Abstract
Non-alcoholic fatty liver disease (NAFLD) is one of the most common chronic liver diseases worldwide. Increasing evidence has shown that microRNAs (miRNAs) play a vital role in the progression of NAFLD. The aim of the present study was to examine the expression level and roles of miR-146a in fatty liver of high-fat diet (HFD) and ob/ob mice and fatty acid-treated hepatic cells using RT-qPCR and western blot analysis. The results showed that the expression of miR-146a was significantly decreased in the livers of high-fat diet (HFD) and ob/ob mice and free fatty acid-stimulated cells by RT-qPCR. Overexpression of hepatic miR-146a improved glucose and insulin tolerance as well as lipid accumulation in the liver by promoting the oxidative metabolism of fatty acids. In addition, the overexpression of miR-146a increased the amount of mitochondria and promoted mitochondrial respiration in hepatocytes. Similarly, inhibition of miR-146a expression levels significantly reduced mitochondrial numbers in AML12 cells as well as the expression of mitochondrial respiration related genes. Additionally, MED1 was a direct target of miR-146a and restoring MED1 abolished the metabolic effects of miR-146a on lipid metabolism and mitochondrial function. Therefore, results of the present study identified a novel function of miR-146a in glucose and lipid metabolism in targeting MED1, suggesting that miR-146a serves as a potential therapeutic target for metabolic syndrome disease.
Keywords: metabolic syndrome disease, non-alcoholic fatty liver disease, miR-146a, MED1, mitochondria
Introduction
The liver is a critical organ in vertebrate animals that is involved in multiple functions related to digestion, detoxification, fluid and electrolyte balance, and haemostasis (1). It plays vital roles in controlling lipid and glucose homeostasis to meet energy needs in response to different metabolic stresses (1). Non-alcoholic fatty liver disease (NAFLD) is one of the most common liver diseases worldwide (2). NAFLD is defined by hepatic steatosis in the absence of apparent liver disease or excessive alcohol intake. The factors leading to NAFLD remain to be fully elucidated. Insulin resistance, excess lipid accumulation and inflammation are known to be involved in the disease process (3,4). Recently, several reports have implicated hepatic mitochondrial dysfunction in the progression of NAFLD (5-7). As the cell powerhouse, mitochondria play an important role in lipid metabolism. Enhanced mitochondrial fatty acid oxidation (FAO), oxidative phosphorylation (OXPHOS) stimulation and tricarboxylic acid (TCA) cycle induction are important in overcoming the hepatic triglyceride (TG) burden (5). By contrast, mitochondrial dysfunction, concomitant with molecular and structural alterations, may result in metabolic disturbances and may contribute to NAFLD progression (8). It has been reported that hepatic mitochondria are structurally and molecularly altered in NAFLD (9-13). Studies on the correlation between mitochondrial dysfunction and NAFLD are likely to provide new strategies for treating hepatic steatosis.
MicroRNAs (miRNAs) are small non-coding RNAs that regulate gene expression at the post-transcriptional level (14). It has been shown that miRNAs participate in proliferation (15,16), apoptosis (17), inflammation (18), cancer (19) and metabolism (20,21). miR-146a-5p (miR-146a), first described in humans by Taganov et al in 2006 (22), is a member of the miR-146 family. A large proportion of the literature regarding miR-146a has focused on inflammation (22,23). In addition, mounting evidence shows that miR-146a plays important roles in cardiovascular disease (24-26). Recently, Jin et al reported that miR-146a was significantly decreased in non-alcoholic steatohepatits (NASH) and that overexpression of miR-146a was capable of improving NASH by targeting HDMCP (27). In addition, miR-146a has been found to play an important role in liver cancer (28-30). However, there are few literature reports regarding whether miR-146a plays a role in the development of insulin resistance and NAFLD. Therefore, we aimed to explore the expression level of miR-146a in fatty liver and fatty acid-treated hepatic cells, and the relationship between miR-146a and fatty liver and fatty acid oxidation, thus providing new insight into the mechanism and treatment of fatty liver.
In this study, we found that miR-146a was decreased in the liver of NAFLD mice and FFA-stimulated cells, and that miR-146a improved glucose metabolism. Moreover, overexpression of hepatic miR-146a attenuated lipid accumulation in the liver of high fat diet (HFD) mice by increasing the mitochondrial density and respiratory capacity. Mechanistic studies revealed that miR-146a regulated mitochondrial function through its direct target gene. In conclusion, we identified a novel function of miR-146a showing that miR-146a could alleviate the metabolic disease in HFD mice by targeting MED1 and enhancing mitochondrial function. These findings indicate that miR-146a is a critical regulator of glucose and lipid homeostasis, and may serve as a potential therapeutic target for hepatic steatosis.
Materials and methods
Ethics statement
All animal protocols were approved by the Animal Experimental Ethical Inspection Committee of Bengbu Medical College (approval no. DWLL-2017-046). All in vivo experiments described in this study were in accordance with institutional guidelines for the care and use of animals.
Animals and treatments
The mice used for in vivo studies were all males aged 6-10 weeks. All 43 mice were C57BL/6 and were maintained in specific pathogen-free conditions under a consistent light-dark cycle (lights on at 6:00 a.m. and off at 6:00 p.m.) with free access to water and normal chow diet (SLACOM) Mice with similar ages or from the same litters had priority of use. High-fat diet (D12492, Research Diets) was used to feed 10 eight-week-old mice for 3 months. During the experiments, the mice were monitored daily. Any mice with significantly abnormal signs of rapid weight loss, inability to eat or drink, clinical symptomatology, toxicity, or unresponsiveness were recorded, and the data from these mice were excluded for statistical analysis following the laboratory animal welfare guidelines of Bengbu Medical College.
Bioinformatics analysis
The website of TargetScan (http://www.targetscan.org/vert_72/) was used to predict the related targets of miR-146a.
Metabolic measurements
Glycogen and triglyceride levels of liver of mice were measured with a Glycogen Assay kit (BioVision, K646-100) and triglyceride assay kit (TriglycerideAssaykit, Sigma Aldrich Co.), respectively. FFA concentrations and insulin levels of serum of mice were analyzed with the NEFA C test kit (Wako Pure Chemical Industries Ltd.) and insulin ELISA kit (Crystal Chem Inc.). ATP and triglyceride levels of AML12 cells were determined with the Cell Titer-Glo Luminescent kit (Promega) and triglyceride assay kit (Triglyceride Assay kit), respectively. All experimental procedures were performed according to the manufacturer's instructions.
Oxygen consumption and glycolysis were measured using a XF24 instrument (Seahorse Bioscience Inc.). AML12 cells were cultured in 20 wells of an XF24 microplate. The Mito Stress Test kit was obtained from Seahorse Bioscience. The oxygen consumption rate (OCR) was determined following the manufacturer's instructions. Furthermore, the basal respiration and respiratory capacity were calculated as described previously (31).
Prior to the fasting blood glucose tests, mice were deprived of food for 6 h. Blood glucose levels were measured at the indicated time points using a glucometer glucose test strip (LifeScan). For the glucose tolerance test, the mice were deprived of food for 16 h, and then given 2 g glucose per kilogram of body weight intraperitoneally. Blood (~10 µl) glucose levels were detected from tail vein after 15, 30, 60 and 120 min using a glucometer glucose test strip (LifeScan). For the insulin tolerance test, mice were deprived of food for 6 h, and then given 1.25 units insulin per kilogram of body weight intraperitoneally. Blood (~10 µl) was collected from the tail and glucose levels were measured at the indicated time points by a method similar to the one described above for the glucose tolerance test.
Adenovirus recombination and administration
The recombinant adenoviruses for miR-146a were generated using the AdEasy Adenoviral Vector System (Stratagene) in 293A cells. Briefly, the stem loop sequence of miR-146a (5′-AGCTCTGAGAACTGAATTCCATGGGTTATATCAATGTCAGACCTGTGAAATTCAGTTCTTCAGCT-3′) and NC (5′-TCACAACCTCCTAGAAAGAGTAGATCACAACCTCCTAGAAAGAGTAGA-3′) was cloned into pShuttle-CMV, and then pShuttle-miR-146a was recombined into the pAd-Easy1 vector. The recombined vector was then transfected into 293A cells to obtain the first generation virus. The virus was further amplified in 293A cells and purified by caesium chloride gradient centrifugation (100,000 × g, 4°C, 90 min) to obtain a high purity adenovirus. Then 1×109 PFU of the virus were administered to each mouse through tail vein injection. A total of 27 mice were used for adenovirus administration. After 14 days of virus injection, mice were intraperitoneally injected with 0.5% pentobarbital sodium (45 mg/kg) and blood was taken from the heart. Tissues were harvested without restricting the mice to food or water, snap-frozen in liquid nitrogen immediately after resection and stored at -80°C.
