binding site. This potential pharmacological regulation of the RyR by trans-resveratrol assuming two different affinity-driven binding sites that cannot be accessed by trans-resveratrol simultaneously, a high affinity binding site that is only accessible at low Ca2+ concentrations and a low affinity binding site becoming accessible at higher Ca2+ concentrations, is illustrated in the summary diagram of Fig. 7B.
For both models of the newly discovered mechanism of action that direct binding of trans-resveratrol to the RyR potentiates channel activity presented in Fig. 7, trans-resveratrol increases Ca2+ release from the ER at the single channel level when cytoplasmic Ca2+ concentrations are clamped to specific levels when Ca2+-mediated RyR inhibition resulting from the channel activity itself cannot be observed. As RyRs are CICR channels with inhibitory, low affinity Ca2+ binding sites, the highly localized increase in cytosolic Ca2+ release from the ER resulting from trans-resveratrol mediated potentiation of channel activity potentially results in Ca2+ transients with above average amplitudes and shorter duration followed by a prolonged period of reduced Ca2+ leakage from the ER, all of which are hallmarks of healthy neuronal signaling (34). The total amount of Ca2+ released from the ER would be lower after trans-resveratrol treatment giving rise to potential therapeutic applications to attenuate Ca2+ homeostasis in chronic or acute neurodegenerative diseases (34).
Therapeutic limitations of trans-resveratrol lie in its limited oral bioavailability and short half-life in vivo. Single 25 mg oral doses of trans-resveratrol in healthy male subjects achieved low concentrations (~30 nM) of free trans-resveratrol in plasma between 0.5 – 2 h after dosing while conjugated trans-resveratrol peaked at 2 μM independent of the route of administration (oral or intraperitoneal; 51, 84–85). Continuous oral administration or an increased dosage improved free trans-resveratrol plasma levels in human subjects (86–88). Synthesizing methylated analogs of trans-resveratrol has been an approach to increase bioavailability. Rodent studies with pterostilbene and 3,5,4’-trimethoxy-resveratrol report plasma levels 60% and 90% greater than the trans-resveratrol control respectively, concluding that chemical modification of trans-resveratrol is a viable option for improving its bioavailability (89–90). Improving bioavailability or delivery techniques by reducing its clearance rate, increasing transport across the blood brain barrier, and/or establishing targeted delivery could enhance trans-resveratrol’s neuroprotective potential even further, ultimately contributing to a novel therapeutic approach or supplementary treatment approach for diseases affecting the nervous system.
Supplementary Material
Acknowledgements
Research reported in this publication was supported in part by grants from the National Eye Institute (EY014227 and EY022774), the Institute on Aging (AG027956), the National Center for Research Resources and National Institute of General Medical Sciences (RR027093) of the National Institutes of Health (PK). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Additional support by the Felix and Carmen Sabates Missouri Endowed Chair in Vision Research and a Challenge Grant from Research to Prevent Blindness (PK) is gratefully acknowledged.
Footnotes
Publisher's Disclaimer: This Author Accepted Manuscript is a PDF file of a an unedited peer-reviewed manuscript that has been accepted for publication but has not been copyedited or corrected. The official version of record that is published in the journal is kept up to date and so may therefore differ from this version.
