1. Introduction
The analgesic properties of epoxy fatty acids (EpFA) have been investigated for the past decade. Polyunsaturated long chain fatty acids (PUFAs) such as omega-3 docosahexaenoic acid and eicosapentaenoic acid as well as the omega-6 arachidonic acid are acted on by the oxidative metabolism of cytochrome P450 (CYP450) enzymes to form different classes of EpFA. All of these classes, which include several regioisomers and their enantiomers per parent PUFA, are substrates of the soluble epoxide hydrolase (sEH; EC 3.3.2.10) enzyme. The sEH transforms the EpFA into their corresponding diols which essentially inactivates them [1]. The EpFA are bioactive cell signaling mediators with several beneficial effects including anti-hypertension, anti-inflammation as well as analgesia [2, 3]. Development of potent small molecule inhibitors of sEH that degrade EpFA enabled the observation of these biological effects in vivo. This allowed the examination of EpFA for their analgesic effects, which typically requires an intact living organism to model pain. The analgesia elicited by sEH inhibitors (sEHI) is unique in that it is active against both acute inflammatory and chronic pain conditions. The pharmacokinetics of the sEHI TPPU have been well described in rodent laboratory species as well as primates [4–6]. The efficacy of TPPU has also been demonstrated in several preclinical models of chronic painful neuropathy [7–9]. Because of the potent efficacy and potential to treat chronic pain, it is important to fully characterize the pharmacodynamics of the sEHI. Yaksh et al. describe the defining property of an analgesic as its ability to change pain perception without disturbing other sensory perception and motor skills [10]. The current therapies that treat chronic pain such as opioids (i.e., morphine, fentanyl) and gabapentinoids (i.e., gabapentin, pregabalin) have well known dose limiting side effects. Opioids are known to cause respiratory depression, severe constipation and addiction and their abuse is an ongoing crisis in the United States. Gabapentinoids were originally intended as γ-aminobutyric acid (GABA) analogs but are now identified as calcium channel binders. The antinociceptive mechanism of action of gabapentinoids still remains in question [11], however, their side effects including sedation and dizziness are well described. Even over-the-counter analgesics such as nonsteroidal anti-inflammatory drugs (NSAIDs) which are used for chronic pain conditions but with inadequate efficacy are limited by side effects of gastrointestinal erosion and cardiovascular risks. Besides the adverse side effects that negatively impact the health of patients, there is also the possibility to develop tolerance to the drug effects. The National Institute on Drug Abuse indicates tolerance as a state requiring a higher dose to have the same effect in an organism. Analgesics that are used to combat chronic pain such as opioids and tricyclic anti-depressants and other classes of drugs used by chronic pain patients such as benzodiazepines are able to induce tolerance [12, 13]. Tolerance to opioids is well documented in humans as well as preclinical experiments and there is increasing clinical evidence of tolerance and physical dependence to gabapentinoids [14–16]. Acquired tolerance is evidenced by increased dosing needed to maintain the same effect and this results in a decreased response to the same repeated dose which is the paradigm we used for all the tested compounds.
The risk of adverse events and side effects described above increase with the repeated dosing these analgesics and tolerance is known to develop particularly for opioid analgesics. Because the sEHI are effective against chronic pain and are proposed for long term dosing, we compared the sEHI to positive controls for the development of tolerance. Here we describe our results of assessing the sEHI TPPU for functional parameters with repeated dosing and investigate if tolerance develops with the extended dosing regimen.
2. Results
2.1. Tolerance and analgesia
von Frey
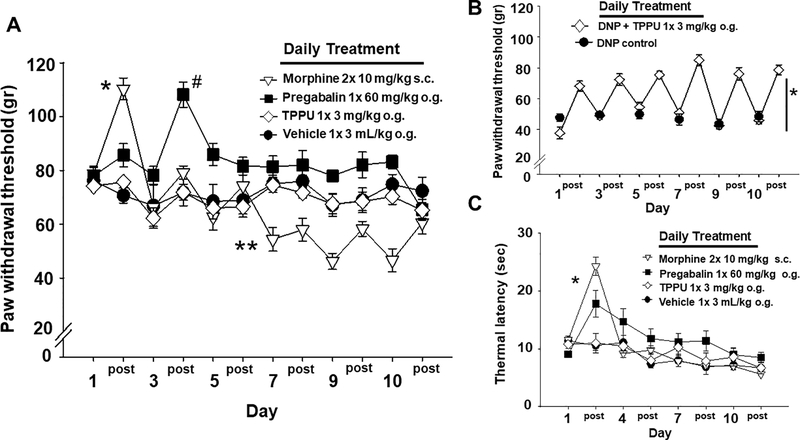
Tolerance to repeated dosing was determined for the compounds and was assessed with several nociceptive tests in naïve rats. The von Frey assay was used to determine the mechanical withdrawal thresholds (MWTs) and the data are depicted as the baseline prior to dosing (labeled per day number) and the measure at 60 minutes post dosing (post) on each of the days. Morphine, as expected, and pregabalin treatments demonstrated robust increases in MWTs, though the timing of the responses to these two drugs differed (Figure 1A). In naïve rats TPPU did not significantly alter MWT responses and remained indistinguishable from the vehicle treated responses (Two Way Analysis of Variance with Holm-Sidak post-hoc, n=6–8/group, p= 0.216 on Day 1). There was a significant interaction between the day and treatment with morphine showing significant increase on Day 1 (p ≤ 0.001) and decrease on Day 7 (p=0.005) while pregabalin showed a significant increase on Day 3 (p ≤ 0.001). The significant decrease from baseline von Frey scores for the morphine treated group indicates opioid induced hyperalgesia may have occurred. Thus, the opioid tolerance developed quickly and was sustained with averaged responses over 10 days while TPPU failed to alter MWTs in healthy animals. Because TPPU did not induce threshold changes in healthy naïve rats, whether it would induce tolerance was still in question, therefore we further explored the repeat dosing paradigm in a model of diabetic neuropathy.
