Abstract
Due to the heavy energetic demands to maintain bone homeostasis, the skeletal system is closely tied to whole-body metabolism via neuronal and hormonal mediators. Glucose, amino acids and fatty acids are the chief fuel sources for bone resident cells during its remodeling. Lipids, which can be mobilized from intracellular depots in the bone marrow, can be a potent source of fatty acids. Thus, while it has been suggested that adipocytes in the bone marrow act as ‘filler’ and are detrimental to skeletal homeostasis, we propose that marrow lipids are in fact essential for proper bone functioning. As such, we examine the prevailing evidence regarding the storage, use, and export of lipids within the skeletal niche, including from both in vitro and in vivo model systems. We also highlight the numerous challenges that remain to fully appreciate the relationship of lipid turnover to skeletal homeostasis.
Keywords: bone, osteoblasts, adipocytes, energy, fat, cholesterol, diet
Introduction
The bone marrow and its skeletal components expend a tremendous amount of daily energy to regulate hematopoietic turnover, acquire and maintain bone mass, and actively participate in whole-body metabolism (Confavreaux et al., 2009). Given the terrestrial nature of our existence, the human adult skeleton undergoes the daily stress of microfracture formation that requires repeated repair. This involves the active removal of bone by osteoclasts, which is exquisitely coordinated by a subsequent replenishment of new bone by osteoblasts in a process called ‘bone coupling’ or remodeling (Tang et al., 2009). The entire remodeling cycle takes approximately 120 days, and 10% of the human skeleton is normally remodeled every year (Feng X and MacDonald J, 2011). Remodeling requires the differentiation of mesenchymal stem-cell derived osteoblasts with subsequent collagen synthesis, the recruitment of hematopoietic-derived osteoclasts necessary for bone resorption and the communication of skeletal signals to those cells from the deeply embedded osteocytes, derived from terminally differentiated osteoblasts. In sum this translates into major energy demands. Fortunately, the skeleton receives up to 10% of overall cardiac output, and thus circulating substrates could meet that demand under normal circumstances (Riddle and Clemens, 2017). However, as we discuss below, lipids within the marrow niche are also available and can be utilized to provide additional fuel during bone remodeling, cell differentiation or states of active metabolic stress. In this perspective we maintain that fatty acids and various lipids are essential for skeletal health, and that perturbations in substrate utilization within the marrow affect whole-body metabolism and can be linked to chronic diseases such as obesity and osteoporosis.
The Energy Demands and Resources of the Skeletal System
The currency for cellular energy is adenosine triphosphate (ATP), which is generated by the hydrolysis of high energy bonds, although ATP can be produced from other sources including nicotinamide adenine dinucleotide phosphate (NADPH). Cellular energy, or what is now often referred to as cellular ‘bioenergetics’, is a highly sophisticated, evolutionarily conserved series of metabolic reactions that sense substrate availability and derive sufficient ATP to feed its cellular needs, including cell survival, proliferation, and function. Importantly, for the skeleton these cellular networks are closely tied to whole body metabolism via afferent signaling to the brain, liver and peripheral fat depots as well as efferent networks back to the skeletal niche (Confavreaux, et a.l 2009; Liu et al., 2016; Motyl, et al., 2017). The cells found within the bone marrow niche, defined in this perspective as osteoblasts, osteoclasts, adipocytes, osteocytes, and skeletal stem cells, derive cellular energy almost exclusively from glucose, amino acids, and lipids. With respect to glucose, during states of high marrow demand, i.e. anemia, granulocyte-colony stimulating factor (gCSF) activation or treatment, stress, hemolysis thalassemia or sepsis, there is a marked increase in glucose utilization by a full range of hematopoietic elements (Oburoglu et al., 2016). These changes are of sufficient magnitude that they now can be visualized and accurately estimated by 2-deoxy-[fluorine-18]fluoro-D-glucose (FDG)- positron emission tomography (PET) scans (Asabella et al., 2019). All of the cells in the skeletal niche utilize glucose at rest and in response to stress, as well as during states of high bone remodeling, such as acquisition of peak bone mass or during intermittent PTH (iPTH) treatment for osteoporosis (Esen et al., 2015). Not surprisingly, the use of glucose as a primary fuel relies on its relative ease of access from the extracellular fluid and rapidity of ATP generation during glycolysis. Yet because fatty acids have more stored energy per mole than glucose, fatty acid oxidation as a source of ATP is particularly relevant during states of intra- or extracellular glucose starvation and early in the differentiation process (Guntur, 2018). Although lipids provide a rich source of fatty acids, until recently little attention was focused on lipid metabolism in the skeletal niches. In part this was due to the belief that lipid-laden cells in the marrow were inert, or ‘fillers’, that occupied space left by absent hematopoietic and skeletal elements. Importantly, seminal studies from the Karsenty group in the last decade, demonstrating the link between the skeleton and whole-body energy metabolism, provided the momentum to re-examine how fuel is used in the niche and how that relates to homeostatic mechanisms in the brain and elsewhere (Lee et al., 2018; Karsenty and Olsen, 2016). A closer look at the niche cells and their relationship to lipid storage, utilization and export provide important clues as to the essential nature of fatty acids in the marrow.
Lipids in the Skeletal Niche
There is no evidence in healthy rodents or other mammals that ectopic lipid is deposited in the bone marrow independent of intracellular structures. Rather, well defined lipid droplets are found in skeletal and mesenchymal stem and mature cells as well as macrophages of the skeletal niche. These droplets have historically been viewed as a passive means for mature adipocytes to sequester free fatty acids (Martin and Parton, 2006). But lipid droplets are incredibly dynamic structures. Notably lipid droplets of varying size are present in most living cells, although their function has remained largely underappreciated, particularly in skeletal biology. Lipid droplets were first observed in bone cells as early as 1965, specifically when Enlow and colleagues described them in osteoblasts around the vicinity of the Haversian canal (Enlow et al, 1965). While our lab has recently described lipid droplets in osteoblast progenitors in vitro (Rendina-Ruedy et al., 2017), others have noted them in vivo with steroid treatment (Kawai et al., 1985), ethanol excess (Maurel et al., 2012; Wang et al., 2003), and ageing (McGee-Lawrence et al., 2016). The most notable clinical example is steroid-induced osteonecrosis of the femoral head, which histologically demonstrates the accumulation of large lipid droplets in osteocytes, a phenomenon the authors’ coined ‘fatty necrosis of osteocytes’ (Maurel et al., 2012). Additionally, alcohol has been shown to increase TAG storage in bone marrow stromal cells (BMSCs) under osteogenic conditions (in vitro), and the accumulation of lipid droplets in osteocytes (in vivo) (Maurel et al., 2012; Wang et al., 2003). More recently, deletion of histone deacetylase (Hdac)-3 in BMSC-derived osteoblasts has been shown to increase intracellular lipid droplets (McGee-Lawrence et al., 2016). The authors describe that both ageing and dexamethasone treatment, two scenarios in which bone mass are decreased, are also associated with lower Hdac3 expression and increased lipid droplets in osteoblasts (McGee-Lawrence et al., 2016). Given the intimate, albeit somewhat elusive relationship between cells of the osteoblast lineage and bone marrow adipocytes, increased lipid droplet accumulation in osteoblasts and/or osteocytes, are also conditions associated with an expansion of bone marrow adipose tissue and low bone mass.
While the function of lipid droplets has not been fully clarified in osteoblastic cells, these organelles are the defining morphological feature of bone marrow adipocytes. Marrow adipocytes can occupy more than 70% of the marrow space in older individuals and that proportion increases in conditions such as osteoporosis, chronic renal failure and diabetes mellitus (Kirkland et al., 2002; McCabe, 2009; Cawthorn et al., 2014). In rodents, marrow fat also increases with age, particularly in the long bones and thus the mouse represents a good model for studying the dynamics of these cells. However, their location within the marrow space, the difficulty in isolating these cells from the rodent, the cross-sectional nature of the observations and the relatively crude histomorphometric methods for studying these adipocytes are impediments to understanding their function.
Notwithstanding, there is considerable indirect evidence these cells can expand or contract in response to metabolic, nutritional, hormonal and environmental cues. But there is an important caveat. Virtually all studies in mice and most in humans are cross-sectional, hence some degree of extrapolation is required to assume that a measurement at a single time can be extrapolated back to the genetically same organism at baseline. For example, in cross-sectional studies of postmenopausal women, loss of estrogen leads to a remarkable increase in bone marrow adipose tissue volume that is reduced relative to controls by estrogen replacement (Limonard et al., 2015). But, only a handful of longitudinal studies have been undertaken in humans using paired bone biopsies or magnetic resonance spectroscopy to assess real-time changes in size and number of adipocytes within the marrow (Cohen et al., 2013, Fazeli et al., 2018). A similar limitation occurs in mouse experiments. Scheller et al. (2015) showed that cold exposure in mice results in a reduction in marrow adipocyte size and number, again through cross-sectional studies. A high-fat diet in C57BL/6J mice results in the rapid accumulation of marrow adipocytes (Doucette et al., 2015; Rendina-Ruedy et al., 2015; Tencerova et al., 2018). And intra-cerebroventricular leptin administration decreases adipocyte size (Hamrick et al., 2007). Thus the presence of these large, unilocular lipid droplets implies that bone marrow adipocytes possess lipid droplets with identical cellular mechanisms relative to the storage, mobilization and utilization of fatty acids as peripheral adipocytes. Importantly, as noted, the droplets are not ectopic but rather represent true adipocytes as confirmed from lineage tracing studies using dual reporter systems and perilipin staining (Sebo et al., 2019). However, still up for debate is the question of whether pre-adipocytes in the marrow may possess the machinery for adipogenesis without the presence of lipid droplets.
Based on their structure it is likely marrow adipocytes could act as a large reservoir for fatty acid storage in states of excess fuel, as well as a source of adipokines that could mediate skeletal remodeling in a paracrine fashion and systemic metabolism in an endocrine manner. Indeed, adiponectin has been proposed as one such adipokine, linking whole body homeostasis to marrow adiposity (Cawthon et al, 2014). Another cytokine released from marrow adipocytes that has potent local effects on skeletal remodeling is RANKL, which drives osteoclastogenesis and ultimately bone resorption (Fan et al., 2017). Importantly, RANKL expression in the marrow adipocyte is much higher than in peripheral adipose depots supporting the tenet that these fat cells have a unique signature reflective of their site of origin (Fan et al, 2017).
One of the biggest challenges in defining the role of lipid droplets in bone marrow adipocytes has been the inability to have a fully representative ex vivo model that simulates in vivo conditions in order to better understand their function. Progress has been made in developing 3D adipocyte culture systems with silk scaffolds that have distinct gene expression differences from conventional 2D cultures, but the differences in oxygen concentration ex vivo vs in situ, as well as the notable absence of immune and endothelial cells, limit interpretation (Fairfield et al., 2019). Moreover, none of these models consider the importance of the vascular network as both a means of supplying progenitors, and as a necessary element for the function of mature adipocytes.