References
- 1.Duncan RS; Goad DL; Grillo MA; Kaja S; Payne AJ; Koulen P, Control of Intracellular Calcium Signaling as a Neuroprotective Strategy. Molecules 2010, 15 (3), 1168–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.LaFerla FM, Calcium Dyshomeostasis and Intracellular Signalling in Alzheimer’s Disease . Nat. Rev. Neurosci 2002, 3 (11), 862–72. [DOI] [PubMed] [Google Scholar]
- 3.Niittykoski M; Kalesnykas G; Larsson KP; Kaarniranta K; Akerman KE; Uusitalo H, Altered Calcium Signaling in an Experimental Model of Glaucoma. Invest. Ophthalmol. Vis. Sci 2010, 51 (12), 6387–93. [DOI] [PubMed] [Google Scholar]
- 4.Bruno AM; Huang JY; Bennett DA; Marr RA; Hastings ML; Stutzmann GE, Altered Ryanodine Receptor Expression in Mild Cognitive Impairment and Alzheimer’s Disease. Neurobiol. Aging 2012, 33 (5), 1001 e1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Stutzmann GE; Caccamo A; LaFerla FM; Parker I, Dysregulated Ip3 Signaling in Cortical Neurons of Knock-in Mice Expressing an Alzheimer’s-Linked Mutation in Presenilin1 Results in Exaggerated Ca2+ Signals and Altered Membrane Excitability. J. Neurosci 2004, 24 (2), 508–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lanner JT; Georgiou DK; Joshi AD; Hamilton SL, Ryanodine Receptors: Structure, Expression, Molecular Details, and Function in Calcium Release. Cold Spring Harb. Perspect. Biol 2010, 2 (11), a003996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wang YJ; Chan MH; Chen L; Wu SN; Chen HH, Resveratrol Attenuates Cortical Neuron Activity: Roles of Large Conductance Calcium-Activated Potassium Channels and Voltage-Gated Sodium Channels. J. Biomed. Sci 2016, 23 (1), 47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.McCalley AE; Kaja S; Payne AJ; Koulen P, Resveratrol and Calcium Signaling: Molecular Mechanisms and Clinical Relevance. Molecules 2014, 19 (6), 7327–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Novakovic R; Radunovic N; Markovic-Lipkovski J; Cirovic S; Beleslin-Cokic B; Ilic B; Ivkovic B; Heinle H; Zivanovic V; Gojkovic-Bukarica LJ, Effects of the Polyphenol Resveratrol on Contractility of Human Term Pregnant Myometrium. Mol. Hum. Reprod 2015, 21 (6), 545–51. [DOI] [PubMed] [Google Scholar]
- 10.Li HF; Chen SA; Wu SN, Evidence for the Stimulatory Effect of Resveratrol on Ca(2+)-Activated K+ Current in Vascular Endothelial Cells. Cardiovasc. Res 2000, 45 (4), 1035–45. [DOI] [PubMed] [Google Scholar]
- 11.Gopalakrishnan M; Molinari EJ; Shieh CC; Monteggia LM; Roch JM; Whiteaker KL; Scott VE; Sullivan JP; Brioni JD, Pharmacology of Human Sulphonylurea Receptor Sur1 and Inward Rectifier K(+) Channel Kir6.2 Combination Expressed in Hek-293 Cells. Br. J. Pharmacol 2000, 129 (7), 1323–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hambrock A; de Oliveira Franz CB; Hiller S; Grenz A; Ackermann S; Schulze DU; Drews G; Osswald H, Resveratrol Binds to the Sulfonylurea Receptor (Sur) and Induces Apoptosis in a Sur Subtype-Specific Manner. J. Biol. Chem 2007, 282 (5), 3347–56. [DOI] [PubMed] [Google Scholar]
- 13.Brini M; Cali T; Ottolini D; Carafoli E, Neuronal Calcium Signaling: Function and Dysfunction. Cell. Mol. Life Sci 2014, 71 (15), 2787–814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Berridge MJ, Neuronal Calcium Signaling. Neuron 1998, 21 (1), 13–26. [DOI] [PubMed] [Google Scholar]
