Abstract
Hemodynamic effectiveness of methylene blue (MB) was tested in patients with refractory distributive shock. A retrospective analysis of 20 critically-ill patients who developed refractory shock was performed. Patients were divided into two study groups as responders with positive hemodynamic response to MB administration (defined as 10% decrease of norepinephrine dose) and non-responders. Hemodynamic, outcome data and baseline tissue hypoxia-related parameters including ratio of central venous-to-arterial carbon dioxide tension to arterio-venous oxygen content (P(v-a)CO2/C(a-v)O2) were compared between the groups. There were 9 (45%) responders and 11 (55%) non-responders to single bolus of MB administration. Dose of MB did not differ between responders and non-responders (1.3 ± 0.5 vs. 1.3 ± 0.4 mg/kg respectively, P = 0.979). MB responders had lower baseline P(v-a) CO2/C(a-v)O2 (1.79 ± 0.73 vs. 3.24 ± 1.18, P = 0.007), higher pH (7.26 ± 0.11 vs. 7.16 ± 0.10, P = 0.037) and lower lactate levels at 12 hours post MB administration (3.4 ± 2.7 vs. 9.9 ± 2.2 mmol/L, P = 0.002) compared to non-responders. Methylene blue represents a non-adrenergic vasopressor with only limited effectiveness in patients with refractory distributive shock. Profound tissue hypoxia with high degree of anaerobic metabolism was associated with the loss of hemodynamic responsiveness to its administration.
Subject terms: Bacterial infection, Combination drug therapy, Outcomes research
Introduction
A circulatory shock with hypotension and the need for high vasopressor support in critically ill patients represents a state with excessive mortality exceeding 50%1. Its further progression into refractory shock, defined as norepinephrine requirements above 0.5 µg/kg/min2, is associated with even higher mortality3. Generally, any type of shock is associated with systemic tissue hypoperfusion leading to the development of systemic inflammatory response syndrome (SIRS) and generalized vasodilation, thus always progressing to some degree of distributive shock. Cytokine-induced hyperproduction of nitrous oxide, activation of endothelial ATP-sensitive potassium channels, vasopressin deficiency, and relative corticosteroid insufficiency are the key pathophysiological factors involved in this process2.
Recently, new rescue methods have been introduced in the management of refractory shock including the use of low-dose corticosteroid supplementation and non-adrenergic vasopressors administration such as vasopressin, angiotensin II and methylene blue4,5. Clinical trials have reported variable and limited hemodynamic responsiveness to these agents with documented dismal outcome in non-responders5,6, and the true effect on mortality remains unknown7.
Methylene blue (MB) is a reduction agent traditionally used in the treatment of methemoglobinaemia, however, it has been increasingly used in the management of refractory distributive shock. Its mechanism of action is based on the inhibition of the nitric oxide-cyclic guanosine monophosphate pathway leading to the increased vasomotor tone in the arterioles. The evidence consisting of case reports and several small clinical trials suggests that MB can be successfully used in patients with vasoplegic syndrome after surgery with the use of cardio-pulmonary bypass (CPB), septic shock or lethal dihydropyridine intoxication8. Nevertheless, similarly to the other non-adrenergic vasopressors, there is a lack of data on the actual MB hemodynamic responsiveness in patients with already established refractory shock and the factors that may influence it.
In this retrospective study, we evaluated the hemodynamic effectiveness of MB in the treatment of refractory distributive shock and its impact on clinical outcomes in a mixed population of medical and surgical patients.
Methods
Ethical approval and informed consent
1. Ethical approval for this study (201/19 S-IV) was provided by the Ethical Committee of General University Hospital, Prague, Czech Republic (Chairperson Prof. J. Sedivy) on 21st February 2019.
2. Data acquisition and analysis were carried out in accordance with our institution´s local guidelines and regulations.
3. Due to the retrospective design of the study and according to national legislation Ethical Committee decided that patients´ informed consent was not required.
Patient population
We searched our institution´s intensive care registry for any use of methylene blue (methylthionium chloride) as a vasopressor agent in the whole history of recorded data. Subsequently, identified patients´ data were collected and reviewed from hospital records. Only patients with refractory distributive shock with norepinephrine dose of more than 0.5 µg/kg/min and measured cardiac index of more than 2.4 L/min/m2 were included in the study.
Study design
Patients were divided into two study groups. The first group comprised of patients who responded to MB administration by lowering the norepinephrine dose by 10% within the first 2 hours after the intervention. The patients who did not respond to the treatment were in the second group. In the previous studies, mean arterial pressure (MAP) change was used to assess vasopressor responsiveness5,6. However, in our institution, there is a nurse-driven protocol of norepinephrine infusion aiming for the MAP of 70 mmHg, therefore we used norepinephrine dose change instead. The demographic and medical history data, outcome data and baseline tissue hypoxia-related biochemical parameters including arterial lactate, pH, base deficit (BD), central venous oxygen saturation (ScvO2), central venous-to-arterial carbon dioxide tension difference (P(v-a)CO2 gap), ratio of central venous to arterial carbon dioxide tension to arterio-venous oxygen content (P(v-a)CO2/C(a-v) O2) and ionized calcium concentration were compared between the groups. Also, post-intervention 12-hour courses of norepinephrine doses, mean arterial pressures (MAP), arterial lactate and ionized calcium concentrations were compared between the groups.
Statistical analysis
Statistica v.12 software (StatSoft, USA) was used for statistical analysis. The Kolmogorov-Smirnov test was used to verify the normality of distribution of continuous variables. Differences between the groups were assessed using Student’s t-test for normally distributed variables, which are expressed as mean ± standard deviation. A chi-square test, followed by Fisher’s exact test as appropriate, was used for comparisons of categorical data, which are expressed as counts and frequencies. 30-day survival was compared using the Kaplan-Meier method and the Mantel-Cox test was used to test survival differences between the study groups. P values < 0.05 were considered statistically significant.
Results
Twenty patients who received a single intravenous bolus of MB for the treatment of refractory distributive shock were identified throughout the period from February 2011 to January 2019. Nine patients (45%) responded positively to MB administration and 11 patients (55%) were non-responders with an insignificant hemodynamic response. In responders, 6 patients (66.7%) were in post-CPB SIRS shock, 2 (22.2%) patients had septic shock and 1 patient (11.1%) suffered dihydropyridine intoxication induced distributive shock. In non-responders, there were only patients with post-CPB SIRS shock. Two patients (22.2%) in the MB responders group were on veno-arterial extracorporeal membrane oxygenation (ECMO). In these patients, MB was administered due to severe hypotension despite maximal ECMO blood flow rates of 6 L/min. Hemodynamic monitoring included the use of Swan-Ganz catheter in 12 (60%), Vigileo FloTracTM monitoring (Edwards Lifesciences) in 4 (20%) and transthoracic/transesophageal echocardiography in 4 patients (20%).
There was no significant difference in demographic or medical history data between the study groups (Table 1). All patients were intubated and mechanically ventilated, the degree of organ dysfunction assessed by SOFA and APACHE II score at the onset of refractory shock also did not differ between the study groups (Table 2). The dose of MB (1.3 ± 0.5 vs. 1.3 ± 0.4 mg/kg respectively, P = 0.979) and time from the onset of a refractory shock to intervention (12.7 ± 2.8 vs. 10.6 ± 6.8 hours respectively, P = 0.553) did not differ between responders and non-responders. There was not a significant difference in the use of other non-adrenergic vasopressors. A continuous infusion of vasopressin in the dose 4 IU/hour was administered in all responders and in 10 patients (92.9%) in the non-responder group (P = 1.00). Low-dose corticosteroid supplementation (intravenous boluses of hydrocortisone in the total daily dose of 2 mg/kg) was used in 4 (44.4%) responders and 3 (27.3%) non-responders (P = 0.642).
Table 1.
Patient´s characteristics.
| Responders, n = 9 | Non-responders, n = 11 | P-value | |
|---|---|---|---|
| Age (years) | 57.9 ± 19.3 | 70.3 ± 8.1 | 0.068 |
| Gender - males | 5 (55.6%) | 6 (54.5%) | 0.091 |
| BMI (kg/m2) | 26.5 ± 5 | 26.3 ± 3.2 | 0.947 |
| Hypertension | 2 (22.2%) | 8 (72.7%) | 0.070 |
| Diabetes mellitus | 0 | 3 (27.3%) | 0.218 |
| COPD | 2 (22.2%) | 3 (27.3%) | 1.000 |
| Renal insufficiency | 2 (22.2%) | 4 (36.4%) | 0.642 |
| Peripheral vascular disease | 2 (22.2%) | 2 (18.2%) | 1.000 |
| CAD | 1 (11.1%) | 5 (45.5%) | 0.157 |
| LV EF (%) | 54.3 ± 14.2 | 58.1 ± 11.8 | 0.526 |
| NYHA I. | 3 (33.3%) | 0 | 0.074 |
| II. | 0 | 1 (9.1%) | |
| III. | 6 (66.6%) | 7 (63.6%) | |
| IV. | 0 | 3 (27.3%) |
BMI, body mass index; COPD, chronic obstructive pulmonary disease; CAD, coronary artery disease; LV EF, left ventricular ejection fraction; NYHA, New York Heart Association heart failure classification.
Table 2.
Patient´s baseline biochemical parameters and outcome data.
| Responders, n = 9 | Non-responders, n = 11 | P-value | |
|---|---|---|---|
| Methylene blue dose (mg/kg) | 1.3 ± 0.5 | 1.3 ± 0.4 | 0.979 |
| Low dose corticosteroids | 4 (44.4%) | 3 (27.3%) | 0.642 |
| Vasopressin | 9 (100%) | 10 (90.9%) | 1.000 |
| Inotropes | |||
| - dobutamine | 2 (22.2%) | 3 (27.3%) | 0.795 |
| - milrinone | 0 | 1 (9.1%) | 0.918 |
| - levosimendan | 0 | 1 (9.1%) | 0.918 |
| Baseline biochemical parameters | |||
| - arterial lactate (mmol/L) | 8.6 ± 5.6 | 11.4 ± 7.7 | 0.386 |
| - arterial pH | 7.26 ± 0.11 | 7.16 ± 0.10 | 0.037* |
| - base deficit (mmol/L) | 5.5 ± 5.1 | 12.9 ± 3.2 | 0.001* |
| - ScvO2 (%) | 68.6 ± 11.8 | 67.8 ± 13 | 0.821 |
| - P(v-a)CO2 gap (mmHg) | 7.84 ± 2.2 | 7.62 ± 2.42 | 0.842 |
| - P(v-a)CO2/C(a-v)O2 (mmHg/mL) | 1.79 ± 0.73 | 3.24 ± 1.18 | 0.007* |
| - ionized calcium (mmol/L) | 0.90 ± 0.05 | 0.94 ± 0.13 | 0.517 |
| SOFA score | 14.2 ± 3.0 | 14.9 ± 1.8 | 0.556 |
| APACHE II score | 31.3 ± 4.7 | 32.1 ± 3.9 | 0.703 |
| Mortality within 24 hours | 0 | 5 (45.5%) | 0.038* |
| ICU mortality | 4 (44.4%) | 11 (100%) | 0.008* |
| 30-day mortality | 4 (44.4%) | 11 (100%) | 0.008* |
| ICU length of stay (days) | 12.6 ± 11.9 | 2.5 ± 3.3 | 0.015* |
| Duration of mechanical ventilation (hours) | 135 ± 111 | 51 ± 64 | 0.047* |
| ARDS | 1 (11.1%) | 2 (18.2%) | 1.000 |
| AKI | 8 (88.9%) | 9 (81.8%) | 1.000 |
| CRRT | 2 (22.2%) | 2 (18.2%) | 1.000 |
| VA-ECMO | 2 (22.2%) | 0 | 0.189 |
ScvO2, central venous oxygen saturation; P(v-a) CO2 gap, venous-to-arterial carbon dioxide tension difference; P(v-a)CO2/C(a-v)O2, ratio of venous-to-arterial carbon dioxide tension to arterio-venous oxygen content; SOFA, Sequential organ failure assessment; APACHE II, Acute physiology and chronic health assessment; ICU, intensive care unit; ARDS, Adult respiratory distress syndrome; AKI, acute kidney injury; CRRT, continuous renal replacement therapy; VA-ECMO, veno-arterial extracorporeal membrane oxygenation.
*P < 0.05.
Baseline norepinephrine doses did not differ between the groups, however after MB administration responders had significantly lower requirements in comparison to non-responders during the following 12 hours (Fig. 1). Mean arterial pressures did not differ between the study groups in the monitored 12-hour period (Fig. 2).
Figure 1.
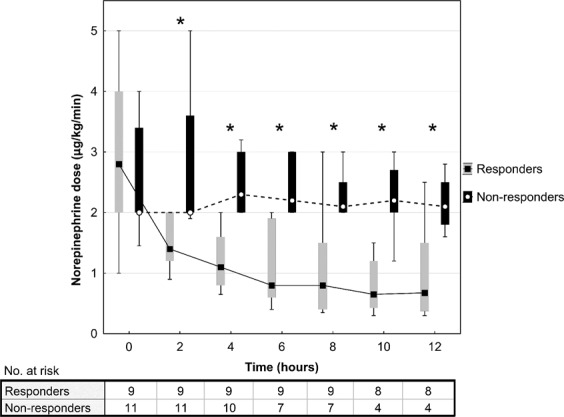
Norepinephrine requirements after methylene blue administration. Data are presented as box plot diagrams. The box represents the range of values (25th–75th percentile) with the horizontal line indicating the median, the whiskers depict adjacent values. *P < 0.05.
Figure 2.
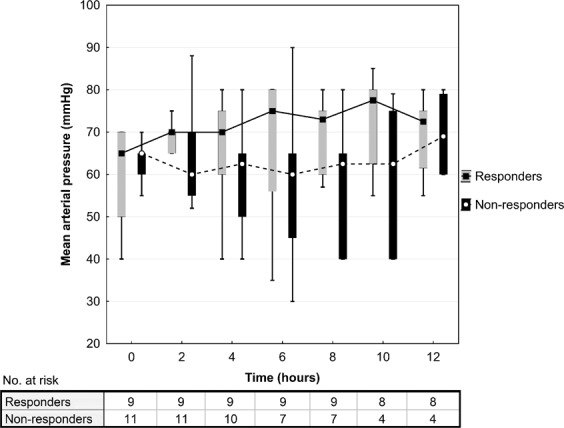
Mean arterial pressure after methylene blue administration. Data are presented as box plot diagrams. The box represents the range of values (25th–75th percentile) with the horizontal line indicating the median, the whiskers depict adjacent values.
There was no significant difference in the outcome data between the study groups (Table 2), except for the lower mortality within 24 hours from administration in the responders (0% vs. 45.5% in non-responders, P = 0.038) and lower ICU/30-day mortalities in the responders (both 44.4% vs. 100% in non-responders, P = 0.008). This was confirmed by Kaplan-Meier analysis that also showed a significant difference in 30-day survival between the study groups (P < 0.001) (Fig. 3). Due to this excessive 24-hour mortality in MB non-responders, responders had also significantly longer ICU stay (12.6 ± 11.9 vs. 2.5 ± 3.3 days respectively, P = 0.015) and duration of mechanical ventilation (135 ± 111 vs. 51 ± 64 hours respectively, P = 0.047) compared to non-responders.
Figure 3.
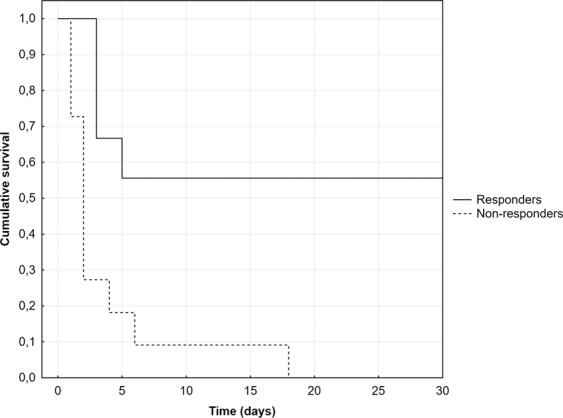
30-day survival: comparison between the methylene blue responders and non-responders.
In biochemical data analysis, there was a significantly higher baseline pH (7.26 ± 0.11 vs. 7.16 ± 0.10 respectively, P = 0.037) and lower BD (5.5 ± 5.1 vs. 12.9 ± 3.2 respectively, P = 0.001) in responders compared to non-responders (Table 2). Also, responders had lower P(v-a)CO2/C(a-v)O2 compared to non-responders (1.79 ± 0.73 vs. 3.24 ± 1.18 respectively, P = 0.007). Arterial lactate levels differed significantly between the responders and non-responders at 9 hours (4.2 ± 3.3 vs. 10.2 ± 1.5 mmol/L respectively, P = 0.007) and at 12 hours (3.4 ± 2.7 vs. 9.9 ± 2.2 mmol/L respectively, P = 0.002) after MB administration (Fig. 4). Ionized calcium concentrations did not differ between the study groups at any time point during the post-intervention 12-hour period (Fig. 5).
Figure 4.
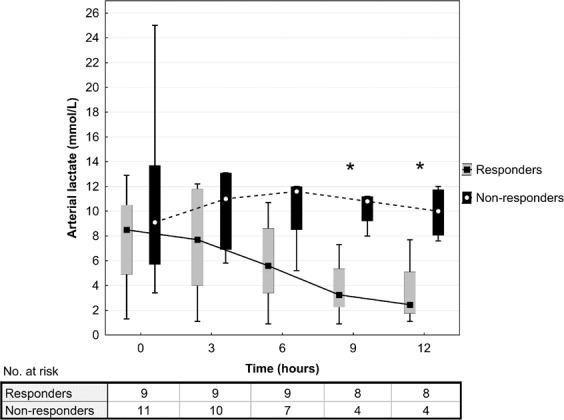
Arterial lactate concentrations after methylene blue administration. Data are presented as box plot diagrams. The box represents the range of values (25th–75th percentile) with the horizontal line indicating the median, the whiskers depict adjacent values. *P < 0.05.
Figure 5.
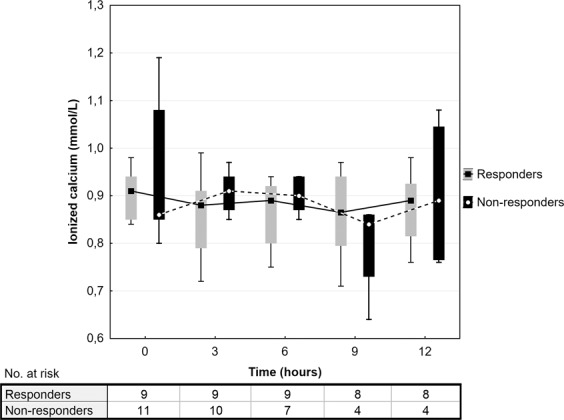
Ionized calcium concentrations after methylene blue administration. Data are presented as box plot diagrams. The box represents the range of values (25th–75th percentile) with the horizontal line indicating the median, the whiskers depict adjacent values.
Discussion
Our study showed limited hemodynamic responsiveness to MB administration of 45% in patients with refractory distributive shock. Those who responded had significantly improved survival compared to those with no hemodynamic response. Also, non-responders were in a more profound state of tissue hypoxia documented by significant metabolic acidosis and higher P(v-a)CO2/C(a-v)O2 compared to responders.
Methylene blue has been increasingly used as a non-adrenergic vasopressor in refractory shock management in various clinical scenarios including post-CPB, septic or dihydropyridine induced refractory shock. Many experimental studies reported beneficial hemodynamic effect, however clinical evidence is based only on case reports, small observational studies, and a few controlled trials8. The effect on patients´ outcome and mortality remains uncertain due to the study small sample sizes, which was also confirmed by the recent meta-analysis4. The majority of the above-mentioned studies compared MB administration to placebo or no treatment. Only one study reported responsiveness rate of 55% in an established distributive shock9, which is similar to our data. The lack of MB responsiveness could be caused by several factors including MB dosing regimen, a time delay between the onset of refractory shock and MB administration and finally by the actual severity of the patient´s clinical condition at the time of intervention.
Despite multiple performed studies, the adequate dosing regimen of MB remains unclear. The currently recommended dosing of 1–2 mg/kg used in experimental and clinical studies was originally based on the doses used for methemoglobinaemia treatment8. Also, various dosing regimens including single bolus, repeated boluses or bolus followed by infusion have been reported. In our study, only single boluses of MB were used in all patients and the doses did not differ significantly between responders and non-responders (Table 2). Therefore, we speculate that there should be other factors determining the final hemodynamic responsiveness to MB treatment.
Another parameter that may influence the MB responsiveness could be a time delay between the onset of refractory shock and MB administration. A time factor represents a key element in the concept of septic shock management as early interventions improve patients´ outcome10. This is supported by the recent study, in which vasoplegic cardiac surgical patients with early peroperative MB administration had improved outcome including mortality compared to the patients with late postoperative intervention11. This is in contrast with our study, in which the time from the onset of refractory shock (defined as norepinephrine dose more than 0.5 ug/kg/min) did not differ between the study groups (12.7 ± 2.8 hours in responders vs. 10.6 ± 6.8 hours in non-responders, P = NS). Thus, the time factor alone had no impact on the final MB responsiveness and the patient´s outcome.
Furthermore, global tissue hypoxia in shock states is the major trigger for the development of multiple organ dysfunction syndrome12. The commonly used indicators of tissue hypoxia including arterial lactate and central venous oxygen saturation may be unreliable in specific clinical situations. For instance, postoperative hyperlactatemia in cardiac surgery may not reflect only the presence of tissue hypoxia but can also represent a “wash-out” lactate after prolonged CPB, myocardial ischemia-reperfusion injury, or be the result of uncorrected hyperglycemia13. Therefore, novel parameters of tissue hypoxia including P(v-a)CO2 gap and P(v-a)CO2/C(a-v)O2 have been increasingly used in clinical practice. P(v-a)CO2 gap reflects flow-dependent CO2 removal from the microcirculation and represents a marker of global tissue hypoperfusion14. On the contrary, P(v-a)CO2/C(a-v)O2 is a surrogate of the respiratory quotient and is considered to be a marker of global tissue hypoxia and anaerobic metabolism15. In our study, MB non-responders had significantly increased.
P(v-a)CO2/C(a-v)O2 compared to responders (3.24 ± 0.11 vs. 1.79 ± 0.73 mmHg/mL respectively, P = 0.007) suggesting a deeper state of tissue hypoxia with higher degree of anaerobic metabolism. In the previous studies, high P(v-a)CO2/C(a-v)O2 has been associated with poor lactate clearance16 and with the condition of oxygen supply-dependency15, which are both associated with poor clinical outcome. This is in agreement with our study, where MB non-responders had significantly higher ICU mortality (100% vs. 44.4% in responders, P = 0.008) and lack of lactate clearance (Fig. 4) compared to responders. Additionally, MB non-responders were in more profound metabolic acidosis compared to responders (Table 2). Acidosis itself potentiates vasopressor hypo-responsiveness in refractory shock by activating ATP-sensitive potassium channels in vascular myocytes. This disables an increase of intracellular calcium and leads to further vasodilation17.
Finally, it has been reported that a low concentration of ionized calcium is common in patients with septic shock and its severity correlates with patient mortality18. Another study suggested that the correction of hypocalcaemia in critically ill patients reduced the doses of inotropes and vasopressors by improving myocardial contractility and vascular tonus19. However, recent human studies did not confirm these findings20 and even higher mortality was reported when calcium was supplemented in an animal experiment21. In our study, ionized calcium levels did not differ between MB responders and non-responders, even though they were mildly lowered in both groups with median values between 0.8–0.9 mmol/L during 12-hour post-intervention interval (Fig. 5). Therefore, we also conclude that ionized calcium concentration did not have a significant effect on MB responsiveness.
Based on our data, we assume that the patient´s responsiveness to MB may depend on the degree of actual tissue hypoxia and anaerobic metabolism. The mortality benefit seen in MB responders may likely be related to the lesser degree of tissue hypoxia rather than to the sole effect of MB and the decrease of vasopressor requirements. Thus, MB responsiveness could only be an indicator of the patient´s better clinical condition with the absence of irreversible hypoxic damage in the tissues. Nevertheless, we suggest that MB should be used as a rescue treatment of refractory distributive shock in patients who do not respond to standard treatment of volume expansion and catecholamine support. We also propose that MB should be administered early in the course of shock state before the profound tissue ischemic changes develop and vasoplegic syndrome progresses to its refractory phase. Further randomized controlled trials are required to elucidate the role of severity of tissue hypoxia on MB responsiveness in refractory shock, adequate MB dosing, MB effectiveness in different types of distributive shock and finally its effect on overall mortality of critically ill patients.
Methylene blue has a favorable safety profile. Minor side effects represent shortness of breath, nausea, vomiting, and discoloration of body fluids. Serious inadvertent effects including acute hemolytic anaemia22, detrimental effect on pulmonary functions23 and potentially fatal serotonin syndrome24 were reported only when high doses of MB were used. Except for the blue discoloration of body skin and urine, we have not detected any of these complications, probably due to the fact that the MB dose in our study was relatively low (1.3 mg/kg in both groups) and all patients were intubated and mechanically ventilated.
Our study has some limitations. First, the major one represents its retrospective design and the small number of patients, especially in interpreting outcome and mortality data. Second, due to the variable methods of hemodynamic monitoring including three different modalities we do not have sufficient data to demonstrate the effect of MB administration on the cardiac index and systemic vascular resistance. Third, the majority of patients in both study groups were classified as post-CPB SIRS shock. It is generally accepted that different pathogenetic mechanisms are involved in different types of vasodilatory shock to a variable extent25. Besides the inhibition of the nitric oxide-cyclic guanosine monophosphate pathway, the most significant factors include proinflammatory cytokines-induced impairment of endothelium-dependent dilatation, inhibition of vasodilator antiplatelet prostanoid26, activation of ATP-sensitive potassium channels and vasopressin deficiency25. Therefore, the hemodynamic response to MB may be variable in different types of distributive shock, which our study did not fully cover. Additionally, a relationship to prostaglandin inhibition may also associate with potentiation of coagulation in septic shock with a negative impact on capillary perfusion and patient´s outcome27. Fourth, the doses of norepinephrine used were extremely high in both study groups often exceeding the dose of 2 µg/kg/min. The pharmacodynamic effect of norepinephrine is based on the existence of the threshold concentration of 1000 pg/mL with a subsequent linear increase in effect as a function of the logarithm of concentrations17. The maximum effect is reached at the doses of 100 to 1000 times the threshold concentration and differs from one patient to another17. Thus, such extreme norepinephrine doses may be ineffective in some patients and the positive effect of MB could be then partially confounded by just simply lowering ineffectively high doses of norepinephrine.
In conclusion, methylene blue represents a non-adrenergic vasopressor with limited effectiveness in patients with refractory distributive shock. The development of profound tissue hypoxia and a high degree of anaerobic metabolism was associated with the loss of hemodynamic responsiveness to its administration.
Acknowledgements
Supported by MZ CZ - DRO-VFN64165.
Author contributions
M.P. participated in the design and coordination of the study, data collection and manuscript writing. P.K. participated in the data collection. H.D. participated in the data collection. J.K. participated in the study design, data collection and helped to draft the manuscript. M.L. participated in the data collection and created the figures. P.M. helped to draft the manuscript. M.B. performed statistical analysis. All authors read and approved the final manuscript.
Data availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.De Backer D, et al. SOAP II Investigators. Comparison of dopamine and norepinephrine in the treatment of shock. N. Engl. J. Med. 2010;362(9):779–89. doi: 10.1056/NEJMoa0907118. [DOI] [PubMed] [Google Scholar]
- 2.Bassi, E., Park, M., Azevedo, L. C. Therapeutic strategies for high-dose vasopressor-dependent shock. Crit. Care. Res. Pract. 654708 (2013). [DOI] [PMC free article] [PubMed]
- 3.Jenkins CR, Gomersall CD, Leung P, Joynt GM. Outcome of patients receiving high dose vasopressor therapy: a retrospective cohort study. Anaesth. Intensive Care. 2009;37(2):286–9. doi: 10.1177/0310057X0903700212. [DOI] [PubMed] [Google Scholar]
- 4.Belletti A, et al. Non-Adrenergic Vasopressors in Patients with or at Risk for Vasodilatory Shock. A Systematic Review and Meta-Analysis of Randomized Trials. PLoS One. 2015;10(11):e0142605. doi: 10.1371/journal.pone.0142605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Khanna A, et al. Angiotensin II for the Treatment of Vasodilatory Shock. N. Engl. J. Med. 2017;377(5):419–430. doi: 10.1056/NEJMoa1704154. [DOI] [PubMed] [Google Scholar]
- 6.Sacha GL, et al. Predictors of response to fixed-dose vasopressin in adult patients with septic shock. Ann. Intensive Care. 2018;8(1):35. doi: 10.1186/s13613-018-0379-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Avni T, et al. Vasopressors for the Treatment of Septic Shock: Systematic Review and Meta-Analysis. PLoS One. 2015;10(8):e0129305. doi: 10.1371/journal.pone.0129305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jang DH, Nelson LS, Hoffman RS. Methylene blue for distributive shock: a potential new use of an old antidote. J. Med. Toxicol. 2013;9(3):242–9. doi: 10.1007/s13181-013-0298-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Park BK, et al. The effects of methylene blue on hemodynamic parameters and cytokine levels in refractory septic shock. Korean J. Intern. Med. 2005;20(2):123–8. doi: 10.3904/kjim.2005.20.2.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rhodes A, et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Intensive Care Med. 2017;43(3):304–377. doi: 10.1007/s00134-017-4683-6. [DOI] [PubMed] [Google Scholar]
- 11.Mehaffey JH, et al. Methylene Blue for Vasoplegic Syndrome After Cardiac Operation: Early Administration Improves Survival. Ann. Thorac. Surg. 2017;104(1):36–41. doi: 10.1016/j.athoracsur.2017.02.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Huang YC. Monitoring oxygen delivery in the critically ill. Chest. 2005;128(5 Suppl 2):554S–560S. doi: 10.1378/chest.128.5_suppl_2.554S. [DOI] [PubMed] [Google Scholar]
- 13.O’Connor E, Fraser JF. The interpretation of perioperative lactate abnormalities in patients undergoing cardiac surgery. Anaesth. Intensive Care. 2012;40(4):598–603. doi: 10.1177/0310057X1204000404. [DOI] [PubMed] [Google Scholar]
- 14.Mallat J, Vallet B. Difference in venous-arterial carbon dioxide in septic shock. Minerva Anestesiol. 2015;81(4):419–25. [PubMed] [Google Scholar]
- 15.Mallat J, et al. Ratios of central venous-to-arterial carbon dioxide content or tension to arteriovenous oxygen content are better markers of global anaerobic metabolism than lactate in septic shock patients. Ann. Intensive Care. 2016;6(1):10. doi: 10.1186/s13613-016-0110-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.He HW, Liu DW, Long Y, Wang XT. High central venous-to-arterial CO2 difference/arterial-central venous O2 difference ratio is associated with poor lactate clearance in septic patients after resuscitation. J. Crit. Care. 2016;31(1):76–81. doi: 10.1016/j.jcrc.2015.10.017. [DOI] [PubMed] [Google Scholar]
- 17.Levy B, et al. Vascular hyporesponsiveness to vasopressors in septic shock: from bench to bedside. Intensive Care Med. 2010;36(12):2019–29. doi: 10.1007/s00134-010-2045-8. [DOI] [PubMed] [Google Scholar]
- 18.Vadstrup S, et al. Correlation between severity of septic conditions and circulating levels of ionized calcium. Intensive Care Med. 1989;15(5):329–30. [PubMed] [Google Scholar]
- 19.Alegre M, Vincent JL. Dopamine dependence in hypocalcemic patients. Intensive Care Med. 1990;16(7):463–5. doi: 10.1007/BF01711228. [DOI] [PubMed] [Google Scholar]
- 20.Forsythe RM, et al. Parenteral calcium for intensive care unit patients. Cochrane Database Syst Rev. 2008;8(4):CD006163. doi: 10.1002/14651858.CD006163.pub2. [DOI] [PubMed] [Google Scholar]
- 21.Zaloga GP, et al. Low dose calcium administration increases mortality during septic peritonitis in rats. Circ Shock. 1992;37(3):226–9. [PubMed] [Google Scholar]
- 22.Goluboff N, Wheaton R. Methylene blue induced cyanosis and acute hemolytic anemia complicating the treatment of methemoglobinemia. J. Pediatr. 1961;58:86–9. doi: 10.1016/S0022-3476(61)80064-4. [DOI] [PubMed] [Google Scholar]
- 23.Weingartner R, et al. Blockade of the action of nitric oxide in human septic shock increases systemic vascular resistance and has detrimental effects on pulmonary function after a short infusion of methylene blue. Braz. J. Med. Biol. Res. 1999;32(12):1505–13. doi: 10.1590/S0100-879X1999001200009. [DOI] [PubMed] [Google Scholar]
- 24.Héritier Barras AC, Walder B, Seeck M. Serotonin syndrome following Methylene Blue infusion: a rare complication of antidepressant therapy. J. Neurol. Neurosurg. Psychiatry. 2010;81(12):1412–3. doi: 10.1136/jnnp.2009.172221. [DOI] [PubMed] [Google Scholar]
- 25.Landry DW, Oliver JA. The pathogenesis of vasodilatory shock. N Engl J Med. 2001;23;345(8):588–95. doi: 10.1056/NEJMra002709. [DOI] [PubMed] [Google Scholar]
- 26.Laffey JG, Boylan JF, Cheng DC. The systemic inflammatory response to cardiac surgery: implications for the anesthesiologist. Anesthesiology. 2002;97(1):215–52. doi: 10.1097/00000542-200207000-00030. [DOI] [PubMed] [Google Scholar]
- 27.Hingorani AD, et al. Acute systemic inflammation impairs endothelium-dependent dilatation in humans. Circulation. 2000;102:994–9. doi: 10.1161/01.CIR.102.9.994. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.


