Abstract
Women residing at high altitudes deliver infants of lower birth weight than at sea level. Birth weight correlates with placental system A-mediated amino acid transport capacity, and severe environmental hypoxia reduces system A activity in isolated trophoblast and the mouse placenta. However, the effect of high altitude on human placental amino acid transport remains unknown. We hypothesized that microvillous membrane (MVM) system A and system L amino acid transporter activity is lower in placentas of women living at high altitude compared with low-altitude controls. Placentas were collected at term from healthy pregnant women residing at high altitude (HA; >2,500 m; n = 14) or low altitude (LA; <1,700 m; n = 14) following planned, unlabored cesarean section. Birth weight, but not placenta weight, was 13% lower in HA pregnancies (2.88 ± 0.11 kg) compared with LA (3.30 ± 0.07 kg, P < 0.01). MVM erythropoietin receptor abundance, determined by immunoblot, was greater in HA than in LA placentas, consistent with lower placental oxygen levels at HA. However, there was no effect of altitude on MVM system A or L activity, determined by Na+-dependent [14C]methylaminoisobutyric acid uptake and [3H]leucine uptake, respectively. MVM abundance of glucose transporters (GLUTs) 1 and 4 and basal membrane GLUT4 were also similar in LA and HA placentas. Low birth weights in the neonates of women residing at high altitude are not a consequence of reduced placental amino acid transport capacity. These observations are in general agreement with studies of IUGR babies at low altitude, in which MVM system A activity is downregulated only in growth-restricted babies with significant compromise.
NEW & NOTEWORTHY Babies born at high altitude are smaller than at sea level. Birth weight is dependent on growth in utero and, in turn, placental nutrient transport. We determined amino acid transport capacity in placentas collected from women resident at low and high altitude. Altitude did not affect system A amino acid transport across the syncytiotrophoblast microvillous membrane, suggesting that impaired placental amino acid transport does not contribute to reduced birth weight in this high-altitude population.
Keywords: fetal growth, glucose transporter, maternal-fetal exchange, trophoblast
INTRODUCTION
Women residing at high altitude deliver infants of lower birth weight than their sea level counterparts (20). This reduction in birth weight is attributable to reduced intrauterine growth rate and not associated with shortened gestation length, which is similar in low- and high-altitude populations. Compared with sea level, the incidence of intrauterine growth restriction (IUGR) is almost fourfold higher at high altitude (29). The defining physiological challenge of high altitude is reduced inspired partial pressure of oxygen (Po2). Whereas the partial pressure of oxygen in the atmosphere declines linearly with increasing altitude, arterial oxygen saturation () does not decline appreciably until ~2500 m due to the sigmoidal shape of the hemoglobin-oxygen dissociation curve. Therefore, high altitude is conventionally defined as >2,500 m (inspired Po2 <120 mmHg) (19). Birth weights in high-altitude populations within the United States are typically 7–13% lower compared with low altitude controls and are associated with reduced maternal and reduced uterine artery blood flow (15–17, 27, 34). Umbilical venous oxygen partial pressure is also reduced at high altitude but may be compensated by increased fetal hematocrit and hemoglobin oxygen affinity, such that umbilical venous oxygen content and fetal oxygen consumption remain uncompromised (22). Therefore, the mechanism by which human fetuses grow more slowly at high altitude remains poorly understood.
Low birth weight at high altitude may be explained by impaired placental nutrient transport capacity. In general agreement with this possibility, placental size and glucose transporter abundance are reduced at high altitude (28, 32). High-altitude placentas have also been reported to have increased phosphorylation of eukaryotic initiation factor-2α (EIF2α), a readout for endoplasmic reticulum stress, and reduced activity of mechanistic target of rapamycin (mTOR) signaling (31). Trophoblast mTOR is a positive regulator of system A and system L amino acid transporters (24). System A is a sodium-dependent transporter of small neutral amino acids, and system L amino acid transporters mediate essential amino acid exchange in the maternal-facing microvillous membrane (MVM) (23, 24). These transporters actively accumulate amino acids in the syncytiotrophoblast, establishing fetal circulating amino acid concentrations that are two- to threefold higher than circulating maternal concentrations and may be critical for normal fetal growth (3). Both placental mTOR and amino acid transport capacity correlate with birth weight at sea level and are downregulated in pregnancies complicated by severe IUGR (2, 5, 10, 14, 18, 25). Moreover, severe environmental hypoxia reduces system A amino acid transport in primary human trophoblast cells (ambient Po2 8–23 mmHg) (21) and in the mouse placenta (inspired Po2 76–99 mmHg) (7). In this first investigation of human placental amino acid transporter activity at high altitude, we hypothesized that MVM system A and system L amino acid transporter activity are lower in placentas of women living at high altitudes compared with low-altitude controls. We assessed system A and system L activity by measuring the rate of radiolabeled amino acid tracer uptake in isolated MVM vesicles. We also determined MVM and basal membrane glucose transporter (GLUT1 and GLUT4) abundance and readouts of placental endoplasmic reticulum stress.
MATERIALS AND METHODS
Subjects and placenta collection.
