Abstract
The development of lateral roots in Arabidopsis thaliana is strongly dependent on signaling directed by the AUXIN RESPONSE FACTOR7 (ARF7), which in turn activates LATERAL ORGAN BOUNDARIES DOMAIN (LBD) transcription factors (LBD16, LBD18 and LBD29). Here, the product of PRH1, a PR-1 homolog annotated previously as encoding a pathogen-responsive protein, was identified as a target of ARF7-mediated auxin signaling and also as participating in the development of lateral roots. PRH1 was shown to be strongly induced by auxin treatment, and plants lacking a functional copy of PRH1 formed fewer lateral roots. The transcription of PRH1 was controlled by the binding of both ARF7 and LBDs to its promoter region.
Author summary
In Arabidopsis thaliana AUXIN RESPONSE FACTOR7 (ARF7)-mediated auxin signaling plays a key role in lateral roots (LRs) development. The LATERAL ORGAN BOUNDARIES DOMAIN (LBD) transcription factors (LBD16, LBD18 and LBD29) act downstream of ARF7-mediated auxin signaling to control LRs formation. Here, the PR-1 homolog PRH1 was identified as a novel target of both ARF7 and LBDs (especially the LBD29) during auxin induced LRs formation, as both ARF7 and LBDs were able to bind to the PRH1 promoter. This study provides new insights about how auxin regulates lateral root development.
Introduction
The architecture of the root system depends on the density of lateral roots (LRs) formed along with the extent of root branching. LRs are initiated from mature pericycle cells lying adjacent to the xylem pole, referred to in Arabidopsis thaliana as the xylem pole pericycle [1, 2]. A subset of these cells, namely the founder cells, undergo a series of highly organized divisions to form an LR primordium, which eventually develops into an LR. The entire process of LR development, from its initiation to its emergence, is regulated by auxin [1, 3, 4]. The accumulation of auxin in protoxylem cells results in the priming of neighboring pericycle cells to gain founder cell identity, forming an LR pre-branch site [5]. This auxin signaling is mediated by the proteins ARF7 and ARF19, which act in the IAA14/28-dependent pathway [6, 7]. LR initiation depends on the transcriptional activation, through the involvement of the transcription factor LBD16 and LBD18, of either E2Fa or CDKA1;1 and CYCB1;1 [8, 9]. Other downstream targets such as EXP14 and EXP17 and the products of certain cell wall loosening-related genes are also either directly or indirectly regulated by LBD18 to promote the emergence of an LR [10–12]. LBD18 has been also found to control LR formation through its interaction with GIP1, which results in the transcriptional activation of EXP14 [13].
LBD transcription factors are among the most well studied downstream targets of ARF7 and ARF19 [14]. ARF7 has also been found to control LR emergence through its regulation of IDA and genes which encode leucine-rich repeat receptor-like kinases such as HAE and HSL2 [15]. These interactions take place in the overlaying tissues, thereby influencing cell wall remodeling and cell wall degradation during LR emergence [15].
Here, the product of PRH1 (At2g19990), a homolog of PR-1 which has been annotated as a pathogen responsive protein [16], was identified as a novel target of ARF7-LBDs mediated auxin signaling. We provided evidence that PRH1 acted downstream of ARF7 and LBDs to control auxin-regulated LR formation.
Results
PRH1 is a target of ARF7-mediated auxin signaling
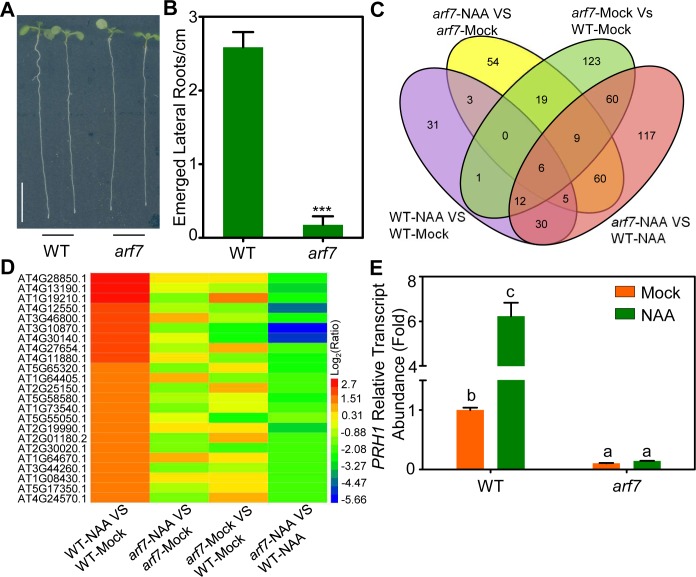
To identify additional targets involved in ARF7-mediated auxin signaling and LR development (Fig 1A and 1B), a comparison was made between the 8-day-old wild type (WT) or arf7 mutant seedlings exposed to 10 μM naphthalene acetic acid (NAA) for 4 hours respectively (Figs 1C and S1A). Based on differential comparisons (WT and arf7 with or without NAA treatment) all of the detected genes were presented in S1 Table as the raw data and the genes changed at least 2-fold (P < 0.0001, log2fold change >1) were selected as differentially expressed genes (DEGs) shown in S2 Table. The DEGs were selected based on the comparisons (WT-NAA VS WT-Mock, arf7-NAA VS arf7-Mock, arf7-Mock VS WT-Mock, arf7-NAA VS WT-NAA), which were induced by auxin and regulated by ARF7. To narrow down the selection of candidate genes, we further adjusted the screening criteria (P < 0.0001, log2fold change >1.5 in WT-NAA VS WT-Mock), which reduces the DEGs to 23 (Fig 1D and S1B Fig). To further explore whether these genes are involved in LR development, we then examined the expression patterns of these 23 candidate genes in LR initiation using the Arabidopsis eFP Browser website (http://bar.utoronto.ca/efp/cgi-bin/efpWeb.cgi). After this analysis, eight of the candidate DEGs were expressed in the process of LR initiation induction, and then we focused on the PRH1 (At2g19990), which displayed the highest induction by auxin while the induction was absent in arf7, for further investigations (Fig 1E, S1B and S1C Fig).
Fig 1. PRH1 is a downstream target of ARF7-mediated auxin signaling.
(A) Eight-day-old seedlings of WT and arf7. Scale bar: 1 cm. (B) The ratio between the number of the emerged lateral roots and the length of the primary root of WT and arf7 seedlings. Data shown as mean±SE, three biological replicates in the experiment, 20 plant seedlings for each repeat. ***: the mutant’s performance differs significantly (P<0.001) from that of WT. (C) Venn diagram indicating the overlap of differentially expressed genens between the four datasets (Col-NAA VS Col-Mock, arf7-NAA VS arf7-Mock, arf7-Mock VS Col-Mock and arf7-NAA VS Col-NAA) based on RNA-seq. RNA-seq analysis: RNA was extracted from the primary roots of eight-day-old seedlings exposed or not exposed to 10 μM NAA for 4 hours. NAA: naphthalene acetic acid. (D) Heat map of the differentially expressed genes induced by auxin and regulated by ARF7 according to RNA-seq data. (E) The relative transcript abundance of PRH1 in the roots of eight-day-old WT and arf7 seedlings exposed (green) or not exposed (orange) to 10 μM NAA for 4 hours. The relative transcript abundance is relative to the untreated WT. Values represent averages of three biological replicates in the experiment, and the total mRNA was extracted from about 100 seedlings for each repeat. Error bars represent SE. Different letters atop the columns indicate significant (P<0.001) differences in abundance.
PRH1, a homolog of PR-1 has been annotated as encoding a pathogen-responsive protein and was also named as PR-1-LIKE [16]. There are 11 members in the family in Arabidopsis, and most of them are defense-related genes (S2A Fig) [17–19]. Expression of PRH1 is not salicylic acid responsive and the biochemical function is unknown [16]. Considering that PRH1 was induced by auxin (Fig 1E), the auxin induced GH3.5 was used as a positive control and the PRH1 expression was examined through time-coursed treatment with auxin (treated with 10 μM naphthalene acetic acid, NAA). The result showed that PRH1 is transcriptionally regulated by auxin and there was a significant induction after 2 hours of auxin treatment (S2B Fig). The GUS expression analysis in 8-day-old seedlings carrying a PRH1pro:GUS transgene implied that PRH1pro:GUS is expressed in the cells overlying/surrounding the lateral root primordium (LRP), especially at endodermis or cortex cells overlying the primordium in WT background (Fig 2A; S2C Fig), and the level of GUS activity was greatly enhanced following exposure of the plants to auxin (Fig 2B). When the PRH1pro:GUS transgene was expressed in the arf7 mutant background, the PRH1pro:GUS expression was strongly suppressed, and auxin treatment only slightly induced the expression of PRH1pro:GUS (Fig 2C and 2D). Consistently, PRH1 expression was completely non-responsive to auxin treatment in arf7 according to our qPCR assays (Fig 1E). The conclusion was that the auxin-responsive PRH1 is mainly expressed in the cells overlying/surrounding the LRP and its expression is dependent on ARF7-mediated auxin signaling.
