Abstract
Introduction: Cabazitaxel (CBZ) is used worldwide for castration-resistant prostate cancer after docetaxel treatment. In July 2014 the drug was approved in Japan with the same induction dose used for Caucasian patients. In this study, we examined and compared the results of an initial low-dose CBZ treatment in patients admitted to our hospital.
Patients and Methods: Between July 2014 and August 2018, sixteen mCRPC patients were enrolled and underwent a low-dose CBZ treatment at our hospital. We compared the results with those of a Japanese metastatic docetaxel- and castration-resistant prostate cancer Phase I study.
Results: The median patient age was 77 years (range, 53–84 years). Of the 16 patients, eight (50%) had a lymph node metastasis and 11 (68.8%) had a distant metastasis, 10 of whom had only a bone metastasis. The median dose of CBZ was 30 mg (range, 20–32 mg) and the median number of CBZ cycles was 2.5 (range, 1–18). The PSA level of 9 (56.3%) patients decreased after CBZ treatment, including 4 (25%) who showed a decrease to <50%. The median time interval in which the PSA level decreased was 2 months (range, 1–18 months). The observed adverse events (AE) were neutropenia (31.3%), febrile neutropenia (6.3%), fatigue (43.8%), nausea (18.8%), diarrhea (12.5%), decreased appetite (25%), dysgeusia (6.3%), white blood cell count decrease (43.8%), platelet count decrease (12.3%), and anemia (75%). However, no patient listed an AE as the reason for discontinuing the treatment.
Conclusions: Even at a low dose, CBZ could improve the PSA value in patients with CRPC previously treated with docetaxel. Dose reduction and prophylactic administration of sustained G-CSF were also safe treatment options. Further studies involving an introduction period including a modulation of duration and dose are necessary, especially in Japanese patients.
Keywords: prostate cancer, castration resistant prostate cancer, cabazitaxcel
Introduction
Prostate cancer is the sixth most common cause of cancer-related death in men and the third most common cause of death in developed countries1). It is also the fourth highest cause of cancer-related death in Japanese men2). Moreover, prostate cancer incidence in Japan has increased in the last decade. Therefore, prostate cancer control is among the most important issues in Japan.
Androgen-deprivation therapy (ADT) is the first-line treatment for advanced metastatic prostate cancer. However, the treatment response is not very effective, with many cases of this disease progressing to metastatic castration-resistant prostate cancer (mCRPC) during ADT3). Some new agents targeting CRPC, including abiraterone, enzalutamide, sipuleucel-T, and radium-223, have been developed4,5,6), used worldwide, and approved in Japan. These agents may be preferable to chemotherapy in patients with a poor tolerance due to predisposing factors. However, in patients with rapidly progressing disease, visceral metastases, or those with a poor response to an initial ADT, chemotherapy is preferred.
Docetaxel, a taxane, is a well-established anti-cancer drug in combination with prednisone and is an effective treatment for mCRPC. Docetaxel therapy is associated with an overall improved survival, quality of life, and pain relief in mCRPC patients3). However, mCRPC generally develops resistance to docetaxel during treatment.
Cabazitaxel (CBZ), a next generation taxane, results in survival benefits in patients with progressing mCRPC after docetaxel7, 8). In July 2014 CBZ was approved in Japan with the same induction dose used for Caucasian patients. A phase III CBZ study (the TROPIC trial) showed that CBZ significantly prolonged the overall survival of CRPC patients who received docetaxel treatment8). However, some patients discontinued chemotherapy due to adverse events (AEs) or death, and for some them the activities of daily living (ADL) decreased after chemotherapy. In a phase I dose-escalation CBZ study in patients with mCRPC in Japan (TED11576), the safety, tolerability, and anti-tumor activity in Japanese patients were found to be comparable to previous studies’ results in Caucasian patients. However, the incidence of AEs, such as neutropenia and febrile neutropenia, was relatively high9). According to this Japanese study, 20.5% patients discontinued a full-dose CBZ therapy due to AEs10).
Therefore, a safer and less complicated administration of CBZ therapy is required, especially in an aging society like Japan. To overcome this problem, a low-dose administration regimen is thought to be one of the most convenient solutions. Thus, in our present study, we examined the results of an initial low-dose CBZ treatment in patients with mCRPC and evaluated the safety and efficacy of low-dose CBZ by comparing our results with those of a previous Japanese phase I clinical study (TED11576).
