Abstract
Cognitive impairment is a common complication observed after a stroke. Currently there are no definitively proven pharmacologic therapies for recovery from post-stroke cognitive impairment and vascular dementia. In this meta-analysis, we evaluated the efficacy and safety of cholinesterase inhibitors in their improvement of cognition in patients with post-stroke cognitive impairment and vascular dementia. We conducted a meta-analysis using seven eligible studies from 305 published articles. We investigated the differences in Mini–Mental State Examination (MMSE) and Alzheimer’s Disease Assessment Scale-Cognitive subscale (ADAS-Cog) scores, before and after cholinergic augmentation in patients with post-stroke cognitive impairment and vascular dementia. MMSE and ADAS-cog scores were also compared during the subsequent follow-up periods. MMSE score of patients with post-stroke cognitive impairment was increased after cholinergic augmentation throughout the 24 weeks with mean differences [MD] of 3.000, 1.732, 1.578 1.516, and 1.222, at 4, 4–8, 8–12, 12–18, and 18–24 weeks, respectively. In addition, ADAS-cog scores decreased at 6, 12, 18, and 24 weeks by pharmaceutical augmentation, but not with placebo with mean differences [MD] of -2.333, -2.913, -2.767, -2.416, and -1.859, respectively. This meta-analysis shows that acetylcholinesterase inhibitors maintain a stable pattern of improved cognitive function in patients with post stroke cognitive impairment and vascular dementia without the increased risk of side effects.
Introduction
Post-stroke cognitive impairment is a common complication observed after stroke. The prevalence of dementia within the first year after stroke ranges from 9% to 30% [1]. However, the prevalence of cognitive impairment with no dementia may be much higher. Post-stroke cognitive impairment can also be a risk factor for vascular dementia. The prevalence of new-onset dementia shortly after a first incidence of stroke is about 10% after a recurrent stroke excluding pre-stroke dementia is about 30% [2]. Stroke itself is one of the main causes of vascular dementia after a stroke [2].
Recovery after stroke arises spontaneously and may last weeks, even years especially for the recovery of language skills and cognition [3]. After 1 year, only 10% of the stroke patients with cognitive impairment with no dementia recover [4]. Pharmacotherapy accelerates spontaneous recovery of post-stroke cognitive impairment, and enhancement of cognition might further facilitate functional recovery. Pharmacological interventions modulating stroke-induced disruption of diverse neurotransmitters may further enhance cognition through brain plasticity and long-term strengthening [5].
Cholinergic agents, such as donepezil, rivastigmine, and galantamine, are commonly used to treat vascular dementia. While evidence exists from large randomized controlled trials on the efficacy of these cholinergic agents in the treatment of Alzheimer’s dementia, their efficacy in the treatment of post stroke cognitive impairment remains less defined. A meta-analysis by Kavirajan and Schneider found that cholinesterase inhibitors might produce small benefits in cognition in patients with mild to moderate vascular dementia; however, this evidence was not significant enough for widespread use of acetylcholinesterase inhibitors [6]. Furthermore, recent drug trials reported that cholinergic boosting using donepezil had a beneficial effect in chronic stroke patients with aphasia after a 16-weeks treatment regimen [7]. Another pilot study suggested that donepezil might lead to improved functional recovery in the immediate post-stroke period [8]. We therefore aimed to evaluate the efficacy and adverse effects of these pharmacological interventions in the treatment of post-stroke cognitive impairment and vascular dementia.
Materials and methods
Published study search and selection criteria
Relevant articles were obtained by searching the PubMed and MEDLINE databases for studies published prior to November 15, 2019. The database was searched using the following key words and search string: ‘Stroke’ AND ‘cholinesterase inhibitors’ OR ‘donepezil’ OR ‘rivastigmine’ OR ‘galantamine’. The titles and the abstracts of all searched articles were screened including review articles in order to find additional eligible studies. The search results were then reviewed and included if (1) the study was performed on human patients, and (2) there was information for the MMSE or ADAS-cog scores after cholinergic augmentation in patients with infarction or vascular dementia. Case reports or non-original articles, and non-English language publications were excluded.
