Abstract
Introduction: The management of Peyronie’s disease (PD) has remained a therapeutic dilemma for physicians and there is no gold standard treatment. In this paper, we decided to investigate the beneficial effect of the intralesional administration of verapamil compared with the intralesional administration of verapamil plus a low-intensity laser (LIL).
Methods: Research was activated from May 2016 to May 2018 and a total of 38 men aged 18 years and older completed the investigation. The subjects were randomly divided into 2 groups. Group 1 was composed of 22 patients that were treated only by verapamil (5 mg) plus a sham laser weekly for 6 weeks, and group 2 consisted of 22 patients that received a laser, using the BTL–6000 HIGH-INTENSITY LASER 12 W machine and the same protocol of intralesional verapamil injection. The visual analogue scale (VAS) was used to evaluate pain during an erection, penile ultrasonography was used to measure plaque size, the penile curvature angle degree was measured using the photographs taken during an erection, and the International Index of Erectile Function questionnaire was used to assess erectile function. The follow-up treatment lasted for nine months, with visits performed in the 3rd and 9th months.
Results: All study parameters decreased significantly after treatment in both arms, but the reduction in pain and penile curvature improvements in combination therapy revealed more significant changes in 3 months (p = .035, p=.032). Nevertheless, these improvements were not seen in the follow-up session after 9 months.
Conclusion: A laser appears to be safe treatment modality in carefully-selected patients with PD. It has moderate efficacy in the short term.
Keywords: Erectile dysfunction, Laser therapy, Intralesional injections, Penile curvature, Verapamil, Peyronie’s disease
Introduction
Peyronie’s disease (PD) is marked by pain on an erection, acquired penile curvature, decreased erectile capacity, and difficulty in vaginal penetration.1,2 The prevalence of PD in the general population ranges from 0.39% to 7.1%, but it goes up to 20.3% in diabetic patients with Erectile Dysfunction (ED).3 Its pathogenesis stands poorly understood and various theories have been proposed in this regard.4 Most urologists support the logic of a combination of microtrauma during sexual activity in a susceptible case, which would lead to deregulation of the scarring process and formation of the plaque.5-7
PD presents with 2 phases: the active phase, in which the penile deformity may vary with time and can last up to 18 months, and the stable phase, when the alteration of the penile form stabilizes.8 Treatment for PD is individualized according to the phase of the disease. During the active phase, noninvasive treatments are used and a surgical procedure is reserved to treat penile deformities during the stable phase of PD.9
Treatment choices include oral medications, topical agents, intralesional injections, mechanical stretching or vacuum devices, and recently, encouraging results from extra corporeal shockwave therapy (ESWT) and laser therapy were reported.10-12 The management of this disease has remained a therapeutic dilemma for urologists and there is no gold standard treatment.2,13 A laser carries light energy to injured cells to stimulate the local release of nitric oxide (a vasodilator agent), to improve cellular energy by stimulating mitochondria to accelerate ATP production, to increase metabolic activity and cell growth, improving wound healing, and to reduce acute and chronic pain.14,15 All of these laser characteristics can reverse abnormal fibrotic reaction in PD pathophysiology.13
For the first time in 1995 Johnson et al,16 used a low-level gallium-aluminum-arsenide (GaAlAs) laser to treat patients with PD. After that in 2008, Candebat Montero et al17 evaluated the result of the management of PD with a helium-neon laser (HeNe). Both studies revealed improvement in symptoms, a decrease in the size of the fibrous plaque, and a decrease of penile curvature. The authors recommended incorporating this treatment as another therapeutic option in PD.
In this paper, we aimed to investigate the beneficial effect of intralesional administration of verapamil compared with intralesional administration of verapamil plus a low-intensity laser (LIL) to evaluate the clinical effectiveness in patients with PD.
Methods
Population
The present research was activated in the field from May 2016 to May 2018, and a total of 38 men completed the investigation.