Patients and Methods
Between July 2014 and August 2018, sixteen patients with mCRPC were enrolled and treated with a low-dose CBZ treatment at our hospital. No patient had undergone radical prostatectomy, and the prostate-specific antigen (PSA) level had increased in all the patients despite medical castration with at least one cycle of docetaxel therapy combined with ADT. All the patients had shown a complete or partial response during previous multiple treatments and had an Eastern Cooperative Oncology Group performance status score of 0 or 1 prior to CBZ treatment.
Our low-dose weekly CBZ therapy protocol was to repeat the treatment every 28 days (guidelines recommend treatment every 21 days)11). The administration interval was extended on the basis of each case.
The CBZ dose was <20 mg/m2 depending on the attending clinician (guidelines recommend a dose of 25 mg/m2). To avoid AEs before the administration of CBZ, a combination of steroids and antiemetics was used: 6.6 mg of dexamethasone and 5 mg of d-chlorpheniramine maleate. To avoid neutropenia after CBZ administration, pegfilgrastim (PEG), a sustained-type granulocyte colony-stimulating factor (G-CSF), was administered 1 or 2 days after CBZ administration; PEG was administered to 15 of the 16 patients, but not to the first enrolled patient.
All the patients were administered 10 mg of prednisolone while undergoing chemotherapy.
The first administration course was performed in hospitalized patients. From the second course onwards, CBZ chemotherapy was performed on an outpatient basis.
The chemotherapy’s efficacy was determined on the basis of the decrease of PSA level, computed tomography (CT) results, and/or bone scintigraphy, and an overall survival (OS) from the starting date of the prostate cancer therapy. The change in PSA level was evaluated using a Waterfall plot and the OS was evaluated using the Kaplan-Meier curve.
We evaluated the AEs according to the National Cancer Institute Common Terminology Criteria for adverse events (CTCAE), version 5.0. Statistical analyses were performed using EZR 1.37 (Division of Hematology, Saitama Medical Center, Jichi Medical University).
The study was approved by the institutional review board of the Anjo Kosei Hospital (review board number: R17-056).
Results
The median age of the 16 patients at the initiation of CBZ treatment was 77 (range, 53–84 years), and the median PSA level at the initiation of the primary therapy was 141 ng/ml (range, 6.8–3,376 ng/ml). The Gleason score was 6 in one patient, 7 in two, 8 in three, 9 in three, 10 in one, and unknown in five. Of the 16 patients, eight (50%) had a lymph node metastasis and 11 (68.8%) had a distant metastasis, 10 of whom only had a bone metastasis. There was one case of lung metastasis in this cohort. There were 13 cases classified as high or very high-risk according to the National Comprehensive Cancer Network (NCCN) guidelines (Gleason score of 8–10 or T3a–4 or initial PSA >20 ng/ml)11).
The median dose of docetaxel was 745 mg (range, 140–1,920) and the median PSA level before and after the CBZ administration was 195.7 ng/ml (range, 4.2–2,041 ng/ml) and 160.15 ng/ml (range, 4.4–1,809.2 ng/ml). The median CBZ dose was 30 mg (range, 20–32 mg) and the median number of CBZ cycles was 2.5 (range, 1–18).
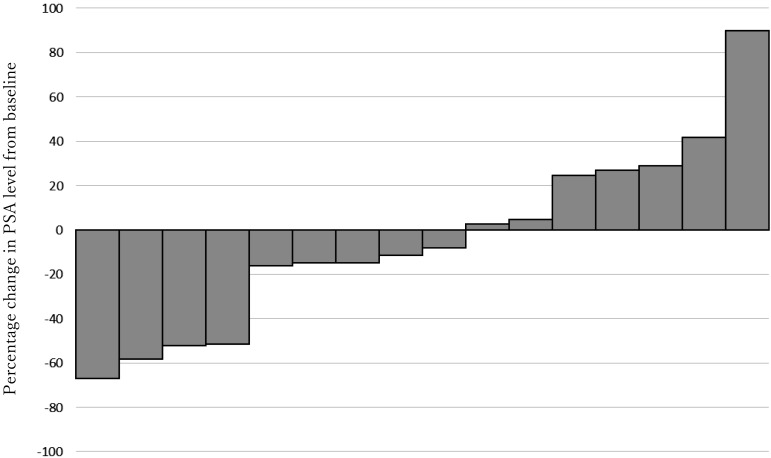
The PSA level of nine (56.3%) patients decreased after CBZ treatment, including four (25%) who showed a decrease to <50% (Figure 1). The PSA level decreased with a median time interval of 2 months (range, 1–18 months).
Figure 1.
Waterfall plot showing maximum percentage change in prostate-specific antigen (PSA) level from baseline.