Data extraction
Data from all eligible studies were extracted by two independent authors. The data extracted from each of the eligible studies included; the paper reference, first author’s name, year of publication, study location, regimen of pharmaceutical augmentation, number and age of the patients, MMSE, ADAS-cog scores, or Clinician’s Interview-Based Impression of Change-Plus (CIBIC-Plus), and any complications that may have risen after cholinergic augmentation or placebo treatment. In addition, the assessment for the quality of nonrandomized studies in the meta-analysis was performed using the Newcastle-Ottawa Scale.
Statistical analyses
All data were analyzed using the Comprehensive Meta-Analysis software package (Biostat, Englewood, NJ). We analyzed the mean differences of MMSE and ADAS-cog scores, before and after cholinergic augmentation in patients with post-stroke cognitive impairment and vascular dementia. The change of MMSE and ADAS-cog scores during various follow-up periods were also compared. Complications such as minor or severe adverse effects, and deaths were also noted. Since the eligible studies included different populations, a random-effects model was deemed more suitable than a fixed-effects model. Heterogeneity between the studies was checked using the Q and I2 statistics, and presented using P-values. Sensitivity analysis was conducted to assess the heterogeneity of eligible studies and the impact of each study on the combined effect. Additionally, the meta-regression test was performed in order to elucidate the heterogeneity between subgroups. To assess for publication bias, Begg’s funnel plot and Egger’s test were used. The results were considered statistically significant at P < 0.05.
Results
Selection and characteristics
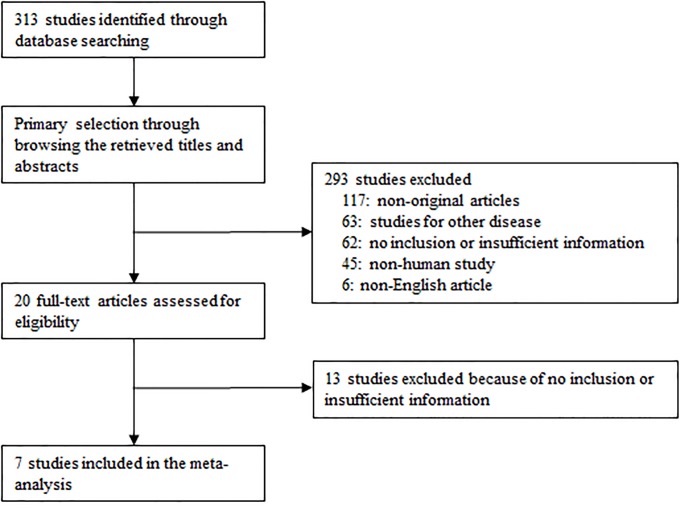
We searched the PubMed database using the before mentioned keywords (see Material and methods) and identified 305 reports. Among them, 117 articles were excluded because of non-original articles, 63 articles for insufficient information and 63 articles due to their study of other diseases, and further 51 articles because they used animals or cell lines (n = 43) and not written in English (n = 6). The remaining 7 studies were included in the meta-analysis (Fig 1 and Table 1) [9–15].
Fig 1. Flow of information through the different phases of the eligible studies.