Plasmid construction
pcDNA3.1 and pmirGLO were used to generate pcDNA3.1-MED1 and pmirGLO-MED1-3′UTR, respectively. Then, miR-146a primers containing EcoRI/NotI or SacI/XbaI cloning sites were used in PCRs for the generation of pcDNA3.1-MED1 (F: 5′-ATGAAGGCTCAGGGGGAAACCGAGG-3′ R: 5′-CTAATTGCCAATCAGGGCCA-3′) or pmirGLO-MED1-3′UTR (F: 5′-CCTAACTTTCTAAACAGACA-3′ R: 5′-ATGCTAACTCCAACAACCTG-3′). Mutation of MED1-3′UTR was performed by a KOD-Plus mutagenesis kit (Toyobo) following the manufacturer's instructions. The binding sites for miR-146a were changed to 5′-TCAAGAG-3′. All the plasmids constructed in this study were confirmed by DNA sequencing. MiR-146a mimic (5′-UGAGAACUGAAUUCCAUGGGUU-3′) and inhibitor (5′-AACCCAUGGAAUUCAGUUCUCA-3′) were purchased from GenePharma. Nonsense sequences were used as mimic control (5′-UCACAACCUCCUAGAAAGAGUAGA-3′) and antagomir control (5′-UCUACUCUUUCUAGGAGGUUGUGA-3′).
Cell culture and reporter assay
A mouse hepatocyte cell line (AML12) (SCSP-550, Stem Cell Bank, Chinese Academy of Sciences) was cultured in Dulbecco's modified Eagle's medium supplemented with 10% FBS, 1 mM glutamine, 100 U/ml penicillin, and 100 mg/ml streptomycin. Cells were maintained in a humidified incubator at 37°C and 5% CO2 atmosphere.
A dual luciferase reporter assay was performed according to the manufacturer's instructions. miR-146a mimics (100 nM) or control (100 nM) were co-transfected with pmirGLO-MED1-3′UTR WT or pmirGLO-MED1-3′UTR MUT in AML12 cells. Forty-eight hours after transfection, the cells were lysed in 1.5 ml centrifuge tube and relative luciferase activity was analyzed with the Dual-Luciferase Reporter Assay System (Promega) on a luminometer (Promega).
Histological analysis
For H&E staining, liver tissues were fixed with 4% paraformaldehyde. Individual lobes of liver biopsy material were placed in processing cassettes, dehydrated through a serial alcohol gradient, and embedded in paraffin wax blocks. Before immunostaining, liver tissue sections were dewaxed in xylene, rehydrated through decreasing concentrations of ethanol, and washed in PBS prior to staining with H&E. For Oil-red O staining, the fixed liver tissue was dehydrated by sucrose solution. Liver sections (8 µm) were stained with Oil-red O for 8 min, differentiated with 75% ethanol after PBS buffer washing, and stained with hematoxylin for 90 sec. Images were obtained with a light microscope (Olympus) and then quantified using Image Pro Plus software (version 6.0).
RT-qPCR and mtDNA copy number analysis
RNA from cells or tissues was extracted with TRIzol reagent (Invitrogen). cDNA was synthesized by reverse transcription of mRNA using the PrimeScript RT reagent kit (Takara). For miRNA detection, total RNA was reverse-transcribed using a miR-146a-specific stem-loop primer (Applied Biosystems) and subsequently measured via a real-time PCR using miR-146a-specific primer (Applied Biosystems). qPCR was performed on a 7900 HT Fast Real-time PCR System (Applied Biosystems) using SYBR-Green (Roche). Relative expression levels of miR-146a and mRNA were calculated with normalization to U6 or GAPDH values, respectively, using the 2−ΔΔCq method (32). The mtDNA copy number was determined by comparing the ratio of mtDNA to nDNA(18S rRNA) by RT-qPCR as described previously (33). The PCR amplification procedure was: Step 1: 95°C for 5 min; Step 2: 95°C for 25 sec; Step 3: 60°C for 20 sec; Step 2-3 repeats for 40 cycles. Primer sequences used are shown in Table SI.
Western blotting
MED1 proteins were enriched by immunoprecipitation before detection using western blot analysis. Cells or tissues were lysed in IP lysis buffer (Beyotime, P0013) supplemented with phosphatase inhibitor for 30 min on ice. Subsequently, the cell lysate was centrifuged, and the supernatant was incubated with the indicated antibodies and Protein A/G PLUS-agarose (Santa Cruz Biotechnology sc-2003) at 4°C overnight. The immunoprecipitates were separated by SDS-PAGE (7.5%). After electrophoresis, the protein was transferred onto nitrocellulose membranes with 300 mA for 3 h and blocked by 5% skim milk then probed with the various primary antibodies (MED1, Cell Signaling Technology, no. 51613, rabbit, polyclonal antibodies, 1:1,000; Histone H3, Ab clonal: A2348, rabbit, polyclonal antibodies, 1:1,000) at 4°C overnight. On the second day, the membranes were washed with TBST buffer and probed with secondary antibodies (Jackson ImmunoResearch, 111-035-003, Peroxidase AffiniPure goat anti-rabbit IgG (H+L), polyclonal antibodies) diluted with 5% skim milk at 1:1,500. Detection was performed via measurement of the chemiluminescent signal with SuperSignal Ultra. Protein quantification was analysed by Quantity One software (Bio-Rad) and the intensity values were normalized to β-actin. Antibodies against MED1 (Cell Signaling Technology, no. 51613, 1:1,000) and Histone H3 (Ab clonal: A2348) were used for western blotting.
Statistical analysis
Differences among multiple groups were assessed using One-way analysis of variance followed by Tukey's post-hoc comparisons. Differences between two groups were tested by the Student's t-test. The software used were Excel and GraphPad Prism 5.0 (GraphPad). Data are presented as mean ± SD. P<0.05 was considered statistically significant. All experiments were repeated at least twice, and representative results are shown.
Results
miR-146a is decreased in the liver of NAFLD mice and FFA stimulated cells
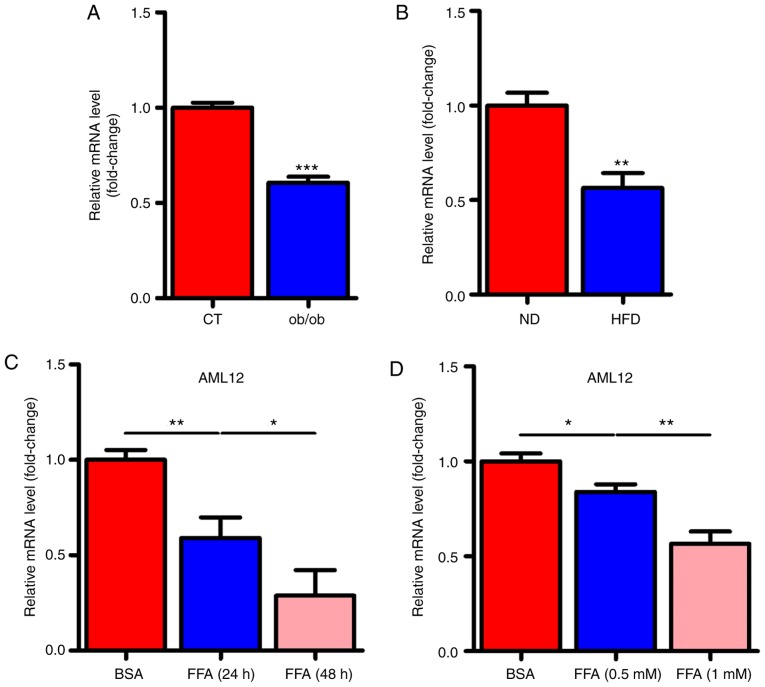
To determine whether miR-146a played a role in the development of metabolic syndrome, we examined the expression levels of miR-146a in the liver of ob/ob and HFD mice. Notably, the expression levels of miR-146a were all significantly decreased in the liver of both ob/ob mice (Fig. 1A) and HFD mice (Fig. 1B). Given that free fatty acid (FFA) in the liver is an important signalling factor, we used FFA to treat AML12 cells to verify whether FFA affected miR-146a. As expected, FFA treatment significantly decreased the expression level of miR-146a in AML12 cells in a time- and dose-dependent manner (Fig. 1C and D). These results indicated that miR-146a is FFA-responsive and may be involved in the development of metabolic syndrome.
Figure 1.
The expression level of miR-146a is decreased in the liver of NAFLD mice and FFA stimulated cells. (A) The relative expression level of miR-146a in the liver of ob/ob mice or control (CT) mice (n=3). (B) The relative expression level of miR-146a in the liver of high-fat diet mice (HFD) or normal diet mice (ND) (n=3). (C) The relative expression level of miR-146a in AML12 cells treated with BSA or FFA for 24 or 48 h (n=3). (D) The relative expression level of miR-146a in AML12 cells treated with BSA, FFA (0.5 mM) or FFA (1 mM) (n=3). Means ± SD are shown. *P<0.05; **P<0.01; ***P<0.001.
