Abstract
We previously demonstrated that auraptene (AUR), a natural coumarin derived from citrus plants, exerts anti-inflammatory effects in the brain, resulting in neuroprotection in some mouse models of brain disorders. The present study showed that treatment with AUR significantly increased the release of glial cell line-derived neurotrophic factor (GDNF), in a dose- and time-dependent manner, by rat C6 glioma cells, which release was associated with increased expression of GDNF mRNA. These results suggest that AUR acted as a neuroprotective agent in the brain via not only its anti-inflammatory action but also its induction of neurotrophic factor. We also showed that (1) the AUR-induced GDNF production was inhibited by U0126, a specific inhibitor of mitogen-activated protein kinase/extracellular signal-regulated kinase (ERK) 1/2, and by H89, a specific inhibitor of protein kinase A (PKA); and (2) AUR induced the phosphorylation of cAMP response element-binding protein (CREB), a transcription factor located within the nucleus. These results suggest that AUR-stimulated gdnf gene expression was up-regulated through the PKA/ERK/CREB pathway in C6 cells.
Keywords: auraptene, glial cell line-derived neurotrophic factor, GDNF, neuroprotective effect
1. Introduction
Auraptene (7-geranyloxycoumarin, AUR) is a coumarin derivative isolated from the peel of certain citrus fruits, and it is the most abundant form of naturally occurring geranyloxycoumarins. Numerous studies revealed that it has valuable pharmacological actions in peripheral tissues, such as anti-carcinogenic, anti-inflammatory, anti-Helicobacter, and anti-genotoxic ones [1,2]. We recently reported that (1) AUR can pass through the blood–brain barrier; (2) it exerts anti-inflammatory effects in the brain as well as in peripheral tissues, consequently exerting neuroprotective effects in the brain; and (3) its neuroprotective effects might be mediated by the suppression of the inflammatory response, as it effectively suppresses hyper-activation of microglia/astrocytes and hyper-expression of cyclooxygenase-2 and pro-inflammatory cytokines [3,4,5,6]. On the other hand, the fact that astrocytes have the ability to support neurons by secreting neurotrophic factors such as glial cell line-derived neurotrophic factor (GDNF), brain-derived neurotrophic factor (BDNF), and nerve growth factor have been well documented [7]. GDNF, in particular, has been reported to inhibit microglial activation [8]. Therefore, the most important object of the present study was to investigate whether AUR would have the ability to induce GDNF expression in astrocytes. As astrocytes, we used rat C6 glioma cells (rat glial tumor cell line), which have been frequently and extensively used as an alternative to astrocytes in primary culture [9,10,11,12,13], because these cells have similarities with primary astrocytes in culture [14,15] and have ability to synthesize/secrete high levels of GDNF [9,10,11].
GDNF was originally found as a trophic agent for midbrain dopamine neurons [16], and therefore the expectation was soon raised that it might be useful as a therapeutic agent to treat Parkinson’s disease (PD) [17]. In fact, many studies have shown that GDNF has the therapeutic benefits for various animal models of PD [18,19]. Later, it became clear that GDNF is expressed abundantly throughout the brain and that it also has a neuroprotective effect on noradrenergic neurons in the locus ceruleus of rodents [20,21,22].
In this study we successfully found that AUR had the ability to induce GDNF synthesis/secretion in C6 cells and then investigated the signal transduction molecule(s) at work that allowed AUR to induce gdnf gene expression.
2. Results
2.1. Effects of AUR on the Viability of C6 Cells
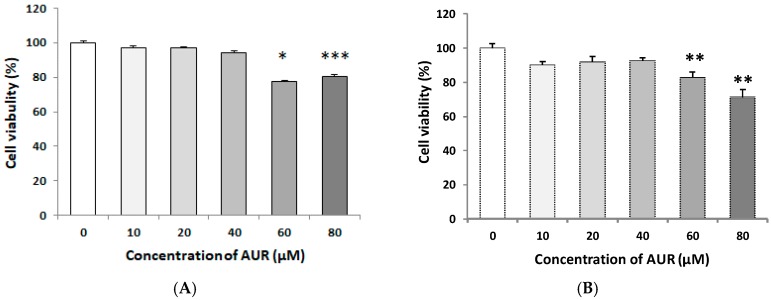
We initially evaluated the effect of 24 h-exposure to AUR on the cell viability. For this, C6 cells were seeded on a 96-well plate and cultured for 24 h in a medium containing 10% fetal bovine serum (FBS), and then treated with 10–80 μM AUR for 24 h in the same medium. Other cells on a 96-well plate were cultured for 24 h in a medium containing 10% FBS, and thereafter for another 24 h in medium containing 2% FBS. The cells were then treated with 10~80 μM AUR for 24 h in a medium containing 2% FBS. The results of MTT assay showed no differences in cell number between non-treated cells and those incubated with AUR (10–40 μM) both in medium containing 10% FBS (Figure 1A) and 2% FBS (Figure 1B). However, a decrease in cell viability was observed when the concentration of AUR was at or exceeded 60 μM in both medium. Based on these results, we chose 10–40 μM AUR for use in subsequent experiments. During the viability experiment, no apparent morphological changes (such as flattening and development of cell processes) were observed for cells treated at any of the concentrations tested.
Figure 1.
Effects of treatment with auraptene (AUR) on C6 cell viability in medium containing 10% fetal bovine serum (FBS) (A) or 2% FBS (B). Cells were treated with various concentrations (0–80 μM) of AUR for 24 h. The results are presented as the mean ± SEM (n = 4). Significance difference in values between the non-treated (0 μM) and AUR-treated cells: * p < 0.05; ** p < 0.01; *** p < 0.001.