Fig 2. Histological localization and expression of PRH1pro:GUS transgene during LR formation.
(A) and (B) PRH1 is expressed in the cells overlying/surrounding the lateral root primordium(LRP) in WT seedlings not exposed to auxin(A), and the intensity of its expression is strongly enhanced when the plants were treated with 10 μM naphthalene acetic acid (NAA) for 4 hours (B). (C) and (D) GUS activity is suppressed in non-treated arf7 seedlings (C), and even more so in treated ones (D). NAA: naphthalene acetic acid. Before GUS staining the seedlings were soaked for 4 hours in liquid half strength Murashige and Skoog (MS) medium, which containing 10 μM NAA or not. The developmental stages (I through VII and E) of the LRs are indicated in the top left corner. GUS (β-glucuronidase) signals are shown in blue. Stages I to VII of primordia were based on the classification by Malamy and Benfey [1], E: Emerged lateral roots, Scale bars: 50 μm.
PRH1 acts downstream of ARF7 to regulate LR development
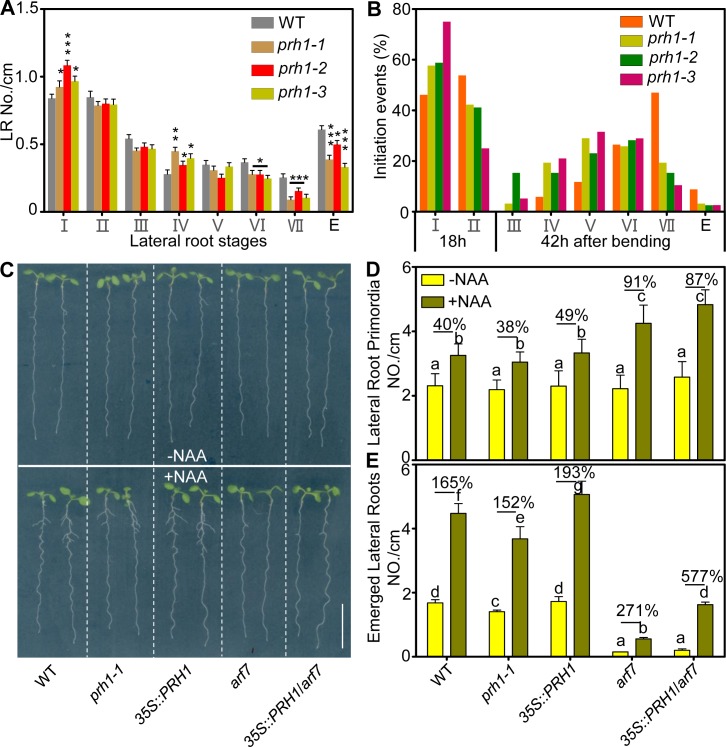
To address if auxin-induced PRH1 expression is involved into LR development, we examined the LR phenotypes in both prh1 mutants and the PRH1 over-expression lines (Fig 3A and 3B; S3B Fig). The third allele of prh1 (phr1-3) generated via Crispr-cas9 system [20] was also used for the LR phenotype analysis (S3A Fig). The primordia numbers of 8-day-old seedlings at various stages were counted of all these three prh1 mutant alleles. The results showed that primordium development was affected. The numbers at stage I and Ⅳ were increased and at the later periods (stage Ⅵ, Ⅶ and emerged) were reduced (Fig 3A). Examination of LR development in prh1 mutants showed that the absence of a functional copy of PRH1 inhibited the formation of emerged LRs (Fig 3E). The expression level of PRH1 in its T-DNA insertion mutants and the over-expression lines were all verified (S3C Fig). The weak phenotype of prh1-2 in the emerged stage might be result from the down-regulation of PRH1 in this allele (Fig 3A; S3C Fig). Moreover the PRH1pro:PRH1 construct was transformed in the prh1-1 mutant, the expression of PRH1 could complement the LR defects in prh1-1 (S4 Fig). Though LR development was not greatly affected by the over-expression of PRH1 in a WT background (S3B Fig), over-expression of PRH1 could significantly increase the LR numbers of arf7 mutant plants in the presence of exogenous NAA treatment (Fig 3C–3E).
Fig 3. PRH1 acts downstream of ARF7-mediated lateral root (LR) development.
(A) Density of primordia at given stages. Stages I to VII of primordia were based on the classification by Malamy and Benfey [1], E: emerged lateral roots, *,**, ***: means differ significantly (P<0.05, P<0.01, P<0.001) from the WT control. (B) The synchronized initiation of lateral root primordia were induced using gravitropic stimulation at the site of root bending in the 3-day-old seedlings of WT, prh1-1, prh1-2 and prh1-3. Phenotypic analysis of lateral root (LR) emergence was achieved after 18 h and 42 h gravistimulus compared with WT (Col-0) and the LR emergence is delayed in prh1 mutants. Lateral root primordium stages (from I to VII according to previous descriptions from 1997 Benfey [1]) were analyzed from 30 seedlings. (C) Eight-day-old seedlings of WT, arf7, prh1-1 mutants and transgenic lines harboring either 35S::PRH1 or 35S::PRH1/arf7. The upper panel shows the seedlings grown on 1/2 MS medium without NAA addition and the lower panel shows the seedlings grown on 1/2 MS medium containing 30 nM NAA. Bar: 1 cm. (D and E) Density of lateral root primordia (D) and emerged lateral roots (E) of the 8-d-old seedlings grown on the half strength Murashige and Skoog (MS) medium containing 30 nM NAA or not. The percentage values represent the increase with NAA treatment. The density was the ratio between lateral roots number and the length of primary root. NAA: naphthalene acetic acid. Different letters atop the columns indicate significant (P<0.05) differences in abundance. Data shown as means±SE, three biological replicates in the experiment, 20 plant seedlings for each repeat.
To further confirm the role of PRH1 in LR formation, we did LR analysis of prh1 mutants through a gravistimulation-based bioassay [21–23]. After synchronizing LR formation with a gravistimulus for 18 h, we observed a delayed transition from stage I to stage II of LR development which was shown by the increased LR I (lateral root stage I) ratio and decreased LR II (lateral root stage II) ratio in prh1 mutants (Fig 3B). Then we examined the phenotype of mutant seedlings after a gravistimulus for 42 h, and the lateral root initiation rates reduced both in stage VII and emerged ones, but increased in the earlier stages (stage IV or V) (Fig 3B). In summary, PRH1 acts downstream of ARF7 to regulate LR development.
ARF7 directly regulates PRH1 transcription
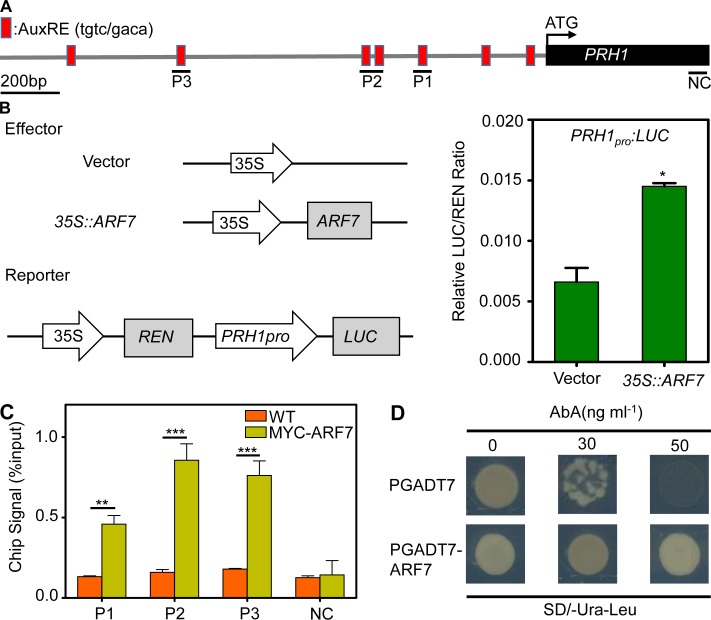
The observation that the abundance of PRH1 transcript was reduced in the arf7 mutant (Fig 1D) suggested that the gene was transcriptionally regulated by the auxin response factor ARF7. The gene’s promoter sequence harbors the known auxin response elements (AuxREs) TGTC (Fig 4A). A transient expression assay carried out in A. thaliana leaf protoplasts showed that ARF7 was able to activate a construct comprising the PRH1 promoter fused with LUC. The co-expression of PRH1pro:LUC and 35S::ARF7 had a large positive effect on the intensity of the luminescence signal (Fig 4B), consistent with the idea that ARF7 is able to activate the PRH1pro:LUC construct. A ChIP-qPCR assay confirmed the association of ARF7 with the AuxRE elements present on the PRH1 promoter (Fig 4A and 4C), and a yeast one hybrid assay showed that ARF7 was able to bind to the PRH1 promoter (Fig 4D). The conclusion was that the effect of ARF7 binding to the PRH1 promoter was regulating the gene’s transcription, thereby influencing LR development.
