Abstract
Rapid anthropogenic alterations caused by urbanization are increasing temperatures in urban cores, a phenomenon known as the urban heat island (UHI) effect. Two cicada species, Cryptotympana atrata and Hyalessa fuscata (Hemiptera: Cicadidae), are abundant in metropolitan Seoul where their population densities correlate strongly with UHI intensities. Such a positive correlation between cicada density and UHI intensity may be possible if cicada abundance is linked to a certain thermal tolerance. We tested this hypothesis by investigating variation in morphology and thermal responses of two cicada species along a thermal gradient in Seoul and surrounding areas. The morphological responses were measured by metrics such as length, thorax width and depth, and mass. The thermal responses were measured in terms of minimum flight temperature, maximum voluntary temperature and heat torpor temperature. First, we observed a species-specific variation in thermal responses, in which C. atrata displayed a higher thermal threshold for maximum voluntary and heat torpor temperatures than H. fuscata. Second, a positive association between temperature conditions and body sizes were displayed in females H. fuscata, but not in either conspecific males or C. atrata individuals. Third, C. atrata exhibited similar thermal responses regardless of habitat temperature, while H. fuscata in warmer areas showed an increase in heat tolerance. In addition, H. fuscata individuals with bigger thorax sizes were more heat-tolerant than those with smaller thoraxes. Overall, our research is the first to detect a variation in thermal responses and body size of H. fuscata individuals at a local scale. More investigations would be needed to better understand the adaptation mechanisms of insects linked to UHI effects.
Subject terms: Behavioural ecology, Ecophysiology
Introduction
Temperature is one of the most important abiotic factors affecting daily activities and the life history of cicadas1–6. As temperature directly dictates chemical rates and metabolic processes in vivo, a habitat with fluctuating temperatures could drive insects to manage the high energy costs of maintaining body temperature, Tb, within a certain range to coordinate reproductive activity4. The surrounding conditions may also have an effect on reproductive success and mortality7–10. If so, cicadas should select favorable thermal conditions to obtain optimal body temperatures via behavioral- or physiological mechanisms4,11.
The urban heat island (UHI) effect, in which increased temperatures are associated with urban areas, is a ubiquitous consequence of microclimatic perturbation12 due to human activities and energy consumption, along with modification of landscapes and urban geometry13–17. Elevated temperatures in urban environments are regarded as detrimental as they reduce plant photosynthesis capability18 and biological diversity19,20. Nevertheless, for urban herbivorous insects such as cicadas, such warm habitats may prove advantageous. First, post-morphogenesis development of cicada eggs require a large degree of thermal accumulation2. Second, growth rates of cicada nymphs demonstrate a positive association with climate: those inhabiting warmer regions are more likely to grow faster than counterparts in cooler regions21. This may facilitate the development of larger body size in cicadas in the former group22, which in turn directly promotes greater fecundity of females23 and indirectly contributes to higher mating success in males24. Third, the emergence of final instars is stimulated by soil temperature, with warmer conditions triggering an earlier phenology of those instars25. Such earlier eclosion of male over female cicadas6 works advantageously for males as it maximizes mating success in those multiple copulating individuals26.
Recent studies have shown that some urban insect species adapt rapidly to warm city cores by increasing thermal tolerance. Herbivorous insects with high thermal tolerance capacity are better able to adapt to urban environments27. Specific physiological tolerances and thermoregulating behaviors of insects vary with their physical habitat conditions28. Accumulating evidence of an ability to track localized thermal profile clines in cities suggests that urban insects can evolve adaptive traits in response to such rapid environmental changes and become overrepresented in cities29,30. In several forest ant species in North America, higher critical thermal maxima correlate positively with population abundances in warm urban plots31,32. Additionally, urban ants possess higher heat tolerance than do rural ants33,34, which indicates adaptive plasticity or local thermal adaptation to microclimatic change in urban organisms.
Two cicada species, Cryptotympana atrata and Hyalessa fuscata (Hemiptera: Cicadidae), are widely distributed on the Korean Peninsula. These cicadas emerge annually from June to early October for key life history events, such as mate attraction, pair formation, and oviposition. The distribution of C. atrata ranges from East Asia to the northern part of Indo-China, whereas that of H. fuscata overlaps East Asia and encompasses the Far East of Russia35. An enumeration survey of cicada exuviae depicts more prevalent population densities in urban and suburban habitats relative to countryside habitats36. In particular, they constitute a major portion of cicada species in metropolitan Seoul, where their population densities correlate highly with UHI intensities37. C. facialis, a closely related species to C. atrata, has shown rapid population expansion in urbanized areas in Japan, owing to superior thermal adaptation to urban conditions2. Provided that the prevalence of C. atrata and H. fuscata populations in urban area is due to greater thermal tolerance to urban conditions, their thermal responses can be expected to be associated with the localized clines of habitat temperatures intensities.
Our aim was to investigate variations in morphology and thermal responses of two cicada species, C. atrata and H. fuscata, in accordance with the temperature of their habitats. We collected cicadas along a thermal gradient and assessed their morphometry and thermal responses. Given that a positive correlation already exists between cicada population densities and ambient temperatures, we predicted positive associations between UHI intensities and (1) morphological characteristics and (2) thermal responses of those cicadas.
Methodology
Sample collection
We sampled populations of C. atrata and H. fuscata from July 15 to August 5, 2016, in metropolitan Seoul and the vicinity in the Republic of Korea. Metropolitan Seoul covers more than 600 km2 with diverse landscape features, generating a mosaic of heterogeneous UHI intensities for cicadas. Approximately 10 million people live within city limits, with another 10 million in the surrounding suburban areas.
