Abstract
Melanoma accounts for 7% of all cancers in patients aged 15 to 19 years but is an unexpected malignancy in younger patients. The prevalence of malignant melanoma is very rare in patients aged 1 to 4 years but, certain non-modifiable risk factors such as xeroderma pigmentosa, congenital melanocytic nevus syndrome and other inherited traits increase the risk for its development in these young patients. Recent genomic studies have identified characteristics of pediatric melanoma that differ from conventional melanoma seen in adults. This review informs on the types of melanoma seen in children and adolescents, discusses similarities and differences in melanoma between children and adults, and discusses the role of imaging in the care of these patients.
Introduction
Malignant melanoma in children and adolescents accounts for about 0.4% of all cases of melanoma seen in the US each year. Despite its rarity, melanoma is one of the most common tumors in 15–19 year olds accounting for 7% of all cancers in this age group [1]. Recent reports suggest that the incidence of pediatric melanoma is decreasing particularly in teenagers [2]. Although the prevalence of malignant melanoma is very rare in patients aged 1 to 4 years compared with teenagers, certain non-modifiable risk factors such as such as xeroderma pigmentosa, congenital melanocytic nevus syndrome and other inherited traits increase the risk for its development in these young patients [3–6].
Until recently, pediatric cases of melanoma have been grouped together with adult cases. Genomic studies from our center and others have identified characteristics of pediatric melanoma that differ from conventional melanoma seen in adults [7].
Risk factors for the development of malignant melanoma
Patient phenotype.
Well-recognized characteristics of children who have an increased risk for developing melanoma include fair skin, blue or green eyes, and sun sensitivity [8–11]. However, additional risks are associated with rare syndromes [9, 12, 13], immunosuppression [4, 8, 14, 15], increased number of melanocytic nevi [3, 10, 16–18], family history [8, 11, 14, 15, 19–23], prior chemotherapy [15], and hereditary retinoblastoma [24].
Congenital melanocytic nevus (CMN).
Classification of CMN has been adopted as lesion size anticipated to be reached at adulthood: small (<1.5 cm), medium (1.5–19.9 cm) and large (≥ 20 cm) [3, 25, 26]. The presence of a congenital melanocytic nevus [Figure 1] has been associated with an increased risk for the development of malignant melanoma [17, 26–28]. The approximate lifetime risk of developing melanoma in these patients is 6% [7, 29]. Notably, the majority of melanomas arise within the large congenital nevus, but unknown primary sites and distance primary sites have also been reported [29]. The greatest risk for developing melanoma and rate of death from disease is greatest in giant congenital melanocytic nevi measuring 40 cm or more [7, 28, 29]. These patients, thus, must be closely monitored for life for the development of melanoma [16].
Figure 1.
Clinical image of a young child with a giant congenital melanocytic nevus of the trunk and buttock.
Xeroderma Pigmentosa.
This autosomal recessive disorder is characterized by marked photosensivity due to a defect in ultraviolet ray-induced DNA repair [3, 9, 12]. Patients with xeroderma pigmentosa have a greater than 1000 times increased risk of developing sun-related skin cancers. The median age at the time of diagnosis of any skin cancer (melanomatous and non-melanomatous) is 8 years and multiple skin cancers are common [9]. Malignant melanoma will develop in about 5% of patients with xeroderma pigmentosa, at a median age of 19 years [30, 31]. The distribution of melanoma in patients with xeroderma pigmentosa differs from the general population in that lesions more often involve the face, head, neck (P=0.007) and fewer involve the trunk (P=0.36) [31].
Familial melanoma.
Familial malignant melanoma comprises between 5 and 10% of cases [3, 8, 19, 20, 22]. The Familial Melanoma Syndrome is characterized by at least two cases of melanoma in first- or second-degree relatives or the presence of two or more melanomas in the same patient. A defect in two proteins of the tumor suppressor gene cyclin-dependent kinase inhibitor 2A (CDKN2A) has been identified as the primary high-risk susceptibility gene [20, 21], found in up to 10% of families with two and 30 to 40% of families with more cases of melanoma [11, 20, 22]. Pediatric cases of melanoma are found 6 to 28 times more often in these families than in the general population, regardless of the CDKN2A status [22]. In addition, families who are CDNK2A positive are significantly more likely to develop multiple melanomas than those who are CDNK2A negative [22].
Immunosuppression.
Children with inherited immunosuppression carry a three to six times greater risk for developing malignant melanoma than of the general population [3, 15, 32]. Individuals who have undergone an organ transplantation carry a four-times greater risk and those post- bone marrow transplantation, a five-times greater risk of developing malignant melanoma [15, 21, 32]. The risk of any second cancer was greatest in children younger than 10 years at the time of bone marrow transplantation, estimated to be 36.6 times that expected [32].
Melanoma in children versus adults
The rarity of malignant cutaneous melanoma occurring in pediatric patients may, at least in part, contribute to the frequent delay in diagnosis in children and adolescents [5, 8, 33, 34]. Further, the clinical presentation of conventional melanoma in children often differs from such lesions in adults. Particularly in younger children, melanoma commonly presents as a changing uniformly pigmented or in an amelanotic lesion, often bleeding [8, 35]. Compared to adults, lesions are often thicker and of a more advanced stage in children at the time of diagnosis [8, 34–38]. In one study, approximately 11% (16 of 150) of pediatric cases were misdiagnosed on initial pathologic examination [36]. Conventional melanoma (discussed below) may be seen in all ages of patients and is the same type of melanoma seen in adults [8, 39]. To further complicate the care of pediatric patients with melanoma is the lack of established evidence-based treatment guidelines. As such, pediatric patients are typically treated according to adult guidelines [40, 41] despite differences in the histopathology of several lesions. Thus, pediatric patients with malignant melanoma present a clinical, diagnostic and therapeutic challenge [36].
Pediatric malignant melanoma may be classified into prepubertal (i.e., congenital, infantile, and childhood) and post-pubertal, or adolescent types [4, 42], based upon the patient age at the time of diagnosis [15, 42].