Table 1. Main characteristics of the eligible studies.
| Study | Location | Subgroup | Regimen | Number of patients | Male | Female | Age (Mean) | MMSE (Mean±SD) | ADAS-cog (Mean±SD) | CIBIC-Plus (Mean±SD) |
|---|---|---|---|---|---|---|---|---|---|---|
| Black 2003 | UK | VD | Placebo | 199 | 115 | 84 | 74.2 | 21.7±0.3 | 20.1±0.7 | |
| Donepezil 5mg/day | 198 | 111 | 87 | 73.7 | 21.9±0.3 | 21.2±0.8 | ||||
| Donepezil 10mg/day | 206 | 107 | 99 | 73.9 | 21.8±0.3 | 20.9±0.7 | ||||
| Chang 2011 | Korea | PSCI | Placebo | 4 | 2 | 2 | 55 (28–74) | 24.8 (24–26) | ||
| Donepezil | 6 | 4 | 2 | 55.5 (45–69) | 24.2 (23–36) | |||||
| Moretti 2008 | Italy | PSCI | Rivastigmine | 50 | 19 | 31 | 74.23 | 18.6±2.1 | ||
| VD | Rivastigmine | 50 | 24 | 26 | 73.23 | 20.7±2.0 | ||||
| Narasimhalu 2010 | Singapore | PSCI | Placebo | 25 | 12 | 13 | 69.4 (48–84) | 23.9±3.2 | 30.4±14.1 | |
| Rivastigmine | 25 | 5 | 20 | 68.1 (57–81) | 23.7±3.4 | 29.9±13.1 | ||||
| Pratt 2002 | Various | VD | Placebo | 392 | 220 | 172 | 74.3 (41–91) | 21.6±4.2 | 22.3±11.1 | |
| Roman GC 2010 | US | VD | Placebo | 326 | 176 | 150 | 72.3 | 23.6±0.3 | 21.7±0.6 | 3.6±0.05 |
| Donepezil | 648 | 398 | 250 | 73.4 | 23.5±0.2 | 21.8±0.4 | 3.6±0.03 | |||
| Wilkinson 2003 | Various | VD | Placebo | 193 | 105 | 88 | 74.4 | 22.2±0.3 | 18.8±0.7 | |
| Donepezil 5mg/day | 208 | 130 | 78 | 74.7 | 21.8±0.3 | 20.8±0.7 | ||||
| Donepezil 10mg/day | 2115 | 134 | 81 | 75.7 | 21.5±0.3 | 20.6±0.7 |
Numbers in parentheses represent percentage
Abbreviations: MMSE, mini–mental state examination; ADAS-cog, Alzheimer’s Disease Assessment Scale-Cognitive subscale; CIBIC-Plus, Clinician’s Interview-Based Impression of Change-Plus; UK, United Kingdom; VD, vascular dementia; PSCI, post-stroke cognitive impairment; US, United States
Meta-analysis for the correlation between MMSE or ADAS-cog score/acetylcholinesterase inhibitor
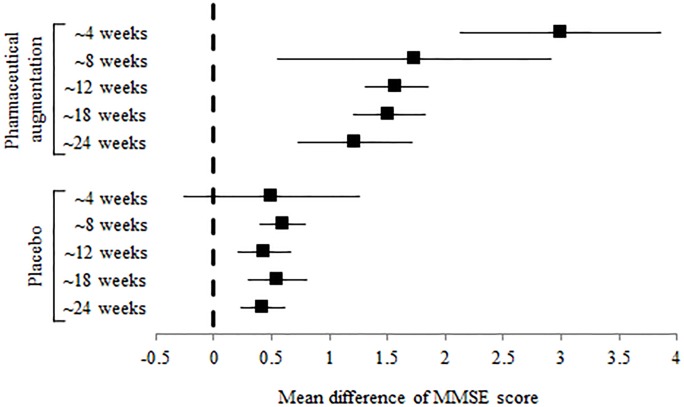
Mean differences (MD) of MMSE score in post-stroke cognitive impairment and vascular dementia with cholinergic augmentation were significantly increased throughout the 24 weeks: 3.000 (95% confidence interval [CI] 2.135 to 3.865) at 4 weeks, 1.732 (95% CI 0.555 to 2.910) at 4–8 weeks, 1.578 (95% CI 1.308 to 1.848) at 8–12 weeks, 1.516 (95% CI 1.203 to 1.829) at 12–18 weeks, and 1.222 (95% CI 0.727 to 1.718) at 18–24 weeks (Table 2 and Fig 2). Only minimal change of MMSE score was observed compared to placebo in post-stroke cognitive impairment and vascular dementia after 4 weeks (Table 2 and Fig 2). In addition, there was no significant impact of each study on estimated values.