- 15.Contreras L; Drago I; Zampese E; Pozzan T, Mitochondria: The Calcium Connection. Biochim. Biophys. Acta 2010, 1797 (6–7), 607–18. [DOI] [PubMed] [Google Scholar]
- 16.Rizzuto R; De Stefani D; Raffaello A; Mammucari C, Mitochondria as Sensors and Regulators of Calcium Signalling. Nat. Rev. Mol. Cell Biol 2012, 13 (9), 566–78. [DOI] [PubMed] [Google Scholar]
- 17.Berridge MJ; Bootman MD; Roderick HL, Calcium Signalling: Dynamics, Homeostasis and Remodelling. Nat. Rev. Mol. Cell Biol 2003, 4 (7), 517–29. [DOI] [PubMed] [Google Scholar]
- 18.Clapham DE, Calcium Signaling. Cell 2007, 131 (6), 1047–58. [DOI] [PubMed] [Google Scholar]
- 19.Altschafl BA; Beutner G; Sharma VK; Sheu SS; Valdivia HH, The Mitochondrial Ryanodine Receptor in Rat Heart: A Pharmaco-Kinetic Profile. Biochim. Biophys. Acta 2007, 1768 (7), 1784–95. [DOI] [PubMed] [Google Scholar]
- 20.Beutner D; Voets T; Neher E; Moser T, Calcium Dependence of Exocytosis and Endocytosis at the Cochlear Inner Hair Cell Afferent Synapse. Neuron 2001, 29 (3), 681–90. [DOI] [PubMed] [Google Scholar]
- 21.Blaustein MP; Juhaszova M; Golovina VA; Church PJ; Stanley EF, Na/Ca Exchanger and Pmca Localization in Neurons and Astrocytes: Functional Implications. Ann. N. Y. Acad. Sci 2002, 976, 356–66. [DOI] [PubMed] [Google Scholar]
- 22.De Stefani D; Patron M; Rizzuto R, Structure and Function of the Mitochondrial Calcium Uniporter Complex. Biochim. Biophys. Acta 2015, 1853 (9), 2006–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hashambhoy YL; Greenstein JL; Winslow RL, Role of Camkii in Ryr Leak, Ec Coupling and Action Potential Duration: A Computational Model. J. Mol. Cell. Cardiol 2010, 49 (4), 617–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chelu MG; Sarma S; Sood S; Wang S; van Oort RJ; Skapura DG; Li N; Santonastasi M; Muller FU; Schmitz W; Schotten U; Anderson ME; Valderrabano M; Dobrev D; Wehrens XH, Calmodulin Kinase Ii-Mediated Sarcoplasmic Reticulum Ca2+ Leak Promotes Atrial Fibrillation in Mice. J. Clin. Invest 2009, 119 (7), 1940–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kook SY; Jeong H; Kang MJ; Park R; Shin HJ; Han SH; Son SM; Song H; Baik SH; Moon M; Yi EC; Hwang D; Mook-Jung I, Crucial Role of Calbindin-D28k in the Pathogenesis of Alzheimer’s Disease Mouse Model. Cell Death Differ 2014, 21 (10), 1575–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Baimbridge KG; Celio MR; Rogers JH, Calcium-Binding Proteins in the Nervous System. Trends Neurosci 1992, 15 (8), 303–8. [DOI] [PubMed] [Google Scholar]
- 27.Heizmann CW; Braun K, Changes in Ca(2+)-Binding Proteins in Human Neurodegenerative Disorders. Trends Neurosci 1992, 15 (7), 259–64. [DOI] [PubMed] [Google Scholar]
- 28.Wojda U; Salinska E; Kuznicki J, Calcium Ions in Neuronal Degeneration. IUBMB Life 2008, 60 (9), 575–90. [DOI] [PubMed] [Google Scholar]
- 29.Stull JT, Ca2+-Dependent Cell Signaling through Calmodulin-Activated Protein Phosphatase and Protein Kinases Minireview Series. J. Biol. Chem 2001, 276 (4), 2311–2. [DOI] [PubMed] [Google Scholar]
- 30.Verkhratsky A; Petersen OH, The Endoplasmic Reticulum as an Integrating Signalling Organelle: From Neuronal Signalling to Neuronal Death. Eur. J. Pharmacol 2002, 447 (2–3), 141–54. [DOI] [PubMed] [Google Scholar]
- 31.Koulen P; Janowitz T; Johenning FW; Ehrlich BE, Characterization of the Calcium-Release Channel/Ryanodine Receptor from Zebrafish Skeletal Muscle. J. Membr. Biol 2001, 183 (3), 155–63. [DOI] [PubMed] [Google Scholar]