Types of pediatric melanomaCongenital
Melanoma is considered to be congenital when diagnosed in utero or at birth [15, 42]. The extreme rarity of this disease, its often delayed diagnosis and slow metastasis which may be misdiagnosed at a later date as ‘acquired’ disease, precludes accurate estimation of the incidence [42]. In patients with congenital malignant melanoma, approximately one-third of cases of malignant melanoma develop within the central nervous system [43, 44]. Melanoma involving the central nervous system is particularly aggressive with patient survival of <60% at 18 months from diagnosis [42, 43]. A recent study by Kinsler et al, found that the greatest risk for developing melanoma at any site was the presence of any abnormality of the central nervous system and survival was most dependent upon whether or not melanoma developed in the central nervous system. A 12% incidence of CNS melanoma was identified in patients with any congenital abnormality of the central nervous system compared with 1–2% of those with a normal screening MR [Figure 2]. Thus, a role for performing screening MR of the central nervous system during the first year of life in patients with a single large or multiple congenital melanocytic nevi warrants further study [43, 45]. Congenital melanoma may also arise in large or giant congenital nevi or in other isolated lesions. Though rare, de novo melanoma, unrelated to a congenital melanocytic nevus may occur [33, 42]. These are characterized by NRAS mutations [7, 46].
Figure 2.
This 2 year-old boy with congenital hydrocephalus requiring shunting was diagnosed with neurocutaneous melanosis. He developed progressive neurologic deficits including a tilt and eventual inability to walk.
2A Clinical photograph of the multiple congenital nevi on the patient’s legs, representative of those distributed over his trunk, face and extremities.
2B and 2C Axial, B, and sagittal, C, non-contrast T1-weight image showing foci of T1-hyperintensity in the patient’s left frontal lobe (arrows), representative of multiple such lesions in bilateral frontal and temporal lobes, and stable since birth.
2D Sagittal contrast-enhanced image through the lumbar spine obtained during evaluation for progressive neurologic deterioration shows an enhancing intradural mass extending from L2 to L4, histologically proven to be malignant melanoma. Note associated syringomyelia.
Melanoma arising in association with large congenital nevus
A congenital melanocytic nevus is considered to be large when the predicted adult diameter is between 20 and 40 cm and giant, when the predicted adult diameter exceeds 40 cm or 5% or more of the total body surface area upon reaching adulthood [7, 29]. Patients with a giant congenital melanocytic nevus have an estimated lifetime risk for developing melanoma ranging from 5% to 40% [14, 47, 48]. As mentioned above, most – but not all - melanomas arising in large or giant congenital melanocytic nevi occur within the lesion [7, 29, 49]. Recent investigations have revealed that 80% of melanomas arising in association with a congenital melanocytic nevus have a mutation of the NRAS oncogene [7, 43, 46, 50] and have epigenetic activation of the TERT promoter gene [7, 43, 44]. A meta-analysis by Krengel et al [29] found that the overall incidence of melanoma was low when all sizes of congenital melanocytic nevi were included and that the median age for development of melanoma in these patients was 7 years. The investigators further found that the melanoma risk was strongly tied to the size of the nevus [29]. Similarly, a meta-analysis of eight studies (432 patient with large congenital melanocytic nevi) comprising 2578 patients found that 2% of the patients developed melanoma; 74% were in nevi that exceeded 40 cm and 94% of cases had satellite lesions [51].
Spitzoid melanoma
Spitzoid tumors are more commonly found in patients younger than 20 years of age and more often in children than in adolescents [7, 52]. Spitzoid melanomas commonly present as an amelanotic polyp, papule or nodules; pink or red in color and often mistaken for a wart or pyogenic granuloma [7, 35, 37, 53, 54]. These lesions often have locoregional nodal metastases and rarely develop distant metastases; outcomes are excellent [7, 55, 56]. The TERT promoter mutations are usually absent in these tumors which may explain the rarity of distant dissemination. However, those with a TERT promoter mutation have an aggressive clinical course [55].
Conventional (adult) melanoma
Typically, the melanoma seen in adolescents is that of conventional melanoma as seen in adults and managed according to adult guidelines [7, 15, 39, 57]. These tumors rarely develop in children and only occasionally develop in periadolescent patients [7]. Those in whom these tumors occur are phenotypically characterized by ‘at risk’ features: Caucasian, fair skin, sun sensitivity, multiple nevi [7, 58]. As in adults, these tumors frequently have activating mutations in promoters of serine/threonine-protein kinase B-Raf (BRAF) and TERT as well as inactivating mutations in tumor suppressor genes of CDKN2A and phosphatase and tensin homolog (PTEN) [7, 57, 59] and are associated with more aggressive disease and distant metastases [59].
Imaging pediatric and adolescent melanoma
Stage at the time of diagnosis is most predictive of outcome [33, 34, 60]. Disease free and overall survival is strongly correlated with the thickness of the lesion, presence or absence of ulceration, sentinel lymph node involvement, and number of positive lymph nodes [61]. Malignant melanoma can metastasize anywhere in the body by direct extension, lymphatic spread and hematogenous dissemination. Local disease is determined by clinical and pathologic stage as defined by the staging system developed by the American Joint Committee on Cancer (AJCC) [60, 62].
Determination of regional disease involvement focuses on lymph node involvement. Nodal metastases tend to occur in lesions that are thicker and larger [33, 60]. Clinical assessment for nodal disease is inaccurate as evidenced by 20% of clinically normal regional lymph nodes being histologically positive for microscopic disease [63] and about 20% of clinically abnormal nodes are negative for metastatic disease [63].
A recent report on over 500 Brazilian children and young adult patients with melanoma identified male gender, lesion thickness greater than 4mm, ulceration and nodular histologic subtype as risk factors for developing metastatic disease [64]. Notably, 24% of children with lesions 1 to 2 mm thick but not ulcerated had sentinel lymph node metastases compared with 4% of young adults (P<.001) [64].
Sentinel node lymphoscintigraphy plays a key role in directing surgical resection and nodal biopsy. As lymph node metastases from melanoma are frequently microscopic, lymphoscintigraphy is probably the most sensitive imaging study for directing sentinel node biopsy and is, thus, useful for initial staging in children, adolescents and adults [63, 65–67]. Knowledge of the draining beds and ‘in transit’ paths of lymphatic spread is critical for directing the imaging phase of the procedure. The prognostic significance of lymphoscintigraphy with sentinel node biopsy is inconsistent but pediatric patients with positive sentinel lymph node biopsy had thicker lesions and were more likely to have ulceration [38, 68, 69]. Children with spitzoid melanoma have a much higher incidence (up to 40%) of sentinel lymph node metastases but outcome remains favorable [56]. Distant sites of recurrence are also more frequent in the lymph node biopsy positive group (71% compared with 49%, respectively, p=0.06) [61]. Lymphoscintigraphy with sentinel node biopsy may be prognostically important even in thin lesions [70]. However, a negative sentinel node biopsy does not guarantee against disease recurrence which most often is seen as distant metastases [61, 71].