2.2. Effects of AUR on GDNF Content of Conditioned Media
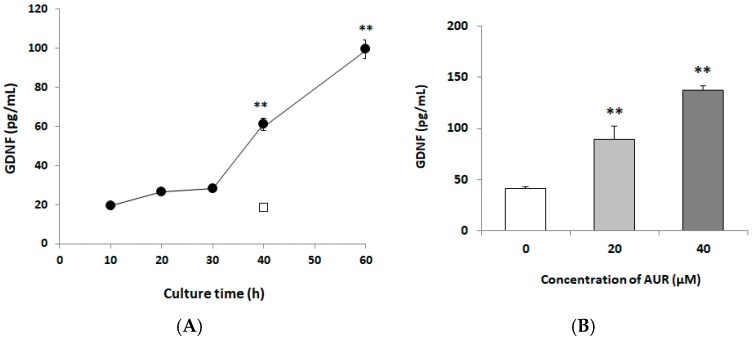
To examine the effect of AUR-treatment on the release of GDNF, we incubated C6 cells with 10 μM AUR for 0~60 h. As shown in Figure 2A, a significant increase in GDNF release by AUR was detectable at 40 h (** p < 0.01), which release remained elevated up to 60 h (** p < 0.01). To assess the concentration-dependency of AUR on the release of GDNF from C6 cells, we treated the cells with 20 or 40 μM AUR for 40 h. As shown in Figure 2B, a significant increase in GDNF release (** p < 0.01) was detectable at either concentration. These results thus showed that AUR induced GDNF release in a time-dependent and dose-dependent manner.
Figure 2.
Effects of treatment with AUR on glial cell line-derived neurotrophic factor (GDNF) content in the medium of C6 cells. (A) Cells were incubated with 10 μM AUR for various times (10–60 h) (●) or without AUR for 40 h (□). Significance difference in values between the non-treated cells (40 h) and other cells: ** p < 0.01; (B) Cells were incubated with various concentrations (0, 20, and 40 μM) of AUR for 40 h. Significance difference in values between the non-treated (0 μM) and AUR-treated cells: ** p < 0.01. In (A) and (B), the results are presented as the mean ± SEM (n = 4).
2.3. Effects of AUR on GDNF Levels in Cell Lysates
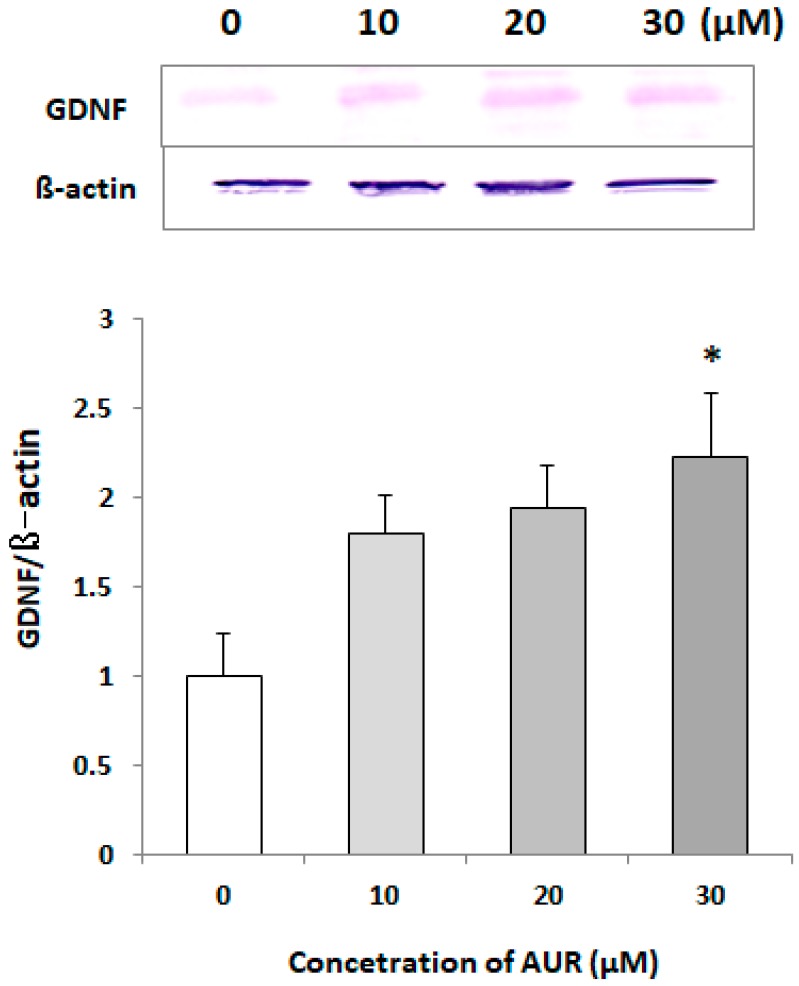
To examine the effect of treatment with AUR on GDNF expression in C6 cells, we treated them with various concentrations (0, 10, 20, and 30 μM) of AUR for 40 h. The results of immunoblot analysis (Figure 3) showed that the GDNF content in the control cell lysate was low but that significant induction occurred after 40 h of treatment with 30 μM AUR (* p < 0.05).
Figure 3.
Effects of AUR-treatment with AUR on GDNF content in C6 cells. Cells were incubated with various concentrations (0, 10, 20, and 30 μM) of AUR for 50 h. The results are presented as the mean ± SEM (n = 3). Significance difference in values between the non-treated and AUR-treated cells: * p < 0.05.