Figure 1.
TPPU mediates effective analgesia without tolerance. A) Tolerance develops with opioids and gabapentinoids. Initial doses of morphine dramatically raised mechanical withdrawal thresholds (MWTs) in naïve rats. The pre- and 60 min post-treatment measures per day depicted in the graph show the diminishing response to treatment over 10 days with morphine indicating tolerance with significant increases on Day 1 (* p ≤ 0.001) and decreases on Day7 (** p=0.005) suggesting the development of opioid induced hyperalgesia. Pregabalin also increased MWT but later (Day 3, # p ≤ 0.001), after which subsequent dosing had no effect but did not lower MWT below baseline. TPPU was indistinguishable from vehicle in this assay in naïve animals. TPPU did not alter MWT early in the time course and it also did not induce any apparent hyperalgesia with repeated dosing. B) In rats with diabetic neuropathic pain (DNP), TPPU significantly increased MWTs over the 10 days of treatment compared to controls (* p ≤ 0.001). The analgesic response from pre-treatment (numbered day) to measures assessed 60 min post treatment (‘post’ per numbered day) did not diminish with repeated administration of the sEHI indicating that tolerance did not develop. C) TPPU dosed to healthy rats does not alter thermal sensitivity. In contrast, both morphine and pregabalin significantly (* p ≤ 0.001) increased the hotplate (52.5°C) latency early in the time course (Day 1) compared to the vehicle indicating an initial response which diminished with repeated dosing (Days 4, 7, 10). The sEHI TPPU showed no change compared to vehicle over the entire time course. DNP rats loose thermal sensitivity altering responses and therefore were not assessed with the thermal assay.
TPPU was tested in a streptozocin induced neuropathic pain model in rats. This demonstrated both the analgesic efficacy of TPPU in the diabetic neuropathy model and the lack of tolerance to the compound with repeated dosing in a chronic pain model (Figure 2B). A daily baseline (labeled per sequential day) and a 60 minutes post treatment interval (post) were assessed daily and compared to controls. This treatment significantly increased MWTs (One Way Repeated Measures Analysis of Variance Holm-Sidak post-hoc, n=5/group, p<0.001) daily from pre to post treatment with no decrease in treatment effect up to 10 days of dosing. The blood concentrations revealed average exposures of 2430 ±404 ng/mL TPPU in blood and average of 87±19 ng/g protein in the brain tissue on Day 10. The potency of TPPU on the rat recombinant sEH enzyme has been previously reported to be 4.6 ± 0.4 nM [7]. Given our knowledge that the potency of TPPU varies with the EpFA substrate class [12] and that we likely must inhibit the majority of the sEH for efficacy, the slow tight binding kinetics and high target occupancy lead to powerful inhibition of the enzyme by TPPU. Thus, the concentrations were more than adequate for enzyme inhibition in the diabetic rats and no observable adverse effects were present over the 10 days of treatment.
Figure 2.
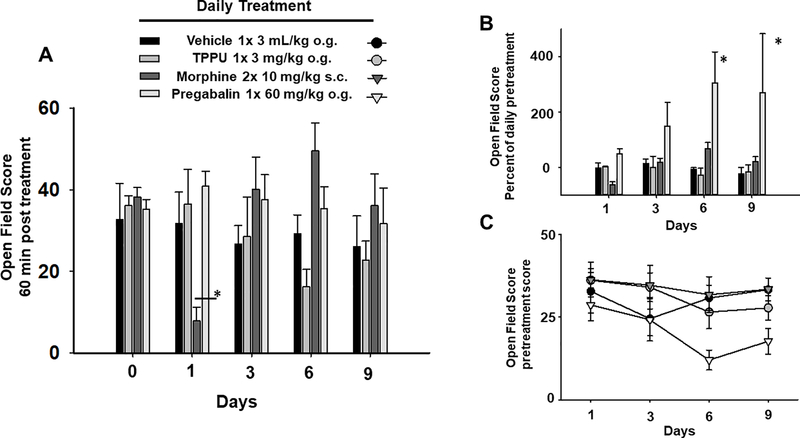
The sEH inhibitor TPPU does not induce significant change in the open field behavior of the healthy animals unlike the positive controls. A) The open field scores were assessed 60 minutes post treatment on alternate days and, as repeated daily dosing progressed, morphine first depressed locomotion (Day 1, * p ≤ 0.001) and then displayed typical hyperlocomotion (Day 3–6) which also showed tolerance development by Day 9. B) Pregabalin showed significant increases (* p ≤ 0.001) in pre to post open field scores without tolerance (calculated as 60 min post score/pretreatment score *100) while TPPU showed no change from vehicle. C) With continued treatment pregabalin negatively impacted the pretreatment daily baseline scores depicted here and rats responded to the drug administration with typical disinhibition (increased motility) resulting in the large increases in the calculated scores in Fig 2B.
