Abstract
Objectives
Prenatal cadmium (Cd) exposure may be associated with attention-deficit/hyperactivity disorder (ADHD) in children. Therefore, the objective of this study was to examine the relationship between Cd exposure during gestation and ADHD at 6 years of age.
Methods
As part of an ongoing cohort study (the Environment and Development of Children study), 479 mother-child pairs from Seoul, Korea were included for analysis between 2008 and 2011. The whole blood concentration of Cd was analyzed using atomic absorption spectrophotometry. The parents were surveyed about ADHD behaviors in their children at age 6. Multivariable linear regression models were used to investigate the relationship between prenatal exposure to Cd and ADHD at 6 years of age.
Results
Increased prenatal Cd concentrations were associated with increased scores for ADHD for girls, but not for boys, at age 6. A 2-fold increase in the prenatal Cd level was significantly associated with a 22.3% (95% confidence interval, 11.6 to 34.1) increase in ADHD in girls at 6 years of age, as indicated by the linear regression model.
Conclusions
Our results identified significant associations between prenatal Cd exposure and ADHD scores in 6-year-old girls.
Keywords: Cadmium, Preschool child, Attention-deficit/hyperactivity disorder, Republic of Korea
INTRODUCTION
Cadmium (Cd), a heavy metal, is dispersed throughout the earth’s soil both naturally and from human activities such as the burning of fossil fuels, the incineration of waste, industrial smelting procedures, and the use of phosphate-containing fertilizers [1]. Among smokers, tobacco use is the dominant source of Cd exposure; however, for the general non-smoking population, diet (specifically, the consumption of leafy vegetables, root crops, grains, organ meats, and seafood) is the primary source of exposure [1,2]. Cd is known to cause nephrotoxicity [3], and growing evidence also suggests that exposure to Cd may have adverse neurodevelopmental consequences. Animal studies have described the effects of Cd on electrophysiological parameters of the brain, changes in neurotransmitter function, and adverse neurobehavioral outcomes [4-8]. In children, studies have reported associations between increased Cd levels and outcomes such as mental retardation [9,10], lower verbal intelligence quotient [11], learning disabilities [12-14], poor reading performance [15], and behavioral problems [16]. However, other human studies have shown contradictory results, failing to detect significant associations between exposure to Cd and neurodevelopmental outcomes [17-19].
Evidence suggests that the prevalence of attention-deficit/hyperactivity disorder (ADHD) is greater in males than in females [20]. In the Attention-Deficit Hyperactivity Disorder Observational Research in Europe study, which was a large 24-month longitudinal observational study of children aged 6-18 years that was conducted in 10 European countries, the sex distribution of the study subjects varied by country, ranging from a 1:3 to a 1:16 ratio of girls to boys [21]. With regard to individuals with ADHD, such a disparity not only exists in childhood, but also persists into adulthood, as ADHD is more commonly diagnosed in adult males than adult females. A worldwide meta-regression analysis of 11 studies of adults with ADHD found that although the ratio of males to females with ADHD decreased with increasing age, a sex-based difference was still present in adults aged 19 years and over [22]. A pooled analysis of studies of ADHD outcomes estimated the rate of persistence of ADHD into adulthood as approximately 15% when limited to those who meet the full criteria for ADHD. However, when cases consistent with the Diagnostic and Statistical Manual of Mental Disorders, 4th edition (DSM-IV) definition of ADHD in partial remission were included, the persistence into adulthood was estimated to be as high as about 60% [23]. Two studies (1 in males and 1 in females) on the persistence of ADHD reported that 11 years after the initial diagnosis, 35.0% of males and 33.3% of females continued to meet the DSM-IV criteria for ADHD, indicating that the persistence of ADHD is similar between the sexes [24]. Despite the apparent sex-based differences, research has mainly focused on ADHD in boys, and studies of ADHD in girls are still limited [25].
In the present study, we used an ongoing cohort of 6-year-old children to examine ADHD symptoms associated with prenatal exposure to Cd, using a linear regression model. In addition, we examined sex-specific effects on ADHD.