2.4. Effects of AUR on GDNF mRNA Expression in C6 Cells
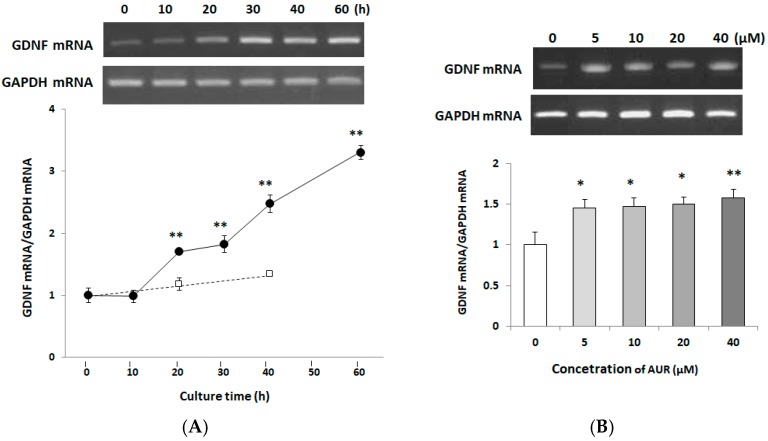
We next investigated the time course of GDNF mRNA expression in C6 cells. C6 cells were treated with 10 μM AUR for 0~60 h. Figure 4A shows that AUR started to enhance the GDNF mRNA expression at 20 h of treatment (** p < 0.01), and continued to increase it even up to 60 h. We then examined the dose-dependency of AUR on GDNF mRNA expression when the cells were treated with 5, 10, 20, or 40 μM AUR for 40 h. Figure 4B shows that GDNF mRNA significantly increased by AUR-treatment. These results showed that AUR enhanced GDNF mRNA expression in a time-dependent manner and in a somewhat dose-dependent one.
Figure 4.
Effects of treatment with AUR on GDNF mRNA content in C6 cells. (A) Cells were incubated with 10 μM AUR for various times (0–60 h) (●) or without AUR for 20 or 40 h (□). Significance difference in values between the non-treated and AUR-treated cells: ** p < 0.01; (B) Cells were incubated with various concentrations (0, 5, 10, 20, and 40 μM) of AUR for 40 h. Significance difference in values between the none-treated (0 μM) and AUR-treated cells: * p < 0.05; ** p < 0.01. In (A) and (B), the results are presented as the mean ± SEM (n = 6).
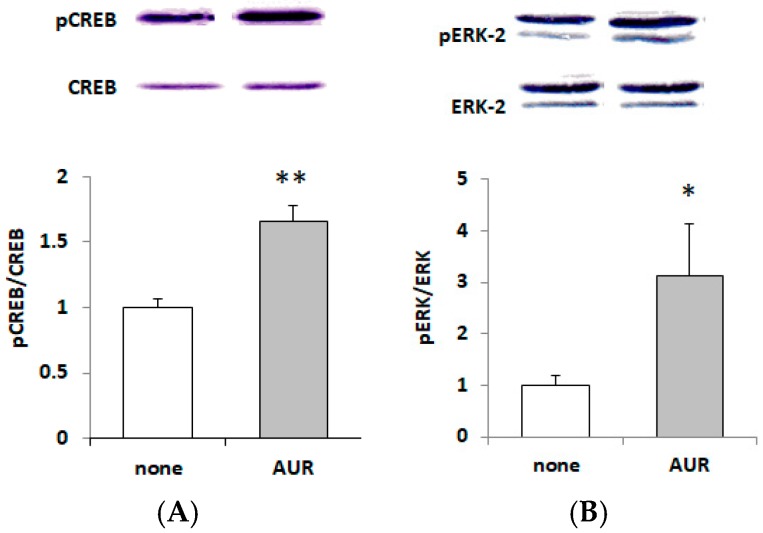
2.5. Effects of AUR on the Phosphorylation of CREB and ERK
The phosphorylation of cAMP response element-binding protein (CREB), a transcription factor located within the nucleus, has been reported to be associated with GDNF expression as well as that of BDNF [14,23]. We thus examined whether AUR could elicit phosphorylation of CREB in C6. When C6 cells were treated with 20 μM AUR for 40 h, the ratio of phosphorylated CREB (pCREB) to total CREB (CREB) was significantly (** p < 0.01) increased (Figure 5A).
Figure 5.
Effects of treatment with AUR on phosphorylation of cAMP response element-binding protein (CREB) (A) and extracellular signal-regulated kinase 2 (ERK2) (B) in C6 cells. Cells were incubated with 20 μM AUR for 40 h. The density ratio of phosphorylated components to total components of untreated cultures (none) was expressed as 1.0. The results are given as the mean ± SEM (n = 3). Significance difference in values between the none-treated and AUR-treated cells: * p < 0.05; ** p < 0.01.
As the signaling pathway involved in the regulation of GDNF expression, extracellular signal-regulated kinases 1/2 (ERK1/2), which are components of the mitogen-activated protein kinase (MAPK) signaling pathway, are well known [13]. Figure 5B shows that AUR increased the ratio of phosphorylated ERK2 (pERK2) to total ERK2 (ERK2). Among ERK isoforms (ERK1; 44 kDa and ERK2; 42 kDa), we analyzed the ratio of phosphorylated ERK2 (pERK2) to total ERK2 (ERK2), because a less important/dispensable role was reported for ERK1 [24]. The results shown in Figure 5B indicate that AUR directly and significantly (* p < 0.05) activated ERK1/2.