Hot plate
The naïve rats tested in the von Frey assay were also assessed for response to thermal stimulation. Rats with diabetic neuropathic pain loose thermal sensitivity over time likely due to c-fiber degradation and were therefore not assessed in the thermal stimulus assay. However, as anticipated, repeat administration of morphine in healthy rats demonstrated an initial response measured as performance on a hot plate (52.5°C) with a significant increase in latency (time to respond) followed by a decrease in effect with repeated dosing (Figure 1C) (Kruskal-Wallis One Way Analysis of Variance on Ranks, H = 15.453 with 3 degrees of freedom, Dunn’s post-hoc, n=6–8/group, p = 0.001 compared to vehicle). Pregabalin also displayed an initial increase and a diminishing response with subsequent dosing. Notably, TPPU did not demonstrate similar modulation of thermal response compared either class of analgesic and was not different from vehicle control in the same assay. Thus, the sEHI TPPU which is robustly analgesic in several chronic pain models [7–9] demonstrated no effect in healthy rats in early stages of repeat dosing. Both morphine and pregabalin displayed first a robust response compared to control followed by a decrease in the thresholds with continued repeat dosing indicating the development of tolerance. The non-significant trend in both vehicle and sEHI responses to decline over 10 days suggest the effect of habituation or training and are not likely due to extra sensitivity to the stimulus.
2.2. Assessing Gait
Open Field
Open field assays were used to gauge the effect of the extended dosing on the exploration and motor function of the animals. The scores from the open field assays revealed the significantly depressed locomotion (Day 1) and then hyperlocomotion (Day 6) of the morphine treated rats with repeated dosing (Figure 2A, (Two Way Analysis of Variance with Holm-Sidak post-hoc, n=4–6/group, p<0.001). Morphine adminstration typically results in hyperlocomotor activity in rodents [13]. Further analysis revealed pregabalin significantly altered motility and exploration compared to vehicle (Figure 2B, Kruskal-Wallis One Way Analysis of Variance on Ranks, H = 13.479 with 3 degrees of freedom, Dunn’s post-hoc, n=6–8/group, p = 0.004). The change due to pregabalin treatment far exceeded the morphine response and increased open field activity over the entire time course per the calculated value (60 min post treatment score/pretreatment score *100) (Figure 2B). It should be noted that the pregabalin also greatly affected the test day pretreatment scores which were used to calculate this change (Figure 2C). A recent study using pregabalin in human patients with diabetic neuropathy revealed no significant pain relief but also increased gait abnormalities including increased stride length and velocity under treatment [14]. In our study in naïve rats the pregabalin actually negatively impacted the pretreatment baseline as well as increasing movement at 1 hr post treatment in the open field over the 10 day time course. The streptozocin induced rats were also assessed for their open field scores. Due to their compromised health status this was limited to Day 5 and on the final day of treatment (Day 10) 60 minutes post treatment. The Day 5 average open field score was 25 ±2.5 and remained the similar measured on Day 10 with average of 25.2± 2.9 after 10 daily treatments at 3 mg/kg TPPU (n=6 male SD STZ rats). Repeated TPPU treatment, which exhibited effective analgesia in these rats with no development of tolerance in the von Frey assay, demonstrated no change in open field scores after the course of treatment.
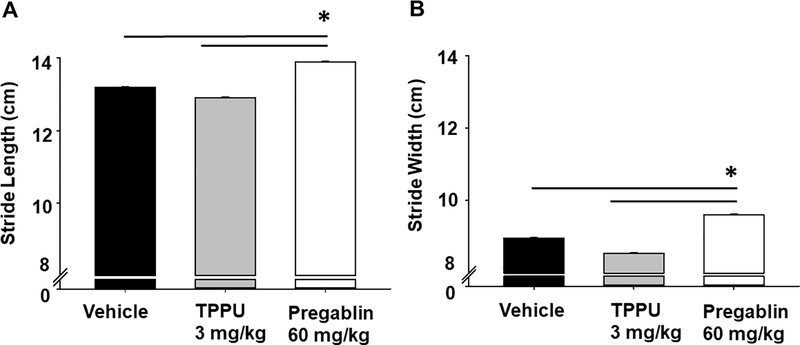
Gaiting is another sensitive measure of motor skill effects related to disease but also of drugs and substances that impair motor coordination as well as having a high translatable value from animals to humans [15]. In healthy rats, and similar to the results of the open field, the gaiting was significantly altered by pregabalin but not TPPU. The stride length in pregabalin treated animals increased compared to both vehicle and TPPU (Fig 3A) (Kruskal-Wallis One Way Analysis of Variance on Ranks with Dunn’s post-hoc, n=9/group p ≤ 0.001, H = 27.445 with 2 degrees of freedom). The gait width was similarly significantly increased by pregabalin compared to vehicle and TPPU (Fig 3B) (Kruskal-Wallis One Way Analysis of Variance on Ranks with Dunn’s post-hoc, p ≤ 0.001, H = 23.418 with 2 degrees of freedom). The sEHI TPPU did not significantly alter gait length or width in the rats compared to the vehicle control.