- 32.Marambaud P; Dreses-Werringloer U; Vingtdeux V, Calcium Signaling in Neurodegeneration. Mol. Neurodegener 2009, 4, 20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mattson MP, Calcium and Neurodegeneration. Aging Cell 2007, 6 (3), 337–50. [DOI] [PubMed] [Google Scholar]
- 34.Thibault O; Gant JC; Landfield PW, Expansion of the Calcium Hypothesis of Brain Aging and Alzheimer’s Disease: Minding the Store. Aging Cell 2007, 6 (3), 307–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gennarelli TA; Graham DI, Neuropathology of the Head Injuries. Semin. Clin. Neuropsychiatry 1998, 3 (3), 160–175. [PubMed] [Google Scholar]
- 36.Foster TC, Calcium Homeostasis and Modulation of Synaptic Plasticity in the Aged Brain. Aging Cell 2007, 6 (3), 319–25. [DOI] [PubMed] [Google Scholar]
- 37.Brewer LD; Thibault O; Staton J; Thibault V; Rogers JT; Garcia-Ramos G; Kraner S; Landfield PW; Porter NM, Increased Vulnerability of Hippocampal Neurons with Age in Culture: Temporal Association with Increases in Nmda Receptor Current, Nr2a Subunit Expression and Recruitment of L-Type Calcium Channels. Brain Res 2007, 1151, 20–31. [DOI] [PubMed] [Google Scholar]
- 38.Murchison D; Griffith WH, Calcium Buffering Systems and Calcium Signaling in Aged Rat Basal Forebrain Neurons. Aging Cell 2007, 6 (3), 297–305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Toescu EC; Verkhratsky A, The Importance of Being Subtle: Small Changes in Calcium Homeostasis Control Cognitive Decline in Normal Aging. Aging Cell 2007, 6 (3), 267–73. [DOI] [PubMed] [Google Scholar]
- 40.Buchholz JN; Behringer EJ; Pottorf WJ; Pearce WJ; Vanterpool CK, Age-Dependent Changes in Ca2+ Homeostasis in Peripheral Neurones: Implications for Changes in Function. Aging Cell 2007, 6 (3), 285–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Alexianu ME; Ho BK; Mohamed AH; La Bella V; Smith RG; Appel SH, The Role of Calcium-Binding Proteins in Selective Motoneuron Vulnerability in Amyotrophic Lateral Sclerosis. Ann. Neurol 1994, 36 (6), 846–58. [DOI] [PubMed] [Google Scholar]
- 42.Iacopino AM; Christakos S, Corticosterone Regulates Calbindin-D28k Mrna and Protein Levels in Rat Hippocampus. J. Biol. Chem 1990, 265 (18), 10177–80. [PubMed] [Google Scholar]
- 43.Hakamata Y; Nakai J; Takeshima H; Imoto K, Primary Structure and Distribution of a Novel Ryanodine Receptor/Calcium Release Channel from Rabbit Brain. FEBS Lett 1992, 312 (2–3), 229–35. [DOI] [PubMed] [Google Scholar]
- 44.Rybalchenko V; Grillo MA; Gastinger MJ; Rybalchenko N; Payne AJ; Koulen P, The Unliganded Long Isoform of Estrogen Receptor Beta Stimulates Brain Ryanodine Receptor Single Channel Activity Alongside with Cytosolic Ca2+. J. Recept. Signal Transduct. Res 2009, 29 (6), 326–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Koulen P; Thrower EC, Pharmacological Modulation of Intracellular Ca(2+) Channels at the Single-Channel Level. Mol. Neurobiol 2001, 24 (1–3), 65–86. [DOI] [PubMed] [Google Scholar]
- 46.Zhang L; Kelley J; Schmeisser G; Kobayashi YM; Jones LR, Complex Formation between Junctin, Triadin, Calsequestrin, and the Ryanodine Receptor. Proteins of the Cardiac Junctional Sarcoplasmic Reticulum Membrane. J. Biol. Chem 1997, 272 (37), 23389–97. [DOI] [PubMed] [Google Scholar]