METHODS
Population
The present study was based on the Environment and Development of Children (EDC) study, a prospective cohort study. The participants chosen for the EDC study were mothers and children who previously participated in the Congenital Anomaly Study (CAS), another cohort study of birth outcomes. In the CAS cohort study, pregnant women from the metropolitan areas of Seoul and Incheon, Korea, who received prenatal care at local hospitals were recruited. The total number of enrolled pregnant females was 13 484, and subjects were recruited during the second trimester of pregnancy. Of these females, 11 085 remained in the study and were followed up until childbirth, between August 2008 and July 2011. Blood and urine samples were collected at the time of enrollment, after subjects had fasted for more than 8 hours. A questionnaire about participants’ demographics and lifestyles was administered by trained nurses. In the CAS cohort, 115 children were found to have congenital anomalies. These children, as well as those with invalid addresses (n=218), were excluded from recruitment, and a total of 10 752 mothers were eligible for the EDC birth cohort.
A sample size of 610 (effect size, 0.017; α, 0.05; power, 0.90) was calculated to be sufficient to examine the association between Cd exposure and other characteristics of the children. The sample size was then inflated by 5%, to 645, to allow for dropouts. Between 2012 and 2015, we contacted 2085 mothers chosen at random from the 10 752 EDC participants, until 726 mother-child pairs were enrolled in the present study (response rate, 31%). We conducted follow-up assessments when the children were approximately 6 years of age, between March 2015 and December 2017. The children underwent health examinations at the Seoul National University Hospital in Jongno-gu, Seoul, Korea. The mothers’ depressive symptoms and the children’s dietary habits were assessed using the Center for Epidemiologic Studies Depression Scale and food frequency questionnaires, respectively.
Neurobehavioral tests for ADHD were administered to the 6-year-old children between 2015 and 2017. The Korean version of the ADHD rating scale (K-ARS) was used to measure ADHD symptoms. At the follow-up examination, urine and blood samples were collected, and physical examinations were conducted, after the children had fasted for more than 8 hours. For prenatal measurements, we assessed blood lead (Pb) and Cd concentrations in whole blood samples during the second trimester of pregnancy, between 14 weeks and 27 weeks (mean, 20 weeks) of gestation. Whole blood heavy metal levels were quantified using atomic absorption spectrophotometry.
After excluding those who did not complete the K-ARS assessment (n=77), those without maternal (n=63) or child (n=22) blood Cd measurements, and those with missing covariate values (n=2), 479 mother-child pairs were included in the present analyses.
Assessments of Attention-deficit/Hyperactivity Symptoms
The parents completed a questionnaire about demographic data and related factors, including socioeconomic status, secondhand smoking exposure, parental age, parental education level, and medical and obstetric histories of themselves and their children. Parental education level was classified as high school graduate or less (≤12 years of education) or university or above (more education than a high school graduate). We used a previously validated translated version of the ADHD rating scale (ARS) called the K-ARS to assess ADHD symptoms. The ARS, which was developed to assess the severity of ADHD symptoms [26], consists of 18 items that reflect the DSM-IV diagnostic criteria for ADHD. Since the K-ARS is a translated version of the ARS and therefore contains identical content to the ARS, it was selected as an adequate method for use on Korean subjects. Each item of the K-ARS is rated on a 4-point Likert scale (0-3 points, total score 0-54), with potential responses of “never or rarely,” “sometimes,” “often,” or “very often.”
Measurements of Blood Lead and Cadmium
Whole blood was collected from the subjects in trace element ethylenediaminetetraacetic acid tubes, and the samples were analyzed using atomic absorption spectrophotometry (240Z AA Zeeman spectrometer, Agilent, Santa Clara, CA, USA), using the Environmental Calibration Standard (Part #5183-4688, Agilent), with ammonium phosphate monobasic (20% NH4H2PO4, Sigma-Aldrich, St. Louis, MO, USA) and Triton X-100 (Sigma-Aldrich) as reagents. Once collected, the samples were frozen and stored at -20°C. Before analysis, the blood samples were brought to room temperature and pretreated using a Vortex Genie 2 mixer (Scientific Industries, Bohemia, NY, USA) and an ES 220A balance (Precisa Gravimetrics AG, Dietikon, Switzerland). The samples were diluted with a matrix modifier reagent (Triton X-100 and ammonium hydrogen phosphate monobasic), then assayed using the matrix-matched calibration curve method (the standard addition method). The method detection limit (MDL) was calculated according to the following formula:
MDL = t(n-1, 1-α=0.99) × S
where:
MDL=method detection limit
t(n-1, 1-α=0.99) =Student’s t-value appropriate for a single-tailed 99th-percentile t statistic and a standard deviation estimate with n−1 degrees of freedom
S = sample standard deviation of the replicate spiked-sample analyses
Seven samples were used to calculate the MDL, so 3.14 was multiplied by the sample standard deviation (as found in Addendum Table 1 of the Definition and Procedure for the Determination of the Method Detection Limit, Revision 2, United States Environmental Protection Agency). The MDL values for lead and cadmium were found to be 0.091 μg/dL and 0.031 μg/L, respectively.