Figure 3.
TPPU does not alter the gaiting of healthy rats while pregabalin does. Compounds were assessed to affect gaiting as an outcome of their CNS effects and changes in motor skill. Measurement of gait demonstrated A) stride length and B) stride width were altered in naïve rats 1.5 hr post oral gavage of pregabalin but not TPPU. The pregabalin dose resulted in significant differences (p ≤ 0.001) while TPPU showed no change from vehicle.
3. Discussion
The sEH inhibitor TPPU was assessed in a blind comparison with other analgesic compounds which are the leading therapies for chronic pain conditions to test if tolerance occurs with repeated dosing. Loss of efficacy over time (tolerance) or the abuse potential of the molecule (reward) are factors driving drug development of analgesics [10]. We have previously demonstrated the lack of rewarding effects of sEHI including in mice with genetic knockout of the sEH target in operant tests [8, 16]. As analgesics the sEHI are proposed for long term use to treat chronic pain conditions. The treatment of pain is greatly limited by the paucity of potent analgesics that lack severe side effects. Despite the almost assured occurrence of physical dependence and tolerance, opioid analgesics are still prescribed for chronic pain disorders due to the failure of other classes of analgesics like gabapentinoids to treat these pain conditions. The utility of opioids for chronic and moderate to severe acute pain is the absence of the ‘ceiling effect’ of other analgesics such as NSAIDs, meaning increased dosing results in increased effect. This benefit of opioids leads directly to one of the major safety problems of opioids. Because opioids have no ceiling effect they can be dose escalated to maintain pain relief, with an associated increased risk of other dangerous side effects [17]. The incidence of opioid side effects then may become even more problematic because the tolerance to the analgesia mediated by opioids occurs at a faster pace than other effects [18]. In light of the particular side effects of opioid dependence and addiction there is a current drive to develop alternative analgesics with broad efficacy. Therefore, we tested the sEHI TPPU, which has demonstrated analgesic effects in chronic pain models, to assess for the occurrence of tolerance with repeated daily administration compared to morphine and pregabalin using nociceptive assays. We employed a von Frey assay with a rigid tip to assess daily mechanical withdrawal thresholds in naive rats (Fig 1A) and diabetic neuropathic rats (Fig 1B) over the time course. We also measured thermal latency to assess the change in hyperalgesia indicative of tolerance in naïve rats (Fig 1C) and limited test days to avoid the behavioral conditioning of frequently repeated hot plate trials. To assess tolerance, the doses were held constant and the change in response monitored in an adaptation of the classical setting of a right-shift (increased dose) to induce the same level of response in a dose-response curve. The administration route and doses of the included positive controls were chosen for equipotent analgesia to maintain blinding between positive controls. The results in naïve rat demonstrated that tolerance developed in morphine and pregabalin treated rats because both mechanical and thermal scores revealed an initial threshold increase that diminished over the course of daily treatments for 10 days. Conversely, TPPU did not induce changes in nociception tested in the von Frey or hot plate assays in healthy rats. This outcome of sEHI lacking effect in the absence of a painful state has been observed repeatedly in nociceptive assays [8, 19, 20].
Tolerance to opioids as analgesics is well known and often approached clinically by increasing opioid doses or opioid rotation. However, these strategies are not always sufficient to safely control pain given the on-target side effects of opioids. What is most significant in these cases is that differential tolerance occurs which is the rapid tolerance to the analgesic effects of opioids compared to slower adaptation with the respiratory and gastrointestinal effects [21]. Thus, the side effects often do not have the same trajectory of analgesic tolerance making them dangerous in the case of respiratory depression (mostly with substance abusers) and also largely uncomfortable as with constipation (because gut motility experiences little tolerance compared to nausea and sedation) [22]. Recently, with the discovery of biased agonism of opioid receptors, there are also novel mμ opioid G protein coupled receptors (GPCR) biased agonists being developed to mitigate some of the side effects of classical agonists such as morphine. It has been demonstrated that TRV130 which produces GPCR biased signaling induces less constipation while maintaining analgesia in rodents [23]. However, in mice and humans, behavioral effects demonstrate the GPCR bias may not be effective in eliminating reward potential [24, 25] and may also cause opioid induced hyperalgesia [26]. The euphoric effects (reward potential) of opioids in part may drive drug seeking behavior [27]. Thus, although there are advances in limiting on-target side effects of opioids, alternatives are still needed to effectively treat pain [26] and sEHI are poised to meet this need.