Hepatic miR-146a regulates glucose homeostasis
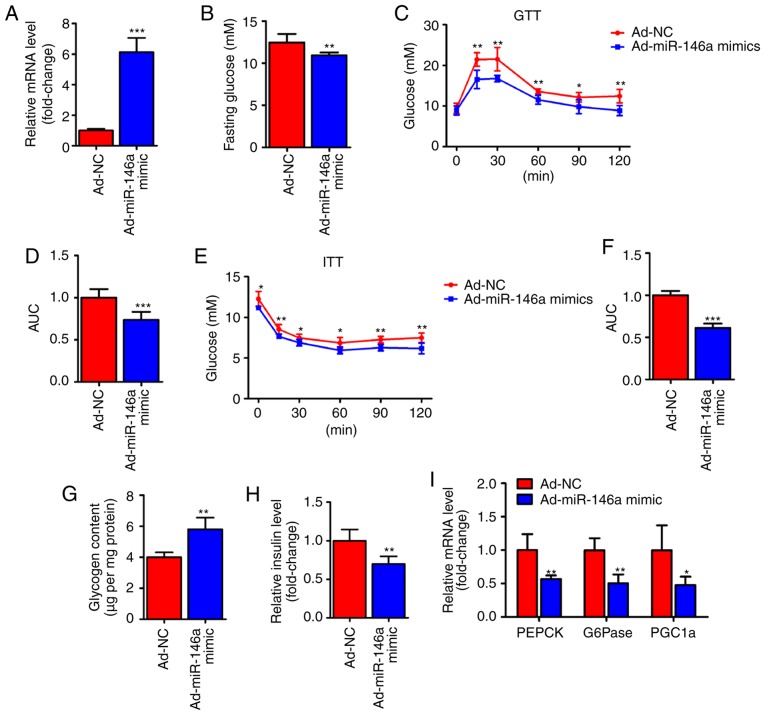
To test whether miR-146a is capable of regulating hepatic glucose metabolism, an adenoviral miR-146a (Ad-miR-146a mimic) was used to overexpress miR-146a in the livers of HFD mice (Fig. 2A). Notably, we found that the fasting serum glucose level was significantly decreased on the fifth day after the infection of Ad-miR-146a mimic. Furthermore, overexpression of miR-146a could improved the glucose and insulin sensitivity of HFD mice (Fig. 2C-F), further indicating that miR-146a plays an important role in glucose metabolism. The liver mainly modulates the body's glucose homeostasis through the regulation of hepatic glycogen synthesis and gluconeogenesis (1). Therefore, we analyzed hepatic glycogen content and gluconeogenesis-associated genes in the liver of two groups using RT-qPCR. Consistent with previous results (Fig. 2B-F), glycogen content analysis revealed that the glycogen level was increased in the livers of miR-146a overexpressing HFD mice (Fig. 2G) and the serum insulin level was also significantly decreased after the overexpression of hepatic miR-146a by Ad-miR146a mimic (Fig. 2H). Accordingly, the expression levels of gluconeogenic genes (PEPCK, G6Pase and PGC1a) were significantly reduced in the livers of miR-146a-overex-pressing HFD mice (Fig. 2I). Together, these results indicate that miR-146a regulates glucose metabolism in the liver and that overexpression of hepatic miR-146a may alleviate type II diabetes.
Figure 2.
miR-146a regulates glucose metabolism in the liver of high-fat diet mice. (A) The relative miR-146a expression level in the liver of HFD mice infected with Ad-miR-146a mimic or Ad-NC (n=6). (B) The fasting glucose level of serum in HFD mice infected with Ad-miR-146a mimic or Ad-NC (n=6). (C-F) Glucose tolerance test (C) and insulin tolerance test (E) analysis of HFD mice infected with Ad-miR-146a mimic or Ad-NC. The area under the curve (AUC) of GTT (D) and ITT (F) were calculated (n=6). (G) Glycogen content analysis in the livers of HFD mice infected with Ad-miR-146a mimic or Ad-NC (n=6). (H) Serum insulin levels in HFD mice infected with Ad-NC or Ad-miR-146a mimic (n=6). (I) The expression levels of gluconeogenic genes (PEPCK, G6Pase and PGC1a) in the livers of HFD mice infected with Ad-miR-146a mimic or Ad-NC. Means ± SD are shown. *P<0.05; **P<0.01; ***P<0.001.
miR-146a improves lipid metabolism in the liver of HFD mice
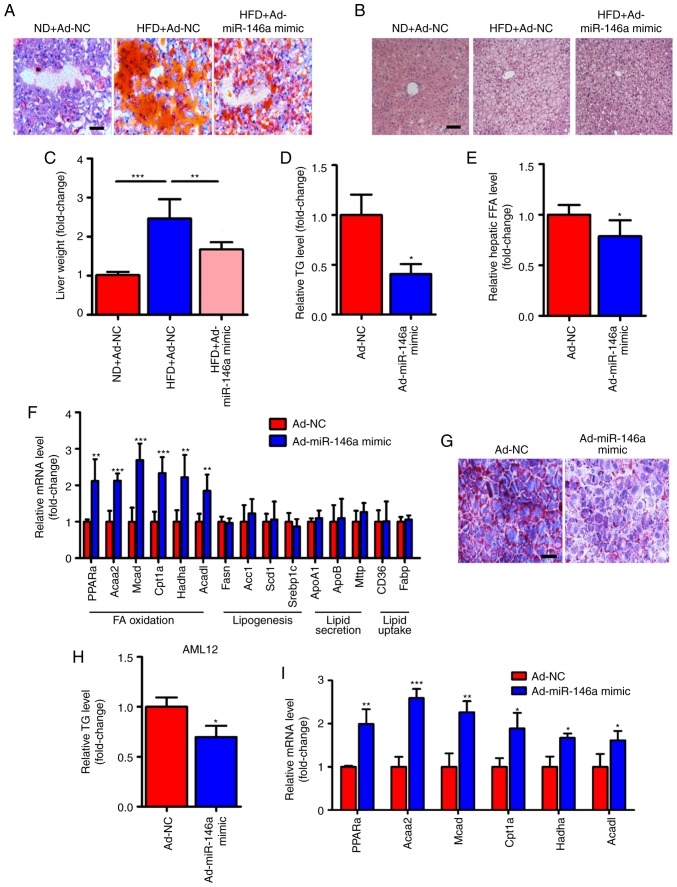
In addition to the improvement of glucose metabolism, we also found that the lipid metabolism was also improved in HFD mice after hepatic miR-146a overexpression by infection with Ad-miR-146a mimic. We observed that lipid accumulation was markedly reduced in the livers of HFD mice infected with Ad-miR-146a mimic by Oil-red O staining (Fig. 3A) and H&E staining (Fig. 3B). Similarly, the liver weight and hepatic triglycerides were all significantly decreased after the overexpression of hepatic miR-146a in HFD mice (Fig. 3C and D). Moreover, the overexpression of hepatic miR-146a also significantly decreased hepatic FFA levels (Fig. 3E). NAFLD develops as a consequence of metabolic dysregulation of de novo lipogenesis, lipid uptake, fatty acid oxidation, and lipid secretion (34). We further analyxed genes related to lipid metabolism in the liver. Of note, we found that the mRNA expression levels of genes related to fatty acid oxidation (PPARa, Acaa2, Mcad, Cpt1a, Hadha and Acadl) were all significantly increased but not for genes related to de novo lipogenesis (Fasn, Acc1, Scd1 and Srebp1c), lipid uptake (CD36 and Fabp), and lipid secretion (ApoA1, ApoB and Mttp) (Fig. 3F). Additionally, we found that overexpression of miR-146a reduced lipid accumulation in AML12 cells by transfection of miR-146a mimic (Fig. 3G and H). Moreover, signicantly increased levels of fatty acid oxidation-related genes (PPARa, Acaa2, Mcad, Cpt1a, Hadha and Acadl) were found in AML12 cells after transfection of miR-146a mimic (Fig. 3I). These results suggest that miR-146a may improve hepatic lipid metabolism and may play an important role in fatty acid oxidation.
Figure 3.
miR-146a attenuates lipid accumulation in the livers of HFD mice. (A-C) Representative images of Oil-red O staining (A) and H&E staining (B) of liver sections and relative liver weight (n=5) (C) of normal diet fed mice (ND) infected with Ad-NC, HFD mice infected with Ad-NC or Ad-miR-146a mimic. Scale bar, 100 µm. (D) Relative hepatic triglyceride (TG) levels of HFD mice infected with Ad-NC or Ad-miR-146a mimic (n=3). (E) Relative hepatic free fatty acid (FFA) in the HFD mice infected with Ad-NC or Ad-miR-146a mimic (n=5). (F) The relative mRNA levels of genes related to fatty acid oxidation (PPARa, Acaa2, Mcad, Cpt1a, Hadha and Acadl), de novo lipogenesis (Fasn, Acc1, Scd1 and Srebp1c), lipid secretion (ApoA1, ApoB and Mttp) and lipid uptake (CD36 and Fabp) in the livers of HFD mice infected with Ad-NC or Ad-miR-146a mimic (n=5 to 6). (G) Representative images of Oil-red O staining of AML12 cells transfected with NC or miR-146a mimic. Scale bar, 100 µm. (H) Quantitative analysis of cellular TAG content in AML12 cells transfected with NC or miR-146a mimic (n=3). (I) The relative mRNA levels of genes related to fatty acid oxidation in AML12 cells transfected with NC or miR-146a mimic (n=3). Means ± SD are shown. *P<0.05; **P<0.01; ***P<0.001.