Compared with planar techniques, contemporary SPECT/CT [Figure 3]may provide increased conspicuity of positive lymph nodes, in transit lesions and provide improved mapping for surgical planning which may also reduce costs related to the length of the surgical procedure in children and adults [72, 73]. In general, routine staging with computed tomography or whole body PET-CT have not been shown to be valuable in further staging of asymptomatic patients with positive sentinel lymph nodes due to false positive findings, cost and low sensitivity, specificity and positive predictive value [62, 74].
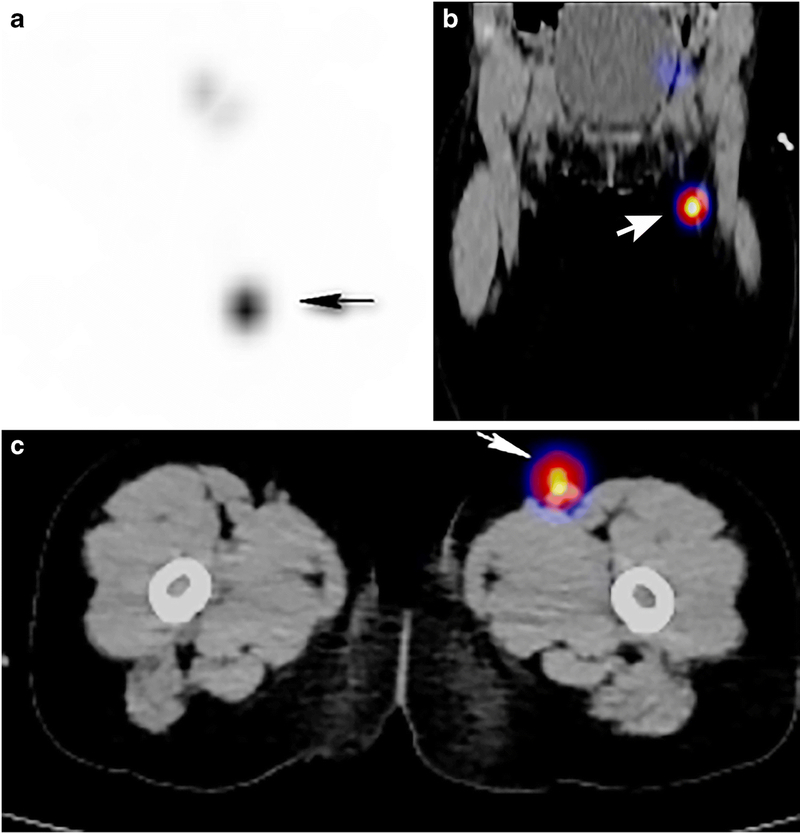
Figure 3.
3 year-old girl with spitzoid melanoma left lower leg.
3A Coronal planar image from lymphoscintigram showing focal metabolic activity in the proximal medial left thigh.
3B and 3C Coronal, B, and axial, C, fused images from the SPECT CT shows to exquisitely better advantage the relationship between the sentinel node and adjacent structures, enhancing surgical planning.
Dedicated ultrasound of regional lymph nodes has been reported as having an overall sensitivity ranging from 24% to 60% and 97% specificity for detection of known nodal metastases [62, 75, 76] which improved when tumor deposits were at least 4.5mm in size and when imaging the neck [62, 75]. Voit et al [77] investigated the use of dedicated ultrasound in over 1000 stage I and II patients with melanoma. The investigators found that using the ultrasound criteria of peripheral perfusion, balloon nodal shape, and loss of central nodal echoes, ultrasound correctly identified at least half of sentinel lymph nodes prior to lymphoscintigraphy. Machet et al also found ultrasound to be superior in sensitivity (92.9% compared to 71.4%) and similar in specificity (99.6% versus 97.8%) to clinical examination in 373 patients (pediatric and adult) with Stage I or II melanoma. The investigators found that the ‘balloon shape’ of a lymph node corresponded to advanced metastatic disease [Figure 5]. Ultrasound alone – without fine needle aspiration - had a sensitivity of 71% for detection of metastatic involvement. Of these, 72% were confirmed by ultrasound directed fine needle aspiration to represent metastases. However, these authors do not advocate routine follow-up ultrasound imaging [78, 79] as they found that only 7% of patients received a benefit from ultrasound results of earlier detection of nodal involvement or avoidance of surgery while 6% suffered stress or unnecessary removal of a benign lymph node [78].
Figure 5.
7 year-old girl had been diagnosed with spitzoid melanoma right upper arm. She underwent wide local reexcision including an in transit nodule, and axillary lymph node sampling several months after initial diagnosis. 18F-FDG PET-CT was performed as a means of closely monitoring for additional disease recurrence. Targeted ultrasound was used for assessing a clinically palpable nodule and for lesion localization for surgical removal.
2A Fused coronal whole body maximum intensity pixel image from 18F-FDG PET-CT showing two tiny metabolically active skin nodules right upper arm obtained 4 months after definitive surgery.
2B and C Fused axial 18F-FDG PET-CT images which correlate with the tiny nodules shown in 2A. 2B shows residual activity through the primary resection scar (more superior nodule) and 2C, through the more subtle and inferior nodule. Routine ultrasound through the region of the scar was negative at this time.