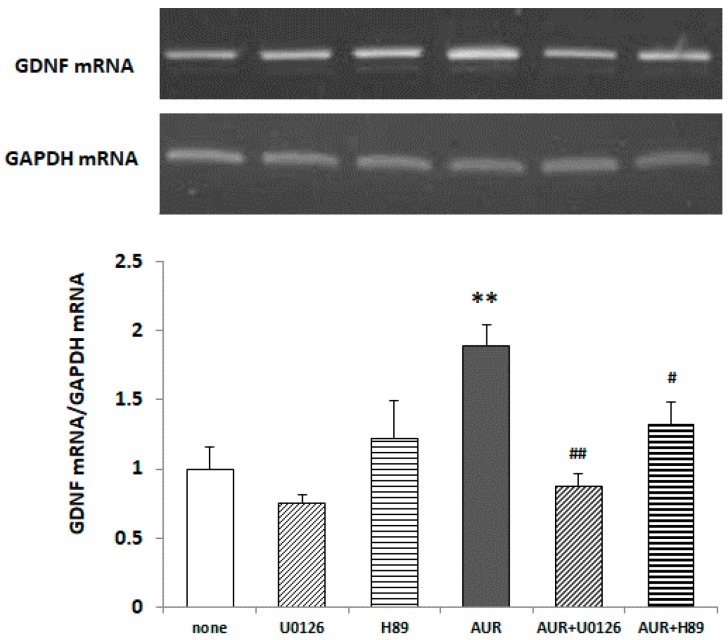
2.6. Effects of Inhibitors of ERK/PKA on AUR-Induced GDNF mRNA Expression in C6 Cells
To examine whether the phosphorylation of ERK caused by AUR was responsible for GDNF expression, we pretreated C6 cells with 10 μM U0126 (a specific inhibitor of MAPK/ERK kinase 1; MEK1) for 30 min and subsequently treated them with 20 μM AUR for 40 h. Figure 6 shows that AUR-induced GDNF mRNA expression was significantly (## p < 0.01) inhibited by U0126, indicating that the action of AUR on GDNF synthesis in astrocytes was mediated in an ERK/CREB-dependent manner.
Figure 6.
Effects of U0126 and H89 on AUR-induced GDNF mRNA content in C6 cells. Cells were preincubated with or without 10 μM U0126 or 1 μM H89 for 30 min and then incubated with 20 μM AUR for 40 h. The results are presented as the mean ± SEM (n = 4). Significance difference in values between the non-treated and AUR-treated cells: ** p < 0.05; significant difference in values between the AUR-treated and AUR/inhibitor-treated cells: # p < 0.05; ## p < 0.01.
In PC12 cells, the Ca2+-induced nuclear translocation of ERK to the nucleus requires protein kinase A (PKA) activation [25]. So we pretreated C6 cells with 1 μM H89 (a specific inhibitor of PKA) for 30 min and subsequently treated them with 20 μM AUR for 40 h. Figure 6 shows that AUR-induced GDNF mRNA expression was significantly (# p < 0.05) inhibited by H89, indicating that the action of AUR on GDNF synthesis in astrocytes was mediated in an ERK/PKA/CREB-dependent manner. Figure 6 also shows that there was no effect of either inhibitor tested alone on the basal level of GDNF mRNA.
3. Discussion
The present study revealed that AUR had the ability to induce GDNF release in C6 cells (Figure 2), and that the AUR-induced GDNF release was associated with increased expression of GDNF mRNA (Figure 4). In order to eliminate the possibility that this release of GDNF was due to a leakage of cells as a consequence of AUR’s toxicity or an increase of cell population as a consequence of AUR’s growth promoting ability, we treated cells with AUR at the concentration of 10~40 μM in the present experiments (Figure 1). In experiments using this concentration range, we did not observe any differences (viability, morphological change etc.) between none-treated cells and AUR-treated cells up to 60 h. On the other hands, recent study showed that AUR was cytotoxic against U87 cells, human malignant glioblastoma (GBM) cell line, dose- and time-dependently with IC50 values of 108.9 μg/mL (namely, 0.365 μM) and 79.17 μg/mL (namely, 0.266 μM) obtained for 24 and 48 h-treatments [26]. Considering these results and our findings suggests the possibility that AUR exerts reversible effects on U87 cells, devastating type of astrocytic tumor, and C6 cells, astrocytoma cells that resemble normal astrocytes.
We also found presently that (1) AUR had the ability to directly elicit phosphorylation of ERK1/2 and CREB (Figure 5); (2) the AUR-induced GDNF mRNA expression was significantly inhibited by U0126, a MEK1 inhibitor, and H89, a PKA inhibitor (Figure 6), suggesting that AUR induced GDNF expression was mediated by the MAPK/ERK signaling pathway and PKA activation. As the mechanism underlying the upregulation of GDNF expression, the PI3K/Akt pathway [27] and PKC pathway [28] have also been reported to be involved. As far as we examined, neither the direct phosphorylation of Akt in response to AUR nor the inhibition of induction of GDNF by LY294002 (a specific inhibitor of PI3K) was observed (data not shown). The involvement of the PKC pathway on GDNF induction is yet still uncertain. In any event, the findings here suggest that AUR could cause the induction of GDNF expression in astrocytes, at least partly, by activating the PKA/ERK/CREB pathway.