Lipid Utilization for Mitochondrial Respiration
Given the presence of lipid droplets in osteoblasts, osteocytes and bone marrow adipocytes, it is likely that lipids can be broken down and mobilized via lipolysis when cellular energy is in demand. Lipolysis is a generalized term describing the catabolism of lipids via mechanisms involving (1) lipid droplet-associated cytoplasmic lipases, and/or (2) autophagosome sequestration of lipids and subsequent degradation by lysosomal acid lipase (Lipa), termed lipophagy (Zhou et al., 2019). The more commonly described lipolysis involves cytoplasmic lipases acting on the lipid droplet in three distinct, sequential steps: (1) adipose triglyceride lipase (ATGL) hydrolyzes TAGs to release a fatty acid, yielding diacylglycerol (DAG); (2) hormone sensitive lipase (HSL) which then converts DAG to monoacylglcerol (MAG), again releasing a fatty acid; (3) while the last fatty acid is liberated from glycerol via monoglyceride lipase (MGL) (Lampidonis et al., 2011). Both mechanisms of lipolysis yield the same products, free fatty acids and free glycerol. While glycerol can be transported out of the cell via aquaporins, free fatty acids undergo β-oxidation and subsequent oxidative phosphorylation (Beitz et al., 2015). Conversely, if the cells do not need free fatty acids for ATP generation, these can be shuttled out of the cells and enter remote cells or circulate to more distant tissues/cells. Marrow adipose tissue was recently shown to undergo cytoplasmic lipase-mediated lipolysis, however this unique depot appeared to be less responsive to β-adrenergic stimulation compared with white adipose tissue (WAT) (Scheller et al., 2018). Therefore, while the process of lipolysis occurs in marrow adipocytes and is thought to occur in cells of the osteoblast lineage, the stimulus and/or responsiveness may be unique to these cell populations. A conventional paradigm in this field is that the location of adipose depots dictates the function of the adipocytes that reside within those tissues. However, because it is unknown how the marrow adipocyte works, it remains uncertain whether resistance to beta adrenergic stimuli is an inherent and unique property of these bone marrow cells.
It should be noted that while fatty acids are the most studied substrate for lipid droplet biogenesis, glucose (and/or fructose) may also be used as the lipid droplet core precursor via de novo lipogenesis. This process involves the uptake of glucose and the classic biochemical pathways to generate pyruvate. Once pyruvate enters the mitochondria and has been converted to acetyl-CoA, it is then condensed to form citrate via citrate synthase. At this particular step feedback from multiple enzymes determine whether citrate will continue through the tricarboxylic acid (TCA) or Krebs cycle, or be shuttled out in to the cytosol for the generation of TAGs. In general, these processes include lipogenesis that begins with citrate desaturation and elongation and concludes with the esterification of fatty acids to yield TAGs, packaged within the core of the lipid droplet (Ferramosca and Zara, 2014). Importantly the formation of TAGs also requires a glycerol backbone, which can be generated from dihydroxyacetone phosphate (DHAP), produced during glycolysis (in adipocytes), or directly via glycerol kinase (in hepatocytes). Recently, inhibition of the rate limiting enzyme of de novo lipogenesis, fatty acid synthase (FAS or FASN), has been shown to increase bone formation and protect from ovariectomy (OVX)-induced bone loss, while also decreasing bone marrow adipose tissue (Bermeo et al., 2019). Moreover, circulating levels of FAS have been shown to relate to adipose tissue as well as bone biomarkers in pre-pubertal children (Prats-Puig et al., 2012). Although osteoblasts and chondrocytes have been metabolically characterized to rely heavily on glucose (Lee et al., 2018; Wei et al., 2015), it still remains unclear whether glucose utilization could contribute significantly to de novo lipogenesis, a prospect that would need to be proven through tracer flux and metabolomic studies.
Lipolysis implies that fatty acid liberation can then be used by the cell or its neighbors to generate energy. Exogenous and endogenous fatty acids destined for ATP generation must first be activated and esterified to coenzyme A (CoA) via acyl-CoA synthetase long chain family members (ACSLs). Following these reactions the ‘carnitine shuttle’ transports fatty acyl groups from the cytosol into the mitochondria (Figure 2a). The carnitine needed for this transport is easily obtained from dietary sources or by de novo synthesis; however it is interesting to note that carnitine levels often decrease with age. As such, carnitine supplementation in an ageing, OVX rat model of post-menopausal osteoporosis was able to increase bone mineral density (BMD) (Murad, 2016). While these data are compelling, recent data specifically targeting carnitine palmitoyltransferase II (CPTII), the obligate enzyme required to facilitate fatty acid entry in to the mitochondria, in osteoblasts, results in compromised bone acquisition in female mice (Kim et al., 2017a). Importantly, these bone-specific changes were associated with altered circulating lipid levels and body composition, emphasizing the central role of the skeleton in influencing whole body metabolism (Kim et al., 2017a).
Figure 2.
Cells within the skeletal niche utilize long chain free fatty acids (LCFA) for energy generation via two complimentary cellular catabolic processes of the carnitine shuttle and β-oxidation. (A) Long chain fatty acids (LCFA) destined for adenosine triphosphate (ATP) generation must first be activated and esterified to coenzyme A (CoA) via acyl-CoA synthetase long chain family members (ACSLs), which requires ATP and coenzyme A (CoA) yielding adenosine monophosphate (ANP) and inorganic pyrophosphate (PPi). Following these reactions the ‘carnitine shuttle’ transports fatty acyl groups via carnitine palmitoyltransferase I (CPTI) and II (CPTII), along with carnitine-acylcarnitine translocase (CACT) in to the mitochondria. (B) β-oxidation then utilizes two carbons of acyl-CoA by a series of reactions involving oxidation, hydration and cleavage ultimately generating acetyl-CoA from fatty acyl CoA. In additional to acetyl-CoA that can enter the Krebs cycle, β-oxidation also generates reducing equivalents FADH2 and NADH. Every round of β-oxidation utilizes two carbons (−2C) to generate acetyl-CoA. Additionally cells within the skeletal niche can acquire cholesterol via low density lipoprotein (LDL)-LDL receptor (LDLR) endocytosis or the cholesterol biosynthetic pathway. (C) Circulating LDL particles will interact with LDL receptor (LDLR) on the cell surface which results in LDL-LDLR endocytosis. Within the cell these endosomes will fuse with the lysosome whereby lysosomal acid lipase (LAL) hydrolyzes cholesterol esters (CE) and triacylglycerols (TAGS) to free cholesterol and free fatty acids, respectively, while the LDLR is recycled back to the cell surface. Both sterol regulatory element-binding protein (SREBP) and proprotein convertase subtilisin/kexin type 9 (PCSK9) regulate intracellular cholesterol via modulation of LDLR expression. (D) Endogenous cholesterol biosynthetic pathway which utilizes 2 acetyl CoA molecules to yield acetoacetyl-CoA via acyl-CoA cholesterol acyltransferase (ACAT). The rate limiting step next converts 3-hydroxy-3-methyl-glutaryl-coenzyme A (HMG-CoA) to mevalonate via HMG-CoA reductase (HMGCR). Subsequent steps include isopentyl pyrophosphate (PP), farnesyl PP to squalene. Final stages will include the cyclization of squalene to ultimately form cholesterol. Cholesterol lowering statins impair HMGCR, while anti-resorptive bisphosphonates inhibit farnesyl disphosphate synthase.
Following fatty acyl-CoA entry in to the mitochondria, β-oxidation occurs to generate acetyl-CoA, which enters the TCA, followed by ATP generation via oxidative phosphorylation. Briefly, β-oxidation utilizes two carbons of acyl-CoA by a series of reactions involving oxidation, hydration and cleavage (Figure 2b), ultimately generating acetyl-CoA. In additional to acetyl-CoA that can enter the Krebs cycle, β-oxidation also generates reducing equivalents FADH2 and NADH that can be coupled to the electron transport chain and ATP generation. Every round of β-oxidation utilizes two carbons to generate acetyl-CoA as well as additional reducing equivalents, and thus fatty acid length and saturation status determine the amount of ATP yield. In sum, catabolic metabolism of fatty acids yields more ATP than other substrates.
Given the energy demands of bone formation, fatty acids would seem to be ideal substrate candidates for osteoblast ATP generation. But it wasn’t until 1987 that Fleisch and colleagues identified fatty acids as a substrate capable of supplying energy to bone tissue and isolated cells (Adamek et al., 1987). In fact, the skeleton, by radiotracer studies accumulates a significant proportion of fatty acids, comparable to that observed for adipose tissue and muscle (Niemeier et al., 2005). In that same vein, one of the seminal studies in this area was undertaken by Frey et. al. who examined the role of fatty acids in osteoblastogenesis and bone formation using genetic models of the Wnt-low density lipoprotein receptor-related protein (LRP)-5 signaling pathway (Frey et al., 2015). Mice lacking the Lrp5 co-receptor specifically in osteoblasts and osteocytes exhibited low bone mass but also showed an increase in body fat with corresponding reductions in energy expenditure. Conversely, mice expressing a high bone mass mutant Lrp5 allele were leaner with reduced plasma triglyceride and free fatty acid levels. In this context, Wnt-initiated signals downstream of Lrp5, regulated the activation of β-catenin and thereby induced the expression of key enzymes required for fatty acid β-oxidation. Importantly, gain of function or loss of function mutations in Lrp5 not only affected the skeletal phenotype, but also whole body metabolism (Frey et al., 2015). These same authors subsequently demonstrated the importance of fatty acid metabolism during peak bone acquisition by disrupting CPTII activity (Kim et al., 2017a). Low bone mass, dyslipidemia and changes in body composition resulted from osteoblast specific genetic deletion of Cpt2 supporting the hypothesis that fatty acids are critical substrates for skeletal acquisition and maintenance. However, it remains uncertain whether fatty acid utilization as fuel is purely dependent on extracellular sources, or includes mobilization of fatty acids from lipid droplets in osteoblasts. Moreover, there is a certain context specificity to fuel utilization, such that the cells retain the flexibility to choose substrates depending on their need and most importantly their differentiation status (Guntur et al., 2018). Indeed, an osteoblast comes in many forms, from the progenitor pre-osteoblast and lining cell, to the plump collagen synthesizing, endosteal mature osteoblast. Hence, more elucidation of the energy needs at each time point of differentiation is required to firmly define when and how lipid metabolism affects osteoblast differentiation.