Inclusion and Exclusion criteria
The inclusion criteria were patients aged ≥18 years with PD confirmed by the presence of stable plaque as detected via ultrasonography, no medical treatment for the previous 6 months, penile deviation of ≤60°, pain in an erection or during intercourse, and the ability to perform vaginal intercourse. Patients were excluded if they were in an active stage and in the presence of coagulopathy disease. In addition, they were excluded if they had systolic blood pressure <90 mmHg, symptomatic orthostatic hypotension, previous surgical treatment of PD, erectile dysfunction due to hormonal, psychological, vascular or neurological causes, a history of allergy to verapamil, and chordee.
Randomization and Blinding
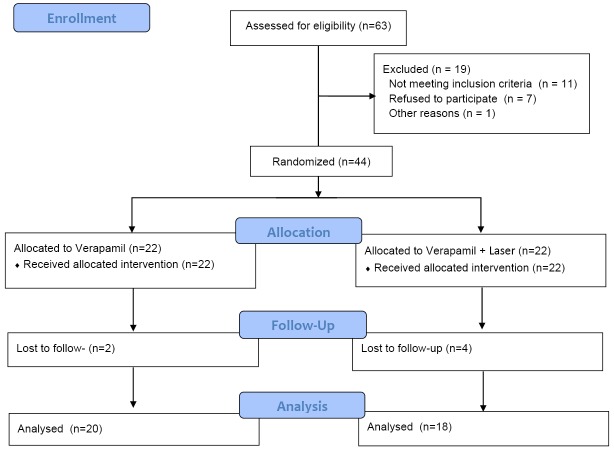
The patients were divided into 2 groups by computed random block randomization. Group 1 was composed of 22 patients treated only by verapamil (5 mg) plus a sham laser (just turn on the light of laser), and group 2 consisted of 22 patients that received a laser and the same protocol of intralesional verapamil injection (Figure 1). The allocation was conducted with non-opaque envelopes to ensure concealment.
Figure 1.
Patients’ Flowchart.
Study Protocol
An accurate demographic, clinical history and physical examination were recorded during the first visit. The subjective pain during an erection was evaluated by means of the visual analogue scale (VAS); with 0 as no pain and 10 as maximum pain, 4 classes of pain were identified: severe pain (8-10), mild pain (5-7), low-mild pain (2-4), and low/no pain (0-1).18 The plaque size was measured (in mm) with penile ultrasonography (with a 7.5 to 12 MHz linear transducer for small organs and superficial lesions). The degree of the penile curvature angle was measured with photographs taken in 3 positions during an erection. The severity of the deformity, which was assessed using the Kelami modified classification,19 was divided into 2 types: mild penile deformity (≤30°) and moderate penile deformity (31°-60°). To assess erectile function, the International Index of Erectile Function (IIEF-5) questionnaire was used.20 According to the IIEF-5 questionnaire, the classes of ED were identified as follows: severe (5-7), moderate (8-11), mild to moderate (12-16), mild (17-21), and normal (22-25).
The patients underwent 6 weekly sessions of the laser, using the BTL–6000 HIGH-INTENSITY LASER 12 W machine (BTL Company, UK). The chosen protocol was the Biostimulation mode, the power of 0.50 W, the dosage of 50 J/cm2, 1 cm2 area coverage, and 100-second exposure duration.
The follow-up visits were performed at 3 and 9 months.
Dropout
During the study, a total of 6 patients withdrew the therapy within 4 weeks. Two of the 22 patients in group 1 discontinued therapy due to their immigration to another city and 1 patient due to little or no therapy effectiveness. In group 2, 4 patients discontinued therapy within 5 weeks: 3 patients due to little or no therapy effectiveness and one patient due to injection pain. A total of 3 patients, 1 patient in group 1 and 2 patients in group 2, received surgical correction for their dissatisfaction despite medical treatment.