Fig 4. PRH1 is regulated by ARF7 at the transcriptional level.
(A) Structure of PRH1 promoter and the fragments used in the CHIP-qPCR assay. AuxREs are indicated by red squares, and black lines show the promoter regions containing the AuxREs used in this assay. NC: negative control. AuxREs: auxin response elements. (B) ARF7 transactivates the PRH1 promoter in A. thaliana leaf protoplasts. The left hand panel is a schematic of the effector (35S::ARF7) and reporter (PRH1pro:LUC) constructs. The empty vector pBI221 was used as a negative control; the right hand panel shows the ratio of ARF7 drived LUC and the empty vector (negative control) to 35S promoter drived REN respectively. LUC: firefly luciferase activity, REN: renilla luciferase activity. Values shown as means±SE, three biological replicates in the experiment. *: means differ significantly (P<0.05) from the negative control. (C) ARF7 is associated with the PRH1 promoter according to a CHIP-qPCR assay. Chromatin isolated from a plant harboring 35S::MYC-ARF7 and a WT mock control was immunoprecipitated with anti-MYC antibody following the amplification of regions P1, P2 and P3. The coding region segment NC was used as the negative control. The ChIP signal represents the ratio of bound promoter fragments (P1-P3) after immunoprecipitation to total input without immunoprecipitation. Values shown as means±SE, three biological replicates in the experiment. **, ***: means differ significantly (P<0.01, P<0.001) from the WT control. (D) Physical interaction of ARF7 with the PRH1 promoter according to a Y1H assay. The plasmid pGADT7-ARF7 was introduced into Y1H Gold cells harboring the reporter gene PRH1pro:AbAr and the cells were grown on SD/-Ura-Leu medium in the presence of 30 or 50 ng/mL aureobasidin A (AbA). The empty vector pGADT7 was used as a negative control.
Auxin-induced PRH1 expression is regulated by LBD29
Members of the LBD family are known to function downstream of ARF7 to control LR initiation and emergence [14, 24–27]. When the transcription level of PRH1 was investigated in the three single lbd mutants lbd16 (Salk_040739), lbd18 (Salk_038125) and lbd29 (Salk_071133) combined with or without exogenous auxin treatment, the observation was that the gene was strongly repressed in the single mutants. Though the auxin induced PRH1 expression was normal in lbd16 or lbd18, the induction was strongly reduced in lbd29, indicating LBD29 plays an important role in auxin induced PRH1 expression (S5A Fig). Consistently, the over-expression of LBD29 strongly up-regulates PRH1 expression (S5B Fig). In addition, we also observed the up-regulation of PRH1 expression in LBD16, LBD18 or LBD29 over-expression lines (S5B Fig). These results suggest that LBDs regulate the transcription of PRH1, auxin induced expression of PRH1 is dependent on LBD29.
Since the auxin influx carrier LIKE-AUXIN3 (LAX3) has been reported to act downstream of ARF7-LBD29 auxin signaling module to facilitate LR emergence [27, 28], we also investigated the relationship between LAX3 and PRH1 through qPCR analysis. The results showed that the absence of PRH1 did not affect the expression of LAX3 with or without auxin treatment (S5C Fig). However, in lax3 which has defects in auxin influx, the auxin-induced PRH1 expression was reduced (S5D Fig).
LBDs regulate PRH1 by binding to its promoter
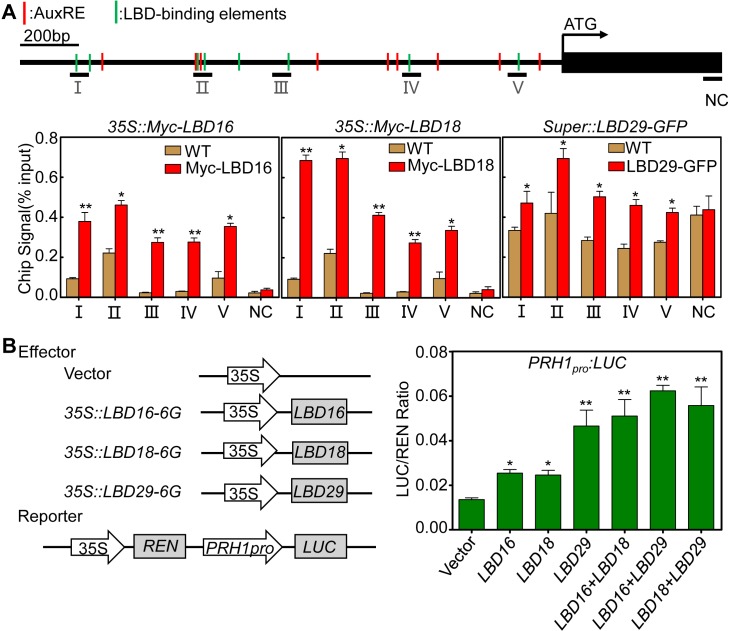
The transcriptional response of PRH1 to the loss-of-function and over-expression of certain LBDs implied that these transcription factors can also regulate PRH1. The PRH1 promoter has the typical LBD protein binding elements including CGGCG and CACGTG (S6A Fig). Although the yeast one hybrid assay produced no direct evidence for the binding of LBD proteins to the PRH1 promoter (S6B Fig), but the ChIP-qPCR assay did support the association of LBD16, LBD18 and LBD29 with the PRH1 promoter (Fig 5A). Thus a transient expression assay in A. thaliana leaf protoplasts was used to investigate whether LBD16, LBD18 and/or LBD29 was able to influence the expression of the PRH1pro:LUC transgene. The outcome of co-expressing this construct with each of 35S::LBD16, 35S::LBD18 or 35S::LBD29 was to enhance the expression of PRH1pro:LUC (Fig 5B). When a pair of effector transgenes (either 35S::LBD16 plus 35S::LBD18, 35S::LBD18 plus 35S::LBD29 or 35S::LBD16 plus 35S::LBD29) was introduced, the PRH1pro:LUC expression was even more strongly activated (Fig 5B). The overall conclusion was that the LBDs regulate PRH1 expression by directly binding to its promoter.
Fig 5. The transcription of PRH1 is activated by LBD.
(A) LBDs associated with the promoter of PRH1 according to a CHIP-qPCR assay. The upper panel shows the structure of the PRH1 promoter, and five uniformly distributed sites (black lines) were selected for the CHIP-qPCR assay. Sites II and III include the LBD29 and LBD18 binding motif (See S6 Fig). NC: negative control. The red lines indicate the AuxREs (auxin response elements) and the green lines indicate LBD-binding elements [12, 44]. The lower panel demonstrates the ratio of bound promoter fragments (Ⅰ-Ⅴ) after immunoprecipitation to total input without immunoprecipitation. The samples were derived from WT seedlings (negative control) and those harboring one of the transgenes 35S::Myc-LBD16 (Myc-LBD16), 35S::Myc-LBD18 (Myc-LBD18), Super::LBD29-GFP (LBD29-GFP) [44, 49]. Values shown as means±SE, three biological replicates in the experiment. * Represents a comparison of the signal arising from the Myc/GFP-fused LBD with WT in the corresponding genomic region. *, **: means differ significantly (P<0.05, P<0.01) from the WT control. (B) LBDs transactivate the PRH1 promoter in A. thaliana leaf protoplasts as shown by a transient dual-luciferase assay. The reporter gene (PRH1 promoter driving LUC) was co-transformed with one or two of the constructs 35S::LBD16, 35S::LBD18 and 35S::LBD29. The empty vector pBI221 was used as negative control. Values shown as means±SE, three biological replicates in the experiment. *, **: means differ significantly (P<0.05, P<0.01) from the WT control.
LBD regulated LR development is partially dependent on PRH1
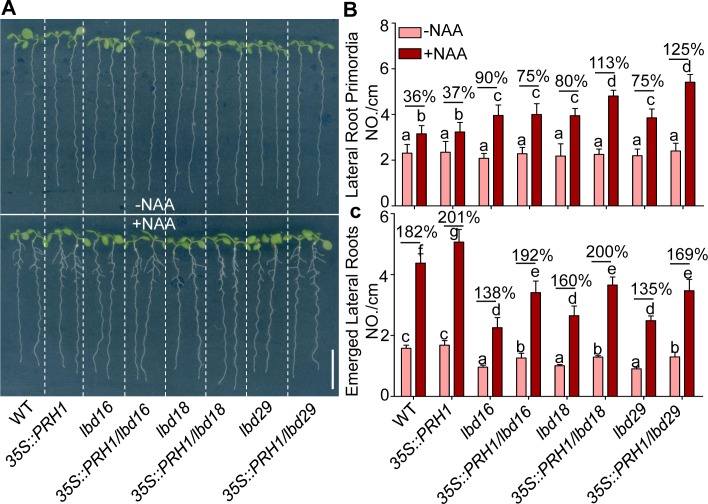
To further study if LBDs regulated PRH1 transcription is involved into LBD-mediated LR development, we crossed 35S::PRH1 with lbd16, lbd18 or lbd29 mutants and analyzed LR phenotypes of the generated double mutants. Compared to lbd16, lbd18, lbd29 single mutant plants, the generated 35S::PRH1/lbd16, 35S::PRH1 lbd18 or 35S::PRH1/lbd29 double mutants have increased LR numbers, especially in the presence of auxin treatment (Fig 6), indicating that PRH1 is involved into LBD-mediated LR development.