We previously reported that AUR exerts powerful neuroprotective effects in vivo in the brain, probably because it suppresses inflammatory responses in the brain of a mouse model of global cerebral ischemia [3,5] and in lipopolysaccharide (LPS)-induced systemic inflammatory model mice [4,6]. In addition, we recently reported that the peel of C. kawachiensis, a rich source of AUR, is neuroprotective in LPS-induced systemic inflammatory model mice [4], senescence-accelerated mice [29], global cerebral ischemia model mice [30], type-2 diabetic db/db mice [31], and an LPS-induced mouse model of PD [32]. We also showed that the AUR-rich fruit juice of C. kawachiensis, made by the addition of peel paste to the raw juice, significantly suppressed the ischemia-induced neuronal cell death in the hippocampus of ischemic mice [33] and that this juice contributed to the preservation of cognitive function of healthy volunteers [34]. In these reports, we confirmed that the basis of this neuroprotective ability might have been anti-inflammatory effect of AUR. The findings obtained presently might offer a novel mechanism through which AUR and the peel of C. kawachiensis may exert their neuroprotective effects, i.e., the induction of GDNF in concert with the suppression of inflammation. In other words, the present findings suggest that the induction of GDNF might be another part of the mechanism accounting for the neuroprotective effects of AUR and the peel of C. kawachiensis. We are planning to confirm the AUR-induced GDNF expression in vivo using various disease model mice.
Accumulating recent studies showed that GDNF has the ability to improve brain function and that it plays an important role in various neuropsychiatric disorders such as depression, Alzheimer’s disease (AD), and aging [35]. As for AD, (1) the administration of GDNF protects against aluminum-induced apoptosis in rabbits by up-regulating Bcl-2 and Bcl-XL and inhibiting mitochondrial Bax translocation [36]; (2) Lenti-GDNF gene therapy protects against AD-like neuropathology in 3xTg-AD mice [37]; and (3) the level of mature GDNF peptide is down-regulated in the postmortem middle temporal gyrus of AD patients [38]. A recent review stated that various phytochemicals, including bioactive compounds in food and beverages, can protect neuronal cells by neurotrophic-factor mimetic activities in the brain [39]. The present findings taken together with the above findings supports our proposition [34] that AUR-rich C. kawachiensis peels might be a functional food for aging and neurodegenerative disorders.
4. Materials and Methods
4.1. Chemical and Reagents
AUR was kindly supplied by Ushio ChemiX Corp. (Omaezaki, Shizuoka, Japan). U0126 and H89 were purchased from Calbiochem Corp. (San Diego, CA, USA). These compounds were dissolved in dimethyl sulfoxide (DMSO). The final concentration of DMSO in all culture media was below 0.1%.
4.2. Cell Culture
All cell culture materials such as Dulbecco’s modified Eagle medium (DMEM), FBS, and antibiotics were purchased from Thermo Fisher Scientific (Waltham, MA, USA) as previously described [13]. C6 cells were maintained in a medium containing 10% FBS. For the MTT assay, cells were seeded in a 96-well plate at a density of 1 × 104 cells/well. For other experiments, cells were seeded in 35-mm culture dishes at a density of 2.5 × 105 cells/dish. Cells were cultured for 24 h in medium containing 10% FBS, and then for a further 24 h in medium containing 2% FBS, after which the cells were treated for the indicated times with samples in medium containing 2% FBS.
4.3. Determination of Cell Viability
Cell viability was determined by the use of the MTT assay, as previously described [40]. All assays were performed 4 times independently.
4.4. ELISA
C6 cells in 35-mm culture dishes were treated with AUR at the desired concentrations and times. The culture supernatants were centrifuged at 2000× g for 20 min to remove particulates before analysis. GDNF contents in cell culture supernatants were measured by using the GDNF Emax® ImmunoAssay System (Promega Corp., Madison, WI, USA).
4.5. Immunoblot Analysis
Cell extracts of C6 cells grown in 35-mm culture dishes were prepared as previously described [40]. Equal amounts of proteins (20 μg for pCREB and 10 μg for others) were separated on SDS-polyacrylamide gels and electroblotted onto an Immuno-Blot™ PVDF membrane (Bio-Rad Lab., Hercules, CA, USA). As primary antibodies, rabbit polyclonal antibodies against 44/42 ERK1/2 and phospho-44/42 MAPK (Thr202/Tyr204) were purchased from Cell Signaling Tech. Inc. (Woburn, MA, USA). Rabbit polyclonal antibodies against actin and GDNF were purchased from Sigma-Aldrich Company Ltd. (St. Louis, MO, USA) and Abcam plc. (Cambridge, Cambridgeshire, UK), respectively. As secondary antibody, alkaline phosphatase-linked anti-rabbit IgG (Cell Signaling) were used. Immunoreactive bands were detected by use of the NCB/BCIP reagent (Roche Diagnotics GmbH, Mannheim, Germany).
4.6. Total RNA Extraction and RT-PCR
C6 cells in 35-mm culture dishes were treated with AUR at the desired concentrations and times. Total RNA was prepared from the cells by use of Isogen II, a reagent for extracting total RNA (Nippon Gene Co., Ltd., Tokyo, Japan) and transcribed into cDNA by using a PrimeScript™ RT-PCR Kit (Takara Bio. Inc., Kusatsu, Shiga, Japan). The synthesized cDNA was amplified by PCR using each primer pair. The following primer pairs were used: 5’-GAG AGG AAT CGG CAG GCT GCA GCT G-3’ (25 mer) and 5’-CAG ATA CAT CCA CAT CGT TTA GCG G-3’ (25 mer) for GDNF; 5’-CGG AGT CAA CGG ATT TGG TCG TAT-3’ (24 mer) and 5’-AGC CTT CTC CAT GGT GGT GGA GAC-3’ (24 mer) for glyceraldehyde-3-phosphate dehydrogenase (GAPDH). The number of PCR cycles and specific annealing temperatures were 34 cycles and 60 °C for GDNF, and 26 cycles and 55 °C for GAPDH. Reaction products (337 bp for GDNF and 309 bp for GAPDH) were electrophoresed on 2% agarose gels containing ethidium bromide and photographed under UV light.