Statistical Analysis
Blood Cd concentrations were transformed by log2 (base 2) to obtain a normal distribution. In order to investigate the relationship between prenatal Cd concentrations and ADHD scores at age 6, we used generalized additive models. In the examination of the regression curve for the generalized additive model, we assumed a linear relationship. We constructed a regression model for Cd exposure and ADHD scores to estimate the overall linear effects of Cd exposure on ADHD scores.
We modeled scores on the K-ARS as a Poisson distribution and estimated the effects of Cd exposure on the total and subcategory scores for ADHD. All models controlled for potential covariates including sex (male or female), parity, birth weight (kg), mother’s body mass index (BMI, kg/m2), mother’s education level, mother’s age, and mother’s smoking status. To examine potential sex-based differences, we stratified our samples by sex. In our sensitivity analysis, we included concurrent child blood Cd concentration measured at the age of 6 and maternal blood Pb concentration in our main model.
The statistical analyses were conducted using SAS version 9.4 (SAS Institute, Inc., Cary, NC, USA) and R version 3.2.1 (https://cran.r-project.org/bin/windows/base/old/3.2.1/). Two-tailed p-values of less than 0.05 were considered to indicate statistical significance.
Ethics Statement
We obtained informed consent from all participants, and the study protocol was approved by the Institutional Review Board of the Seoul National University College of Medicine (IRB No. 1201-010-392).
RESULTS
The characteristics of the study population regarding maternal and child age, sex, BMI, maternal education level, parity, secondhand tobacco smoke exposure, K-ARS scores, and maternal blood heavy metal (Pb and Cd) levels are presented in Table 1. The mean maternal age of the 479 mothers was 31.4 years, and 84.3% of the mothers had an education level of university or above. Only 4.4% of the mothers were current smokers. The mean age of the children was 71.1 months, and 53.4% were male. Furthermore, 60.9% of the boys and 61.0% of the girls were the first-born child in the family. Regarding ADHD symptoms, the mean total K-ARS score was 6.9 for boys, which was higher than the mean total K-ARS score for girls (5.4). This relationship was also observed when symptoms were classified by type (attention-deficit or hyperactivity); the K-ARS scores in boys and girls for the attention-deficit type were 3.9 and 3.0, respectively, and those for the hyperactivity type were 3.0 and 2.3, respectively. The mean maternal blood levels of Pb and Cd were 1.4 μg/dL and 0.7 μg/L, respectively. No statistically significant differences in any demographic or clinical factors were observed according to sex.
Table 1.
Characteristics of participants (n=479)
| Characteristics | Overall | Boys | Girls |
|---|---|---|---|
| Total | 479 (100) | 256 (53.4) | 223 (46.6) |
| Age (mo) | 71.1±1.6 | 71.1±1.6 | 71.1±1.6 |
| Body mass index (kg/m2) | 15.8±1.7 | 15.8±1.7 | 15.8±1.7 |
| Maternal age (y) | 31.4±3.6 | 31.5±3.6 | 31.2±3.5 |
| Maternal blood cadmium level (μg/L) | 0.7±0.3 | 0.7±0.3 | 0.7±0.3 |
| Maternal blood lead level (μg/dL) | 1.4±0.4 | 1.4±0.5 | 1.3±0.3 |
| Maternal education level | |||
| High school or less | 75 (15.6) | 40 (15.6) | 35 (15.7) |
| University or above | 404 (84.3) | 216 (84.4) | 188 (84.3) |
| Current smoker | 31 (6.4) | 19 (7.4) | 12 (5.4) |
| Secondhand smoke exposure | |||
| No | 87 (18.2) | 44 (17.2) | 43 (19.3) |
| Yes | 392 (81.8) | 212 (82.8) | 180 (80.7) |
| Parity | |||
| First child | 292 (61.0) | 156 (60.9) | 136 (61.0) |
| Second child or above | 187 (39.0) | 100 (39.0) | 87 (39.0) |
| K-ARS scores | |||
| Total | 6.2±5.8 | 6.9±5.7 | 5.4±5.9 |
| Attention deficit | 3.4±3.2 | 3.9±3.1 | 3.0±3.3 |
| Hyperactivity | 2.7±3.1 | 3.0±3.0 | 2.3±3.0 |
Values are presented as mean±standard deviation or number (%).