Distant metastases may be detected by diagnostic computed tomography and 18F-FDG PET/PET-CT. The value of 18F-FDG PET/PET-CT alone has been inconsistently shown for disease detection [62]. Further, the timing of 18F-FDG PET/PET-CT during the staging process must be considered - prior to or after lymphoscintigraphy. The sensitivity of 18F-FDG PET/PET-CT is partially dependent upon tumor burden [80]; melanoma metastases are often microscopic and, thus, may be too small for detection. 18F-FDG PET-CT has been reported to have 21% sensitivity for lymph node metastases in an adult series of patients [81]. However, in a meta-analysis of 74 publications comprised of 10,528 patients imaged with CT, ultrasonography, 18F-FDG PET and PET-CT for initial staging and follow-up of melanoma, 18F-FDG PET-CT was found to have the highest sensitivity of 80% and 87% specificity for detection of distant metastases both at diagnosis and at follow-up (86% sensitivity and 91% specificity, respectively) [Figure 4] [62, 76]. Pfannenberg et al found that 18F-FDG PET-CT in adult patients had an overall accuracy of 87% compared with whole body MR of 79% [82] from the German 18F-FDG PET-CT registry and that the results of 18F-FDG PET-CT prompted a change in planned management in 46% of 750 adult patients with melanoma [83]. As mentioned above, Xing et al found ultrasonography to be most sensitive (96%) and specific (99%) for identifying lymph node metastases [76, 84]. Other investigators reported that US may be considered for detection or monitoring of regional nodal disease when sentinel node biopsy is not performed at initial staging [62, 76, 78]. Brain MR has been shown to be more sensitive than 18F-FDG PET/PET-CT or CT for the detection of intracranial metastases [63, 85, 86].
Figure 4.
14 year-old girl with history of having undergone shave biopsy of a nodular lesion mid back which was histologically diagnosed as malignant melanoma, nodular type. She subsequently underwent wide local resection primary site of disease, bilateral axillary node biopsies and biopsy of left breast nodule (not shown), all found to represent metastatic melanoma.
5A Coronal 18F-FDG PET-CT image showing foci of metabolic activity in both axilla (arrows).
5B and 5C Axial fused PET-CT images through the axillae corresponding to the sites of metabolic activity seen in Figure 5A (long arrows). Note also subtle metabolic activity in the superficial soft tissues of the back (short arrow), site of shave biopsy of primary malignant melanoma.
5D Axial contrast-enhance CT image through the axillae show bilaterally enlarged abnormal axillary lymph nodes (arrows), found histologically to represent sites of metastatic melanoma.
5E and 5F Dedicated ultrasound images of axillary lymph nodes (E, right, and, F, left) for localization and characterization for interventional radiology fine needle aspiration. Note rounded ‘balloon shape’, loss of central hilum, bilaterally. Peripheral hypervascularity of the left axillary node is also demonstrated. Ultrasound findings indicative of tumor involvement.
For AJCC stage I and II disease, the National Comprehensive Cancer Network (NCCN) recommends no additional imaging. If imaging is needed in patients of any age, it should target specific signs and symptoms [87]. A high false positivity rate associated with FDG-PET-CT and low sensitivity for detecting occult distant metastases or regional metastases following a positive sentinel lymph node, limits the utility of PET-CT in low stage disease [88, 89].
For more advanced Stage III and IV disease, imaging should also target specific signs and symptoms but may be incorporated to detect disease recurrence or metastatic disease at 3 to 12 month intervals. Additional staging and follow-up imaging as recommended by the American Joint Commission on Cancer depends upon the histologic stage of disease [87]. A 2014 meta-analysis of studies reported between 1990 and 2012 comprised of over 600 patients found 18F-FDG PET to have an overall sensitivity of 89.4%, specificity of 88.8% in detecting distant metastases in Stage III disease [90]. Thus, for Stage III and IV disease, 18F-FDG PET-CT is reported to be the most sensitive and specific imaging modality in pediatric and adults patients for detecting occult distant metastases in the trunk and extremities [62, 84, 91]. MR brain is recommended for detection of brain metastases [62, 86]. CT may be helpful in identifying occult metastases in adolescent and adult patients with melanoma of the head and neck prior to lymphoscintigraphy [92].
A recent investigation of 1152 adult patients with melanoma comparing 18FFDG PET-MR with DWI, 18F-FDG PET-CT, and sentinel node lymphoscintigraphy and biopsy concluded that PET-MR was unable to differentiate positive from negative lymph node involvement and that both 18F-FDG PET-MR (even with the addition of DWI) and 18F-FDG PET-CT had low sensitivity (23.5% and 17.7%, respectively) with high specificity (95.6% and 96.9%, respectively) for detection of nodal involvement [93]. Whole body MR with or without DWI may have a role in detecting skeletal metastases which may occur in 23–49% of patients [85, 94–96] but has limitations in regards to detecting pulmonary metastases as reported in young adult and adult patients [82, 95]. Whole body MR may be more accurate than 18F-FDG PET-CT in identifying metastases in the liver and brain, as reported in adults [82].
Inasmuch as there is a paucity of guidelines for imaging pediatric patients with melanoma, and as pediatric melanoma typically differs histologically from adult melanoma, we recently rereviewed our experience. In our prior report we advocated routine CT chest-abdomen-pelvis for staging and follow-up of pediatric patients with thick melanoma or those with unknown primary lesions as we found clinically occult lesions in 25% of cases [97]. With the current availability of more refined subtyping of melanoma in children, we found that in children with spitzoid melanoma (especially those who lacked the TERT promoter mutation) required very little imaging at diagnosis and for follow-up, beyond initial staging. These patients, staged with sentinel node lymphoscintigraphy and biopsy, had favorable outcomes. The only death in the patient group with spitzoid melanoma was TERT promoter positive. However, those pediatric patients who had conventional melanoma, warranted imaging according to adult guidelines [98].
Surveillance imaging
Guidelines for routine off therapy surveillance are sparse, report inconsistent results, are retrospective and based upon predominantly adult populations. A recent report by Berger et al [99] indicates that patient symptoms were the most frequent means of detecting recurrence in a large group predominately of adults, with stage II melanoma; the site of recurrence was most often regional lymph nodes. This finding is an important aspect of long-term follow-up of patients with low stage disease. Ultrasound of regional lymph nodes is advocated as a non-invasive and sensitive method that can augment physical examination as reported in adolescent and adult patients [100, 101]. In our own [unpublished] experience, ultrasound interrogation of the primary site, of the residual scar and of any palpable nodule serves as a low cost, low risk method for assessing for local recurrence and is useful for localizing palpable nodules [Figure 5].
There is little consistent evidence that routine follow-up imaging provides significant gain in survival, even in patients with more advanced (Stage III) disease [101]. Reuth et al reported that patients with stage III surgically resected melanoma who have survived for at least 8 years, might not benefit from surveillance any more intensive than those with low stage disease [102]. Parks et al, however, found that routine CT chest-abdomen-pelvis detected 75% of asymptomatic distant sites of disease in 466 patients with Stage III or IV resected disease, enrolled on adjuvant immunotherapy trials [103].