In respect to lipid metabolism in the marrow adipocyte, not surprisingly, their metabolism has yet to be rigorously described (Sebo et al., 2019) due to the difficulty in isolating these cells and working with them in vitro. However, their defining feature, the lipid droplet, allows us to formulate some hypotheses that can be readily testable. For example, just as in other adipose depots, bone marrow adipocytes are presumed to use both glycolysis and oxidative phosphorylation. Importantly, several groups have now shown that intermittent administration of PTH to mice or humans can reduce bone marrow adipose tissue volume (Cohen et al., 2013; Fan et al., 2017) even though PTH drives glycolysis in most circumstances (Esen et al., 2015). As such, our lab recently demonstrated that acute treatment with PTH was capable of increasing bone marrow adipocyte mitochondria respiration (Maridas et al., 2019). Conversely, while prolonged treatment with PTH was still able to induce MAT lipolysis, the liberated free fatty acids were not being utilized for mitochondrial respiration but instead shuttled out of the marrow adipocytes (Maridas et al., 2019). Perhaps most importantly these liberated fatty acids were shown to be directly taken up by bone marrow stromal cells in culture (Maridas et al., 2019). These data demonstrated a direct relationship whereby bone marrow adipocytes were capable of supplying neighboring osteoblasts with fatty acid substrates for energy utilization. That tenet is also plausible because of the topography of the bone marrow niche, in which the adipocytes are often found adjacent to bone lining cells on the endosteal surface (Fan et al., 2017). But, to date it remains to be determined whether this occurs in vivo, and whether bone marrow adipocytes can supply fatty acids to other cell types that reside in the niche.
As a cautionary note, it is important to consider that not all ‘fatty acids’ are created equally and thus data interpretation can be convoluted. For example, multiple studies have used palmitate to study fatty acid’s impact on osteoblasts (Gunaratnam et al., 2013; Yang et al., 2018; Yeh et al., 2014). One study in particular reported that palmitate increased osteoblast cell death via autophagy-apoptosis pathway (Gunaratnam et al., 2013). While these data provide insight, it is equally important to frame these data within the context of the study. For example, palmitate has also been shown to significantly increase mitochondrial reactive oxygen species (ROS) production, which correlates with mitochondrial dysfunction and induction of apoptosis (Yuzefovych et al., 2010). Conversely, the unsaturated free fatty acid oleate does not have this effect on mitochondria and can actually ameliorate palmitate-induced mitochondrial damage (Gillet et al., 2015; Yuzefovych et al., 2010). Therefore, the types of free fatty acids used at both the in vitro and in the in vivo level should be carefully scrutinized before broad conclusions are drawn about fatty acid use in the niche
Lipid Droplets and their relationship to Exogenous Dietary Fats
Lipid metabolism begins in its simplest form through exogenous, dietary triacylglycerol (TAG) and cholesteryl esters (CE) that are packaged in chylomicrons and circulate throughout the lymphatic system (Dash et al., 2015). Chylomicrons then interact and essentially ‘dock’ on to the cell surface via apolipoproteins embedded in their membrane. This interaction facilitates the function of extracellular lipases, including lipoprotein lipase (LPL) and hepatic lipase (HL), which hydrolyze free fatty acids from the TAG-core of the chylomicrons, allowing entry of the TAGs into the cell via specific transport proteins (Ramasamy, 2014).
While the liver remains the primary site for post-prandial chylomicron clearance, surprisingly, bone tissue clears 20% of chylomicron remnants relative to liver (Niemeier et al. 2008). Importantly, chylomicrons can be delivered to and directly taken up by bone cells in vivo due in part to the rich vascular supply in the marrow (Riddle and Clemens, 2017). At least one group has proposed that bone and the bone marrow can concentrate chylomicron remnants, serving a similar function as the sinusoidal endothelial cells of the liver (Niemeier et al., 2008). This purported function primarily has been discussed within the context of vitamin K’s role in the carboxylation status of osteocalcin, but certainly point to an intimate relationship between bone and exogenous lipids (Neimeier et al., 2005). Additionally, while bone cells do not express high amounts of LPL, an unbiased genome-wide expression analyses (GWAS) identified HL (encoded by the Lipc gene) as a candidate differentially regulated during osteoblastogenesis (Bartelt et al., 2014). Hence, the hydrolysis of free fatty acids from TAGs within the chylomicron core, presumably by HL on osteoblastic cells, and/or circulating free fatty acids permits the uptake by cells in the bone marrow niche. That step is mediated by CD36 which is expressed on cells of the osteoclast (Koduru et al., 2018) and osteoblast (Staines et al., 2014) lineage as well as marrow adipocytes (Tabe et al., 2017). As such, genetic deletion of CD36 in mice results in a low bone-mass phenotype due to a reduction in osteoblast numbers and low bone formation rates (Kevorkova et al., 2013). While the global nature of the deletion may be confounded by other factors, ex vivo and in vitro cultures corroborated the cell-autonomous role of CD36 as bone marrow stromal progenitors exhibited impaired osteoblastogenesis (Kevorkova et al., 2013). Much less is known relative to other fatty acid transport proteins (e.g., fatty acid transport proteins, FATP1–5 or fatty acid binding proteins, such as FABP4) in osteoblasts; however, osteoblasts do express a number of fatty acid transport genes. Taken together, these lines of evidence support the capacity for cells of the bone marrow niche, including osteoblasts and marrow adipocytes, to modulate circulating chylomicrons.
Obesity, Type 2 diabetes and the Marrow Niche
The relationship between obesity and bone turnover is complex, so it is not surprising that the impact of Type 2 diabetes (T2D) on skeletal metabolism is also complicated. For example, increased adiposity or ‘obesity’ is believed to exert a somewhat osteoprotective role in that additional weight serves as an anabolic signal for bone (Cao, 2011). This added ‘loading’ of the skeleton may favor osteoblastogenesis and bone formation. While it remains tempting to speculate that obesity also supports bone formation by increasing the bioavailability of exogenous lipids via chylomicron remnants, this has not been established in vivo. Moreover, given the systemic nature of increased adiposity, obesity often results in many derangements of whole-body metabolism including hyperglycemia and marked inflammatory responses. It follows that one of the most striking health consequences related to the prevalence of obesity has been the staggering increase in cases of T2D. Over the last two decades, studies designed to determine whether T2D influenced fracture risk and thus altered remodeling, were based solely on assessment of areal BMD and revealed mixed results, with the preponderance of the evidence indicating that patients were not at increased risk (van Daele et al., 1995; Piepkorn et al., 1997; Rishaug et al., 1995; Stolk et al., 1996). However, subsequent studies with fracture as the primary outcome variable have challenged these initial findings and the clinical evidence now indicates: (i) patients with T2D have an increased risk of fracture, independent of BMD; (ii) fracture risk in T2D is underestimated when using BMD; and (iii) fracture risk increases with increasing duration of T2D (Farr et al., 2014; Janghorbani et al.; de Liefde et al., 2005; Nicodemus et al., 2001; Schwartz, 2011; Schwartz et al., 2001).
The apparent disconnect between BMD and fracture risk in T2D continues to perplex researchers and clinicians alike, although, a consistent finding appears to be that diabetic patients and obese animal models of T2D demonstrate impaired bone turnover, particularly reduced bone formation. In fact, these observations have led to the sweeping conclusion that fatty acids/lipids are lipotoxic and ‘bad’ for bone. However, it is also conceivable that the inability to appropriately utilize these substrates results in impaired osteogenesis, not lipotoxicity per se. Therefore instances of increased circulating lipid profiles (i.e., obesity) do not always extrapolate to increased lipid catabolic metabolism for cells in the skeletal niche. Similarly T2D is often accompanied by elevated LDL, non-esterified fatty acids (NEFA), and triglycerides (TG) (Mooradian, 2009). In addition to this dyslipidemia various tissues also accumulate and expand lipid droplets including WAT, hepatocytes, and myocytes (Barnes, 2011; Li et al., 2015; Richard and Lingvay, 2011). While this dysfunction in cellular lipid metabolism involves an increase in circulating lipid substrates, it is also accompanied by alterations in lipolysis and β-oxidation (Duncan et al., 2007; Muoio and Newgard, 2008). Thus, it could be hypothesized that the inability to properly break down and utilize fatty acid substrates is particularly detrimental to overall cellular function. Taken together, these data underscore the importance of not only the availability of fatty acid substrates, but also a cell’s ability to mobilize these stores for ATP generation, which could be compromised during obesity-related metabolic derangements.
IV. Bone Metabolic ‘Competition’ Modulates Whole-Body Metabolism
The concept of “metabolic competition” was originally proposed as a mechanism to explain the nature of cancer growth. Briefly, this tenet was based on the notion that cancer cells, in their hypermetabolic state, out-compete other cells within the tumor microenvironment for metabolic substrates (Chang et al., 2015). If indeed this competition occurs it would paralyze regulatory T-cells due to lack of substrate from tumor-imposed metabolic restrictions within the niche (Chang et al., 2015). Recent studies in multiple myeloma suggest that tumor infiltration of the niche is associated with utilization of marrow adipose lipids, and that myeloma cells can even exhibit adipogenic characteristics, often referred to as ‘adipomimicry’ (Caers et al., 2007; Liu et al., 2015; Reagan et al., 2016; Tirado-Vélez et al., 2012; Trotter et al., 2016, Reagan MR, personal communication, 2019). This could render osteoblasts without the necessary fuel to maintain bone remodeling and thus may be responsible for the reduction in bone formation seen in myeloma patients (Hameed et al., 2014).
Here we consider ‘competition’ in the context of bone remodeling and whole-body metabolism. For example, alterations in bone homeostasis could drive changes in systemic lipid metabolism (Figure 3). To test this hypothesis it is reasonable to examine instances of high or low bone turnover and the association with subsequent global metabolic parameters. Several animal studies have demonstrated that during states of high bone formation, glucose sensitivity is enhanced (Kim et al., 2017b). By shunting fuel to the skeletal niche, there may be enhanced insulin sensitivity, which may also be related to release of osteocalcin from the skeletal matrix (Lee and Karsenty, 2008; Liu, 2016). Similarly, in states of calorie restriction in mice, adiponectin secretion from the marrow adipocyte, drove an increase in insulin sensitivity (Cawthorn et al., 2014). One possible mechanism for the increase in adiponectin secretion in anorexia and other calorie deficient states could be increased energy utilization by the marrow to preserve bone cell function under nutritional stress.
Figure 3.
Implications for bone homeostasis impacting whole body lipid metabolism relative to metabolic competition hypothesis. (Top) During times of active or high bone formation cells of the osteoblast lineage are rapidly utilizing lipids (i.e., fatty acids, triacylglycerol or TAGs, and low density lipoprotein or LDL) and lipid precursors (glucose), drawing them out of the circulation. This action thereby decreases their deposition and accumulation in peripheral cells (tissues) including adipocytes (adipose tissue), hepatocytes (liver), and myocytes (muscle). (Bottom) Alternatively, when bone formation is low, these lipids and lipid substrates are now readily available in the circulation and subsequently become stored in peripheral tissues.