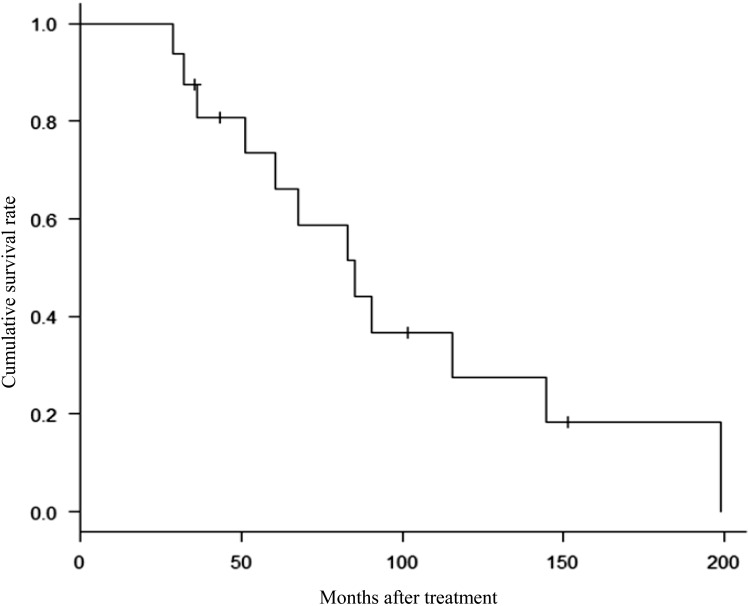
The OS from the beginning of the prostate cancer treatment is shown in Figure 2. The median OS was 85 months. The 5-year survival rate was 73.4% (95% confidence interval [CI], 43.5–89.2).
Figure 2.
Kaplan-Meier curve showing overall survival (OS) after the start of androgen-deprivation therapy.
The observed AEs were neutropenia (31.3%), febrile neutropenia (6.3%), fatigue (43.8%), nausea (18.8%), diarrhea (12.5%), decreased appetite (25%), dysgeusia (6.3%), white blood cell count decrease (43.8%), platelet count decrease (12.3%), and anemia (75%). However, no patient listed an AE as the reason for discontinuing treatment.
Discussion
According to the phase III TROPIC trial, 25 mg/m2 CBZ, in combination with prednisone, for treating CRPC patients previously treated with docetaxel8) was approved in 2010. However, severe AEs were frequently reported, and the administration of lower doses was tested. In the phase III PROSELICA study, the non-inferiority and safety of 20 mg/m2 CBZ were compared with those of 25 mg/m2 CBZ. PSA level changes of >50% were significantly more common in the 25 mg/m2 group (42.9%) than in the 20 mg/m2 group (29.5%, P=0.001). However, there was no significant difference in the median progression-free survival (2.9 months and 3.5 months in the 20 mg/m2 and 25 mg/m2 groups, respectively; hazard ratio [HR], 1.099; 95% CI, 0.974–1.240) or median OS (13.4 months and 14.5 months in the 20 mg/m2 and 25 mg/m2 groups, respectively; HR, 1.024; 95% CI, 0.989–1.184)12). Moreover, Japanese patients are more susceptible to myelotoxicity due to CBZ therapy compared to Western patients13). Therefore, the optimal CBZ dose for Japanese patients’ needs to be evaluated.
Our study is the first to demonstrate the feasibility of a low-dose CBZ treatment in Japanese CRPC patients after docetaxel administration in our hospital by comparing our results with those of a phase I study in Japan (TED 11576). Our low-dose CBZ treatment was safer than a full-dose CBZ therapy, considering the frequency of all AEs. In the phase I CBZ study in Japan, the AEs were neutropenia (100%), febrile neutropenia (54.5%), fatigue (54.5%), nausea (52.3%), diarrhea (50%), decreased appetite (40.9%), dysgeusia (27.3%), white blood cell decreases (100%), platelet count decrease (72.7%), and anemia (100%)13). In contrast, in our study, all the AEs were less frequent after our low-dose CBZ therapy, and we believe that the low incidence of AEs contributes to the patient’s stable ADL.
Regarding the efficacy, in the phase I study in Japan, 61% patients had a PSA response, including 31.7% of patients who had >50% decline in PSA levels. Considering that these patients received a full-dose CBZ treatment, the results of our study are not inferior. Therefore, after comparing our results with those of the phase I study in Japan, we believe that our low-dose CBZ chemotherapy had fewer AEs and was effective for Japanese patients.