Table 2. The mean difference of MMSE score according to the pharmaceutical augmentation.
| Number of Subsets | Heterogeneity test [P-value] | Random effect [95% Confidence interval] | Egger’s Test [P-value] | |
|---|---|---|---|---|
| in 4 weeks | ||||
| Placebo | 1 | 1.000 | 0.500 [-0.259, 1.259] | - |
| Pharmaceutical augmentation | 1 | 1.000 | 3.000 [2.135, 3.865] | - |
| 4–8 weeks | ||||
| Placebo | 3 | 0.913 | 0.600 [0.403, 0.796] | 0.870 |
| Pharmaceutical augmentation | 3 | < 0.001 | 1.732 [0.555, 2.910] | 0.107 |
| Donepezil 5mg/day | 2 | < 0.001 | 2.208 [-0.466, 4.882] | - |
| Donepezil 10mg/day | 1 | 1.000 | 0.968 [0.588, 1.347] | - |
| 8–12 weeks | ||||
| Placebo | 2 | 0.446 | 0.437 [0.206, 0.668] | - |
| Pharmaceutical augmentation | 2 | 0.300 | 1.578 [1.308, 1.848] | - |
| Donepezil 5mg/day | 1 | 1.000 | 1.387 [0.938, 1.836] | - |
| Donepezil 10mg/day | 1 | 1.000 | 1.677 [1.361, 1.994] | - |
| 12–18 weeks | ||||
| Placebo | 2 | 0.742 | 0.552 [0.302, 0.801] | - |
| Pharmaceutical augmentation | 2 | 0.686 | 1.516 [1.203, 1.829] | - |
| Donepezil 5mg/day | 1 | 1.000 | 1.581 [1.138, 2.023] | - |
| Donepezil 10mg/day | 1 | 1.000 | 1.452 [1.009, 1.894] | - |
| 18–24 weeks | ||||
| Placebo | 4 | 0.434 | 0.421 [0.233, 0.610] | 0.901 |
| Pharmaceutical augmentation | 5 | < 0.001 | 1.222 [0.727, 1.718] | 0.331 |
| Donepezil 5mg/day | 2 | 0.778 | 1.113 [0.652, 1.575] | - |
| Donepezil 10mg/day | 2 | 0.688 | 1.589 [1.270, 1.908] | - |
Abbreviations: MMSE, mini–mental state examination
Fig 2. The mean difference of MMSE(mini–mental state examination) score according to the pharmaceutical augmentation.
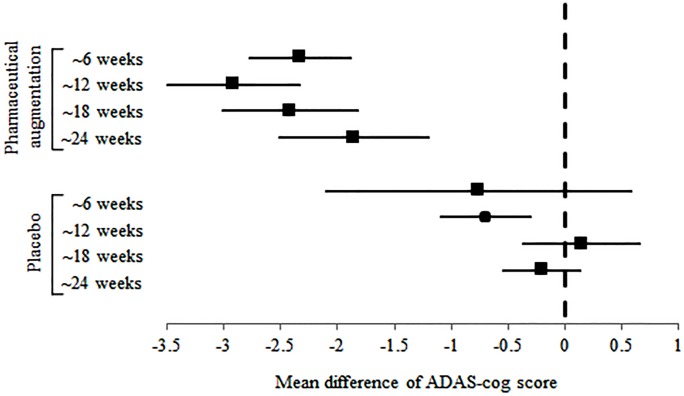
Next, the changes of ADAS-cog scores were evaluated with and without cholinergic augmentation. No studies were found comparing ADAS-cog in patients with post stroke cognitive impairment, and only the ADAS-cog score of patients with vascular dementia was comparable. ADAS-cog scores by cholinergic augmentation were found to be decreased by: MD of -2.333 (95% CI -2.778 to -1.889) at 6 weeks, MD of -2.913 (95% CI -3.490 to -2.335) at 12 weeks, MD of -2.416 (95% CI -3.009 to -1.824) at 18 weeks, and MD of -1.859 (95% CI -2.514 to -1.204) at 24 weeks and this decreased pattern was maintained for 24 weeks (Table 3 and Fig 3). In patients with vascular dementia, however, no effects of placebo on ADAS-cog scores were observed at follow-ups at 6, 18, and 24 weeks (MD -0.763, 95% CI -2.104 to 0.578; MD 0.148, 95% CI -0.365 to 0.133; and MD -0.204, 95% CI -0.541 to 0.133, respectively), but were observed at 12 weeks (MD -0.699, 95% CI -1.092 to -0.307).