miR-146a regulates mitochondrial function of the hepatocytes
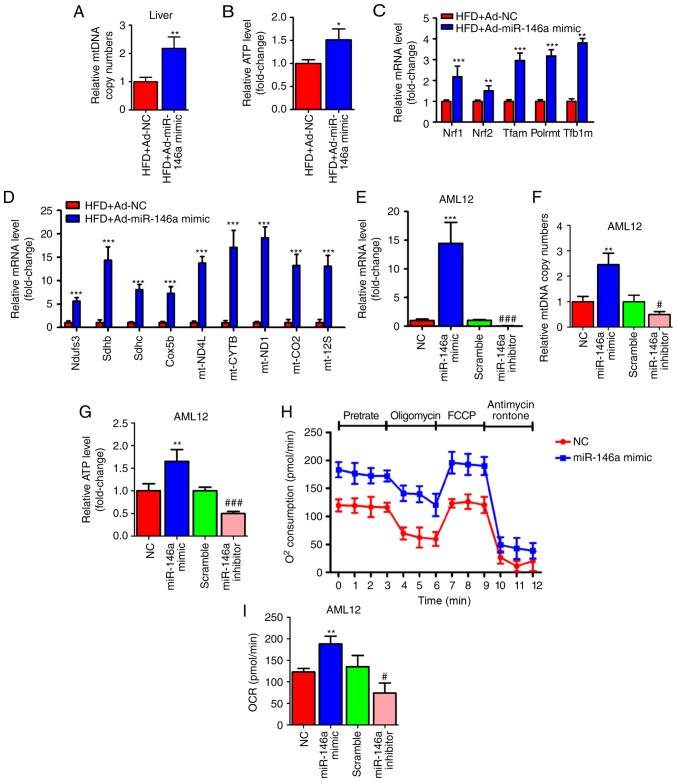
Since we found that the expression level of fatty acid oxidation-related genes in the liver was significantly increased after the overexpression of miR-146a (Fig. 3F) and mitochondria play a key role in energy metabolism by generating most of the energy via the tricarboxylic acid cycle and oxidative phosphorylation (5), we examined whether the overexpression of hepatic miR-146a could regulate mitochondrial function in the liver of HFD mice. As expected, we found that the mtDNA copy numbers were significantly increased in the livers of HFD mice after infection of Ad-miR-146a-mimic (Fig. 4A), which indicated that miR-146a could increase the amount of mitochondria. Consistently, overexpression of hepatic miR-146a could increase the ATP level in the liver of HFD mice (Fig. 4B). Given that the nuclear respiratory factors, Nrf1 and Nrf2 are the key transcription factors required for mitochondrial respiration, mtDNA transcription and replication (35-38), we then measured the mRNA levels of Nrf1 and Nrf2 and their downstream target genes (Tfam, Plormt, Tfb1m), related to mtDNA maintenance (39-41), and found that the expression levels of these genes were all significantly increased in the liver after infection with Ad-miR-146a-mimic in HFD mice (Fig. 4C). Moreover, the levels of genes involved in the oxidative phosphorylation (OXPHOS) pathway in both the nucleus (Ndufs3, Sdhb, Sdhc, Cox5b) and mitochondria (ND4L, CYTB, ND1, COII, 12SrRNA) (42-44) were significantly increased in the liver after HFD mice were infected with Ad-miR-146a mimic (Fig. 4D). Furthermore, chemosynthetic mimic and inhibitor of miR-146a were used to up- and downregulate, respectively, the expression of miR-146a in AML12 cells (Fig. 4E). Consistent with the previous results, miR-146a mimic and inhibitor increased and decreased mtDNA copy numbers and ATP levels in AML12 cells, respectively (Fig. 4F and G). Additionally, a Seahorse Bioscience extracellular flux analyser was used to investigate the cellular oxygen consumption rate, an indicator of mitochondrial oxidation. As shown in Fig. 4H and I, bioenergetics profiles and the calculated oxygen consumption rates indicated that overexpression of miR-146a was able to increase the basal and maximal mitochondrial respiratory capacity, which was consistent with the miR-146a overexpression-mediated induction of fatty acid oxidation in the liver of HFD mice (Fig. 3F). These results indicated that miR-146a increased the amount of mitochondria and promote mitochondrial oxidative phosphorylation and ATP production.
Figure 4.
miR-146a regulates mitochondrial function of the hepatocytes. (A and B) Relative mtDNA copy numbers (A) and ATP levels (B) in the livers of high fat diet mice (HFD) infected with Ad-NC or Ad-miR-146a mimic (n=5). (C) The relative mRNA expression of nuclear respiratory factors (Nrf1 and Nrf2) and their downstream target genes (Tfam, Plormt and Tfb1m) in the liver of high fat diet mice (HFD) infected with Ad-NC or Ad-miR-146a-mimic (n=5). (D) Relative mRNA expression levels of oxidative phosphorylation pathway-related genes in nucleus (Ndufs3, Sdhb, Sdhc and Cox5b) and mitochondria (ND4L, CYTB, ND1, COII and 12 SrRNA) in the liver of high-fat diet mice (HFD) a infected with Ad-NC or Ad-miR-146a mimic (n=5). (E-G) The relative expression level of miR-146a (E) mtDNA copy numbers (F) and ATP levels (G) in AML12 cells transfected with NC or miR-146a-mimic, or transfected with scramble control or miR-146a inhibitor (n=3). (H) Oxygen consumption of AML12 cells transfected with miR-146a mimic or NC was analysed with a Seahorse XF24 extracellular flux analyser (n=3). (I) Basal respiration and respiratory capacity oxygen consumption rates were measured and calculated (n=3). Means ± SD are shown. *P<0.05; **P<0.01; ***P<0.001, vs. NC, #P<0.05; ###P<0.001, vs. scramble control.
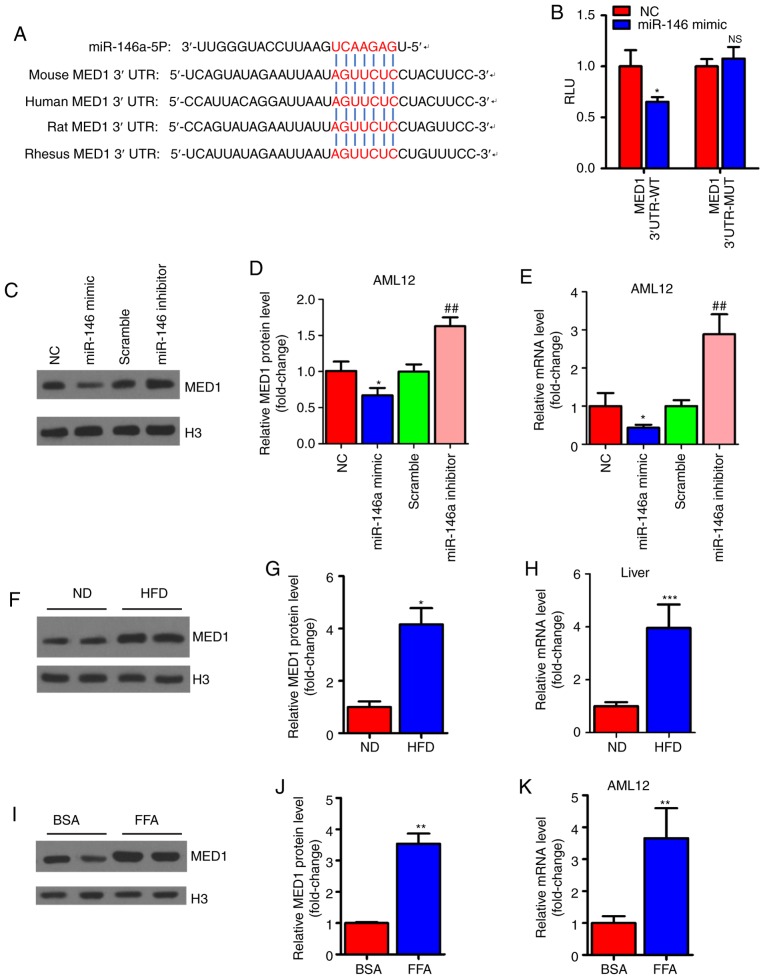
MED1 is a direct target gene of miR-146a
To explore the molecular mechanism involved in the regulation of mitochondrial function by miR-146a, we predicted the target genes of miR-146a using TargetScan (http://www.targetscan.org/vert_72/) to identify related putative targets. The results showed that MED1, a key subunit of the mammalian mediator complex involved in mitochondrial function (45), was predicted as a target gene of miR-146a and contained a conserved binding site (Fig. 5A). For validation, the mouse-complete MED1 3′UTR containing the microRNA response element (MRE) sequence was cloned into a luciferase reporter. Reporter activity analysis revealed that overexpression of miR-146a could decrease the luciferase activity of the reporter but had no effect on the reporter containing the mutated MED1 3′UTR (Fig. 5B). Similarly, the mRNA and protein levels were significantly decreased after miR-146a mimic administration but increased with miR-146a inhibitor transfection in AML12 cells (Fig. 5C-E). As we expected, we found that the protein and mRNA expression levels of MED1 were increased in the livers of HFD mice (Fig. 5F-H). Furthermore, the mRNA levels and protein levels of MED1 were increased in FFA-treated AML12 cells in vitro (Fig. 5I-K). These results indicate that MED1 is a direct target gene of miR-146a in murine hepatocytes.