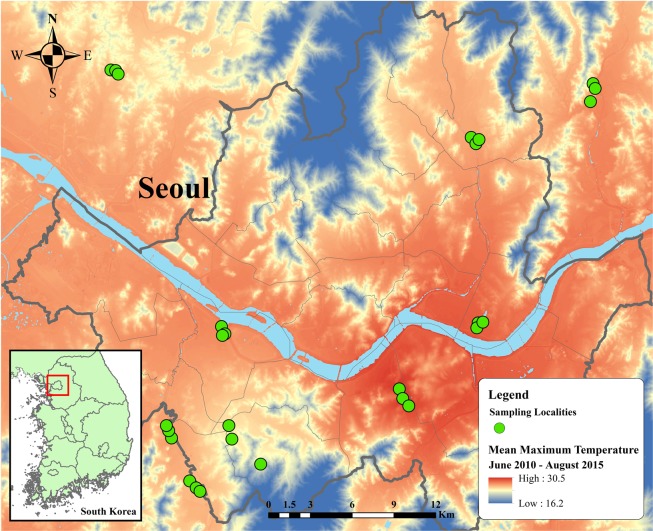
The sampling method and selection of study areas followed 12 sampling areas by Nguyen, et al.37. From there, three areas were excluded due to low sampling densities and difficulty in collecting cicadas. In order to verify our sampling design and its suitability to our research question, we compared those nine areas in terms of the abiotic factors related to urbanization such as greenness, wetness, imperviousness and isothermal. Greenness is a measure of photosynthetically active vegetation, wetness represents soil and vegetation moisture38, and imperviousness measures building footprints, pavements and asphalt. Isothermal is mean diurnal range divided by temperature annual range, extracted from worldclim 1.439. A one-way analysis of variance in which greenness, wetness, imperviousness, and isothermal were response variables was conducted. As expected, isothermal was the only variable that was significantly different among the nine areas (Supplementary Material 1). We concluded that temperature was the critical factor that differentiated among those nine area, thus verifying our sampling scheme (Fig. 1).
Figure 1.
Distribution of sampling areas within and surrounding metropolitan Seoul. Thermal map of average maximum temperature from June 1, 2010, to August 31, 2015, in metropolitan Seoul constructed by ArcMap 10.5. Temperature ranges from 16.2 to 30.5 °C, indicated by dark blue to red regions. Green circles designate sampling localities. Inset map in the left bottom corner illustrates the regional location of metropolitan Seoul in the Republic of Korea.
Weather data were compiled from Korea Meteorological Administration records. We calculated average maximum ambient temperature (TaMax) of each area during summer periods from June 1, 2010, to August 31, 2015. Given that mean, minimum, and maximum ambient temperatures are employed in UHI studies40, we relied on maximum ambient temperature, as it better represents the actual high ambient temperature that the cicadas encounter in their environment than the mean or minimum ambient temperature. Nine areas were randomly sampled and each area was sampled twice. Cicadas were collected from 8:00 a.m. to 12:00 p.m. at residential complexes in each area, and were subjected to thermal-response experiments within the day of capture. Information regarding sampling areas and the number of each species collected at each area are provided in Supplementary Material 2.
Measurement of thermal responses
Minimum flight temperature (MFT), maximum voluntary temperature (MVT), and heat torpor temperature (HTT) are conservative measures of thermal adaptation of cicadas to a habitat4,41,42 (see43 for a summary of thermal responses in 118 taxa of North American cicadas). MFT represents the lowest body temperature with fully coordinated activity. MVT is the upper thermoregulatory point at which thermoregulation takes precedence over other behaviors. HTT is the upper limit beyond which cicadas sink into a state of torpor. MVT increases as a habitat becomes warmer in some cases, whereas HTT is strictly related to the thermal condition of a habitat4,41–44. The MFT-to-HTT range indicates the fully active thermal breadth (TbRange) of each species in relation to a certain habitat condition.
Thermal responses of each individual were assessed in a single assay with no rest time between treatments under laboratory conditions. First, each cicada was cooled to a torpid state by keeping it at −20 °C. We checked each individual’s Tb every three minutes to ensure they did not freeze, as this might affect HTT42. As soon as the individual was torpid, we assessed MFT by dropping the insect from a height of 2.5 m. If the insect could not perform the expected behavior, it was allowed to gradually warm up at ambient temperature for one minute before being reexamined. After that, the insect was placed under a heat lamp to obtain MVT. The Tb at which the individual moved away from the heat source and started to seek shade was determined to be its MVT. The cicada was continuously heated under the heat lamp until no movement was observed, at which point its Tb indicated HTT. The procedure was not lethal, as individuals could recover to normal active conditions after several minutes. We acknowledged that this assay may have induced stress to some extent in cicadas. However, such measurement of thermal responses have been performed over several decades43, and further investigation is necessary to assess tentative influences of this assay on the thermal performance of cicadas.
The evaluation of Tb in cicadas is commonly conducted inside the mesothorax43. Here, we chose to assess Tb from both the mesothorax and the pronotum. Indeed, pronotom may represent an evaporative cooling site45. Evaporative cooling is a key physiological thermoregulation mechanism in cicadas, as it provides a major cooling effect by dissipating excess heat45–47 and facilitates the cicadas’ endurance of high ambient temperatures4. The temperature of the pronotum therefore may signal a critical thermal threshold for the individual to regulate Tb within its functionally active range and adopt necessary thermoregulation strategies to prevent excessive increase in Tb.
All temperature measurements were performed using a digital thermometer with a k-type thermocouple (Omega; model #: 450–AKT; Norwalk, Connecticut, USA) sensitive to ± 0.25 °C. The total live body mass of each individual was determined using an Adventurer Pro Analytical (Ohaus; New York, USA) scale sensitive to ± 0.0001 g. We also measured body length, mesothorax width, and mesothorax depth using Digital Calipers (Insize Co., Ltd.; Georgia, USA) sensitive to ± 0.02 mm.
Statistical analysis
Comparative thermal responses of C. atratra and H. fuscata
A first constrained multivariate analysis, i.e., redundancy analysis48 (RDA), was performed to compare thermal responses of MFT, MVT, HTT and TbRange between C. atrata and H. fuscata. Analyses were conducted separately for temperature measurements from the pronotum and mesonotum. C. atrata and H. fuscata responded differently to heat experiments. Therefore, we conducted hereafter analyses separately for each species.
Variation in morphological characters of each cicada species
First, we applied an RDA to evaluate the responses of the morphological characteristics measured by total mass (mass), body length (length), mesothorax width (width), and mesothorax depth (depth) to sex and TaMax. Second, intersexual morphological differences were assessed by performing t-tests for normally distributed data of mass and Kruskal-Wallis tests for non-normally distributed data of length, width and depth. Furthermore, we also conducted linear regressions to examine the effect of temperature on each of the morphological characteristics, separating analysis for males and females. We examined the assumption of homogeneity of variance of residuals of each linear regression model via a diagnostic plot of predicted values versus standardized residuals.