Summary
Malignant melanoma is a complex malignancy with the potential for widespread and asymptomatic metastases. Its behavior and subtypes in young children vary from conventional melanoma seen in adults. Imaging serves a role in caring for these patients, especially those with advanced disease, but must be considered in light of disease stage, melanoma subtype and modality limitations. Once staged, guidelines for imaging focus on assessment of local and regional nodal involvement and organ- or specific symptoms. Risks, benefits, imaging costs, radiation exposure, false positive rates [99, 101, 104], must all be considered, particularly when caring for pediatric patients.
Acknowledgement:
I thank Vella Laws-Bell for manuscript preparation and Dr. Alberto Pappo for critical review.
References
- 1.Howlader N, Noon AM, Kropcho M, et al. (2017) SEER Cancer Statistics Review National Cancer Institute, pp 1975–2014. [Google Scholar]
- 2.Campbell LB, Kreicher KL, Gittleman HR, et al. (2015) Melanoma Incidence in Children and Adolescents: Decreasing Trends in the United States. J Pediatr 166:1505–1513. [DOI] [PubMed] [Google Scholar]
- 3.Wong JR, Harris JK, Rodriguez-Galindo C, et al. (2013) Incidence of childhood and adolescent melanoma in the United States: 1973–2009. Pediatrics 131:846–854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jen M, Murphy M, Grant-Kels JM (2009) Childhood melanoma. Clin Dermatol 27:529–536. [DOI] [PubMed] [Google Scholar]
- 5.Indini A, Brecht I, Del Vecchio M, et al. (2018) Cutaneous melanoma in adolescents and young adults. Pediatr Blood Cancer 65:e27292. [DOI] [PubMed] [Google Scholar]
- 6.St. Jude Children’s Research Hospital (2018) Melanoma. St. Jude Children’s Research Hospital; Memphis, TN. [Google Scholar]
- 7.Bahrami A, Barnhill RL (2018) Pathology and genomics of pediatric melanoma: A critical reexamination and new insights. Pediatr Blood Cancer 65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Stefanaki C, Chardalias L, Soura E, et al. (2017) Paediatric melanoma. J Eur Acad Dermatol Venereol 31:1604–1615. [DOI] [PubMed] [Google Scholar]
- 9.Kraemer KH, Lee MM, Andrews AD, et al. (1994) The role of sunlight and DNA repair in melanoma and nonmelanoma skin cancer. The xeroderma pigmentosum paradigm. Arch Dermatol 130:1018–1021. [PubMed] [Google Scholar]
- 10.Kopf AW, Kripke ML, Stern RS (1984) Sun and malignant melanoma. J Am Acad Dermatol 11:674–684. [DOI] [PubMed] [Google Scholar]
- 11.Williams ML, Sagebiel RW (1994) Melanoma risk factors and atypical moles. West J Med 160:343–350. [PMC free article] [PubMed] [Google Scholar]
- 12.Downard CD, Rapkin LB, Gow KW (2007) Melanoma in children and adolescents. Surg Oncol 16:215–220. [DOI] [PubMed] [Google Scholar]
- 13.Schultz KAP, Rednam SP, Kamihara J, et al. (2017) PTEN, DICER1, FH, and Their Associated Tumor Susceptibility Syndromes: Clinical Features, Genetics, and Surveillance Recommendations in Childhood. Clin Cancer Res 23:e76–e82. [DOI] [PubMed] [Google Scholar]
- 14.Rhodes AR, Weinstock MA, Fitzpatrick TB, et al. (1987) Risk factors for cutaneous melanoma. A practical method of recognizing predisposed individuals. JAMA 258:3146–3154. [PubMed] [Google Scholar]
- 15.Pappo AS (2003) Melanoma in children and adolescents. Eur J Cancer 39:2651–2661. [DOI] [PubMed] [Google Scholar]
- 16.Levy R, Lara-Corrales I (2016) Melanocytic Nevi in Children: A Review. Pediatr Ann 45:e293–298. [DOI] [PubMed] [Google Scholar]
- 17.Bittencourt FV, Marghoob AA, Kopf AW, et al. (2000) Large congenital melanocytic nevi and the risk for development of malignant melanoma and neurocutaneous melanocytosis. Pediatrics 106:736–741. [DOI] [PubMed] [Google Scholar]
- 18.Masaki T, Wang Y, DiGiovanna JJ, et al. (2014) High frequency of PTEN mutations in nevi and melanomas from xeroderma pigmentosum patients. Pigment Cell Melanoma Res 27:454–464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kopf AW, Hellman LJ, Rogers GS, et al. (1986) Familial malignant melanoma. JAMA 256:1915–1919. [PubMed] [Google Scholar]
- 20.Sa BCS, Moredo LF, Gomes EE, et al. (2018) Hereditary melanoma: a five-year study of Brazilian patients in a cancer referral center - phenotypic characteristics of probands and pathological features of primary tumors. An Bras Dermatol 93:337–340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kefford RF, Newton Bishop JA, Bergman W, et al. (1999) Counseling and DNA testing for individuals perceived to be genetically predisposed to melanoma: A consensus statement of the Melanoma Genetics Consortium. J Clin Oncol 17:3245–3251. [DOI] [PubMed] [Google Scholar]
- 22.Goldstein AM, Stidd KC, Yang XR, et al. (2018) Pediatric Melanoma in Melanoma-Prone Families. Cancer. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Goldstein AM, Xiao Y, Sampson J, et al. (2017) Rare germline variants in known melanoma susceptibility genes in familial melanoma. Hum Mol Genet 26:4886–4895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wong JR, Morton LM, Tucker MA, et al. (2014) Risk of subsequent malignant neoplasms in long-term hereditary retinoblastoma survivors after chemotherapy and radiotherapy. J Clin Oncol 32:3284–3290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Viana ACL, Goulart EMA, Gontijo B, et al. (2017) A prospective study of patients with large congenital melanocytic nevi and the risk of melanoma. An Bras Dermatol 92:200–205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kopf AW, Bart RS, Hennessey P (1979) Congenital nevocytic nevi and malignant melanomas. J Am Acad Dermatol 1:123–130. [DOI] [PubMed] [Google Scholar]