Healthy pregnant women delivering at term were recruited as part of a larger study with informed consent and ethical approval from the Institutional Review Boards of the University of Colorado and the Catholic Health Initiatives Institute for Research and Innovation. Placentas were collected following planned, unlabored cesarean sections from women residing at high altitudes (elevation 2,849 ± 230 m; n = 14) and low-altitude controls (elevation <1,700 m; n = 14). In addition to residential altitude, study subject inclusion criteria were maternal age from 18 to 45 yr, prepregnant body mass index <30 kg/m2, and singleton pregnancy. Exclusion criteria included maternal smoking, any form of maternal diabetes, pre-eclampsia, infection, fetal anomaly, or recreational drug or alcohol usage during pregnancy. The low-altitude controls reported here were matched to high-altitude subjects based on ethnicity, infant sex, and maternal prepregnancy body mass index. Clinical characteristics of the study cohort are given in Table 1. Birth weight percentile was determined based on the Fenton growth chart (4). Following delivery at St. Anthony’s Summit Medical Center in Frisco, CO (elevation 2,793 m), placental specimens were transported on ice over an ∼2-h period to the University of Colorado Anschutz Medical Campus (elevation 1,640 m). Control tissues collected at the University of Colorado Hospital were similarly stored on ice before processing.
Table 1.
Clinical characteristics of study subjects
| Low Altitude | High Altitude | |
|---|---|---|
| Maternal age, yr | 31 ± 1 (25–38) | 31 ± 1 (23–43) |
| Prepregnancy body mass index | 23.6 ± 0.8 (19–30) | 23.6 ± 1.4 (18–34) |
| Ethnicity (no. Hispanic) | 5 | 5 |
| Gestational age at delivery, wk | 39.0 ± 0.01 (39.0–39.1) | 39.0 ± 0.2 (37.1–39.6) |
| Birth weight, kg | 3.30 ± 0.07 (2.87–3.77) | 2.88 ± 0.11 (2.23–3.66)* |
| Birth weight percentile, % | 49 ± 19 (21–85) | 24 ± 21 (2–74)** |
| Placenta weight, g | 673 ± 36 (434–873) | 588 ± 56 (464–998) |
| Infant sex (no. female) | 5 | 5 |
Continuous variables presented as mean ± SD (minimum-maximum; n = 14).
P < 0.05 vs. low altitude by Student’s t test;
P < 0.05 vs. low altitude by Mann-Whitney U-test.
Isolation of syncytiotrophoblast microvillous membrane.
Placental villous tissue (∼100 g) was dissected and homogenized in ice-cold buffer (HEPES-Tris, sucrose) with protease and phosphatase inhibitors. Syncytiotrophoblast membrane vesicles were isolated from the homogenate using differential ultracentrifugation and Mg2+-precipitation, as described previously (8, 11). Briefly, crude homogenate was centrifuged at low speed to remove large tissue debris, and then the supernatant was subjected to ultracentrifugation (125,000 g, 30 min, 4°C) to separate the membrane fraction. The resultant pellet was resuspended, and MgCl2 was added and recentrifuged to precipitate the syncytiotrophoblast basal plasma membrane. The pellet was homogenized and layered onto a sucrose gradient and centrifuged for 60 min at 144,000 g at 4°C. The supernatant from the slow spin following MgCl2 precipitation (containing MVM) and the basal plasma membrane (BM)-containing layer from the sucrose gradient spin were then subjected to two further ultracentrifugation steps (125,000 g, 30 min, 4°C) to isolate the final MVM and BM vesicles. The final membrane pellets were resuspended and vesiculated using a Dounce homogenizer. Microvillous plasma membrane enrichment of the final isolate was confirmed by its relatively greater alkaline phosphatase activity compared with crude homogenate. Average relative enrichments were similar for placentas collected at low (13.3 ± 1.3) and high altitude (12.1 ± 0.7, P > 0.05, Student’s t test). Basal plasma membrane relative enrichment was confirmed by Western blot for voltage-dependent, anion-selective channel 1 (VDAC1) and was also similar for low- (23.9 ± 3.6) and high-altitude placentas (21.5 ± 2.8, P < 0.05).
System A and system L amino acid transporter activity.
Microvillous membrane system a and system L amino acid transporter activities were determined, using a rapid filtration approach, from the rates of uptake of radiolabeled [14C]methylaminoisobutyric acid ([14C]MeAIB) and [3H]leucine, respectively. Mannitol-loaded microvillous membrane vesicles (∼200 μg protein) were incubated at 37°C with 150 μM [14C]MeAIB and 375 nM [3H]leucine in a sodium-containing buffer (150 mM NaCl, 10 mM HEPES, pH 7.4). At time points ≤30s after the initiation of incubation, tracer uptake was stopped by vacuum filtering the vesicles and washing with ice-cold PBS. Total radiotracer accumulation within the vesicles was subsequently quantified by liquid scintillation counting. Nonmediated uptake and nonspecific binding of [14C]MeAIB and [3H]leucine with each vesicle sample was also determined from contemporaneous incubations in sodium-free buffer plus excess unlabeled leucine (30 mM). Net sodium-dependent [14C]MeAIB ( = system A activity) and transporter-mediated [3H]leucine uptake were calculated as the difference between total and nonmediated radiotracer accumulation, corrected for vesicle protein content. Transporter-mediated leucine uptake measured in this study represents system L activity because mediated leucine uptake in MVM is almost exclusively due to system L (12).
Transporter protein abundance.