4.7. Statistical Analysis
All results were expressed as means ± SEM. Significant differences in experiments with 2 groups were analyzed by performing Student’s t-test. Experiments with 3 or more groups were subjected to a one-way ANOVA followed by Dunnet’s multiple comparison test. p < 0.05 was taken to be statistically significant.
Acknowledgments
The authors wish to thank Ushio ChemiX Corporation for supplying AUR.
Author Contributions
Conceptualization, Y.F. and S.O.; formal analysis, R.-i.H. and M.N. (Makiko Nakaya); investigation, A.S. and M.N. (Mitsunari Nakajima); writing—original draft preparation, Y.F.; writing—review and editing, Y.F. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- 1.Genovese S., Epifano F. Auraptene: A natural biologically active compound with multiple targets. Curr. Drug Targets. 2011;12:381–386. doi: 10.2174/138945011794815248. [DOI] [PubMed] [Google Scholar]
- 2.DeRosa G., Maffioli P., Sahebkar A. Auraptene and Its Role in Chronic Diseases. Adv. Exp. Med. Biol. 2016;929:399–407. doi: 10.1007/978-3-319-41342-6_19. [DOI] [PubMed] [Google Scholar]
- 3.Okuyama S., Minami S., Shimada N., Makihata N., Nakajima M., Furukawa Y. Anti-inflammatory and neuroprotective effects of auraptene, a citrus coumarin, following cerebral global ischemia in mice. Eur. J. Pharmacol. 2013;699:118–123. doi: 10.1016/j.ejphar.2012.11.043. [DOI] [PubMed] [Google Scholar]
- 4.Okuyama S., Yamamoto K., Mori H., Toyoda N., Yoshimura M., Amakura Y., Yoshida T., Sugawara K., Sudo M., Nakajima M., et al. Auraptene in the Peels of Citrus kawachiensis (Kawachi Bankan) Ameliorates Lipopolysaccharide-Induced Inflammation in the Mouse Brain. Evid. Based Complement. Altern. Med. 2014;2014:1–9. doi: 10.1155/2014/408503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Okuyama S., Morita M., Kaji M., Amakura Y., Yoshimura M., Shimamoto K., Ookido Y., Nakajima M., Furukawa Y. Auraptene Acts as an Anti-Inflammatory Agent in the Mouse Brain. Molecules. 2015;20:20230–20239. doi: 10.3390/molecules201119691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Okuyama S., Semba T., Toyoda N., Epifano F., Genovese S., Fiorito S., Taddeo V.A., Sawamoto A., Nakajima M., Furukawa Y. Auraptene and Other Prenyloxyphenylpropanoids Suppress Microglial Activation and Dopaminergic Neuronal Cell Death in a Lipopolysaccharide-Induced Model of Parkinson’s Disease. Int. J. Mol. Sci. 2016;17:1716. doi: 10.3390/ijms17101716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Toyomoto M., Inoue S., Ohta K., Kuno S., Ohta M., Hayashi K., Ikeda K. Production of NGF, BDNF and GDNF in mouse astrocyte cultures is strongly enhanced by a cerebral vasodilator, ifenprodil. Neurosci. Lett. 2005;379:185–189. doi: 10.1016/j.neulet.2004.12.063. [DOI] [PubMed] [Google Scholar]
- 8.Rocha S.M., Cristovão A.C., Campos F.L., Fonseca C.P., Baltazar G. Astrocyte-derived GDNF is a potent inhibitor of microglial activation. Neurobiol. Dis. 2012;47:407–415. doi: 10.1016/j.nbd.2012.04.014. [DOI] [PubMed] [Google Scholar]
- 9.Suter-Crazzolara C., Unsicker K. GDNF mRNA levels are induced by FGF-2 in rat C6 glioblastoma cells. Mol. Brain Res. 1996;41:175–182. doi: 10.1016/0169-328X(96)00089-7. [DOI] [PubMed] [Google Scholar]
- 10.Golan M., Schreiber G., Avissar S. Antidepressants elevate GDNF expression and release from C₆ glioma cells in a β-arrestin1-dependent, CREB interactive pathway. Int. J. Neuropsychopharmacol. 2011;14:1289–1300. doi: 10.1017/S1461145710001550. [DOI] [PubMed] [Google Scholar]
- 11.Di Benedetto B., Kühn R., Nothdurfter C., Rein T., Wurst W., Rupprecht R. N-desalkylquetiapine activates ERK1/2 to induce GDNF release in C6 glioma cells: A putative cellular mechanism for quetiapine as antidepressant. Neuropharmacology. 2012;62:209–216. doi: 10.1016/j.neuropharm.2011.07.001. [DOI] [PubMed] [Google Scholar]
- 12.Eun C.-S., Lim J.-S., Lee J., Lee S.-P., Yang S.-A. The protective effect of fermented Curcuma longa L. on memory dysfunction in oxidative stress-induced C6 gliomal cells, proinflammatory-activated BV2 microglial cells, and scopolamine-induced amnesia model in mice. BMC Complement. Altern. Med. 2017;17:367. doi: 10.1186/s12906-017-1880-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sawamoto A., Okuyama S., Nakajima M., Furukawa Y. Citrus flavonoid 3,5,6,7,8,3’,4’-heptamethoxy-flavone induces BDNF via cAMP/ERK/CREB signaling and reduces phosphodiesterase activity in C6 glioma cells. Pharmacol. Rep. 2019;71:653–658. doi: 10.1016/j.pharep.2019.03.006. [DOI] [PubMed] [Google Scholar]