- 27.Gari LM, Rivers JK, Kopf AW (1988) Melanomas arising in large congenital nevocytic nevi: a prospective study. Pediatr Dermatol 5:151–158. [DOI] [PubMed] [Google Scholar]
- 28.Fan Y, Lee S, Wu G, et al. (2016) Telomerase Expression by Aberrant Methylation of the TERT Promoter in Melanoma Arising in Giant Congenital Nevi. J Invest Dermatol 136:339–342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Krengel S, Hauschild A, Schafer T (2006) Melanoma risk in congenital melanocytic naevi: a systematic review. Br J Dermatol 155:1–8. [DOI] [PubMed] [Google Scholar]
- 30.Lambert WC, Kuo HR, Lambert MW (1995) Xeroderma pigmentosum. Dermatol Clin 13:169–209. [PubMed] [Google Scholar]
- 31.Kraemer KH, Lee MM, Scotto J (1987) Xeroderma pigmentosum. Cutaneous, ocular, and neurologic abnormalities in 830 published cases. Arch Dermatol 123:241–250. [DOI] [PubMed] [Google Scholar]
- 32.Curtis RE, Rowlings PA, Deeg HJ, et al. (1997) Solid cancers after bone marrow transplantation. N Engl J Med 336:897–904. [DOI] [PubMed] [Google Scholar]
- 33.Wu SJ, Lambert DR (1997) Melanoma in children and adolescents. Pediatr Dermatol 14:87–92. [DOI] [PubMed] [Google Scholar]
- 34.Lam PH, Obirieze AC, Ortega G, et al. (2018) An Age-Based Analysis of Pediatric Melanoma: Staging, Surgery, and Mortality in the Surveillance, Epidemiology, and End Results Database. Am Surg 84:739–745. [PubMed] [Google Scholar]
- 35.Ferrari A, Bono A, Baldi M, et al. (2005) Does melanoma behave differently in younger children than in adults? A retrospective study of 33 cases of childhood melanoma from a single institution. Pediatrics 115:649–654. [DOI] [PubMed] [Google Scholar]
- 36.Aldrink JH, Selim MA, Diesen DL, et al. (2009) Pediatric melanoma: a single-institution experience of 150 patients. J Pediatr Surg 44:1514–1521. [DOI] [PubMed] [Google Scholar]
- 37.Lange JR, Palis BE, Chang DC, et al. (2007) Melanoma in children and teenagers: an analysis of patients from the National Cancer Data Base. J Clin Oncol 25:1363–1368. [DOI] [PubMed] [Google Scholar]
- 38.Mu E, Lange JR, Strouse JJ (2012) Comparison of the use and results of sentinel lymph node biopsy in children and young adults with melanoma. Cancer 118:2700–2707. [DOI] [PubMed] [Google Scholar]
- 39.Brecht IB, De Paoli A, Bisogno G, et al. (2018) Pediatric patients with cutaneous melanoma: A European study. Pediatr Blood Cancer 65:e26974. [DOI] [PubMed] [Google Scholar]
- 40.Tracy ET, Aldrink JH (2016) Pediatric melanoma. Semin Pediatr Surg 25:290–298. [DOI] [PubMed] [Google Scholar]
- 41.Austin MT, Xing Y, Hayes-Jordan AA, et al. (2013) Melanoma incidence rises for children and adolescents: an epidemiologic review of pediatric melanoma in the United States. J Pediatr Surg 48:2207–2213. [DOI] [PubMed] [Google Scholar]
- 42.Richardson SK, Tannous ZS, Mihm MC Jr. (2002) Congenital and infantile melanoma: review of the literature and report of an uncommon variant, pigment-synthesizing melanoma. J Am Acad Dermatol 47:77–90. [DOI] [PubMed] [Google Scholar]
- 43.Kinsler VA, O’Hare P, Bulstrode N, et al. (2017) Melanoma in congenital melanocytic naevi. Br J Dermatol 176:1131–1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Neuhold JC, Friesenhahn J, Gerdes N, et al. (2015) Case reports of fatal or metastasizing melanoma in children and adolescents: a systematic analysis of the literature. Pediatr Dermatol 32:13–22. [DOI] [PubMed] [Google Scholar]
- 45.Waelchli R, Aylett SE, Atherton D, et al. (2015) Classification of neurological abnormalities in children with congenital melanocytic naevus syndrome identifies magnetic resonance imaging as the best predictor of clinical outcome. Br J Dermatol 173:739–750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kinsler VA, Thomas AC, Ishida M, et al. (2013) Multiple congenital melanocytic nevi and neurocutaneous melanosis are caused by postzygotic mutations in codon 61 of NRAS. J Invest Dermatol 133:2229–2236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Tannous ZS, Mihm MC Jr., Sober AJ, et al. (2005) Congenital melanocytic nevi: clinical and histopathologic features, risk of melanoma, and clinical management. J Am Acad Dermatol 52:197–203. [DOI] [PubMed] [Google Scholar]
- 48.Sober AJ, Burstein JM (1995) Precursors to skin cancer. Cancer 75:645–650. [DOI] [PubMed] [Google Scholar]
- 49.Marghoob AA, Schoenbach SP, Kopf AW, et al. (1996) Large congenital melanocytic nevi and the risk for the development of malignant melanoma. A prospective study. Arch Dermatol 132:170–175. [PubMed] [Google Scholar]
- 50.Charbel C, Fontaine RH, Malouf GG, et al. (2014) NRAS mutation is the sole recurrent somatic mutation in large congenital melanocytic nevi. J Invest Dermatol 134:1067–1074. [DOI] [PubMed] [Google Scholar]
- 51.Vourc’h-Jourdain M, Martin L, Barbarot S, et al. (2013) Large congenital melanocytic nevi: therapeutic management and melanoma risk: a systematic review. J Am Acad Dermatol 68:493–498 e491–414. [DOI] [PubMed] [Google Scholar]
- 52.Bartenstein DW, Kelleher CM, Friedmann AM, et al. (2018) Contrasting features of childhood and adolescent melanomas. Pediatr Dermatol 35:354–360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Cordoro KM, Gupta D, Frieden IJ, et al. (2013) Pediatric melanoma: results of a large cohort study and proposal for modified ABCD detection criteria for children. J Am Acad Dermatol 68:913–925. [DOI] [PubMed] [Google Scholar]