- 47.Fill M; Copello JA, Ryanodine Receptor Calcium Release Channels. Physiol. Rev 2002, 82 (4), 893–922. [DOI] [PubMed] [Google Scholar]
- 48.Copello JA; Barg S; Sonnleitner A; Porta M; Diaz-Sylvester P; Fill M; Schindler H; Fleischer S, Differential Activation by Ca2+, Atp and Caffeine of Cardiac and Skeletal Muscle Ryanodine Receptors after Block by Mg2+. J. Membr. Biol 2002, 187 (1), 51–64. [DOI] [PubMed] [Google Scholar]
- 49.Sonnleitner A; Fleischer S; Schindler H, Gating of the Skeletal Calcium Release Channel by Atp Is Inhibited by Protein Phosphatase 1 but Not by Mg2+. Cell Calcium 1997, 21 (4), 283–90. [DOI] [PubMed] [Google Scholar]
- 50.Xu L; Mann G; Meissner G, Regulation of Cardiac Ca2+ Release Channel (Ryanodine Receptor) by Ca2+, H+, Mg2+, and Adenine Nucleotides under Normal and Simulated Ischemic Conditions. Circ. Res 1996, 79 (6), 1100–9. [DOI] [PubMed] [Google Scholar]
- 51.Bastianetto S; Menard C; Quirion R, Neuroprotective Action of Resveratrol. Biochim. Biophys. Acta 2015, 1852 (6), 1195–201. [DOI] [PubMed] [Google Scholar]
- 52.Marambaud P; Zhao H; Davies P, Resveratrol Promotes Clearance of Alzheimer’s Disease Amyloid-Beta Peptides. J. Biol. Chem 2005, 280 (45), 37377–82. [DOI] [PubMed] [Google Scholar]
- 53.Huang TC; Lu KT; Wo YY; Wu YJ; Yang YL, Resveratrol Protects Rats from Abeta-Induced Neurotoxicity by the Reduction of Inos Expression and Lipid Peroxidation. PLoS One 2011, 6 (12), e29102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Rege SD; Geetha T; Griffin GD; Broderick TL; Babu JR, Neuroprotective Effects of Resveratrol in Alzheimer Disease Pathology. Front. Aging Neurosci 2014, 6, 218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Renaud S; de Lorgeril M, Wine, Alcohol, Platelets, and the French Paradox for Coronary Heart Disease. Lancet 1992, 339 (8808), 1523–6. [DOI] [PubMed] [Google Scholar]
- 56.Wu JM; Hsieh TC; Wang Z, Cardioprotection by Resveratrol: A Review of Effects/Targets in Cultured Cells and Animal Tissues. Am. J. Cardiovasc. Dis 2011, 1 (1), 38–47. [PMC free article] [PubMed] [Google Scholar]
- 57.Liu BL; Zhang X; Zhang W; Zhen HN, New Enlightenment of French Paradox: Resveratrol’s Potential for Cancer Chemoprevention and Anti-Cancer Therapy. Cancer Biol. Ther 2007, 6 (12), 1833–6. [DOI] [PubMed] [Google Scholar]
- 58.Bastianetto S; Zheng WH; Quirion R, Neuroprotective Abilities of Resveratrol and Other Red Wine Constituents against Nitric Oxide-Related Toxicity in Cultured Hippocampal Neurons. Br. J. Pharmacol 2000, 131 (4), 711–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Buryanovskyy L; Fu Y; Boyd M; Ma Y; Hsieh TC; Wu JM; Zhang Z, Crystal Structure of Quinone Reductase 2 in Complex with Resveratrol. Biochemistry 2004, 43 (36), 11417–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Sakata Y; Zhuang H; Kwansa H; Koehler RC; Dore S, Resveratrol Protects against Experimental Stroke: Putative Neuroprotective Role of Heme Oxygenase 1. Exp. Neurol 2010, 224 (1), 325–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zhang F; Liu J; Shi JS, Anti-Inflammatory Activities of Resveratrol in the Brain: Role of Resveratrol in Microglial Activation. Eur. J. Pharmacol 2010, 636 (1–3), 1–7. [DOI] [PubMed] [Google Scholar]
- 62.Gong QH; Wang Q; Shi JS; Huang XN; Liu Q; Ma H, Inhibition of Caspases and Intracellular Free Ca2+ Concentrations Are Involved in Resveratrol Protection against Apoptosis in Rat Primary Neuron Cultures. Acta Pharmacol. Sin 2007, 28 (11), 1724–30. [DOI] [PubMed] [Google Scholar]