Fig 6. The over-expression of PRH1 partially alleviates the defective development of lateral roots (LRs) in the lbd mutant.
(A) Eight-day-old seedlings of WT, the mutants lbd16, lbd18 and lbd29, and the transgenic lines harboring 35S::PRH1. The upper panel shows the seedlings grown on 1/2 MS medium without NAA addition and the lower panel shows the seedlings grown on 1/2 MS medium containing 30 nM NAA. Scale bar: 1 cm. (B and C) Density of lateral root primordia (B) and emerged lateral roots (C) in eight-day-old seedlings grown on the 1/2 MS medium containing 30 nM NAA or not. The percentage values represent the increase with NAA treatment. The density was the ratio between lateral roots number and the length of primary root. NAA: naphthalene acetic acid. Data shown as means±SE, three biological replicates in the experiment, 20 plant seedlings for each repeat. Different letters atop the columns indicate significant (P<0.05) differences in abundance.
To determine the subcellular localization of PRH1, the cytosolic marker ADH-RFP [29] or plasma membrane maker ZmCRN-mCherry [30] were coexpressed with PRH1-GFP in N. benthamiana leaf cells (S7 Fig). The green signals from PRH1-GFP were merged with the red signals from ADH-RFP, indicating that PRH1 is localized in the cytosol. Given the LR phenotype of prh1 mutants and its expression pattern, the expression levels of some cell wall formation related genes were detected in prh1-1. We found some EXPANSIN genes, including EXP3 EXP8 EXP11 and EXP14 which were all involved in cell wall loosening [12, 31], were down-regulated in prh1 (S8 Fig). Therefore, PRH1 might regulate LR emergence by influencing the EXPANSIN gene expression.
According to this study together with the previous reports, a proposed model was given in Fig 7. PRH1 is involved in ARF7-LBD dependent auxin signaling pathway regulating lateral root development. PRH1 acts as a downstream gene of both ARF7 and LBDs by the direct transcriptional regulation and then affects the expression of some EXPANSIN genes. Meanwhile LAX3 acts downstream of LBD29 to control IAA accumulation in the epidermits and cortex [32], and thus influences auxin-induced PRH1 expression indirectly.
Fig 7. A working model of the regulation of LR development by PRH1.
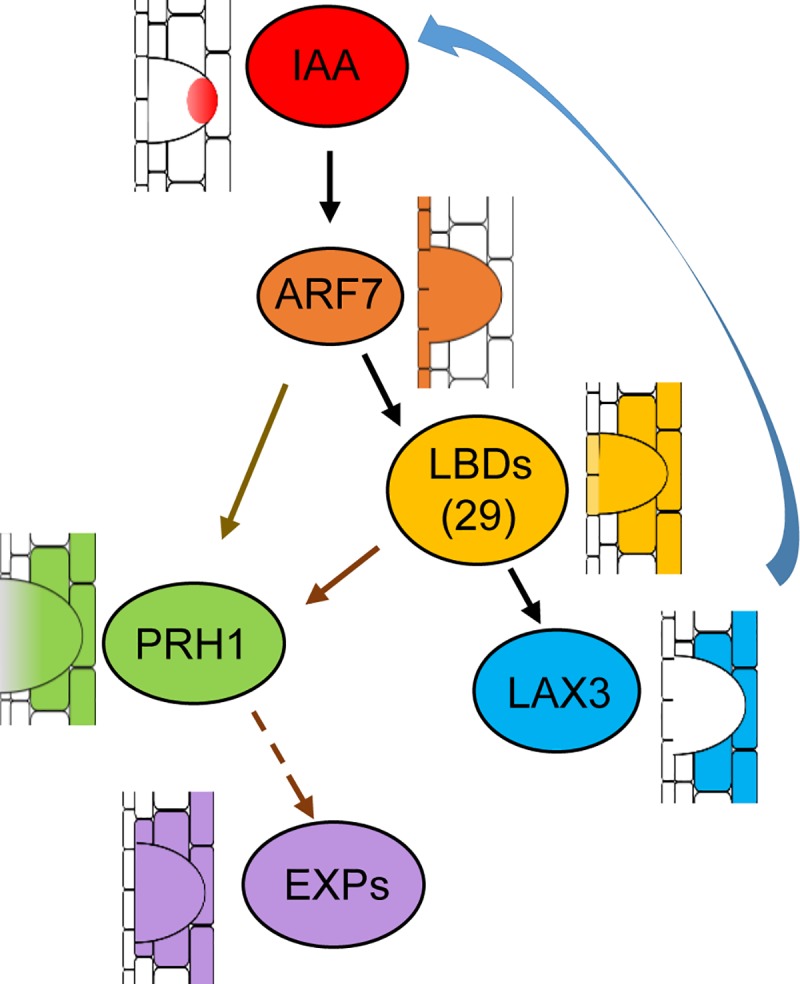
Auxin induced PRH1 expression is dependent on both ARF7 and LBDs. LAX3, which acts downstream of LBD29, also indirectly regulates PRH1 expression through controlling IAA accumulation in the epidermits and cortex. PRH1 may promote LR emergence by regulating the expression of EXPANSIN genes. ARF7 is expressed in pericycle and primordium [33]; LBD16 is expressed in the primordium at earlier stages [9]; LBD18 and LBD29 are expressed in the LR primordium and overlying tissues [27]; LAX3 is expressed in the epidermis and cortex cells above the primordia [27]. The histological localization of PRH1 is in the cells overlying/surrounding the lateral root primordium. PRH1 might regulate LR emergence through influencing the expression of EXPs that affecting cell wall loosening.
Discussion
The product of the auxin-induced gene PRH1 controls LR formation
The role of auxin in LR development has been extensively investigated. During the initiation of LR, auxin signals are transduced through the two distinct AUX/IAA-ARFs modules IAA14-ARF7/ARF19 and IAA12-ARF5 [6, 33–36]. This process establishes LR founder cell identity, which is the initial step towards the formation of a LR. Auxin is also involved in LR emergence, since it controls the activation of IDA, HAE and HSL2 which encode cell wall remodeling enzymes and of genes encoding various expansions in the endodermal cells which lie above the LR primordia [11, 15, 37]. Members of the LBD gene family encode transcription factors, characterized by their LOB domain [38], which act downstream of ARF7-mediated auxin signaling to control LR formation [13, 24–26].
Here, the PR-1 homolog gene PRH1 was identified as a novel target of ARF7-mediated auxin signaling. Otherwise we have compared our RNA-Seq data to the previous datasets of lateral root induction, recommend in Lewis et al [39] and Himanen et al [40]. Consistently, all these studies showed that the expression of PRH1 was induced by auxin and the induction depended on ARF7. Although the over-expression of PRH1 in a WT background was not associated with any LR phenotype, its over-expression in arf7, lbd16, 1bd18 or lbd29 mutant backgrounds led to a partial rescue of the compromised LR phenotype, especially under auxin treatment (Figs 3 and 6). The more striking rescued LR formation by overexpression of PRH1 in lbd mutants than that in arf7 suggests additional factors other than LBDs act downstream of ARF7. The PRH1 promoter directs its product primarily to the cells overlying/surrounding the lateral root primordium (as shown by the site of GUS expression in plants expressing a PRH1pro:GUS transgene), which is consistent with the proposed role of PRH1 in LR formation. Auxin treatment strongly induced the expression of the PRH1pro:GUS transgene in a WT (Fig 2A and 2B), but not in an arf7 background (Fig 2C and 2D), which implied that the auxin-induced expression of PRH1 must depend on the presence of a functional copy of ARF7. LBD proteins, especially LBD29, are also involved in the induction by auxin of PRH1. Experimentally, it was shown that both ARF7 and LBDs were able to bind to the PRH1 promoter (Figs 4 and 5). As some of the LBD-binding and AuxRE elements close to each other in PRH1 promoter (Fig 5A), it will be of future interest to investigate how ARF7 coordinates with LBDs to mediate auxin signaling and therefore to control the expression of PRH1. In addition, the auxin influx carrier LIKE-AUXIN3 (LAX3) which acts downstream of ARF7-LBD29 auxin signaling module to control LR emergence [27, 28], might be involved into auxin-induced PRH1 expression through the regulation of local auxin transport and local auxin accumulations.