Study Variables
The primary treatment goal was assessed by the feasibility of sexual intercourse via the IIEF-5 questionnaire. The secondary outcome measures were the size of the fibrous plaque and improvement in pain during an erection. The occurrence adverse effects of the laser were also investigated.
Statistical Analysis
The statistical analysis was done using the Statistical Package for Social Sciences (SPSS Inc, Chicago, Illinois, USA) version 23 and R statistical software (version 3.5.2). Before conducting the statistical analyses, the collected data were coded to avoid unwanted bias during the analysis. The analytic phase was carried out using the Pearson’s chi-squared test for categorical variables and repeated measures ANOVA for quantitative variables. The angular data were analyzed with the Watson-Williams test and the Wheeler-Watson test. A level of significance was assumed to be <0.05. The missing data were completely at random and then we ignore them.
Results
General
The differences in baseline characteristics between the groups (see Table 1) were not significant. The groups had the same disease characteristics regarding the plaque diameter, the penile curvature angle, pain during an erection, and the IIEF5 score.
Table 1. Clinical and Baseline Characteristics .
| Variable |
Verapamil
(n = 20) |
Verapamil+ Laser
(n = 18) |
P value |
| Age(y) | 49.56± 10.12 | 48.05 ± 9.43 | <0.05 |
| Duration of disease (mon) | 11.03±2.16 | 11.06±2.09 | |
| Diabetes | 3 | 3 | |
| Smoking | 7 | 8 | |
| Hypertension | 2 | 1 | |
| Hyperlipidemia | 1 | 1 |
The history of genital trauma was present in 7 subjects. At the onset of the disease, the primary symptoms were pain in 8 cases, palpable plaque in 8 cases, and penile deformity in 6 cases. Five patients mentioned a sudden onset of the disease. The penile curvature had a dorsal direction in 17 patients, ventral direction in 9 patients, left lateral direction in 7 patients, and right lateral direction in 5 patients.
The localization site of the fibrous plaque, as evaluated by ultrasonography, had the following pattern: dorsolateral in 21 patients, dorsal in 13 patients, ventral in 3 patients, and ventrolateral in one patient.
The outcome measures for each group before and after the intervention are depicted in Table 2.
Table 2. Improvement in Clinical Outcomes Following Laser Therapy .
| Variable | Time | Verapamil (n = 20) | Verapamil+ Laser (n = 18) | P value |
| Plaque diameter (mm) | Before | 16.22 ±9.92 | 15.55 ±6.56 | 0.805 |
| After 3 month | 14.78± 9.82 | 12.10± 6.09 | 0.314 | |
| After 9 month | 13.56± 9.18 | 11.30± 5.89 | 0.368 | |
| P a | 0.001 | 0.0001 | ||
| P b | 0.001, 0.006 | 0.0001, 0.002 | ||
| P c | 0.0001 | 0.001 | ||
| Penile curvature | Before | 31.11±9.634 | 29.75±7.860 | 0.137 |
| After 3 month | 28.89 ±8.498 | 24.75± 5.495 | 0.032 | |
| After 9 month | 25.00± 10.290 | 22.75± 6.584 | 0.107 | |
| P a | >0.1 | >0.1 | ||
| P b | >0.1 | >0.1 | ||
| P c | >0.5 | >0.5 | ||
| Pain during erection | Before | 7.44±1.79 | 7.65±1.87 | 0.732 |
| After 3 month | 6.89 ±1.94 | 5.45 ±2.09 | 0.035 | |
| After 9 month | 6.39± 1.72 | 5.00± 2.10 | 0.033 | |
| P a | 0.004 | 0.001 | ||
| P b | 0.0001, 0.008 | 0.0001, 0.131 | ||
| P c | 0.0001 | 0.0001 | ||
| IIEF5 score | Before | 13.44± 4.93 | 11.80 ±4.41 | 0.285 |
| After 3 month | 14.28± 4.80 | 16.05 ±3.22 | 0.197 | |
| After 9 month | 14.72± 4.59 | 18.85± 3.07 | 0.003 | |
| P a | 0.003 | 0.0001 | ||
| P b | 0.001, 0.190 | 0.0001, 0.0001 | ||
| P c | 0.0001 | 0.001 |
Abbreviation: IIEF, International Index of Erectile Function.
a P value within group before vs 9 months after treatment.
b P value within group.
c Repeated measures ANOVA analysis P value.