Variation in thermal responses of each cicada species
A third RDA was used to assess the thermal responses of MFT, MVT, HTT, and TbRange by sex, TaMax and width. We performed independent RDAs for temperature measurement from the pronotum and mesonotum. Again, we compared intersexual differences in terms of thermal responses, employing t-tests for normally distributed data (MVT, HTT and TB) and Kruskal-Wallis tests for non-normally distributed data (MFT). Linear regressions tested how both male and female thermal responses, measured from pronotum and mesonotum, were influenced by TaMax and width. Diagnostic plots of predicted values versus standardized residuals were visualized to assess the assumption of homoscedasticity of residuals for each linear regression model. RDA results showed no significant thermal responses of C. atrata in the measurement from the pronotum or mesonotum, and it was exempted from follow-up intersexual variation tests.
Intersexual variation in thermal responses of H. fuscata
Finally, an RDA was performed to quantify intersexual differences in thermal responses according to TaMax and width. The Vegan package49 on R Studio (Version 1.0.143) was used for all multivariate analyses, and the statistical significance of the entire model for each variable (marginal test) was evaluated using Monte-Carlo permutation tests (n = 999). Linear regressions were performed with SPSS 22 (IBM Corp.; New York, USA). Results are presented as the mean ± standard deviation.
Ethics declaration
Cicadas are common species in Republic of Korea. Neither C. atrata nor H. fuscata was listed as protected or endangered species in the “List of wildlife species prohibited for collection” issued by the Ministry of Environment, Republic of Korea and in the IUCN Red List. Therefore, no field permit was required for this study.
Results
Comparative thermal responses of C. atratra and H. fuscata
Thermal responses measured at the pronotum (n = 158) showed that C. atrata became fully coordinated at an MFT of 24.81 ± 1.72 °C, reaching the thermoregulation level at an MVT of 37.58 ± 2.01 °C, and becoming heat torpid at an HTT of 46.92 ± 2.71 °C. Body temperatures measured inside the mesonotum (n = 114) indicated that MFT was achieved at 23.88 ± 1.80 °C, MVT at 38.13 ± 1.99 °C, and HTT at 49.56 ± 1.87 °C.
H. fuscata thermal responses assessed from the pronotum (n = 258) showed that the species had an MFT at 25.66 ± 2.06 °C, elevated its thermoregulation level to an MVT of 36.10 ± 1.46 °C, and became heat torpid at an HTT of 46.41 ± 2.58 °C. The responses to heat determined at the mesonotum (n = 197) exhibited MFT at 24.80 ± 1.89 °C, MVT at 36.39 ± 1.70 °C, and HTT at 48.56 ± 2.09 °C.
In general, C. atrata tolerated heat better than H. fuscata in terms of MVT and HTT. The RDA models explained 11.77% and 13.17% of the total variation in temperature measurement from the pronotum and the mesonotum, respectively (Table 1). Both showed that thermal responses were significantly different between C. atrata and H. fuscata. Indeed, for both the pronotum and mesonotum temperature measurements, the species factor was the most significant (p = 0.001), representing the first axis for both ordination diagrams (Fig. 2A,B) and accounting for 80.6% and 76.33% of the inertia, respectively. Among four thermal responses, only MFT was greater for H. fuscata, whereas the others tended to be higher for C. atrata (Fig. 2A,B).
Table 1.
Results of the RDA for comparative thermal responses measured from the pronotum and mesonotum of two cicada species. Percentage of inertia and p-values were calculated for each variable. The statistical significance of the entire model for each variable (marginal tests) was evaluated using Monte-Carlo permutation tests (n = 999).
| Pronotum | Mesonotum | |||
|---|---|---|---|---|
| % inertia | p-value | % inertia | p-value | |
| Full model | 0.001 | 0.001 | ||
| Constrained | 11.77 | 13.17 | ||
| Unconstrained | 88.23 | 86.83 | ||
| Variables effects | ||||
| TaMax | 0.001 | 0.005 | ||
| Sex | 0.252 | 0.002 | ||
| Species | 0.001 | 0.001 | ||
Figure 2.
Ordination diagram of the two first axes of the RDA for comparative thermal responses for C. atrata and H. fuscata for the (A) pronotom and (B) mesonotum. MFT: minimum flighte temperature; MVT: maximum voluntary temperature; HTT: heat torpor temperature; TbRange: thermal breadth. This figure was generated using Vegan package on R Studio version 1.0.143.
Variation in morphological characters of each cicada species
Morphological measurements of 158 C. atrata individuals produced a total mass of 2.90 ± 0.47 g, a body length of 41.63 ± 1.62 cm, a mesothorax depth of 14.61 ± 0.63 cm, and a mesothorax width of 16.59 ± 0.75 cm. The result of the RDA showed that the effect of TaMax and sex did not significantly explain the morphological measurement matrix (Table 2).
Table 2.
Results of the RDA conducted on morphological characteristics for C. atrata and H. fuscata. Percentage of inertia and p-values were calculated for each variable. The statistical significance of the entire model for each variable (marginal tests) was evaluated using Monte-Carlo permutation tests (n = 999). ns: not significant.
| C. atrata | H. fuscata | |||
|---|---|---|---|---|
| % inertia | p-value | % inertia | p-value | |
| Full model | ns | 0.001 | ||
| Constrained | 11.5 | |||
| Unconstrained | 88.5 | |||
| Variables effects | ||||
| TaMax | ns | 0.002 | ||
| Sex | ns | 0.001 | ||
H. fuscata (n = 258) was characterized by a total live mass of 1.60 ± 0.25 g, with a body length of 32.54 ± 1.80 cm, a mesothorax depth of 12.06 ± 1.05 cm, and a mesothorax width of 13.84 ± 1.24 cm. The RDA model explained 11.5% of the recorded variation, in which sex was the principal factor (p = 0.001), represented along the first axis and accounting for 88.72% of the variance (Table 2, Fig. 3). TaMax was also significant (p = 0.002) and was represented along the second axis, accounting for 11.28% of the variance. Females were significantly heavier than males, but shorter in total body length (Supplementary Material 3). H. fuscata individuals exhibited a decrease in mass (Fig. 4A) but an increase in mesothorax size relative to TaMax (Fig. 4B,C). Linear regressions showed that TaMax influenced male and female morphological characteristics differently (Table 3); TaMax was significant for only the mass of males but not for other characteristics; on the contrary, TaMax was significant for almost all characteristics of females, except length.
Figure 3.

Ordination diagram of the two first axes of the RDA for morphologic characteristics of H. fuscata. The position of the variables are represented with convex hull based on sex. The part of the inertia explained by each axis is given. This figure was generated using Vegan package on R Studio version 1.0.143.
Figure 4.
Variation in morphological characters of H. fuscata in response to TaMax divided by sex. Correlation between (A) Mass and TaMax, (B) Depth and TaMax, and (C) Width and TaMax. Asterisks denote significant linear regressions. This figure was generated using SPSS 22.