Table 3. The mean difference of ADAS-cog score according to the pharmaceutical augmentation.
| Number of Subsets | Heterogeneity test [P-value] | Random effect [95% CI] | Egger’s Test [P-value] | |
|---|---|---|---|---|
| 6 weeks | ||||
| Placebo | 2 | < 0.001 | -0.763 [-2.104, 0.578] | - |
| Pharmaceutical augmentation | 2 | 1.000 | -2.333 [-2.778, -1.889] | - |
| Donepezil 5mg/day | 1 | 1.000 | -2.333 [-3.013, -1.654] | - |
| Donepezil 10mg/day | 1 | 1.000 | -2.333 [-2.921, -1.745] | - |
| 12 weeks | ||||
| Placebo | 2 | 0.384 | -0.699 [-1.092, -0.307] | - |
| Pharmaceutical augmentation | 2 | 0.542 | -2.913 [-3.490, -2.335] | - |
| Donepezil 5mg/day | 1 | 1.000 | -2.767 [-3.511, -2.022] | - |
| Donepezil 10mg/day | 1 | 1.000 | -3.133 [-4.048, -2.219] | - |
| 18 weeks | ||||
| Placebo | 2 | 0.197 | 0.148 [-0.365, 0.661] | - |
| Pharmaceutical augmentation | 2 | 0.471 | -2.416 [-3.009, -1.824] | - |
| Donepezil 5mg/day | 1 | 1.000 | -2.267 [-2.985, -1.548] | - |
| Donepezil 10mg/day | 1 | 1.000 | -2.733 [-3.779, -1.688] | - |
| 24 weeks | ||||
| Placebo | 4 | 0.292 | -0.204 [-0.541, 0.133] | 0.775 |
| Pharmaceutical augmentation | 5 | 0.002 | -1.859 [-2.514, -1.204] | 0.081 |
| Donepezil 5mg/day | 2 | 0.265 | -1.924 [-2.507, -1.342] | - |
| Donepezil 10mg/day | 2 | 0.212 | -2.386 [-3.179, -1.592] | - |
Abbreviations: ADAS-cog, alzheimer’s disease assessment scale-cognitive subscale;,CI, confidence interval
Fig 3. The mean difference of ADAS-cog score according to the pharmaceutical augmentation.
The improved rate in CIBIC-Plus was significantly higher in patients with donepezil 5mg/day than placebo group (P < 0.001 in a meta-regression test). However, there was no significant difference of improved rate in CIBIC-Plus between Donepezil 10mg/day and placebo subgroup (P = 0.693 in a meta-regression test). We added the methods and results for CIBIC-Plus in the revised manuscript.
Adverse effects were observed in 0.669 (95% CI 0.269 to 0.917) of patients with placebo and 0.813 (95% CI 0.516 to 0.946) of patients with cholinergic augmentation (Table 4). Severe adverse effects were found in 0.145 (95% CI 0.100 to 0.206) and 0.138 (95% CI 0.119 to 0.160) of patients with placebo and cholinergic augmentation, respectively. Death occurred in 0.018 (95% CI 0.010 to 0.032) and 0.009 (95% CI 0.000 to 0.189) in patients with cholinergic augmentation and placebo, respectively.