Total expression and phosphorylation of selected proteins was determined in MVM, BM, and crude villous homogenates. MVM erythropoietin receptor abundance was determined by capillary-based immunoassay (ProteinSimple; Bio-Techne, San Jose, CA) using a rabbit-derived primary antibody at 1:50 dilution (EPO-R, NBP1-19388; Novus Biologicals, Centennial, CO). Phosphorylated EIF2α was also determined by capillary immunoassay (1:100, no. 3398; Cell Signaling Technology, Danvers, MA). Vinculin abundance was used as a loading control in capillary immunoassays. All other targets were assessed by traditional Western blot, using equal amounts of protein lysate resolved by polyacrylamide gel electrophoresis under reducing, denaturing conditions, and then transferred to polyvinylidene fluoride membrane. Following antigen blocking, microvillous membrane samples were interrogated with specific primary antibodies targeting glucose transporter 1 (GLUT1; 07-1401; Millipore-Sigma) and glucose transporter 4 (GLUT4; no. 2213; Cell Signaling Technology). Markers of endoplasmic reticulum stress, activating transcription factor 4 (ATF4; no. 11815; Cell Signaling Technology), phosphorylated (Ser51) and total eukaryotic initiation factor-2α (EIF2α; nos. 3398 and 5325; Cell Signaling Technology), and phosphorylated (Thr899) and total general control nonderepressible 2 (GCN2; ab75836; Abcam, Cambridge, UK; and no. 3302; Cell Signaling Technology) were determined in placental homogenates. Membranes were subsequently washed and incubated with a species-specific, horseradish peroxidase-conjugated secondary antibody and specific protein bands visualized using enhanced chemiluminescence and an automated imaging system. Membrane amido black staining was used to control for any differences in protein loading or transfer.
Statistics.
All results are presented as the means ± SD. Low- and high-altitude groups were compared using the unpaired Student’s t test. When data did not follow a normal distribution or variances differed between groups, values were logarithmically transformed before statistical analysis. If data transformation did not restore normality or heterogeneity of variances, low- and high-altitude groups were compared by Mann-Whitney U-test. In all cases, statistical significance was taken at the level P < 0.05.
RESULTS
Birth weight was 13% lower in neonates born to women residing at high altitude compared with low-altitude controls (Table 1) and negatively correlated with maternal altitude of residence (Fig. 1). In contrast, maternal altitude of residence had no effect on placental weight (Table 1). By design, there were no differences in ethnicity, infant sex, prepregnancy body mass index, maternal age, or gestational age at delivery between the two groups (Table 1). Two of the 14 high-altitude neonates, but none of the low-altitude neonates, had birth weights below the fifth centile, and therefore, they were defined as IUGR (subjects indicated by crossed circles in Figs. 1–5).
Fig. 1.
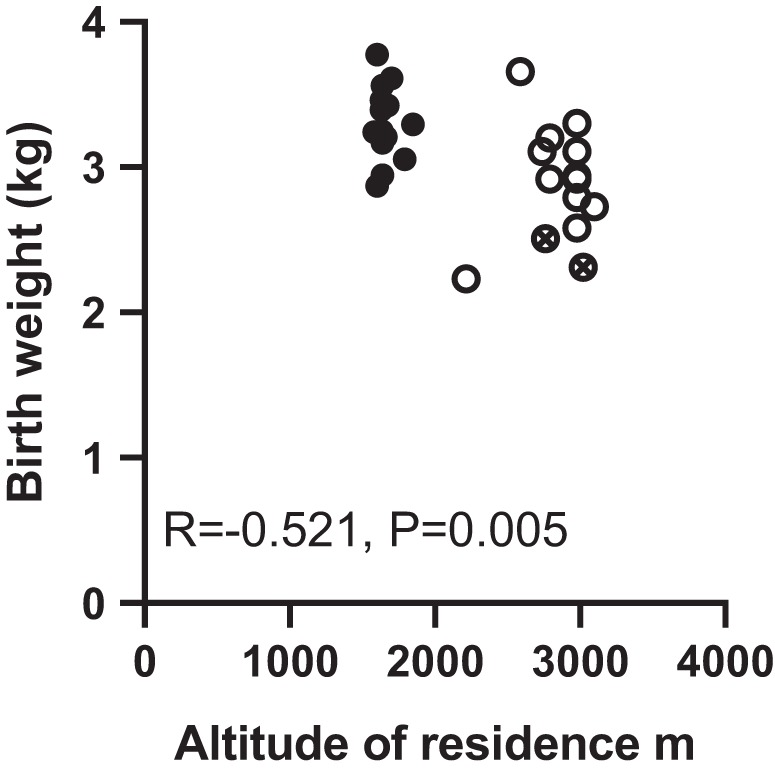
Relationship between maternal altitude of residence and birth weight. Symbols indicate individual low-altitude (●) and high-altitude (○) subjects. Crossed circles indicate placentas collected from pregnancies complicated by intrauterine growth restriction; r and P values for Pearson correlation are given.
Fig. 5.