There are several other aspects of lipid metabolism and bone remodeling that are clinically relevant and may support the principal of ‘metabolic competition’. Teriparatide or (human PTH 1–34) and PTHrP are two of the three FDA-approved anabolic agents to treat severe osteoporosis. While the ability of these agents to increase bone formation has been impressive, albeit of relatively short duration, it remains underappreciated that PTH can also modulate not only marrow adiposity but whole-body lipid metabolism. Jeon et al. (2014) showed one year of teriparatide treatment not only increased lumbar spine BMD, but these changes were accompanied by decreased total cholesterol and triglycerides. Perhaps even more impressive were the decreases in body weight and body mass index (BMI) (Jeon et al., 2014). These systemic effects may be mediated in part via direct PTH effects from lipolysis and fatty acid utilization in WAT, BAT, and MAT (Kir et al., 2014; Larsson et al., 2016; Maridas et al., 2019). Importantly the receptor for PTH and PTHrP, PTH1R, is highly expressed on bone marrow adipocytes (Fan et al., 2017). This could account for a reduction in marrow adipocyte size with intermittent PTH treatment as well as the finding of an enhanced skeletal responsiveness during states of high bone marrow adiposity (Maridas et al., 2019). Moreover, the ability for PTH to stimulate the transfer of fatty acids from bone marrow adipocytes to stromal cells provide further evidence in vitro that marrow lipids may have clinical relevance (Maridas et al., 2019). Furthermore, bone biopsies from osteoporotic and normal controls during hip arthroplasty revealed that osteoporotic subjects not only exhibited lower osteoblastogenesis compared to ‘healthy’ controls, but also exhibited impaired triglyceride metabolism (Dragojevič et al., 2013). Moreover both hormone sensitive lipase and lipoprotein lipase expression were reduced suggesting impaired fatty acid uptake and release from bone cells. This is consistent with preclinical studies using unbiased lipidomic analyses demonstrating profound changes in lipid metabolism within mineralized bone tissue during estrogen deficiency (Zhao et al., 2018). Thus, there is evidence that the decrease in bone formation observed in some osteoporotic patients is associated with impaired lipid metabolism within the skeleton. However, once again it should be noted that these are purely association studies, hence a true cause and effect mechanism has not been established.
In another clinical scenario, loading of the skeleton or enhanced mechanical stimulation is accompanied by improved lipid profiles, as well a reduction in marrow adipose tissue. Conversely, bouts of unloading or weightlessness not only result in a profound deterioration of bone mass but also dyslipidemia and enhanced marrow adiposity (Rendina-Ruedy and Rosen, 2017; Simpson et al., 2016). To this end a recent study demonstrated that microgravity specifically alters osteoblast mitochondria, impairing ATP synthesis and inhibition of fatty acid β-oxidation (Michaletti et al., 2017). Hence, another putative anabolic skeletal stimulus, weight bearing, an activator of some of the same pathways as PTH, also drives global metabolic parameters via changes in peripheral adipose depots.
The Paradox of Marrow Adiposity
In mice, bone marrow clears about 10% of the fatty acid intake after an oral meal, with only muscle, brown fat and liver using a greater percentage of dietary fatty acids (Niemeier et al., 2008). And marrow adipose tissue (MAT) comprises about 10% of the lipid stores by weight in humans, likely reflecting its capacity as a major storage depot for energy (Cawthorn et al., 2014). The relative proportion of MAT to total fat stores may increase significantly as marrow adipocytes expand in both size and number under the influence of a high-fat diet or with calorie restriction (Doucette et al., 2015, Cawthorn et al., 2014). Similarly, there is now emerging evidence that osteoblasts and mechanosensing osteocytes also expand and/or accumulate their lipid droplets in conditions related to excess energy availability or exposure to particular environmental toxins (McGee-Lawrence, 2016; Kawai, 1985). Paradoxically, MAT hyperplasia and hypertrophy occurs during calorie restriction or starvation in most mammals including humans (Cawthorn et al.,2014; Bredella et al.,2009). In anorexia nervosa, the proportion of fat in the marrow relative to other depots exceeds 30% (Cawthorn et al., 2014). Similarly, in patients with Type I diabetes mellitus there is enhanced MAT, possibly reflecting chronic intracellular glucose starvation (McCabe, 2009). In contrast, patients with Type 2 diabetes (T2D) and obesity, with an abundance of exogenous substrates, have variable vertebral marrow fat content based on MRI analyses (de Aruajo et al., 2017).
The paradox that diet-induced obesity in mice and acquired (i.e. calorie restriction) or genetic lipodystrophy are associated with increased marrow adiposity has not been resolved and remains a major challenge in this field. Why would marrow adipocytes store lipids in states of acute or chronic calorie deficiency? One possibility is that this represents a protective mechanism permitting the marrow to have readily available substrate to maintain hematopoiesis and bone remodeling in a manner analogous to what has been reported with lymph tissue during chronic starvation in animals (Elmas et al.,2008; Nelson and Demas,1996). Another hypothesis is that marrow fat fills the space previously occupied by skeletal tissue because of a lineage shift between pre-adipocytes and pre-osteoblasts due to lack of substrate availability. Finally, another theory holds that marrow adiposity during starvation is a function of a unique thermogenic program induced by lack of substrates and designed to maintain temperature in the niche. Although patients with anorexia nervosa have lower body temperature, little evidence has been presented that the marrow adipocyte has beige fat-like properties (Bredella et al., 2009, Sebo et al., 2019). However, experimental approaches to more fully understand the contrast between starvation-induced lipolysis in the peripheral tissue, and lipid storage in the marrow will require innovative approaches that are not currently available.
Cholesterol and its Role in the Niche
For decades epidemiological data have demonstrated a compelling link between serum cholesterol and bone health (Farhat and Cauley, 2008; Makovey et al., 2009; Mandal, 2015; Yerges-Armstrong et al., 2013). Most of these studies have reported an inverse correlation between low density lipoprotein (LDL)-cholesterol and BMD but the mechanism by which cholesterol regulates skeletal remodeling is not clear. All tissues, including cells in the marrow niche, utilize cholesterol for the maintenance of membrane permeability and fluidity. As such, cholesterol homeostasis is a tightly regulated process. The most extensively characterized pathways regulating cholesterol status include (1) exogenous uptake via LDL-LDLR endocytosis (Figure 2c); and (2) the cholesterol biosynthetic pathway, both of which have been described in osteoblastic cells (Figure 2d).
While exogenous dietary cholesterol is initially packaged and circulated in chylomicrons as cholesteryl esters (CE), the majority of these particles are remodeled in the liver and circulate as very LDL or VLDL and LDL. These circulating LDL particles will interact with LDL receptor (LDLR) on the cell surface which results in LDL-LDLR endocytosis. Within the cell these endosomes will fuse with the lysosome whereby lysosomal acid lipase (LAL) hydrolyzes CE to free cholesterol, and LDLR is recycled back to the cell surface. While LAL activity is critical for the release of free cholesterol, sterol regulatory element-binding protein (SREBP) and proprotein convertase subtilisin/kexin type 9 (PCSK9) regulate intracellular cholesterol via modulation of LDLR expression. LAL deficiency leads to cholesteryl ester storage disease (CESD) in adults and Wolman’s syndrome in children (Boldrini et al., 2004). Consistent with LAL’s function, patients with this condition demonstrate markedly abnormal cholesterol metabolism with foamy macrophages and premature atherosclerosis (Desai and Wilson, 2000; Hoffman et al., 1993; Reiner et al.; Tylki-Szymanska and Jurecka, 2014). There is a paucity of data relative to LAL, SREBP, and PCSK9 in cells of the skeletal niche, however, osteoblasts express all three of these genes. In fact, our lab recently reported that the LAL deficiency mouse model (LAL−/−) has low trabecular and cortical bone mass and that these alterations appear to be driven in part by a cell-autonomous role for LAL in osteoblasts (Rendina-Ruedy et al., 2018).
In addition to the epidemiological data relative to cholesterol and bone as well as the LAL deficient model, in vivo studies of mice maintained on an ‘atherogenic’ diet (1.25% cholesterol, 15.8% fat, and 0.5% cholate) demonstrated compromised micro-architectural changes and suppressed bone formation (Parhami et al., 2001). Pelton et al. showed that a high cholesterol diet promoted an osteoporotic phenotype in mice although the mechanism was not delineated (Pelton et al., 2012). One possible mechanism for these changes was that high circulating LDL-cholesterol led to high intracellular free cholesterol in the osteoblast, exerting a negative impact on bone. However, an alternative hypothesis is that the cellular uptake of LDL is impaired, subsequently diminishing intracellular free cholesterol thereby impairing osteoblastic function. This latter explanation is tenable based on experimental model systems. For example, calvarial osteoblasts from LDLR−/− mice demonstrate reduced osteoblast differentiation and mineralization as noted by reduced alkaline phosphatase (ALP) activity and Von Kossa staining (Zhang et al., 2017). Additionally, decreased bone mineral density has been reported in individuals with a mutation in APOB, the apolipoprotein that facilitates LDL-LDLR binding (Yerges-Armstrong et al., 2013.). It should be noted that global LDLR deletion results in an increase in bone mass, but this has been attributed to impaired bone resorption due to a defect in osteoclast fusion (Okayasu et al., 2012).
The mechanism of action for two commonly used classes of agents, statins and bisphosphonates, provides further insights into the link between cholesterol biosynthesis and the skeleton. Independent of LDL-LDLR endocytosis, cells are able to endogenously synthesize cholesterol from acetyl-CoA precursors through a cholesterol biosynthetic pathway. Impairment of this pathway by statins, cholesterol lowering drugs which inhibit the rate limiting enzyme, 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase, have been shown to increase bone formation in vivo by increasing osteoblast activity (Chen et al., 2010; Maeda et al., 2001, 2004). While the precise mechanism of statin treatment on bone density has been somewhat controversial, a recent study using bidirectional Mendelian randomization noted that the positive effects of statins on BMD were due in part to lowered LDL (Zheng et al., 2018). These data imply that cholesterol generation via the biosynthetic pathway exerts a negative impact on osteoblasts, which is somewhat counterintuitive; however, the mechanism may involve the cell’s bioenergetic capacity. For example, while cholesterol is important for osteoblast function, generating endogenous cholesterol from acetyl-CoA requires 99 ATP molecules per cholesterol molecule synthesized, compared to 49 ATP for fatty acid synthesis or 10 ATP for glucose synthesis. Therefore, impaired exogenous cholesterol uptake from LDL would initiate the endogenous cholesterol biosynthetic pathway, compromising the overall biogenetic capacity of osteoblasts during a critical time when ATP generation is required such as during active bone formation. To this point, it is likely that osteocytes rely primarily on this pathway for cholesterol generation, as LDL particles (20–40 μm) are too large to circulate through the canalicular network.