Our present study has several limitations. This study design was retrospective and included a relatively small population. Furthermore, we have not compared low-dose CBZ therapy with a full dose therapy, because few patients were administrated a full CBZ dose in our hospital. Therefore, further prospective large-scale studies in the Japanese population are needed in the near future. Moreover, it is necessary to evaluate ADL over time using methods such as questionnaires, because we did not quantitatively evaluate the patient’s ADL and satisfaction in this study.
Conclusion
Even at a low dose, CBZ could improve the PSA value in patients with CRPC previously treated with docetaxel, indicating that it may be effective for cancer control. Dose reduction and prophylactic administration of sustained G-CSF were also safe treatment options. Nevertheless, further studies involving an introduction period as well as modulation of duration and dose are necessary, especially in Japanese patients.
References
- 1.Jemal A, Bray F, Center MM. Global cancer statistics. CA Cancer J Clin 2011; 61: 69–90. doi: 10.3322/caac.20107 [DOI] [PubMed] [Google Scholar]
- 2.World Health Organization International Agency for Research on Cancer. Global Cancer Observatory. https://gco.iarc.fr. (Accessed May 14, 2019).
- 3.Tannock IF, de Wit R, Berry WR. TAX 327 InvestigatorsDocetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med 2004; 351: 1502–1512. doi: 10.1056/NEJMoa040720 [DOI] [PubMed] [Google Scholar]
- 4.de Bono JS, Logothetis CJ, Molina A. COU-AA-301 InvestigatorsAbiraterone and increased survival in metastatic prostate cancer. N Engl J Med 2011; 364: 1995–2005. doi: 10.1056/NEJMoa1014618 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Parker C, Nilsson S, Heinrich D. ALSYMPCA InvestigatorsAlpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med 2013; 369: 213–223. doi: 10.1056/NEJMoa1213755 [DOI] [PubMed] [Google Scholar]
- 6.Beer TM, Armstrong AJ, Rathkopf DE. PREVAIL InvestigatorsEnzalutamide in metastatic prostate cancer before chemotherapy. N Engl J Med 2014; 371: 424–433. doi: 10.1056/NEJMoa1405095 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mita AC, Denis LJ, Rowinsky EK. Phase I and pharmacokinetic study of XRP6258 (RPR 116258A), a novel taxane, administered as a 1-hour infusion every 3 weeks in patients with advanced solid tumors. Clin Cancer Res 2009; 15: 723–730. doi: 10.1158/1078-0432.CCR-08-0596 [DOI] [PubMed] [Google Scholar]
- 8.Bahl A, Oudard S, Tombal B. TROPIC InvestigatorsImpact of cabazitaxel on 2-year survival and palliation of tumour-related pain in men with metastatic castration-resistant prostate cancer treated in the TROPIC trial. Ann Oncol 2013; 24: 2402–2408. doi: 10.1093/annonc/mdt194 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mukai H, Takahashi S, Nozawa M. TED 11576 investigatorsPhase I dose-escalation and pharmacokinetic study (TED 11576) of cabazitaxel in Japanese patients with castration-resistant prostate cancer. Cancer Chemother Pharmacol 2014; 73: 703–710. doi: 10.1007/s00280-014-2394-z [DOI] [PubMed] [Google Scholar]
- 10.Fukuta F, Kitamura H, Yanase M. Efficacy and safety of docetaxel and prednisolone for castration-resistant prostate cancer: a multi-institutional retrospective study in Japan. Jpn J Clin Oncol 2015; 45: 682–687. doi: 10.1093/jjco/hyv053 [DOI] [PubMed] [Google Scholar]
- 11.National Comprehensive Cancer Network (NCCN) NCCN Clincal Practice Guidelines in Oncology: Prostate Cancer. Version 4.2018. https://www2.tri-kobe.org/nccn/guideline/urological/japanese/prostate.pdf. (Accessed May 14, 2019).
- 12.Eisenberger M, Hardy-Bessard AC, Kim CS. Phase III study comparing a reduced dose of cabazitaxel (20 mg/m2) and the currently approved dose (25 mg/m2) in postdocetaxel patients with metastatic castration-resistant prostate cancer-PROSELICA. J Clin Oncol 2017; 35: 3198–3206. doi: 10.1200/JCO.2016.72.1076 [DOI] [PubMed] [Google Scholar]
- 13.Nozawa M, Mukai H, Takahashi S. Japanese phase I study of cabazitaxel in metastatic castration-resistant prostate cancer. Int J Clin Oncol 2015; 20: 1026–1034. doi: 10.1007/s10147-015-0820-9 [DOI] [PubMed] [Google Scholar]