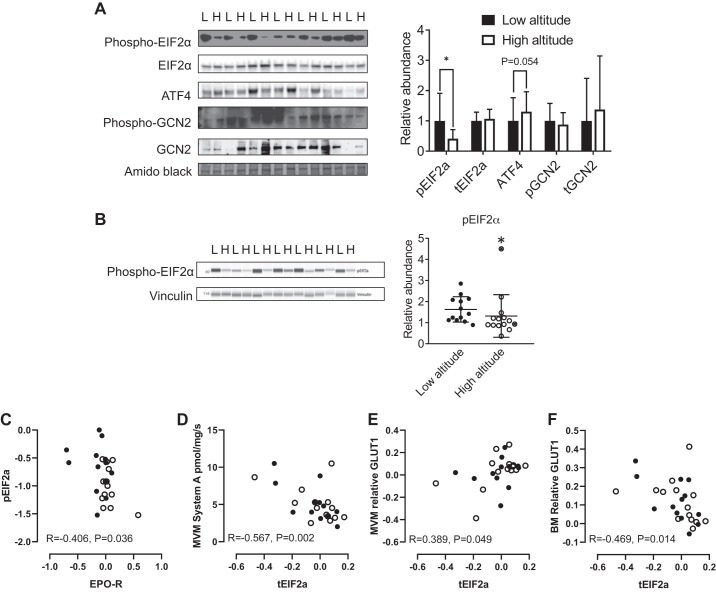
Effect of high altitude on eukaryotic initiation factor-2α (EIF2α) signaling. A: representative blots and relative abundance of total and phosphorylated EIF2α, activating transcription factor 4 (ATF4), and total and phosphorylated general control nonderepressible 2 (GCN2) in homogenates of low-altitude (L; n = 13) and high-altitude (H; n = 14) placentas. Means ± SD. *P < 0.05 vs. low altitude by Mann-Whitney U-test. B: representative capillary electrophoresis image and relative abundance of phosphorylated EIF2α normalized to vinculin in homogenates of high- (n = 14) compared with low-altitude (n = 13) placentas. Symbols indicate individual subjects; bars are means ± SD. Crossed circles indicate placentas collected from pregnancies complicated by intrauterine growth restriction. C–F: correlations of EPO-R, EIF2α, and nutrient transporter activity/abundance in the syncytiotrophoblast membranes; r and P values for Pearson correlations are given (n = 27). ●, Low altitude; ○, high altitude.
MVM EPO-R abundance was greater in high- compared with low-altitude placentas (Fig. 2), consistent with lower placental oxygen levels at high altitude. One high-altitude subject, which was IUGR, was identified as an outlying value in EPO-R abundance, suggesting severe placental hypoxia in this subject (Grubbs’ maximum normalized residual test, log-relative EPO-R abundance 0.58). When this subject was excluded from the analysis, EPO-R abundance remained greater in high-altitude than in low-altitude placentas (P < 0.05, Mann-Whitney U-test). MVM EPO-R abundance negatively correlated with birth weight (r = −0.522, P = 0.004, n = 28) and placenta weight (r = −0.438, P = 0.037, n = 28).
Fig. 2.
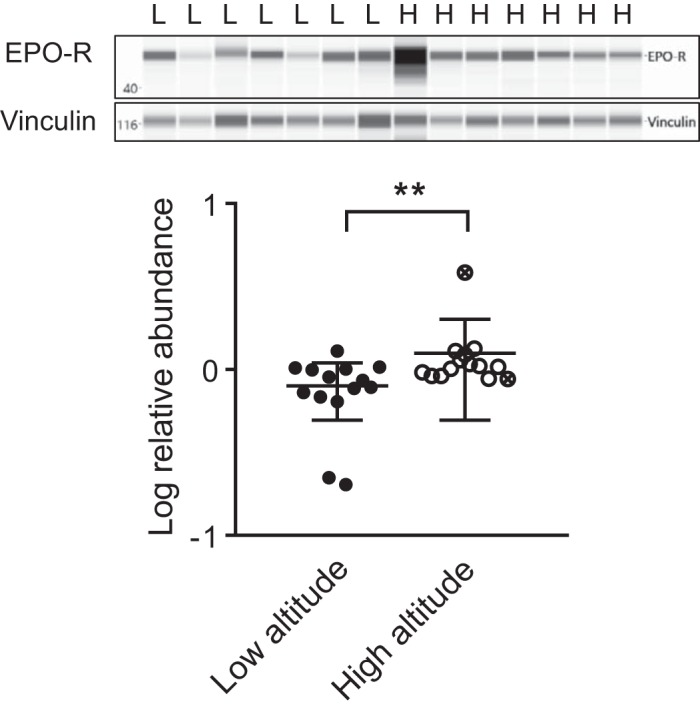
Effect of high altitude on microvillous membrane erythropoietin receptor. Representative image of capillary electrophoresis bands and protein band abundance, normalized to vinculin, in placentas collected at low altitude (L; n = 14) and high altitude (H; n = 14). **P < 0.01 vs. low altitude by Mann-Whitney U-test. Symbols indicate individual subjects; bars are means ± SD. Crossed circles indicate placentas collected from pregnancies complicated by intrauterine growth restriction.