The homo- and heterodimerization of LBD proteins are thought to be an important determinant of LR formation [26, 41]. The AtbZIP59 transcription factor has been reported to form complexes with LBDs to direct auxin-induced callus formation and LR development [42]. All these results suggest that homodimerization and heterodimerization of signaling components are important in LR formation. Here, it was shown that PRH1 was mainly expressed in the cells overlying the primordium (Fig 2) and there was a LR emergence defect in the prh1 mutants. Several of the EXPANSIN genes were down-regulated in prh1-1(S8 Fig), indicating that PRH1 might regulate LR emergence through influencing cell wall loosening. Whether PRH1 regulates LR formation through a direct interaction with LBDs, AtZIP59 and other factors which have been shown to control LR development, is still an open question for the future.
Materials and methods
Plant materials and growing conditions
A. thaliana ecotype Col-0 was used as the wild type (WT), and all mutants and transgenes were constructed in a Col-0 background. The mutants comprised a set of T-DNA insert lines obtained from the Arabidopsis Biological Resource Center (ABRC, Columbus, OH, USA), namely prh1-1 (GK_626H08), prh1-2 (Salk_014249), lbd16 (Salk_040739), lbd18 (Salk_038125) [43] and lbd29 (Salk_071133) [27], along with arf7 (Salk_040394) [33]; lax3 [28]; the transgenic lines were Super::LBD29-GFP [44], 35S::MYC-LBD16, 35S::MYC-LBD18, 35S::PRH1, 35S::PRH1-GFP (PRH1-GFP) and PRH1pro:GUS. Seeds were surface-sterilized by fumigation with chlorine gas for 4 hours, and then plated on solidified half strength Murashige and Skoog (MS) medium. All of the seeds were held for two days at 4°C and finally grown in a greenhouse delivering a 16 h photoperiod with a constant temperature of 22°C.
Transgene constructs and the generation of transgenic lines
The 1,692 nt region upstream of the PRH1 start cordon was used as the PRH1 promoter, and was inserted, along with the PRH1 coding sequence into a pENTRTM/SD/D-TOPOTM plasmid (ThermoFisher). The construct was subsequently recombined with several Gateway destination vectors, namely PK7WG2 [45] for over-expression without any tags, PB7WGF2 for over-expression with an C-GFP tag, PGWB18 for over-expression with an N-Myc tag, PBI221 for over-expression, PKGWFS7.1 [45] for promoter analysis with C-GFP/GUS. Transgenesis was effected using the floral dip method [46], employing transgene constructs harbored by Agrobacterium tumefaciens strain GV3101.
Microscopy
The PRH1 subcellular localization was detected in tobacco leaf cells. The Agrobacterium tumefaciens strain GV3101 containing 35S::PRH1-GFP or the cytosolic marker ADH-RFP [29] or plasma membrane maker ZmCRN-mCherry [30] were incubated shaking until the OD600 reaching 0.6. Centrifuge the cells at 4000g for 5 minutes, then discard the supernatant and resuspend the pellet with induction buffer (0.01mol/L MES, 0.01Mol/L MgCl2, 150 μmol/L acetosyringone) to find OD = 0.4. Mix two kinds of agrobacterium up with a same volume and inject the mixture into 3 week old tobacco leaves. The transfected plants grown in the greenhouse for at least 48 h at 28°C before GFP and RFP caning. The fluorescent signals were visualized by using the LSM880 laser scanning confocal microscope (Zeiss). ZmCRN-mCherry and ADH-RFP signals were detected by using 561 nm laser excitation and 580–675 nm emission, PRH1-GFP signals were detected by using 561 nm laser excitation and 500–530 nm emission.
The 8-d-old seedlings of PRH1pro:GUS were used for GUS staining. The GUS staining assay followed Liu et al. [47]. GUS signal was photographed by using an Olympus BX53 microscope supported with an Olympus DP72 digital camera.
Phenotype analysis
Eight-day-old seedlings were photographed using a Epson Perfection V800 scanner for the whole seedling images and the root length was measured by Image J software. To observe lateral root primordia clearly the seedlings were treated with a serials of solutions (1. 4% HCL, 20% methanol at 57°C for 15 minutes. 2. 7% NaOH, 60% ethanol at room temperature for 15 minutes. 3. 60% ethanol at room temperature for 10 minutes. 4. 40% ethanol at room temperature for 10 minutes. 5. 20% ethanol at room temperature for 10 minutes. 6. 10% ethanol at room temperature for 10 minutes. 7. 25% glycerol, 5% ethanol at room temperature for 10 minutes) before mounted in 50% glycerol on slides. Then the lateral root primordia and the emerged lateral roots were calculated using the Olympus BX53 microscope. The differential stages of lateral roots development have been described by Malamy and Benfey [1].
RNA isolation and quantitative real time PCR (qPCR) analysis
RNA was extracted from the roots of eight-day-old seedlings (some of which had been treated with 10 μM naphthalene acetic acid (NAA), obtained from Sigma (St. Louis, MO, USA)) using a TIANGEN Biotech (Beijing) Co. RNA simple Total RNA kit (www.tiangen.com), following the manufacturer’s protocol. A 2 μg aliquot of the RNA was used as the template for synthesizing the first cDNA strand, using a Fast Quant RT Kit (TIANGEN Biotech (Beijing) Co.). Subsequent qPCRs were processed on an MyiQ Real-time PCR Detection System (Bio-Rad, Hercules, CA, USA), using the Super Real PreMix Plus SYBR Green reagent (TIANGEN Biotech (Beijing) Co.). Each sample was represented by three biological replicates, and each biological replicate by three technical replicates. The AtACTIN2 (At3g18780) sequence was used as the reference. Details of the relevant primer sequences are given in S3 Table.
RNA-Seq analysis
RNA submitted for RNA-seq analysis was isolated from the roots of eight-day-old WT and arf7 seedlings (treated with the 10 μM naphthalene acetic acid (NAA) for 4 hours and the non-treated samples), using a RNeasy Mini Kit (Qiagen, Hilden, Germany). The RNA-seq analysis were processed following the methods given by Lv et al. [48]
Chromatin immunoprecipitation coupled to quantitative PCR (ChIP-qPCR) analysis
DNA harvested from eight-day-old seedlings of WT and transgenic lines harboring either 35S::MYC-ARF7, 35S::MYC-LBD16, 35S::MYC-LBD18 (ARF7, LBD16 and LBD18 were cloned into PGWB18) or Super::LBD29-GFP [44, 49] then they were cross-linked in a solution containing 1% v/v formaldehyde. The ChIP procedure followed that given by Gendrel A-V et al. [50], and employed antibodies recognizing MYC and GFP (Abcam, UK). The immune-precipitated DNA and total input DNA were analyzed by qPCR. The enrichment in WT and transgenic plants was measured as a ratio of the bound promoter fragments over total input respectively. Details of the relevant primers are given in S3 Table.
Yeast one-hybrid (Y1H) assays
Y1H assays were carried out using the Matchmaker Gold yeast one hybrid Library Screening System (Biosciences Clontech, Palo Alto, CA, USA). The PRH1 promoter was inserted into the pAbAi vector, and the bait vector (PRH1pro-pAbAi) was linearized, restricted by BbsI, and introduced into the Y1H Goldeast strain along with the prey vector AD-ARF7. Transformants were grown on SD/-Leu-Ura dropout plates containing different concentrations of aureobasidin A (CAT#630499, Clontech, USA). The primers used for generating the various constructs are listed in S3 Table.
Dual-luciferase transient expression assay in A. thaliana protoplasts
Protoplasts were isolated from three week old WT leaves following Yoo et al. [51]. The ligation of the PRH1 promoter to pGreen0800-LUC was realized by enzyme digestion and ligation [52]. The primers used for this procedure are listed in S3 Table. The PRH1pro:LUC reporter plasmid was transferred into Arabidopsis protoplast cells with one or more effector plasmids (ARF7, LBD16, LBD18, LBD29 –PBI221); the empty vector PBI221 was used as the negative control. The dual-luciferase assay kit (Promega, USA) allows for the quantification of both firefly and renilla luciferase activity. Signals were detected with a Synergy 2 multimode micro-plate (Centro LB960, Berthold, Germany).
Generation of prh1-3 using CRISPR technology
The experimental method was described by Yan et al [20]. The Arabidopsis plants Columbia-0 ecotype were used as wild type (WT) and were transformed with the CRISPR construct by floral dipping. The prh1-3 plants were identified at the T2 stage. The target sequence is presented in S3 Table.
Statistical analysis
Tests were applied to the data to check for normality and homogeneity before attempting an analysis of the variation. The data were analyzed using routines implemented in Prism 5 software (GraphPad Software). The Students’ t-test was applied to compare pairs of means. Values have been presented in the form means±SE; the significance thresholds were 0.05 (*), 0.01 (**) and 0.001 (***). Comparisons between multiple means were made using a one-way analysis of variance and Tukey’s test.
Accession numbers
Sequence data referred to in this paper have been deposited in both the Arabidopsis Genome Initiative and the GenBank/EMBL databases under the accession numbers: ARF7 (At5g20730, NP_851046), PRH1 (At2g19990, NP_179589), LBD16 (At2g42430, NP_565973), LBD18 (At2g45420, NP_850436), LBD29 (At3g58190, NP_191378), LAX3 (At1g77690, NP_177892). The RNA-seq data are available in the Gene Expression Omnibus database under accession number GSE122355.