SD, standard deviation; K-ARS, Korean attention-deficit/hyperactivity disorder rating scale.
Figure 1 shows the association between prenatal blood Cd concentrations and the K-ARS total score. A positive, nearly linear association was observed between prenatal Cd levels and the K-ARS total score at 6 years of age. Therefore, assuming linearity, we used a generalized estimating equation model in our analysis. When we performed an additional analysis assuming a non-linear association, similar results to our main analysis were observed (Supplemental Material 1). Table 2 shows estimations of the relationships between prenatal blood Cd levels and the total and subcategory scores for ADHD as shown by percentage increase in the K-ARS score per 2-fold increase in the prenatal blood Cd level. No significant associations were observed between postnatal (concurrent) Cd exposure at 6 years and K-ARS scores. However, each doubling of the maternal prenatal Cd level was significantly associated with an increase in the K-ARS total score of 22.3% (95% confidence interval [CI], 11.6 to 34.1) in girls only. When investigated separately by symptom type, again in girls only, each doubling of prenatal Cd exposure showed a 21.2% (95% CI, 7.4 to 36.8) increase in the score related to attention-deficit symptoms and a 23.5% (95% CI, 7.1 to 42.3) increase in the score for the hyperactivity symptoms. These associations were not observed for boys. The effect size was significantly larger in girls than in boys (p=0.002). A sensitivity analysis with adjustment for maternal blood Pb levels and concurrent blood Cd levels at age 6 did not change the main findings. In our analysis, maternal Pb levels were not associated with K-ARS scores (Supplemental Material 2). When we included concurrent (6-year-old) blood Cd levels and maternal Pb levels in our model, the main results remained unchanged (Supplemental Material 2). The proportion of current smokers in our population was very low (4.4%), and similar results were seen after we excluded current smokers (Supplemental Material 3).
Figure. 1.
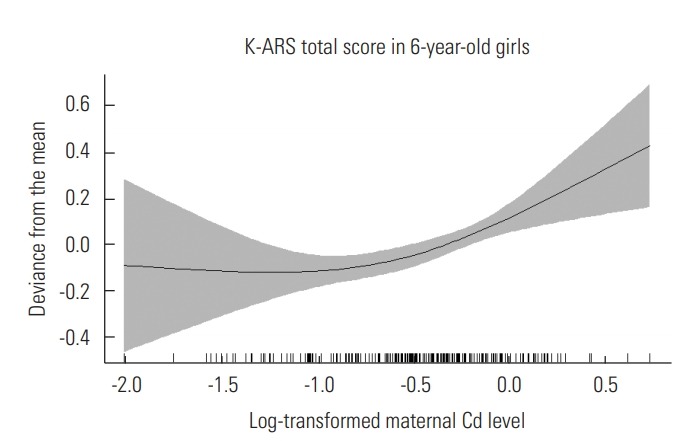
Relationship between log-transformed maternal blood cadmium (Cd) levels and Korean attention-deficit/hyperactivity disorder rating scale (K-ARS) total score in 6-yearold girls (p<0.001).
Table 2.
Percent change in K-ARS scores associated with 2-fold increases in the maternal blood cadmium level in 6-year old children
| Subtype | Sex | % change (95% CI) | p-value |
|---|---|---|---|
| Total | Overall | 6.4 (0.1, 13.2) | 0.045 |
| Male | -0.1 (-8.3, 8.6) | 0.964 | |
| Female | 22.3 (11.6, 34.1) | <0.001 | |
| Attention deficit | Overall | 7.8 (-0.6, 17.0) | 0.070 |
| Male | 1.8 (-9.0, 14.1) | 0.747 | |
| Female | 21.2 (7.4, 36.8) | 0.002 | |
| Hyperactivity | Overall | 4.5 (-4.7, 14.8) | 0.347 |
| Male | -2.9 (-14.6, 10.4) | 0.653 | |
| Female | 23.5 (7.1, 42.3) | 0.004 |
The model was adjusted for sex (male or female), parity, birth weight (kg), mother’s body mass index, mother's education level, mother's age, and mother's passive smoking status.