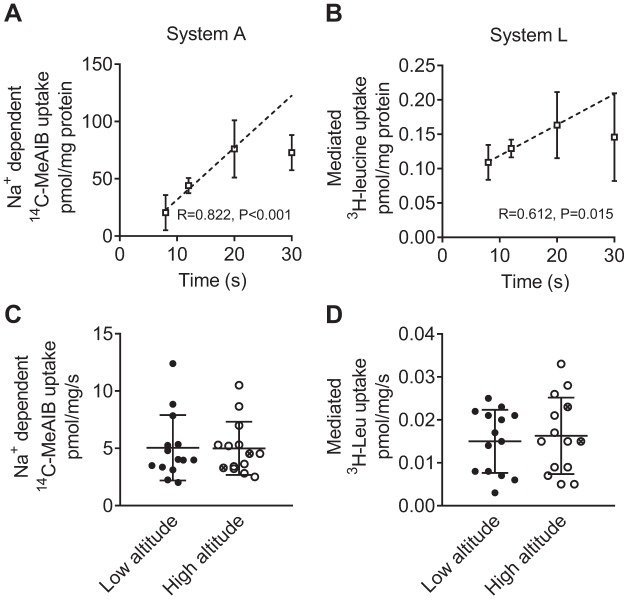
Prior to the determination of MVM amino acid transporter activity, time course experiments using five individual microvillous membrane preparations determined that the initial net uptakes of both [14C]MeAIB and [3H]leucine were linear with respect to incubation time ≤20 s (Fig. 3, A and B). Rates of [14C]MeAIB and [3H]leucine uptake for all study subjects, determined at 10 s of incubation, showed that both system A and system L activity in the MVM were similar in the low- and high-altitude placentas (Fig. 3, C and D).
Fig. 3.
Effect of high altitude on microvillous membrane amino acid transport capacity. A and C: system A activity determined by Na+-dependent uptake of [14C]methylaminoisobutyric acid ([14C]MeAIB). B and D: system L activity determined by mediated [3H]leucine uptake. A and B: net uptake time course over 30 s of incubation with tracer (n = 5 placentas). Dashed line shows linear regression of net amino acid uptake over 20 s. C and D: rates of system A and system L net uptake were determined at 10 s of tracer incubation and did not differ in low- (n = 14) vs. high-altitude placentas (n = 14). Symbols indicate individual subjects; bars are means ± SD. Crossed circles indicate placentas collected from pregnancies complicated by intrauterine growth restriction.
Abundances of the glucose transporters GLUT1 in the MVM and GLUT1 and GLUT4 in the basal plasma membrane (BM) were also similar in low- and high-altitude placentas (Fig. 4). The endoplasmic reticulum stress readout phosphorylated EIF2α was less abundant in homogenates of high- compared with low-altitude placentas (Fig. 5A) and inversely correlated with MVM EPO-R abundance (r = −0.406, P = 0.036, n = 27; Fig. 5C). Phosphorylated EIF2α levels were also lower in high- compared with low-altitude placentas when determined by capillary-based immunoassay, confirming the Western blot findings (Fig. 5B). By contrast, placental abundance of the downstream target of EIF2α, ATF4, tended to be greater in the high-altitude compared with low-altitude residents (Fig. 5A). There was no effect of altitude on total placental EIF2α or on the abundance or phosphorylation of the upstream kinase GCN2 (Fig. 5A). Total but not phosphorylated EIF2α abundance positively correlated with MVM GLUT1 abundance and inversely correlated with MVM system A activity and BM GLUT1 abundance (Fig. 5, D–F). Placental ATF4 abundance was also inversely correlated with BM GLUT1 abundance (r = −0.420, P = 0.029, n = 27).
Fig. 4.
Effect of high altitude on microvillous and basal plasma membrane glucose transporter abundance. A and B: representative blots and relative abundance of glucose transporter 1 (GLUT1) in microvillous (MVM) and basal plasma membranes (BM). C: GLUT4 in basal plasma membranes was similar in low-altitude (n = 13) and high-altitude (n = 14) placentas. Symbols indicate individual subjects; bars are means ± SD. Crossed circles indicate placentas collected from pregnancies complicated by intrauterine growth restriction. Means ± SD. Protein abundance is expressed relative to total protein stained with amido black.
DISCUSSION
This is the first study to determine the effect of high altitude on human placental amino acid transport capacity. We found a 58% increase in EPO-R expression in the syncytiotrophoblast microvillous membrane in high- compared with low-altitude pregnancies, consistent with lower placental oxygen levels at high altitude, but no differences in system A and system L amino acid transport or microvillous membrane glucose transporter abundance between residential altitudes. Therefore, our study suggested that reduced fetal growth at moderately high altitude was not a consequence of altered placental amino acid transport capacity.
The reduction in birth weight in the high-altitude group was consistent with that reported previously at this elevation in geographically similar study populations (15–17, 27, 32). MVM EPO-R expression also increased in line with previous high-altitude studies (32). EPO-R protein is increased in abundance in hypoxic ischemic human tissues (26) and directly upregulated by hypoxia in vitro (6), and therefore, it is used here as a marker of tissue hypoxia. Thus, the inverse correlation of MVM EPO-R expression with birth weight suggests that reduced fetal growth at high altitude is associated with placental hypoxia. EPO-R abundance was highest in the placenta of one of the two IUGR infants included in the high-altitude group but nevertheless remained significantly elevated in the high-altitude group even when this value was omitted.