Supporting information
(A) Numbers of DEGs in arf7 roots compared to WT with auxin treatment (treated with 10 μM naphthalene acetic acid (NAA) for 4 hours) or not. (B) Selected candidate genes from both the up-regulated DEGs in WT by auxin and down-regulated genes in arf7 (the bigger purple circle), and the 8 targets involved in lateral root development further refined by Arabidops is eFP Browser from the 23 selected candidates (the smaller yellow circle). (C) The expression pattern and change folds of 8 targets at the site of lateral roots initiation in WT and slr-1 under the condition of auxin treatment or not.
(TIF)
(A) Phylogenetic tree of PRH1 in Arabidopsis. Numbers in green represent bootstrap values. The key genes PR1 and PRH1 were marked in red. (B) The induction of PRH1 by auxin (10 μM NAA) was monitored in wild-type (Col-0) by qPCR. Values represent averages of three biological replicates in the experiment, and the total RNA was extracted from the primary roots of about 100 seedlings for each repeat. NAA: naphthalene acetic acid. Error bars represent SE. (C) Expression pattern of PRH1 in intact seedlings before (left) and after (right) a 4 hours exposure to 10 μM NAA. GUS signals appear blue. GUS: β-glucuronidase. Bar: 0.5 cm.
(TIF)
(A) A map of the T-DNA insertion and the frameshift mutant of prh1 on chromosome (upper panel), and the single base insertion site in the coding sequence (lower panel). The orange line represents the promoter region, the black box represents the coding region and the blue lines represent the insertion sites. (B) Latral root (LR) number per centimeter (cm) along the primary root of the PRH1 over-expression lines. Values shown as mean±SE, three biological replicates in the experiment, 20 plant seedlings for each repeat. (C) The PRH1 relative transcript aboundance in the primary roots of WT, prh1 mutants and plants over-expressing PRH1. Three biological replicates in the experiment. Total mRNA was extracted from the primary roots of about 100 seedlings for each repeat. Data represent means±SE. **, ***: means differ significantly (P<0.01, P<0.001) from the WT control.
(TIF)
(A) The 8-day-old seedlings of WT, prh1-1 and RH1pro-PRH1 transgenetic lines in both WT and the prh1-1 background. Bar: 1 cm. (B) LR phenotyping was achieved by synchronizing lateral root formation with a gravistimulus for 18 h and 42 h. Primordia stages from I to VII were based on the classification by Malamy and Benfey [1] and the data were analysed from 20 seedlings. (C) Density of primordia at given stages. *,**: means differ significantly (P<0.05, P<0.01) from the WT control. (D) The LR density of WT, prh1-1 and the transgenetic lines. LRP: lateral root primordia. LRE: emerged lateral root, LRT: total lateral roots including the LRP and LRE. Data shown as means±SE, three biological replicates in the experiment, twenty plant seedlings for each repeat. The asterisks indicate means which differ significantly (P<0.05) from one another.
(TIF)
(A) Transcription level of PRH1 in the roots of WT, lbd16, lbd18 and lbd29 seedlings exposed to 10 μM naphthalene acetic acid (NAA) for 4 hours (orange) or not treated (brown) before extracting total mRNA. Different letters atop the columns indicate significant (P<0.05) differences in abundance. (B) Transcription level of PRH1 is enhanced in each of the LBD16, 18 and 29 over-expression lines. Data represent means±SE, three biological replicates in the experiment **, ***: means differ significantly (P<0.01, P<0.001) from the WT. (C) LAX3 expression in prh1-1 in response to auxin. Eight-day-old seedlings were incubated with 10 μM naphthalene acetic acid (NAA) for 4 hours. Total mRNA were extracted from the primary roots and subjected to qPCR. Data are the means±SE, three independent biological replications. Different letters atop the columns indicate significant (P<0.05) differences in abundance. (D) PRH1 expression in lax3 in response to auxin. Eight-day-old seedlings were incubated with 10 μM naphthalene acetic acid (NAA) for 4 hours. Total mRNA were extracted from the primary roots and subjected to qPCR. Data are the means±SE, three independent biological replications. Different letters atop the columns indicate significant (P<0.05) differences in abundance.
(TIF)
(A) Structure of the PRH1 promoter, showing the putative binding motifs of LBD18 (red square) [12] and LBD29 (yellow square) [44]. (B) Yeast one-hybrid binding assay containing the interaction between LBD16, LBD18, LBD29 and PRH1 promoter.
(TIF)
(A and B) Laser-scanning confocal image of PRH1-GFP fusion protein transiently expressed in N. benthamiana leaf cells with ADH-RFP as a cytosolic maker [29] (A) or ZmCRN-mCherry as a plasma membrane maker [30] (B). GFP: green fluorescent protein. RFP: red fluorescent protein. Scale bar: 20 μm.
(TIF)
Eight-day-old seedlings of WT and the prh1-1 mutant were used in this study. Total mRNA were extracted from the primary roots and subjected to qPCR. Data are the means±SE, three independent biological replications. The asterisk means differ significantly (P<0.05) from the WT.
(TIF)
(XLSX)
(XLSX)
(XLSX)
Acknowledgments
We thank Prof. Yuxin Hu, Prof. Jiri Friml, Prof. Eva Benkova, and Malcolm J. Bennett for sharing published materials.
Data Availability
The RNA-seq data are available in the Gene Expression Omnibus database under accession number GSE122355.
Funding Statement
This work is supported by the National Natural Science Foundation of China (Projects 31770305, 31870252 and 31470371), by Qingdao's Leading Technology Innovator Project, by Youth Interdisciplinary Science and Innovative Research Groups of Shandong University (Grant No. 2020QNQT014), by the National Natural Science Foundation of China (No. 31500227), the Fundamental Research Funds of Shandong University (No. 2016GN021). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Malamy JE, Benfey PN. Organization and cell differentiation in lateral roots of Arabidopsis thaliana. Development. 1997;124: 33–44. doi:VL—124 [DOI] [PubMed] [Google Scholar]
- 2.Dubrovsky JG, Doerner PW, Colón-Carmona A, Rost TL. Pericycle Cell Proliferation and Lateral Root Initiation in Arabidopsis. Plant Physiology. 2000;124: 1648–1657. 10.1104/pp.124.4.1648 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Peret B, Larrieu A, Bennett MJ. Lateral root emergence: a difficult birth. Journal of Experimental Botany. 2009;60: 3637–3643. 10.1093/jxb/erp232 [DOI] [PubMed] [Google Scholar]
- 4.Benková E, Bielach A. Lateral root organogenesis—from cell to organ. Current Opinion in Plant Biology. 2010;13: 677–683. 10.1016/j.pbi.2010.09.006 [DOI] [PubMed] [Google Scholar]
- 5.De Smet I, Tetsumura T, De Rybel B, Frey NF d., Laplaze L, Casimiro I, et al. Auxin-dependent regulation of lateral root positioning in the basal meristem of Arabidopsis. Development. 2007;134: 681–690. 10.1242/dev.02753 [DOI] [PubMed] [Google Scholar]
- 6.Fukaki H, Tameda S, Masuda H, Tasaka M. Lateral root formation is blocked by a gain-of-function mutation in the SOLITARY-ROOT/IAA14 gene of Arabidopsis. The Plant Journal. 2002;29: 153–168. 10.1046/j.0960-7412.2001.01201.x [DOI] [PubMed] [Google Scholar]
- 7.Notaguchi M, Wolf S, Lucas WJ. Phloem-Mobile Aux / IAA Transcripts Target to the Root Tip and Modify Root Architecture F. Journal of Integrative Plant Biology. 2012;54: 760–772. 10.1111/j.1744-7909.2012.01155.x [DOI] [PubMed] [Google Scholar]
- 8.Berckmans B, Vassileva V, Schmid SPC, Maes S, Parizot B, Naramoto S, et al. Auxin-Dependent Cell Cycle Reactivation through Transcriptional Regulation of Arabidopsis E2Fa by Lateral Organ Boundary Proteins. The Plant Cell. 2011;23: 3671–3683. 10.1105/tpc.111.088377 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lee HW, Cho C, Kim J. Lateral Organ Boundaries Domain16 and 18 Act Downstream of the AUXIN1 and LIKE-AUXIN3 Auxin Influx Carriers to Control Lateral Root Development in Arabidopsis. Plant Physiology. 2015;168: 1792–1806. 10.1104/pp.15.00578 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kim J, Lee HW. Direct activation of EXPANSIN14 by LBD18 in the gene regulatory network of lateral root formation in Arabidopsis. Plant Signaling & Behavior. 2013;8: e22979 10.4161/psb.22979 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lee HW, Kim J. EXPANSINA17 Up-Regulated by LBD18/ASL20 promotes lateral root formation during the auxin response. Plant and Cell Physiology. 2013;54: 1600–1611. 10.1093/pcp/pct105 [DOI] [PubMed] [Google Scholar]