Table 4. The adverse effects according to the pharmaceutical augmentation.
| Number of Subsets | Heterogeneity test [P-value] | Random effect [95% CI] | Egger’s Test [P-value] | |
|---|---|---|---|---|
| Adverse effect | ||||
| Placebo | 4 | < 0.001 | 0.669 [0.269, 0.917] | 0.426 |
| Pharmaceutical augmentation | 6 | < 0.001 | 0.813 [0.516, 0.946] | 0.055 |
| Severe adverse effect | ||||
| Placebo | 3 | 0.099 | 0.145 [0.100, 0.206] | 0.666 |
| Pharmaceutical augmentation | 4 | 0.541 | 0.138 [0.119, 0.160] | 0.075 |
| Death | ||||
| Placebo | 2 | 0.058 | 0.009 [0.000, 0.189] | - |
| Pharmaceutical augmentation | 2 | 0.408 | 0.018 [0.010, 0.032] | - |
Abbreviations: CI, confidence interval
Discussion
Significant improvements of MMSE score were found between cholinergic augmentation and placebo groups in patients at 4 weeks with post stroke cognitive impairment and vascular dementia. ADAS-cog scores in patients with vascular dementia improved with cholinesterase inhibitor treatment and maintained a stable pattern of improved cognitive function compared to the placebo group through the 24 weeks. A study showed that ADAS-Cog improvement is likely clinically meaningful, whereas many patients with no change in the ADAS-Cog still show meaningful improvement [16]. In addition, the intensity and direction of the initial treatment response appear to be important in informing long-term outcomes [16].
Cholinergic pathways and the neural system are vulnerable to vascular damage and can lead to cognitive impairment. The cholinergic system includes the basal forebrain, substantia innominate, striatum, cerebral cortex (mainly pyramidal neurons and medium-sized neurons in layers III and V of the motor and secondary sensory areas), some brainstem nuclei, and spinal motor neurons [17]. The entire cerebral cortex and the white matter contains a dense network of cholinergic fibers originating from the nucleus basalis of Meynert [18]. Acetylcholinesterase inhibitors have been known to modulate cognitive function by compensating for the lack of intracerebral cholinergic neurotransmitters through inhibition of acetylcholine hydrolysis which is an effective treatment pathway in patients with post-stroke cognitive impairment and vascular dementia [19]. The mechanisms by which acetylcholinesterase inhibitors promote recovery from post stroke cognitive impairment are currently not fully understood, but two mechanisms are mostly considered to be: 1) Acetylcholine acting as a cortical modulator playing a critical role in practice-related plasticity [20] and, 2) the regulation of cerebral circulation influenced by cholinergic mechanisms and acetylcholine improving regional cerebral blood flow in vascular dementia patients [17]. In support of the first mechanism, experimental studies in rodents have shown that cholinergic fibers are associated with structural adaptation and functional recovery, thereby highlighting a key role for acetylcholine in rehabilitation-mediated recovery from traumatic brain injury [21]. For the latter, acetylcholinesterase inhibitor might restore the impaired cognition by improving on mechanisms that couple neuronal activity and vascular status of the brain.
Placebo-treated patients with post stroke cognitive impairment displayed minor improvements on MMSE after 4 weeks and only marginal improvements on ADAS-cog at 12 weeks (Tables 2 and 3). In general, post stroke cognitive impairment and motor dysfunction show recovery during the subacute phase and reach a plateau or level off after several months following the onset [22–23]. Patient with post-stroke cognitive impairment does not worsen without the recurrence of a stroke. As the deterioration of the cognitive function continues, there is a possibility that the pathology of the degenerative disease also plays a role. Our results also indicate that the ADAS-cog score is generally unchanged with the only change being significant at 12 weeks.