The cholesterol biosynthetic pathway is also a target for the most frequently prescribed therapeutic class of osteoporosis drugs, the nitrogen-containing anti-resorptive bisphosphonates. Bisphosphonates inhibit osteoclast mediated bone resorption by blocking farnesyl pyrophosphate synthase in the mevalonate/cholesterol pathway, inducing osteoclast apoptosis (van Beek et al., 1999; Russell, 2007). The effects of bisphosphonates on osteoblast function are less clear but overall bone remodeling is suppressed with these agents (Maruotti et al., 2012), and in vitro, alendronate and zoledronate suppress osteoblast differentiation (Gao et al., 2017; Shimizu et al., 2012), suggesting there may be modest impacts on bone formation due to altered cholesterol biosynthesis.
Unresolved Questions, Challenges and Potential Opportunities
Cells in the marrow niche are evolutionarily programmed for survival and use distinct energy sources during physiologic and pathologic states. Defining the precise relationship between lipids and the skeletal niche in a protected and hypoxic environment leads to several unresolved questions and technological challenges but also potential opportunities for advancing the science. Metabolic flexibility is a characteristic feature of cellular programs. Yet much of what we learned about flexibility in normal and neoplastic cells derives from in vitro and in situ studies. The extracellular environment also determines cell fate, but little attention has been focused on the matrix and the marrow milieu as major contributors. This makes it incumbent to more completely recapitulate the marrow environment, particularly the hypoxic environment. As noted above, new 3-D matrices have shown potential but have their own limitations. Systems such as a ‘marrow on chip’ models would help better define cell-cell interactions and their substrate requirements. In vitro assays for oxygen consumption and glycolysis are limiting because of the artificial nature of the substrates and their mode of presentation. Preliminary studies using 2-photon microscopy of in vivo long bones to assess mitochondrial membrane potential are promising but widespread application has been limited. Single-cell technology has revolutionized our understanding of cellular heterogeneity and progress has been made in sorting pre-adipocytes and pre-osteoblasts as well as mutli-potent stem cells (Silberstein, et al., 2016). Importantly, more efficient ways to sort mature adipocytes from the marrow are essential to define the functional nature of these cells. Currently, fat cells are separated ex vivo from the rest of the marrow by simple centrifugation, but this results in marrow adipocytes that possess significant heterogeneity. Flow cytometry is often used to sort marrow progenitors but mature adipocytes break apart during sorting, leaving few ways to assess cell features ex vivo, In that same vein, there is a need to consider experimentally how the vascular network in the marrow delivers energy sources and also how adjacent stem cells use those substrates to maintain the viability of the niche. For example, seminal work by Prisby et al., (2011) demonstrated that PTH treatment can redirect vascular elements closer to the endosteum during high rates of bone formation. This could represent one mechanism whereby substrate sensing by targeted osteoblasts, might direct vascular invasion. Similarly, bone marrow exhibits a wide gradient of oxygen concentrations and as such, more focus in situ on the importance of hypoxia and its relevance to substrate utilization by niche cells is required. Major steps have been taken to define the early adipocyte and osteoblast progenitor in the marrow through lineage tracing, but even that technique is limited by the purity of the tissue specific promoter and the non-heritable factors that influence genetically identical mouse strains. Finally, and probably the most difficult challenge, is to define the functional aspects of those lipid-laden cells (i.e. marrow adipocytes and some osteoblasts) in situ. That will almost certainly require newer in vivo imaging techniques as well as other approaches using better genetic reporter systems. This is relevant in particular for understanding how T2D, in which insulin resistance is a major feature of peripheral adipocytes, affects the function and response of the marrow adipocyte. In that same vein, a very recent study suggests that some marrow adipocytes may be resistant to beta adrenergic induced lipolysis (Scheller et al., 2019).
Additionally, while we alluded to the ability of cells within the bone marrow niche to undergo lipophagy, to date no studies have investigated the ability of osteoblasts to undergo lipolysis via lipophagy; however, mounting evidence hints at a pivotal role for lipophagy in these cells. For example, multiple studies have reported that autophagy is essential for osteoblast differentiation/maturation, thus impairment of autophagy results in an osteopenic bone phenotype (Liu et al., 2013; Onal et al., 2013; Nollet et al., 2014). However, these studies primarily assessed ‘autophagy’ by visualizing autophagy-related proteins (i.e., phosphotidylethanolamine conjugation of microtubule associated protein light chain 3 or LC3B-II), which are also involved in lipophagy, rendering autophagy and lipophagy indistinguishable from one another (Tanida et al., 2004). Therefore, conclusions based solely on these means could in fact be attributed to the selective autophagy of lipids, or lipophagy. Moreover, transcription factor EB (TFEB), which has recently been shown to regulate osteoblast differentiation via ATF4/CHOP, is critical for lysosomal biogenesis, autophagy regulation, and lipid metabolism, further implicating lipophagy’s importance during osteoblast differentiation (Yoneshima et al., 2016). As such, these data remain provocative and suggest that lipophagy is a critical cellular process occurring within cells of the skeletal niche and further investigation is warranted. Importantly, a better understanding of additional mechanisms of selective autophagy including mitophagy in osteoblasts is an area of notable interest to more fully appreciate how mitochondrial dynamics impact the differentiation program.
In summary, significant advances have been made in understanding the relationship between lipids and bone marrow cells. We propose there is a fine balance metabolically between enough and too much lipid in the marrow niche. In the former, lipids can serve as a fuel source to benefit the work of the osteoblast, particularly during times of stress, whereas in the latter, particularly during substrate excess, lipids may impair the functional capacity of stem and progenitor cells.
Despite the advances noted in this perspective, the immediate opportunities include ways to define better methods for lineage tracing to ascertain the origin of the marrow adipocyte, and to provide functional studies of the bone marrow progenitor adipocyte population. In respect to the former, by identifying specific adipocyte markers, the use of a combination of Cre promoters could be employed to selectively label and/or delete marrow adipocytes in vivo. Notwithstanding, the question remains regarding the relevance of marrow fat to overall metabolic balance; i.e. what are the signals that arise from the CNS and/or peripheral sites that direct fuel utilization in the marrow and how are those signals transmitted back to the brain and other tissues to maintain whole-body homeostasis? If the appropriate in vivo techniques can be perfected in order to analyze fuel use by marrow adipocytes and their relationship to metabolic status, translational applications are likely to follow, as well as novel approaches to better understand the metabolic dysfunction(s) that lead to obesity and/or osteoporosis.
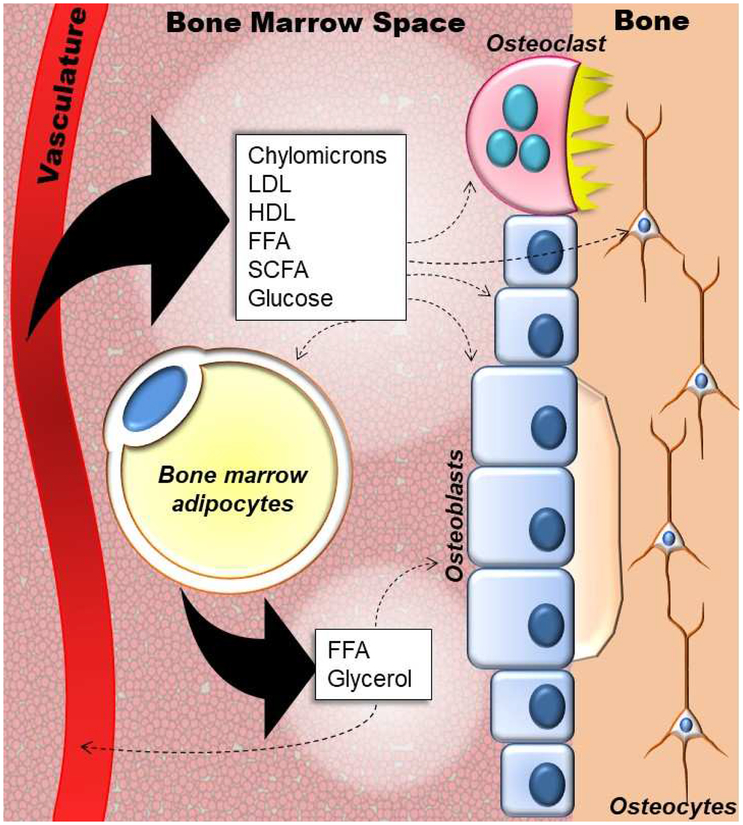
Figure 1.
Schematic diagram of lipids and fatty acids found within the skeletal niche. These include chylomicron remnants, low density lipoproteins (LDL), high density lipoproteins (HDL), free fatty acids (FFA), and short chain fatty acids (SCFA). Glucose is depicted as it can serve as a substrate for fatty acid synthesis via de novo lipogenesis. All or some of these elements are presumed to be available to cells with the bone microenvironment including bone marrow adipocytes (yellow), osteoblasts and bone lining cells (blue), osteoclasts (pink), and osteocytes via vascularization. Bone marrow adipocytes are also capable of providing free fatty acid and glycerol.
In this Perspective, Rendina-Ruedy et al highlight the prevailing evidence regarding the storage, use and export of lipids within the skeletal niche. They outline how lipid utilization helps meet the high energy demands of the bone marrow and its skeletal components needed for its various functions.
Acknowledgments
This project received support from the U.S. National Institutes of Health (NIH) National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) grants DK092759 (C.J.R.), DK112374 (C.J.R.); and National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS) grant AR072123 (E.R.R.).
