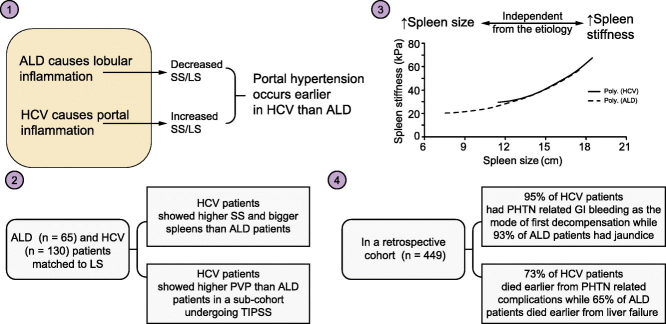
Abstract
Background & Aims
Both liver stiffness (LS) and spleen stiffness (SS) are widely used to non-invasively assess liver fibrosis and portal hypertension, respectively. We aimed to identify the impact of disease etiology, namely the localization of inflammation (portal vs. lobular), on the SS/LS ratio.
Methods
In this multicenter study, LS and SS were prospectively assessed in 411 patients with alcohol-related liver disease (ALD) or hepatitis C virus (HCV) using FibroScan® (Echosens, Paris); changes in these parameters were also studied in response to treatment (alcohol withdrawal, HCV therapy). LS and spleen length (SL) were further analyzed in a retrospective cohort of 449 patients with long-term data on decompensation/death.
Results
Both, SS and SL were significantly higher in HCV compared to ALD (42.0 vs. 32.6 kPa, p≪0.0001, 15.6 vs. 11.9 cm, p≪0.0001) despite a lower mean LS in HCV. Consequently, the SS to LS ratio and the SL to LS ratio were significantly higher in HCV (3.8 vs. 1.72 and 1.46 vs. 0.86, p≪0.0001) through all fibrosis stages. Notably, SL linearly increased with SS and the relation between SS and SL was identical in HCV and ALD. In contrast, livers were much larger in ALD at comparable LS. After treatment, LS significantly decreased in both diseases without significant changes to the SS/LS ratio. In the prognostic cohort, patients with ALD had higher LS values (30.5 vs. 21.3 kPa) and predominantly presented with jaundice (65.2%); liver failure was the major cause of death (p≪0.01). In contrast, in HCV, spleens were larger (17.6 vs. 12.1 cm) while variceal bleeding was the major cause of decompensation (73.2%) and death (p≪0.001).
Conclusion
Both SS/LS and SL/LS ratios are significantly higher in patients with portal HCV compared to lobular ALD. Thus, combined LS and SS or SL measurements provide additional information about disease etiology and disease-specific complications.
Lay summary
Herein, we show that patients with hepatitis C virus infection (HCV) have higher spleen stiffness and portal pressure than patients with alcohol-related liver disease (ALD), within the same fibrosis stage and matched to liver stiffness. Thus, the spleen stiffness to liver stiffness ratio is significantly higher in patients with HCV compared to ALD. Additionally, patients with HCV more commonly progress to portal hypertension-related complications (e.g. variceal bleeding), while patients with ALD more commonly progress to liver failure (e.g. jaundice). The spleen stiffness to liver stiffness ratio is a useful tool to confirm disease etiology and predict disease-specific complications.
Keywords: Cirrhosis, liver decompensation, alcohol detoxification, DAAs, portal hypertension
Graphical abstract
Highlights
-
•
SS/LS ratio is significantly higher in patients with portal HCV compared to lobular ALD.
-
•
Patients with HCV have higher SS and portal pressure than patients with ALD and the same LS.
-
•
Patients with HCV die earlier from portal hypertension-related complications.
-
•
Patients with ALD die earlier from liver failure.
-
•
Combined LS and SS measurements provide extra information about disease etiology and disease-specific complications.
Introduction
The presence of clinically significant portal hypertension has important prognostic implications.[1], [2] Spleen stiffness (SS) is now widely used as novel non-invasive parameter to screen for portal hypertension[3], [4] outscoring liver stiffness (LS) or platelet count in predicting complications of portal hypertension such as the presence of esophageal varices and the risk of variceal bleeding.[5], [6], [7], [8], [9], [10] In fact, combining both LS and SS is currently considered the best predictor of portal hypertension.[3], [11] Although transient elastography (TE) was originally designed to measure LS, it can also be used to assess SS.[3], [9] Limitations are small or normal sized spleens, and obesity. Since SS is typically higher than LS, the upper detection limit of the FibroScan® device at 75 kPa is more rapidly reached, which has resulted in the development of a technically modified FibroScan prototype with an extended measuring range.12
However, less attention has been paid to SS as a function of disease etiology. For instance, LS clearly depends on disease etiology which has resulted in different F4 cirrhosis cut-off values, e.g. for patients with viral hepatitis or cholestatic liver disease.[13], [14] While this is still an ongoing debate, it has been well settled that inflammation is a key modifier of LS for common liver disease such as viral hepatitis or alcohol-related liver disease (ALD).[13], [15], [16], [17] Moreover, resolution of inflammation has been demonstrated to result in lowered LS both upon viral clearance in patients with hepatitis C virus (HCV)18 and alcohol detoxification in patients with ALD.[16], [19] In patients undergoing antiviral treatment, it is not completely clear to what extent the decreased LS is due to resolution of inflammation or fibrosis. In contrast, data on alcohol withdrawal show a clearer picture. Here, withdrawal from alcohol for less than a week can result in complete resolution of inflammation and consequent normalization of aminotransferases and LS. Consequently, the LS resolution in such a short time period is solely due to resolving inflammation. Interestingly, in a recent large multicenter trial with 2,068 patients with biopsy-proven HCV and ALD, it was demonstrated that the LS cut-off values clearly depended on inflammation as measured by aspartate aminotransferase (AST) levels.13 Moreover, identical AST levels caused a much higher increase of LS and related cut-off values in patients with ALD than in those with HCV. These findings are also in agreement with histological established sides of inflammation. Thus, ALD has been characterized mainly as a lobular disease, while chronic hepatitis C is predominantly seen as a portal disease.[20], [21], [22], [23] Consequently, it has been suggested that the localization of inflammation and fibrosis (portal vs. lobular) could be an additional critical factor that determines LS.24
Therefore, we aimed to analyze and compare SS, spleen length (SL) and LS in patients with portal (HCV) and lobular (ALD) chronic liver disease in a multicenter prospective cohort. Second, we studied the changes in these parameters in response to treatment. Finally, based on a retrospective cohort with long-term surveillance, we analyzed the association of LS and SL with clinical end points such hepatic decompensation and death.
Patients and methods
Patient cohorts and clinical data
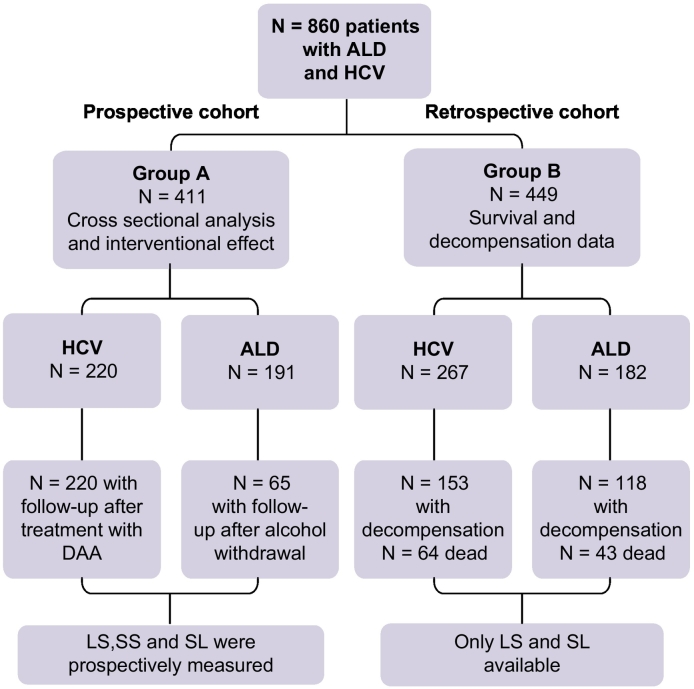
The study design is shown in Fig. 1 encompassing a total of 860 patients with both ALD and HCV. The local ethics committee approved the study protocol and informed consent was obtained from all patients. The overall study consists of 2 major Caucasian cohorts. Group A represents prospectively recruited patients from Heidelberg/Germany (n = 289), Odense/Denmark (n = 96) and Gluj Napoca/Romania (n = 26) with and without treatment while group B consists of a retrospective cohort from Germany (n = 352) and Great Britain (n = 97) with long-term follow-up data, LS parameters, SL and information on liver-related events and survival.
Fig. 1.
Study design.
ALD, alcohol-related liver disease; DAA, direct-acting antiviral; HCV, hepatitis C virus; LS, liver stiffness; SL, spleen length; SS, spleen stiffness.
For group A (411 patients), we prospectively recruited 191 patients with ALD and 220 patients with HCV. All patients were analyzed cross-sectionally for LS, SS, SL, ultrasonographic findings and routine lab parameters. In addition, we studied LS and SS in response to treatment interventions such as alcohol withdrawal or antiviral therapy in 65 patients with ALD and all 220 with HCV. Patients with ALD were 18 to 75 years of age and reported a prior or ongoing use of alcohol exceeding the maximum recommended limit of 3 units per day for men and 2 units per day for women (1 unit = 12 grams of alcohol), with a period of excessive drinking of at least 1 year. Patients with ALD represented a wide range of disease stages, from no fibrosis (F0) to F4 end-stage cirrhosis without clinically defined alcoholic hepatitis (see also Table 1). In patients with ALD, all other potential causes of chronic liver disease such as viral hepatitis, autoimmune disease, iron overload disease and Wilson’s disease were ruled out according to standard guideline recommendations. All patients with HCV had quantitative PCR before treatment and had no other etiology for chronic liver disease except HCV. Patients with HCV and concomitant alcohol use were also excluded. Data from patients with HCV were further verified for non-reported alcohol consumption by analyzing AST/ALT ratio, gamma glutamyltransferase and mean corpuscular volume levels.25
Table 1.
Patient characteristics and cross-sectional comparison of ALD and HCV cohorts.
| Parameter | ALD (n = 191) | HCV (n = 220) | p value⁎ |
|---|---|---|---|
| Age (years) | 55.3 ± 10.1 | 50.7 ± 11.1 | 0.0016 |
| Gender (male) | 73% | 63% | 0.14 |
| BMI (kg/m2) | 26.2 ± 4.3 | 24.1 ± 4.0 | 0.0016 |
| DM (present) | 22% | 27% | 0.3120 |
| Bilirubin total (mg/dl) | 1.1 (0.5-1.7) | 0.7 (0.5-0.9) | ≪0.0001 |
| AST (U/L) | 46 (32-75) | 45 (29-68) | ≪0.0001 |
| ALT (U/L) | 35 (22-51) | 46 (29-68) | ≪0.05 |
| Hemoglobin (g/dl) | 10.7 ± 3.2 | 13.4 ± 2.0 | ≪0.0001 |
| Leucocytes (103/μl) | 7.3 ± 2.9 | 7.5 ± 6.8 | 0.5436 |
| Platelets (103/μl) | 194 ± 85 | 172 ± 80 | 0.095 |
| INR | 1.2 ± 0.39 | 1.1 ± 0.12 | 0.048 |
| Creatinine (mg/dl) | 0.60 ± 0.37 | 0.87 ± 0.65 | 0.0068 |
| APRI | 2.1 ± 1.2 | 1.8 ± 1.1 | 0.0188 |
| Liver size (cm) | 16.1 ± 2.9 | 14.3 ± 1.6 | ≪0.0001 |
| Spleen size (cm) | 11.9 ± 2.9 | 15.6 ± 3.6 | ≪0.0001 |
| Liver stiffness (kPa) | 31.0 ± 25.2 | 14.9 ± 10.3 | ≪0.001 |
| CAP (dB/m) | 285 ± 67 | 298 ± 49 | 0.2692 |
| Spleen stiffness (kPa) | 32.6 ± 21.7 | 42.0 ± 16.6 | ≪0.0001 |
| SL/LS ratio | 0.86 ± 0.79 | 1.46 ± 0.83 | ≪0.0001 |
| SS/LS ratio | 1.72 ± 1.31 | 3.8 ± 3.08 | ≪0.0001 |
| Fibrosis stages (non-invasive) | 0.0028 | ||
| F0 | 13.6% | 15.0% | |
| F1-2 | 22.2% | 30.0% | |
| F3 | 8.5% | 16.8% | |
| F4 | 55.7% | 38.2% | |
ALD, alcohol-related liver disease; ALT, alanine aminotransferase; APRI, AST to platelet ratio index; AST, aspartate aminotransferase; BMI, body mass index; CAP, controlled attenuation parameter; DM, diabetes mellitus; HCV, hepatitis C virus; INR, international normalized ratio; LS, liver stiffness; SS, spleen stiffness.
The statistical test used is unpaired t test.
The effect of alcohol withdrawal was studied in 65 patients for a mean time interval of 5.3 consecutive days following complete normalization of aminotransferase levels. These patients were heavy drinkers that primarily presented for alcohol detoxification to the hospital. Patients with HCV received the combination of sofosbuvir/daclatasvir +/- ribavirin for 3 months. Second LS and SS measurements were obtained after 10 days of treatment in 81 patients and after 4 weeks in 139 patients. At both time points, aminotransferase levels had normalized. Viral clearance was confirmed by quantitative PCR in all patients before the second measurement. In some analysis (see result section), patients with ALD and HCV were matched for LS, age, gender and body mass index (BMI) to further analyze and compare SS, SL, portal pressure, serum bilirubin, international normalized ratio (INR) and platelet count between both groups.
For group B (449 patients), we retrospectively accrued data on 182 patients with ALD and 267 with HCV, including the base line characteristics, sonographic data (liver size, SL and LS) and laboratory features. The patients were followed up for 7 years or until the date of death, with the incidence of various forms of decompensation recorded: jaundice (serum bilirubin level >2 mg/dl associated with yellowish discoloration of skin or sclera and radiological exclusion of biliary obstruction), variceal bleeding (diagnosed by upper gastrointestinal endoscopy), ascites (clinical diagnosis confirmed by ultrasonographic finding) or hepatic encephalopathy (clinical diagnosis). The mode of first decompensation was noted, in addition to the date of death or last visit. All data were retrieved from either admission or follow-up records.
Liver and spleen stiffness measurements
LS and SS were measured by TE (FibroScan, Echosens, Paris, France). TE was performed by physicians with at least 12 months of experience in abdominal ultrasound. TE of the liver was performed on the right lobe of the liver in the intercostal position using the M and XL probe. LS measurements were first started with the M probe with a measuring failure in 3.2% of all patients. In these patients, the XL probe was used according to the established protocols, yielding valid measurements without any exception.25 TE of the spleen was also done using the M (89.5%) or XL (10.5%) probe after determining the spleen size using ultrasonography while the patient was lying in the supine position with the left arm maximally abducted. The tip of the transducer was placed in the left intercostal space, mostly in the posterior axillary line, then 10 successful measurements were obtained.[3], [9] Valid SS measurements were obtained in 82% of patients. The mean success rate of LS measurements was 96% while it was 79% for SS measurements. Liver fibrosis stages were determined using recently established algorithms including the consideration of aminotransferase levels.[13], [25] In addition, liver size and ascites formation were assessed by abdominal ultrasound. Patients with mechanic cholestasis, liver congestion and nodular masses were excluded from the study. Patients with failed LS and SS measurements were also excluded from the analysis.
Statistical analysis
The statistical analysis was performed using SPSS Statistics version 25.0 (IBM, USA). For comparison of groups, we used the independent samples t test, chi-square test (for comparison of observed frequencies) and Mann-Whitney U test where appropriate. We reported the mean and the standard deviation in all parameters except in serum bilirubin, AST and alanine aminotransferase (ALT, not normally distributed) where median and interquartile range are given. For analysis of measurements before and after treatment, we used the paired-samples t test. Results were considered statistically significant when p values were lower than 0.05. All reported p values were uncorrected and 2-tailed. Matching for LS (65 ALD vs. 130 HCV) was performed, choosing 2 patients with HCV for 1 patient with ALD for the following LS ranges: 6–12 kPa, 12–20 kPa, 20–40 kPa and 40–55 kPa. Higher LS values were omitted because no LS-measurements above 55 kPa were observed in the HCV cohort. Patients could be selected so that age, BMI and gender were also not significantly different between the ALD and HCV group.
Results
Patients with HCV exhibit higher SS and SL compared to those with ALD
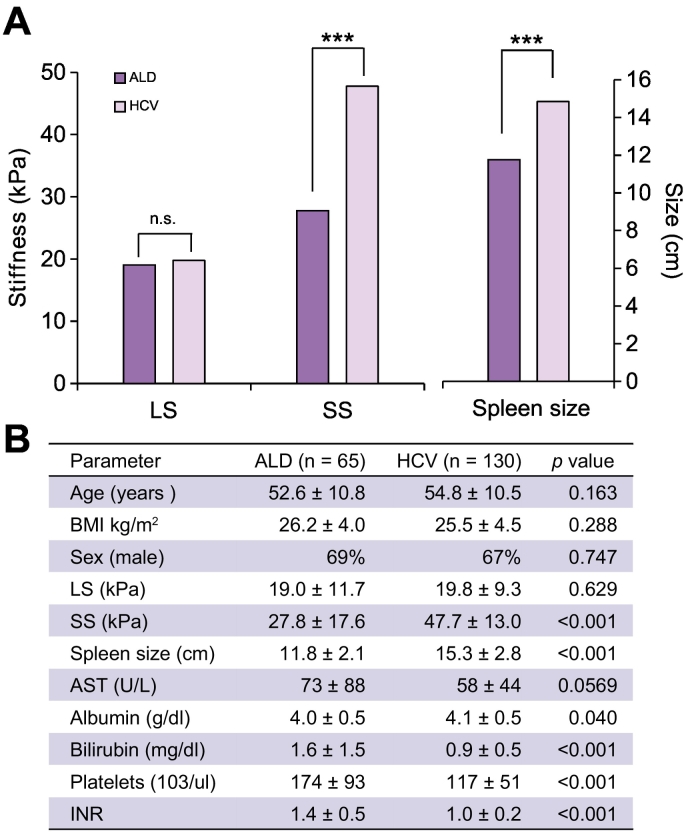
Since there are no established quality criteria for SS, we used all SS measurements with 10 successful measurements and we applied the established criteria used for LS.26 Accordingly, valid or reliable measurements of SS could be obtained in 85% of patients.26 As shown in Table 1, in the overall cross-sectional cohort, SL (15.0 vs. 11.9 cm) and SS (42.0 vs. 32.6 kPa, p≪0.001) were significantly higher in HCV patients despite a significantly lower LS (31.0 vs. 14.9 kPa) compared to ALD. Although patients with HCV and ALD differed in the cross-sectional cohort with regard to age (51 vs. 55 years, p = 0.01) and BMI (24.1 vs. 26.2, p = 0.001), no statistically significant differences were observed for gender and diabetes mellitus. We also noted that serum bilirubin levels and INR were higher in ALD (1.1 vs. 0.7 mg/dl, p≪0.0001 and 1.2 vs. 1.12, p≪0.001, respectively), while mean platelet count was lower in patients with HCV (202 vs. 172 [103/μl], p≪0.05). To rule out cohort-specific differences, we further matched both HCV and ALD cohorts for LS, age and gender and performed further analysis (see Fig. 2A and B). Although no significant differences were observed with regard to LS (19.0 vs. 19.8 kPa, p = 0.63), Fig. 2A demonstrates that spleens were significantly larger and SS was higher in HCV (11.8 vs. 14.9 cm and 27.8 vs. 47.8 kPa, p≪0.00001, respectively). Fig. 2B shows additional important differences between HCV and ALD in the matched cohorts. Thus, patients with HCV exhibited significantly lower platelet counts (195 vs. 139 [103/μl], p≪0.0001), while bilirubin (1.6 vs. 0.9 mg/dl, p≪0.0001) and INR were significantly elevated in ALD (1.4 vs. 1.0, p≪0.0001).
Fig. 2.
Differences between patients with ALD and HCV matched for fibrosis stage, age, gender and BMI. (A) Spleen stiffness and spleen length are significantly higher in HCV. (B) Comparison of other important parameters in the matched cohorts. (unpaired test, ***p ≪0.001).
ALD, alcohol-related liver disease; AST, aspartate aminotransferase; BMI, body mass index; HCV, hepatitis C virus; INR, international normalized ratio; LS, liver stiffness; SS, spleen stiffness.
Similar data were obtained when matching the patients for SS. Likewise, ALD patients had significantly higher LS (34.3 vs. 11.2 kPa, p≪0.001, see also Table S3). Notably, due to these differences, the AST to platelet ratio index (APRI) score, commonly used as a fibrosis score, was higher in patients with ALD without fibrosis than in all other stages with fibrosis (see Table S1). In contrast, in HCV, the APRI score continuously increased with fibrosis stage. Finally, in a subgroup of ALD (n = 17) and HCV (n = 25) matched for LS, we were able to measure portal vein pressure during transjugular portosystemic shunt implantation. Despite similar LS levels (52.4 [ALD] vs. 48.6 kPa [HCV]), patients with HCV had a significantly higher portal vein pressure (33.0 vs. 25.1 mmHg, respectively, p≪0.0001) (data not shown). In conclusion, we demonstrate a significantly elevated SS and SL in patients with HCV compared to ALD, even after matching for LS.
Identical SS to spleen association in HCV and ALD
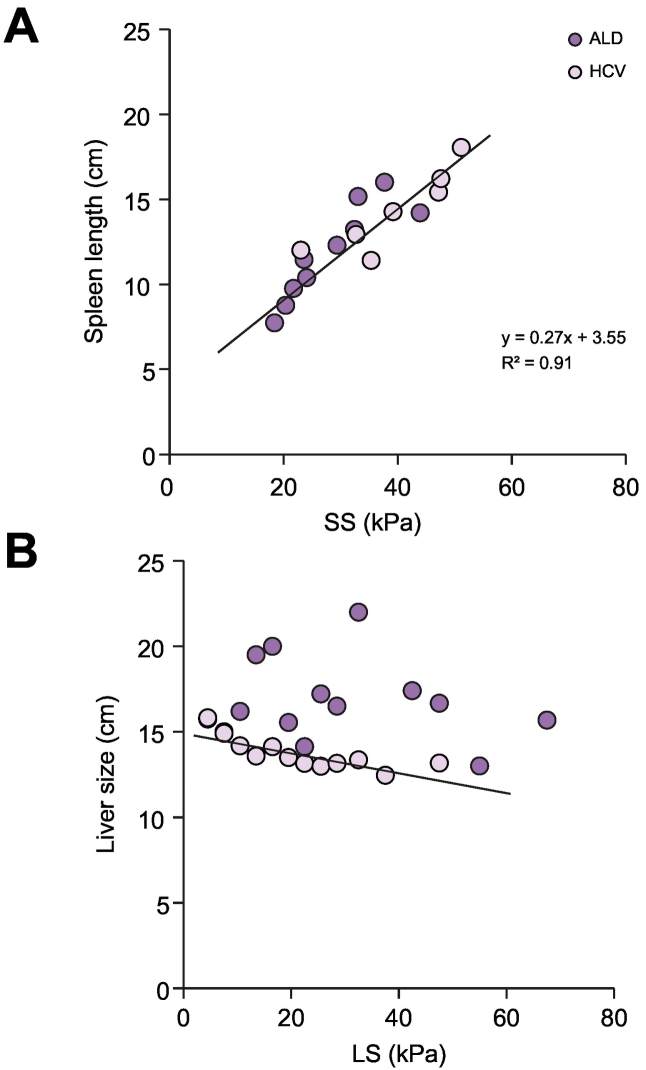
To get further insights into the observed differences in SS and SL between ALD and HCV, we plotted organ size as a function of organ stiffness for both diseases (see Fig. 3A and B). Interestingly, as shown in Fig. 3A, no differences were observed between ALD and HCV with regard to SS and SL. In other words, patients with similar SS showed identical SL irrespective of whether they had HCV or ALD. This indicates that SS and SL seem to be the sole result of portal hypertension while additional e.g. extrahepatic disease-specific confounders are negligible. SL correlated highly with SS, following a linear function (see Fig. 3A) with a coefficient of determination of 0.91 and no significant differences between both diseases. In contrast, liver size and LS differed substantially between HCV and ALD. Thus, in HCV, liver size continuously decreased with increasing LS while livers from patients with ALD were much larger. In summary, these data confirm that the differences between ALD and HCV are predominantly due to differences in liver size and stiffness, while SS and SL do not differ.
Fig. 3.
Relation of organ stiffness to size for liver and spleen. (A) Both in HCV40 and ALD (black), SL is linearly correlated with SS (R2=0.91). (B) Liver size plotted against LS for both HCV and ALD. The liver size gradually decreases with the progression of LS in HCV (see linear regression line). In contrast, large livers are observed with increasing LS in ALD. Regression graphical analysis and coefficient of determination (R2).
ALD, alcohol-related liver disease; HCV, hepatitis C virus; LS, liver stiffness; SL, spleen length; SS, spleen stiffness.
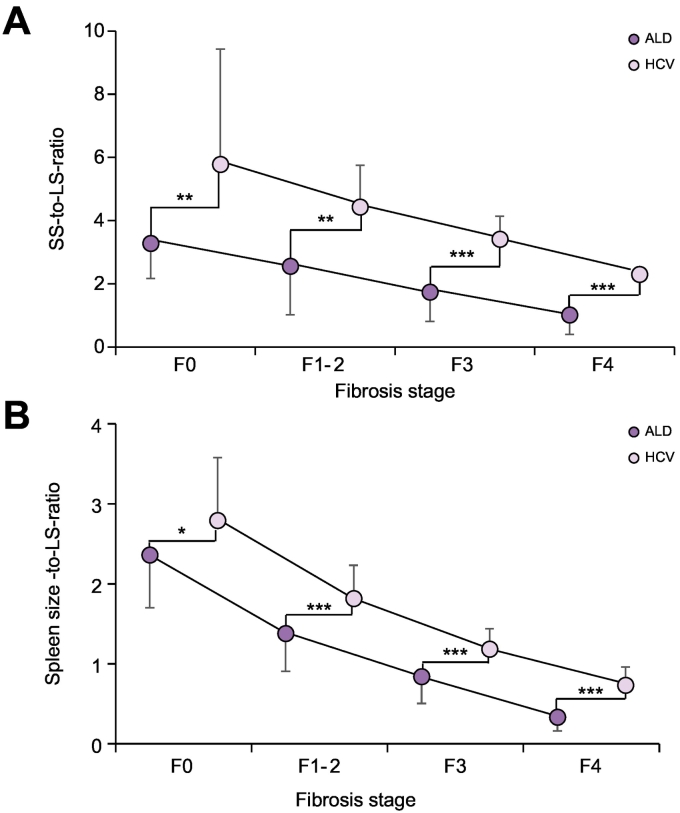
SS/LS ratio is significantly higher in HCV through all fibrosis stages
Our observations of significantly elevated SS to LS, or SL to LS ratio in HCV compared to ALD suggested to us that the underlying etiology had an important role in these measures. Thus, the portal localization of inflammation and fibrosis in HCV could explain the higher SS compared to the higher LS in patients with ALD and pronounced lobular disease. Since the site of inflammation is strongly dependent on fibrosis stage and will eventually encompass the whole liver in both diseases, we further postulated that fibrosis stage might have an important role on the SS/LS ratio. Consequently, we next studied in more detail the SS to LS ratio in patients with various fibrosis stages for both diseases using a recently established non-invasive algorithm.13 Indeed, the SS/LS ratio decreased with increasing fibrosis stage both in ALD and HCV (see Fig. 4A). Notably, SS/LS ratio was highest in early HCV with still normal LS exceeding a ratio of 5. In contrast, the highest SS/LS ratios in ALD reached levels of 2.6, but also continuously decreased with advancing liver disease. Notably, the SS/LS ratio overlapped between high fibrosis with HCV and low fibrosis with ALD. Similar data were obtained for SL (Fig. 4B). When plotting SS and SL directly against LS, both SS and SL were higher in HCV for the total LS range (see Fig. S1A and B). SS/LS ratio was also significantly higher in patients with HCV than ALD (3.3 vs. 1.3 for SS ≥40 kPa, p≪0.001, see Table S4) when stratifying patients according to the SS cut-off values for clinically significant portal hypertension.27 Taken together, both SS and SL to LS ratio are higher in HCV, most likely due to the specific histological site of inflammation/fibrosis.
Fig. 4.
Spleen stiffness or spleen length to liver stiffness ratios for HCV and ALD as a function of fibrosis stage. (A) SS to LS ratio and (B) SL to LS ratio according to fibrosis stage. Fibrosis stage was non-invasively assessed by TE using established AST-adapted cut-off values13 (see methods section). SS, SL to LS-ratios are always significantly higher in HCV in each fibrosis stage. Given are mean and standard deviations (unpaired test, **p≪0.01, ***p≪0.001).
ALD, alcohol-related liver disease; AST, aspartate aminotransferase; HCV, hepatitis C virus; LS, liver stiffness; SL, spleen length; SS, spleen stiffness; TE, transient elastography.
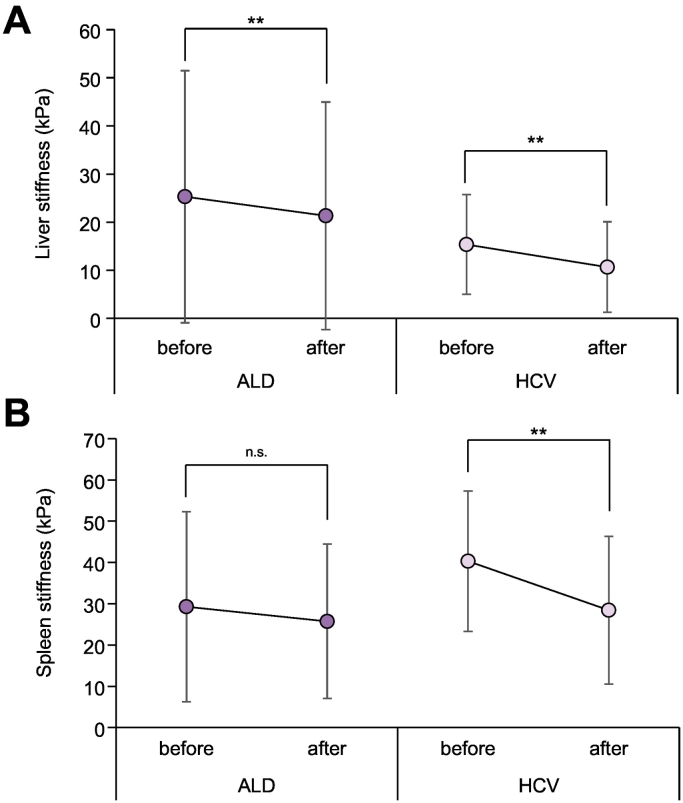
SS/LS ratio is not changed after treatment intervention
We now analyzed whether therapeutic interventions would have an impact on the SS/LS ratio in HCV and ALD. First, we studied the effect of alcohol withdrawal (mean interval 5.3 days) on laboratory markers, LS and SS. For comparison, we used a rather short follow-up protocol where the second measurements were obtained after 10 days and 4 weeks of standard direct-acting antiviral treatment protocol in patients with HCV (see patients and methods). All laboratory parameters including serum bilirubin, AST, ALT, INR and platelet count showed significant improvement after intervention in both diseases (see Table S2). Likewise, LS decreased significantly in both diseases (see Fig. 5). In contrast and although SS also decreased in both diseases after intervention, the decrease was only significant in patients with HCV (29.3 to 25.8 vs. 40.3 to 28.4 kPa, p = 0.18 and p≪0.0001, respectively). Consequently, the change in SS was more significant in patients with HCV than in those with ALD (-11.1 vs. -4.5 kPa, respectively, p≪0.0001, see Fig. 5). Finally, the SS to LS ratio did not change significantly during interventions in both diseases and there were no significant differences among HCV subgroups (not shown). In summary, although all stiffness parameters improved in both diseases after intervention, only patients with HCV showed a significant decrease of SS after treatment. Moreover, no significant changes in the SS/LS ratio were observed during intervention.
Fig. 5.
Change of liver stiffness and spleen stiffness during alcohol withdrawal (black) or HCV treatment40. (A) Liver stiffness significantly declined after intervention in both ALD and HCV (Paired t test, **p≪0.01). (B) In contrast, spleen stiffness only significantly decreased after HCV treatment but not after alcohol detoxification (Paired t test ***p≪0.001).
ALD, alcohol-related liver disease; HCV, hepatitis C virus; LS, liver stiffness; SS, spleen stiffness.
SL and LS correlate with the mode of liver decompensation and death
Finally, in a large retrospective cohort, we assessed whether the specific differences between patients with HCV and ALD would have an impact on the long-term outcome. Since SS was not available in this retrospective cohort, we focused on SL which showed a highly significant correlation with SS (see Fig. 3A). Table 2 shows long-term prognostic data for 182 patients with ALD and 267 with HCV who were clinically followed up for 7 years. Besides survival, final endpoints included signs of decompensation such as variceal bleeding, onset of jaundice, encephalopathy or ascites. Decompensation was noted in 118 patients with ALD and 153 with HCV. With 112 observations (73.2%, p≪0.0001 vs. ALD), variceal bleeding was the most frequent cause of first decompensation in the HCV group. In contrast, jaundice was the most frequent cause of first decompensation with 77 observations in the ALD cohort (65.2%, p≪0.0001). The incidence of variceal bleeding among dead patients was higher in HCV (95.3% vs. 16.2%, p≪0.0001), while the incidence of hepatocellular jaundice was much higher in the ALD group (93% vs. 28.1%, p≪0.0001). This follow-up cohort clearly demonstrates that patients with HCV are most likely to die from complications of portal hypertension, while patients with ALD are significantly more likely to experience liver failure with jaundice as a primary feature.
Table 2.
Patient characteristics of the retrospective cohorts with long-term survival and complications.
| ALD (n = 182) | HCV (n = 267) | p value⁎ | |
|---|---|---|---|
| Age (years) | 57.2 ± 9.2 | 56.3 ± 12.0 | 0.396 |
| Gender (% male) | 77.5% | 74.1% | 0.193 |
| AST (U/L) | 39 (34-79) | 71 (36-105) | ≪0.001 |
| ALT (U/L) | 34 (21-53) | 45 (22-71) | 0.008 |
| Bilirubin (mg/dl) | 1.4 (0.8-2.1) | 0.7 (0.4-1.2) | ≪0.001 |
| Platelets (103/μl) | 155 ± 80 | 95 ± 70 | ≪0.001 |
| Albumin (mg/dl) | 3.69 ± 0.43 | 3.81 ± 0.62 | 0.023 |
| INR | 1.09 ± 0.19 | 1.09 ± 0.26 | 0.989 |
| Creatinine (mg/dl) | 0.89 ± 0.78 | 0.78 ± 0.38 | ≪0.001 |
| Liver size (cm) | 16.8 ± 3.2 | 13.5 ± 2.2 | ≪0.001 |
| Spleen size (cm) | 12.5 ± 2.8 | 17.5 ± 4.2 | ≪0.001 |
| Liver stiffness (kPa) | 32.7 ± 19.7 | 20.1 ± 11.3 | ≪0.001 |
| SL/LS ratio | 0.62 ± 0.55 | 1.15 ± 0.67 | ≪0.001 |
| Mortality | 43 (23.6%) | 64 (23.9%) | |
| Decompensation | 118 (64.8%) | 153 (57.5%) | |
| Mode of first decompensation | |||
| Jaundice | 77 (65.3%) | 6 (3.9%) | ≪0.001 |
| Variceal bleeding | 11 (9.3%) | 112 (73.2%) | ≪0.001 |
| Ascites | 26 (22%) | 32 (20.9%) | 0.739 |
| Encephalopathy | 4 (3.4%) | 3 (2.0%) | 0.329 |
| Incidence of decompensation modes within mortality cohort (over lifetime) | |||
| Jaundice | 40 (93%) | 18 (28%) | ≪0.001 |
| Variceal bleeding | 7 (16.2%) | 61 (95.3%) | ≪0.01 |
| Ascites | 31 (72.1%) | 45 (70.3%) | 0.78 |
| Encephalopathy | 29 (67.4%) | 46 (72%) | 0.34 |
ALD, alcohol-related liver disease; ALT, alanine aminotransferase; AST, aspartate aminotransferase; HCV, hepatitis C virus; INR, international normalized ratio; LS, liver stiffness; SL, spleen length.
The statistical test used is unpaired t test.
Discussion
Herein, we demonstrate for the first time that both the SS to LS ratio as well as the SL to LS ratio are significantly higher in patients with HCV compared to ALD. In addition, they remain higher through all fibrosis stages and during therapeutic interventions. We further confirm in cohorts matched for LS that not only SS, but also the invasively measured portal pressure are higher in patients with HCV. Moreover, the association between SL and SS was comparable between ALD and HCV, while liver size was much larger in patients with ALD compared to HCV. Finally, in line with these observations, a long-term follow-up cohort clearly indicates that patients with HCV are more likely to have complications from portal hypertension, while patients with ALD are prone to develop complications of liver failure.
Our data suggest that non-invasive assessment of liver and spleen stiffness go far beyond the mere definition of fibrosis stage or prediction of portal hypertension. Rather, the SS/LS ratio seems to be determined by histological characteristics of the liver disease, namely the histological site of inflammation and fibrosis. Thus, portal inflammation with portal lymphoid aggregates are the histological hallmark of chronic hepatitis C and are established early on.[21], [28], [29], [30] In contrast, ALD is typically characterized by lobular inflammation in zone 3.[22], [23] Additional portal inflammation will only be observed in ALD during end-stage disease.[13], [31], [32] These established findings have been confirmed in previous histological studies on our ALD cohort.[16], [33] Moreover, a recent large multicenter trial on 2,068 patients with biopsy-proven HCV and ALD indicated important differences between both diseases with regard to LS and its response to inflammation.13 In this study, inflammation translated into a much lower LS elevation in HCV compared to ALD. While these findings have direct implications for non-invasive fibrosis staging at the bedside, the aforementioned histological site of inflammation has been suggested to impact on LS in a recent hypothesis paper.24 Accordingly, lobular inflammation/fibrosis in ALD should cause a more pronounced elevation of LS by intrahepatic congestion comparable to the extrahepatic liver congestion seen in heart failure.34 In contrast, a portal disease such as HCV should have less impact on LS but a more pronounced effect on portal hypertension and SS.
This scenario seems to be confirmed by our findings. First, SL and SS, invasively measured portal pressure and complications of portal hypertensions were always higher or more frequent in patients with HCV. Second, liver size to LS clearly differs between HCV and ALD. While, during fibrosis progression, liver size continuously decreased in patients with HCV, it rather increased in patients with ALD up to a LS of 30 kPa and only thereafter started to decrease. These findings also underscore that the liver is primarily responsible for the differences in SS/LS ratio between HCV and ALD. Third and in contrast to the liver, no differences were observed between HCV and ALD with regard to the relation of length and stiffness of the spleen. In fact and surprisingly, SL performed almost as well as SS as a parameter in ALD and HCV despite other confounders of splenomegaly such as hemolysis or mTOR activation.[35], [36] This also has practical implications since SL can easily be assessed during commonly available abdominal ultrasound.
Our findings could also explain the disease-specific nature of fibrosis scores. For instance, the APRI score (ratio AST/AST upper limit divided by platelet count) that is widely used for fibrosis scoring in viral hepatitis performs poorly in ALD.37 We recently showed that AST levels continuously increase in HCV with fibrosis stage, which is not the case for ALD.13 This is also confirmed in the present study where the APRI score increased with fibrosis score in HCV but not at all in ALD (see also Table S1). Our present data provide a mechanistic rational for these observations. Thus, portal inflammation and fibrosis predominantly elevate portal pressure with subsequent elevation of spleen stiffness/size and reduction of platelets. In contrast, lobular ALD primarily increases LS most likely by intrahepatic congestion while portal pressure increases at later stages. Consequently, our data highlight the fact that fibrosis is not a uniform disease but strongly etiology dependent. In the future, this could lead to more specific fibrosis characterization and, potentially, a novel fibrosis classification.24
We were also surprised to learn that the long-term outcome and complications differ dramatically between HCV and ALD and are in line with our findings on LS and SS. Thus, variceal bleeding is the most common sign of decompensation and cause of death in patients with HCV, while jaundice is the most common sign of decompensation in patients with ALD. The possibility that the SS/LS ratio could help to optimize the diagnosis of liver diseases and predict liver-specific complications remains an attractive idea. Since there was no overlap of SS/LS ratio between ALD and HCV, the SS/LS ratio may be also useful to better characterize the role of additional alcohol consumption in patients with HCV. It remains to be studied whether other liver diseases such as Budd Chiari syndrome, other forms of viral hepatitis including hepatitis B virus, diseases such as Schistosomiasis will also fit into the herein presented concept of histological site and its impact on LS and SS. Single unpublished observations seem to underline the concept. For instance, in patients with liver congestion and heart failure, LS levels of 75 kPa are often observed in the absence of SS elevation reaching SS/LS ratios of 10 kPa/75 kPa = 0.13. In contrast, portal vein thromboses can cause drastically elevated SS up to 75 kPa in the presence of an almost normal LS corresponding to SS/LS ratios of ca. 20. Therefore, the SS/LS ratio could be a useful additional test for the diagnosis of liver disease.
Our study has some limitations which must be considered. We did not have liver biopsies in all patients. However, such studies have been performed and the association between LS and fibrosis score is well established for both HCV and ALD.[13], [38], [39] We also used the conventional FibroScan Touch with measurement failure of SS in 18% which could generate a study bias. It also remains unclear how different elastographic techniques such as acoustic radiation force impulse or magnetic resonance elastography will affect the ratio since they cannot be directly converted to FibroScan assess units and have a larger range of measurement. Echosens has recently introduced a device to allow measurements of higher SS values.12 Finally, the lack of SS in the retrospective cohort makes it impossible to conclude whether SS is a stronger prognostic factor than SL.
In summary, we have demonstrated the disease-specific differences in SS to LS and SL to LS ratio between HCV and ALD. They underscore the role of the intrahepatic histological site of inflammation/fibrosis. We suggest that the SS/LS ratio could be used to confirm the disease etiology and predict disease-specific complications.
Financial support
This study was supported by the Dietmar Hopp Foundation (grant 1DH1811387) to SM and, in part, by a grant of the DFG to SM (MU 1373/9-1). The Danish patient inclusions were funded by Innovation Fund Denmark.
Conflicts of interest
The authors declare no conflicts of interest that pertain to this work.
Please refer to the accompanying ICMJE disclosure forms for further details.
Author contributions
Concept and design: SM & OE. Data collection: All authors. Statistical analysis: JM & OE. Reviewing and revising the paper: SM, MT, IG & HS. Writing the paper: OE, JM & SM
Footnotes
Supplementary data associated with this article can be found, in the online version, at https://doi.org/10.1016/j.jhepr.2019.05.003.
Supplementary data
Supplementary material 1
Supplementary material 2
Supplementary material 3
References
- 1.D'Amico G, Garcia-Tsao G, Pagliaro L. Natural history and prognostic indicators of survival in cirrhosis: A systematic review of 118 studies. J Hepatol. 2006;44:217–231. doi: 10.1016/j.jhep.2005.10.013. [DOI] [PubMed] [Google Scholar]
- 2.Abraldes JG, Araujo IK, Turón F, Berzigotti A. Diagnosing and monitoring cirrhosis: Liver biopsy, hepatic venous pressure gradient and elastography. Gastroenterol Hepatol. 2012;35:488–495. doi: 10.1016/j.gastrohep.2012.02.010. [DOI] [PubMed] [Google Scholar]
- 3.Colecchia A, Montrone L, Scaioli E, Bacchi-Reggiani ML, Colli A, Casazza G. Measurement of Spleen Stiffness to Evaluate Portal Hypertension and the Presence of Esophageal Varices in Patients With HCV-Related Cirrhosis. Gastroenterology. 2012;143:646–654. doi: 10.1053/j.gastro.2012.05.035. [DOI] [PubMed] [Google Scholar]
- 4.Abraldes JG, Reverter E, Berzigotti A. Spleen stiffness: toward a noninvasive portal sphygmomanometer? Hepatology (Baltimore, Md) 2013;57:1278–1280. doi: 10.1002/hep.26239. [DOI] [PubMed] [Google Scholar]
- 5.Abraldes JG, Bureau C, Stefanescu H, Augustin S, Ney M, Blasco H. Noninvasive tools and risk of clinically significant portal hypertension and varices in compensated cirrhosis: The "Anticipate" study. Hepatology (Baltimore, Md) 2016 doi: 10.1002/hep.28824. [DOI] [PubMed] [Google Scholar]
- 6.Kim HY, Jin EH, Kim W, Lee JY, Woo H, Oh S. The Role of Spleen Stiffness in Determining the Severity and Bleeding Risk of Esophageal Varices in Cirrhotic Patients. Medicine (Baltimore) 2015;94 doi: 10.1097/MD.0000000000001031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Takuma Y, Nouso K, Morimoto Y, Tomokuni J, Sahara A, Takabatake H. Prediction of oesophageal variceal bleeding by measuring spleen stiffness in patients with liver cirrhosis. Gut. 2016;65:354–355. doi: 10.1136/gutjnl-2015-309741. [DOI] [PubMed] [Google Scholar]
- 8.Sharma P, Kirnake V, Tyagi P, Bansal N, Singla V, Kumar A. Spleen Stiffness in Patients With Cirrhosis in Predicting Esophageal Varices. Am J Gastroenterol. 2013;108:1101. doi: 10.1038/ajg.2013.119. [DOI] [PubMed] [Google Scholar]
- 9.Stefanescu H, Grigorescu M, Lupsor M, Procopet B, Maniu A, Badea R. Spleen stiffness measurement using fibroscan for the noninvasive assessment of esophageal varices in liver cirrhosis patients. J Gastroenterol Hepatol. 2011;26:164–170. doi: 10.1111/j.1440-1746.2010.06325.x. [DOI] [PubMed] [Google Scholar]
- 10.Abraldes JG, Villanueva C, Aracil C, Turnes J, Hernandez-Guerra M, Genesca J. Addition of Simvastatin to Standard Therapy for the Prevention of Variceal Rebleeding Does Not Reduce Rebleeding but Increases Survival in Patients With Cirrhosis. Gastroenterology. 2016;150:1160-70.e3. doi: 10.1053/j.gastro.2016.01.004. [DOI] [PubMed] [Google Scholar]
- 11.Colecchia A, Colli A, Casazza G, Mandolesi D, Schiumerini R, Reggiani LB. Spleen stiffness measurement can predict clinical complications in compensated HCV-related cirrhosis: A prospective study. J Hepatol. 2014;60:1158–1164. doi: 10.1016/j.jhep.2014.02.024. [DOI] [PubMed] [Google Scholar]
- 12.Bastard C, Miette V, Calès P, Stefanescu H, Festi D, Sandrin L. A Novel FibroScan Examination Dedicated to Spleen Stiffness Measurement. Ultrasound Med Biol. 2018;44:1616–1626. doi: 10.1016/j.ultrasmedbio.2018.03.028. [DOI] [PubMed] [Google Scholar]
- 13.Mueller S, Englert S, Seitz HK, Badea RI, Erhardt A, Bozaari B. Inflammation-adapted liver stiffness values for improved fibrosis staging in patients with hepatitis C virus and alcoholic liver disease. Liver Int. 2015;35:2514–2521. doi: 10.1111/liv.12904. [DOI] [PubMed] [Google Scholar]
- 14.Castera L, Forns X, Alberti A. Non-invasive evaluation of liver fibrosis using transient elastography. J Hepatol. 2008;48:835–847. doi: 10.1016/j.jhep.2008.02.008. [DOI] [PubMed] [Google Scholar]
- 15.Nguyen-Khac E, Thiele M, Voican C, Nahon P, Moreno C, Boursier J. Non-invasive diagnosis of liver fibrosis in patients with alcohol-related liver disease by transient elastography: an individual patient data meta-analysis. Lancet Gastroenterol Hepatol. 2018;3:614–625. doi: 10.1016/S2468-1253(18)30124-9. [DOI] [PubMed] [Google Scholar]
- 16.Mueller S, Millonig G, Sarovska L, Friedrich S, Reimann FM, Pritsch M. Increased liver stiffness in alcoholic liver disease: differentiating fibrosis from steatohepatitis. World J Gastroenterol. 2010;16:966–972. doi: 10.3748/wjg.v16.i8.966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Arena U, Vizzutti F, Corti G, Ambu S, Stasi C, Bresci S. Acute viral hepatitis increases liver stiffness values measured by transient elastography. Hepatology (Baltimore, Md) 2008;47:380–384. doi: 10.1002/hep.22007. [DOI] [PubMed] [Google Scholar]
- 18.Pons M, Santos B, Simon-Talero M, Ventura-Cots M, Riveiro-Barciela M, Esteban R. Rapid liver and spleen stiffness improvement in compensated advanced chronic liver disease patients treated with oral antivirals. Therap Adv Gastroenterol. 2017;10:619–629. doi: 10.1177/1756283X17715198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gelsi E, Dainese R, Truchi R, Marine-Barjoan E, Anty R, Autuori M. Effect of detoxification on liver stiffness assessed by fibroscan((R)) in alcoholic patients. Alcohol Clin Exp Res. 2011;35:566–570. doi: 10.1111/j.1530-0277.2010.01374.x. [DOI] [PubMed] [Google Scholar]
- 20.Scheuer PJ. Classification of chronic viral hepatitis: a need for reassessment. J Hepatol. 1991;13:372–374. doi: 10.1016/0168-8278(91)90084-o. [DOI] [PubMed] [Google Scholar]
- 21.Dhingra S, Ward SC, Thung SN. Liver pathology of hepatitis C, beyond grading and staging of the disease. World J Gastroenterol. 2016;22:1357–1366. doi: 10.3748/wjg.v22.i4.1357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Junge J, Horn T, Vyberg M, Christoffersen P, Svendsen LB. The pattern of fibrosis in the acinar zone 3 areas in early alcoholic liver disease. J Hepatol. 1991;12:83–86. doi: 10.1016/0168-8278(91)90914-w. [DOI] [PubMed] [Google Scholar]
- 23.Theise ND. Histopathology of alcoholic liver disease. Clin Liver Dis. 2013;2:64–67. doi: 10.1002/cld.172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mueller S. Does pressure cause liver cirrhosis? The sinusoidal pressure hypothesis. World J Gastroenterol. 2016;22:10482–10501. doi: 10.3748/wjg.v22.i48.10482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mueller S, Seitz HK, Rausch V. Non-invasive diagnosis of alcoholic liver disease. World J Gastroenterol. 2014;20:14626–14641. doi: 10.3748/wjg.v20.i40.14626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jérôme B, Jean-Pierre Z, Victor dL, Marie-Christine R, Nathalie S, Brigitte L. Determination of reliability criteria for liver stiffness evaluation by transient elastography. Hepatology (Baltimore, Md) 2013;57:1182–1191. doi: 10.1002/hep.25993. [DOI] [PubMed] [Google Scholar]
- 27.Colecchia A, Montrone L, Scaioli E, Bacchi-Reggiani ML, Colli A, Casazza G. Measurement of spleen stiffness to evaluate portal hypertension and the presence of esophageal varices in patients with HCV-related cirrhosis. Gastroenterology. 2012;143:646–654. doi: 10.1053/j.gastro.2012.05.035. [DOI] [PubMed] [Google Scholar]
- 28.Bach N, Thung SN, Schaffner F. The histological features of chronic hepatitis C and autoimmune chronic hepatitis: a comparative analysis. Hepatology (Baltimore, Md) 1992;15:572–577. doi: 10.1002/hep.1840150403. [DOI] [PubMed] [Google Scholar]
- 29.Fischer HP, Willsch E, Bierhoff E, Pfeifer U. Histopathologic findings in chronic hepatitis C. J Hepatol. 1996;24:35–42. [PubMed] [Google Scholar]
- 30.Bedossa P, Poynard T. An algorithm for the grading of activity in chronic hepatitis C. The METAVIR Cooperative Study Group. Hepatology (Baltimore, Md) 1996;24:289–293. doi: 10.1002/hep.510240201. [DOI] [PubMed] [Google Scholar]
- 31.Brunt EM, Tiniakos DG. 2009. CHAPTER 41 Fatty Liver Disease. Surgical Pathology of the GI Tract, Liver, Biliary Tract, and Pancreas; pp. 1087–1114. [Google Scholar]
- 32.Nakano M, Worner TM, Lieber CS. Perivenular fibrosis in alcoholic liver injury: ultrastructure and histologic progression. Gastroenterology. 1982;83:777–785. [PubMed] [Google Scholar]
- 33.Mueller S, Nahon P, Rausch V, Peccerella T, Silva I, Yagmur E. Caspase-cleaved keratin-18 fragments increase during alcohol withdrawal and predict liver-related death in patients with alcoholic liver disease. Hepatology (Baltimore, Md) 2017;66:96–107. doi: 10.1002/hep.29099. [DOI] [PubMed] [Google Scholar]
- 34.Millonig G, Friedrich S, Adolf S, Fonouni H, Golriz M, Mehrabi A. Liver stiffness is directly influenced by central venous pressure. J Hepatol. 2010;52:206–210. doi: 10.1016/j.jhep.2009.11.018. [DOI] [PubMed] [Google Scholar]
- 35.Bose SK, Shrivastava S, Meyer K, Ray RB, Ray R. Hepatitis C Virus Activates the mTOR/S6K1 Signaling Pathway in Inhibiting IRS-1 Function for Insulin Resistance. J Virol. 2012;86:6315. doi: 10.1128/JVI.00050-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rausch V, Silva I, Peccerella T, Mueller S. Masked hemolysis as novel factor contributing to hepatic iron overload in ALD. J Hepatol. 2018;68:S815-S6. [Google Scholar]
- 37.Lieber CS, Weiss DG, Morgan TR, Paronetto F. Aspartate Aminotransferase to Platelet Ratio Index in Patients with Alcoholic Liver Fibrosis. Am J Gastroenterol. 2006;101:1500. doi: 10.1111/j.1572-0241.2006.00610.x. [DOI] [PubMed] [Google Scholar]
- 38.Castera L, Vergniol J, Foucher J, Le Bail B, Chanteloup E, Haaser M. Prospective comparison of transient elastography, Fibrotest, APRI, and liver biopsy for the assessment of fibrosis in chronic hepatitis C. Gastroenterology. 2005;128:343–350. doi: 10.1053/j.gastro.2004.11.018. [DOI] [PubMed] [Google Scholar]
- 39.Thiele M, Detlefsen S, Sevelsted Møller L, Madsen BS, Fuglsang Hansen J, Fialla AD. Transient and 2-Dimensional Shear-Wave Elastography Provide Comparable Assessment of Alcoholic Liver Fibrosis and Cirrhosis. Gastroenterology. 2016;150:123–133. doi: 10.1053/j.gastro.2015.09.040. [DOI] [PubMed] [Google Scholar]
- 40.Hakala BE, White C, Recklies AD. Human cartilage gp-39, a major secretory product of articular chondrocytes and synovial cells, is a mammalian member of a chitinase protein family. J Biol Chem. 1993;268 [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material 1
Supplementary material 2
Supplementary material 3