- 63.Wu XP; Xiong M; Xu CS; Duan LN; Dong YQ; Luo Y; Niu TH; Lu CR, Resveratrol Induces Apoptosis of Human Chronic Myelogenous Leukemia Cells in Vitro through P38 and Jnk-Regulated H2ax Phosphorylation. Acta Pharmacol. Sin 2015, 36 (3), 353–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Huang T; Gao D; Jiang X; Hu S; Zhang L; Fei Z, Resveratrol Inhibits Oxygen-Glucose Deprivation-Induced Mmp-3 Expression and Cell Apoptosis in Primary Cortical Cells Via the Nf-Kappab Pathway. Mol Med Rep 2014, 10 (2), 1065–71. [DOI] [PubMed] [Google Scholar]
- 65.Hogg SJ; Chitcholtan K; Hassan W; Sykes PH; Garrill A, Resveratrol, Acetyl-Resveratrol, and Polydatin Exhibit Antigrowth Activity against 3d Cell Aggregates of the Skov-3 and Ovcar-8 Ovarian Cancer Cell Lines. Obstet. Gynecol. Int 2015, 2015, 279591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Liao PC; Ng LT; Lin LT; Richardson CD; Wang GH; Lin CC, Resveratrol Arrests Cell Cycle and Induces Apoptosis in Human Hepatocellular Carcinoma Huh-7 Cells. J. Med. Food 2010, 13 (6), 1415–23. [DOI] [PubMed] [Google Scholar]
- 67.Varoni EM; Lo Faro AF; Sharifi-Rad J; Iriti M, Anticancer Molecular Mechanisms of Resveratrol. Front Nutr 2016, 3, 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Lee BH; Choi SH; Hwang SH; Kim HJ; Lee JH; Nah SY, Resveratrol Inhibits Gabac Rho Receptor-Mediated Ion Currents Expressed in Xenopus Oocytes. Korean J. Physiol. Pharmacol 2013, 17 (2), 175–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Lee BH; Hwang SH; Choi SH; Shin TJ; Kang J; Lee SM; Nah SY, Resveratrol Enhances 5-Hydroxytryptamine Type 3a Receptor-Mediated Ion Currents: The Role of Arginine 222 Residue in Pre-Transmembrane Domain I. Biol. Pharm. Bull 2011, 34 (4), 523–7. [DOI] [PubMed] [Google Scholar]
- 70.Yu L; Wang S; Kogure Y; Yamamoto S; Noguchi K; Dai Y, Modulation of Trp Channels by Resveratrol and Other Stilbenoids. Mol Pain 2013, 9, 3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Zhang LP; Yin JX; Liu Z; Zhang Y; Wang QS; Zhao J, Effect of Resveratrol on L-Type Calcium Current in Rat Ventricular Myocytes. Acta Pharmacol. Sin 2006, 27 (2), 179–83. [DOI] [PubMed] [Google Scholar]
- 72.Jakab M; Lach S; Bacova Z; Langeluddecke C; Strbak V; Schmidt S; Iglseder E; Paulmichl M; Geibel J; Ritter M, Resveratrol Inhibits Electrical Activity and Insulin Release from Insulinoma Cells by Block of Voltage-Gated Ca+ Channels and Swelling-Dependent Cl- Currents. Cell. Physiol. Biochem 2008, 22 (5–6), 567–78. [DOI] [PubMed] [Google Scholar]
- 73.Samso M; Wagenknecht T; Allen PD, Internal Structure and Visualization of Transmembrane Domains of the Ryr1 Calcium Release Channel by Cryo-Em. Nat. Struct. Mol. Biol 2005, 12 (6), 539–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Welch W; Rheault S; West DJ; Williams AJ, A Model of the Putative Pore Region of the Cardiac Ryanodine Receptor Channel. Biophys. J 2004, 87 (4), 2335–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Ramachandran S; Serohijos AW; Xu L; Meissner G; Dokholyan NV, A Structural Model of the Pore-Forming Region of the Skeletal Muscle Ryanodine Receptor (Ryr1). PLoS Comput. Biol 2009, 5 (4), e1000367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Kim YH; Kim YS; Kang SS; Cho GJ; Choi WS, Resveratrol