Despite evidence of lower placental oxygen levels, there was no alteration in MVM amino acid transport capacity in high-altitude placentas. This finding contrasts with the reduction in system A activity demonstrated in mice and primary human trophoblast cells exposed to severe hypoxia (7, 21). Inspired oxygen partial pressure at the mean altitude of residence of high-altitude subjects in our cohort (∼2,800 m) is ∼110 mmHg, whereas reductions in in vivo amino acid transport were observed in the previous rodent studies at levels of isobaric hypoxia equivalent to inspired oxygen partial pressures of ∼70 mmHg and not at ∼100 mmHg (7). Therefore, the degree of environmental hypoxia experienced by women in our high-altitude cohort was most likely of insufficient severity to reduce placental amino acid transport. Moreover, prolonged environmental hypoxia throughout pregnancy, as in our high-altitude subjects, may allow for compensatory responses in placental transport, in contrast to the relatively acute hypoxia used previously in late pregnancy in mice (7), and in primary trophoblast cells isolated from term human placentas (21). It cannot be excluded that the absence of a difference between low- and high-altitude placentas is explained by the ∼2-h delay between delivery and placental homogenization, during which samples were transported to the laboratory on ice. However, our preliminary studies indicate that storing placental tissue on ice for up to 6 h does not alter nutrient transporter activity in syncytiotrophoblast plasma membrane vesicles. Although we were unable to control for the fact that placentas collected at high altitude were exposed to increased ambient oxygen levels during transport to the low-altitude laboratory, this was most likely outweighed by the effect of post-delivery ischemia in all samples, irrespective of collection location.
We also found no difference in syncytiotrophoblast plasma membrane glucose transporter abundance in the present study, in contrast with the previously reported reduction in BM GLUT1 abundance in high-altitude placentas in a similar study population (32). The discrepancy between these studies may be explained by the somewhat broader variability and lower average altitude of residence of the high-altitude participants in our study (2,849 ± 230 m), compared with the previous report (all resident at 3,100 m) (32). However, the present findings are in line with other studies indicating no hypoxia-induced alteration in MVM or BM GLUT1 abundance or tracer glucose uptake in villous explants at Po2 of ∼20 mmHg (1) or in placentas of severely IUGR newborns (13, 14). This study is the first report of syncytiotrophoblast membrane GLUT4 abundance at high altitude, which is localized exclusively to the BM and may regulate fetal glucose delivery in response to insulin (9).
The lack of decrease of nutrient transporter abundance or activity in high-altitude placentas in this study may be explained, in turn, by the absence of ER stress and reduced EIF2α phosphorylation. Hypoxia- and ER stress-induced phosphorylation of EIF2a generally prevents global protein translation, and therefore, it may be an important regulator of nutrient transporter protein synthesis. Certainly, EIF2α phosphorylation is upregulated in association with reduced MVM system A transporter abundance and activity in IUGR placentas (2). A previous study showed that placental villous tissue collected from women residing at 3,100 m had increased levels of phosphorylated EIF2α compared with sea level placentas (31). Our findings contrast with this observation, which may reflect the differing sample sizes, geographical origin, and ethnicity of the control placentas in the two studies. In the present study, high-altitude hypoxia appeared to suppress placental EIF2α phosphorylation, which was inversely correlated with EPO-R levels. Although this result may reflect the technical limitation of our study, whereby processing of high-altitude placental samples was delayed for ∼2 h after delivery, post-delivery placental ischemia would be expected to increase rather than decrease EIF2α phosphorylation (30). Therefore, endoplasmic reticulum stress may actually be reduced as a consequence of reduced placental accumulation of reactive oxygen species at moderately high altitude (33). Nonetheless, high-altitude placentas in this study tended to have higher levels of the transcription factor ATF4, which is translationally induced by phosphorylated EIF2α and indicative of mild endoplasmic reticulum stress downstream. Irrespective of altitude, interindividual variability in total placental EIF2α was correlated with syncytiotrophoblast amino acid and glucose transport capacity. Therefore, our data support a role for placental ER stress in the regulation of nutrient transport, although they indicate that placental ER stress is not important for determining nutrient transport at moderately high altitudes.
Taken together, our results indicate that reduced intrauterine growth at moderately high altitudes is not a consequence of low placental amino acid or glucose transport capacity per se. Whether the net uptake of nutrients by the human fetus is reduced by more subtle alterations in placental metabolism at altitude remains unknown. Most likely, a combination of physiological adaptations in uteroplacental blood flow, nutrient and oxygen delivery, placental morphology, and fetoplacental metabolism contributes to low birth weight in this environment. Our findings nevertheless confirm that high altitude is linked to low birth weight via placental hypoxia, with important implications for the monitoring of pregnancy outcome in high-altitude populations.
GRANTS
This study was supported by the National Institute of Child Health and Human Development (R01-HD-088590).
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the authors.
AUTHOR CONTRIBUTIONS
O.R.V., T.L.P., L.G.M., and T.J. conceived and designed research; O.R.V., F.T., R.A.L., and C.G.J. performed experiments; O.R.V. and F.T. analyzed data; O.R.V. interpreted results of experiments; O.R.V. prepared figures; O.R.V. drafted manuscript; O.R.V., R.A.L., C.G.J., T.L.P., L.G.M., and T.J. edited and revised manuscript; O.R.V., F.T., R.A.L., C.G.J., T.L.P., L.G.M., and T.J. approved final version of manuscript.
ACKNOWLEDGMENTS
We thank Anita Kramer, Kathryn Erickson, and the staff of the Perinatal Clinical and Translational Research Centre at the University of Colorado Anschutz Medical Campus for technical assistance.
