Abstract
The emergence of livestock-associated (LA)-methicillin-resistant Staphylococcus aureus (MRSA) in livestock animal has become a significant zoonotic concern. In the present study, we investigated nationwide prevalence of LA-MRSA across pork production chain including pig farms, slaughterhouses, and retail markets. A total of 40 MRSA strains were isolated during the investigation and the overall prevalence of MRSA was 3.4% (n = 37), 0.6% (n = 2), and 0.4% (n = 1) in pig farms, slaughterhouses, and retail markets, respectively. Multilocus sequence typing analyses revealed that the 2 most significant clonal lineages in pork production chain in Korea were ST398 (n = 25) and ST541 (n = 6). All of the 40 MRSA isolates were further characterized to investigate key genotypic and phenotypic correlates associated with the emergence and spread of clonal complex 398 (CC398; ST398, and ST541) LA-MRSA. Although the prevalence of swine-associated MRSA was still relatively low and mostly restricted to pig farms, multidrug-resistant CC398 LA-MRSA isolates with new spa types (t18102 and t18103) were identified as a major clonal lineage. The CC398 LA-MRSA strains tended to exhibit increased levels of multiple drug resistance (MDR) phenotype compared with non-CC398 MRSA strains. Of note, in comparison with non-CC398 MRSA isolates, CC398 LA-MRSA isolates exhibited significantly enhanced tetracycline (TET) and zinc resistance. These findings suggested that co-selection pressure associated with MDR phenotype, especially TET resistance, and zinc resistance may have played a significant role in the emergence and persistence of CC398 LA-MRSA in pig farms in Korea.
Keywords: MRSA, swine, CC398, antimicrobial resistance, zinc resistance
INTRODUCTION
Methicillin-resistant Staphylococcus aureus (MRSA) strains adapted to humans have traditionally been recognized as hospital-associated (HA)-MRSA and community-associated (CA)-MRSA [1,2,3]. In addition to HA- and CA-MRSA infections, the incidence of livestock-associated (LA)-MRSA infections has also been increasing in various animal species, especially in pigs in European countries and North America [4,5,6]. Distinct clonal lineages of MRSA have been identified depending on geographical regions. Although sequence type (ST) 398 LA-MRSA has been detected in cattle, horses, sheep, goats, poultry, and even in companion animals, pigs seem to be the major reservoir animal for ST398 MRSA [7,8]. More importantly, ST398 LA-MRSA has also been found in persons who are in direct/indirect contact with MRSA-colonized pigs or foods of animal origin [5,9].
Although there is still limited information on the prevalence of LA-MRSA infections, recent studies have indicated that the most frequent MRSA clones isolated from pork or pigs in Korea are ST398 and ST541 (CC398), followed by ST72 [10,11,12]. It has been indicated that the increase of LA-MRSA (ST398 and ST541) in pigs seem to be associated with the use of antibiotics, especially tetracycline (TET) compounds [5,13]. In addition to TET resistance, zinc resistance among LA-MRSA isolates is thought to be associated with prevalence of ST398 MRSA [14,15]. Thus, the current investigation was designed to assess the national prevalence of MRSA in the pork production chain including pigs, pork meat, workers, and environment of the facilities in pig farms, slaughterhouses, and retail markets. In addition, to investigate epidemiological correlation among the LA-MRSA isolates, multilocus ST (MLST), staphylococcal cassette chromosome mec (SCCmec), spa, agr, and staphylococcal enterotoxin (SE) types were determined. Moreover, to identify potential mechanisms/factors involved in the frequent emergence of LA type of MRSA (CC398; ST398, and ST541) in pig farms, resistance to zinc chloride and TET was examined, and genes that confer the resistant phenotypes (czrC; tetK, tetL, tetM, tetO, and tetS) were identified.
MATERIALS AND METHODS
Collection of samples
A total of 1,587 non-duplicate swab samples or pork meat samples were collected from 19 pig farms (n = 1,009), 7 slaughterhouses (n = 311), and 35 retail markets (n = 267) in 8 provinces in Korea between July 2017 and August 2018. The pig farms, slaughterhouses, and retail markets were selected at random in each of 8 provinces. Pig farm samples were collected from at least 10 different finishing pigs (nasal, skin, rectal, and fecal swabs from each pig) and different farm environment (sewage, floor, fence, and ventilations). Slaughterhouse samples were collected from pig carcasses within 6 h of slaughter. For retail meat samples, 2 to 3 fresh pork meat samples of each type were collected from retail outlets and local markets based on their regional distribution in each province. A total of 152 swab samples were collected from 39 workers in pig farms (n = 34), slaughterhouses (n = 4), or retail markets (n = 1) after a written consent was obtained from each participant. All swab samples and meat samples were stored under 4°C in ice-cooled containers and transported to the laboratory for isolation of MRSA within 24 h of sampling.
Isolation of MRSA
All swab samples were inoculated into 5 mL of Tryptic Soy Broth (TSB; Difco Laboratories, USA) supplemented with 10% NaCl for 24 h at 37°C. Pork meat samples (25 g) were homogenized in the 225 mL of 10% NaCl-TSB and incubated at 37°C. After 24-h incubation, 10 µL aliquots of the pre-enriched media were streaked onto both chromogenic MRSA agar (chromID MRSA SMART agar; bioMérieux, France) and incubated for 16–18 h at 37°C. Up to 3 suspected MRSA colonies from each sample were selected and subcultured on Baired-Parker agar (Difco Laboratories) for further identification. All S. aureus strains were identified by 16S ribosomal RNA sequencing (Cosmogenetech, Korea) and Vitek 2 system (bioMérieux). Coagulase production was confirmed as described in previous study [16]. Two or more S. aureus strains recovered from the same animal or person were considered separate strains when further phenotypic or genotypic analyses revealed different profiles.
Antimicrobial susceptibility assays
Antimicrobial susceptibility tests were performed using the disc diffusion methods according to the 2017 Clinical and Laboratory Standards Institute guidelines [17]. The antimicrobial agents used were ampicillin (AMP; 10 μg), chloramphenicol (30 μg), clindamycin (2 μg), erythromycin (15 μg), cefoxitin (CEF; 30 μg), gentamicin (30 μg), rifampin (5 μg), sulfamethoxazole-trimethoprim (23.73 + 1.25 μg), quinupristin-dalfopristin (15 μg), TET (30 μg), ciprofloxacin (5 μg), mupirocin (200 μg Oxoid, UK). Minimum inhibitory concentrations (MICs) to oxacillin, vancomycin, teicoplanin, tigecycline, and TET were determined by using standard E-test (Bio Mérieux, France) on Meuller-Hinton agar plates according to manufacturer's recommendation. S. aureus ATCC 29213 and S. aureus MW2 strains was used as a reference strain for the antimicrobial susceptibility assays. All the antimicrobial susceptibility assays were performed 3 times.
Detection of mecA and SCCmec typing
Genomic DNA samples were prepared from S. aureus isolates using the previously described method [18].
The presence of the mecA gene was determined in all MRSA isolates as described by Vannuffel et al. [19]. SCCmec types were determined by the multiplex polymerase chain reaction (PCR) method as described previously [20] based on combinations of the ccr and mec regions.