Figure 5.
miR-146a can directly target MED1. (A) Sequence alignment of MED1 3′-UTR and miR-146a in multiple species with seed sequences highlighted in red. (B) The luciferase activity of the reporter containing the MED1 3′UTR-WT or MED1 3′UTR-MUT was determined in AML12 cells transfected with NC or miR-146a mimic (n=3). (C-E) The protein level of MED1 (C) and densitometry analysis (D) and relative mRNA level (E) of MED1 in AML12 cells transfected with NC or miR-146a mimic, or transfected with scramble control or miR-146a inhibitor (n=3). (F-H) The protein level of MED1 (F) densitometry analysis (G) and relative mRNA level (H, n=5) of MED1 in the liver of normal diet mice (ND) or high fat diet mice (HFD). (I-K) The protein level of MED1 (I) densitometry analysis (J) and relative mRNA level (K, n=3) of MED1 in AML12 cells treated with BSA or FFA (1 mM). Means ± SD are shown. *P<0.05; **P<0.01; ***P<0.001, vs. NC, ##P<0.01, vs. scramble.
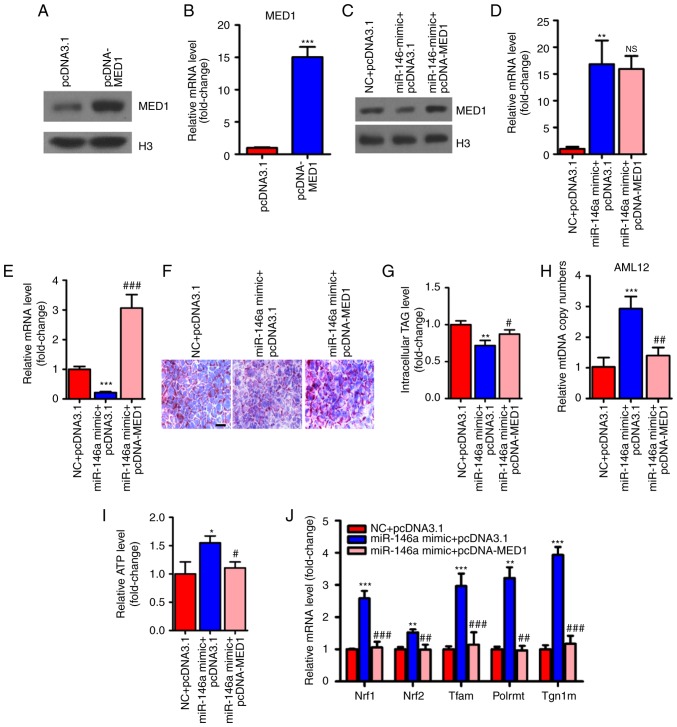
MED1 mediates the metabolic action of miR-146a
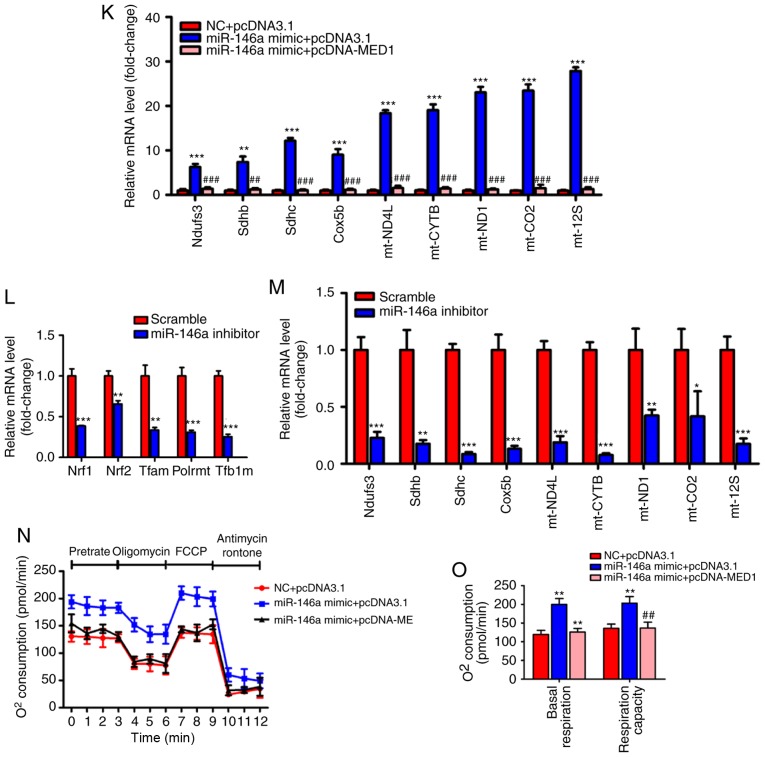
To determine whether MED1 mediated the effect of miR-146a on glucose and lipid metabolism and mitochondrial function, we employed pcDNA3-MED1, which could not be repressed by miR-146a due to the absence of the MED1 3′UTR (Fig. 6A-E). As expected, transient transfection and overexpression of MED1 abolished the effect of miR-146a on lipid accumulation in AML12 cells (Fig. 6F and G). Furthermore, restoring MED1 could also compensate the number of mitochondria and ATP levels after miR-146a mimic administration in AML12 cells (Fig. 6H and I). Consistent with these findings, MED1 attenuated the effect of miR-146a on the expression levels of Nrf1, Nrf2 and their downstream target genes (Fig. 6J), and genes involved in the oxidative phosphorylation (OXPHOS) pathway in both the nucleus (Ndufs3, Sdhb, Sdhc and Cox5b) and mitochondria (ND4L, CYTB, ND1, COII and 12SrRNA) (Fig. 6K) in AML12 cells. In addition, we found that Nrf1, Nrf2 and their downstream target genes and OXPHOS pathway-related genes were significantly decreased with inhibiting the expression of miR-146a (Fig. 6L and M). Notably, the rescue of MED1 abolished the effect of miR-146a on basal and maximal mitochondrial respiratory capacity (Fig. 6N and O). Given that the restoration of MED1 expression could almost fully abrogate the effect of miR-146a on mitochondrial function and lipid metabolism in AML12 cells. We suggest that MED1 may be the primary target gene of miR-146a that mediates the effect of miR-146a on mitochondrial function and lipid metabolism.
Figure 6.
Restoration of MED1 inhibits the metabolic action of miR-146a. (A and B) The protein levels (A) and relative mRNA levels (B, n=3) of MED1 in AML12 cells transfected with pcDNA3.1 or pcDNA-MED1. (C-E) The protein level of MED1 (C) and relative mRNA levels of miR-146a (D, n=3) and MED1 (E, n=3) in AML12 cells cotransfected with NC and pcDNA3.1, miR-146a mimic and pcDNA3.1 or miR-146a mimic and pcDNA-MED1. (F and G) Representative images of Oil-red O staining (F) and cellular TG content analysis (G) of AML12 cells transfected with NC and pcDNA3.1, miR-146a mimics and pcDNA3.1 or miR-146a mimic and pcDNA-MED1. Scale bar, 100 µm. (H and I) The relative mtDNA copy numbers (H) and ATP levels (I) in AML12 cells co-transfected with NC and pcDNA3.1, miR-146a mimic and pcDNA3.1 or miR-146a mimic and pcDNA-MED1 (n=3). (J) The relative mRNA expression level of nuclear respiratory factors (Nrf1 and Nrf2) and their downstream target genes (Tfam, Plormt and Tfb1m) in AML12 cells co-transfected with NC and pcDNA3.1, miR-146a mimic and pcDNA3.1 or miR-146a mimic and pcDNA-MED1 (n=3). Restoration of MED1 inhibits the metabolic action of miR-146a. (K) Relative mRNA expression of oxidative phosphorylation pathway-related genes in the nucleus (Ndufs3, Sdhb, Sdhc and Cox5b) and in mitochondria (ND4L, CYTB, ND1, COII, and 12SrRNA) in AML12 cells co-transfected with NC and pcDNA3.1, miR-146a mimic and pcDNA3.1 or miR-146a mimic and pcDNA-MED1 (n=3). (L) The relative mRNA expression level of nuclear respiratory factors and their downstream target genes in AML12 cells transfected with Scramble control or miR-146a inhibitor (n=3). (M) Relative mRNA expression of oxidative phosphorylation pathway-related genes in the nucleus and in mitochondria in AML12 cells transfected with Scramble control or miR-146a inhibitor (n=3). (N) Oxygen consumption of AML12 cells co-transfected with NC and pcDNA3.1, miR-146a mimic and pcDNA3.1 or miR-146a mimic and pcDNA-MED1 were analyzed with a Seahorse XF24 extracellular flux analyser (n=3). (O) Basal respiration and respiratory capacity oxygen consumption rates were measured and calculated (n=3). Means ± SD are shown. *P<0.05; **P<0.01; ***P<0.001 (vs. control group transfected with NC and pcDNA3.1), #P<0.05; ##P<0.01; ###P<0.001 (vs. the group transfected with miR-146a mimic and pcDNA3.1).