- 12.Lee HW, Kim M-J, Kim NY, Lee SH, Kim J. LBD18 acts as a transcriptional activator that directly binds to the EXPANSIN14 promoter in promoting lateral root emergence of Arabidopsis. The Plant Journal. 2013;73: 212–224. 10.1111/tpj.12013 [DOI] [PubMed] [Google Scholar]
- 13.Lee HW, Park JH, Park MY, Kim J. GIP1 may act as a coactivator that enhances transcriptional activity of LBD18 in Arabidopsis. Journal of Plant Physiology. Elsevier GmbH.; 2014;171: 14–18. 10.1016/j.jplph.2013.11.003 [DOI] [PubMed] [Google Scholar]
- 14.Okushima Y, Fukaki H, Onoda M, Theologis A, Tasaka M. ARF7 and ARF19 Regulate Lateral Root Formation via Direct Activation of LBD/ASL Genes in Arabidopsis. The Plant Cell. 2007;19: 118–130. 10.1105/tpc.106.047761 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kumpf RP, Shi C-L, Larrieu A, Sto IM, Butenko MA, Peret B, et al. Floral organ abscission peptide IDA and its HAE/HSL2 receptors control cell separation during lateral root emergence. Proceedings of the National Academy of Sciences. 2013;110: 5235–5240. 10.1073/pnas.1210835110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Laird J, Armengaud P, Giuntini P, Laval V, Milner JJ. Inappropriate annotation of a key defence marker in Arabidopsis: Will the real PR-1 please stand up? Planta. 2004;219: 1089–1092. 10.1007/s00425-004-1355-x [DOI] [PubMed] [Google Scholar]
- 17.Lu S, Friesen TL, Faris JD. Molecular characterization and genomic mapping of the pathogenesis-related protein 1 (PR-1) gene family in hexaploid wheat (Triticum aestivum L.). Molecular Genetics and Genomics. 2011;285: 485–503. 10.1007/s00438-011-0618-z [DOI] [PubMed] [Google Scholar]
- 18.Chen Y-L, Lee C, Cheng K, Chang W, Huang R, Nam HG, et al. Quantitative Peptidomics Study Reveals That a Wound-Induced Peptide from PR-1 Regulates Immune Signaling in Tomato. The Plant Cell. 2014;26: 4135–4148. 10.1105/tpc.114.131185 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Schumann U, Lee J, Kazan K, Ayliffe M, Wang M. DNA-Demethylase Regulated Genes Show Methylation-Independent Spatiotemporal Expression Patterns. Frontiers in Plant Science. 2017;8: 1–15. 10.3389/fpls.2017.00001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yan L, Wei S, Wu Y, Hu R, Li H, Yang W, et al. High-Efficiency Genome Editing in Arabidopsis Using YAO Promoter-Driven CRISPR/Cas9 System. Molecular Plant. 2015;8: 1820–1823. 10.1016/j.molp.2015.10.004 [DOI] [PubMed] [Google Scholar]
- 21.Péret B, Li G, Zhao J, Band LR, Voß U, Postaire O, et al. Auxin regulates aquaporin function to facilitate lateral root emergence. Nature Cell Biology. 2012;14: 991–998. 10.1038/ncb2573 [DOI] [PubMed] [Google Scholar]
- 22.Voß U, Wilson MH, Kenobi K, Gould PD, Robertson FC, Peer WA, et al. The circadian clock rephases during lateral root organ initiation in Arabidopsis thaliana. Nature Communications. 2015;6: 7641 10.1038/ncomms8641 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lucas M, Godin C, Jay-Allemand C, Laplaze L. Auxin fluxes in the root apex co-regulate gravitropism and lateral root initiation. Journal of Experimental Botany. 2008;59: 55–66. 10.1093/jxb/erm171 [DOI] [PubMed] [Google Scholar]
- 24.Lee HW, Kim NY, Lee DJ, Kim J. LBD18/ASL20 Regulates Lateral Root Formation in Combination with LBD16/ASL18 Downstream of ARF7 and ARF19 in Arabidopsis. Plant Physiology. 2009;151: 1377–1389. 10.1104/pp.109.143685 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lee DJ, Park JW, Lee HW, Kim J. Genome-wide analysis of the auxin-responsive transcriptome downstream of iaa1 and its expression analysis reveal the diversity and complexity of auxin-regulated gene expression. Journal of Experimental Botany. 2009;60: 3935–3957. 10.1093/jxb/erp230 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lee HW, Kang NY, Pandey SK, Cho C, Lee SH, Kim J. Dimerization in LBD16 and LBD18 Transcription Factors Is Critical for Lateral Root Formation. Plant Physiology. 2017;174: 301–311. 10.1104/pp.17.00013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Porco S, Larrieu A, Du Y, Gaudinier A, Goh T, Swarup K, et al. Lateral root emergence in Arabidopsis is dependent on transcription factor LBD29 regulation of auxin influx carrier LAX3. Development. 2016;143: 3340–3349. 10.1242/dev.136283 [DOI] [PubMed] [Google Scholar]
- 28.Swarup K, Benková E, Swarup R, Casimiro I, Péret B, Yang Y, et al. The auxin influx carrier LAX3 promotes lateral root emergence. Nature Cell Biology. 2008;10: 946–954. 10.1038/ncb1754 [DOI] [PubMed] [Google Scholar]
- 29.Yang Y-Z, Ding S, Wang Y, Li C-L, Shen Y, Meeley R, et al. Small kernel2 Encodes a Glutaminase in Vitamin B 6 Biosynthesis Essential for Maize Seed Development. Plant Physiology. 2017;174: 1127–1138. 10.1104/pp.16.01295 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Je B Il, Xu F, Wu Q, Liu L, Meeley R, Gallagher JP, et al. The CLAVATA receptor FASCIATED EAR2 responds to distinct CLE peptides by signaling through two downstream effectors. eLife. 2018;7: 1–21. 10.7554/eLife.35673 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kwon YR, Lee HJ, Kim KH, Hong S-W, Lee SJ, Lee H. Ectopic expression of Expansin3 or Expansinβ1 causes enhanced hormone and salt stress sensitivity in Arabidopsis. Biotechnology Letters. 2008;30: 1281–1288. 10.1007/s10529-008-9678-5 [DOI] [PubMed] [Google Scholar]
- 32.Porco S, Larrieu A, Du Y, Gaudinier A, Goh T, Swarup K, et al. Lateral root emergence in Arabidopsis is dependent on transcription factor LBD29 regulation of auxin influx carrier LAX3. Development. 2016;143: 3340–3349. 10.1242/dev.136283 [DOI] [PubMed] [Google Scholar]
- 33.Okushima Y, Overvoorde PJ, Arima K, Alonso JM, Chan A, Chang C, et al. Functional Genomic Analysis of the AUXIN RESPONSE FACTOR Gene Family Members in Arabidopsis thaliana: Unique and Overlapping Functions of ARF7 and ARF19. The Plant Cell. 2005;17: 444–463. 10.1105/tpc.104.028316 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Vanneste S, De Rybel B, Beemster GTS, Ljung K, De Smet I, Van Isterdael G, et al. Cell Cycle Progression in the Pericycle Is Not Sufficient for SOLITARY ROOT/IAA14-Mediated Lateral Root Initiation in Arabidopsis thaliana. The Plant Cell. 2005;17: 3035–3050. 10.1105/tpc.105.035493 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.De Smet I, Lau S, Voss U, Vanneste S, Benjamins R, Rademacher EH, et al. Bimodular auxin response controls organogenesis in Arabidopsis. Proceedings of the National Academy of Sciences. 2010;107: 2705–2710. 10.1073/pnas.0915001107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Piya S, Shrestha SK, Binder B, Stewart CN, Hewezi T. Protein-protein interaction and gene co-expression maps of ARFs and Aux/IAAs in Arabidopsis. Frontiers in Plant Science. 2014;5: 1–9. 10.3389/fpls.2014.00744 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Laskowski M, Biller S, Stanley K, Kajstura T, Prusty R. Expression Profiling of Auxin-treated Arabidopsis Roots: Toward a Molecular Analysis of Lateral Root Emergence. Plant and Cell Physiology. 2006;47: 788–792. 10.1093/pcp/pcj043 [DOI] [PubMed] [Google Scholar]
- 38.Shuai B, Reynaga-Pen˜a CG, Springer PS. The Lateral Organ Boundaries Gene Defines a Novel, Plant-Specific Gene Family. Plant Physiology. 2002;129: 747–761. 10.1104/pp.010926 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lewis DR, Olex AL, Lundy SR, Turkett WH, Fetrow JS, Muday GK. A Kinetic Analysis of the Auxin Transcriptome Reveals Cell Wall Remodeling Proteins That Modulate Lateral Root Development in Arabidopsis. The Plant Cell. 2013;25: 3329–3346. 10.1105/tpc.113.114868 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Himanen K, Boucheron E, Vanneste S, de Almeida Engler J, Inzé D, Beeckman T. Auxin-Mediated Cell Cycle Activation during Early Lateral Root Initiation. The Plant Cell. 2002;14: 2339–2351. 10.1105/tpc.004960 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pandey SK, Kim J. Coiled-coil motif in LBD16 and LBD18 transcription factors are critical for dimerization and biological function in arabidopsis. Plant Signaling & Behavior. Taylor & Francis; 2018;13: e1411450 10.1080/15592324.2017.1411450 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Xu C, Cao H, Zhang Q, Wang H, Xin W, Xu E, et al. Control of auxin-induced callus formation by bZIP59–LBD complex in Arabidopsis regeneration. Nature Plants. Springer US; 2018;4: 108–115. 10.1038/s41477-017-0095-4 [DOI] [PubMed] [Google Scholar]