K-ARS, Korean attention-deficit/hyperactivity disorder rating scale; CI, confidence interval.
DISCUSSION
In this study, we investigated the relationship between prenatal blood Cd exposure and ADHD symptoms in 6-year-old children. We observed that higher maternal blood Cd concentrations during pregnancy were significantly associated with higher ADHD scores, as measured by the K-ARS questionnaire, in girls.
To date, there is only limited information on the relationship between Cd exposure and ADHD. The published literature lacks significant findings and contains mixed results. A study conducted using National Health and Nutrition Examination Survey data from 1999-2000 reported non-significantly higher odds of ADHD diagnosis in children with higher blood Cd concentrations, but this trend disappeared when the analysis was adjusted for the effects of persistent organic pollutants [19]. Another study reported that a higher frequency of social problems, withdrawal, and attention problems was linked to higher concentrations of Cd in children’s hair at ages 7 years to 16 years [27]. In contrast, another recent analysis of National Health and Nutrition Examination Survey data from 1997-2004 found a non-significant decrease in ADHD risk with increasing levels of urinary Cd in children [28].
Although an association between Cd and ADHD was not consistently found in previous studies, numerous studies have investigated the behavioral effects of early-life Cd exposure and have reported significant findings. In Belgium, a prospective study of children reported a significant association between prenatal exposure to Cd and an increased risk of emotional problems among 7- to 8-year-old boys [29]. Another study in China reported an association between attention and social problems in 7- to 16-year-old children and increased hair Cd levels [27].
Previous animal studies have shown that Cd affects central nervous system metabolism via inhibition of sulfhydryl-containing enzymes. As a result, in chronically exposed individuals, Cd can act as a depressant of various neurotransmitters, including acetylcholine, norepinephrine, and serotonin [30,31]. Changes in the metabolism of the dopamine and serotonin systems were also documented, and these neurotransmitter systems are known to be linked with the etiology of ADHD [32]. Animal studies have also shown that Cd can readily cross the blood-brain barrier [33], from which we can infer that Cd may move directly to the central nervous system and impact the neurodevelopment of young children [34]. Other studies have found evidence that Cd exposure can induce changes in activity levels in animals, although these studies showed inconsistent results, as some reported increased hyperactivity, while others found a decrease in exploratory activity and in time spent in motion [4].
We tested maternal blood Pb levels as a potential covariate in the relationship between blood Cd levels and K-ARS scores, and our analysis found that maternal blood Pb concentrations were not associated with K-ARS scores. Specific domains of neurobehavioral impairment that may be associated with Pb exposure remain an issue of uncertainty in the literature. Previous studies have displayed inconsistent results regarding the contribution of Pb exposure to inattention and impulsivity [35]. Maternal Cd levels, on the other hand, displayed a positive relationship with ADHD in girls when included in the model along with maternal Pb levels and concurrent blood Cd levels in the children.
The main strength of this study is its prospective cohort design. We also controlled for potential confounders, such as blood Pb levels and secondhand smoking status. However, this study also has some limitations. The current study investigated the associations between toxic heavy metal exposure and K-ARS scores, and although we adjusted for maternal variables such as maternal age and education, we were not able to account for the diverse social, cultural, and socioeconomic effects that are known to play a role in the development of ADHD. The K-ARS, which was the screening tool used for the study, is fundamentally a parent-administered tool used to detect symptoms of ADHD in children. As this test was completed by parents of the children in the study, observer bias may have influenced the results. Consequently, there is potential for misclassification resulting from parents over-reporting or under-reporting their children’s symptoms. Given this limitation, we attempted to show the associations between increased maternal levels of Cd and symptom-related scores of ADHD at 6 years of age, although the clinical significance of our findings needs further investigation. Additionally, no children in this cohort were clinically diagnosed with ADHD. The K-ARS scale is known to have high reliability and validity for the screening and diagnosis of ADHD, but it is not a clinical diagnostic tool [36]. Since the diagnosis of ADHD requires a thorough evaluation of consistent symptoms for more than 6 months by a trained physician, the results of this study should be interpreted with caution.