REFERENCES
- 1.Baumann MU, Zamudio S, Illsley NP. Hypoxic upregulation of glucose transporters in BeWo choriocarcinoma cells is mediated by hypoxia-inducible factor-1. Am J Physiol Cell Physiol 293: C477–C485, 2007. doi: 10.1152/ajpcell.00075.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chen Y-Y, Rosario FJ, Shehab MA, Powell TL, Gupta MB, Jansson T. Increased ubiquitination and reduced plasma membrane trafficking of placental amino acid transporter SNAT-2 in human IUGR. Clin Sci (Lond) 129: 1131–1141, 2015. doi: 10.1042/CS20150511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cramer S, Beveridge M, Kilberg M, Novak D. Physiological importance of system A-mediated amino acid transport to rat fetal development. Am J Physiol Cell Physiol 282: C153–C160, 2002. doi: 10.1152/ajpcell.2002.282.1.C153. [DOI] [PubMed] [Google Scholar]
- 4.Fenton TR, Kim JH. A systematic review and meta-analysis to revise the Fenton growth chart for preterm infants. BMC Pediatr 13: 59, 2013. doi: 10.1186/1471-2431-13-59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Glazier JD, Cetin I, Perugino G, Ronzoni S, Grey AM, Mahendran D, Marconi AM, Pardi G, Sibley CP. Association between the activity of the system A amino acid transporter in the microvillous plasma membrane of the human placenta and severity of fetal compromise in intrauterine growth restriction. Pediatr Res 42: 514–519, 1997. doi: 10.1203/00006450-199710000-00016. [DOI] [PubMed] [Google Scholar]
- 6.Heir P, Srikumar T, Bikopoulos G, Bunda S, Poon BP, Lee JE, Raught B, Ohh M. Oxygen-dependent Regulation of Erythropoietin Receptor Turnover and Signaling. J Biol Chem 291: 7357–7372, 2016. doi: 10.1074/jbc.M115.694562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Higgins JS, Vaughan OR, Fernandez de Liger E, Fowden AL, Sferruzzi-Perri AN. Placental phenotype and resource allocation to fetal growth are modified by the timing and degree of hypoxia during mouse pregnancy. J Physiol 594: 1341–1356, 2016. doi: 10.1113/JP271057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Illsley NP, Wang Z-Q, Gray A, Sellers MC, Jacobs MM. Simultaneous preparation of paired, syncytial, microvillous and basal membranes from human placenta. Biochim Biophys Acta 1029: 218–226, 1990. doi: 10.1016/0005-2736(90)90157-J. [DOI] [PubMed] [Google Scholar]
- 9.James-Allan LB, Arbet J, Teal SB, Powell TL, Jansson T. Insulin stimulates GLUT4 trafficking to the syncytiotrophoblast basal plasma membrane in the human placenta. J Clin Endocrinol Metab 104: 4225–4238, 2019. doi: 10.1210/jc.2018-02778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jansson N, Rosario FJ, Gaccioli F, Lager S, Jones HN, Roos S, Jansson T, Powell TL. Activation of placental mTOR signaling and amino acid transporters in obese women giving birth to large babies. J Clin Endocrinol Metab 98: 105–113, 2013. doi: 10.1210/jc.2012-2667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jansson T, Illsley NP. Osmotic water permeabilities of human placental microvillous and basal membranes. J Membr Biol 132: 147–155, 1993. doi: 10.1007/BF00239004. [DOI] [PubMed] [Google Scholar]
- 12.Jansson T, Scholtbach V, Powell TL. Placental transport of leucine and lysine is reduced in intrauterine growth restriction. Pediatr Res 44: 532–537, 1998. doi: 10.1203/00006450-199810000-00011. [DOI] [PubMed] [Google Scholar]
- 13.Jansson T, Wennergren M, Illsley NP. Glucose transporter protein expression in human placenta throughout gestation and in intrauterine growth retardation. J Clin Endocrinol Metab 77: 1554–1562, 1993. doi: 10.1210/jcem.77.6.8263141. [DOI] [PubMed] [Google Scholar]
- 14.Jansson T, Ylvén K, Wennergren M, Powell TL. Glucose transport and system A activity in syncytiotrophoblast microvillous and basal plasma membranes in intrauterine growth restriction. Placenta 23: 392–399, 2002. doi: 10.1053/plac.2002.0826. [DOI] [PubMed] [Google Scholar]
- 15.Julian CG, Galan HL, Wilson MJ, Desilva W, Cioffi-Ragan D, Schwartz J, Moore LG. Lower uterine artery blood flow and higher endothelin relative to nitric oxide metabolite levels are associated with reductions in birth weight at high altitude. Am J Physiol Regul Integr Comp Physiol 295: R906–R915, 2008. doi: 10.1152/ajpregu.00164.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lichty JA, Ting RY, Bruns PD, Dyar E. Studies of babies born at high altitudes. I. Relation of altitude to birth weight. AMA J Dis Child 93: 666–669, 1957. doi: 10.1001/archpedi.1957.02060040668009. [DOI] [PubMed] [Google Scholar]