Inhibits Neuronal Apoptosis and Elevated Ca2+/Calmodulin-Dependent Protein Kinase Ii Activity in Diabetic Mouse Retina. Diabetes 2010, 59 (7), 1825–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Li W; Wang YP; Gao L; Zhang PP; Zhou Q; Xu QF; Zhou ZW; Guo K; Chen RH; Yang HT; Li YG, Resveratrol Protects Rabbit Ventricular Myocytes against Oxidative Stress-Induced Arrhythmogenic Activity and Ca2+ Overload. Acta Pharmacol. Sin 2013, 34 (9), 1164–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Dong Q; Wu Z; Li X; Yan J; Zhao L; Yang C; Lu J; Deng J; Chen M, Resveratrol Ameliorates Cardiac Dysfunction Induced by Pressure Overload in Rats Via Structural Protection and Modulation of Ca(2+) Cycling Proteins. J. Transl. Med 2014, 12, 323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Kim GH; Kim JE; Rhie SJ; Yoon S, The Role of Oxidative Stress in Neurodegenerative Diseases. Exp. Neurobiol 2015, 24 (4), 325–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Efremov RG; Leitner A; Aebersold R; Raunser S, Architecture and Conformational Switch Mechanism of the Ryanodine Receptor. Nature 2015, 517 (7532), 39–43. [DOI] [PubMed] [Google Scholar]
- 81.Song DW; Lee JG; Youn HS; Eom SH; Kim DH, Ryanodine Receptor Assembly: A Novel Systems Biology Approach to 3d Mapping. Prog. Biophys. Mol. Biol 2011, 105 (3), 145–61. [DOI] [PubMed] [Google Scholar]
- 82.Ozawa T, Modulation of Ryanodine Receptor Ca2+ Channels (Review). Mol Med Rep 2010, 3 (2), 199–204. [DOI] [PubMed] [Google Scholar]
- 83.Zalk R; Clarke OB; des Georges A; Grassucci RA; Reiken S; Mancia F; Hendrickson WA; Frank J; Marks AR, Structure of a Mammalian Ryanodine Receptor. Nature 2015, 517 (7532), 44–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Goldberg DM; Yan J; Soleas GJ, Absorption of Three Wine-Related Polyphenols in Three Different Matrices by Healthy Subjects. Clin. Biochem 2003, 36 (1), 79–87. [DOI] [PubMed] [Google Scholar]
- 85.Walle T; Hsieh F; DeLegge MH; Oatis JE Jr.; Walle UK, High Absorption but Very Low Bioavailability of Oral Resveratrol in Humans. Drug Metab. Dispos 2004, 32 (12), 1377–82. [DOI] [PubMed] [Google Scholar]
- 86.Almeida L; Vaz-da-Silva M; Falcao A; Soares E; Costa R; Loureiro AI; Fernandes-Lopes C; Rocha JF; Nunes T; Wright L; Soares-da-Silva P, Pharmacokinetic and Safety Profile of Trans-Resveratrol in a Rising Multiple-Dose Study in Healthy Volunteers. Mol Nutr Food Res 2009, 53 Suppl 1, S7–15. [DOI] [PubMed] [Google Scholar]
- 87.Walle T, Bioavailability of Resveratrol. Ann. N. Y. Acad. Sci 2011, 1215, 9–15. [DOI] [PubMed] [Google Scholar]
- 88.Vitaglione P; Sforza S; Galaverna G; Ghidini C; Caporaso N; Vescovi PP; Fogliano V; Marchelli R, Bioavailability of Trans-Resveratrol from Red Wine in Humans. Mol Nutr Food Res 2005, 49 (5), 495–504. [DOI] [PubMed] [Google Scholar]
- 89.Kapetanovic IM; Muzzio M; Huang Z; Thompson TN; McCormick DL, Pharmacokinetics, Oral Bioavailability, and Metabolic Profile of Resveratrol and Its Dimethylether Analog, Pterostilbene, in Rats. Cancer Chemother. Pharmacol 2011, 68 (3), 593–601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Lin HS; Ho PC, A Rapid Hplc Method for the Quantification of 3,5,4’-Trimethoxy-Trans-Stilbene (Tms) in Rat Plasma and Its Application in Pharmacokinetic Study. J. Pharm. Biomed. Anal 2009, 49 (2), 387–92. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.