Discussion
NAFLD, one of the most common chronic liver diseases worldwide, has recently raised much interest. The factors leading to NAFLD remain to be fully elucidated. Insulin resistance, excess lipid accumulation, inflammation and mitochondrial dysfunction are known to be involved in the disease process (46). MicroRNAs (miRNAs) are small non-coding RNAs that regulate gene expression at the post-transcriptional level. Previous findings have demonstrated that microRNAs play an important role in the progression of metabolic disorders. An early study evaluated 4 upregulated miRNAs (miR-103, miR-31, miR-107 and miR-126-3p) and 2 downregulated miRNAs (miR-100 and miR-29c) in the liver tissue of ob/ob mice and streptozotocin-induced diabetic mice (47). In addition, several studies have shown that miR-34a is significantly elevated and miR-21 and miR-29 were significantly downregulated in the liver of ob/ob mice and NAFLD patients (48-54). Therefore, different microRNAs play different roles in the development of NAFLD while the decrease of microRNA is not a general trend. Identification of essential miRNAs that regulate metabolic processes and investigation of the specific targets of these functional miRNAs will not only increase our knowledge but also provide new strategies for treating metabolic disorders.
Findings of the present study showed that miR-146a play an important role in hepatic glucose and lipid metabolism. We found that miR-146a was decreased in the liver of NAFLD mice and FFA-stimulated cells. He et al reported that miR-146b may be involved in target delivery to hepatocytes to improve NAFLD by acting as a lipid cleaner and inflammation suppressor (55). MiR-146b and miR-146a belong to the same microRNA family with two base inconsistencies in the mature sequence, indicating that the miR-146 family plays an important role in the progression of NAFLD by regulating different signalling pathways. In addition, it is well known that excessive FFA accumulation in the liver leads to an increase in oxidative stress, which is thought to be critical for the pathogenesis of hepatic steatosis (56). Therefore, miR-146a may be involved in the FFA-oxidative stress-fatty liver axis. Azizi et al reported that miR-146a was significantly decreased under increased oxidative stress conditions (57). Therefore, FFA may inhibit the expression level of miR-146a by increasing the level of oxidative stress, but further investigation is needed. In addition, overexpression of hepatic miR-146a may improve glucose tolerance and insulin tolerance in HFD mice. Interestingly, we also found that overexpression of miR-146a could increase mitochondrial density and respiration capacity in AML12 cells.
Mechanistic studies revealed that MED1 is the direct target of miR-146a, and it mediated the regulation of miR-146a on lipid metabolism and mitochondrial function. MED1 is regarded as a key subunit of the mammalian mediator complex. An earlier study showed that MED1 was essential for PPAR-gamma stimulated adipogenesis in mouse embryonic fibroblasts (58). Furthermore, Chen et al reported that muscle-specific MED1 knockout mice showed enhanced insulin and glucose tolerance and resistance to high-fat diet-induced obesity through increasing mitochondrial density and respiratory capacity (59), which is consistent with our findings. Similarly, loss of MED1 in C2C12 cells could trigger mitochondrial biogenesis (60). In addition, Bai et al found that MED1 was required for high-fat diet-induced and PPARγ-induced hepatic steatosis and that loss of MED1 protects against fatty liver under these conditions (61). On the one hand, they found that hepatic MED1 deficiency impaired PPARγ-induced adipogenic gene expression. On the other hand, they showed that FGF21, which increases energy expenditure by increasing fatty acid oxidation, was significantly increased in the liver of MED1 hepatic specific knockout mouse after PPARγ overexpression (61). Of note, MED1 is well known to be a coactivator of PPARα and is necessary for PPARα signaling (62,63). Additionally, the expression levels of these factors seem to be slightly conflicting after the overexpression of hepatic miR-146a. Therefore, the upregulation of fatty acid oxidation-related genes by overexpression of hepatic miR-146a may not occur mainly through PPARα but other genes, such as FGF21 or HFN4α. Notably, whether MED1 mediates the regulation of miR-146a on glucose metabolism still needs to be further explored. It has been reported that there are changes in mitochondrial morphology in diabetic patients or insulin resistance animal models (64). Furthermore, swollen mitochondria and decreased matrix density were observed in rat models of insulin resistance and hypertension (65). These observations strengthen the relationship between mitochondrial function and insulin resistance.
Previous findings have shown that simple steatosis is associated with an upregulation in mitochondrial oxidative function (66-68). However, alterations in mitochondrial energy metabolism could not completely prevent the development of NAFLD (69). Furthermore, reductions in mitochondrial respiratory activity and rates of ATP synthesis were reported in both mice and human models of long-term overnutrition that led to progressive and severe liver disease (8-12). Therefore, it is possible to improve NAFLD by enhancing mitochondrial respiration or the efficiency of ATP production. It is worth noting that miR-146a can also inhibit the development of liver cancer by targeting several genes; therefore, overexpression of miR-146a is a better choice than inhibition of MED1 when patients have both liver cancer and fatty liver. Although there may be other unknown target genes of miR-146a, such treatment may have a negative effect on the human body (70).
In summary, our study demonstrated that overexpression of hepatic miR-146a could improve glucose and lipid metabolism. Additionally, MED1 mediates the effect of miR-146a on mitochondrial function and lipid metabolism. Our findings, not only add a new dimension to our understanding of the relationship between mitochondrial function and NAFLD, but also provide a potential therapeutic target for NAFLD and metabolic syndrome disease.
Supplementary Data
Acknowledgments
Not applicable.
Funding
This study was financially supported by the National Natural Science Foundation of China (grant no. 31700735), the Natural Science Foundation of Anhui province (grant nos. 1808085QH264 and 1908085MB54), the Wanjiang Scholar Award of Anhui Province (China) and the Major Natural Science Research Projects in Anhui Universities (grant no. KJ2019ZD58).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
KL and BZ designed and performed the experiments, analysed the data and wrote the manuscript. DW and YC provided useful advice on the manuscript and analysed the data. LQ and WW and GL designed the experiments and wrote the manuscript. All authors read and approved the manuscript.
Ethics approval and consent to participate
All animal protocols were approved by the Animal Experimental Ethical Inspection Committee of Bengbu Medical College (approval no. DWLL-2017-046).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Trefts E, Gannon M, Wasserman DH. The liver. Curr Biol. 2017;27:R1147–R1151. doi: 10.1016/j.cub.2017.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Targher G, Bertolini L, Padovani R, Rodella S, Tessari R, Zenari L, Day C, Arcaro G. Prevalence of nonalcoholic fatty liver disease and its association with cardiovascular disease among type 2 diabetic patients. Diabetes Care. 2007;30:1212–1218. doi: 10.2337/dc06-2247. [DOI] [PubMed] [Google Scholar]
- 3.Byrne CD, Targher G. NAFLD: A multisystem disease. J Hepatol. 2015;62(1 Suppl):S47–S64. doi: 10.1016/j.jhep.2014.12.012. [DOI] [PubMed] [Google Scholar]
- 4.Cobbina E, Akhlaghi F. Non-alcoholic fatty liver disease (NAFLD)-pathogenesis, classification, and effect on drug metabolizing enzymes and transporters. Drug Metab Rev. 2017;49:197–211. doi: 10.1080/03602532.2017.1293683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ajith TA. Role of mitochondria and mitochondria-targeted agents in non-alcoholic fatty liver disease. Clin Exp Pharmacol Physiol. 2018;45:413–421. doi: 10.1111/1440-1681.12886. [DOI] [PubMed] [Google Scholar]