Molecular typing and sequencing
MLST was performed on all confirmed MRSA isolates as described previously [21]. Briefly, fragments of 7 housekeeping genes (arcC, aroE, glpF, gmk, pta, tpi, and yqiL) were PCR-amplified and sequenced. Then, the alleles and STs were assigned according to the S. aureus MLST database (http://pubmlst.org/saureus/).
The spa is a surface protein that plays an important role in the avoidance of host immune responses by binding to Fc portion of immunoglobulins [22]. The spa repeat regions were amplified using a specific primer set [23], sequenced, and the spa type was determined based on the variable number tandem repeats in the SpaServer database (http://spa.ridom.de/).
The agr operon is a critical regulatory component involved in the control of virulence factors in S. aureus. The agr types (I–IV) of S. aureus isolates were determined by multiplex PCR assays as described by Gilot et al. [24].
Sequencing of all DNA samples was performed at Cosmo Genetech (Korea).
Detection of virulence genes and SEs
The genes for Panton-Valentine leukocidin (PVL; lukF and lukS), toxic shock syndrome toxin-1 (TSST-1), and exfoliative toxins (ETs; eta, etb, and etd) were detected using PCR-based assays as previously described [25,26,27].
The presence of SE genes (sea, seb, sec, sed, and see) in S. aureus isolates was examined by PCR as described before [28].
Detection of TET and zinc resistance genes
The presence of TET resistance genes (tetK, tetL, tetM, tetO, and tetS) and zinc resistance gene (czrC) was determined by PCR-based assays using specific primer sets as described previously [29,30].
Zinc chloride susceptibility assays
The MICs to zinc chloride were determined by the standardized agar dilution assay in Mueller-Hinton agar (MHA) as described previously [29]. Briefly, MHA was adjusted to pH5.5 and then supplemented with zinc chloride in 2-fold dilutions with concentrations ranging from 0.25 to 16 mM to encompass low-to-high resistance levels. Zinc chloride resistance was set as an MIC of > 2 mM based on previous publications [31,32]. A minimum of 3 experiments was performed for each isolate on separate days.
Statistical analysis
All quantitative data were analyzed by Mann-Whitney U test using GraphPad Prism5 software (GraphPad Software, USA; www.Graph Pad.com). A p values of < 0.05 was considered significant.
RESULTS
Prevalence of MRSA in pig farms, slaughterhouses, and retail outlets
A total of 40 MRSA strains (2.4%) were isolated from 1,657 samples taken from pig farms, slaughterhouses, and retail markets during the study period (2017–2018). The prevalence of MRSA was highest in pig farms (3.4%), followed by slaughterhouses (0.6%) and retail markets (0.4%) (Table 1). Among the 40 MRSA strains, 37 MRSA strains (92.5%) were isolated from pig farms in the Chungcheong, Jeolla, Gyeongsang provinces, while only 2 (5%) and 1 (2.5%) of the MRSA strains were isolated from slaughterhouses in Jeolla and retail markets in Gangwon, respectively.
Table 1. Prevalence of MRSA isolates in pig farms, slaughterhouses, and retail markets in Korea.
| Sample origin | Provinces | Total | |||||
|---|---|---|---|---|---|---|---|
| Gyeonggi | Gangwon | Chungcheong | Jeolla | Gyeongsang | |||
| Farms | 0/178 | 0/160 | 11/228 (4.8) | 17/259 (6.6) | 9/254 (3.5) | 37/1,079 (3.4) | |
| Pigs | 0/120 | 0/120 | 7/160 (4.3) | 13/200 (6.5) | 8/200 (4.0) | 28/800 (3.5) | |
| Workers | 0/40 | 0/32 | 4/44 (9.0) | 1/29 (3.4) | 0/24 | 5/169 (3.0) | |
| Environment | 0/18 | 0/18 | 0/24 | 3/30 (10.0) | 1/30 (3.3) | 4/120 (3.3) | |
| Slaughterhouses | 0/40 | 0/43 | 0/43 | 2/142 (1.4) | 0/43 | 2/311 (0.6) | |
| Carcass | 0/40 | 0/40 | 0/40 | 1/120 (0.8) | 0/40 | 1/280 (0.4) | |
| Workers | - | - | - | 1/13 (7.6) | - | 1/13 (7.6) | |
| Environment | - | 0/3 | 0/3 | 0/9 | 0/3 | 0/21 | |
| Retail markets | 0/65 | 1/42 (2.4) | 0/55 | 0/54 | 0/51 | 1/267 (0.4) | |
| Pork meats | 0/58 | 1/42 (2.4) | 0/55 | 0/54 | 0/51 | 1/260 (3.8) | |
| Workers | 0/4 | - | - | - | - | 0/4 | |
| Environment | 0/3 | - | - | - | - | 0/3 | |
| Total | 0/283 | 1/245 (0.4) | 11/326 (3.4) | 19/455 (4.2) | 9/348 (2.6) | 40/1,657 (2.4) | |
Data are shown as number of MRSA/total number of samples (% of MRSA isolation).
MRSA, methicillin-resistant Staphylococcus aureus.
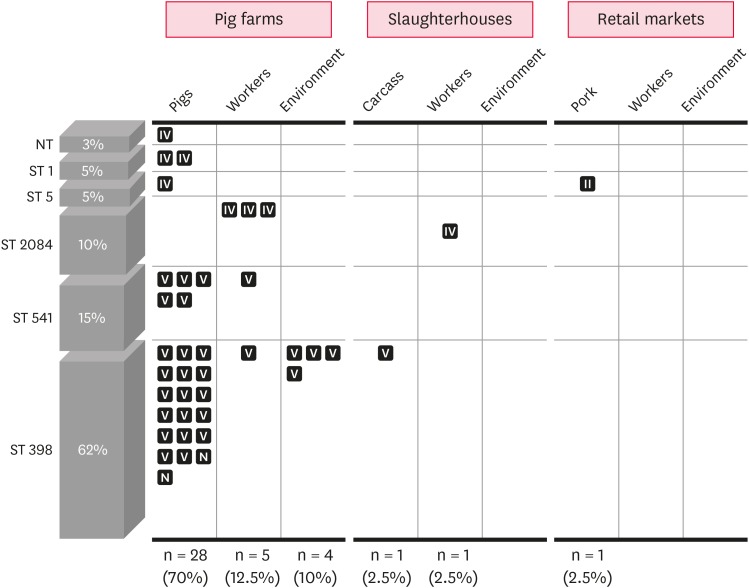
As shown in Fig. 1, 28/40 swine-associated MRSA strains were recovered from pigs, while 5 and 4 MRSA strains were isolated from the farm workers and the environment of pig farms, respectively.
Fig. 1. Prevalence and genetic profiles of MRSA isolates recovered from pig farms, slaughterhouses, and retail markets. Each square represents individual MRSA isolate and staphylococcal cassette chromosome mec type II, IV, V, or N is shown in the square.
MRSA, methicillin-resistant Staphylococcus aureus; NT, non-typeable for multilocus sequence typing; ST, sequence type; N, non-typeable.