Preclinical study results often have difficulty translating to clinical pain relief due to many factors, one of which may be the outcomes which are measured by verbal report in humans and withdrawal from stimuli in laboratory species [28]. Because verbal reports in humans may be complicated by drug seeking behaviors or psychological conditions, other metrics are often sought [27]. In this way, physical functions of humans are also assessed for pain scores, such as lack of mobility in walking, or trouble with daily life skills and working. In fact, a goal of pain treatment in humans is to prevent and/or recover from impaired functioning [29]. Thus, the goal is to prevent functional impairment due to pain and therefore the analgesics should not contribute to impaired central nervous system function in order to relieve pain. Brain penetration of TPPU has previously been demonstrated in healthy animals (5), here we determined the concentration of the inhibitor in blood and brain in diabetic neuropathic rats. At the end of 10 days the blood concentration was high and compound was present in the brain, however, there were no changes in sensitive measures of open field conduct and catwalk gaiting. Thus, the spontaneous locomotion (open field) and motor skill (gaiting) of the TPPU treated animals remained intact during treatment while the inhibitor demonstrated efficacious analgesia and sufficient brain levels of the compound to inhibit the target enzyme at the same dose in neuropathic rats. The penetration of TPPU into the brain suggests that the analgesia could be centrally mediated. But in addition to this, sEH inhibition has previously been demonstrated to reduce inflammatory pain both with intraspinal administration [19] and in the periphery [20]. The broad scope of sEHI mediated analgesia is thought to be due to the ability to reduce oxidative damage to cells by preventing mitochondrial dysfunction and blocking the resulting damage that drives endoplasmic reticulum stress [30]. The data from the experiments here suggest that sEH inhibitors have the therapeutic potential to replace opioids and gabapentinoids for chronic pain disorders as effective analgesics without the development of tolerance or reward.
4. Conclusion
The current opioid epidemic presents several issues, first the need for alternative analgesics that lack abuse potential and second the need for agents with sustained efficacy for chronic pain conditions. The analgesic efficacy of sEH inhibition has been previously demonstrated in acute and chronic pain models. There is also evidence they lack abuse potential. The experiments reported here demonstrated a lack of CNS altering effects in multiple assays, the absence of activity in naïve animals, and the lack of attenuated efficacy with repeat dosing in a neuropathic pain model. Thus, the sEH inhibitors could be a novel strategy to combat chronic pain conditions in humans and a timely alterative to opioid analgesics.
5. Methods and materials
5.1. Animals
Procedures and animal care performed at the University of California, Davis adhered to the guidelines of the National Institutes of Health guide for the care and use of Laboratory animals and were performed in accordance with the protocols approved by the Animal Use and Care Committee (IACUC) of the University of California, Davis. Great care was taken to minimize suffering of the animals and to reduce the number of animals used. Sprague–Dawley male rats (250 to 300 g; Charles River, Wilmington, MA, USA) were housed 2 per cage on corncob bedding all with free access to food and water. They were maintained under a 12 h light/dark at controlled temperature and relative humidity (65–77°F with 35–60% humidity). The animals were randomly divided into groups and behavioral testing was also randomized among groups during testing with assays performed between 9:00 a.m. and 5:00 p.m. Scientists running the experiments were blinded to the treatment protocol at the time of the tests.
5.2. Chemicals
Morphine sulfate formulated in saline (Fisher Scientific, Pittsburgh, PA) was administered by subcutaneous injection, pregabalin (Tocris, Minneapolis, MN) was formulated in water administered by oral gavage, the sEHI TPPU (synthesized in house) was formulated in PEG300 (Sigma Aldrich, St. Louis MO) which was tested as the vehicle and both were administered by oral gavage. Streptozocin (Fisher Scientific, Pittsburgh, PA) was formulated in saline and administered via tail vein catheter.
5.3. Tolerance and analgesia
To assess tolerance, we tested a 10 day repeated 2x daily subcutaneous administration of 10 mg/kg morphine or 1x daily oral gavage of the sEHI TPPU at 3 mg/kg, pregabalin at 60 mg/kg and vehicle 3ml/kg using von Frey, hot plate, open field and assays. The administration frequency of the compounds differed based on half-lives and duration of action. Morphine (3 hr half-life) with a duration of action of approximately 4–5 hours was administered more frequently to induce tolerance [13, 31]. TPPU (10–12 hr half-life), pregabalin (6–12 hrs) and the vehicle were dosed once daily. Rats were acclimated and tested for a baseline before treatment and with a schedule of motor or nociceptive assays daily for 10 days of treatment. After initial increases in threshold responses with drug administration, the decrease in responses with repeated dosing in naive rats indicated the developing tolerance. We tested the rats daily in the von Frey assay using an electronic aesthesiometer with a rigid tip to assess mechanical withdrawal thresholds (MWTs) and on days only on 1, 4 and 7 and 10 for hot plate (52.5°C) latency to avoid the development of behavioral habituation to repeated suprathreshold thermal testing [32]. The von Frey scores are reported as the grams to induce a withdrawal response and the hot plate scores represent the latency in seconds to hind paw lick or jump behaviors. All assays are reported as the mean ± SEM for groups.