All study parameters decreased significantly after treatment in both arms, but the reduction in pain and improvements in penile curvature in combination therapy revealed more significant changes at 3 months after the intervention. However, these improvements were not seen in the follow-up session after 9 months.
The management was well-tolerated. No complications were observed after intralesional application of verapamil. The laser therapy showed only penile bruising in 2 patients. The use of analgesics was not necessary.
Discussion
Determining appropriate treatment is based on a variety of factors such as the phase of the disease and the patient’s erectile status. Optimal patient management needs early detection to enhance functional and psychological outcomes. Non-surgical treatments may be considered for cases in the acute phase of the disease. Surgery is typically reserved for patients with stable disease.21,22
This study showed that 6 sessions of the laser with a 1-week interval improved penile curvature and pain significantly in comparison with single therapy by verapamil.
In 1994, Levine and colleagues published the first series of 16 patients treated with multiple intralesional injections of verapamil.23 This study started many other investigations in order to examine the effect of verapamil on the treatment of PD, and the results from most of them revealed a significant reduction in plaque consistency and penile curvature.1,24-27
Recently, new therapeutic advances in the management of PD have been presented as ESWT. Data from a meta-analysis by Hauck et al28 and Gao et al,29 systematic review by Fojecki et al,30 and also more recent prospective trials31-33 showed that while ESWT was effective with regards to penile pain and improved sexual function compared to natural history, it was not effective in reducing the plaque size or the degree of penile curvature. However, the efficiency of ESWT for PD has been controversial.29 After this innovation, laser therapy was introduced as one of the newest modalities for PD treatment.
PD is considered as an aberrant wound-healing process in response to inflammation.34 The outcome of a prolonged inflammatory response is the total breakdown of the delicately arranged network of elastic fibers and excessive production of collagen fibers.35 A microwave device, a high energy ultrasound, and a shock wave or laser energy can be used to direct the energy to ‘ground zero’ to zap the inflammatory reaction without zapping the adjacent erectile tissue such as in the case of radiation therapy of the plaque.34
In 2008, Montero et al17 performed an experimental study to evaluate the results of the treatment of PD with interferon and a laser from January 2003 to July 2005. Ninety-six patients were included and were divided into 3 treatment groups: interferon, the laser (HeNe) and interferon plus the laser. Treatment lasted for 28 weeks, with reevaluation 6 and 12 months after the end of treatment. Final results with combined interferon and the laser were an improvement in symptoms (84.7%), a decrease in the size of the fibrous plaque (90.6%), and a decrease of penile curvature (87.5%). The authors recommended incorporating this treatment as another therapeutic alternative in PD.
The same result was mentioned in the Johnson study16 in which a low-level GaAlAs laser at a wavelength of 830 nm was used; Treatment consisted of 30 mW, administered over a duty cycle of 100 seconds (3 J), beginning at the base of the penis and extending to the coronal sulcus over the dorsum of the penis at 0.5 cm intervals. An additional duty cycle of 100 seconds was delivered to each 0.5 cm of palpable plaque to treat the patients with symptomatic PD.
Conclusion
Despite some benefit of the laser in regard to penile curvature and pain reduction, it should be emphasized that pain usually resolves itself spontaneously with time. In a carefully selected group of men with PD, the laser appears to be safe and has effective modality treatment in the short term.