- 6.Petrosillo G, Portincasa P, Grattagliano I, Casanova G, Matera M, Ruggiero FM, Ferri D, Paradies G. Mitochondrial dysfunction in rat with nonalcoholic fatty liver involvement of complex I, reactive oxygen species and cardiolipin. Biochim Biophys Acta. 2007;1767:1260–1267. doi: 10.1016/j.bbabio.2007.07.011. [DOI] [PubMed] [Google Scholar]
- 7.Wei Y, Rector RS, Thyfault JP, Ibdah JA. Nonalcoholic fatty liver disease and mitochondrial dysfunction. World J Gastroentero. 2008;14:193–199. doi: 10.3748/wjg.14.193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Satapati S, Sunny NE, Kucejova B, Fu X, He TT, Méndez-Lucas A, Shelton JM, Perales JC, Browning JD, Burgess SC. Elevated TCA cycle function in the pathology of diet-induced hepatic insulin resistance and fatty liver. J Lipid Res. 2012;53:1080–1092. doi: 10.1194/jlr.M023382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cortez-Pinto H, Chatham J, Chacko VP, Arnold C, Rashid A, Diehl AM. Alterations in liver ATP homeostasis in human nonalcoholic steatohepatitis-A pilot study. JAMA. 1999;282:1659–1664. doi: 10.1001/jama.282.17.1659. [DOI] [PubMed] [Google Scholar]
- 10.Schmid AI, Szendroedi J, Chmelik M, Krssak M, Moser E, Roden M. Liver ATP synthesis is lower and relates to insulin sensitivity in patients with type 2 diabetes. Diabetes Care. 2011;34:448–453. doi: 10.2337/dc10-1076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Abdelmalek MF, Lazo M, Horska A, Bonekamp S, Lipkin EW, Balasubramanyam A, Bantle JP, Johnson RJ, Diehl AM, Clark JM, et al. Higher dietary fructose is associated with impaired hepatic adenosine triphosphate homeostasis in obese individuals with type 2 diabetes. Hepatology. 2012;56:952–960. doi: 10.1002/hep.25741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Szendroedi J, Chmelik M, Schmid AI, Nowotny P, Brehm A, Krssak M, Moser E, Roden M. Abnormal hepatic energy homeostasis in type 2 diabetes. Hepatology. 2009;50:1079–1086. doi: 10.1002/hep.23093. [DOI] [PubMed] [Google Scholar]
- 13.Fritsch M, Koliaki C, Livingstone R, Phielix E, Bierwagen A, Meisinger M, Jelenik T, Strassburger K, Zimmermann S, Brockmann K, et al. Time course of postprandial hepatic phosphorus metabolites in lean, obese, and type 2 diabetes patients. Am J Clin Nutr. 2015;102:1051–1058. doi: 10.3945/ajcn.115.107599. [DOI] [PubMed] [Google Scholar]
- 14.Ambros V. The functions of animal microRNAs. Nature. 2004;431:350–355. doi: 10.1038/nature02871. [DOI] [PubMed] [Google Scholar]
- 15.Hackl H, Burkard TR, Sturn A, Rubio R, Schleiffer A, Tian S, Quackenbush J, Eisenhaber F, Trajanoski Z. Molecular processes during fat cell development revealed by gene expression profiling and functional annotation. Genome Biol. 2005;6:R108. doi: 10.1186/gb-2005-6-13-r108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yuan Y, Zeng ZY, Liu XH, Gong DJ, Tao J, Cheng HZ, Huang SD. MicroRNA-203 inhibits cell proliferation by repressing ΔNp63 expression in human esophageal squamous cell carcinoma. BMC Cancer. 2011;11:57. doi: 10.1186/1471-2407-11-57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lima RT, Busacca S, Almeida GM, Gaudino G, Fennell DA, Vasconcelos MH. MicroRNA regulation of core apoptosis pathways in cancer. Eur J Cancer. 2011;47:163–174. doi: 10.1016/j.ejca.2010.11.005. [DOI] [PubMed] [Google Scholar]
- 18.Liston A, Linterman M, Lu LF. MicroRNA in the adaptive immune system, in sickness and in health. J Clin Immunol. 2010;30:339–346. doi: 10.1007/s10875-010-9378-5. [DOI] [PubMed] [Google Scholar]
- 19.Esquela-Kerscher A, Slack FJ. Oncomirs-microRNAs with a role in cancer. Nat Rev Cancer. 2006;6:259–269. doi: 10.1038/nrc1840. [DOI] [PubMed] [Google Scholar]
- 20.Liang T, Liu C, Ye Z. Deep sequencing of small RNA repertoires in mice reveals metabolic disorders-associated hepatic miRNAs. PLoS One. 2013;8:e80774. doi: 10.1371/journal.pone.0080774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rottiers V, Naar AM. MicroRNAs in metabolism and metabolic disorders. Nat Rev Mol Cell Biol. 2012;13:239–250. doi: 10.1038/nrm3313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Taganov KD, Boldin MP, Chang KJ, Baltimore D. NF-kappaB-dependent induction of microRNA miR-146, an inhibitor targeted to signaling proteins of innate immune responses. Proc Natl Acad Sci USA. 2006;103:12481–12486. doi: 10.1073/pnas.0605298103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cobb BS, Hertweck A, Smith J, O'Connor E, Graf D, Cook T, Smale ST, Sakaguchi S, Livesey FJ, Fisher AG, Merkenschlager M. A role for Dicer in immune regulation. J Exp Med. 2006;203:2519–2527. doi: 10.1084/jem.20061692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Liu X, Dong Y, Chen S, Zhang G, Zhang M, Gong Y, Li X. Circulating MicroRNA-146a and MicroRNA-21 predict left ventricular remodeling after ST-elevation myocardial infarction. Cardiology. 2015;132:233–241. doi: 10.1159/000437090. [DOI] [PubMed] [Google Scholar]
- 25.Xiong XD, Cho M, Cai XP, Cheng J, Jing X, Cen JM, Liu X, Yang XL, Suh Y. A common variant in pre-miR-146 is associated with coronary artery disease risk and its mature miRNA expression. Mutat Res. 2014;761:15–20. doi: 10.1016/j.mrfmmm.2014.01.001. [DOI] [PubMed] [Google Scholar]
- 26.Jazdzewski K, Murray EL, Franssila K, Jarzab B, Schoenberg DR, de la Chapelle A. Common SNP in pre-miR-146a decreases mature miR expression and predisposes to papillary thyroid carcinoma. Proc Natl Acad Sci USA. 2008;105:7269–7274. doi: 10.1073/pnas.0802682105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jin X, Liu J, Chen YP, Xiang Z, Ding JX, Li YM. Effect of miR-146 targeted HDMCP up regulation in the pathogenesis of nonalcoholic steatohepatitis. PLoS One. 2017;12:e0174218. doi: 10.1371/journal.pone.0174218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bleau AM, Redrado M, Nistal-Villan E, Villalba M, Exposito F, Redin E, de Aberasturi AL, Larzabal L, Freire J, Gomez-Roman J, Calvo A. miR-146a targets c-met and abolishes colorectal cancer liver metastasis. Cancer Lett. 2018;414:257–267. doi: 10.1016/j.canlet.2017.11.008. [DOI] [PubMed] [Google Scholar]
- 29.Sun X, Zhang J, Hou Z, Han Q, Zhang C, Tian Z. miR-146a is directly regulated by STAT3 in human hepatocellular carcinoma cells and involved in anti-tumor immune suppression. Cell Cycle. 2015;14:243–252. doi: 10.4161/15384101.2014.977112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zhang X, Ye ZH, Liang HW, Ren FH, Li P, Dang YW, Chen G. Down-regulation of miR-146a-5p and its potential targets in hepatocellular carcinoma validated by a TCGA- and GEO-based study. FEBS Open Bio. 2017;7:504–521. doi: 10.1002/2211-5463.12198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Canto C, Jiang LQ, Deshmukh AS, Mataki C, Coste A, Lagouge M, Zierath JR, Auwerx J. Interdependence of AMPK and SIRT1 for metabolic adaptation to fasting and exercise in skeletal muscle. Cell Metab. 2010;11:213–219. doi: 10.1016/j.cmet.2010.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 33.Schneider L, Giordano S, Zelickson BR, S Johnson M, A Benavides G, Ouyang X, Fineberg N, Darley-Usmar VM, Zhang J. Differentiation of SH-SY5Y cells to a neuronal phenotype changes cellular bioenergetics and the response to oxidative stress. Free Radic Biol Med. 2011;51:2007–2017. doi: 10.1016/j.freeradbiomed.2011.08.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Liu W, Cao H, Yan J, Huang R, Ying H. 'Micro-managers' of hepatic lipid metabolism and NAFLD. Wiley Interdiscip Rev RNA. 2015;6:581–593. doi: 10.1002/wrna.1295. [DOI] [PubMed] [Google Scholar]
- 35.Bonawitz ND, Clayton DA, Shadel GS. Initiation and beyond: Multiple functions of the human mitochondrial transcription machinery. Mol Cell. 2006;24:813–825. doi: 10.1016/j.molcel.2006.11.024. [DOI] [PubMed] [Google Scholar]
- 36.Piantadosi CA, Carraway MS, Babiker A, Suliman HB. Heme oxygenase-1 regulates cardiac mitochondrial biogenesis via Nrf2-mediated transcriptional control of nuclear respiratory factor-1. Circ Res. 2008;103:1232–1240. doi: 10.1161/01.RES.0000338597.71702.ad. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Scarpulla RC, Vega RB, Kelly DP. Transcriptional integration of mitochondrial biogenesis. Trends Endocrinol Metab. 2012;23:459–466. doi: 10.1016/j.tem.2012.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Suliman HB, Sweeney TE, Withers CM, Piantadosi CA. Co-regulation of nuclear respiratory factor-1 by NFkappaB and CREB links LPS-induced inflammation to mitochondrial biogenesis. J Cell Sci. 2010;123:2565–2575. doi: 10.1242/jcs.064089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Campbell CT, Kolesar JE, Kaufman BA. Mitochondrial transcription factor A regulates mitochondrial transcription initiation, DNA packaging, and genome copy number. Biochim Biophys Acta. 2012;1819:921–929. doi: 10.1016/j.bbagrm.2012.03.002. [DOI] [PubMed] [Google Scholar]