5.4. Diabetic peripheral neuropathy (DPN) model
Rats were assessed for their baseline MWT nociceptive scores and then induced with type I diabetes with a single 55 mg/kg streptozocin i.v. injection and allowed to develop neuropathic pain. Once allodynia was confirmed with a von Frey assay 5–7 days after streptozocin injection, the rats were treated for 10 days with TPPU 3 mg/kg orally exactly as the naïve animals in previous experiments. The von Frey scores were measured to assess the treatment over 10 days. Thermal sensitivity was not assessed because it is subject to change over the time course in the diabetic neuropathy model due to c-fiber loss. For the graphs the daily pre- and 60 minute post-treatment scores are depicted to reveal the change from normal baseline in naïve animals or the painful neuropathic baseline compared to treatment in diabetic neuropathic animals.
5.5. Blood concentration determination
Prior to the begin of treatment of the diabetic rats, 10 uL of whole blood was collected via tail vein puncture. Then a post treatment regime blood sample was taken on Day 10 after behavioral assessments and the animals were euthanized and sampled for brain tissue. The blood concentrations before and after treatment and brain tissue concentrations on Day 10 were analyzed per previously reported LC-MS/MS methods [33].
5.6. Gaiting assessment
We assessed motor skills in naïve rats with the open field assay every third day (Days 1, 3, 6, 9) and diabetic neuropathic rats at the middle and end of treatment (to reduce handling stress). For the open field assay rats were placed in an open-field arena (40W x 40L x 30H cm) of a 16-square grid clear acrylic open top chamber and observed for 2 min continuously. Activity was assessed and scored as the sum of lines each animal crossed with both hind paws and number of rears as a function of time. The scores are a sum of the lines crossed with both hind paws and total number of rearing. For the calculated assay results, the scores were the daily treatment score divided by the daily baseline score multiplied by 100 to obtain the percent of baseline measure.
For the ‘catwalk’ gaiting assay naïve rats were habituated to handling by trained staff for 3 consecutive days prior to testing. On the first day of the of 2 trials, rats were administered test compounds or oral gavage randomly and 90 minutes later were assessed for gait by staff blinded to the treatments. For the test, rats were brought into the test area and placed one at a time on a runway tunnel lined with white paper and a dark goal box at the end. Rat paws were dabbed with nontoxic ink (front and back different colors) and allowed to enter and walk the length of the tunnel. The resulting footprints were then scored manually for stride length and width (measured in centimeters) by trained staff blinded to treatments. The measures were assessed with a straight line from toe to heel of paw print (vertical line) and a line below the most lateral toe on either side of the print (horizontal line) determining the center of the paw print where the two lines intersect. The rats were assessed twice daily on two separate test days and the scores reported as the sum of these trials. After the completion of the assay the data were decoded, grouped by treatments and subjected to statistical analysis. Morphine was not assessed in this assay and location due to logistics imposed by regulatory controls.
5.7. Statistics
Statistical analysis was performed using Systat Softare Inc. Sigmaplot 14.0 for Windows. All results are expressed as mean ± standard error of the mean (SEM). Data were tested for normality and homogeneity of variance. If the variance was homogeneous, one-way or two-way ANOVA followed by Holms-Sidak post hoc analysis were used. Nonparametric data were analyzed using the Kruskal-Wallis test followed by Dunn’s post hoc or the Mann-Whitney U test. P < 0.05 was considered significant.
Soluble epoxide hydrolase (sEH) inhibition mediates analgesia against chronic pain.
Small molecule sEH inhibitors to not induce tolerance in chronic pain models.
sEH inhibitors do not alter behavior in control animals.
Acknowledgments
This work was supported by the National Institute of Environmental Health Sciences (NIEHS) Grant R01 ES002710, NIEHS Superfund Research Program P42 ES004699, National Institute of Neurological Disorders and Stroke (NINDS) U54 NS079202-01 and Grants NIH 5T32DC008072-05 and 4T32HL086350-09 (to K.W.) and NIEHS T32 ES007059 (to J.A.). Partial support for clinical developmental of sEH inhibitors for human medicine comes from the NIEHS SBIR Program R44ES025598 and the NIH NINDS Blueprint Neurotherapeutics Network UH2NS094258. The gaiting studies were conducted at the UC Davis MIND Institute IDDRC Rodent Behavior Core.