- 17.Lorca RA, Lane SL, Bales ES, Nsier H, Yi H, Donnelly MA, Euser AG, Julian CG, Moore LG. High Altitude Reduces NO-Dependent Myometrial Artery Vasodilator Response During Pregnancy. Hypertension 73: 1319–1326, 2019. doi: 10.1161/HYPERTENSIONAHA.119.12641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Maiiendran D, Donnai P, Glazier JD, D’Souza SW, Boyd RD, Sibley CP. Amino acid (system A) transporter activity in microvillous membrane vesicles from the placentas of appropriate and small for gestational age babies. Pediatr Res 34: 661–665, 1993. doi: 10.1203/00006450-199311000-00019. [DOI] [PubMed] [Google Scholar]
- 19.Moore LG. Altitude-aggravated illness: examples from pregnancy and prenatal life. Ann Emerg Med 16: 965–973, 1987. doi: 10.1016/S0196-0644(87)80742-4. [DOI] [PubMed] [Google Scholar]
- 20.Moore LG, Niermeyer S, Zamudio S. Human adaptation to high altitude: regional and life-cycle perspectives. Am J Phys Anthropol 107, Suppl 27: 25–64, 1998. doi:. [DOI] [PubMed] [Google Scholar]
- 21.Nelson DM, Smith SD, Furesz TC, Sadovsky Y, Ganapathy V, Parvin CA, Smith CH. Hypoxia reduces expression and function of system A amino acid transporters in cultured term human trophoblasts. Am J Physiol Cell Physiol 284: C310–C315, 2003. doi: 10.1152/ajpcell.00253.2002. [DOI] [PubMed] [Google Scholar]
- 22.Postigo L, Heredia G, Illsley NP, Torricos T, Dolan C, Echalar L, Tellez W, Maldonado I, Brimacombe M, Balanza E, Vargas E, Zamudio S. Where the O2 goes to: preservation of human fetal oxygen delivery and consumption at high altitude. J Physiol 587: 693–708, 2009. doi: 10.1113/jphysiol.2008.163634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rosario FJ, Dimasuay KG, Kanai Y, Powell TL, Jansson T. Regulation of amino acid transporter trafficking by mTORC1 in primary human trophoblast cells is mediated by the ubiquitin ligase Nedd4-2. Clin Sci (Lond) 130: 499–512, 2016. doi: 10.1042/CS20150554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rosario FJ, Kanai Y, Powell TL, Jansson T. Mammalian target of rapamycin signalling modulates amino acid uptake by regulating transporter cell surface abundance in primary human trophoblast cells. J Physiol 591: 609–625, 2013. doi: 10.1113/jphysiol.2012.238014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Shibata E, Hubel CA, Powers RW, von Versen-Hoeynck F, Gammill H, Rajakumar A, Roberts JM. Placental system A amino acid transport is reduced in pregnancies with small for gestational age (SGA) infants but not in preeclampsia with SGA infants. Placenta 29: 879–882, 2008. doi: 10.1016/j.placenta.2008.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sirén AL, Knerlich F, Poser W, Gleiter CH, Brück W, Ehrenreich H. Erythropoietin and erythropoietin receptor in human ischemic/hypoxic brain. Acta Neuropathol 101: 271–276, 2001. doi: 10.1007/s004010000297. [DOI] [PubMed] [Google Scholar]
- 27.Unger C, Weiser JK, McCullough RE, Keefer S, Moore LG. Altitude, low birth weight, and infant mortality in Colorado. JAMA 259: 3427–3432, 1988. doi: 10.1001/jama.1988.03720230037027. [DOI] [PubMed] [Google Scholar]
- 28.van Patot MC, Valdez M, Becky V, Cindrova-Davies T, Johns J, Zwerdling L, Jauniaux E, Burton GJ. Impact of pregnancy at high altitude on placental morphology in non-native women with and without preeclampsia. Placenta 30: 523–528, 2009. doi: 10.1016/j.placenta.2009.04.002. [DOI] [PubMed] [Google Scholar]
- 29.Yip R. Altitude and birth weight. J Pediatr 111: 869–876, 1987. doi: 10.1016/S0022-3476(87)80209-3. [DOI] [PubMed] [Google Scholar]
- 30.Yung HW, Colleoni F, Atkinson D, Cook E, Murray AJ, Burton GJ, Charnock-Jones DS. Influence of speed of sample processing on placental energetics and signalling pathways: implications for tissue collection. Placenta 35: 103–108, 2014. doi: 10.1016/j.placenta.2013.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yung HW, Cox M, Tissot van Patot M, Burton GJ. Evidence of endoplasmic reticulum stress and protein synthesis inhibition in the placenta of non-native women at high altitude. FASEB J 26: 1970–1981, 2012. doi: 10.1096/fj.11-190082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zamudio S, Baumann MU, Illsley NP. Effects of chronic hypoxia in vivo on the expression of human placental glucose transporters. Placenta 27: 49–55, 2006. doi: 10.1016/j.placenta.2004.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zamudio S, Kovalenko O, Vanderlelie J, Illsley NP, Heller D, Belliappa S, Perkins AV. Chronic hypoxia in vivo reduces placental oxidative stress. Placenta 28: 846–853, 2007. doi: 10.1016/j.placenta.2006.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zamudio S, Palmer SK, Dahms TE, Berman JC, McCullough RG, McCullough RE, Moore LG. Blood volume expansion, preeclampsia, and infant birth weight at high altitude. J Appl Physiol (1985) 75: 1566–1573, 1993. doi: 10.1152/jappl.1993.75.4.1566. [DOI] [PubMed] [Google Scholar]