- 14.Kumar S., Weingarten D.P., Callahan J.W., Sachar K., deVellis J. Regulation of mRNAs for three enzymes in the glial cell model C6 cell line. J. Neurochem. 1984;43:1455–1463. doi: 10.1111/j.1471-4159.1984.tb05408.x. [DOI] [PubMed] [Google Scholar]
- 15.Mangoura D., Sakellaridis N., Jones J., Vernadakis A. Early and late passage C-6 glial cell growth: Similarities with primary glial cells in culture. Neurochem. Res. 1989;14:941–947. doi: 10.1007/BF00965927. [DOI] [PubMed] [Google Scholar]
- 16.Lin L., Doherty D., Lile J., Bektesh S., Collins F. GDNF: A glial cell line-derived neurotrophic factor for midbrain dopaminergic neurons. Science. 1993;260:1130–1132. doi: 10.1126/science.8493557. [DOI] [PubMed] [Google Scholar]
- 17.Tomac A., Lindqvist E., Lin L.-F.H., Ögren S.O., Young D., Hoffer B.J., Olson L. Protection and repair of the nigrostriatal dopaminergic system by GDNF in vivo. Nature. 1995;373:335–339. doi: 10.1038/373335a0. [DOI] [PubMed] [Google Scholar]
- 18.Airaksinen M.S., Saarma M. The GDNF family: Signalling, biological functions and therapeutic value. Nat. Rev. Neurosci. 2002;3:383–394. doi: 10.1038/nrn812. [DOI] [PubMed] [Google Scholar]
- 19.Tenenbaum L., Humbert-Claude M. Glial Cell Line-Derived Neurotrophic Factor Gene Delivery in Parkinson’s Disease: A Delicate Balance between Neuroprotection, Trophic Effects, and Unwanted Compensatory Mechanisms. Front. Neuroanat. 2017;11:2336. doi: 10.3389/fnana.2017.00029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pöyhönen S., Er S., Domanskyi A., Airavaara M. Effects of Neurotrophic Factors in Glial Cells in the Central Nervous System: Expression and Properties in Neurodegeneration and Injury. Front. Physiol. 2019;10:486. doi: 10.3389/fphys.2019.00486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Arenas E., Trupp M., Åkerud P., Ibañez C.F. GDNF prevents degeneration and promotes the phenotype of brain noradrenergic neurons in vivo. Neuron. 1995;15:1465–1473. doi: 10.1016/0896-6273(95)90024-1. [DOI] [PubMed] [Google Scholar]
- 22.Pascual A., Hidalgo-Figueroa M., I Piruat J., Pintado C.O., Gómez-Díaz R., López-Barneo J. Absolute requirement of GDNF for adult catecholaminergic neuron survival. Nat. Neurosci. 2008;11:755–761. doi: 10.1038/nn.2136. [DOI] [PubMed] [Google Scholar]
- 23.Koyama Y., Egawa H., Osakada M., Baba A., Matsuda T. Increase by FK960, a novel cognitive enhancer, in glial cell line-derived neurotrophic factor production in cultured rat astrocytes. Biochem. Pharmacol. 2004;68:275–282. doi: 10.1016/j.bcp.2004.03.023. [DOI] [PubMed] [Google Scholar]
- 24.Buscà R., Pouysségur J., Lenormand P. ERK1 and ERK2 map kinases: Specific roles or functional redundancy? Front. Cell. Dev. Biol. 2016;4:53. doi: 10.3389/fcell.2016.00053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Impey S., Obrietan K., Wong S.T., Poser S., Yano S., Wayman G., Deloulme J.C., Chan G., Storm D.R. Cross talk between ERK and PKA is required for Ca2+ stimulation of CREB-dependent transcription and ERK nuclear translocation. Neuron. 1998;21:869–883. doi: 10.1016/S0896-6273(00)80602-9. [DOI] [PubMed] [Google Scholar]
- 26.Afshari A.R., Roshan M.K., Soukhtanloo M., Ghorbani A., Rahmani F., Jalili-Nik M., Vahedi M.M., Hoseini A., Sadeghnia H.R., Mollazadeh H., et al. Cytotoxic effects of auraptene against a human malignant glioblastoma cell line. Avicenna J. Phytomed. 2019;9:334–346. [PMC free article] [PubMed] [Google Scholar]
- 27.Bao X.-Q., Wang L., Yang H.-Y., Hou L.-Y., Wang Q.-S., Zhang D. Induction of glial cell line-derived neurotrophic factor by the squamosamide derivative FLZ in astroglia has neuroprotective effects on dopaminergic neurons. Brain Res. Bull. 2019 doi: 10.1016/j.brainresbull.2019.10.008. [DOI] [PubMed] [Google Scholar]
- 28.Yu H., Oh-Hashi K., Tanaka T., Sai A., Inoue M., Hirata Y., Kiuchi K. Rehmannia glutinosa induces glial cell line-derived neurotrophic factor gene expression in astroglial cells via cPKC and ERK1/2 pathways independently. Pharmacol. Res. 2006;54:39–45. doi: 10.1016/j.phrs.2006.01.014. [DOI] [PubMed] [Google Scholar]