Ethical Considerations
The present study was reviewed and approved by the Research Ethics Committee of Shahid Beheshti University of Medical Sciences (IR.SBMU.RETECH.REC.1396, 285) and the Iranian center of Randomized clinical trial (identifier: IRCT201710088146N27; http://en.irct.ir/trial/8585). Full written informed consent was obtained from all patients before their inclusion in the study.
Conflict of Interests
The authors declare no conflict of interest.
Funding
This research received a specific grant from the Laser Application in Medical Science Research Center (grant No. 9328), Shahid Beheshti University of Medical Sciences, Tehran, Iran.
Acknowledgments
We are grateful to Urology and Physical Medicine and Rehabilitation ward personnel for their assistance in the current investigation.
Please cite this article as follows: Allameh F, Razzaghi M, Rayegani SM, Fallah-Karkan M, Ranjbar A, Rahavian A, et al. Laser therapy for Peyronie’s disease: a randomized control double-blind pilot study. J Lasers Med Sci. 2019;10(suppl 1):S37-S42. doi:10.15171/jlms.2019.S7.
References
- 1.Mehrsai AR, Namdari F, Salavati A, Dehghani S, Allameh F, Pourmand G. Comparison of transdermal electromotive administration of verapamil and dexamethasone versus intra‐lesional injection for Peyronie’s disease. Andrology. 2013;1(1):129–32. doi: 10.1111/j.2047-2927.2012.00018.x. [DOI] [PubMed] [Google Scholar]
- 2.Muñoz-Rangel CA, Fernandez-Vivar E, Bañuelos-Gallo RA, Gonzalez-Ojeda A, Macias-Amezcua MD, Chavez-Tostado M. et al. Minimally invasive therapy using intralesional onabotulinumtoxin A in Peyronie’s disease. Urol J. 2015;12(2):2105–10. doi: 10.22037/uj.v12i2.2894. [DOI] [PubMed] [Google Scholar]
- 3.Pavone C, D’Amato F, Dispensa N, Torretta F, Magno C. Smoking, diabetes, blood hypertension: possible etiologic role for Peyronie’s disease? Analysis in 279 patients with a control group in Sicily. Arch Ital Urol Androl. 2015;87(1):20–4. doi: 10.4081/aiua.2015.1.20. [DOI] [PubMed] [Google Scholar]
- 4.Moreland R, Nehra A. Pathophysiology of Peyronie’s disease. Int J Impot Res. 2002;14(5):406–10. doi: 10.1038/sj.ijir.3900875. [DOI] [PubMed] [Google Scholar]
- 5.Tapscott AH, Hakim LS. Office-based management of impotence and Peyronie’s disease. Urol Clin North Am. 2013;40(4):521–43. doi: 10.1016/j.ucl.2013.07.003. [DOI] [PubMed] [Google Scholar]
- 6.Mirzazadeh M, Fallahkarkan M, Hosseini J. Penile fracture epidemiology, diagnosis and management in Iran: a narrative review. Transl Androl Urol. 2017;6(2):158–66. doi: 10.21037/tau.2016.12.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Karami H, Hassanzadeh-Hadad A, Fallah-Karkan M. Comparing monotherapy with tadalafil or tamsulosin and their combination therapy in men with benign prostatic hyperplasia: a randomized clinical trial. Urol J. 2016;13(6):2920–6. doi: 10.22037/uj.v13i6.3666. [DOI] [PubMed] [Google Scholar]
- 8.Jarow JP, Lowe FC. Penile trauma: an etiologic factor in Peyronie’s disease and erectile dysfunction. J Urol. 1997;158(4):1388–90. doi: 10.1016/s0022-5347(01)64222-8. [DOI] [PubMed] [Google Scholar]
- 9.Toscano LJr, Rezende MV, Mello LF, Pires L, Paulillo D, Glina S. A prospective, randomized, single-blind study comparing intraplaque injection of thiocolchicine and verapamil in Peyronie’s disease: a pilot study. Int Braz J Urol. 2016;42(5):1005–9. doi: 10.1590/S1677-5538.IBJU.2015.0598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gaspar S, Dias J, Martins F, Lopes T. Recent surgical advances in Peyronie’s disease. Acta Med Port. 2016;29(2):131–8. doi: 10.20344/amp.6317. [DOI] [PubMed] [Google Scholar]
- 11.Claro JA, Passerotti CC, Figueiredo Neto AC, Nardozza AJr, Ortiz V, Srougi M. An alternative non-invasive treatment for Peyronie’s disease. Int Braz J Urol. 2004;30(3):199–204. doi: 10.1590/s1677-55382004000300004. [DOI] [PubMed] [Google Scholar]
- 12.Razzaghi MR, Fallah Karkan M, Ghiasy S, Javanmard B. Laser application in Iran urology: a narrative review. J Lasers Med Sci. 2018;9(1):1–6. doi: 10.15171/jlms.2018.01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.El-Sakka AI, Salabas E, Dinçer M, Kadioglu A. The pathophysiology of Peyronie’s disease. Arab J Urol. 2013;11(3):272–7. doi: 10.1016/j.aju.2013.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tsai SR, Hamblin MR. Biological effects and medical applications of infrared radiation. J Photochem Photobiol B. 2017;170:197–207. doi: 10.1016/j.jphotobiol.2017.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mussttaf RA, Jenkins DFL, Jha AN. Assessing the impact of low level laser therapy (LLLT) on biological systems: a review. Int J Radiat Biol. 2019;95(2):120–43. doi: 10.1080/09553002.2019.1524944. [DOI] [PubMed] [Google Scholar]
- 16.Johnson DE, John EJr, Harris JM, Hawkins JH. Low-level laser therapy for Peyronie’s disease Lasers in Surgery: Advanced Characterization, Therapeutics, and Systems V. International Society for Optics and Photonics. 1995 doi: 10.1117/12.209091. [DOI] [Google Scholar]
- 17.Candebat Montero LH, Miranda Reyes PL, Díaz García F, González Ferro I, Barbosa Ramos F, Codorniu Furet J. Peyronie’s disease: treatment with interferon and laser. Arch Esp Urol. 2008;61(3):413–23. doi: 10.4321/s0004-06142008000300008. [Spanish]. [DOI] [PubMed] [Google Scholar]
- 18.Williamson A, Hoggart B. Pain: a review of three commonly used pain rating scales. J Clin Nurs. 2005;14(7):798–804. doi: 10.1111/j.1365-2702.2005.01121.x. [DOI] [PubMed] [Google Scholar]
- 19.Kelâmi A. Classification of congenital and acquired penile deviation. Urol Int. 1983;38(4):229–33. doi: 10.1159/000280897. [DOI] [PubMed] [Google Scholar]
- 20.Pakpour AH, Zeidi IM, Yekaninejad MS, Burri A. Validation of a translated and culturally adapted Iranian version of the International Index of Erectile Function. J Sex Marital Ther. 2014;40(6):541–51. doi: 10.1080/0092623X.2013.788110. [DOI] [PubMed] [Google Scholar]
- 21.Hussein AA, Alwaal A, Lue TF. All about Peyronie’s disease. Asian J Urol. 2015;2(2):70–8. doi: 10.1016/j.ajur.2015.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Allameh F, Azghandi S, Karkan MF. Is Chemotherapy related with erectile dysfunction in non-urologic cancer patients? Int J Cancer Manag. 2018;11(9):e82529. doi: 10.5812/ijcm.82529. [DOI] [Google Scholar]
- 23.Levine LA, Merrick PF, Lee RC. Intralesional verapamil injection for the treatment of Peyronie’s disease. J Urol. 1994;151(6):1522–4. doi: 10.1016/s0022-5347(17)35291-6. [DOI] [PubMed] [Google Scholar]