- 54.Livestro DP, Kaine EM, Michaelson JS, et al. (2007) Melanoma in the young: differences and similarities with adult melanoma: a case-matched controlled analysis. Cancer 110:614–624. [DOI] [PubMed] [Google Scholar]
- 55.Lee S, Barnhill RL, Dummer R, et al. (2015) TERT Promoter Mutations Are Predictive of Aggressive Clinical Behavior in Patients with Spitzoid Melanocytic Neoplasms. Sci Rep 5:11200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lallas A, Kyrgidis A, Ferrara G, et al. (2014) Atypical Spitz tumours and sentinel lymph node biopsy: a systematic review. Lancet Oncol 15:e178–183. [DOI] [PubMed] [Google Scholar]
- 57.Lu C, Zhang J, Nagahawatte P, et al. (2015) The genomic landscape of childhood and adolescent melanoma. J Invest Dermatol 135:816–823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Strouse JJ, Fears TR, Tucker MA, et al. (2005) Pediatric melanoma: risk factor and survival analysis of the surveillance, epidemiology and end results database. J Clin Oncol 23:4735–4741. [DOI] [PubMed] [Google Scholar]
- 59.Seynnaeve B, Lee S, Borah S, et al. (2017) Genetic and Epigenetic Alterations of TERT Are Associated with Inferior Outcome in Adolescent and Young Adult Patients with Melanoma. Sci Rep 7:45704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Gershenwald JE, Scolyer RA (2018) Melanoma Staging: American Joint Committee on Cancer (AJCC) 8th Edition and Beyond. Ann Surg Oncol 25:2105–2110. [DOI] [PubMed] [Google Scholar]
- 61.Wagner JD, Ranieri J, Evdokimow DZ, et al. (2003) Patterns of initial recurrence and prognosis after sentinel lymph node biopsy and selective lymphadenectomy for melanoma. Plast Reconstr Surg 112:486–497. [DOI] [PubMed] [Google Scholar]
- 62.Stodell M, Thompson JF, Emmett L, et al. (2017) Melanoma patient imaging in the era of effective systemic therapies. Eur J Surg Oncol 43:1517–1527. [DOI] [PubMed] [Google Scholar]
- 63.Donohoe K, Atkins M, Vora S (2017) Imaging studies in melanoma. UpToDate. [Google Scholar]
- 64.Cherobin A, Wainstein AJA, Colosimo EA, et al. (2018) Prognostic factors for metastasis in cutaneous melanoma. An Bras Dermatol 93:19–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Toro J, Ranieri JM, Havlik RJ, et al. (2003) Sentinel lymph node biopsy in children and adolescents with malignant melanoma. J Pediatr Surg 38:1063–1065. [DOI] [PubMed] [Google Scholar]
- 66.Moncayo VM, Alazraki AL, Alazraki NP, et al. (2017) Sentinel Lymph Node Biopsy Procedures. Semin Nucl Med 47:595–617. [DOI] [PubMed] [Google Scholar]
- 67.Jaukovic L, Sijan G, Rajovic M, et al. (2015) Lymphoscintigraphy and sentinel lymph node biopsy, in cutaneous melanoma staging and treatment decisions. Hell J Nucl Med 18:146–151. [DOI] [PubMed] [Google Scholar]
- 68.Kim J, Sun Z, Gulack BC, et al. (2016) Sentinel lymph node biopsy is a prognostic measure in pediatric melanoma. J Pediatr Surg 51:986–990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Parida L, Morrisson GT, Shammas A, et al. (2012) Role of lymphoscintigraphy and sentinel lymph node biopsy in the management of pediatric melanoma and sarcoma. Pediatr Surg Int 28:571–578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ranieri JM, Wagner JD, Wenck S, et al. (2006) The prognostic importance of sentinel lymph node biopsy in thin melanoma. Ann Surg Oncol 13:927–932. [DOI] [PubMed] [Google Scholar]
- 71.O’Connell EP, O’Leary DP, Fogarty K, et al. (2016) Predictors and patterns of melanoma recurrence following a negative sentinel lymph node biopsy. Melanoma Res 26:66–70. [DOI] [PubMed] [Google Scholar]
- 72.Doepker MP, Yamamoto M, Applebaum MA, et al. (2017) Comparison of Single-Photon Emission Computed Tomography-Computed Tomography (SPECT/CT) and Conventional Planar Lymphoscintigraphy for Sentinel Node Localization in Patients with Cutaneous Malignancies. Ann Surg Oncol 24:355–361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Stoffels I, Muller M, Geisel MH, et al. (2014) Cost-effectiveness of preoperative SPECT/CT combined with lymphoscintigraphy vs. lymphoscintigraphy for sentinel lymph node excision in patients with cutaneous malignant melanoma. Eur J Nucl Med Mol Imaging 41:1723–1731. [DOI] [PubMed] [Google Scholar]
- 74.Holtkamp LHJ, Read RL, Emmett L, et al. (2017) Futility of imaging to stage melanoma patients with a positive sentinel lymph node. Melanoma Res 27:457–462. [DOI] [PubMed] [Google Scholar]
- 75.Sanki A, Uren RF, Moncrieff M, et al. (2009) Targeted high-resolution ultrasound is not an effective substitute for sentinel lymph node biopsy in patients with primary cutaneous melanoma. J Clin Oncol 27:5614–5619. [DOI] [PubMed] [Google Scholar]
- 76.Xing Y, Bronstein Y, Ross MI, et al. (2011) Contemporary diagnostic imaging modalities for the staging and surveillance of melanoma patients: a meta-analysis. J Natl Cancer Inst 103:129–142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Voit CA, Gooskens SL, Siegel P, et al. (2014) Ultrasound-guided fine needle aspiration cytology as an addendum to sentinel lymph node biopsy can perfect the staging strategy in melanoma patients. Eur J Cancer 50:2280–2288. [DOI] [PubMed] [Google Scholar]
- 78.Machet L, Nemeth-Normand F, Giraudeau B, et al. (2005) Is ultrasound lymph node examination superior to clinical examination in melanoma follow-up? A monocentre cohort study of 373 patients. Br J Dermatol 152:66–70. [DOI] [PubMed] [Google Scholar]