Footnotes
Declaration of Interests
The authors declare no competing interests.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Adamek G, Felix R, Guenther HL, and Fleisch H 1987. Fatty acid oxidation in bone tissue and bone cells in culture. Characterization and hormonal influences. Biochem.J 248, 129–137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Araújo IM, Salmon CE, Nahas AK, Nogueira-Barbosa MH, Elias J Jr, de Paula FJ (2017) Marrow adipose tissue spectrum in obesity and type 2 diabetes mellitus. Eur J Endocrinol. 176(1):21–30. [DOI] [PubMed] [Google Scholar]
- Barnes AS (2011). The epidemic of obesity and diabetes: trends and treatments. Texas Hear. Inst. J 38, 142–144. [PMC free article] [PubMed] [Google Scholar]
- Bartelt A, Beil FT, Müller B, Koehne T, Yorgan TA, Heine M, Yilmaz T, Rüther W, Heeren J, Schinke T, et al. (2014). Hepatic lipase is expressed by osteoblasts and modulates bone remodeling in obesity. Bone 62, 90–98. [DOI] [PubMed] [Google Scholar]
- van Beek E, Pieterman E, Cohen L, Löwik C, and Papapoulos S (1999). Farnesyl Pyrophosphate Synthase Is the Molecular Target of Nitrogen-Containing Bisphosphonates. Biochem. Biophys. Res. Commun 264, 108–111. [DOI] [PubMed] [Google Scholar]
- Beitz E, Golldack A, Rothert M, von Bülow J. (2015). Challenges and achievements in the therapeutic modulation of aquaporin functionality. Pharmacol Ther. 155:22–35. [DOI] [PubMed] [Google Scholar]
- Bermeo S, Al Saedi A, Vidal C, Khalil M, Pang M, Troen BR, Myers D, and Duque G (2019). Treatment with an inhibitor of fatty acid synthase attenuates bone loss in ovariectomized mice. Bone 122, 114–122. [DOI] [PubMed] [Google Scholar]
- Boldrini R, Devito R, Biselli R, Filocamo M, and Bosman C (2004). Wolman disease and cholesteryl ester storage disease diagnosed by histological and ultrastructural examination of intestinal and liver biopsy. Pathol.Res.Pract 200, 231–240. [DOI] [PubMed] [Google Scholar]
- Bredella MA, Fazeli PK, Miller KK, Misra M, Torriani M, Thomas BJ, Ghomi RH, Rosen CJ, Klibanski A. (2009). Increased bone marrow fat in anorexia nervosa. J Clin Endocrinol Metab. 94(6):2129–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caers J, Deleu S, Belaid Z, De Raeve H, Van Valckenborgh E, De Bruyne E, DeFresne M-P, Van Riet I, Van Camp B, and Vanderkerken K (2007). Neighboring adipocytes participate in the bone marrow microenvironment of multiple myeloma cells. Leukemia 21, 1580–1584. [DOI] [PubMed] [Google Scholar]
- Cao JJ (2011). Effects of obesity on bone metabolism. J. Orthop. Surg. Res 6, 30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cawthorn WP, Scheller EL, Learman BS, Parlee SD, Simon BR, Mori H, Ning X, Bree AJ, Schell B, Broome DT, et al. (2014). Bone marrow adipose tissue is an endocrine organ that contributes to increased circulating adiponectin during caloric restriction. Cell Metab. 20, 368–375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang C-H, Qiu J, O’Sullivan D, Buck MD, Noguchi T, Curtis JD, Chen Q, Gindin M, Gubin MM, van der Windt GJW, et al. (2015). Metabolic Competition in the Tumor Microenvironment Is a Driver of Cancer Progression. Cell 162, 1229–1241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen P-Y, Sun J-S, Tsuang Y-H, Chen M-H, Weng P-W, and Lin F-H (2010). Simvastatin promotes osteoblast viability and differentiation via Ras/Smad/Erk/BMP-2 signaling pathway. Nutr. Res 30, 191–199. [DOI] [PubMed] [Google Scholar]
- Cohen A, Stein EM, Recker RR, Lappe JM, Dempster DW, Zhou H, Cremers S, McMahon DJ, Nickolas TL, Müller R, et al. (2013). Teriparatide for Idiopathic Osteoporosis in Premenopausal Women: A Pilot Study. J. Clin. Endocrinol. Metab 98, 1971–1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Confavreux CB, Levine RL, Karsenty G 2009. A paradigm of integrative physiology, the crosstalk between bone and energy metabolisms. Mol Cell Endocrinol;310(1–2):21–9. doi: 10.1016/j.mce.2009.04.004. Epub 2009 Apr 17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Daele PL, Stolk RP, Burger H, Algra D, Grobbee DE, Hofman A, Birkenhäger JC, and Pols HA (1995). Bone density in non-insulin-dependent diabetes mellitus. The Rotterdam Study. Ann. Intern. Med 122, 409–414. [DOI] [PubMed] [Google Scholar]
- Dash S, Xiao C, Morgantini C, Lewis GF. (2015). New Insights into the Regulation of Chylomicron Production. Annu Rev Nutr. 35:265–94. [DOI] [PubMed] [Google Scholar]
- Desai NK, and Wilson DP (2016). Lysosomal Acid Lipase Deficiency in ENDOTEXT, eds by Wilson DP; www.endotext.com, [Google Scholar]
- Doucette CR, Horowitz MC, Berry R, MacDougald OA, Anunciado-Koza R, Koza RA, and Rosen CJ (2015). A High Fat Diet Increases Bone Marrow Adipose Tissue (MAT) But Does Not Alter Trabecular or Cortical Bone Mass in C57BL/6J Mice. J.Cell Physiol 230, 2032–2037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dragojevič J, Zupan J, Haring G, Herman S, Komadina R, and Marc J (2013). Triglyceride metabolism in bone tissue is associated with osteoblast and osteoclast differentiation: a gene expression study. J. Bone Miner. Metab 31, 512–519. [DOI] [PubMed] [Google Scholar]
- Duncan RE, Ahmadian M, Jaworski K, Sarkadi-Nagy E, and Sul HS (2007). Regulation of lipolysis in adipocytes. Annu. Rev. Nutr 27, 79–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elmas C, Erdogan D, Take G, Ozogul C, Nacar A, Koksal M. (2008) Ultrastructure of the thymus in diabetes mellitus and starvation. Adv Ther. 25(1):67–76. [DOI] [PubMed] [Google Scholar]
- Enlow DH, Conklin JL, and Bang S (1965). Observations on the occurrence and the distribution of lipids in compact bone. Clin.Orthop.Relat Res 38, 157–169. [DOI] [PubMed] [Google Scholar]
- Esen E, Lee SY, Wice BM, Long F. (2015). PTH Promotes Bone Anabolism by Stimulating Aerobic Glycolysis via IGF Signaling. J Bone Miner Res. 30(11):2137. [DOI] [PubMed] [Google Scholar]
- Fairfield H, Falank C, Farrell M, Vary C, Boucher JM, Driscoll H, Liaw L, Rosen CJ, Reagan MR. (2019). Development of a 3D bone marrow adipose tissue model. Bone. 118:77–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fan Y, Hanai J-I, Le PT, Dempster DW, Rosen CJ, Lanske B, Bi R, Maridas D, Demambro V, Figueroa CA, et al. (2017). Parathyroid Hormone Directs Bone Marrow Mesenchymal Cell Fate. Cell Metab. 25, 661–672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farhat GN, and Cauley JA (2008). The link between osteoporosis and cardiovascular disease. Clin. Cases Miner. Bone Metab 5, 19. [PMC free article] [PubMed] [Google Scholar]
- Farr JN, Drake MT, Amin S, Melton LJ, McCready LK, and Khosla S (2014). In Vivo Assessment of Bone Quality in Postmenopausal Women With Type 2 Diabetes. J. Bone Miner. Res 29, 787–795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fazeli PK, Faje A, Bredella MA, Polineni S, Russell S, Resulaj M, Rosen CJ, Klibanski A. (2018). Changes in marrow adipose tissue with short-term changes in weight in premenopausal women with anorexia nervosa. Eur J Endocrinol. 2018 December 1 pii: EJE-18-0824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng X and MacDonald JM. (2011) Disorders of Bone Remodeling. Ann Rev Pathology 6: 121–145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferramosca A, Zara V. (2014) Dietary fat and hepatic lipogenesis: mitochondrial citrate carrier as a sensor of metabolic changes. Adv Nutr. 5(3):217–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frey JL, Li Z, Ellis JM, Zhang Q, Farber CR, Aja S, Wolfgang MJ, Clemens TL, and Riddle RC (2015). Wnt-Lrp5 Signaling Regulates Fatty Acid Metabolism in the Osteoblast. [DOI] [PMC free article] [PubMed]
- Gao S, Zheng G, Wang L, Liang Y, Zhang S, Lao X, Li K, and Liao G (2017). Zoledronate suppressed angiogenesis and osteogenesis by inhibiting osteoclasts formation and secretion of PDGF-BB. PLoS One 12, e0179248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gillet C, Spruyt D, Rigutto S, Dalla Valle A, Berlier J, Louis C, Debier C, Gaspard N, Malaisse WJ, Gangji V, et al. (2015). Oleate Abrogates Palmitate-Induced Lipotoxicity and Proinflammatory Response in Human Bone Marrow-Derived Mesenchymal Stem Cells and Osteoblastic Cells. Endocrinology 156, 4081–4093. [DOI] [PubMed] [Google Scholar]
- Gunaratnam K, Vidal C, Gimble JM, and Duque G(2014) Mechanisms of Palmitate-Induced Lipotoxicity in Human Osteoblasts. Endocrinology.155: 105–116. [DOI] [PubMed] [Google Scholar]
- Gunaratnam K, Vidal C, Boadle R, Thekkedam C, and Duque G (2013). Mechanisms of palmitate-induced cell death in human osteoblasts. Biol.Open 2, 1382–1389 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guntur AR, Gerencser AA, Le PT, DeMambro VE, Bornstein SA, Mookerjee SA, Maridas DE, Clemmons DE, Brand MD, Rosen CJ. (2018). Osteoblast-like MC3T3-E1 Cells Prefer Glycolysis for ATP Production but Adipocyte-like 3T3-L1 Cells Prefer Oxidative Phosphorylation. J Bone Miner Res. 33(6):1052–1065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hameed A, Brady JJ, Dowling P, Clynes M, and O’Gorman P (2014). Bone disease in multiple myeloma: pathophysiology and management. Cancer Growth Metastasis 7, 33–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamrick MW, Della Fera MA, Choi YH, Hartzell D, Pennington C, Baile CA. (2007). Injections of leptin into rat ventromedial hypothalamus increase adipocyte apoptosis in peripheral fat and in bone marrow. Cell Tissue Res. 327(1):133–41. [DOI] [PubMed] [Google Scholar]
- Hoffman EP, Barr ML, Giovanni MA, and Murray MF (2015). Lysosomal Acid Lipase Deficiency in Gene Reviews ed by Adams MP. [Google Scholar]
- Janghorbani M, Feskanich D, Willett WC, and Hu F Prospective study of diabetes and risk of hip fracture: the Nurses’ Health Study. Diabetes Care 29, 1573–1578. [DOI] [PubMed] [Google Scholar]
- Jeon YK, Kim KM, Kim KJ, Kim IJ, Lim S-K, and Rhee Y (2014). The Anabolic Effect of Teriparatide is Undermined by Low Levels of High-Density Lipoprotein Cholesterol. Calcif. Tissue Int 94, 159–168. [DOI] [PubMed] [Google Scholar]