- 43.Fan M, Xu C, Xu K, Hu Y. LATERAL ORGAN BOUNDARIES DOMAIN transcription factors direct callus formation in Arabidopsis regeneration. Cell Research. 2012;22: 1169–1180. 10.1038/cr.2012.63 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Xu C, Cao H, Xu E, Zhang S, Hu Y. Genome-Wide Identification of Arabidopsis LBD29 Target Genes Reveals the Molecular Events behind Auxin-Induced Cell Reprogramming during Callus Formation. Plant and Cell Physiology. 2018;59: 749–760. 10.1093/pcp/pcx168 [DOI] [PubMed] [Google Scholar]
- 45.Karimi M, Inzé D, Depicker A. GATEWAYTM vectors for Agrobacterium- mediated plant transformation. Trends in Plant Science. 2002;7: 193–195. 10.1016/s1360-1385(02)02251-3 [DOI] [PubMed] [Google Scholar]
- 46.Clough SJ, Bent AF. Floral dip: a simplified method for Agrobacterium -mediated transformation ofArabidopsis thaliana. The Plant Journal. 1998;16: 735–743. 10.1046/j.1365-313x.1998.00343.x [DOI] [PubMed] [Google Scholar]
- 47.Liu G, Gao S, Tian H, Wu W, Robert HS, Ding Z. Local Transcriptional Control of YUCCA Regulates Auxin Promoted Root-Growth Inhibition in Response to Aluminium Stress in Arabidopsis. Yu H, editor. PLOS Genetics. 2016;12: e1006360 10.1371/journal.pgen.1006360 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lv B, Tian H, Zhang F, Liu J, Lu S, Bai M, et al. Brassinosteroids regulate root growth by controlling reactive oxygen species homeostasis and dual effect on ethylene synthesis in Arabidopsis. Muday GK, editor. PLOS Genetics. 2018;14: e1007144 10.1371/journal.pgen.1007144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Chinnusamy V, Ohta M, Kanrar S, Lee B-H, Hong X, Agarwal M, et al. ICE1: a regulator of cold-induced transcriptome and freezing tolerance in Arabidopsis. Genes & development. 2003;17: 1043–54. 10.1101/gad.1077503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gendrel A-V, Lippman Z, Martienssen R, Colot V. Profiling histone modification patterns in plants using genomic tiling microarrays. Nature Methods. 2005;2: 213–218. 10.1038/nmeth0305-213 [DOI] [PubMed] [Google Scholar]
- 51.Yoo S-D, Cho Y-H, Sheen J. Arabidopsis mesophyll protoplasts: a versatile cell system for transient gene expression analysis. Nature Protocols. 2007;2: 1565–1572. 10.1038/nprot.2007.199 [DOI] [PubMed] [Google Scholar]
- 52.Hellens RP, Allan AC, Friel EN, Bolitho K, Grafton K, Templeton MD, et al. Transient expression vectors for functional genomics, quantification of promoter activity and RNA silencing in plants. Plant Methods. 2005;1: 1–14. 10.1186/1746-4811-1-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(A) Numbers of DEGs in arf7 roots compared to WT with auxin treatment (treated with 10 μM naphthalene acetic acid (NAA) for 4 hours) or not. (B) Selected candidate genes from both the up-regulated DEGs in WT by auxin and down-regulated genes in arf7 (the bigger purple circle), and the 8 targets involved in lateral root development further refined by Arabidops is eFP Browser from the 23 selected candidates (the smaller yellow circle). (C) The expression pattern and change folds of 8 targets at the site of lateral roots initiation in WT and slr-1 under the condition of auxin treatment or not.
(TIF)
(A) Phylogenetic tree of PRH1 in Arabidopsis. Numbers in green represent bootstrap values. The key genes PR1 and PRH1 were marked in red. (B) The induction of PRH1 by auxin (10 μM NAA) was monitored in wild-type (Col-0) by qPCR. Values represent averages of three biological replicates in the experiment, and the total RNA was extracted from the primary roots of about 100 seedlings for each repeat. NAA: naphthalene acetic acid. Error bars represent SE. (C) Expression pattern of PRH1 in intact seedlings before (left) and after (right) a 4 hours exposure to 10 μM NAA. GUS signals appear blue. GUS: β-glucuronidase. Bar: 0.5 cm.
(TIF)
(A) A map of the T-DNA insertion and the frameshift mutant of prh1 on chromosome (upper panel), and the single base insertion site in the coding sequence (lower panel). The orange line represents the promoter region, the black box represents the coding region and the blue lines represent the insertion sites. (B) Latral root (LR) number per centimeter (cm) along the primary root of the PRH1 over-expression lines. Values shown as mean±SE, three biological replicates in the experiment, 20 plant seedlings for each repeat. (C) The PRH1 relative transcript aboundance in the primary roots of WT, prh1 mutants and plants over-expressing PRH1. Three biological replicates in the experiment. Total mRNA was extracted from the primary roots of about 100 seedlings for each repeat. Data represent means±SE. **, ***: means differ significantly (P<0.01, P<0.001) from the WT control.
(TIF)
(A) The 8-day-old seedlings of WT, prh1-1 and RH1pro-PRH1 transgenetic lines in both WT and the prh1-1 background. Bar: 1 cm. (B) LR phenotyping was achieved by synchronizing lateral root formation with a gravistimulus for 18 h and 42 h. Primordia stages from I to VII were based on the classification by Malamy and Benfey [1] and the data were analysed from 20 seedlings. (C) Density of primordia at given stages. *,**: means differ significantly (P<0.05, P<0.01) from the WT control. (D) The LR density of WT, prh1-1 and the transgenetic lines. LRP: lateral root primordia. LRE: emerged lateral root, LRT: total lateral roots including the LRP and LRE. Data shown as means±SE, three biological replicates in the experiment, twenty plant seedlings for each repeat. The asterisks indicate means which differ significantly (P<0.05) from one another.
(TIF)
(A) Transcription level of PRH1 in the roots of WT, lbd16, lbd18 and lbd29 seedlings exposed to 10 μM naphthalene acetic acid (NAA) for 4 hours (orange) or not treated (brown) before extracting total mRNA. Different letters atop the columns indicate significant (P<0.05) differences in abundance. (B) Transcription level of PRH1 is enhanced in each of the LBD16, 18 and 29 over-expression lines. Data represent means±SE, three biological replicates in the experiment **, ***: means differ significantly (P<0.01, P<0.001) from the WT. (C) LAX3 expression in prh1-1 in response to auxin. Eight-day-old seedlings were incubated with 10 μM naphthalene acetic acid (NAA) for 4 hours. Total mRNA were extracted from the primary roots and subjected to qPCR. Data are the means±SE, three independent biological replications. Different letters atop the columns indicate significant (P<0.05) differences in abundance. (D) PRH1 expression in lax3 in response to auxin. Eight-day-old seedlings were incubated with 10 μM naphthalene acetic acid (NAA) for 4 hours. Total mRNA were extracted from the primary roots and subjected to qPCR. Data are the means±SE, three independent biological replications. Different letters atop the columns indicate significant (P<0.05) differences in abundance.
(TIF)
(A) Structure of the PRH1 promoter, showing the putative binding motifs of LBD18 (red square) [12] and LBD29 (yellow square) [44]. (B) Yeast one-hybrid binding assay containing the interaction between LBD16, LBD18, LBD29 and PRH1 promoter.
(TIF)
(A and B) Laser-scanning confocal image of PRH1-GFP fusion protein transiently expressed in N. benthamiana leaf cells with ADH-RFP as a cytosolic maker [29] (A) or ZmCRN-mCherry as a plasma membrane maker [30] (B). GFP: green fluorescent protein. RFP: red fluorescent protein. Scale bar: 20 μm.
(TIF)
Eight-day-old seedlings of WT and the prh1-1 mutant were used in this study. Total mRNA were extracted from the primary roots and subjected to qPCR. Data are the means±SE, three independent biological replications. The asterisk means differ significantly (P<0.05) from the WT.
(TIF)
(XLSX)
(XLSX)
(XLSX)
Data Availability Statement
The RNA-seq data are available in the Gene Expression Omnibus database under accession number GSE122355.