- 79.Machet L, Perrinaud A, Giraudeau B (2005) Routine ultrasonography in melanoma follow-up? Lancet Oncol 6:2; discussion 3. [DOI] [PubMed] [Google Scholar]
- 80.Wagner JD, Schauwecker DS, Davidson D, et al. (2001) FDG-PET sensitivity for melanoma lymph node metastases is dependent on tumor volume. J Surg Oncol 77:237–242. [DOI] [PubMed] [Google Scholar]
- 81.Wagner JD, Schauwecker D, Davidson D, et al. (2005) Inefficacy of F-18 fluorodeoxy-D-glucose-positron emission tomography scans for initial evaluation in early-stage cutaneous melanoma. Cancer 104:570–579. [DOI] [PubMed] [Google Scholar]
- 82.Pfannenberg C, Aschoff P, Schanz S, et al. (2007) Prospective comparison of 18F-fluorodeoxyglucose positron emission tomography/computed tomography and whole-body magnetic resonance imaging in staging of advanced malignant melanoma. Eur J Cancer 43:557–564. [DOI] [PubMed] [Google Scholar]
- 83.Pfannenberg C, Gueckel B, Wang L, et al. (2018) Practice-based evidence for the clinical benefit of PET/CT-results of the first oncologic PET/CT registry in Germany. Eur J Nucl Med Mol Imaging. [DOI] [PubMed] [Google Scholar]
- 84.Xing Y, Cromwell KD, Cormier JN (2012) Review of diagnostic imaging modalities for the surveillance of melanoma patients. Dermatol Res Pract 2012:941921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Stecco A, Trisoglio A, Soligo E, et al. (2018) Whole-Body MRI with Diffusion-Weighted Imaging in Bone Metastases: A Narrative Review. Diagnostics (Basel) 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Petralia G, Padhani A, Summers P, et al. (2013) Whole-body diffusion-weighted imaging: is it all we need for detecting metastases in melanoma patients? Eur Radiol 23:3466–3476. [DOI] [PubMed] [Google Scholar]
- 87.NCCN (2018) Cutaneous Melanoma.
- 88.Wagner JD (2006) Fluorodeoxyglucose positron emission tomography for melanoma staging: refining the indications. Ann Surg Oncol 13:444–446. [DOI] [PubMed] [Google Scholar]
- 89.Wagner JD (2003) PET detection of melanoma metastases in lymph nodes. J Nucl Med 44:486. [PubMed] [Google Scholar]
- 90.Rodriguez Rivera AM, Alabbas H, Ramjaun A, et al. (2014) Value of positron emission tomography scan in stage III cutaneous melanoma: a systematic review and meta-analysis. Surg Oncol 23:11–16. [DOI] [PubMed] [Google Scholar]
- 91.Reinhardt MJ, Joe AY, Jaeger U, et al. (2006) Diagnostic performance of whole body dual modality 18F-FDG PET/CT imaging for N- and M-staging of malignant melanoma: experience with 250 consecutive patients. J Clin Oncol 24:1178–1187. [DOI] [PubMed] [Google Scholar]
- 92.Hafstrom A, Silfverschiold M, Persson SS, et al. (2017) Benefits of initial CT staging before sentinel lymph node biopsy in patients with head and neck cutaneous melanoma. Head Neck 39:2301–2310. [DOI] [PubMed] [Google Scholar]
- 93.Schaarschmidt BM, Grueneisen J, Stebner V, et al. (2018) Can integrated 18F-FDG PET/MR replace sentinel lymph node resection in malignant melanoma? Eur J Nucl Med Mol Imaging. [DOI] [PubMed] [Google Scholar]
- 94.Akslen LA, Hove LM, Hartveit F (1987) Metastatic distribution in malignant melanoma. A 30-year autopsy study. Invasion Metastasis 7:253–263. [PubMed] [Google Scholar]
- 95.Mosavi F, Ullenhag G, Ahlstrom H (2013) Whole-body MRI including diffusion-weighted imaging compared to CT for staging of malignant melanoma. Ups J Med Sci 118:91–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Laurent V, Trausch G, Bruot O, et al. (2010) Comparative study of two whole-body imaging techniques in the case of melanoma metastases: advantages of multi-contrast MRI examination including a diffusion-weighted sequence in comparison with PET-CT. Eur J Radiol 75:376–383. [DOI] [PubMed] [Google Scholar]
- 97.Kaste SC, Pappo AS, Jenkins JJ 3rd, et al. (1996) Malignant melanoma in children: imaging spectrum. Pediatr Radiol 26:800–805. [DOI] [PubMed] [Google Scholar]
- 98.Halalsheh H, Kaste SC, Navid F, et al. (2018) The role of routine imaging in pediatric cutaneous melanoma. Pediatr Blood Cancer 65:e27412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Berger AC, Ollila DW, Christopher A, et al. (2017) Patient Symptoms Are the Most Frequent Indicators of Recurrence in Patients with American Joint Committee on Cancer Stage II Melanoma. J Am Coll Surg 224:652–659. [DOI] [PubMed] [Google Scholar]
- 100.Rueth NM, Cromwell KD, Cormier JN (2015) Long-term follow-up for melanoma patients: is there any evidence of a benefit? Surg Oncol Clin N Am 24:359–377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Rueth NM, Xing Y, Chiang YJ, et al. (2014) Is surveillance imaging effective for detecting surgically treatable recurrences in patients with melanoma? A comparative analysis of stage-specific surveillance strategies. Ann Surg 259:1215–1222. [DOI] [PubMed] [Google Scholar]
- 102.Rueth NM, Groth SS, Tuttle TM, et al. (2010) Conditional survival after surgical treatment of melanoma: an analysis of the Surveillance, Epidemiology, and End Results database. Ann Surg Oncol 17:1662–1668. [DOI] [PubMed] [Google Scholar]
- 103.Park TS, Phan GQ, Yang JC, et al. (2017) Routine Computer Tomography Imaging for the Detection of Recurrences in High-Risk Melanoma Patients. Ann Surg Oncol 24:947–951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Baker JJ, Meyers MO, Frank J, et al. (2014) Routine restaging PET/CT and detection of initial recurrence in sentinel lymph node positive stage III melanoma. Am J Surg 207:549–554. [DOI] [PubMed] [Google Scholar]