Table 3.
Linear regression analysis to assess the effect of TaMax on morphological characters of H. fuscata males and females.
| Dependent variable | Independent variable | B | SE | t | P |
|---|---|---|---|---|---|
| Male | |||||
| Mass | Intercept | 3.34 | 0.72 | 4.58 | <0.001 |
| TaMax | −0.61 | 0.02 | −2.49 | 0.014 | |
| Length | Intercept | 29.18 | 6.27 | 4.65 | <0.001 |
| TaMax | 0.14 | 0.21 | 0.65 | 0.514 | |
| Depth | Intercept | 8.95 | 4.06 | 2.21 | 0.029 |
| TaMax | 0.11 | 0.14 | 0.80 | 0.427 | |
| Width | Intercept | 8.05 | 4.77 | 1.69 | 0.094 |
| TaMax | 0.20 | 0.16 | 1.24 | 0.216 | |
| Female | |||||
| Mass | Intercept | 4.39 | 1.07 | 4.12 | <0.001 |
| TaMax | −0.09 | 0.04 | −2.54 | 0.012 | |
| Length | Intercept | 24.02 | 6.84 | 3.51 | 0.001 |
| TaMax | 0.26 | 0.23 | 1.14 | 0.258 | |
| Depth | Intercept | −4.35 | 4.05 | −1.07 | 0.286 |
| TaMax | 0.55 | 0.14 | 4.02 | <0.001 | |
| Width | Intercept | −4.53 | 4.86 | −0.93 | 0.354 |
| TaMax | 0.61 | 0.16 | 3.75 | <0.001 | |
Variation in thermal responses of each cicada species
Although C. atrata individuals exhibited some changes in their thermal responses, the species responded similarly to heat regardless of habitat conditions. The results of the RDA showed that both sex and TaMax had no significant effect on thermal responses obtained from the pronotum (p > 0.05, Table 4). For the mesonotum, sex was the only significant factor explaining the thermal responses (p = 0.029).
Table 4.
Results of the RDA conducted on thermal responses for C. atrata and H. fuscata. Percentage of inertia and p-values were calculated for each variable. The statistical significance of the entire model for each variable (marginal tests) was evaluated using Monte-Carlo permutation tests (n = 999). ns: not significant.
| C. atrata – Pronotum | C. atrata – Mesonotum | H. fuscata – Pronotum | H. fuscata – Mesonotum | |||||
|---|---|---|---|---|---|---|---|---|
| % inertia | p-value | % inertia | p-value | % inertia | p-value | % inertia | p-value | |
| Full model | 0.786 | 0.057 | 0.001 | 0.001 | ||||
| Constrained | ns | 6.01 | 11.76 | 11.25 | ||||
| Unconstrained | ns | 95 | 88.24 | 88.75 | ||||
| Variables effects | ||||||||
| TaMax | ns | 0.269 | 0.001 | 0.04 | ||||
| Sex | ns | 0.029 | 0.011 | 0.001 | ||||
| Width | ns | 0.093 | 0.011 | 0.001 | ||||
The RDA model showed that thermal responses from the pronotum were mostly influenced by TaMax (p = 0.001), followed by sex (p = 0.011) and width (p = 0.011) (Table 4). The model explained 11.76% of the total inertia, of which 87.32% was explained by axis 1 and 12.57% by axis 2 (Fig. 5A). Regarding the mesonotum, the thermal responses were mostly significantly influenced by sex (p = 0.001) and width (p = 0.001), followed by TaMax (p = 0.04) (Table 4, Fig. 5B). The thermal responses measured at the mesonotum were driven primarily by width rather than other factors (Supplementary Material 4).
Figure 5.
Ordination diagram of the two first axes of the RDA performed on MFT, MVT, HTT and TbRange measured at the (A) pronotum and (B) mesonotum separately for H. fuscata. The positions of the variables are represented with convex hulls based on sex. The part of the inertia explained by each axis is given. This figure was generated using Vegan package on R Studio version 1.0.143.
Intersexual variation in thermal responses of H. fuscata
For measurement from the pronotum, males and females had similar MFT and MVT. However, females tolerated heat significantly better (Fig. 6A) and had wider thermal ranges than males (Fig. 6B). Both sexes significantly increased their HTT and TbRange as TaMax increased (Supplementary Material 5), but no relationships were found between TaMax with either MFT or MVT (Table 5). Width displayed a significant negative effect on MFT of both sexes (Supplementary Material 6), while this factor was significantly positively correlated with HTT and TbRange of both males and females (Fig. 7).
Figure 6.
Intersexual variation in thermal responses of H. fuscata obtained from the pronotum according to TaMax. Correlation between (A) Heat torpor temperature and TaMax, (B) Thermal breadth and TaMax. Asterisks denote significant linear regressions. This figure was generated using SPSS 22.
Table 5.
Linear regression analysis to assess the effect of TaMax and Width on thermal responses of males and females H. fuscata obtained from the pronotum.
| Dependent variable | Independent variable | B | SE | t | P |
|---|---|---|---|---|---|
| Male | |||||
| MFT | Intercept | 40.45 | 8.25 | 4.90 | <0.001 |
| TaMax | −0.31 | 0.28 | −1.10 | 0.272 | |
| Width | −0.39 | 0.15 | −2.59 | 0.011 | |
| MVT | Intercept | 31.56 | 5.78 | 5.46 | <0.001 |
| TaMax | 0.20 | 0.19 | 1.02 | 0.310 | |
| Width | −0.1 | 0.11 | −0.94 | 0.351 | |
| HTT | Intercept | 4.02 | 10.11 | 0.40 | 0.691 |
| TaMax | 1.64 | 0.34 | 4.83 | <0.001 | |
| Width | −0.47 | 0.18 | −2.54 | 0.012 | |
| TB | Intercept | −36.42 | 13.24 | −2.75 | 0.007 |
| TaMax | 1.94 | 0.44 | 4.38 | <0.001 | |
| Width | −0.08 | 0.24 | −0.33 | 0.743 | |
| Female | |||||
| MFT | Intercept | 38.17 | 7.81 | 4.89 | <0.001 |
| TaMax | −0.27 | 0.28 | −1.0 | 0.320 | |
| Width | −0.33 | 0.14 | −2.35 | 0.020 | |
| MVT | Intercept | 28.98 | 5.94 | 4.88 | <0.001 |
| TaMax | 0.29 | 0.21 | 1.38 | 0.169 | |
| Width | −0.11 | 0.11 | −0.99 | 0.323 | |
| HTT | Intercept | 0.49 | 8.52 | 0.06 | 0.954 |
| TaMax | 1.45 | 0.30 | 4.83 | <0.001 | |
| Width | 0.22 | 0.16 | 1.43 | 0.154 | |
| TB | Intercept | −37.68 | 11.95 | −3.15 | 0.002 |
| TaMax | 1.73 | 0.42 | 4.10 | <0.001 | |
| Width | 0.56 | 0.22 | 2.55 | 0.012 | |
Figure 7.
Relationship between thermal responses of H. fuscata obtained from the mesonotum and width. Correlation between (A) Heat torpor temperature and width, (B) Thermal breadth and width. Asterisks denote significant linear regressions. This figure was generated using SPSS 22.
Discussion
In sum, our analyses suggest a local adaptation of thermal responses and thorax sizes in H. fuscata populations distributed along a thermal gradient in metropolitan Seoul, but not for the other cicada species. Although the results of our redundancy analysis showed high values of unconstrained variance (>85% in all of them), significant differences in thermal responses between two cicadas and within H. fuscata were determined. Specifically, thermal responses of C. atrata measured by MFT, MVT, HTT and TbRange indicated a better tolerance to heat stimuli than H. fuscata. Furthermore, no relationship was observed between the ambient temperature of the habitat and either morphology or thermal physiology of C. atrata. On the contrary, H. fuscata from habitats with higher ambient temperature had substantially enlarged thoraxes, endured heat better, and held wider fully active thermal ranges.
Although ambient temperature was significant for the pronotum’s HTTs and TBs of both sexes (Table 5), it was marginally significant for the mesonotum’s TB of males and HTT of females (Supplementary Material 3). To justify the effect of ambient temperature on thermal responses of H. fuscata, we conducted the RDA and linear regression again, using Mass instead of Width as a covariate (results not shown). RDA result shows that TaMax was significant for thermal responses of H. fuscata regardless of pronotum or mesonotum temperature. Furthermore, the result of linear regression analysis displays a consistent result between pronotum and mesonotum temperatures. We therefore conclude that overall H. fuscata increased their thermal tolerance in accordance to the increase in ambient temperature.
The increase in heat tolerance of H. fuscata resembles other research on urban-adapted insects, which indicates a close association between thermal tolerance and localized thermal clines29,30,33. Research on thermal responses of cicadas across a wide geographic range provides evidence that cicadas are more tolerant of warmer environments41–43. Cicadas of 38 species inhabiting Mediterranean habitats display an elevated HTT in accordance with the local thermal characteristics, regardless of taxonomic position or the diversity of particular plant species50. Our results further imply a localized thermal acclimatization of H. fuscata.
Here, we observed interspecific differences in thermal responses between C. atrata and H. fuscata, in which the warmer the habitat, the greater thermal responses C. atrata exhibited compared to the other species. This disparity can be explained by variation in species geographical origins: C. atrata originated in subtropical regions, whereas H. fuscata originated in tundra regions35. Additionally, segregation in microhabitat niches may contribute to how each species utilizes its habitat for thermoregulation. C. atrata perches mainly on top of the canopy, where it is exposed to solar radiation, whereas H. fuscata is found mainly on tree trunks in shaded environments51. As a result, adaptation to individual thermal regimes has led to variation in thermal responses between these species, a pattern that is well-discerned in other cicadas inhabiting tropical habitats52.
In line with other studies on thermal responses of cicadas, we found HTT depends strongly on the maximum environmental thermal regime43,44,53. This positive relationship was observed in H. fuscata, but not in C. atrata, regardless of habitat conditions. Such difference may be partially due to the origins of the two species. C. atrata is, therefore, more prone to experiencing higher thermal regimes and is adapted to high thermal conditions in metropolitan Seoul, thus exhibiting no difference in thermal tolerance across heterogeneous ambient temperatures. Environmental constraints applied to populations of the acorn ant, Temnothorax curvispinosus, are greater in lower latitudes, causing a reduction in evolutionary thermal responses relative to populations at higher latitudes29.
In contrast to C. atrata, distributions of H. fuscata at higher latitudes expose this species to colder environments, and the warmer conditions of metropolitan Seoul may induce a thermal acclimatization to warmer temperatures. Not only does urban warming seem to select for thermophilic species, but it also extends thermal tolerance ranges by elevating heat tolerance30. Here, as a function of ambient temperature, the thermal range of H. fuscata was extended toward warmer habitats. Better heat tolerance and wider thermal active ranges promote colonization of microhabitat niches generated by urbanization.
Besides thermal responses, our study suggest contrast relationships between thermal conditions and morphological characters of H. fuscata females, while thorax sizes increased as ambient temperature increased, total mass decreased. According to Bergman size clines, warmer environments usually trigger the growth rate of ectotherms. As a results, those from warmer environments tend to be bigger than the ones from colder environments54. The decrease of thorax sizes of females H. fuscata from warm to cooler habitats in this study shows support to Bergmann size clines. Besides, females cicadas developed at cooler environments were heavier than those at warmer environments. The flies examined by Crill et al.55 also exhibited similar changes in body dry mass to our study. Accordingly, only female flies developed at higher temperature were heavier in terms of dry mass whereas male flies were insensitive to their developmental temperature. However, the underlying mechanism is unclear. In our study, we observe such an inverse effect of temperature on thorax size and mass of females H. fuscata. The evolutionary explanation for this phenomenon remains elusive, thus we aim to elucidate it in further study. Besides, more investigations are needed to better understand this phenomenon and to clarify if these first results are linked to an exceptional population of small individuals in the coolest areas or if this could be a general trend.
Insect metabolic activity is accelerated under exposure to higher temperatures, which triggers an increase in body size56. Furthermore, warming climates have been proposed as causes of escalating growth rates of cicada nymphs underground, resulting in larger body sizes within a fixed development period22. Females of the scale insect Melanasimpis tenebricosa inhabiting warmer tree canopies exhibit larger body size than those living in cooler tree canopies57. An increase of 2.5 °C in rearing temperature promotes the development of larger females in the southern green stink bug, Nezara viridula58. There is also evidence that larger body size may be driven by local adaptation to warmer habitats. In periodical cicadas, for instance, the more southerly diverged M. tredecim experiences higher temperatures as adults possess larger bodies22. Larger bodies provide multiple ecological and evolutionary advantages by contributing positively to survival, fecundity, and mating success59. As a result, a larger size could result from selection for greater fecundity in female cicadas23.
The relationship between body size and thermal tolerance has been explored in various terrestrial insects. Although there are cases where superior heat tolerance is found in smaller60,61 or intermediate-sized insects62, larger species tend to tolerate higher temperatures better than smaller species do57,58,62,63. In our study, larger H. fuscata individuals had higher heat tolerance. Consequently, better thermal tolerance capacity is suggested to provide higher fitness to female cicadas3. Although body size of male H. fuscata was not correlated with ambient temperature, it was found to be associated with acoustic properties of cicada songs64–66, which act as premating signals to attract conspecific mates. Furthermore, songs of warmer males are higher in intensity and are able to travel further in the air67, increasing the transmission of their mating signals to prospective mates.
Conclusion
Our thermal tolerance experiments indicate a local adaptation of thermal responses and thorax sizes along a thermal gradient of H. fuscata in metropolitan Seoul. Whether such variation in heat tolerance is caused by phenotypic plasticity or evolutionary adaptation to environmental conditions is unclear. However, acclimatization to anthropogenic perturbation as a consequence of urbanization may be partially responsible. This is the first study to notice variation in thermal tolerance of cicadas at the local urban scale. Our research highlights the importance of taking localized thermal regimes into consideration when examining species-specific responses to escalating urban warming caused by urbanization.
Supplementary information
Acknowledgements
We thank Jeong Hyunsuu for her assistance in sample collection and the experiments, and Desiree Anderson for the heat map construction. This work was supported by the National Research Foundation of Korea (Grant Number: 2015R1A4A1041997). H.S. was funded by the Korea Research Fellowship Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (KRF project grant number: 2016H1D3A1938095).
Author contributions
H.Q.N. designed the study; H.Q.N., Y.B., S.J., S.C., I.Y.K. and J.H. collected samples and conducted experiments; H.Q.N. and H.S. performed statistical analyses and drafted the initial manuscript; H.Q.N., H.S. and Y.J. critically reviewed and revised the manuscript. All authors have seen and approved the final version of this manuscript.
Data availability
The datasets analysed during the current study are available in the Mendeley Data repository, DOI: http://dx.doi.org/10.17632/4z4n7p5gxt.2#file-ae6d6c15-bf0a-4fde-8f32-4f62ebf8f96c.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
is available for this paper at 10.1038/s41598-020-58276-0.
References
- 1.Fonseca P, Revez MA. Temperature dependence of cicada songs (Homoptera, Cicadoidea) Journal of comparative Physiology A. 2002;187:971–976. doi: 10.1007/s00359-001-0267-5. [DOI] [PubMed] [Google Scholar]
- 2.Moriyama M, Numata H. Diapause and prolonged development in the embryo and their ecological significance in two cicadas, Cryptotympana facialis and Graptopsaltria nigrofuscata. Journal of insect physiology. 2008;54:1487–1494. doi: 10.1016/j.jinsphys.2008.08.008. [DOI] [PubMed] [Google Scholar]
- 3.Toolson EC. Comparative thermal physiological ecology of syntopic populations of Cacama valvata and Tibicen bifidus (Homoptera: Cicadidae): modeling fitness consequences of temperature variation. American Zoologist. 1998;38:568–582. doi: 10.1093/icb/38.3.568. [DOI] [Google Scholar]
- 4.Sanborn, A. Cicada thermoregulation (Hemiptera, Cicadoidea). (na, 2002).
- 5.Sanborn AF, Maté S. Thermoregulation and the effect of body temperature on call temporal parameters in the cicada Diceroprocta olympusa (Homoptera: Cicadidae) Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology. 2000;125:141–148. doi: 10.1016/S1095-6433(99)00169-5. [DOI] [PubMed] [Google Scholar]
- 6.Sato Y, Sato S. Spring temperature predicts the long-term molting phenology of two cicadas, Cryptotympana facialis and Graptopsaltria nigrofuscata (Hemiptera: Cicadidae) Annals of the Entomological Society of America. 2015;108:494–500. doi: 10.1093/aesa/sav036. [DOI] [Google Scholar]
- 7.Battisti A, et al. Expansion of geographic range in the pine processionary moth caused by increased winter temperatures. Ecological applications. 2005;15:2084–2096. doi: 10.1890/04-1903. [DOI] [Google Scholar]
- 8.Dale AG, Frank SD. The effects of urban warming on herbivore abundance and street tree condition. PloS one. 2014;9:e102996. doi: 10.1371/journal.pone.0102996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Laws AN, Belovsky GE. How will species respond to climate change? Examining the effects of temperature and population density on an herbivorous insect. Environmental Entomology. 2010;39:312–319. doi: 10.1603/EN09294. [DOI] [PubMed] [Google Scholar]
- 10.Rueda L, Patel K, Axtell R, Stinner R. Temperature-dependent development and survival rates of Culex quinquefasciatus and Aedes aegypti (Diptera: Culicidae) Journal of medical entomology. 1990;27:892–898. doi: 10.1093/jmedent/27.5.892. [DOI] [PubMed] [Google Scholar]
- 11.Heath James Edward. Temperature Responses of the Periodical "17-year" Cicada, Magicicada cassini (Homoptera, Cicadidae) American Midland Naturalist. 1967;77(1):64. doi: 10.2307/2423426. [DOI] [Google Scholar]
- 12.Oke TR. City size and the urban heat island. Atmospheric Environment (1967) 1973;7:769–779. doi: 10.1016/0004-6981(73)90140-6. [DOI] [Google Scholar]
- 13.Nowak, D. J. & Dwyer, J. F. In Handbook of urban and community forestry in the Northeast 11–25 (Springer, 2000).
- 14.Oke, T. In Wind climate in cities (eds. Cermark, J. E., Davenport, A. G., Plate, E. J. & Viegas, D. X.) 81–107 (Springer, 1995).
- 15.Hart MA, Sailor DJ. Quantifying the influence of land-use and surface characteristics on spatial variability in the urban heat island. Theoretical and applied climatology. 2009;95:397–406. doi: 10.1007/s00704-008-0017-5. [DOI] [Google Scholar]
- 16.Santamouris M. Heat island research in Europe: the state of the art. Advances in building energy research. 2007;1:123–150. doi: 10.1080/17512549.2007.9687272. [DOI] [Google Scholar]
- 17.Ryu Y-H, Baik J-J. Quantitative analysis of factors contributing to urban heat island intensity. Journal of Applied Meteorology and Climatology. 2012;51:842–854. doi: 10.1175/JAMC-D-11-098.1. [DOI] [Google Scholar]
- 18.Agrawal, M. In Urban Ecology 603–607 (Springer, 1998).