Abbreviations
- CNS
central nervous system
- CYP450
cytochrome P450
- EpFA
epoxy fatty acids
- ER Stress
endoplasmic reticulum stress
- GPCR
G protein coupled receptors
- GABA
γ-aminobutyric acid
- MWT
mechanical withdrawal thresholds
- NSAIDs
non-steroidal anti-Inflammatory drugs
- PUFAs
polyunsaturated long chain fatty acids
- sEH
soluble epoxide hydrolase
- sEHI
soluble epoxide hydrolase inhibitors
- TPPU
1-trifluoromethoxyphenyl-3-(1-propionylpiperidin-4-yl) urea
Conflict of Interest Statement: The University of California holds patents on the sEH inhibitors used in this study as well as their use to treat inflammation, inflammatory pain, and neuropathic pain. BD Hammock is a co-founder and K Wagner is an employee of EicOsis L.L.C., a startup company advancing sEH inhibitors into the clinic.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Spector AA, Kim HY. Cytochrome P450 epoxygenase pathway of polyunsaturated fatty acid metabolism. Biochim Biophys Acta. 2015;1851(4):356–365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chiamvimonvat N, Ho CM, Tsai HJ, Hammock BD. The soluble epoxide hydrolase as a pharmaceutical target for hypertension. J Cardiovasc Pharmacol. 2007;50(3):225–237. [DOI] [PubMed] [Google Scholar]
- 3.Kodani SD, Hammock BD. The 2014 Bernard B. Brodie Award Lecture Epoxide Hydrolases: Drug Metabolism to Therapeutics for Chronic Pain. Drug metabolism and disposition: the biological fate of chemicals. 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Harris TR, Bettaieb A, Kodani S, Dong H, Myers R, Chiamvimonvat N, Haj FG, Hammock BD. Inhibition of soluble epoxide hydrolase attenuates hepatic fibrosis and endoplasmic reticulum stress induced by carbon tetrachloride in mice. Toxicol Appl Pharmacol. 2015;286(2):102–111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ostermann AI, Herbers J, Willenberg I, Chen R, Hwang SH, Greite R, Morisseau C, Gueler F, Hammock BD, Schebb NH. Oral treatment of rodents with soluble epoxide hydrolase inhibitor 1-(1-propanoylpiperidin-4-yl)-3-[4-(trifluoromethoxy)phenyl]urea (TPPU): Resulting drug levels and modulation of oxylipin pattern. Prostaglandins Other Lipid Mediat. 2015;121(Pt A):131–137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ulu A, Appt S, Morisseau C, Hwang SH, Jones PD, Rose TE, Dong H, Lango J, Yang J, Tsai HJ, Miyabe C, Fortenbach C, Adams MR, Hammock BD. Pharmacokinetics and in vivo potency of soluble epoxide hydrolase inhibitors in cynomolgus monkeys. Br J Pharmacol. 2012;165(5):1401–1412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sasso O, Wagner K, Morisseau C, Inceoglu B, Hammock BD, Piomelli D. Peripheral FAAH and soluble epoxide hydrolase inhibitors are synergistically antinociceptive. Pharmacol Res. 2015;97:7–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wagner K, Lee KS, Yang J, Hammock BD. Epoxy fatty acids mediate analgesia in murine diabetic neuropathy. Eur J Pain. 2017;21(3):456–465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wagner KM, McReynolds CB, Schmidt WK, Hammock BD. Soluble epoxide hydrolase as a therapeutic target for pain, inflammatory and neurodegenerative diseases. Pharmacol Ther. 2017;180:62–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yaksh TL, Hunt MA, dos Santos GG. Development of New Analgesics: An Answer to Opioid Epidemic. Trends in Pharmacological Sciences. 2018;39(12):1000–1002. [DOI] [PubMed] [Google Scholar]
- 11.Patel R, Dickenson AH. Mechanisms of the gabapentinoids and α (2) δ−1 calcium channel subunit in neuropathic pain. Pharmacology Research & Perspectives. 2016;4(2):e00205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Morisseau C, Inceoglu B, Schmelzer K, Tsai HJ, Jinks SL, Hegedus CM, Hammock BD. Naturally occurring monoepoxides of eicosapentaenoic acid and docosahexaenoic acid are bioactive antihyperalgesic lipids. J Lipid Res. 2010;51(12):3481–3490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Grecksch G, Bartzsch K, Widera A, Becker A, Hollt V, Koch T. Development of tolerance and sensitization to different opioid agonists in rats. Psychopharmacology (Berl). 2006;186(2):177–184. [DOI] [PubMed] [Google Scholar]
- 14.Karmakar S, Rashidian H, Chan C, Liu C, Toth C. Investigating the role of neuropathic pain relief in decreasing gait variability in diabetes mellitus patients with neuropathic pain: a randomized, double-blind crossover trial. J Neuroeng Rehabil. 2014;11:125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Piesla MJ, Leventhal L, Strassle BW, Harrison JE, Cummons TA, Lu P, Whiteside GT. Abnormal gait, due to inflammation but not nerve injury, reflects enhanced nociception in preclinical pain models. Brain Res. 2009;1295:89–98. [DOI] [PubMed] [Google Scholar]
- 16.Wagner K, Yang J, Inceoglu B, Hammock BD. Soluble epoxide hydrolase inhibition is antinociceptive in a mouse model of diabetic neuropathy. The journal of pain : official journal of the American Pain Society. 2014;15(9):907–914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Khan MI, Walsh D, Brito-Dellan N. Opioid and adjuvant analgesics: compared and contrasted. The American journal of hospice & palliative care. 2011;28(5):378–383. [DOI] [PubMed] [Google Scholar]
- 18.Hayhurst CJ, Durieux ME. Differential Opioid Tolerance and Opioid-induced Hyperalgesia: A Clinical Reality. Anesthesiology: The Journal of the American Society of Anesthesiologists. 2016;124(2):483–488. [DOI] [PubMed] [Google Scholar]
- 19.Inceoglu B, Jinks SL, Ulu A, Hegedus CM, Georgi K, Schmelzer KR, Wagner K, Jones PD, Morisseau C, Hammock BD. Soluble epoxide hydrolase and epoxyeicosatrienoic acids modulate two distinct analgesic pathways. Proc Natl Acad Sci U S A. 2008;105(48):18901–18906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wagner K, Inceoglu B, Dong H, Yang J, Hwang SH, Jones P, Morisseau C, Hammock BD. Comparative efficacy of 3 soluble epoxide hydrolase inhibitors in rat neuropathic and inflammatory pain models. Eur J Pharmacol. 2013;700(1–3):93–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dumas EO, Pollack GM. Opioid Tolerance Development: A Pharmacokinetic/Pharmacodynamic Perspective. The AAPS Journal. 2008;10(4):537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cahill CM, Walwyn W, Taylor AMW, Pradhan AAA, Evans CJ. Allostatic Mechanisms of Opioid Tolerance Beyond Desensitization and Downregulation. Trends Pharmacol Sci. 2016;37(11):963–976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.DeWire SM, Yamashita DS, Rominger DH, Liu G, Cowan CL, Graczyk TM, Chen XT, Pitis PM, Gotchev D, Yuan C, Koblish M, Lark MW, Violin JD. A G protein-biased ligand at the mu-opioid receptor is potently analgesic with reduced gastrointestinal and respiratory dysfunction compared with morphine. J Pharmacol Exp Ther. 2013;344(3):708–717. [DOI] [PubMed] [Google Scholar]
- 24.Soergel DG, Subach RA, Sadler B, Connell J, Marion AS, Cowan CL, Violin JD, Lark MW. First clinical experience with TRV130: pharmacokinetics and pharmacodynamics in healthy volunteers. J Clin Pharmacol. 2014;54(3):351–357. [DOI] [PubMed] [Google Scholar]
- 25.Manglik A, Lin H, Aryal DK, McCorvy JD, Dengler D, Corder G, Levit A, Kling RC, Bernat V, Hubner H, Huang XP, Sassano MF, Giguere PM, Lober S, Da D, Scherrer G, Kobilka BK, Gmeiner P, Roth BL, Shoichet BK. Structure-based discovery of opioid analgesics with reduced side effects. Nature. 2016;537(7619):185–190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Araldi D, Ferrari LF, Levine JD. Mu-opioid Receptor (MOR) Biased Agonists Induce Biphasic Dose-dependent Hyperalgesia and Analgesia, and Hyperalgesic Priming in the Rat. Neuroscience. 2018;394:60–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Severino AL, Shadfar A, Hakimian JK, Crane O, Singh G, Heinzerling K, Walwyn WM. Pain Therapy Guided by Purpose and Perspective in Light of the Opioid Epidemic. Frontiers in psychiatry. 2018;9:119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Deuis JR, Dvorakova LS, Vetter I. Methods Used to Evaluate Pain Behaviors in Rodents. Frontiers in Molecular Neuroscience. 2017;10:284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Negus SS. Addressing the Opioid Crisis: The Importance of Choosing Translational Endpoints in Analgesic Drug Discovery. Trends in Pharmacological Sciences. 2018;39(4):327–330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Inceoglu B, Bettaieb A, Haj FG, Gomes AV, Hammock BD. Modulation of mitochondrial dysfunction and endoplasmic reticulum stress are key mechanisms for the wide-ranging actions of epoxy fatty acids and soluble epoxide hydrolase inhibitors. Prostaglandins Other Lipid Mediat. 2017;133:68–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fabian G, Bozo B, Szikszay M, Horvath G, Coscia CJ, Szucs M. Chronic morphine-induced changes in mu-opioid receptors and G proteins of different subcellular loci in rat brain. J Pharmacol Exp Ther. 2002;302(2):774–780. [DOI] [PubMed] [Google Scholar]
- 32.Morgan MM, Reid RA, Saville KA. Functionally selective signaling for morphine and fentanyl antinociception and tolerance mediated by the rat periaqueductal gray. PLoS One. 2014;9(12):e114269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wan D, Yang J, McReynolds CB, Barnych B, Wagner KM, Morisseau C, Hwang SH, Sun J, Blocher R, Hammock BD. In vitro and in vivo Metabolism of a Potent Inhibitor of Soluble Epoxide Hydrolase, 1-(1-Propionylpiperidin-4-yl)-3-(4-(trifluoromethoxy)phenyl)urea. Front Pharmacol. 2019;10:464. [DOI] [PMC free article] [PubMed] [Google Scholar]