Adverse effects and severe adverse effects were near significant in both placebo and cholinergic augmentation groups. Death in the cholinergic augmentation groups was two-fold, but the total number of deaths was very small in both groups. It is therefore not a necessary concern as to when a patient with a stroke undergoes cholinergic augmentation in order to avoid severe adverse effects and a possible death. A recent study of 33 patients found that donepezil is safe when initiated within the 24 hours of a stroke onset [24]., thereby confirming that administration of acetylcholinesterase inhibitor after an acute stroke is safe and well tolerated.
This meta-analysis has some limitations. There are no detailed indicators that can be used to establish risk and benefit from the different patterns and severity of cognitive impairment after a stroke. First, the total number of trials included in the analysis was very small, with only two trials on cholinergic enhancement in patients with cognitive impairment after a stroke. Therefore, our meta-analysis can be more meaningful. The other trials were on cholinergic enhancement in patients with vascular dementia. Second, vascular dementia is heterogeneous because it groups together a broad category of patients with a variety of cerebrovascular diseases that include impaired cognitive function. In these five clinical trials, 61% to 70% of patients with vascular dementia experienced more than one transient ischemic attack or stroke, suggesting that there were a significant number of patients affected with post stroke cognitive impairment. However, the information of patients with post stroke cognitive impairment could not be obtained from included studies. Third, only ADAS-Cog scores and MMSE score changes were compared in the present meta-analysis. However, other evaluating methods for cognitive status of patients could not find from included studies. Fifth, the detailed information for the effect of short term treatment could not be obtained due to too low number of included studies.
In conclusion, our analysis of placebo-controlled studies suggests that acetylcholinesterase inhibitors are helpful for vascular dementia and post-stroke cognitive impairment. We demonstrate that cholinergic augmentation is well tolerated in patients with cognitive impairment and vascular dementia. Further longitudinal studies with a longer follow-up assessment are vital to further evaluate the efficacy of cholinesterase inhibitors in the early treatment of post-stroke cognitive impairment.
Supporting information
(DOC)
(DOCX)
Acknowledgments
The authors cordially thank Prof. Soo Joo Lee MD, Ph.D, and Prof. Jung Soo Pyo MD, Ph.D for consultation. They would also like to express their gratitude to study group for meta-analysis for their supports and collaborations.
Data Availability
All relevant data are within the Supporting Information file.
Funding Statement
The authors received no specific funding for this work.
References
- 1.Srikanth VK, Anderson JF, Donnan GA, Saling MM, Didus E, et al. (2004) Progressive dementia after first-ever stroke A community-based follow-up study. Neurology. 63:785–92. 10.1212/01.wnl.0000137042.01774.33 [DOI] [PubMed] [Google Scholar]
- 2.Pendlebury ST, Rothwell PM (2009) Prevalence, incidence, and factors associated with pre-stroke and post-stroke dementia: a systematic review and meta-analysis. Lancet Neurol. 8:1006–1018. 10.1016/S1474-4422(09)70236-4 [DOI] [PubMed] [Google Scholar]
- 3.Cramer SC (2018) Treatments to Promote Neural Repair after Stroke. J Stroke. 20:57–70. 10.5853/jos.2017.02796 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rasquin SM, Lodder J, Verhey FR (2005) Predictors of reversible mild cognitive impairment after stroke: a 2-year follow-up study. J Neurol Sci. 229–230:21–25. 10.1016/j.jns.2004.11.015 [DOI] [PubMed] [Google Scholar]
- 5.Berthier ML, Pulvermüller F, Dávila G, Casares NG, Gutiérrez A (2011) Drug therapy of post-stroke aphasia: a review of current evidence. Neuropsychol Rev. 21:302–317. 10.1007/s11065-011-9177-7 [DOI] [PubMed] [Google Scholar]
- 6.Kavirajan H, