Abstract
Aims/hypothesis
Prediabetes is associated with postprandial hypertriacylglycerolaemia. Resistance exercise acutely lowers postprandial plasma triacylglycerol (TG); however, the changes in lipid metabolism that mediate this reduction are poorly understood. The aim of this study was to identify the constitutive metabolic mechanisms underlying the changes in postprandial lipid metabolism after resistance exercise in obese men with prediabetes.
Methods
We evaluated the effect of a single bout of whole-body resistance exercise (seven exercises, three sets, 10–12 repetitions at 80% of one-repetition maximum) on postprandial lipid metabolism in ten middle-aged (50±9 years), overweight/obese (BMI: 33 ± 3 kg/m2), sedentary men with prediabetes (HbA1c >38 but <48 mmol/mol [>5.7% but <6.5%]), or fasting plasma glucose >5.6 mmol/l but <7.0 mmol/l or 2 h OGTT glucose >7.8 mmol/l but <11.1 mmol/l). We used a randomised, crossover design with a triple-tracer mixed meal test (ingested [(13C4)3]tripalmitin, i.v. [U-13C16]palmitate and [2H5]glycerol) to evaluate chylomicron-TG and total triacylglycerol-rich lipoprotein (TRL)-TG kinetics. We used adipose tissue and skeletal muscle biopsies to evaluate the expression of genes regulating lipolysis and lipid oxidation, skeletal muscle respirometry to evaluate oxidative capacity, and indirect calorimetry to assess whole-body lipid oxidation.
Results
The single bout of resistance exercise reduced the lipaemic response to a mixed meal in obese men with prediabetes without changing chylomicron-TG or TRL-TG fractional clearance rates. However, resistance exercise reduced endogenous and meal-derived fatty acid incorporation into chylomicron-TG and TRL-TG. Resistance exercise also increased whole-body lipid oxidation, skeletal muscle mitochondrial respiration, oxidative gene expression in skeletal muscle, and the expression of key lipolysis genes in adipose tissue.
Conclusions/interpretation
A single bout of resistance exercise improves postprandial lipid metabolism in obese men with prediabetes, which may mitigate the risk for cardiovascular disease and type 2 diabetes.
Keywords: Chylomicron, Lipid oxidation, Lipoprotein, Obesity, Postprandial, Prediabetes, Resistance exercise, Stable isotope tracer, VLDL
Introduction
Sedentary behaviour is associated with dyslipidaemia and insulin resistance, type 2 diabetes and cardiovascular disease (CVD) (1–3). There are also strong correlations between postprandial triacylglycerol (TG) concentrations and the risk of future cardiovascular events, as well as chylomicron remnant concentrations and the progression of coronary lesions, indicating that postprandial dyslipidaemia may be a potent contributor to CVD and a key target for treatment (4,5). Endurance and resistance-type exercise acutely lower fasting and postprandial plasma TG concentrations in men and women who are healthy, obese or have type 2 diabetes, and are part of the standard of care for patients with obesity, prediabetes, and type 2 diabetes (6–8). However, these improvements may not be equivalent between men and women. There is evidence for sexual dimorphism in lipid metabolism in healthy individuals, with men demonstrating more pronounced postprandial lipaemia, but greater improvements in postprandial TG, lipid oxidation and resting metabolic rate after exercise than women (9–11). Overall, the mechanisms of reduced postprandial lipaemia after a prior bout of exercise are incompletely understood.
Increased VLDL-TG and chylomicron-TG plasma clearance and oxidation play a central role in the reduction of postprandial TG after aerobic exercise (12,13). Aerobic exercise has been shown to reduce VLDL-TG by increasing VLDL size (assessed as the VLDL-TG/VLDL-apolipoprotein B-100 ratio), which increases VLDL affinity for lipoprotein lipase (LPL) (12). Moreover, using an isotopically labelled test meal, it was previously shown that aerobic exercise reduces endogenous, but not exogenous (meal-derived), TG concentrations in obese women (14). It is unclear if aerobic exercise affects the TG content of chylomicron-TG, but it does not change chylomicron LPL affinity, which may explain why chylomicron concentrations do not change to the same extent as VLDL-TG after aerobic exercise (12,15). In addition, since VLDL-TG secretion accounts for 18–20% of normal postprandial TG variation, changes in lipoprotein secretion (VLDL and/or chylomicrons) may also contribute to reduced postprandial lipaemia after aerobic exercise (16).
In contrast, much less is known about the effects of resistance exercise on postprandial lipid metabolism. In healthy, and metabolically normal, obese men and women, resistance exercise acutely (30 min post exercise) increases total postprandial NEFA oxidation, and augments VLDL-TG plasma clearance, but not secretion, after an overnight fast (14,17). In men and women with type 2 diabetes, post-meal resistance exercise reduced postprandial plasma TG by reducing the VLDL-TG concentration (8). In obese women, use of an isotopically labelled test meal demonstrated that resistance exercise increases exogenous postprandial lipid oxidation, reduces postprandial endogenous TG concentrations, and increases postprandial endogenous NEFA concentrations (14). However, because a separate tracer was not used to track endogenous NEFA kinetics, measures of adipose tissue lipolysis, endogenous NEFA clearance or insulin resistance—key determinants of postprandial lipaemia—were not measured (18). Moreover, chylomicrons and VLDL were not separated for tracer enrichment analysis, so it is not known if there are differential effects of resistance exercise on chylomicron vs VLDL handling or clearance.
Therefore, we aimed to perform a comprehensive analysis of postprandial lipid metabolism following an acute bout of resistance exercise in overweight/obese men with prediabetes. Using a novel approach employing three stable isotope tracers, we hypothesised that resistance exercise would reduce postprandial TG levels by improving insulin sensitivity (which may reduce adipose tissue lipolysis and reduce NEFA release into the plasma), reducing lipoprotein endogenous and exogenous NEFA concentrations in triacylglycerol-rich lipoprotein (TRL)-TG, increasing whole-body and skeletal muscle mitochondrial lipid oxidation to augment lipoprotein and NEFA clearance, and increasing the expression of genes regulating lipolysis and lipid oxidation in adipose tissue and skeletal muscle.
Methods
Participants
Participants were recruited from Washington University in St Louis (WUSTL) School of Medicine Diabetes Clinic, the WUSTL Volunteers for Health, the Center for Community Based Research databases, and advertising in the St Louis metropolitan area from August 2015 to May 2018. Potential participants completed a detailed screening evaluation including a history and physical examination, serum blood chemistries (HbA1c, a lipid panel, a comprehensive metabolic panel and a complete blood count) and an OGTT (see electronic supplementary material (ESM) Methods for further details). Participants were included if they were male, aged 30–65 years with a BMI of 28–45 kg/m2, and met the criteria for prediabetes (HbA1c >38 but <48 mmol/mol [>5.7% but <6.5%]), or fasting plasma glucose >5.6 mmol/l but <7.0 mmol/l or 2 h OGTT glucose >7.8 mmol/l but <11.1 mmol/l) (19). Participants were excluded if they were diagnosed with type 2 diabetes, used insulin or other blood-sugar-lowering medications, had participated in regular exercise (≥2 times/week) within the previous 6 months (assessed by questionnaire during the initial phone interview), had a history of pulmonary or cardiovascular disease, coagulation disorders, anaemia or orthopaedic, neurological, metabolic or other medical condition that would prohibit them from participating in the exercise and metabolic testing protocol. Participants also completed a one-repetition maximum (1RM) strength assessment, daily physical activity monitoring and body composition measurements using dual x-ray absorptiometry (DXA, see ESM Methods for further details). The study was approved by the WUSTL Human Research Protection Office (protocol no. 20160495) and all participants provided written informed consent prior to enrolment. All procedures were completed in accordance with the ethical principles outlined in the Declaration of Helsinki.
Participant demographics
Participants had a mean age of 50 years, were obese and demonstrated insulin resistance with compensatory increases in beta cell function. Participants also presented with elevated mean total triacylglycerol and LDL-cholesterol concentrations and a low HDL-cholesterol concentration, and were sedentary for the majority of the day (Table 1).
Table 1.
Participant demographics
| Measure | Mean (SD) |
|---|---|
| Age (years) | 50 (9) |
| Height (cm) | 178 (6) |
| Weight (kg) | 105 (12) |
| BMI (kg/m2) | 33 (3) |
| Fat mass (kg) | 33 (6.4) |
| Lean mass (kg) | 67 (6.2) |
| Body fat (%) | 31.6 (4) |
| HbA1c (mmol/mol) | 39 (3.4) |
| HbA1c (%) | 5.7 (0.5) |
| Fasting glucose (mmol/1) | 5.9 (0.7) |
| 2-hr OGTT glucose (mmol/1) | 9.1 (0.5) |
| Fasting insulin (pmol/1) | 3.2 (0.86) |
| HOMA2-IR | 2.6 (1.2) |
| HOMA2-%B | 139.4 (56) |
| HOMA2-%S | 46 (16) |
| Triacylglycerol (mmol/1) | 1.9 (0.27) |
| Total cholesterol (mmol/1) | 4.8 (0.83) |
| HDL-cholesterol (mmol/1) | 0.99 (0.13) |
| LDL-cholesterol (mmol/1) | 3.0 (0.75) |
| Time sedentary (%) | 69 (19.2) |
| Time light activity (%) | 21 (11.4) |
| Time moderate activity (%) | 10 (9.5) |
| Time vigorous activity (%) | 0 (0) |
Times spent in each activity were derived from accelerometry data (see ESM methods). 2h OGTT is plasma glucose at 2 h after a 75 g oral glucose challenge. HOMA2 was calculated using the HOMA2 calculator (see ESM methods). HOMA2-%B, HOMA2 of steady state beta cell function; HOMA2-%S, HOMA2 of insulin sensitivity
Study protocol
Lipid metabolism study
Each participant completed two lipid metabolism studies (exercise and rest) in randomised order, approximately 1 week apart (Fig. 1). Treatment order (rest or exercise) was assigned through simple random sampling without replacement (see Electronic supplementary material (ESM) Methods). Between studies participants were instructed to maintain their normal diet and daily activity level. During the exercise study, participants completed a 1 h bout of whole-body resistance exercise 270 min before meal ingestion; for the rest study, participants rested in a chair for 1 h.
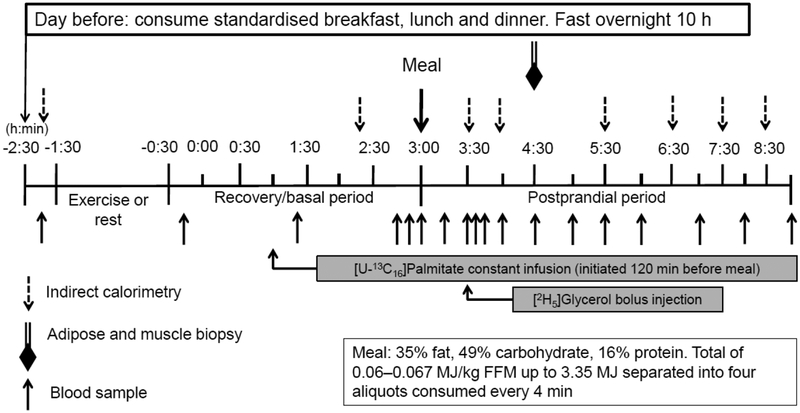
Fig. 1.
Study protocol
The day prior to each lipid metabolism study, participants were provided a standardised breakfast (2.18 MJ), lunch (3.02 MJ), afternoon snack (0.75 MJ), and dinner (3.26 MJ) from the WUSTL Clinical and Translational Research Unit (CTRU) Bionutrition Service. They consumed a liquid formula (Ensure; Ross Laboratories, Columbus, OH, USA) containing 1.05 MJ at ~22:00 hours to ensure complete filling of hepatic glycogen stores. The carbohydrate, fat and protein contents for each meal are provided in the ESM. Participants then fasted overnight (10 h) except for water and reported to the CTRU at 07:30 hours the following morning. After admission, an i.v. catheter was inserted into the antecubital vein to collect baseline blood samples and for later for tracer infusions.
During the rest period, participants rested quietly in the semi-recumbent position for 60 min from 09:00 hours to 10:00 hours. During the exercise study, participants performed 1 h (from 09:00 hours – 10:00 hours) of resistance exercise consisting of three sets of 10–12 repetitions at 80% of 1RM of the following seven exercises: leg press, knee extension, chest press, shoulder press, seated row, pull down and bicep curl (See ESM).
Following the rest or resistance exercise period, a second catheter was inserted into a hand vein on the contralateral arm; the hand was warmed with a warming box (55°C) to collect arterialised blood samples. To allow time for catheter insertion, the study timer was started (study time 0) 30 min post exercise or post rest. At 60 min, a constant infusion of [U-13C16]palmitate (5.3 nmol·kg−1·min−1) bound to human albumin was initiated and continued until the end of the study. At 180 min, participants were given a liquid meal (Boost Plus; Fremont, MI, USA) consisting of 0.08 MJ/kg fat-free mass (47.8% carbohydrate, 36.1% fat, and 16.1% protein). [1,1,1,2,2,2,3,3,3,4,4,4-(13C4)3]Tripalmitin was added to the test meal and thoroughly mixed. A description of meal preparation is included in the ESM. At 210 min, a 75 μmol/kg body weight bolus of [1,1,2,3,3-2H5]glycerol was administered by injection to the final seven participants who completed the study. Blood samples were collected at −10, 160, 170, 180, 196, 210, 215, 225, 240, 270, 300, 330, 360, 420, 480, and 540 min (Fig. 1). All stable isotope tracers were obtained from Cambridge Isotope Laboratories (Tewksbury, MA, USA).
At 270 min (90 min post meal), biopsy samples were obtained from the vastus lateralis muscle and abdominal subcutaneous adipose tissue. The whole-body lipid oxidation rate was measured at baseline (−90 min), immediately before the resistance exercise/rest period and at 140, 210, 240, 330, 390, 450, and 510 min using indirect calorimetry, as previously described (20).
Sample processing
Methods for isolating TRL and chylomicron fractions, and measuring palmitate and glycerol enrichments are described in the ESM Methods. Total plasma, TRL, and chylomicron-TG concentrations were measured using a colorimetric assay (Wako Diagnostics, Richmond VA, USA). VLDL-TG concentrations were measured by subtracting chylomicron-TG concentration from TRL-TG concentration. Plasma insulin was measured using electrochemiluminescence immunoassay (Elecsys, Roche Diagnositics, Mannheim, Germany). Plasma glucose was measured using the glucose oxidase method (Yellow Springs Instrument, Yellow Springs, OH, USA).
Tissue biopsies, mitochondrial respiration and RT-qPCR
Biopsy samples from the vastus lateralis muscle and abdominal subcutaneous adipose tissue were collected under local anaesthesia as previously described (21,22). A 6–10 mg sample of skeletal muscle was used for mitochondrial high-resolution respirometry analysis (Oxygraph-2k, Oroboros Instruments, Innsbruck, Austria; see ESM methods and ESM Tables 1 and 2). Remaining muscle and adipose tissue samples were irrigated with saline (150 mmol/l NaCl), flash frozen in liquid nitrogen and stored at −80° until analysis. Gene expression was determined using RT-qPCR (see ESM methods and ESM Table 3). Data were log2-transformed for statistical analyses and back-transformed for presentation.
Calculations
The concentrations of [13C4]palmitate and [U-13C16]palmitate in TRL-TG, chylomicron-TG, and plasma palmitate were calculated using palmitate concentration and tracer enrichments in the respective pools (see ESM Methods). Basal (fasted) palmitate rate of appearance (Ra) into plasma was calculated by dividing the tracer infusion rate by the plasma palmitate tracer to tracee ratio (TTR) (23). The basal adipose tissue insulin resistance index (ATIRI) was calculated as the product of basal palmitate Ra and plasma insulin concentration (23).
The total AUC was calculated using the trapezoid rule (24); incremental AUCs (iAUCs) were obtained after adjusting for basal values (24). Fractional clearance rates (FCR) of TRL-TG and chylomicron-TG were determined using the monoexponential decay rate of TG 2H5-glycerol from 2–6 h after the start of the meal (1.5–5.5 h after the bolus of 2H5-glycerol tracer) as previously described (25).
Statistical analysis
Differences in outcome measures between rest and exercise were assessed using repeated measures ANOVA with treatment (rest/RE) and time (before/after meal) entered as repeated factors, and order (rest>RE or RE>rest) as a between-subjects factor. Significant interactions between treatment and time were further analysed using paired t-tests. The Benjamini–Hochberg procedure was used to control for multiple comparisons for all repeated measures. AUCs, respiration, lipid oxidation and gene expression measures were analysed using paired t-tests (if normally distributed) or the Wilcoxon signed-rank test (if non-normally distributed) between rest and exercise. An α level of 0.05 was used for significance. Cohen’s d (paired t-tests), r (Wilcoxon signed-rank tests) and partial η2 (repeated measures ANOVA) were to calculate effect size. Previous studies have reported effect sizes ranging from 0.78 (reduced plasma TG by 14–24%) to 1.85 for the impact of resistance exercise on postprandial triacylglycerol concentrations (7,26). Based on these findings, using an effect size of 1.0, we determined a sample size of ten participants was needed to detect a significant treatment effect at a two-tailed α level of <0.05 with 80% power (See ESM Statistical Analysis and Sample Size Determination). All analyses were completed in SPSS version 25 (IBM, Armonk, NY, USA) on al 10 subjects unless otherwise specified.
Results
Basal period NEFA kinetics and insulin resistance
Basal (fasted state prior to the meal test) plasma fatty acid concentrations, palmitate rates of appearance (Ra) and disappearance (Rd), and ATIRI were not significantly different between rest and resistance exercise (Table 2).
Table 2.
Postprandial iAUC, basal NEFA kinetics and postprandial lipoprotein kinetics
| Measure | Rest | Exercise | p value | Effect size |
|---|---|---|---|---|
| Basal NEFA concentration (mmol/1) | 0.91 [0.65–2.75; 0.42] | 0.88 [0.45–1.83; 0.40] | 0.44 | 0.17 |
| Basal palmitate concentration (mmol/1) | 0.24 [0.16–0.67; 0.05] | 0.22 [0.14–0.43; 0.16] | 0.57 | 0.13 |
| Basal palmitate Ra (μmol [kg FM]−1 min−1) | 5.3 (1.7) | 6.5 (2.4) | 0.15 | 0.57 |
| Basal palmitate Rd (μmol [kg FFM]−1 min−1) | 2.4 [1.5–4.6; 1.6] | 2.5 [1.9–7.1; 2.2] | 0.16 | 0.31 |
| Basal ATIRI (μmol/min × pmol/1) | 394 (231) | 454 (215) | 0.41 | 0.24 |
| Postprandial chylomicron-TG iAUC (mmol/1 × 360 min) | 43.9 [18.4–174.9; 77.8] | 33.5 [21.4–140.7; 38.8] | 0.02 | 0.51 |
| Postprandial VLDL-TG iAUC (mmol/1 × 360 min) | 100 (85) | 59 (79) | 0.06 | 0.68 |
| Postprandial TRL-TG iAUC (mmol/1 × 360 min) | 126.6 [51.1–346.4; 157.2] | 96.3 [−20–295.7; 92.5] | 0.01 | 0.58 |
| Postprandial plasma NEFA iAUC (mmol/1 × 360 min) | −113 [−195−−25; 79] | −79 [−432 – −40; 81] | 0.96 | 0.01 |
| Postprandial total plasma TG iAUC (mmol/1 × 360 min) | 135 [26.8–354.3; 174] | 97.9 [23.3–318.1; 185.2] | 0.01 | 0.63 |
| Postprandial plasma palmitate iAUC (mmol/1 × 360 min) | −30 (16) | −35 (30) | 0.61 | 0.19 |
| Postprandial plasma insulin iAUC (pmol/1 × 360 min) | 4108 (924) | 3282 (1187) | 0.03 | 0.83 |
| Postprandial TRL-TG FCR (pools/h) | 0.25 (0.17) | 0.33 (0.19) | 0.21 | 0.46 |
| Postprandial chylomicron-TG FCR (pools/h) | 0.42 (0.22) | 0.49 (0.21) | 0.34 | 0.32 |
Data are presented as mean (±SD) for normally distributed data, or median [minimum–maximum; interquartile range] for non-normally distributed data. ATIRI, adipose tissue insulin resistance index (higher = more insulin resistant). Rd, rate of disappearance. FM, fat mass. FFM, fat-free mass. Chylo, chylomicron. FCR calculations were made from n=7 participants (see Methods). The iAUC is negative for postprandial plasma NEFA, representing the area above the curve extending up to the baseline (180 min) value as an index of postprandial NEFA suppression. Basal NEFA and palmitate concentration is the concentration at 180 min. Significant difference between rest and exercise if p<0.05 for paired t-tests (parametric) or Wilcoxon signed-rank test (non-parametric). Effect sizes are calculated using Cohen’s d (paired t-tests) or r (Wilcoxon signed-rank test)
Postprandial total plasma triacylglycerol, NEFA and insulin concentration
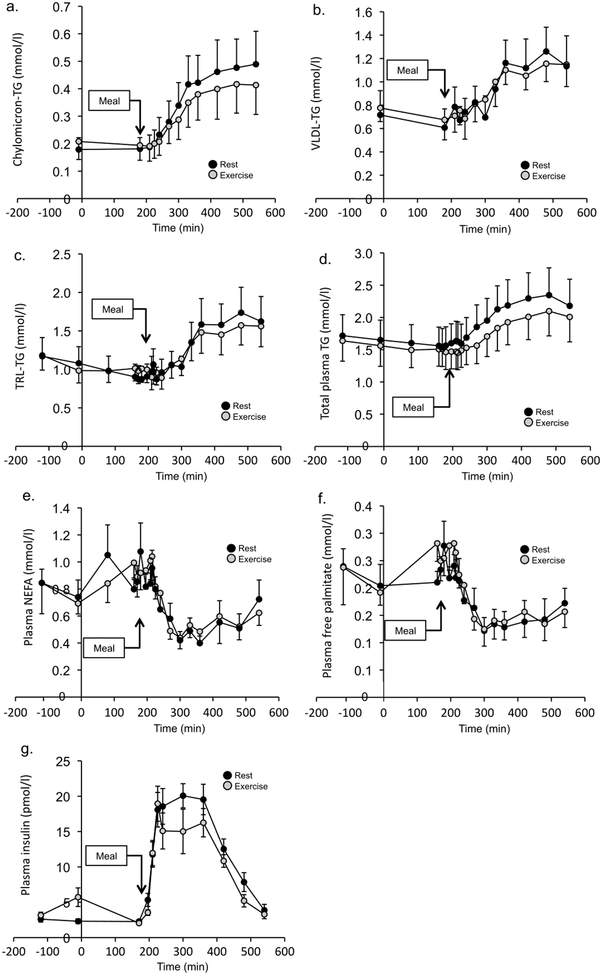
Changes in chylomicron-TG, VLDL-TG, TRL-TG, total plasma TG, plasma NEFA and plasma free palmitate are shown in Fig. 2. Although there was no statistically significant difference in plasma TG concentration after rest and resistance exercise at any given time point, the postprandial iAUC for chylomicron-TG, and TRL-TG concentrations were significantly lower after resistance exercise than after rest (Table 2). The iAUC was also significantly reduced after resistance exercise for total plasma TG. VLDL-TG concentration iAUC was not lower after resistance exercise than rest (Table 2). The postprandial plasma NEFA concentration iAUC following RE was not significantly different to that after rest; however, the plasma insulin concentration iAUC was significantly lower after resistance exercise than after rest (Table 2).
Fig. 2.
TG and NEFA concentration time courses. (a–f) Postprandial TG concentrations in the rest and resistance exercise conditions for chylomicron-TG (a), VLDL-TG (b), TRL-TG (c), total plasma TG (d), plasma NEFA (e) and plasma free palmitate (f). (g) Plasma insulin concentration is shown in in the rest and resistance exercise conditions. Values are mean ± SEM (n=10)
Meal-derived postprandial lipid tracer kinetics
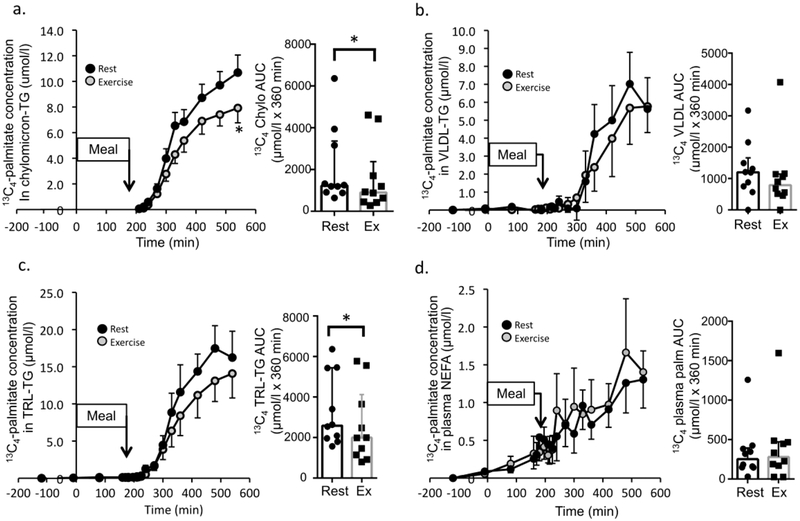
Meal-derived [13C4]palmitate enrichment and concentration in chylomicron-TG, VLDL-TG, and TRL-TG, and the plasma free palmitate pool were quantified throughout the postprandial period to determine meal-derived (exogenous) lipid handling. [13C4]palmitate concentration in chylomicron-TG (treatment × time interaction, p=0.014, partial η2=0.23) and TRL-TG (main effect of treatment, p=0.044, partial η2=0.17) were significantly lower after resistance exercise than rest (Fig. 3a,c). [13C4]Palmitate TTR (ESM Fig. 1c) and concentration (Fig. 3d) in the plasma free palmitate pool, and the [13C4]palmitate concentration in VLDL-TG (Figure 3b) was not significantly different between the resistance exercise and rest conditions. The [13C4]palmitate TTR was not different after resistance exercise in chylomicron-TG or TRL-TG (ESM Figure 1a,b). Total AUC for exogenous palmitate concentration in chylomicron-TG (p=0.039, d=0.85) and TRL-TG (p=0.036, d=0.78) was significantly lower following resistance exercise compared with rest (Figure 3a,c).
Fig. 3.
Meal-derived lipid tracer metabolism. The [13C4]palmitate concentration is shown for chylomicron-TG (a), VLDL-TG (b), TRL-TG (c) and in plasma NEFA (d) (n=10). The corresponding AUC (median ± interquartile range) for the concentration data are shown in the bar graph immediately to the right in (a–d) (n=10). ‘Meal’ indicates timing of mixed meal consumption. Time course values are mean ± SEM. * Denotes a time point significantly different between rest and exercise (time course data, repeated measures ANOVA) or a significant difference between rest and exercise for tracer concentration AUC (Wilcoxon signed-rank test) (p<0.05).
Infused postprandial lipid tracer concentrations
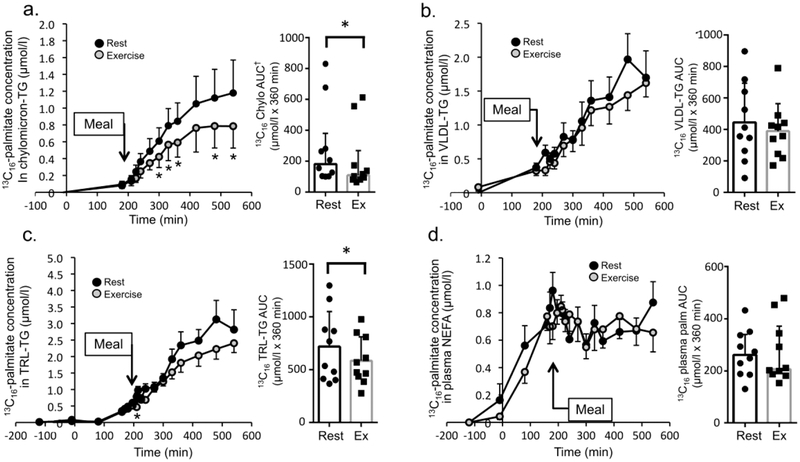
[U-13C16]Palmitate TTR and concentration in chylomicron-TG, VLDL-TG and TRL-TG, and the plasma free palmitate pool were quantified throughout the postprandial period to evaluate utilisation of plasma fatty acids. [U-13C16]Palmitate TTR in chylomicron-TG (ESM Fig. 1d, main effect of treatment, p=0.010, partial η2=0.62), [U-13C16]palmitate concentration in chylomicron-TG (Fig. 4a, interaction treatment × time, p=0.004, partial η2=0.32), and [U-13C16]palmitate concentration in TRL-TG (Fig. 4c, interaction treatment × time, p=0.020, partial η2=0.28) was significantly lower after resistance exercise than after rest. The [U-13C16]palmitate concentration in VLDL-TG (Fig. 4b, p=0.22) was not significantly different between the rest and resistance exercise conditions. The [U-13C16]palmitate TTR was not different between rest and resistance exercise for TRL-TG or plasma free palmitate (ESM Fig. 1e,f,). However, the AUC for [U-13C16]palmitate TTR (ESM Fig. 1d, p=0.012, d=1.06) and the AUC for [U-13C16]palmitate concentration in chylomicron-TG (Fig. 4a, p=0.008, d=1.10) was lower following resistance exercise than after rest, as was the AUC for [U-13C16]palmitate concentration in TRL-TG (Fig. 4c, p=0.014, d=0.963). The AUC for [U-13C16]palmitate concentration in VLDL-TG (Fig. 4b, p=0.16,) and in plasma NEFA (Fig. 4d, p=0.89) were not significantly different between the resistance exercise and rest conditions. The lack of a difference in the postprandial free [U-13C16]palmitate TTR (ESM Fig. 1f) or the plasma palmitate concentration (p=0.63, Fig. 2f) between rest and resistance exercise indicates that there was no significant difference in the postprandial plasma palmitate Ra between conditions.
Fig. 4.
Infused lipid tracer metabolism. The [U-13C16]palmitate concentration is shown for chylomicron-TG (a), VLDL-TG (b), TRL-TG (c), and in plasma NEFA (d) (n=10). The corresponding AUC (mean ± SD unless otherwise noted) for the concentration data are shown in the bar graph immediately to the right in (a–d). ‘Meal’ indicates timing of mixed meal consumption. Time course values are mean ± SEM. * Denotes time point significantly different between rest and exercise (time course data), or a significant difference between rest and exercise for tracer concentration AUC (paired t test for normally distributed data or Wilcoxon signed-rank test for non-normally distributed data) (p<0.05). † Denotes use of median ± interquartile range for non-normally distributed data
Postprandial lipoprotein FCR
The FCR for TRL-TG (p=0.21) and chylomicron-TG (p=0.34) were not significantly different between the resistance exercise and rest conditions (Table 2).
Lipid oxidation
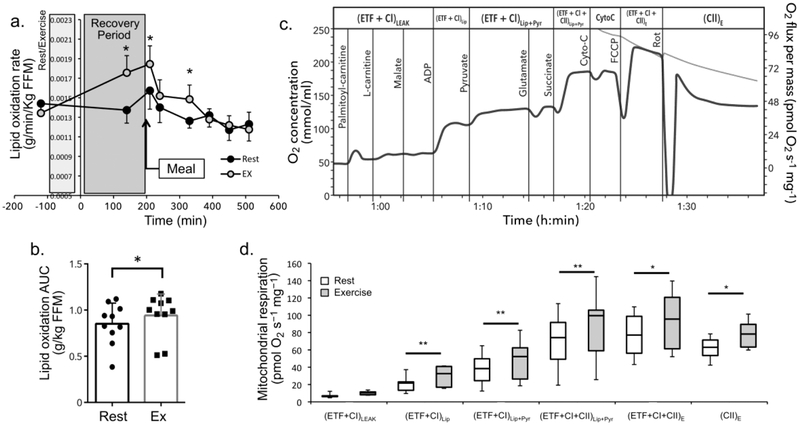
The whole-body lipid oxidation rate was significantly elevated (significant interaction treatment × time p=0.004, partial η2=0.34) at 140, 210, and 330 min after resistance exercise compared with after rest (Fig. 5a). The lipid oxidation AUC was significantly greater after resistance exercise than after rest (p=0.049, d=0.69) (Fig. 5b).
Fig. 5.
Changes in lipid oxidation after resistance exercise. (a) Lipid oxidation rate (g min−1 [kg FFM]−1, mean ± SEM) in the rest and exercise conditions. ‘Meal’ indicates timing of mixed meal consumption. Grey box labelled ‘Rest/exercise’ denotes timing of the rest or exercise session. Box labelled ‘Recovery period’ denotes timing of the 180 min recovery period. There was a significant interaction between treatment and time for lipid oxidation rate (see Results for details). (b) AUC (from −120 to 540 min, mean ± SD) for total lipid oxidation in the rest and exercise conditions. (c) An example of a respiration curve recorded from permeabilised muscle fibres from the vastus lateralis. Vertical lines denote the timing of substrate titrations. The substrate added at each time point is listed to the left of each line. Subsequent substrates were added only after O2 flux reached a plateau as shown. Respiratory states are listed along the top. For corresponding definitions, see ESM Table 1. (d) Box plots displaying oxygen consumption for the respiratory states assessed using the substrate-uncoupler-inhibitor titration (SUIT) protocol (see ESM Table 1). Resistance exercise significantly increased complex I-supported lipid [(ETF + CI)Lip] and lipid + pyruvate respiration [(ETF + CI)Lip+Pyr], as well as lipid + pyruvate respiration through complexes I and II [(ETF + CI + CII)Lip+Pyr], maximal electron transport capacity [(ETF + CI + CII)E] (uncoupled respiration) and complex II-supported respiration [(CII)E] (measured after inhibition of complex I with rotenone). ETF, electron transfer flavoprotein. * Denotes significantly different than rest at p<0.05, **p<0.01, (n=10)
Skeletal muscle mitochondrial respiration
Muscle biopsies were obtained from eight participants. A representative trace of mitochondrial respiration is shown in Fig. 5c. Maximal lipid oxidation (p=0.004, d=1.46), complex I-supported lipid + pyruvate oxidation (p=0.003, d=1.53), oxidative phosphorylation through complex I and II (p=0.005, d=1.38), electron transport capacity (p=0.017, d=1.09), and complex II-supported respiration (p=0.022, d=1.03) rates were higher following resistance exercise compared with rest (Fig. 5d). Leak respiration was not significantly different between conditions (p=0.260) (Fig. 5d). Oxidative phosphorylation control ratio and respiratory control ratio was significantly higher following resistance exercise compared with the rest condition; however, the phosphorylation control ratio and the leak control ratio were not different between resistance exercise and rest (ESM Table 4). Complex I-supported lipid respiration normalised to maximal oxidative phosphorylation, complex I-supported lipid respiration normalised to maximal electron transport capacity, the lipid to glucose oxidation ratio, and the free oxidative phosphorylation capacity were significantly higher following resistance exercise than following rest (ESM Table 4).
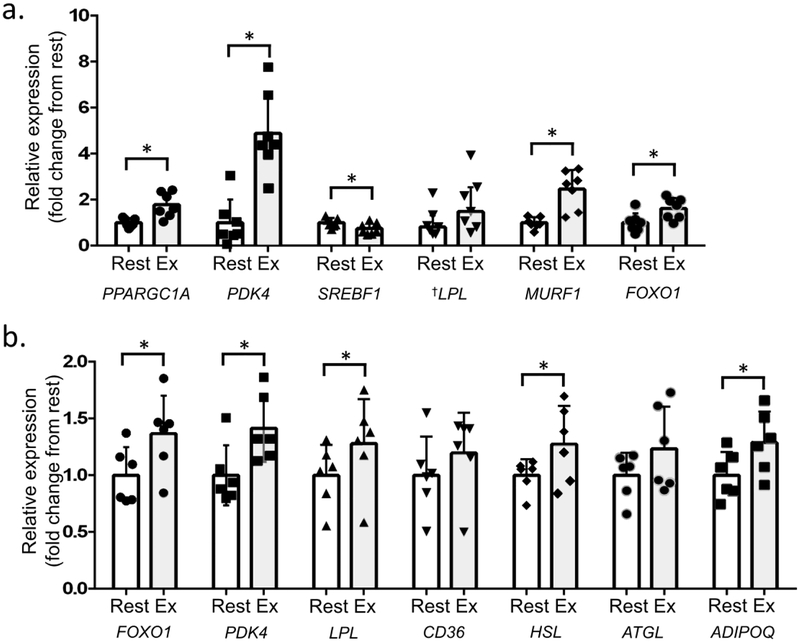
Skeletal muscle and adipose tissue gene expression
Total RNA was isolated from muscle biopsy samples from eight participants. However, RNA in one sample was degraded; therefore, data were analysed from samples for qPCR analysis for seven participants only. PPARGC1A (p=0.010, d=1.49), PDK4 (p=0.008, d=1.54), FOXO1 (p=0.037, d=0.98), and MURF1 (also known as TRIM63, p=0.002, d=1.63) expression in skeletal muscle was significantly higher following resistance exercise compared with rest (Fig. 6a). LPL expression in skeletal muscle was higher after resistance exercise compared with after rest, but it did not reach statistical significance (p=0.064) (Fig. 6a). SREBF1 expression in skeletal muscle was significantly lower after resistance exercise than after rest (p=0.020, d=1.23) (Fig. 6a). Expression of skeletal muscle FASN, ACACA, CD36, and CPT1A were not significantly different between conditions (not shown).
Fig. 6.
Skeletal muscle and abdominal subcutaneous adipose tissue gene expression. (a) Resistance exercise significantly increased skeletal muscle expression of PPARGC1A, PDK4, FOXO1 and MURF1, and non-significantly increased LPL gene expression. The exercise session also significantly reduced expression of SREBF1 relative to rest (n=7 for skeletal muscle gene expression). (b) Resistance exercise significantly increased adipose tissue expression of FOXO1, PDK4, LPL, HSL and ADIPOQ, but did not significantly change CD36 or ATGL expression (n=6 for all genes). Values are mean fold change above rest (mean ± SD) unless otherwise indicated. * Significant difference between rest and resistance exercise at p<0.05 for paired t tests (parametric) or Wilcoxon signed-rank tests (non-parametric). See results for corresponding p values. † Denotes use of median ± interquartile range for non-normally distributed data
Abdominal subcutaneous tissue biopsies were collected from six participants. Adipose FOXO1 (d=1.47), PDK4 (d=1.57), LPL (d=1.04), HSL (d=1.95) and adiponectin ADIPOQ (d=1.41) expression was significantly higher following exercise than following rest (all p<0.05, Fig. 6b). Adipose tissue CD36 expression was not changed (p=0.068) following resistance exercise compared with following rest (Fig. 6b). Adipose triglyceride lipase (ATGL) expression (p=0.110), likewise, was not significantly different between conditions (Fig. 6b).
Discussion
We found that a single 1 h bout of resistance exercise 4.5 h prior to consumption of a mixed meal reduced total plasma TG, chylomicron-TG iAUC and TRL-TG iAUC but did not reduce VLDL-TG iAUC in men with prediabetes. In addition, we demonstrated that resistance exercise before a meal reduces the incorporation of both meal-derived and endogenous plasma fatty acids into TRL-TG and chylomicron-TG. Resistance exercise also increased whole-body postprandial lipid oxidation, which was further supported by increases in skeletal muscle lipid oxidation gene expression, lipoprotein lipase gene expression and skeletal muscle mitochondrial lipid respiration. Moreover, we demonstrated the novel finding that a single bout of resistance exercise significantly increased the expression of genes that enhance lipid metabolism in subcutaneous adipose tissue. Taken together, these data establish that resistance exercise acutely reduces postprandial lipaemia in men with prediabetes and provides insight into some of the underlying mechanisms. Additional studies of chronic resistance exercise training are needed to extend these findings.
In this study, a prior bout of resistance exercise reduced [13C4]palmitate (exogenous) and [U-13C16]palmitate (plasma) tracer concentration in chylomicron-TG. The mechanisms of the reduction in meal-derived palmitate are unclear, but we did identify a ~17% increase in chylomicron-TG FCR after resistance exercise, as well as significant increases in whole-body and mitochondrial lipid oxidation, suggesting that improved clearance might be a contributing mechanism. The reduction in mean-derived palmitate might also be associated with a decline in splanchnic blood flow following exercise, which could reduce gastrointestinal motility and gastric emptying, thereby delaying the absorption of palmitate from the meal (27). Alternatively, increased intestinal fatty acid oxidation, which was shown to occur after aerobic exercise, could reduce the substrate available for triacylglycerol synthesis and chylomicron secretion (28–30).
The mechanism of the reduced incorporation of plasma fatty acids (endogenous) in chylomicrons is also unclear. We confirmed previous speculation that plasma NEFA can be taken up by the enterocyte from the portal system and packaged into chylomicrons, with plasma free palmitate making a ~40% contribution to chylomicron-TG (the 13C16 palmitate TTR plateau in chylomicrons is ~40% of the plasma free palmitate TTR) (31). However, the postprandial palmitate Ra was not affected by resistance exercise, nor was postprandial plasma NEFA concentration, so reduced systemic NEFA availability for enterocyte uptake and TG production was probably not a significant contributing factor to the reduced plasma palmitate in chylomicrons. These results suggest that changes in chylomicron formation or enterocyte lipid handling may contribute to reduced postprandial lipaemia following resistance exercise.
Further insight into intestinal lipid handling after resistance exercise is provided by the TTR of exogenous meal-derived (13C4) and plasma (13C16) palmitate in chylomicron-TG. The enrichments of the initial sources of these fatty acids were not different between rest and exercise (test meal [(13C4)3]tripalmitin preparation; plasma [U-13C16]palmitate infusion). Therefore, the lower TTR after resistance exercise compared with rest indicates increased isotopic dilution of both meal-derived and plasma palmitate by unlabelled palmitate in chylomicron-TG. The sources of unlabelled palmitate in chylomicrons are not clear, but there is mounting evidence that pre-existing lipids in the gastrointestinal tract appear in chylomicrons when new dietary fat and nutrients are absorbed (32,33). Thus, resistance exercise appears to alter the relative contributions of meal-derived and plasma fatty acids compared with pre-existing unlabelled fatty acids to chylomicron-TG production, resulting in greater isotopic dilution of the test meal 13C4- and plasma [U-13C16]palmitate tracers. This mechanism could also contribute to the reduced meal-derived and plasma palmitate tracer concentration in chylomicron-TG if a greater portion of NEFA entering the enterocytes was diverted to replenish the cytosolic lipid droplet pool (34–36).
With regard to VLDL-TG, previous evidence suggests that a single bout (1 h) of resistance exercise and aerobic exercise (performed 30 min before a mixed meal) reduces postprandial VLDL-TG in obese women (14). Similarly, in our study, we found a reduction in the incremental increase in plasma VLDL-TG after a meal (Table 2). Although it did not reach statistical significance, we identified a ~30% increase in TRL-TG FCR, suggesting that increased clearance could contribute to reduced VLDL-TG iAUC (since VLDL-TG accounted for ~80% of TRL-TG).
We also found that a single session of resistance exercise increased adipose tissue expression of FOXO1, HSL and LPL. In adipose tissue, FOXO1 activates a gene network involved in lipid mobilisation, glucose sparing, and inhibition of lipid storage/fat accumulation (37,38). Recent studies also showed that upregulation of forkhead box protein O1 (FOXO1) in adipocytes during nutrient restriction enhances lysosomal degradation of lipid droplets (lipophagy), which generates NEFA that are used for cellular beta oxidation (39). Increased lipophagy and fatty acid oxidation in adipose tissue could contribute to the finding that postprandial palmitate rates of appearance were not significantly higher than basal rates.
An additional mechanism for the blunted appearance of NEFA after resistance exercise would include increased NEFA recycling back into adipose TG (40). We found that a single session of resistance exercise also increased the expression of PDK4 in adipose tissue; here, the protein product, pyruvate dehydrogenase kinase 4, stimulates glyceroneogenesis to promote re-esterification of NEFA, limit excessive plasma NEFA release and divert glucose away from oxidation (41,42). The activation of genes involved in both adipose tissue lipolysis (FOXO1, HSL) and NEFA re-esterification (PDK4) could reflect increased activity of the adipose tissue TG–NEFA cycle, which involves immediate re-esterification of the majority of NEFA liberated through adipose tissue lipolysis (43,44).
We observed increases in whole-body lipid oxidation rate, skeletal muscle mitochondrial lipid respiration, as well as coupled and uncoupled respiration, complex I- and complex II-supported respiration, free oxidative phosphorylation capacity, and the respiratory acceptor control ratio (an index of sensitivity to ADP). We also found a significant increase in the ratio of lipid to carbohydrate oxidation in skeletal muscle mitochondria, suggesting a greater contribution from NEFA for oxidation after resistance exercise. Similarly, skeletal muscle LPL mRNA content increased following resistance exercise (though not significantly), suggesting increased TG hydrolysis and NEFA uptake may have accompanied higher lipid oxidation.
To our knowledge, this is the first study to demonstrate that acute resistance exercise increases the contribution of lipid oxidation to overall energy production. This increase occurs concurrently with enhanced skeletal muscle expression of PPARGC1A (the protein product of which, peroxisome proliferator-activated receptor-γ coactivator-1α, upregulates genes coding for many proteins of tricarboxylic acid cycle, β-oxidation and fatty acid transport) (45). In skeletal muscle, resistance exercise also upregulated the genes encoding PDK4 (which stimulates NEFA oxidation and shunts glucose toward glycogen storage) and FOXO1 (which stimulates PDK4 and PGC1α expression), while reducing expression of SREBP1 (encoding sterol regulatory element binding transcription factor 1, which inhibits fatty acid synthase) (46–48).
Finally, resistance exercise increased the expression of the adiponectin gene in adipose tissue (an adipokine associated with larger lipid droplet formation, increased insulin sensitivity and enhanced adipocyte NEA oxidation), which is typically downregulated in insulin-resistant conditions (49,50). Higher adiponectin expression might have contributed to the reduced insulin concentration as well as the increase in whole-body lipid oxidation following resistance exercise.
Limitations
There are limitations to this study. First, we limited our analysis to overweight/obese, sedentary, middle-aged men with prediabetes. Further research is needed to investigate changes in lipid metabolism in response to resistance exercise in women, or in populations of different ages and activity levels. Additionally, we did not isolate VLDL for tracer enrichment analysis. Therefore, we used differences between TRL-TG and chylomicron metabolism to draw conclusions about VLDL metabolism. Our study was a priori powered to detect treatment effects of ~1 SD for paired differences between exercise and rest on postprandial plasma TG, lipoprotein clearance, NEFA Ra and Rd, lipid oxidation and mitochondrial respiration, and gene expression. However our results indicated were underpowered to detect differences in TRL-TG and chylomicron FCR and basal NEFA Ra and Rd. (effect sizes ranging .32-.57 for these outcomes).
Conclusion
We found that a single bout of resistance exercise reduces the lipaemic response to a mixed meal, and this reduction was mediated by diminished meal-derived and plasma fatty acid contributions to circulating lipoproteins, increased whole-body lipid oxidation, increased skeletal muscle mitochondrial lipid oxidation, and increased adipose tissue and skeletal muscle lipid oxidation gene expression. These changes might reduce the risk of cardiovascular disease or progression to type 2 diabetes in men with prediabetes.
Supplementary Material
Research in context.
What is already known about this subject?
Prediabetes is associated with postprandial dyslipidaemia, which increases the risk for cardiovascular disease and type 2 diabetes
Endurance and resistance-type exercise acutely lower fasting and postprandial plasma triacylglycerol (TG) concentrations in men and women who are healthy, obese or have type 2 diabetes
Aerobic exercise reduces postprandial TG by changing lipoprotein composition, and increasing lipoprotein clearance secondary to changes in lipoprotein affinity for lipoprotein lipase
What is the key question?
What are the constitutive metabolic mechanisms underlying the changes in postprandial lipid metabolism after acute resistance exercise in overweight/obese men with prediabetes?
What are the new findings?
A prior bout of acute resistance exercise reduces postprandial TG concentrations in obese men with prediabetes
Acute resistance exercise reduces the incorporation of both meal-derived and endogenous plasma fatty acids into triacylglycerol-rich lipoprotein- and chylomicron TGs
Resistance exercise also increases postprandial whole-body lipid oxidation, skeletal muscle mitochondrial respiration, oxidative gene expression in skeletal muscle and the expression of key lipolysis genes in adipose tissue
How might this impact on clinical practice in the foreseeable future?
This research provides mechanistic evidence to support the role of resistance exercise in the management of prediabetes, as well as the prevention of cardiovascular disease and type 2 diabetes in men with prediabetes
Acknowledgements
The authors would like to thank M. Giuffrida and K. Gratza for collection of muscle and adipose tissue biopsies, L. Ruvinov for preparing all stable isotope tracers for infusion for this study, S. Waller for preparing the standardised meals and the liquid test meals, T. Pietka for assistance with mitochondrial respiration analysis and F. Custodio and J. Shew for help with sample preparation for GC-MS analysis (all from the Center for Human Nutrition, Washington University School of Medicine, St Louis, MO, USA). We also thank the nursing staff at the Washington University Institute for Clinical and Translational Sciences Clinical Research Unit (Washington University School of Medicine, St Louis, MO, USA) for their assistance with all study procedures.
Parts of this study were presented in poster form (poster 733-B) at the American Diabetes Association 78th Scientific Sessions in 2018
Funding
This work was supported by the National Institutes of Health T32 HD007434-25 (principal investigator Lang), UL1TR000448 (principal investigator Evanoff), and P30 DK056341 (Washington University Nutrition Obesity Research Center); the Foundation for Physical Therapy Promotion of Doctoral Studies II Scholarship (AJB); and the Cambridge Isotopes Laboratories Research Grant Program (WTC).
Abbreviations
- ATIRI
Adipose tissue insulin resistance index
- CTRU
Clinical and Translational Research Unit
- CVD
Cardiovascular disease
- FCR
Fractional clearance rate
- FOXO1
Forkhead box protein O1
- iAUC
Incremental AUC
- Ra
Rate of appearance
- TG
Triacylglycerol
- TRL
Triacylglycerol-rich lipoprotein
- TTR
Tracer to tracee ratio
- WUSTL
Washington University in St Louis
Footnotes
Publisher's Disclaimer: This Author Accepted Manuscript is a PDF file of a an unedited peer-reviewed manuscript that has been accepted for publication but has not been copyedited or corrected. The official version of record that is published in the journal is kept up to date and so may therefore differ from this version.
Data availability
The datasets generated/analysed during this study are available from the corresponding author on reasonable request.
Duality of interest
The authors declare that there is no duality of interest associated with this manuscript.
References
- 1.Hamilton MT, Hamilton DG, Zderic TW (2007) Role of low energy expenditure and sitting in obesity, metabolic syndrome, type 2 diabetes, and cardiovascular disease. Diabetes 56(11):2655–2667. [DOI] [PubMed] [Google Scholar]
- 2.Sardinha LB, Magalhães JP, Santos DA, Júdice PB (2017) Sedentary patterns, physical activity, and cardiorespiratory fitness in association to glycemic control in type 2 diabetes patients. Front Physiol 8:262. doi: 10.3389/fphys.2017.00262 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lahjibi E, Heude B, Dekker JM, et al. (2013) Impact of objectively measured sedentary behaviour on changes in insulin resistance and secretion over 3 years in the RISC study: interaction with weight gain. Diabetes Metab 39(3):217–225 [DOI] [PubMed] [Google Scholar]
- 4.Bansal S, Buring JE, Rifai N, Mora S, Sacks FM, Ridker PM (2007) Fasting compared with nonfasting triglycerides and risk of cardiovascular events in women. JAMA 298(3):309–316 [DOI] [PubMed] [Google Scholar]
- 5.Karpe F, Steiner G, Uffelman K, Olivecrona T, Hamsten A (1994) Postprandial lipoproteins and progression of coronary atherosclerosis. Atherosclerosis 106(1):83–97 [DOI] [PubMed] [Google Scholar]
- 6.Colberg SR, Sigal RJ, Yardley JE, et al. (2016) Physical activity/exercise and diabetes: a position statement of the american diabetes association. Diabetes Care 39(11): 2065–2079 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Petitt DS, Arngrímsson SA, Cureton KJ (2002) Effect of resistance exercise on postprandial lipemia. J Appl Physiol 94: 694–700. doi: 10.1152/japplphysiol.00377.2002 [DOI] [PubMed] [Google Scholar]
- 8.Heden TD, Winn NC, Mari A, et al. (2015) Postdinner resistance exercise improves postprandial risk factors more effectively than predinner resistance exercise in patients with type 2 diabetes. Journal of Applied Physiology 118(5): 624–634. doi: 10.1152/japplphysiol.00917.2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cohn JS, McNamara JR, Cohn SD, Ordovas JM, Schaefer EJ (1988) Postprandial plasma lipoprotein changes in human subjects of different ages. J Lipid Res 29:469–479 [PubMed] [Google Scholar]
- 10.Shannon KA, Shannon RM, Clore JN, Gennings C, Warren BJ, Potteiger JA (2005) Resistance exercise and postprandial lipemia: the dose effect of differing volumes of acute resistance exercise bouts. Metabolism 54:756–63. [DOI] [PubMed] [Google Scholar]
- 11.Henderson GC (2014) Sexual dimorphism in the effects of exercise on metabolism of lipids to support resting metabolism. Front Endocrinol (Lausanne) 5:162. doi: 10.3389/fendo.2014.00162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gill JM, Frayn KN, Wootton SA, Miller GJ, Hardman AE (2000) Prior exercise and postprandial substrate extraction across the human leg. Am J Physiol Endocrinol Metab 279(5):E1020–E1028. [DOI] [PubMed] [Google Scholar]
- 13.Gill JM, Frayn KN, Wootton SA, Miller GJ, Hardman AE (2001) Effects of prior moderate exercise on exogenous and endogenous lipid metabolism and plasma factor VII activity. Clin Sci (Lond) 100(5):517–27 [PubMed] [Google Scholar]
- 14.Davitt PM, Arent SM, Tuazon MA, Golem DL, Henderson GC (2013) Postprandial triglyceride and free fatty acid metabolism in obese women after either endurance or resistance exercise. J Appl Physiol 114: 1743–1754. [DOI] [PubMed] [Google Scholar]
- 15.Gill JM, Al-Mamari A, Ferrell WR, et al. (2006) Effects of a moderate exercise session on postprandial lipoproteins, apolipoproteins and lipoprotein remnants in middle-aged men. Atherosclerosis 185(1):87–96. [DOI] [PubMed] [Google Scholar]
- 16.Adiels M, Mardinoglu A, Taskinen MR, Borén J (2015) Kinetic studies to elucidate impaired metabolism of triglyceride-rich lipoproteins in humans. Front Physiol 6:342. doi: 10.3389/fphys.2015.00342 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Magkos F, Tsekouras YE, Prentzas KI, et al. (2008) Acute exercise-induced changes in VLDL-triglyceride kinetics leading to hypotriglyceridemia manifest more readily after resistance than endurance exercise. J Appl Physiol 105: 1228–1236 [DOI] [PubMed] [Google Scholar]
- 18.Dias CB, Moughan PJ, Wood LG, Singh H, Garg ML (2017) Postprandial lipemia: factoring in lipemic response for ranking foods for their healthiness. Lipids Health Dis 16(1):178. doi: 10.1186/s12944-017-0568-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Huang Y, Cai X, Mai W, Li M, Hu Y (2016) Association between prediabetes and risk of cardiovascular disease and all cause mortality: systematic review and metaanalysis. The BMJ 355:i5953. doi: 10.1136/bmj.i5953 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cade WT, Spencer CT, Reeds DN, et al. (2013) Substrate metabolism during basal and hyperinsulinemic conditions in adolescents and young-adults with Barth syndrome. J Inherit Metab Dis 36:91–101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Smith GI, Villareal DT, Lambert CP, Reeds DN, Mohammed BS, Mittendorfer B (2009) Timing of the initial muscle biopsy does not affect the measured muscle protein fractional synthesis rate during basal, postabsorptive conditions. J Appl Physiol (1985) 108(2):363–368. doi: 10.1152/japplphysiol.00957.2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fabbrini E, Yoshino J, Yoshino M, et al. (2015) Metabolically normal obese people are protected from adverse effects following weight gain. J Clin Invest 125(2):787–795. doi: 10.1172/JCI78425 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Fabbrini E, Magkos F, Conte C, et al. (2012) Validation of a novel index to assess insulin resistance of adipose tissue lipolytic activity in obese subjects. J Lipid Res 53(2):321–324. doi: 10.1194/jlr.D020321 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Carstensen M, Thomsen C, Hermansen K (2003) Incremental area under response curve more accurately describes the triglyceride response to an oral fat load in both healthy and type 2 diabetic subjects. Metabolism 52(8):1034–1037. [DOI] [PubMed] [Google Scholar]
- 25.Patterson BW, Mittendorfer B, Elias N, Satyanarayana R, Klein S (2002) Use of stable isotopically labeled tracers to measure very low density lipoprotein-triglyceride turnover. J Lipid Res 43: 223–233 [PubMed] [Google Scholar]
- 26.Zotou E, Magkos F, Koutsari C, et al. (2010) Acute resistance exercise attenuates fasting and postprandial triglyceridemia in women by reducing triglyceride concentrations in triglyceride-rich lipoproteins. Eur J Appl Physiol 2010; 110: 869–874 [DOI] [PubMed] [Google Scholar]
- 27.Rehrer NJ, Smets A, Reynaert H, Goes E, De Meirleir K (2001) Effect of exercise on portal vein blood flow in man. Med Sci Sports Exerc 33:1533–1537 [DOI] [PubMed] [Google Scholar]
- 28.Hung Y, Linden MA, Gordon A, Scott Rector R, Buhman KK (2015) Endurance exercise training programs intestinal lipid metabolism in a rat model of obesity and type 2 diabetes. Physiological Reports 3(1):e12232. doi: 10.14814/phy2.12232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kimura R, Takahashi N, Murota K (2011) Activation of peroxisome proliferator-activated receptor-α (PPARα) suppresses postprandial lipidemia through fatty acid oxidation in enterocytes. Biochem Biophys Res Commun 410(1):1–6 [DOI] [PubMed] [Google Scholar]
- 30.Pavlic M, Xiao C, Szeto L, Patterson BW, Lewis GF (2010) Insulin acutely inhibits intestinal lipoprotein secretion in humans in part by suppressing plasma free fatty acids. Diabetes 59:580–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chavez-Jauregui RN, Mattes RD, Parks EJ (2010) Dynamics of fat absorption and effect of sham feeding on postprandial lipema. Gastroenterology 139(5):1538–1548 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Robertson MD, Henderson RA, Vist GE, Rumsey RD (2002) Extended effects of evening meal carbohydrate-to-fat ratio on fasting and postprandial substrate metabolism. Am. J. Clin. Nutr 75:505–510. [DOI] [PubMed] [Google Scholar]
- 33.Xiao C, Stahel P, Carreiro AL, Buhman KK, Lewis GF (2018) Recent advances in triacylglycerol mobilization by the gut. Trends Endocrinol Metab 29: 151–163 [DOI] [PubMed] [Google Scholar]
- 34.Zhu B Lee KK Buhman JX Cheng (2009) A dynamic, cytoplasmic triacylglycerol pool in enterocytes revealed by ex vivo and in vivo coherent anti-Stokes Raman scattering imaging. J Lipid Res 50 pp. 1080–1089 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Vaugelade P, Posho L, Darcy-Vrillon B, Bernard F, Morel MT, Duée PH (1994) Intestinal oxygen uptake and glucose metabolism during nutrient absorption in the pig. Proc Soc Exp Biol Med 207(3):309–16. [DOI] [PubMed] [Google Scholar]
- 36.Soayfane Z, Tercé F, Cantiello M, et al. (2016) Exposure to dietary lipid leads to rapid production of cytosolic lipid droplets near the brush border membrane. Nutr Metab (Lond) 13: 48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Nakae J, Cao Y, Oki M, Orba Y, Sawa H (2008) Forkhead transcription factor FoxO1 in adipose tissue regulates energy storage and expenditure. Diabetes 57(3):563–576 [DOI] [PubMed] [Google Scholar]
- 38.Chakrabarti P, Kandror KV (2009) FoxO1 controls insulin-dependent adipose triglyceride lipase (ATGL) expression and lipolysis in adipocytes. JBC 284(20):13296–13300. doi: 10.1074/jbc.C800241200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lettieri Barbato D, Tatulli G, Aquilano K, Ciriolo MR (2013) FoxO1 controls lysosomal acid lipase in adipocytes: implication of lipophagy during nutrient restriction and metformin treatment. Cell Death Dis 4: e861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Granneman JG, Burnazi M, Zhu Z, Schwamb LA (2003) White adipose tissue contributes to UCP1-independent thermogenesis. Am J Physiol Endocrinol Metab 285:E1230–E1236 [DOI] [PubMed] [Google Scholar]
- 41.Wan Z, Frier BC, Williams DB, Wright DC (2012) Epinephrine induces PDK4 mRNA expression in adipose tissue from obese, insulin resistant rats. Obesity (Silver Spring) 20(2):453–456. [DOI] [PubMed] [Google Scholar]
- 42.Cadoudal T, Distel E, Durant S, et al. (2008) Pyruvate dehydrogenase kinase 4: regulation by thiazolidinediones and implication in glyceroneogenesis in adipose tissue. Diabetes 57(9):2272–2279. doi: 10.2337/db08-0477 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Reshef L, Olswang Y, Cassuto H, et al. (2003) Glyceroneogenesis and the triglyceride/fatty acid cycle. J Biol Chem 278(33):30413–30416. [DOI] [PubMed] [Google Scholar]
- 44.Vallerand AL, Zamecnik J, Jones PJ, Jacobs I (1999) Cold stress increases lipolysis, FFA Ra and TG/FFA cycling in humans. Aviat Space Environ Med 1999 70(1):42–50. [PubMed] [Google Scholar]
- 45.Wu Z, Puigserver P, Andersson U (1999) Mechanisms controlling mitochondrial biogenesis and respiration through the thermogenic coactivator PGC-1. Cell 9;98(1):115–124 [DOI] [PubMed] [Google Scholar]
- 46.Constantin-Teodosiu D (2013) Regulation of muscle pyruvate dehydrogenase complex in insulin resistance: effects of exercise and dichloroacetate. Diabetes Metab J 37(5):301–314. doi: 10.4093/dmj.2013.37.5.301 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Furuyama T, Kitayama K, Yamashita H, Mori N (2003) Forkhead transcription factor FOXO1 (FKHR)-dependent induction of PDK4 gene expression in skeletal muscle during energy deprivation. Biochem J 375(Pt 2):365–371. doi: 10.1042/BJ20030022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Nadeau KJ, Ehlers LB, Aguirre LE (2006) Exercise training and calorie restriction increase SREBP-1 expression and intramuscular triglyceride in skeletal muscle. Am J Physiol Endocrinol Metab 291(1):E90–E98. Epub 2006 Jan 31 [DOI] [PubMed] [Google Scholar]
- 49.Kim JY, van de Wall E, Laplante M, et al. (2007) Obesity-associated improvements in metabolic profile through expansion of adipose tissue. J Clin Invest 117(9):2621–2637. doi: 10.1172/JCI31021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Slutsky N, Vatarescu M, Haim Y, et al. (2016) Decreased adiponectin links elevated adipose tissue autophagy with adipocyte endocrine dysfunction in obesity. Int J Obes (Lond) 40(6):912–20. doi: 10.1038/ijo.2016.5 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.