- 19.Stringer LD, Stephens AE, Suckling DM, Charles JG. Ant dominance in urban areas. Urban Ecosystems. 2009;12:503–514. doi: 10.1007/s11252-009-0100-4. [DOI] [Google Scholar]
- 20.Willigalla C, Fartmann T. Patterns in the diversity of dragonflies (Odonata) in cities across Central Europe. European Journal of Entomology. 2012;109:235–245. doi: 10.14411/eje.2012.031. [DOI] [Google Scholar]
- 21.White, J. A. & Lloyd, M. Growth rates of 17 and 13-year periodical cicadas. American Midland Naturalist, 127–143 (1975).
- 22.Koyama T, et al. Geographic body size variation in the periodical cicadas Magicicada: implications for life cycle divergence and local adaptation. Journal of evolutionary biology. 2015;28:1270–1277. doi: 10.1111/jeb.12653. [DOI] [PubMed] [Google Scholar]
- 23.Karban R. Evolution of prolonged development: a life table analysis for periodical cicadas. The American Naturalist. 1997;150:446–461. doi: 10.1086/286075. [DOI] [PubMed] [Google Scholar]
- 24.Karban Richard. Sexual Selection, Body Size and Sex-related Mortality in the Cicada Magicicada cassini. American Midland Naturalist. 1983;109(2):324. doi: 10.2307/2425413. [DOI] [Google Scholar]
- 25.Smith DM, Kelly JF, Finch DM. Cicada emergence in southwestern riparian forest: influences of wildfire and vegetation composition. Ecological Applications. 2006;16:1608–1618. doi: 10.1890/1051-0761(2006)016[1608:CEISRF]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- 26.Saisho Y. Mathematical observations on the relation between eclosion periods and the copulation rate of cicadas. Mathematical biosciences and engineering: MBE. 2010;7:443–453. doi: 10.3934/mbe.2010.7.443. [DOI] [PubMed] [Google Scholar]
- 27.Hamblin AL, Youngsteadt E, López-Uribe MM, Frank SD. Physiological thermal limits predict differential responses of bees to urban heat-island effects. Biology letters. 2017;13:20170125. doi: 10.1098/rsbl.2017.0125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Schowalter, T. D. Insect ecology: an ecosystem approach. (Academic Press, 2016).
- 29.Diamond SE, Chick LD, Perez A, Strickler SA, Martin RA. Evolution of thermal tolerance and its fitness consequences: parallel and non-parallel responses to urban heat islands across three cities. Proc. R. Soc. B. 2018;285:20180036. doi: 10.1098/rspb.2018.0036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Piano Elena, De Wolf Katrien, Bona Francesca, Bonte Dries, Bowler Diana E., Isaia Marco, Lens Luc, Merckx Thomas, Mertens Daan, van Kerckvoorde Marc, De Meester Luc, Hendrickx Frederik. Urbanization drives community shifts towards thermophilic and dispersive species at local and landscape scales. Global Change Biology. 2017;23(7):2554–2564. doi: 10.1111/gcb.13606. [DOI] [PubMed] [Google Scholar]
- 31.Diamond SE, et al. A physiological trait-based approach to predicting the responses of species to experimental climate warming. Ecology. 2012;93:2305–2312. doi: 10.1890/11-2296.1. [DOI] [PubMed] [Google Scholar]
- 32.Stuble KL, et al. Foraging by forest ants under experimental climatic warming: a test at two sites. Ecology and evolution. 2013;3:482–491. doi: 10.1002/ece3.473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Angilletta MJ, et al. Urban physiology: city ants possess high heat tolerance. PLoS One. 2007;2:e258. doi: 10.1371/journal.pone.0000258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Diamond SE, Chick L, Perez A, Strickler SA, Martin RA. Rapid evolution of ant thermal tolerance across an urban-rural temperature cline. Biological Journal of the Linnean Society. 2017;121:248–257. doi: 10.1093/biolinnean/blw047. [DOI] [Google Scholar]
- 35.Lee YJ. Revised synonymic list of Cicadidae (Insecta: Hemiptera) from the Korean Peninsula, with the description of a new species and some taxonomic remarks. Proceedings of the Biological Society of Washington. 2008;121:445–467. doi: 10.2988/08-09.1. [DOI] [Google Scholar]
- 36.Kim TE, Oh S-Y, Chang E, Jang Y. Host availability hypothesis: complex interactions with abiotic factors and predators may best explain population densities of cicada species. Animal Cells and Systems. 2014;18:143–153. doi: 10.1080/19768354.2014.906501. [DOI] [Google Scholar]
- 37.Nguyen HQ, Andersen DK, Kim Y, Jang Y. Urban heat island effect on cicada densities in metropolitan Seoul. PeerJ. 2018;6:e4238. doi: 10.7717/peerj.4238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lobser S, Cohen W. MODIS tasselled cap: land cover characteristics expressed through transformed MODIS data. International Journal of Remote Sensing. 2007;28:5079–5101. doi: 10.1080/01431160701253303. [DOI] [Google Scholar]
- 39.Hijmans RJ, Cameron SE, Parra JL, Jones PG, Jarvis A. Very high resolution interpolated climate surfaces for global land areas. International Journal of Climatology: A Journal of the Royal Meteorological Society. 2005;25:1965–1978. doi: 10.1002/joc.1276. [DOI] [Google Scholar]
- 40.Tzavali A, Paravantis JP, Mihalakakou G, Fotiadi A, Stigka E. Urban heat island intensity: a literature review. Fresenius Environmental Bulletin. 2015;24:4535–4554. [Google Scholar]
- 41.Sanborn, A. F. & Phillips, P. K. Thermal responses of the Diceroprocta cinctifera species group (Homoptera: Cicadidae). The Southwestern Naturalist, 136–139 (1996).
- 42.Sanborn AF, Heath J. Thermal responses of periodical cicadas: within and between brood parity (Hemiptera: Cicadidae: Magicicada spp.) Open Access Insect Physiol. 2009;1:13–20. doi: 10.2147/OAIP.S7211. [DOI] [Google Scholar]
- 43.Sanborn AF, Heath JE, Heath MS, Phillips PK. Thermal adaptation in North American cicadas (Hemiptera: Cicadidae) Journal of thermal biology. 2017;69:v–xviii. doi: 10.1016/j.jtherbio.2017.07.011. [DOI] [PubMed] [Google Scholar]
- 44.Sanborn AF, Phillips PK, Heath JE, Heath MS. Influence of altitude, habitat and microhabitat on thermal adaptation of cicadas from Southwest Texas (Hemiptera: Cicadidae) Journal of Thermal Biology. 2011;36:386–389. doi: 10.1016/j.jtherbio.2011.07.001. [DOI] [Google Scholar]
- 45.Sanborn AF, Heath JE, Heath MS. Thermoregulation and evaporative cooling in the cicada Okanagodes gracilis (Homoptera: Cicadidae). Comparative biochemistry and physiology. Comparative physiology. 1992;102:751–757. doi: 10.1016/0300-9629(92)90736-a. [DOI] [PubMed] [Google Scholar]
- 46.Harrison, J. F., Woods, H. A. & Roberts, S. P. Ecological and environmental physiology of insects. Vol. 3 (Oxford University Press, 2012).
- 47.Toolson EC. Water profligacy as an adaptation to hot deserts: water loss rates and evaporative cooling in the Sonoran Desert cicada, Diceroprocta apache (Homoptera: Cicadidae) Physiological Zoology. 1987;60:379–385. doi: 10.1086/physzool.60.4.30157899. [DOI] [Google Scholar]
- 48.Rao, C. R. The use and interpretation of principal component analysis in applied research. Sankhyā: The Indian Journal of Statistics, Series A, 329–358 (1964).
- 49.Oksanen J. Multivariate analysis of ecological communities in R: vegan tutorial. R Doc. 2015;43:11–12. [Google Scholar]
- 50.Sanborn AF, Phillips PK, Heath JE, Heath MS. Comparative thermal adaptation in cicadas (Hemiptera: Cicadidae) inhabiting Mediterranean ecosystems. Journal of Thermal Biology. 2011;36:150–155. doi: 10.1016/j.jtherbio.2011.01.002. [DOI] [Google Scholar]
- 51.Lee Y. A list of Cicadidae (Homoptera) in Korea. Cicada. 1999;15:1–16. [Google Scholar]
- 52.Sanborn AF, Heath JE, Phillips PK, Heath MS, Noriega FG. Thermal adaptation and diversity in tropical ecosystems: evidence from cicadas (Hemiptera, Cicadidae) PLoS One. 2011;6:e29368. doi: 10.1371/journal.pone.0029368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sanborn A. Comparative thermoregulation of sympatric endothermic and ectothermic cicadas (Homoptera: Cicadidae: Tibicen winnemanna and Tibicen chloromerus) Journal of Comparative Physiology A. 2000;186:551–556. doi: 10.1007/s003590000110. [DOI] [PubMed] [Google Scholar]
- 54.Van Voorhies WA. On the adaptive nature of Bergmann size clines: a reply to Mousseau, Partridge and Coyne. Evolution. 1997;51:635–640. doi: 10.2307/2411140. [DOI] [PubMed] [Google Scholar]
- 55.Crill WD, Huey RB, Gilchrist GW. Within-and between‐generation effects of temperature on the morphology and physiology of Drosophila melanogaster. Evolution. 1996;50:1205–1218. doi: 10.1111/j.1558-5646.1996.tb02361.x. [DOI] [PubMed] [Google Scholar]
- 56.Ray C. The application of Bergmann’s and Allen’s rules to the poikilotherms. Journal of morphology. 1960;106:85–108. doi: 10.1002/jmor.1051060104. [DOI] [PubMed] [Google Scholar]
- 57.Dale AG, Frank SD. Warming and drought combine to increase pest insect fitness on urban trees. PloS one. 2017;12:e0173844. doi: 10.1371/journal.pone.0173844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Musolin DL, Tougou D, Fujisaki K. Too hot to handle? Phenological and life-history responses to simulated climate change of the southern green stink bug Nezara viridula (Heteroptera: Pentatomidae) Global Change Biology. 2010;16:73–87. doi: 10.1111/j.1365-2486.2009.01914.x. [DOI] [Google Scholar]
- 59.Kingsolver JG, Huey RB. Size, temperature, and fitness: three rules. Evolutionary Ecology Research. 2008;10:251–268. [Google Scholar]
- 60.Brans KI, et al. The heat is on: Genetic adaptation to urbanization mediated by thermal tolerance and body size. Global change biology. 2017;23:5218–5227. doi: 10.1111/gcb.13784. [DOI] [PubMed] [Google Scholar]
- 61.Geerts A, et al. Rapid evolution of thermal tolerance in the water flea Daphnia. Nature Climate Change. 2015;5:665. doi: 10.1038/nclimate2628. [DOI] [Google Scholar]
- 62.Oberg E, Del Toro I, Pelini S. Characterization of the thermal tolerances of forest ants of New England. Insectes sociaux. 2012;59:167–174. doi: 10.1007/s00040-011-0201-y. [DOI] [Google Scholar]
- 63.Baudier KM, Mudd AE, Erickson SC, O’donnell S. Microhabitat and body size effects on heat tolerance: implications for responses to climate change (army ants: Formicidae, Ecitoninae) Journal of Animal Ecology. 2015;84:1322–1330. doi: 10.1111/1365-2656.12388. [DOI] [PubMed] [Google Scholar]
- 64.Bennet-Clark H, Young D. The scaling of song frequency in cicadas. Journal of Experimental Biology. 1994;191:291–294. doi: 10.1242/jeb.191.1.291. [DOI] [PubMed] [Google Scholar]
- 65.Sueur J, Aubin T. Specificity of cicada calling songs in the genus Tibicina (Hemiptera: Cicadidae) Systematic Entomology. 2003;28:481–492. doi: 10.1046/j.1365-3113.2003.00222.x. [DOI] [Google Scholar]
- 66.Shieh B-S, Liang S-H, Liao C-Y, Chiu Y-W. Song frequency correlates with latitude and individual body size in the cicada Mogannia formosana Matsumura (Hemiptera: Cicadidae) acta ethologica. 2017;20:147–155. doi: 10.1007/s10211-017-0258-3. [DOI] [Google Scholar]
- 67.Sanborn AF. Body temperature and the acoustic behavior of the cicada Tibicen winnemanna (Homoptera: Cicadidae) Journal of insect behavior. 1997;10:257–264. doi: 10.1007/BF02765558. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets analysed during the current study are available in the Mendeley Data repository, DOI: http://dx.doi.org/10.17632/4z4n7p5gxt.2#file-ae6d6c15-bf0a-4fde-8f32-4f62ebf8f96c.