Our study is the first to suggest a significant dose-dependent relationship between prenatal Cd concentrations and ADHD scores at the age of 6 years. The strengths of our study are its prospective cohort design, as well as its consideration of multiple covariates, including prenatal Pb concentrations and concurrent blood Cd concentrations in the children at age 6. However, given the nature of parent-reported K-ARS scores, the results of this study should be interpreted cautiously. Additional prospective epidemiological studies should be performed to further replicate and clarify the possible association observed in this study.
Acknowledgments
None.
CONFLICT OF INTEREST
The authors have no conflicts of interest associated with the material presented in this paper.
FUNDING
This research was supported by a grant from the Ministry of Food and Drug Safety in 2018 (18162MFDS121) and from the Center for Environmental Health through the Ministry of Environment.
AUTHOR CONTRIBUTIONS
Conceptualization: WK, YCH. Data curation: YHL, BNK, CHS, YAL, JIK, YCH. Formal analysis: WK, YJ, YHL, YCH. Funding acquisition: YCH. Methodology: WK, YHL, YCH. Project administration: YCH. Visualization: YCH. Writing - original draft: WK. Writing - review & editing: WK, YJ, YCH, YHL, BNK, CHS, YAL, JIK.
SUPPLEMENTAL MATERIALS
Supplemental materials are available at https://doi.org/10.3961/jpmph.19.175.
Percentage change in K-ARS scores associated with quartile increase of maternal blood cadmium.
Percentage change in K-ARS scores associated with 2-fold increases in maternal blood lead and cadmium levels in 6-year old children.
Percentage change in K-ARS scores associated with 2-fold increases in maternal blood cadmium levels in 6-year old children. (excluded current smokers)
REFERENCES
- 1.Agency for Toxic Substances and Disease Registry Toxicological profile for cadmium. 2012 [cited 2019 Jun 1]. Available from: https://www.atsdr.cdc.gov/ToxProfiles/tp5.pdf. [PubMed]
- 2.Järup L, Akesson A. Current status of cadmium as an environmental health problem. Toxicol Appl Pharmacol. 2009;238(3):201–208. doi: 10.1016/j.taap.2009.04.020. [DOI] [PubMed] [Google Scholar]
- 3.European Food Safety Authority (EFSA) Cadmium in food‐scientific opinion of the Panel on Contaminants in the Food Chain. EFSA J. 2009;7(3):980. doi: 10.2903/j.efsa.2008.653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ali MM, Murthy RC, Chandra SV. Developmental and longterm neurobehavioral toxicity of low level in-utero cadmium exposure in rats. Neurobehav Toxicol Teratol. 1986;8(5):463–468. [PubMed] [Google Scholar]
- 5.Dési I, Nagymajtényi L, Schulz H. Behavioural and neurotoxicological changes caused by cadmium treatment of rats during development. J Appl Toxicol. 1998;18(1):63–70. doi: 10.1002/(sici)1099-1263(199801/02)18:1<63::aid-jat475>3.0.co;2-z. [DOI] [PubMed] [Google Scholar]
- 6.Lehotzky K, Ungváry G, Polinák D, Kiss A. Behavioral deficits due to prenatal exposure to cadmium chloride in CFY rat pups. Neurotoxicol Teratol. 1990;12(2):169–172. doi: 10.1016/0892-0362(90)90130-5. [DOI] [PubMed] [Google Scholar]
- 7.Nagymajtényi L, Schulz H, Dési I. Behavioural and functional neurotoxicological changes caused by cadmium in a three-generational study in rats. Hum Exp Toxicol. 1997;16(12):691–699. doi: 10.1177/096032719701601201. [DOI] [PubMed] [Google Scholar]
- 8.Nation JR, Bourgeois AE, Clark DE, Baker DM, Hare MF. The effects of oral cadmium exposure on passive avoidance performance in the adult rat. Toxicol Lett. 1984;20(1):41–47. doi: 10.1016/0378-4274(84)90180-2. [DOI] [PubMed] [Google Scholar]
- 9.Jiang HM, Han GA, He ZL. Clinical significance of hair cadmium content in the diagnosis of mental retardation of children. Chin Med J (Engl) 1990;103(4):331–334. [PubMed] [Google Scholar]
- 10.Marlowe M, Errera J, Jacobs J. Increased lead and cadmium burdens among mentally retarded children and children with borderline intelligence. Am J Ment Defic. 1983;87(5):477–483. [PubMed] [Google Scholar]
- 11.Thatcher RW, Lester ML, McAlaster R, Horst R. Effects of low levels of cadmium and lead on cognitive functioning in children. Arch Environ Health. 1982;37(3):159–166. doi: 10.1080/00039896.1982.10667557. [DOI] [PubMed] [Google Scholar]
- 12.Capel ID, Pinnock MH, Dorrell HM, Williams DC, Grant EC. Comparison of concentrations of some trace, bulk, and toxic metals in the hair of normal and dyslexic children. Clin Chem. 1981;27(6):879–881. [PubMed] [Google Scholar]
- 13.Ely DL, Mostardi RA, Woebkenberg N, Worstell D. Aerometric and hair trace metal content in learning-disabled children. Environ Res. 1981;25(2):325–339. doi: 10.1016/0013-9351(81)90035-9. [DOI] [PubMed] [Google Scholar]
- 14.Pihl RO, Parkes M. Hair element content in learning disabled children. Science. 1977;198(4313):204–206. doi: 10.1126/science.905825. [DOI] [PubMed] [Google Scholar]
- 15.Thatcher RW, McAlaster R, Lester ML, Cantor DS. Comparisons among EEG, hair minerals and diet predictions of reading performance in children. Ann N Y Acad Sci. 1984;433:87–96. doi: 10.1111/j.1749-6632.1984.tb14761.x. [DOI] [PubMed] [Google Scholar]
- 16.Marlowe M, Cossairt A, Moon C, Errera J, MacNeel A, Peak R, et al. Main and interaction effects of metallic toxins on classroom behavior. J Abnorm Child Psychol. 1985;13(2):185–198. doi: 10.1007/BF00910641. [DOI] [PubMed] [Google Scholar]
- 17.Cao Y, Chen A, Radcliffe J, Dietrich KN, Jones RL, Caldwell K, et al. Postnatal cadmium exposure, neurodevelopment, and blood pressure in children at 2, 5, and 7 years of age. Environ Health Perspect. 2009;117(10):1580–1586. doi: 10.1289/ehp.0900765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gillberg C, Norén JG, Wahlström J, Rasmussen P. Heavy metals and neuropsychiatric disorders in six-year-old children. Aspects of dental lead and cadmium. Acta Paedopsychiatr. 1982;48(5):253–263. [PubMed] [Google Scholar]
- 19.Lee DH, Jacobs DR, Porta M. Association of serum concentrations of persistent organic pollutants with the prevalence of learning disability and attention deficit disorder. J Epidemiol Community Health. 2007;61(7):591–596. doi: 10.1136/jech.2006.054700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Willcutt EG. The prevalence of DSM-IV attention-deficit/hyperactivity disorder: a meta-analytic review. Neurotherapeutics. 2012;9(3):490–499. doi: 10.1007/s13311-012-0135-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Faraone SV, Biederman J, Mick E. The age-dependent decline of attention deficit hyperactivity disorder: a meta-analysis of follow-up studies. Psychol Med. 2006;36(2):159–165. doi: 10.1017/S003329170500471X. [DOI] [PubMed] [Google Scholar]
- 22.Fayyad J, Sampson NA, Hwang I, Adamowski T, Aguilar-Gaxiola S, Al-Hamzawi A, et al. The descriptive epidemiology of DSM-IV adult ADHD in the World Health Organization World Mental Health Surveys. Atten Defic Hyperact Disord. 2017;9(1):47–65. doi: 10.1007/s12402-016-0208-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Faraone SV, Biederman J, Mick E. The age-dependent decline of attention deficit hyperactivity disorder: a meta-analysis of follow-up studies. Psychol Med. 2006;36(2):159–165. doi: 10.1017/S003329170500471X. [DOI] [PubMed] [Google Scholar]
- 24.Biederman J, Petty CR, Clarke A, Lomedico A, Faraone SV. Predictors of persistent ADHD: an 11-year follow-up study. J Psychiatr Res. 2011;45(2):150–155. doi: 10.1016/j.jpsychires.2010.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Biederman J, Petty CR, O’Connor KB, Hyder LL, Faraone SV. Predictors of persistence in girls with attention deficit hyperactivity disorder: results from an 11-year controlled follow-up study. Acta Psychiatr Scand. 2012;125(2):147–156. doi: 10.1111/j.1600-0447.2011.01797.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.DuPaul GJ, Reid R, Anastopoulos AD, Lambert MC, Watkins MW, Power TJ. Parent and teacher ratings of attention-deficit/hyperactivity disorder symptoms: factor structure and normative data. Psychol Assess. 2016;28(2):214–225. doi: 10.1037/pas0000166. [DOI] [PubMed] [Google Scholar]
- 27.Bao QS, Lu CY, Song H, Wang M, Ling W, Chen WQ, et al. Behavioural development of school-aged children who live around a multi-metal sulphide mine in Guangdong province, China: a cross-sectional study. BMC Public Health. 2009;9:217. doi: 10.1186/1471-2458-9-217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ciesielski T, Weuve J, Bellinger DC, Schwartz J, Lanphear B, Wright RO. Cadmium exposure and neurodevelopmental outcomes in U.S. children. Environ Health Perspect. 2012;120(5):758–763. doi: 10.1289/ehp.1104152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sioen I, Den Hond E, Nelen V, Van de Mieroop E, Croes K, Van Larebeke N, et al. Prenatal exposure to environmental contaminants and behavioural problems at age 7-8 years. Environ Int. 2013;59:225–231. doi: 10.1016/j.envint.2013.06.014. [DOI] [PubMed] [Google Scholar]
- 30.Singhal RL, Merali Z, Hrdina PD. Aspects of the biochemical toxicology of cadmium. Fed Proc. 1976;35(1):75–80. [PubMed] [Google Scholar]
- 31.Stowe HD, Wilson M, Goyer RA. Clinical and morphologic effects of oral cadmium toxicity in rabbits. Arch Pathol. 1972;94(5):389–405. [PubMed] [Google Scholar]
- 32.Faraone SV, Perlis RH, Doyle AE, Smoller JW, Goralnick JJ, Holmgren MA, et al. Molecular genetics of attention-deficit/hyperactivity disorder. Biol Psychiatry. 2005;57(11):1313–1323. doi: 10.1016/j.biopsych.2004.11.024. [DOI] [PubMed] [Google Scholar]
- 33.Andersson H, Petersson-Grawé K, Lindqvist E, Luthman J, Oskarsson A, Olson L. Low-level cadmium exposure of lactating rats causes alterations in brain serotonin levels in the offspring. Neurotoxicol Teratol. 1997;19(2):105–115. doi: 10.1016/s0892-0362(96)00218-8. [DOI] [PubMed] [Google Scholar]
- 34.Provias JP, Ackerley CA, Smith C, Becker LE. Cadmium encephalopathy: a report with elemental analysis and pathological findings. Acta Neuropathol. 1994;88(6):583–586. doi: 10.1007/BF00296497. [DOI] [PubMed] [Google Scholar]
- 35.Goodlad JK, Marcus DK, Fulton JJ. Lead and attention-deficit/hyperactivity disorder (ADHD) symptoms: a meta-analysis. Clin Psychol Rev. 2013;33(3):417–425. doi: 10.1016/j.cpr.2013.01.009. [DOI] [PubMed] [Google Scholar]
- 36.Park JI, Shim SH, Lee M, Jung YE, Park TW, Park SH, et al. The validities and efficiencies of Korean ADHD rating scale and Korean child behavior checklist for screening children with ADHD in the community. Psychiatry Investig. 2014;11(3):258–265. doi: 10.4306/pi.2014.11.3.258. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Percentage change in K-ARS scores associated with quartile increase of maternal blood cadmium.
Percentage change in K-ARS scores associated with 2-fold increases in maternal blood lead and cadmium levels in 6-year old children.
Percentage change in K-ARS scores associated with 2-fold increases in maternal blood cadmium levels in 6-year old children. (excluded current smokers)