- 24.Levine LA, Goldman KE, Greenfield JM. Experience with intraplaque injection of verapamil for Peyronie’s disease. J Urol. 2002;168(2):621–6. doi: 10.1016/s0022-5347(05)64691-5. [DOI] [PubMed] [Google Scholar]
- 25.Rehman J, Benet A, Melman A. Use of intralesional verapamil to dissolve Peyronie’s disease plaque: a long-term single-blind study. Urology. 1998;51(4):620–6. doi: 10.1016/s0090-4295(97)00700-0. [DOI] [PubMed] [Google Scholar]
- 26.Bennett NE, Guhring P, Mulhall JP. Intralesional verapamil prevents the progression of Peyronie’s disease. Urology. 2007;69(6):1181–4. doi: 10.1016/j.urology.2007.02.042. [DOI] [PubMed] [Google Scholar]
- 27.Shirazi M, Haghpanah A, Badiee M, Afrasiabi M, Haghpanah S. Effect of intralesional verapamil for treatment of Peyronie’s disease: a randomized single-blind, placebo-controlled study. Int Urol Nephrol. 2009;41(3):467–71. doi: 10.1007/s11255-009-9522-4. [DOI] [PubMed] [Google Scholar]
- 28.Hauck EW, Mueller UO, Bschleipfer T, Schmelz HU, Diemer T, Weidner W. Extracorporeal shock wave therapy for Peyronie’s disease: exploratory meta-analysis of clinical trials. J Urol. 2004;171(2):740–5. doi: 10.1097/01.ju.0000108060.30363.8d. [DOI] [PubMed] [Google Scholar]
- 29.Gao L, Qian S, Tang Z, Li J, Yuan J. A meta-analysis of extracorporeal shock wave therapy for Peyronie’s disease. Int J Impot Res. 2016;28(5):161–6. doi: 10.1038/ijir.2016.24. [DOI] [PubMed] [Google Scholar]
- 30.Fojecki GL, Tiessen S, Osther PJ. Extracorporeal shock wave therapy (ESWT) in urology: a systematic review of outcome in Peyronie’s disease, erectile dysfunction and chronic pelvic pain. World J Urol. 2017;35(1):1–9. doi: 10.1007/s00345-016-1834-2. [DOI] [PubMed] [Google Scholar]
- 31.Palmieri A, Imbimbo C, Longo N, Fusco F, Verze P, Mangiapia F. et al. A first prospective, randomized, double-blind, placebo-controlled clinical trial evaluating extracorporeal shock wave therapy for the treatment of Peyronie’s disease. Eur Urol. 2009;56(2):363–9. doi: 10.1016/j.eururo.2009.05.012. [DOI] [PubMed] [Google Scholar]
- 32.Hatzichristodoulou G, Meisner C, Gschwend JE, Stenzl A, Lahme S. Extracorporeal shock wave therapy in Peyronie’s disease: results of a placebo-controlled, prospective, randomized, single-blind study. J Sex Med. 2013;10(11):2815–21. doi: 10.1111/jsm.12275. [DOI] [PubMed] [Google Scholar]
- 33.Chung E. Peyronie’s disease and low intensity shock wave therapy: Clinical outcomes and patient satisfaction rate in an open-label single arm prospective study in Australian men. Korean J Urol. 2015;56(11):775–80. doi: 10.4111/kju.2015.56.11.775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lue TF. Peyronie’s disease: an anatomically-based hypothesis and beyond. Int J Impot Res. 2002;14(5):411–3. doi: 10.1038/sj.ijir.3900876. [DOI] [PubMed] [Google Scholar]
- 35.El-Sakka AI, Hassoba HM, Chui RM, Bhatnagar RS, Dahiya R, Lue TF. An animal model of Peyronie’s-like condition associated with an increase of transforming growth factor beta mRNA and protein expression. J Urol. 1997;158(6):2284–90. doi: 10.1016/s0022-5347(01)68236-3. [DOI] [PubMed] [Google Scholar]