Profiles of MLST, spa, SCCmec, and agr types
All 40 MRSA strains were subjected to sequencing of 7 housekeeping genes (arcC, aroE, glpF, gmk, pta, tpi, and yqiL) for MLST analyses. As shown in Table 2 and Fig. 1, MLST indicated a total of 5 STs (ST1, ST5, ST2084, ST541, and ST398) among the isolates, except for one isolate (for which spa type was t901). The 2 most significant clonal lineages among the 40 MRSA isolates were ST398 (n = 25) and ST541 (n = 6), accounting for 62.5% and 15% of the isolates, respectively. Interestingly, except for 2 non-typeable isolates from pigs, all ST398 and ST541 MRSA isolates harbored SCCmec type V. Although MRSA isolates with the major LA-MRSA lineage, CC398 (ST398 and ST541), were recovered from pig farms (pigs, farm workers, and environment within facilities) or carcass samples in slaughterhouses, none of the CC398 MRSA isolates were recovered from samples from retail markets. All other MRSA isolates with non-CC398 (ST1, ST5, and ST2084) genotype had SCCmec IV or SCCmec II.
Table 2. Genotypes, antimicrobial resistance profiles, virulence factors, and zinc chloride susceptibility of methicillin-resistant Staphylococcus aureus strains.
| Isolates | MLST | Origin | spa | SCCmec | agr | Places | Resistance phenotype | tet | TET MICs (µg/mL) | czrC | Zinc MICs (µg/mL) | ETs TSST PVL | SEs |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PKFA-1101 | ST398 | Pig | t18102 | V | I | F-1 | AMP, CHL, CEF, TET, CIP | tet(M), tet(K) | 64 | + | 10 | ||
| PKFA-181 | Pig | t18102 | V | I | F-1 | AMP, CHL, CEF, TET, CIP | tet(M), tet(K) | 64 | + | 10 | |||
| PKFA-191 | Pig | t18102 | V | I | F-1 | AMP, CHL, CEF, TET, CIP | tet(M), tet(K) | 64 | + | 10 | |||
| PKFA-111 | Pig | t18102 | V | I | F-1 | AMP, CHL, CEF, TET, CIP | tet(M), tet(K) | 64 | + | 10 | |||
| PKFA-171 | Pig | t18102 | V | I | F-1 | AMP, CHL, CEF, TET, CIP | tet(K) | 32 | + | 10 | |||
| PKFA-151 | Pig | t571 | V | I | F-1 | AMP, CHL, CEF, TET, CIP | tet(M), tet(K) | 64 | + | 10 | |||
| PJFA-3101 | Pig | t571 | NT | I | F-2 | AMP, CHL, CLI, ERY, CEF, GEN, SYN, TET, CIP | tet(M), tet(K) | 32 | + | 10 | |||
| PJFA-561 | Pig | t18103 | V | I | F-3 | AMP, CHL, CLI, ERY, CEF, GEN, SYN, TET, CIP | tet(M), tet(K) | 64 | + | 4 | sea | ||
| PJFA-511 | Pig | t18103 | V | I | F-3 | AMP, CHL, CLI, ERY, CEF, GEN, SYN, TET, CIP | tet(M), tet(L) | 64 | + | 4 | |||
| PJFA-521M | Pig | t18103 | V | I | F-3 | AMP, CHL, CLI, ERY, CEF, GEN, SYN, TET, CIP | tet(M), tet(L) | 64 | + | 4 | |||
| PJFA-531M | Pig | t18103 | V | I | F-3 | AMP, CHL, CLI, ERY, CEF, GEN, SYN, TET, CIP | tet(M), tet(L) | 64 | − | 4 | |||
| PJFA-541M | Pig | t18103 | V | I | F-3 | AMP, CHL, CLI, ERY, CEF, GEN, SYN, TET, CIP | tet(M), tet(L) | 128 | + | 4 | etb | ||
| PJFA-551M | Pig | t18103 | V | I | F-3 | AMP, CHL, CLI, ERY, CEF, GEN, SYN, TET, CIP | tet(M), tet(L) | 64 | + | 4 | etb | ||
| PJFA-581M | Pig | t18103 | V | I | F-3 | AMP, CHL, CLI, ERY, CEF, GEN, SYN, TET, CIP | tet(M), tet(L) | 64 | + | 4 | |||
| PJFA-591M | Pig | t18103 | V | I | F-3 | AMP, CHL, CLI, ERY, CEF, GEN, SYN, TET, CIP | tet(M), tet(L) | 128 | − | 4 | etb | ||
| PJFA-5101M | Pig | t18103 | V | I | F-3 | AMP, CHL, CLI, ERY, CEF, GEN, SYN, TET, CIP | tet(M), tet(L) | 64 | + | 4 | |||
| PKFA-163 | Pig | t571 | NT | I | F-1 | AMP, CHL, CLI, ERY, CEF, GEN, TET, CIP | tet(M), tet(L) | 32 | + | 12 | |||
| PJFA-573 | Pig | t18103 | V | I | F-3 | AMP, CHL, CLI, ERY, CEF, GEN, SYN, TET, CIP | tet(M), tet(K) | 64 | + | 6 | sea | ||
| PKFA-124 | Pig | t18102 | V | I | F-1 | AMP, CHL, CEF, TET, CIP | tet(M), tet(K) | 64 | + | 10 | |||
| PJFH-522M | Worker | t18103 | V | I | F-3 | AMP, CHL, CLI, ERY, CEF, GEN, TET, CIP | tet(M), tet(L) | 64 | − | 4 | |||
| PKFE-102-1 | Environ. | t18102 | V | I | F-1 | AMP, CHL, CEF, TET, CIP | tet(M), tet(K) | 64 | + | 10 | |||
| PJFE-502 | Environ. | t18103 | V | I | F-3 | AMP, CHL, CLI, ERY, CEF, GEN, SYN, TET, CIP | tet(M), tet(L) | 64 | − | 4 | |||
| PJFE-503 | Environ. | t18103 | V | I | F-3 | AMP, CHL, CLI, ERY, CEF, GEN, SYN, TET, CIP | tet(M), tet(L) | 128 | + | 4 | |||
| PJFE-504 | Environ. | t18103 | V | I | F-3 | AMP, CHL, CLI, ERY, CEF, GEN, SYN, TET, CIP | tet(M), tet(L) | 128 | + | 4 | |||
| PJSM-251E | Carcass | t571 | V | I | S-1 | AMP, CHL, CEF, TET, CIP | tet(M), tet(K) | 64 | + | 10 | sec, sed | ||
| PCFA-221 | ST541 | Pig | t034 | V | I | F-4 | AMP, CLI, ERY, CEF, TET | tet(K) | 64 | + | 6 | ||
| PCFA-231 | Pig | t034 | V | I | F-4 | AMP, CLI, ERY, CEF, TET | tet(K) | 16 | − | 4 | |||
| PCFA-263 | Pig | t034 | V | I | F-4 | AMP, CLI, ERY, CEF, TET | tet(K) | 64 | + | 6 | |||
| PCFA-254 | Pig | t034 | V | I | F-4 | AMP, CLI, ERY, CEF, TET | tet(K) | 16 | + | 6 | |||
| PCFA-244 | Pig | t034 | V | I | F-4 | AMP, CLI, ERY, CEF, TET | tet(M), tet(K), tet(L) | 16 | + | 6 | |||
| PCFH-226 | Worker | t034 | V | I | F-4 | AMP, CLI, ERY, CEF, TET | tet(M), tet(K), tet(L) | 64 | + | 6 | |||
| PCFH-321 | ST2084 | Worker | t664 | IV | I | F-5 | AMP, ERY, CEF | < 0.25 | − | 4 | |||
| PCFH-322 | Worker | t5440 | IV | I | F-5 | AMP, ERY, CEF | < 0.25 | − | 4 | ||||
| PCFH-326 | Worker | t664 | IV | I | F-5 | AMP, ERY, CEF, STX | < 0.25 | − | 4 | ||||
| PJSH-331E | Worker | t5440 | IV | I | S-2 | AMP, CEF | 0.5 | − | 4 | sed | |||
| PCFA-3102 | ST5 | Pig | t9353 | IV | I | F-5 | AMP, CLI, ERY, CEF, TET, CIP, MUP | tet(M), tet(K) | 16 | − | 4 | tsst | |
| PGMM-711 | Pork | t2460 | II | II | R-1 | AMP, CLI, ERY, CEF, GEN, TET, CIP | tet(M) | 16 | − | 4 | tsst | ||
| PCFA-442 | ST1 | Pig | t128 | IV | III | F-12 | AMP, CEF | < 0.25 | + | 4 | pvl | sea | |
| PJFA-423 | Pig | t128 | IV | III | F-6 | AMP, CEF, GEN, STX, SYN, CIP | < 0.25 | + | 4 | pvl | sea, sec | ||
| PJFA-393 | NT | Pig | t901 | IV | I | F-2 | AMP, ERY, CEF | < 0.25 | + | 6 |
MLST, multilocus sequence typing; ST, sequence type; SCCmec, staphylococcal cassette chromosome mec; MIC, minimum inhibitory concentrations; ET, exfoliative toxin; TSST, toxic shock syndrome toxin; PVL, Panton-Valentine leukocidin; SE, staphylococcal enterotoxin; NT, non-typeable; F, farms; S, slaughterhouses; R, retail markets; AMP, ampicillin; CHL, chloramphenicol; CLI, clindamycin; ERY, erythromycin; CEF, cefoxitin; GEN, gentamicin; RIF, rifampicin; SXT, sulfamethoxazole-trimethoprim; SYN, quinupristin-dalfopristin; MUP, mupirocin; TET, tetracycline; CIP, ciprofloxacin.
In addition to the MLST and SCCmec types, sequence analyses of spa in the 40 MRSA isolates revealed ten different spa types: t18102, t18103, t571, t034, t664, t5440, t128, t9353, t901, and t2460. The majority of ST398 MRSA strains isolated from Gyeongsang and Jeolla provinces were t18102 and t18103 types, respectively (Table 2). All of the 6 ST541 MRSA strains isolated from Chungcheong province had spa type of t034. All MRSA isolates were assigned to agr type I, except for 2 ST1 and a ST5 isolates with agr type of III and II, respectively.
Based on the profiles of MLST, SCCmec, spa, and agr types, the most prevalent genotypes observed in these MRSA isolates were ST398-SCCmec V-t18102, ST398-SCCmec V-t18103, and ST541-SCCmec V-t034.
Antimicrobial resistance profiles and TET resistance genes
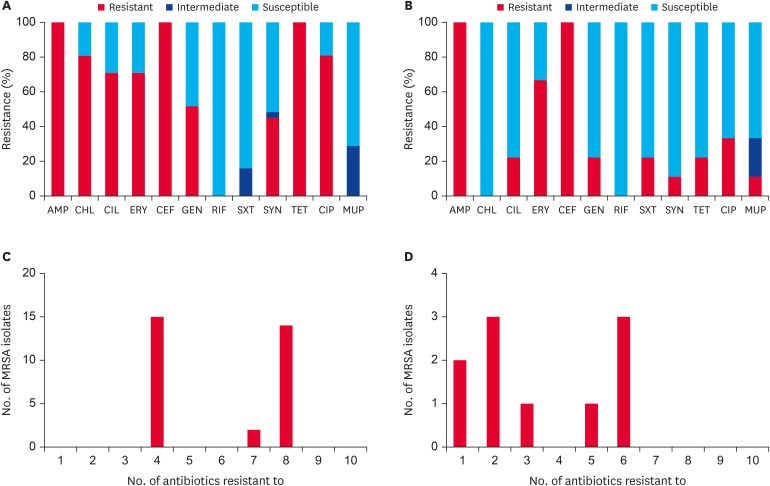
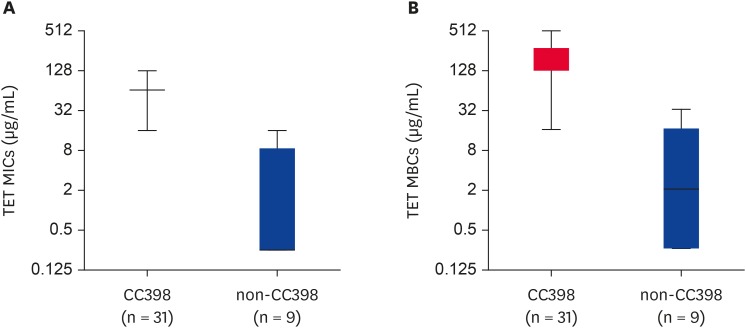
All 40 MRSA strains exhibited resistance to AMP and CEF (Table 2 and Fig. 2A and C), and they were all susceptible to rifampin, vancomycin, teicoplanin, and tigecycline (data not shown). Interestingly, all CC398 (ST398 and ST541) MRSA isolates displayed a multidrug resistance (MDR) phenotype (resistant to ≥ 3 antimicrobial subclasses) with 100% resistance to TET (Table 2). All these CC398 MRSA isolates carried 1 or more TET resistance genes (tetM, tetK, or tetL) (Table 2). None of the 40 MRSA isolates carried tetO and tetS for TET resistance phenotype. As shown in Fig. 3A and B, the TET MICs and minimum bactericidal concentrations (MBCs) were significantly higher for CC398 MRSA isolates than for non-CC398 MRSA isolates (p < 0.0001). Except for 2 ST5 MRSA isolates, which harbored tetM and/or tetK with TET MICs of 16 µg/mL, all non-CC398 MRSA isolates displayed TET MICs of ≤ 0.5 µg/mL.
Fig. 2. Antimicrobial susceptibility and frequency of MDR phenotype. Antimicrobial susceptibility of (A) CC398 and (B) non-CC398 MRSA isolates and frequency of a MDR phenotype among (C) CC398 and (D) non-CC398 MRSA isolates. Susceptibility assays were performed using the disc diffusion methods according to the 2017 Clinical and Laboratory Standards Institute guidelines [17].
MDR, multidrug resistance; MRSA, methicillin-resistant Staphylococcus aureus; AMP, ampicillin; CHL, chloramphenicol; CLI, clindamycin; ERY, erythromycin; CEF, cefoxitin; GEN, gentamicin; RIF, rifampicin; SXT, trimethoprim-sulfamethoxazole; SYN, quinupristin-dalfopristin; TET, tetracycline; CIP, ciprofloxacin; MUP, mupirocin.
Fig. 3. Box and Whisker plots of TET MICs (A) and MBCs (B) for CC398 and non-CC398 MRSA isolates. The median TET MICs for CC398 and non-CC398 MRSA isolates were 64.59 µg/mL and 3.778 µg/mL, respectively (p = 0.0001). The median TET MBCs for CC398 and non-CC398 MRSA isolates were 192.5 µg/mL and 7.667 µg/mL, respectively (p = 0.0004).
MIC, minimum inhibitory concentration; MBC, minimum bactericidal concentration; MRSA, methicillin-resistant Staphylococcus aureus; TET, tetracycline.
Zinc chloride susceptibility and PCR detection of czrC
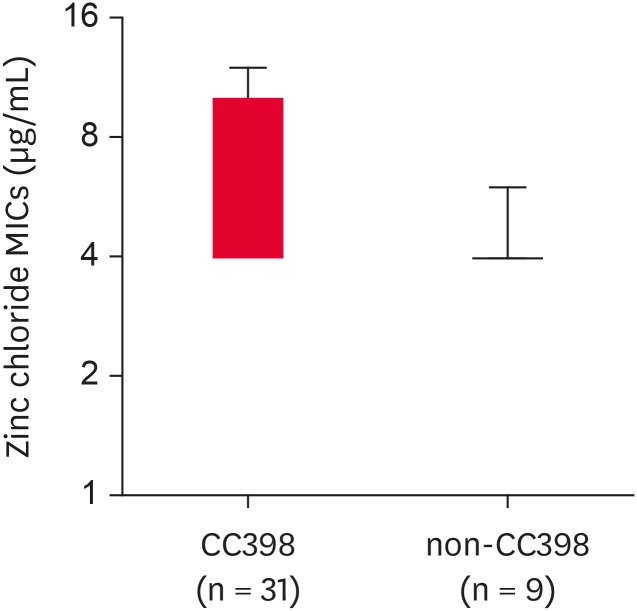
Previous studies have indicated that zinc resistance phenotype in LA-MRSA may be associated with certain clonal lineages of MRSA isolates such as CC398 MRSA [32]. Interestingly, all 40 MRSA isolates displayed zinc chloride MICs of > 2 mM, indicating zinc resistance phenotype (Table 2). Despite the absence of the czrC gene, 1 isolate of ST541 MRSA, 4 isolates of ST2084 MRSA, and 2 isolates of ST5 MRSA exhibited a zinc resistant phenotype. Of note, 12 of the 25 ST398 MRSA (48%) and 5 of the 6 ST541 (83.3%) MRSA isolates had zinc chloride MICs of ≥ 6 mM. A high level zinc chloride resistance (MICs of ≥ 10 mM) was observed only among czrC-positive ST398 MRSA isolates. As presented in Fig. 4, when the MRSA strains were compared as groups of CC398 and non-CC398 strains, the CC398 MRSA strains exhibited significantly higher levels of zinc chloride MICs as compared to the non-CC398 MRSA strains (median zinc chloride MICs of 6.581 vs. 4.222 µg/mL, p = 0.0027).
Fig. 4. Box and Whisker plots of zinc chloride MICs for CC398 and non-CC398 MRSA isolates. The median zinc chloride MICs for CC398 and nonCC398 MRSA isolates were 6.581 µg/mL and 4.222 µg/mL, respectively (p = 0.0027).
MIC, minimum inhibitory concentration; MRSA, methicillin-resistant Staphylococcus aureus.
Virulence factors and SE genes
An analysis of ETs in the 40 MRSA isolates revealed that only 3 ST398 MRSA isolates (7.5%) from pigs were positive for etb.
As shown in Table 2, only 3 CC398 MRSA isolates (3 ST398 MRSA isolates) carried at least 1 of the SE genes; 2 ST398 MRSA harbored sea, 1 ST398 MRSA isolate was double-positive for sec and sed, and none of the ST541 MRSA isolates had SE genes. Three of the non-CC398 MRSA isolates were positive for at least 1 of the SE genes; 1 ST2084 had sed, and 2 ST1 MRSA were positive for sea and sea/sec. Although the 2 ST1 MRSA isolates were also positive for PVL, none of the CC398 MRSA isolates carried genes for PVL and TSST-1.
DISCUSSION
The emergence and spread of LA-MRSA in food-producing animals have raised public health concerns particularly on the possible transmission to humans [33,34,35]. Although LA-MRSA has primarily been associated with livestock animals, especially pigs [8], there is a considerable number of reports where humans are colonized by LA-MRSA through occupational contact [7,33,34,35,36]. In this study, the overall prevalence of MRSA in pigs, farm workers, and farm facilities was 2.2%, 3.2%, and 2.8% (30/1,340, 6/186, and 4/144), respectively. Similar to the prevalence rates reported in 2 previous studies in Korea (3.2% [10] and 4.4% [11]), the prevalence of swine-associated MRSA in this study was still much lower than the prevalence in North Americas (26–36%) [37,38] and several European countries such as Spain (20.7%) [39], Germany (49%) [40], and Netherlands (38.7%) [41]. Unlike the similar levels of MRSA prevalence in pigs, the prevalence of MRSA in pig farmers (15.4%, 6 out of 39 pig farmers) observed in this study was somewhat lower than that (16.7%) in another study conducted in Korea during 2012–2016, in which only nasal swab samples were collected from pig farmers [11]. This might be attributed to several factors such as the sampling and isolation methods, farm management system, pig density, and geographical regions [4]. The prevalence of LA-MRSA was significantly decreased in slaughterhouses (0.6%) and retail markets (0.4%). Recently, Moon et al. [12] also reported a low level of MRSA prevalence in pig carcasses (0.6%) from slaughterhouses. These results indicate that transmission of LA-MRSA from pig farms to slaughterhouses and then to retail markets is still limited in Korea. However, the high prevalence of MRSA, especially swine-associated MRSA, in persons in contact with livestock or food of animal origin has been frequently observed in many countries [35,42,43]. Thus, continued efforts are required to identify the potential transmission routes of MRSA.
The spread of LA-MRSA CC398 (ST398 and ST541) in the swine population and pork production system has been linked to an increased number of MRSA infections in humans [44]. In contrast to other Asian countries, where the widespread colonization of ST9 LA-MRSA has been observed [35,45], the most frequent pig-associated MRSA in Korea has been ST398 and ST541 with spa type of t571 and t034 (ST398-t571 and ST541-t034), respectively [10,11]. In line with the previous observations [11,32,46], 25 (62.5%) and 6 (15%) of the 40 MRSA isolates were ST398 and ST541, respectively, mostly carrying SCCmec V. However, in contrast to the prior investigation [10,11,12], 21/25 ST398 MRSA strains isolated in this study had spa type of t18103 or t18102. The detection of different spa types in MRSA isolates with identical STs may be attributed to the diversification of spa through adaptation to different environments. The detection of ST398 MRSA isolates with varied spa types (ST398-t18103 and ST398-t18102) warrants further investigation and monitoring. Furthermore, although the incidence is still limited, the detection of LA-MRSA isolates with identical genotypes (ST398-t571-SCCmecV-agr I) both in pig farms and slaughterhouses suggests potential transmission of LA-MRSA from live pigs to pork supply chains.
As shown in Table 2 and Fig. 2 all CC398 MRSA isolates exhibited multidrug-resistant phenotype with 100% resistance to TET. Similar to the findings of previous studies [12,13,15], these LA-MRSA isolates carried 1 or more genes that could confer TET resistance (tetK, tetL, and/or tetM). It has been proposed that TET resistance is a potential phenotypic marker for CC398 LA-MRSA isolates (ST398 and ST541 LA-MRSA) [9]. Although the history of TET usage in the pig farms in the current study is not available, TET compounds have been frequently used to control respiratory diseases in weaning pigs in Korea. As shown by the results in Fig. 3A and B, CC398 MRSA isolates showed significantly higher levels of MICs (p = 0.0001) and MBCs (p = 0.0004) to TET than non-CC398 MRSA isolates, indicating that the CC398 MRSA isolates can have selective advantage under TET pressure. Thus, in combination with MDR phenotype, it is speculated that the CC398 LA-MRSA strains isolated in this study may have been selected by various antimicrobial agents, especially TET.
In addition to the resistance to TET, zinc resistance mediated by the czrC gene has been associated with the high prevalence of CC398 LA-MRSA in pigs. As the czrC and mecA genes are colocalized within the SCCmec V element [47], the use of zinc as a feed additive to prevent diarrhea in weaning pigs may favor the selection of czrC in CC398 LA-MRSA. As shown in Fig. 4, the levels of zinc chloride MICs were significantly higher for the CC398 LA-MRSA group than for the non-CC398 MRSA group (p < 0.01), suggesting that the use of dietary zinc in feed may contribute to the prevalence of CC398 LA-MRSA in pigs in Korea by promoting the selection and maintenance of the SCCmec V element in these clonal lineages. Zinc chloride MIC data, in combination with the TET susceptibility (Fig. 3) and SCCmec typing data (Table 2), indicate that co-selection pressure associated with TET usage and zinc supplementation in feed may play an important role in the prevalence of CC398 LA-MRSA in the swine population in Korea. Future studies are necessary with a larger collection of MRSA strains to elucidate the role of zinc and TET cross-resistance in the selection and maintenance of CC398 LA-MRSA in pig farms.
The acquisition of SE genes, PVL genes (lukF and lukS), TSST-1, and ET genes (eta, etb, and etd) by LA-MRSA isolates may pose a considerable threat to public health. However, in comparison with non-CC398 MRSA isolates, CC398 LA-MRSA isolates did not acquire these virulence genes more frequently, and none of the CC398 LA-MRSA isolates harbored the PVL and TSST-1 genes in our current study.
It is important to recognize that our current study have several limitations. Our data were generated based on a limited number of LA-MRSA isolates recovered from 2017–2018. Thus, future studies with a larger sample of LA-MRSA over an extended period of time are required. Moreover, information on antibiotic usage, especially TET, and zinc concentration in the feed additives in pig farms were not available in the present study. However, our current study is unique in that it is the first national investigation to systematically sample pigs, humans, and facility environment of different sectors in the pork production chain including pig farms, slaughterhouses, and retail markets. In addition, this study is the first to report the detection of ST398-t18102 and ST398-t18103 LA-MRSA isolates in pigs and farm workers in Korea.
In conclusion, our results suggest that 1) the prevalence of swine-associated MRSA is lower in Korea than in other European and Asian countries, which is similar to the findings of previous studies [10,11]; 2) the prevalence of CC398 LA-MRSA (ST398 and ST541) appears to be correlated with increased levels of MDR phenotype, especially TET resistance; and 3) in addition to TET resistance, CC398 LA-MRSA isolates tend to exhibit cross-resistance to zinc, suggesting its potential role in the prevalence and persistence of swine-associated CC398 LA-MRSA in Korea.
Footnotes
Funding: This research was supported by a fund by Research of Korea Centers for Disease Control and Prevention (2017NER54060 to S.J.Y.). This research was also supported by the Chung-Ang University Graduate Research Scholarship (S.H.B) in 2017.
Conflict of Interest: The authors declare no conflicts of interest.
- Conceptualization: Yang SJ, Park KT.
- Data curation: Yang SJ, Back SH.
- Formal analysis: Yang SJ, Back SH.
- Funding acquisition: Yang SJ.
- Investigation: Back SH, Eom HS, Lee HH, Lee GY.
- Methodology: Yang SJ, Park KT.
- Project administration: Back SH, Eom HS.
- Writing - original draft: Yang SJ.
- Writing - review & editing: Yang SJ, Park KT.
References
- 1.Bartels MD, Boye K, Rhod Larsen A, Skov R, Westh H. Rapid increase of genetically diverse methicillin-resistant Staphylococcus aureus, Copenhagen, Denmark. Emerg Infect Dis. 2007;13:1533–1540. doi: 10.3201/eid1310.070503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kim ES, Song JS, Lee HJ, Choe PG, Park KH, Cho JH, Park WB, Kim SH, Bang JH, Kim DM, Park KU, Shin S, Lee MS, Choi HJ, Kim NJ, Kim EC, Oh MD, Kim HB, Choe KW. A survey of community-associated methicillin-resistant Staphylococcus aureus in Korea. J Antimicrob Chemother. 2007;60:1108–1114. doi: 10.1093/jac/dkm309. [DOI] [PubMed] [Google Scholar]
- 3.Sahibzada S, Abraham S, Coombs GW, Pang S, Hernández-Jover M, Jordan D, Heller J. Transmission of highly virulent community-associated MRSA ST93 and livestock-associated MRSA ST398 between humans and pigs in Australia. Sci Rep. 2017;7:5273. doi: 10.1038/s41598-017-04789-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gonçalves da Silva A, Baines SL, Carter GP, Heffernan H, French NP, Ren X, Seemann T, Bulach D, Kwong J, Stinear TP, Howden BP, Williamson DA. A phylogenomic framework for assessing the global emergence and evolution of clonal complex 398 methicillin-resistant Staphylococcus aureus. Microb Genom. 2017;3:e000105. doi: 10.1099/mgen.0.000105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sørensen AI, Toft N, Boklund A, Espinosa-Gongora C, Græsbøll K, Larsen J, Halasa T. A mechanistic model for spread of livestock-associated methicillin-resistant Staphylococcus aureus (LA-MRSA) within a pig herd. PLoS One. 2017;12:e0188429. doi: 10.1371/journal.pone.0188429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Witte W, Kresken M, Braulke C, Cuny C. Increasing incidence and widespread dissemination of methicillin-resistant Staphylococcus aureus (MRSA) in hospitals in central Europe, with special reference to German hospitals. Clin Microbiol Infect. 1997;3:414–422. doi: 10.1111/j.1469-0691.1997.tb00277.x. [DOI] [PubMed] [Google Scholar]
- 7.Crombé F, Argudín MA, Vanderhaeghen W, Hermans K, Haesebrouck F, Butaye P. Transmission dynamics of methicillin-resistant Staphylococcus aureus in pigs. Front Microbiol. 2013;4:57. doi: 10.3389/fmicb.2013.00057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sharma M, Nunez-Garcia J, Kearns AM, Doumith M, Butaye PR, Argudín MA, Lahuerta-Marin A, Pichon B, AbuOun M, Rogers J, Ellis RJ, Teale C, Anjum MF. Livestock-associated methicillin-resistant Staphylococcus aureus (LA-MRSA) clonal complex (CC) 398 isolated from UK animals belong to European lineages. Front Microbiol. 2016;7:1741. doi: 10.3389/fmicb.2016.01741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Price LB, Stegger M, Hasman H, Aziz M, Larsen J, Andersen PS, Pearson T, Waters AE, Foster JT, Schupp J, Gillece J, Driebe E, Liu CM, Springer B, Zdovc I, Battisti A, Franco A, Zmudzki J, Schwarz S, Butaye P, Jouy E, Pomba C, Porrero MC, Ruimy R, Smith TC, Robinson DA, Weese JS, Arriola CS, Yu F, Laurent F, Keim P, Skov R, Aarestrup FM. Staphylococcus aureus CC398: host adaptation and emergence of methicillin resistance in livestock. MBio. 2012;3:e00305-11. doi: 10.1128/mBio.00305-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lim SK, Nam HM, Jang GC, Lee HS, Jung SC, Kwak HS. The first detection of methicillin-resistant Staphylococcus aureus ST398 in pigs in Korea. Vet Microbiol. 2012;155:88–92. doi: 10.1016/j.vetmic.2011.08.011. [DOI] [PubMed] [Google Scholar]
- 11.Moon DC, Jeong SK, Hyun BH, Lim SK. Prevalence and characteristics of methicillin-resistant Staphylococcus aureus isolates in pigs and pig farmers in Korea. Foodborne Pathog Dis. 2019;16:256–261. doi: 10.1089/fpd.2018.2509. [DOI] [PubMed] [Google Scholar]
- 12.Moon DC, Tamang MD, Nam HM, Jeong JH, Jang GC, Jung SC, Park YH, Lim SK. Identification of livestock-associated methicillin-resistant Staphylococcus aureus isolates in Korea and molecular comparison between isolates from animal carcasses and slaughterhouse workers. Foodborne Pathog Dis. 2015;12:327–334. doi: 10.1089/fpd.2014.1868. [DOI] [PubMed] [Google Scholar]
- 13.Larsen J, Clasen J, Hansen JE, Paulander W, Petersen A, Larsen AR, Frees D. Copresence of tet(K) and tet(M) in livestock-associated methicillin-resistant Staphylococcus aureus clonal complex 398 is associated with increased fitness during exposure to sublethal concentrations of tetracycline. Antimicrob Agents Chemother. 2016;60:4401–4403. doi: 10.1128/AAC.00426-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Moodley A, Nielsen SS, Guardabassi L. Effects of tetracycline and zinc on selection of methicillin-resistant Staphylococcus aureus (MRSA) sequence type 398 in pigs. Vet Microbiol. 2011;152:420–423. doi: 10.1016/j.vetmic.2011.05.025. [DOI] [PubMed] [Google Scholar]
- 15.Sieber RN, Skov RL, Nielsen J, Schulz J, Price LB, Aarestrup FM, Larsen AR, Stegger M, Larsen J. Drivers and dynamics of methicillin-resistant livestock-associated Staphylococcus aureus CC398 in pigs and humans in Denmark. MBio. 2018;9:e02142-18. doi: 10.1128/mBio.02142-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zdovc I, Ocepek M, Pirs T, Krt B, Pinter L. Microbiological features of Staphylococcus schleiferi subsp. coagulans, isolated from dogs and possible misidentification with other canine coagulase-positive staphylococci. J Vet Med B Infect Dis Vet Public Health. 2004;51:449–454. doi: 10.1111/j.1439-0450.2004.00792.x. [DOI] [PubMed] [Google Scholar]
- 17.Clinical and Laboratory Standards Institute (CLSI) Performance Standards for Antimicrobial Susceptibility Testing. CLSI document M100-S27. Wayne: CLSI; 2017. [Google Scholar]
- 18.Dyer DW, Iandolo JJ. Rapid isolation of DNA from Staphylococcus aureus. Appl Environ Microbiol. 1983;46:283–285. doi: 10.1128/aem.46.1.283-285.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Vannuffel P, Gigi J, Ezzedine H, Vandercam B, Delmee M, Wauters G, Gala JL. Specific detection of methicillin-resistant Staphylococcus species by multiplex PCR. J Clin Microbiol. 1995;33:2864–2867. doi: 10.1128/jcm.33.11.2864-2867.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Boye K, Bartels MD, Andersen IS, Møller JA, Westh H. A new multiplex PCR for easy screening of methicillin-resistant Staphylococcus aureus SCCmec types I–V. Clin Microbiol Infect. 2007;13:725–727. doi: 10.1111/j.1469-0691.2007.01720.x. [DOI] [PubMed] [Google Scholar]
- 21.Enright MC, Day NP, Davies CE, Peacock SJ, Spratt BG. Multilocus sequence typing for characterization of methicillin-resistant and methicillin-susceptible clones of Staphylococcus aureus. J Clin Microbiol. 2000;38:1008–1015. doi: 10.1128/jcm.38.3.1008-1015.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Uhlén M, Guss B, Nilsson B, Gatenbeck S, Philipson L, Lindberg M. Complete sequence of the staphylococcal gene encoding protein A. A gene evolved through multiple duplications. J Biol Chem. 1984;259:1695–1702. [PubMed] [Google Scholar]
- 23.Harmsen D, Claus H, Witte W, Rothgänger J, Claus H, Turnwald D, Vogel U. Typing of methicillin-resistant Staphylococcus aureus in a university hospital setting by using novel software for spa repeat determination and database management. J Clin Microbiol. 2003;41:5442–5448. doi: 10.1128/JCM.41.12.5442-5448.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gilot P, Lina G, Cochard T, Poutrel B. Analysis of the genetic variability of genes encoding the RNA III-activating components Agr and TRAP in a population of Staphylococcus aureus strains isolated from cows with mastitis. J Clin Microbiol. 2002;40:4060–4067. doi: 10.1128/JCM.40.11.4060-4067.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Becker K, Roth R, Peters G. Rapid and specific detection of toxigenic Staphylococcus aureus: use of two multiplex PCR enzyme immunoassays for amplification and hybridization of staphylococcal enterotoxin genes, exfoliative toxin genes, and toxic shock syndrome toxin 1 gene. J Clin Microbiol. 1998;36:2548–2553. doi: 10.1128/jcm.36.9.2548-2553.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lina G, Piémont Y, Godail-Gamot F, Bes M, Peter MO, Gauduchon V, Vandenesch F, Etienne J. Involvement of Panton-Valentine leukocidin-producing Staphylococcus aureus in primary skin infections and pneumonia. Clin Infect Dis. 1999;29:1128–1132. doi: 10.1086/313461. [DOI] [PubMed] [Google Scholar]
- 27.Mehrotra M, Wang G, Johnson WM. Multiplex PCR for detection of genes for Staphylococcus aureus enterotoxins, exfoliative toxins, toxic shock syndrome toxin 1, and methicillin resistance. J Clin Microbiol. 2000;38:1032–1035. doi: 10.1128/jcm.38.3.1032-1035.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hu DL, Omoe K, Shimoda Y, Nakane A, Shinagawa K. Induction of emetic response to staphylococcal enterotoxins in the house musk shrew (Suncus murinus) Infect Immun. 2003;71:567–570. doi: 10.1128/IAI.71.1.567-570.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cavaco LM, Hasman H, Stegger M, Andersen PS, Skov R, Fluit AC, Ito T, Aarestrup FM. Cloning and occurrence of czrC, a gene conferring cadmium and zinc resistance in methicillin-resistant Staphylococcus aureus CC398 isolates. Antimicrob Agents Chemother. 2010;54:3605–3608. doi: 10.1128/AAC.00058-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ng LK, Martin I, Alfa M, Mulvey M. Multiplex PCR for the detection of tetracycline resistant genes. Mol Cell Probes. 2001;15:209–215. doi: 10.1006/mcpr.2001.0363. [DOI] [PubMed] [Google Scholar]
- 31.Aarestrup FM, Hasman H. Susceptibility of different bacterial species isolated from food animals to copper sulphate, zinc chloride and antimicrobial substances used for disinfection. Vet Microbiol. 2004;100:83–89. doi: 10.1016/j.vetmic.2004.01.013. [DOI] [PubMed] [Google Scholar]
- 32.Hau SJ, Frana T, Sun J, Davies PR, Nicholson TL. Zinc resistance within swine-associated methicillin-resistant Staphylococcus aureus isolates in the united states is associated with multilocus sequence type lineage. Appl Environ Microbiol. 2017;83:e00756-17. doi: 10.1128/AEM.00756-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Smith TC, Wardyn SE. Human infections with Staphylococcus aureus CC398. Curr Environ Health Rep. 2015;2:41–51. doi: 10.1007/s40572-014-0034-8. [DOI] [PubMed] [Google Scholar]
- 34.van der Mee-Marquet NL, Corvaglia A, Haenni M, Bertrand X, Franck JB, Kluytmans J, Girard M, Quentin R, François P. Emergence of a novel subpopulation of CC398 Staphylococcus aureus infecting animals is a serious hazard for humans. Front Microbiol. 2014;5:652. doi: 10.3389/fmicb.2014.00652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wang W, Liu F, Baloch Z, Zhang CS, Ma K, Peng ZX, Yan SF, Hu YJ, Gan X, Dong YP, Bai Y, Li FQ, Yan XM, Ma AG, Xu J. Genotypic characterization of methicillin-resistant Staphylococcus aureus isolated from pigs and retail foods in China. Biomed Environ Sci. 2017;30:570–580. doi: 10.3967/bes2017.076. [DOI] [PubMed] [Google Scholar]
- 36.Lekkerkerk WS, van Wamel WJ, Snijders SV, Willems RJ, van Duijkeren E, Broens EM, Wagenaar JA, Lindsay JA, Vos MC. What is the origin of livestock-associated methicillin-resistant Staphylococcus aureus clonal complex 398 isolates from humans without livestock contact? an epidemiological and genetic analysis. J Clin Microbiol. 2015;53:1836–1841. doi: 10.1128/JCM.02702-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Khanna T, Friendship R, Dewey C, Weese JS. Methicillin resistant Staphylococcus aureus colonization in pigs and pig farmers. Vet Microbiol. 2008;128:298–303. doi: 10.1016/j.vetmic.2007.10.006. [DOI] [PubMed] [Google Scholar]
- 38.Smith TC, Male MJ, Harper AL, Kroeger JS, Tinkler GP, Moritz ED, Capuano AW, Herwaldt LA, Diekema DJ. Methicillin-resistant Staphylococcus aureus (MRSA) strain ST398 is present in midwestern U.S. swine and swine workers. PLoS One. 2009;4:e4258. doi: 10.1371/journal.pone.0004258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Gómez-Sanz E, Torres C, Lozano C, Fernández-Pérez R, Aspiroz C, Ruiz-Larrea F, Zarazaga M. Detection, molecular characterization, and clonal diversity of methicillin-resistant Staphylococcus aureus CC398 and CC97 in Spanish slaughter pigs of different age groups. Foodborne Pathog Dis. 2010;7:1269–1277. doi: 10.1089/fpd.2010.0610. [DOI] [PubMed] [Google Scholar]
- 40.Tenhagen BA, Fetsch A, Stührenberg B, Schleuter G, Guerra B, Hammerl JA, Hertwig S, Kowall J, Kämpe U, Schroeter A, Bräunig J, Käsbohrer A, Appel B. Prevalence of MRSA types in slaughter pigs in different German abattoirs. Vet Rec. 2009;165:589–593. doi: 10.1136/vr.165.20.589. [DOI] [PubMed] [Google Scholar]
- 41.de Neeling AJ, van den Broek MJ, Spalburg EC, van Santen-Verheuvel MG, Dam-Deisz WD, Boshuizen HC, van de Giessen AW, van Duijkeren E, Huijsdens XW. High prevalence of methicillin resistant Staphylococcus aureus in pigs. Vet Microbiol. 2007;122:366–372. doi: 10.1016/j.vetmic.2007.01.027. [DOI] [PubMed] [Google Scholar]
- 42.Grøntvedt CA, Elstrøm P, Stegger M, Skov RL, Skytt Andersen P, Larssen KW, Urdahl AM, Angen Ø, Larsen J, Åmdal S, Løtvedt SM, Sunde M, Bjørnholt JV. Methicillin-resistant Staphylococcus aureus CC398 in humans and pigs in Norway: a “One Health” perspective on introduction and transmission. Clin Infect Dis. 2016;63:1431–1438. doi: 10.1093/cid/ciw552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Verkade E, Kluytmans J. Livestock-associated Staphylococcus aureus CC398: animal reservoirs and human infections. Infect Genet Evol. 2014;21:523–530. doi: 10.1016/j.meegid.2013.02.013. [DOI] [PubMed] [Google Scholar]
- 44.Becker K, Ballhausen B, Kahl BC, Köck R. The clinical impact of livestock-associated methicillin-resistant Staphylococcus aureus of the clonal complex 398 for humans. Vet Microbiol. 2017;200:33–38. doi: 10.1016/j.vetmic.2015.11.013. [DOI] [PubMed] [Google Scholar]
- 45.Li J, Jiang N, Ke Y, Feßler AT, Wang Y, Schwarz S, Wu C. Characterization of pig-associated methicillin-resistant Staphylococcus aureus . Vet Microbiol. 2017;201:183–187. doi: 10.1016/j.vetmic.2017.01.017. [DOI] [PubMed] [Google Scholar]
- 46.Kim ES, Lee HJ, Chung GT, Lee YS, Shin DH, Jung SI, Song KH, Park WB, Kim NJ, Park KU, Kim EC, Oh MD, Kim HB. Molecular characterization of methicillin-resistant Staphylococcus aureus isolates in Korea. J Clin Microbiol. 2011;49:1979–1982. doi: 10.1128/JCM.00098-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Agersø Y, Hasman H, Cavaco LM, Pedersen K, Aarestrup FM. Study of methicillin resistant Staphylococcus aureus (MRSA) in Danish pigs at slaughter and in imported retail meat reveals a novel MRSA type in slaughter pigs. Vet Microbiol. 2012;157:246–250. doi: 10.1016/j.vetmic.2011.12.023. [DOI] [PubMed] [Google Scholar]