- 40.Falkenberg M, Gaspari M, Rantanen A, Trifunovic A, Larsson NG, Gustafsson CM. Mitochondrial transcription factors B1 and B2 activate transcription of human mtDNA. Nat Genet. 2002;31:289–294. doi: 10.1038/ng909. [DOI] [PubMed] [Google Scholar]
- 41.Larsson NG, Wang J, Wilhelmsson H, Oldfors A, Rustin P, Lewandoski M, Barsh GS, Clayton DA. Mitochondrial transcription factor A is necessary for mtDNA maintenance and embryogenesis in mice. Nat Genet. 1998;18:231–236. doi: 10.1038/ng0398-231. [DOI] [PubMed] [Google Scholar]
- 42.Dhar SS, Ongwijitwat S, Wong-Riley MT. Nuclear respiratory factor 1 regulates all ten nuclear-encoded Subunits of cytochrome c oxidase in neurons. J Biol Chem. 2008;283:3120–3129. doi: 10.1074/jbc.M707587200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kelly DP, Scarpulla RC. Transcriptional regulatory circuits controlling mitochondrial biogenesis and function. Genes Dev. 2004;18:357–368. doi: 10.1101/gad.1177604. [DOI] [PubMed] [Google Scholar]
- 44.Scarpulla RC. Transcriptional paradigms in mammalian mitochondrial biogenesis and function. Physiol Rev. 2008;88:611–638. doi: 10.1152/physrev.00025.2007. [DOI] [PubMed] [Google Scholar]
- 45.Jia Y, Viswakarma N, Reddy JK. Med1 subunit of the mediator complex in nuclear receptor-regulated energy metabolism, liver regeneration, and hepatocarcinogenesis. Gene Expr. 2014;16:63–75. doi: 10.3727/105221614X13919976902219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Leite NC, Salles GF, Araujo AL, Villela-Nogueira CA, Cardoso CR. Prevalence and associated factors of non-alcoholic fatty liver disease in patients with type-2 diabetes mellitus. Liver Int. 2009;29:113–119. doi: 10.1111/j.1478-3231.2008.01718.x. [DOI] [PubMed] [Google Scholar]
- 47.Li S, Chen X, Zhang H, Liang X, Xiang Y, Yu C, Zen K, Li Y, Zhang CY. Differential expression of microRNAs in mouse liver under aberrant energy metabolic status. J Lipid Res. 2009;50:1756–1765. doi: 10.1194/jlr.M800509-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ding J, Li M, Wan X, Jin X, Chen S, Yu C, Li Y. Effect of miR-34a in regulating steatosis by targeting PPARα expression in nonalcoholic fatty liver disease. Sci Rep. 2015;5:13729. doi: 10.1038/srep13729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Xu Y, Zalzala M, Xu J, Li Y, Yin L, Zhang Y. A metabolic stress-inducible miR-34a-HNF4 α pathway regulates lipid and lipoprotein metabolism. Nat Commun. 2015;6:7466. doi: 10.1038/ncomms8466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Derdak Z, Villegas KA, Harb R, Wu AM, Sousa A, Wands JR. Inhibition of p53 attenuates steatosis and liver injury in a mouse model of non-alcoholic fatty liver disease. J Hepatol. 2013;58:785–791. doi: 10.1016/j.jhep.2012.11.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ahn J, Lee H, Jung CH, Ha T. Lycopene inhibits hepatic steatosis via microRNA-21-induced downregulation of fatty acid-binding protein 7 in mice fed a high-fat diet. Mol Nutr Food Res. 2012;56:1665–1674. doi: 10.1002/mnfr.201200182. [DOI] [PubMed] [Google Scholar]
- 52.Sun C, Huang F, Liu X, Xiao X, Yang M, Hu G, Liu H, Liao L. miR-21 regulates triglyceride and cholesterol metabolism in non-alcoholic fatty liver disease by targeting HMGCR. Int J Mol Med. 2015;35:847–853. doi: 10.3892/ijmm.2015.2076. [DOI] [PubMed] [Google Scholar]
- 53.He Y, Huang C, Lin X, Li J. MicroRNA-29 family, a crucial therapeutic target for fibrosis diseases. Biochimie. 2013;95:1355–1359. doi: 10.1016/j.biochi.2013.03.010. [DOI] [PubMed] [Google Scholar]
- 54.Pogribny IP, Starlard-Davenport A, Tryndyak VP, Han T, Ross SA, Rusyn I, Beland FA. Difference in expression of hepatic microRNAs miR-29c, miR-34a, miR-155, and miR-200b is associated with strain-specific susceptibility to dietary nonalcoholic steatohepatitis in mice. Lab Invest. 2010;90:1437–1446. doi: 10.1038/labinvest.2010.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.He S, Guo W, Deng F, Chen K, Jiang Y, Dong M, Peng L, Chen X. Targeted delivery of microRNA 146b mimic to hepatocytes by lactosylated PDMAEMA nanoparticles for the treatment of NAFLD. Artif Cells Nanomed Biotechnol. 2018;46(Suppl 2):S217–S228. doi: 10.1080/21691401.2018.1453830. [DOI] [PubMed] [Google Scholar]
- 56.Paradies G, Paradies V, Ruggiero FM, Petrosillo G. Oxidative stress, cardiolipin and mitochondrial dysfunction in nonalcoholic fatty liver disease. World J Gastroentero. 2014;20:14205–14218. doi: 10.3748/wjg.v20.i39.14205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Azizi R, Soltani-Zangbar MS, Sheikhansari G, Pourmoghadam Z, Mehdizadeh A, Mahdipour M, Sandoghchian S, Danaii S, Koushaein L, Samadi Kafil H, Yousefi M. Metabolic syndrome mediates inflammatory and oxidative stress responses in patients with recurrent pregnancy loss. J Reprod Immunol. 2019;133:18–26. doi: 10.1016/j.jri.2019.05.001. [DOI] [PubMed] [Google Scholar]
- 58.Ge K, Guermah M, Yuan CX, Ito M, Wallberg AE, Spiegelman BM, Roeder RG. Transcription coactivator TRAP220 is required for PPAR gamma 2-stimulated adipogenesis. Nature. 2002;417:563–567. doi: 10.1038/417563a. [DOI] [PubMed] [Google Scholar]
- 59.Chen W, Zhang X, Birsoy K, Roeder RG. A muscle-specific knockout implicates nuclear receptor coactivator MED1 in the regulation of glucose and energy metabolism. Proc Natl Acad Sci USA. 2010;107:10196–10201. doi: 10.1073/pnas.1005626107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Yu J, Xiao Y, Liu J, Ji Y, Liu H, Xu J, Jin X, Liu L, Guan MX, Jiang P. Loss of MED1 triggers mitochondrial biogenesis in C2C12 cells. Mitochondrion. 2014;14:18–25. doi: 10.1016/j.mito.2013.12.004. [DOI] [PubMed] [Google Scholar]
- 61.Bai L, Jia Y, Viswakarma N, Huang J, Vluggens A, Wolins NE, Jafari N, Rao MS, Borensztajn J, Yang G, Reddy JK. Transcription coactivator mediator subunit MED1 is required for the development of fatty liver in the mouse. Hepatology. 2011;53:1164–1174. doi: 10.1002/hep.24155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Kornberg RD. The molecular basis of eukaryotic transcription. Proc Natl Acad Sci USA. 2007;104:12955–12961. doi: 10.1073/pnas.0704138104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Malik S, Roeder RG. Dynamic regulation of pol II transcription by the mammalian Mediator complex. Trends Biochem Sci. 2005;30:256–263. doi: 10.1016/j.tibs.2005.03.009. [DOI] [PubMed] [Google Scholar]
- 64.Lieber CS, Leo MA, Mak KM, Xu Y, Cao Q, Ren C, Ponomarenko A, DeCarli LM. Model of nonalcoholic steatohepatitis. Am J Clin Nutr. 2004;79:502–509. doi: 10.1093/ajcn/79.3.502. [DOI] [PubMed] [Google Scholar]
- 65.Kim JA, Wei Y, Sowers JR. Role of mitochondrial dysfunction in insulin resistance. Circ Res. 2008;102:401–414. doi: 10.1161/CIRCRESAHA.107.165472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Miele L, Grieco A, Armuzzi A, Candelli M, Forgione A, Gasbarrini A, Gasbarrini G. Hepatic mitochondrial beta-oxidation in patients with nonalcoholic steatohepatitis assessed by 13C-octanoate breath test. Am J Gastroenterol. 2003;98:2335–2336. doi: 10.1111/j.1572-0241.2003.07725.x. [DOI] [PubMed] [Google Scholar]
- 67.Iozzo P, Bucci M, Roivainen A, Någren K, Järvisalo MJ, Kiss J, Guiducci L, Fielding B, Naum AG, Borra R, et al. Fatty acid metabolism in the liver, measured by positron emission tomography, is increased in obese individuals. Gastroenterology. 2010;139:846–856. e1–6. doi: 10.1053/j.gastro.2010.05.039. [DOI] [PubMed] [Google Scholar]
- 68.Sunny NE, Parks EJ, Browning JD, Burgess SC. Excessive hepatic mitochondrial TCA cycle and gluconeogenesis in humans with nonalcoholic fatty liver disease. Cell Metab. 2011;14:804–810. doi: 10.1016/j.cmet.2011.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Sunny NE, Bril F, Cusi K. Mitochondrial adaptation in nonalcoholic fatty liver disease: Novel mechanisms and treatment strategies. Trends Endocrinol Metab. 2017;28:250–260. doi: 10.1016/j.tem.2016.11.006. [DOI] [PubMed] [Google Scholar]
- 70.Rong M, He R, Dang Y, Chen G. Expression and clinicopathological significance of miR-146a in hepatocellular carcinoma tissues. Ups J Med Sci. 2014;119:19–24. doi: 10.3109/03009734.2013.856970. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.