- 29.Okuyama S., Kotani Y., Yamamoto K., Sawamoto A., Sugawara K., Sudo M., Ohkubo Y., Tamanaha A., Nakajima M., Furukawa Y. The peel ofCitrus kawachiensis(kawachi bankan) ameliorates microglial activation, tau hyper-phosphorylation, and suppression of neurogenesis in the hippocampus of senescence-accelerated mice. Biosci. Biotechnol. Biochem. 2018;82:869–878. doi: 10.1080/09168451.2018.1433993. [DOI] [PubMed] [Google Scholar]
- 30.Okuyama S., Yamamoto K., Mori H., Sawamoto A., Amakura Y., Yoshimura M., Tamanaha A., Ohkubo Y., Sugawara K., Sudo M., et al. Neuroprotective effect of Citrus kawachiensis (Kawachi Bankan) peels, a rich source of naringin, against global cerebral ischemia/reperfusion injury in mice. Biosci. Biotechnol. Biochem. 2018;82:1216–1224. doi: 10.1080/09168451.2018.1456320. [DOI] [PubMed] [Google Scholar]
- 31.Okuyama S., Shinoka W., Nakamura K., Kotani M., Sawamoto A., Sugawara K., Sudo M., Nakajima M., Furukawa Y. Suppressive effects of the peel of Citrus kawachiensis (Kawachi Bankan) on astroglial activation, tau phosphorylation, and inhibition of neurogenesis in the hippocampus of type 2 diabetic db/db mice. Biosci. Biotechnol. Biochem. 2018;82:1384–1395. doi: 10.1080/09168451.2018.1469396. [DOI] [PubMed] [Google Scholar]
- 32.Okuyama S., Kanzaki T., Kotani Y., Katoh M., Sawamoto A., Nakajima M., Furukawa Y. Continual treatment with the peels of Citrus kawachiensis (Kawachi Bankan) protects the dopaminergic neuronal cell death in a lipopolysaccharide-induced model of Parkinson’s disease. J. Nutr. Sci. Vitaminol. 2019;65:205–208. doi: 10.3177/jnsv.65.205. [DOI] [PubMed] [Google Scholar]
- 33.Okuyama S., Katoh M., Kanzaki T., Kotani Y., Amakura Y., Yoshimura M., Fukuda N., Tamai T., Sawamoto A., Nakajima M., et al. Auraptene/Naringin-Rich Fruit Juice of Citrus kawachiensis (Kawachi Bankan) Prevents Ischemia-Induced Neuronal Cell Death in Mouse Brain through Anti-Inflammatory Responses. J. Nutr. Sci. Vitaminol. 2019;65:66–71. doi: 10.3177/jnsv.65.66. [DOI] [PubMed] [Google Scholar]
- 34.Igase M., Okada Y., Ochi M., Igase K., Ochi H., Okuyama S., Furukawa Y., Ohyagi Y. Auraptene in the peels of Citrus Kawachiensis (Kawachibankan) contributes to the preservation of cognitive function: A randomized, placebo-controlled, double-blind study in healthy volunteers. J. Prev. Alz. Dis. 2017;3:1–5. doi: 10.14283/jpad.2017.47. [DOI] [PubMed] [Google Scholar]
- 35.Budni J., Bellettini-Santos T., Mina F., Garcez M.L., Zugno A.I. The involvement of BDNF, NGF and GDNF in aging and Alzheimer’s disease. Aging Dis. 2015;6:331–341. doi: 10.14336/AD.2015.0825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ghribi O., Herman M.M., Forbes M.S., DeWitt D.A., Savory J. GDNF Protects against Aluminum-Induced Apoptosis in Rabbits by Upregulating Bcl-2 and Bcl-XL and Inhibiting Mitochondrial Bax Translocation. Neurobiol. Dis. 2001;8:764–773. doi: 10.1006/nbdi.2001.0429. [DOI] [PubMed] [Google Scholar]
- 37.Revilla S., Ursulet S., Álvarez-López M.J., Castro-Freire M., Perpiña U., García-Mesa Y., Bortolozzi A., Giménez-Llort L., Kaliman P., Cristòfol R., et al. Lenti-GDNF Gene Therapy Protects Against Alzheimer’s Disease-Like Neuropathology in 3xTg-AD Mice and MC65 Cells. CNS Neurosci. Ther. 2014;20:961–972. doi: 10.1111/cns.12312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Airavaara M., Pletnikova O., Doyle M.E., Zhang Y.E., Troncoso J.C., Liu Q.-R. Identification of Novel GDNF Isoforms and cis-Antisense GDNFOS Gene and Their Regulation in Human Middle Temporal Gyrus of Alzheimer Disease*. J. Boil. Chem. 2011;286:45093–45102. doi: 10.1074/jbc.M111.310250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Naoi M., Inaba-Hasegawa K., Shamoto-Nagai M., Maruyama W. Neurotrophic function of phytochemicals for neuroprotection in aging and neurodegenerative disorders: Modulation of intracellular signaling and gene expression. J. Neural Transm. 2017;124:1515–1527. doi: 10.1007/s00702-017-1797-5. [DOI] [PubMed] [Google Scholar]
- 40.Furukawa Y., Sawamoto A., Yamaoka M., Nakaya M., Hieda Y., Choshi T., Hatae N., Okuyama S., Nakajima M., Hibino S. Effects of Carbazole Derivatives on Neurite Outgrowth and Hydrogen Peroxide-Induced Cytotoxicity in Neuro2a Cells. Molecules. 2019;24:1366. doi: 10.3390/molecules24071366. [DOI] [PMC free article] [PubMed] [Google Scholar]