- Karsenty G, Olson EN. (2016). Bone and Muscle Endocrine Functions: Unexpected Paradigms of Inter-organ Communication. Cell. 2016 March 10;164(6):1248–1256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawai K, Tamaki A, and Hirohata K (1985). Steroid-induced accumulation of lipid in the osteocytes of the rabbit femoral head. A histochemical and electron microscopic study. J.Bone Jt. Surg.Am 67, 755–763. [PubMed] [Google Scholar]
- Kevorkova O, Martineau C, Martin-Falstrault L, Sanchez-Dardon J, Brissette L, and Moreau R (2013). Low-Bone-Mass Phenotype of Deficient Mice for the Cluster of Differentiation 36 (CD36). PLoS One 8, e77701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim SP, Li Z, Zoch ML, Frey JL, Bowman CE, Kushwaha P, Ryan KA, Goh BC, Scafidi S, Pickett JE, et al. (2017a). Fatty acid oxidation by the osteoblast is required for normal bone acquisition in a sex- and diet-dependent manner. JCI Insight 2 (16). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim SP, Frey JL, Li Z, Kushwaha P, Zoch ML, Tomlinson RE, Da H, Aja S, Noh HL, Kim JK, et al. (2017b). Sclerostin influences body composition by regulating catabolic and anabolic metabolism in adipocytes. Proc. Natl. Acad. Sci 114, E11238–E11247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kir S, White JP, Kleiner S, Kazak L, Cohen P, Baracos VE and Spiegelman BM (2014). Tumour-derived PTH-related protein triggers adipose tissue browning and cancer cachexia. Nature 513, 100–104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koduru SV, Sun B-H, Walker JM, Zhu M, Simpson C, Dhodapkar M, and Insogna KL (2018). The contribution of cross-talk between the cell-surface proteins CD36 and CD47-TSP-1 in osteoclast formation and function. J. Biol. Chem 293, 15055–15069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lampidonis AD, Rogdakis E, Voutsinas GE, Stravopodis DJ. (2011) The resurgence of Hormone-Sensitive Lipase (HSL) in mammalian lipolysis. Gene. 15;477(1–2):1–11. [DOI] [PubMed] [Google Scholar]
- Larsson S, Jones HA, Göransson O, Degerman E, and Holm C (2016). Parathyroid hormone induces adipocyte lipolysis via PKA-mediated phosphorylation of hormone-sensitive lipase. Cell. Signal 28, 204–213. [DOI] [PubMed] [Google Scholar]
- Lee S-Y, Dale Abel E, and Long F (2018). Glucose metabolism induced by Bmp signaling is essential for murine skeletal development. Nat. Commun 16: 9;(1) 4831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee NK, Karsenty G. (2008). Reciprocal regulation of bone and energy metabolism. Trends Endocrinol Metab. 19(5):161–6. [DOI] [PubMed] [Google Scholar]
- Li Y, Xu S, Zhang X, Yi Z, and Cichello S (2015). Skeletal intramyocellular lipid metabolism and insulin resistance. Biophys. Reports 1, 90–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Liefde II, van der Klift M, de Laet CEDH, van Daele PLA, Hofman A, and Pols HAP (2005). Bone mineral density and fracture risk in type-2 diabetes mellitus: the Rotterdam Study. Osteoporos. Int 16, 1713–1720. [DOI] [PubMed] [Google Scholar]
- Limonard EJ, Veldhuis-Vlug AG, Van DL, Runge JH, Tanck MW, Endert E, Heijboer AC, Fliers E, Hollak CE, Akkerman EM, et al. (2015). Short-Term Effect of Estrogen on Human Bone Marrow Fat. J.Bone Miner.Res 30, 2058–2066. [DOI] [PubMed] [Google Scholar]
- Liu F, Fang F, Yuan H et al. (2013). Suppression of autophagy by FIP200 deletion leads to osteopenia in mice through the inhibition of osteoblast terminal differentiation. J Bone Miner Res. 28(11):2414–2430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu JM, Rosen CJ, Ducy P, Kousteni S, Karsenty G. (2016). Regulation of Glucose Handling by the Skeleton: Insights From Mouse and Human Studies. Diabetes. November;65(11):3225–3232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Z, Xu J, He J, Liu H, Lin P, Wan X, Navone NM, Tong Q, Kwak LW, Orlowski RZ, et al. (2015). Mature adipocytes in bone marrow protect myeloma cells against chemotherapy through autophagy activation. Oncotarget 6, 34329–34341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maeda T, Matsunuma A, Kawane T, and Horiuchi N (2001). Simvastatin Promotes Osteoblast Differentiation and Mineralization in MC3T3-E1 Cells. Biochem. Biophys. Res. Commun 280, 874–877. [DOI] [PubMed] [Google Scholar]
- Maeda T, Matsunuma A, Kurahashi I, Yanagawa T, Yoshida H, and Horiuchi N (2004). Induction of osteoblast differentiation indices by statins in MC3T3-E1 cells. J. Cell. Biochem 92, 458–471. [DOI] [PubMed] [Google Scholar]
- Makovey J, Chen JS, Hayward C, Williams FMK, and Sambrook PN (2009). Association between serum cholesterol and bone mineral density. Bone 44, 208–213. [DOI] [PubMed] [Google Scholar]
- Mandal CC (2015). High Cholesterol Deteriorates Bone Health: New Insights into Molecular Mechanisms. Front Endocrinol.(Lausanne) 6, October 23:165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maridas DE, Rendina-Ruedy E, Helderman RC, DeMambro VE, Brooks D, Guntur AR, Lanske B, Bouxsein ML, and Rosen CJ (2019). Progenitor recruitment and adipogenic lipolysis contribute to the anabolic actions of parathyroid hormone on the skeleton. FASEB J. 33, 2885–2898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin S, and Parton RG (2006). Lipid droplets: a unified view of a dynamic organelle. Nat.Rev.Mol.Cell Biol 7, 373–378. [DOI] [PubMed] [Google Scholar]
- Maruotti N, Corrado A, Neve A, and Cantatore FP (2012). Bisphosphonates: effects on osteoblast. Eur. J. Clin. Pharmacol 68, 1013–1018. [DOI] [PubMed] [Google Scholar]
- Maurel DB, Pallu S, Jaffre C, Fazzalari NL, Boisseau N, Uzbekov R, Benhamou CL, and Rochefort GY (2012). Osteocyte apoptosis and lipid infiltration as mechanisms of alcohol-induced bone loss. Alcohol Alcohol 47, 413–422. [DOI] [PubMed] [Google Scholar]
- McCabe LR. (2009). Switching Fat from the periphery to the bone marrow. Why in Type I Diabetes Mellitus. Expert Rev Endocrinol Metab. 4(3):203–207. [DOI] [PubMed] [Google Scholar]
- McGee-Lawrence ME, Carpio LR, Schulze RJ, Pierce JL, McNiven MA, Farr JN, Khosla S, Oursler MJ, and Westendorf JJ (2016). Hdac3 Deficiency Increases Marrow Adiposity and Induces Lipid Storage and Glucocorticoid Metabolism in Osteochondroprogenitor Cells. J.Bone Miner.Res 31, 116–128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michaletti A, Gioia M, Tarantino U, and Zolla L (2017). Effects of microgravity on osteoblast mitochondria: a proteomic and metabolomics profile. Sci. Rep 7, 15376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Motyl KJ, Guntur AR, Carvalho AL, Rosen CJ. (2017). Energy Metabolism of Bone. Toxicol Pathol:45:887–893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mooradian AD (2009). Dyslipidemia in type 2 diabetes mellitus. Nat. Rev. Endocrinol 5, 150–159. [DOI] [PubMed] [Google Scholar]
- Muoio DM, and Newgard CB (2008). Fatty acid oxidation and insulin action: when less is more. Diabetes 57, 1455–1456. [DOI] [PubMed] [Google Scholar]
- Murad HAS (2016). L-Carnitine, but not coenzyme Q10, enhances the anti-osteoporotic effect of atorvastatin in ovariectomized rats. J. Zhejiang Univ. Sci. B 17, 43–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson RJ, Demas GE. (1996) Seasonal changes in immune function. Q Rev Biol. 71(4):511–48. [DOI] [PubMed] [Google Scholar]
- Nicodemus KK, Folsom AR, and Iowa Women’s Health Study (2001). Type 1 and type 2 diabetes and incident hip fractures in postmenopausal women. Diabetes Care 24, 1192–1197. [DOI] [PubMed] [Google Scholar]
- Niccoli Asabella A, Altini C, Nappi AG, Lavelli V, Ferrari C, Marzullo A, Loiodice A, Rubini G. (2019). Sickle cell diseases: What can nuclear medicine offer? Hell J Nucl Med. 2019 Jan-Apr;22(1):2–3. [DOI] [PubMed] [Google Scholar]
- Niemeier A, Niedzielska D, Secer R, Schilling A, Merkel M, Enrich C, Rensen PC, and Heeren J (2008). Uptake of postprandial lipoproteins into bone in vivo: impact on osteoblast function. Bone 43, 230–237. [DOI] [PubMed] [Google Scholar]
- Niemeier A, Kassem M, Toedter K, Wendt D, Ruether W, Beisiegel U, Heeren J. (2005) Expression of LRP1 by human osteoblasts: a mechanism for the delivery of lipoproteins and vitamin K1 to bone. J Bone Miner Res. 20(2):283–93. [DOI] [PubMed] [Google Scholar]
- Nollet M, Santucci-Darmanin S, Breuil V et al. (2014). Autophagy in osteoblasts is involved in mineralization and bone homeostasis. Autophagy. 10(11):1965–1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oburoglu L, Romano M, Taylor N, Kinet S. (2016). Metabolic regulation of hematopoietic stem cell commitment and erythroid differentiation. Curr Opin Hematol. 23(3):198–205. [DOI] [PubMed] [Google Scholar]
- Okayasu M, Nakayachi M, Hayashida C, Ito J, Kaneda T, Masuhara M, Suda N, Sato T, and Hakeda Y (2012). Low-density Lipoprotein Receptor Deficiency Causes Impaired Osteoclastogenesis and Increased Bone Mass in Mice because of Defect in Osteoclastic Cell-Cell Fusion. J. Biol. Chem 287, 19229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Onal M, Piemontese M, Xiong J et al. (2013). Suppression of autophagy in osteocytes mimics skeletal aging. J Biol Chem. 288(24):17432–17440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papackova Z and Cahova M. (2015). Fatty Acid Signaling: The New Function of Intracellular Lipases. Int J Mol Sci. 16(2):3831–3855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parhami F, Tintut Y, Beamer WG, Gharavi N, Goodman W, and Demer LL (2001). Atherogenic High-Fat Diet Reduces Bone Mineralization in Mice. J. Bone Miner. Res 16, 182–188. [DOI] [PubMed] [Google Scholar]
- Pelton K, Krieder J, Joiner D, Freeman MR, Goldstein SA, and Solomon KR (2012). Hypercholesterolemia promotes an osteoporotic phenotype. Am. J. Pathol 181, 928–936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piepkorn B, Kann P, Forst T, Andreas J, Pfützner A, and Beyer J (1997). Bone Mineral Density and Bone Metabolism in Diabetes Mellitus. Horm. Metab. Res 29, 584–591. [DOI] [PubMed] [Google Scholar]
- Prats-Puig A, Puig T, Sitjar C, Mas-Parareda M, Grau-Cabrera P, Soriano-Rodríguez P, Montesinos-Costa M, Diaz M, de Zegher F, Ibáñez L, et al. (2012). Soluble fatty acid synthase relates to bone biomarkers in prepubertal children. Osteoporos. Int 23, 2053–2058. [DOI] [PubMed] [Google Scholar]
- Prisby R, Guignandon A, Vanden-Bossche A, Mac-Way F, Linossier MT, Thomas M, Laroche N, Malaval L, Langer M, Peter ZA, Peyrin F, Vico L, Lafage-Proust MH. (2011). Intermittent PTH(1–84) is osteoanabolic but not osteoangiogenic and relocates bone marrow blood vessels closer to bone-forming sites. J Bone Miner Res. 26(11):2583–96. [DOI] [PubMed] [Google Scholar]
- Ramasamy I (2014). Recent advances in physiological lipoprotein metabolism. Clin Chem Lab Med. 52(12):1695–727. [DOI] [PubMed] [Google Scholar]
- Reagan M, Falank C, Fairfield H, McDonald M, Croucher P, and Rosen CJ (2016). Multiple Myeloma Progression: Dependence on Bone Marrow Adipose Tissue. Blood 128. [Google Scholar]
- Reiner Z, Guardamagna O, Nair D, Soran H, Hovingh K, Bertolini S, Jones S, Coric M, Calandra S, Hamilton J, et al. Lysosomal acid lipase deficiency--an under-recognized cause of dyslipidaemia and liver dysfunction. Atherosclerosis 235, 21–30. [DOI] [PubMed] [Google Scholar]
- Rendina-Ruedy E, and Rosen CJ (2017). Bone–Fat Interaction. Endocrinol. Metab. Clin. North Am 46, 41–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rendina-Ruedy E, Hembree KD, Sasaki A, Davis MR, Lightfoot SA, Clarke SL, Lucas EA, and Smith BJ (2015). A Comparative Study of the Metabolic and Skeletal Response of C57BL/6J and C57BL/6N Mice in a Diet-Induced Model of Type 2 Diabetes. J.Nutr.Metab 2015, 758080 Epub: Jun 3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rendina-Ruedy E, Guntur AR, and Rosen CJ (2017). Intracellular Lipid Droplets Support Osteoblast Function. Adipocyte. 6(3): 250–258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rendina-Ruedy E, Duta-Mare M, Kratky D, and Rosen CJ (2018). Lysosomal Acid Lipase and Its Role in Osteoblast Differentiation. ASMBR Annual Meeting Abstrac: M-0288. [Google Scholar]
- Richard J, and Lingvay I (2011). Hepatic steatosis and Type 2 diabetes: current and future treatment considerations. Expert Rev. Cardiovasc. Ther 9, 321–328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rishaug U, Birkeland KI, Falch JA, and Vaaler S (1995). Bone mass in non-insulin-dependent diabetes mellitus. Scand. J. Clin. Lab. Invest 55, 257–262. [DOI] [PubMed] [Google Scholar]
- Riddle RC and Clemens TR. (2017). Bone Cell Bioenergetics and Skeletal Homeostasis. Physiol Reviews 97(2): 667–698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russell RGG (2007). Bisphosphonates: Mode of Action and Pharmacology. Pediatrics 119, S150–S162. [DOI] [PubMed] [Google Scholar]
- Scheller EL, Doucette CR, Learman BS, Cawthorn WP, Khandaker S, Schell B, Wu B, Ding SY, Bredella MA, Fazeli PK, et al. (2015). Region-specific variation in the properties of skeletal adipocytes reveals regulated and constitutive marrow adipose tissues. Nat.Commun 6:7808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scheller E, Khandaker S, Learman BS, Cawthorn WP, Anderson LM, Pham HA, Robles H, Wang Z, Li Z, Parlee SD, et al. (2019). Bone marrow adipocytes resist lipolysis and remodelig in response to B-adrenergic stimulation. Bone. 118:32–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz AV (2011). Association of BMD and FRAX Score With Risk of Fracture in Older Adults With Type 2 Diabetes. JAMA 305, 2184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz AV, Sellmeyer DE, Ensrud KE, Cauley JA, Tabor HK, Schreiner PJ, Jamal SA, Black DM, and Cummings SR (2001). Older Women with Diabetes Have an Increased Risk of Fracture: A Prospective Study. J. Clin. Endocrinol. Metab 86, 32–38. [DOI] [PubMed] [Google Scholar]
- Sebo ZL, Rendina-Ruedy E, Ables GP, Lindskog DM, Rodeheffer MS, Fazeli PK, and Horowitz MC (2019). Bone Marrow Adiposity: Basic and Clinical Implications. Endocr. Rev Epub ahead of publication April 25: 00138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimizu E, Tamasi J, and Partridge NC (2012). Alendronate affects osteoblast functions by crosstalk through EphrinB1-EphB. J. Dent. Res 91, 268–274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silberstein L, Goncalves KA, Kharchenko PV, Turcotte R, Kfoury Y, Mercier F, Baryawno N, Severe N, Bachand J, Spencer JA, Papazian A, Lee D, Chitteti BR, Srour EF, Hoggatt J, Tate T, Lo Celso C, Ono N, Nutt S, Heino J, Sipilä K, Shioda T, Osawa M, Lin CP, Hu GF, Scadden DT. Proximity-Based Differential Single-Cell Analysis of the Niche to Identify Stem/Progenitor Cell Regulators Cell Stem Cell. 2016. October 6;19(4):530–543 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simpson EJ, Debevec T, Eiken O, Mekjavic I, and Macdonald IA (2016). PlanHab: the combined and separate effects of 16 days of bed rest and normobaric hypoxic confinement on circulating lipids and indices of insulin sensitivity in healthy men. J. Appl. Physiol 120, 947–955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Staines KA, Zhu D, Farquharson C, and MacRae VE (2014). Identification of novel regulators of osteoblast matrix mineralization by time series transcriptional profiling. J. Bone Miner. Metab 32, 240–251. [DOI] [PubMed] [Google Scholar]
- Stolk RP, Van Daele PL, Pols HA, Burger H, Hofman A, Birkenhäger JC, Lamberts SW, and Grobbee DE (1996). Hyperinsulinemia and bone mineral density in an elderly population: The Rotterdam Study. Bone 18, 545–549. [DOI] [PubMed] [Google Scholar]
- Tang Y, Wu X, Lei W, Panh L, Wan C, Shi Z, Zhao L, Nagy TR, Peng X, Hu J, Feng X, Van Hul W, Wan M, Cao X. (2009). TGF-beta1-induced migration of bone mesenchymal stem cells couples bone resorption with formation. Nat Med 15(7):757–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabe Y, Yamamoto S, Saitoh K, Sekihara K, Monma N, Ikeo K, Mogushi K, Shikami M, Ruvolo V, Ishizawa J, et al. (2017). Bone Marrow Adipocytes Facilitate Fatty Acid Oxidation Activating AMPK and a Transcriptional Network Supporting Survival of Acute Monocytic Leukemia Cells. Cancer Res. 77, 1453–1464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanida I, Ueno T, Kominami E. (2004). LC3 conjugation system in mammalian autophagy. Int J Biochem Cell Biol. 36(12):2503–2518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tencerova M, Figeac F, Ditzel N, Taipaleenmäki H, Nielsen TK, and Kassem M (2018). High-Fat Diet-Induced Obesity Promotes Expansion of Bone Marrow Adipose Tissue and Impairs Skeletal Stem Cell Functions in Mice. J Bone Miner Res 33, 1154–1165. [DOI] [PubMed] [Google Scholar]
- Tirado-Vélez JM, Joumady I, Sáez-Benito A, Cózar-Castellano I, and Perdomo G (2012). Inhibition of fatty acid metabolism reduces human myeloma cells proliferation. PLoS One 7, e46484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trotter TN, Gibson JT, Sherpa TL, Gowda PS, Peker D, and Yang Y (2016). Adipocyte-Lineage Cells Support Growth and Dissemination of Multiple Myeloma in Bone. Am. J. Pathol 186, 3054–3063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tylki-Szymanska A, and Jurecka A (2014). Lysosomal acid lipase deficiency: wolman disease and cholesteryl ester storage disease. Pril.(Makedon.Akad.Nauk Umet.Odd.Med.Nauki) 35, 99–106. [PubMed] [Google Scholar]
- Wang Y, Li Y, Mao K, Li J, Cui Q, and Wang GJ (2003). Alcohol-induced adipogenesis in bone and marrow: a possible mechanism for osteonecrosis. Clin.Orthop.Relat Res 213–224. [DOI] [PubMed] [Google Scholar]
- Wei J, Shimazu J, Makinistoglu MP, Maurizi A, Kajimura D, Zong H, Takarada T, Iezaki T, Pessin JE, Hinoi E, et al. (2015). Glucose Uptake and Runx2 Synergize to Orchestrate Osteoblast Differentiation and Bone Formation. Cell 161, 1576–1591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang L, Guan G, Lei L, Lv Q, Liu S, Zhan X, Jiang Z, and Gu X (2018). Palmitic acid induces human osteoblast-like Saos-2 cell apoptosis via endoplasmic reticulum stress and autophagy. Cell Stress Chaperones 23, 1283–1294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yeh L-CC, Ford JJ, Lee JC, and Adamo ML (2014). Palmitate attenuates osteoblast differentiation of fetal rat calvarial cells. Biochem. Biophys. Res. Commun 450, 777–781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yerges-Armstrong LM, Shen H, Ryan KA, Streeten EA, Shuldiner AR, and Mitchell BD (2013). Decreased bone mineral density in subjects carrying familial defective apolipoprotein B-100. J.Clin.Endocrinol.Metab 98, E1999–E2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoneshima E, Okamoto K, Sakai E, Nishishita K, Yoshida N, Tsukuba T. (2016). The Transcription Factor EB (TFEB) Regulates Osteoblast Differentiation Through ATF4/CHOP-Dependent Pathway. J Cell Physiol. 231(6):1321–1333. [DOI] [PubMed] [Google Scholar]
- Yuzefovych L, Wilson G, and Rachek L (2010). Different effects of oleate vs. palmitate on mitochondrial function, apoptosis, and insulin signaling in L6 skeletal muscle cells: role of oxidative stress. Am. J. Physiol. Metab 299, E1096–E1105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang N, Zhang Y, Lin J, Qiu X, Chen L, Pan X, Lu Y, Zhang J, Wang Y, Li D, et al. (2017). Low-density lipoprotein receptor deficiency impaired mice osteoblastogenesis Biosci. Trends 11, 658–666. [DOI] [PubMed] [Google Scholar]
- Zhao H, Li X, Zhang D, Chen H, Chao Y, Wu K, Dong X, and Su J (2018). Integrative Bone Metabolomics—Lipidomics Strategy for Pathological Mechanism of Postmenopausal Osteoporosis Mouse Model. Sci. Rep 8, Nove 7 (8); 16456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng J, Brion M-J, Kemp JP, Warrington NM, Borges M-C, Hemani G, Richardson T, Qiao Z, Haycock P, Ala-Korpela M, et al. (2018). The effect of plasma lipids and lipid lowering interventions on bone mineral density: a Mendelian randomization study. [DOI] [PubMed] [Google Scholar]
- Zhou K, Yao P, He J, Zhao H. (2019). Lipophagy in nonliver tissues and some related diseases: Pathogenic and therapeutic implications. J Cell Physiol. 234(6):7938–7947. [DOI] [PubMed] [Google Scholar]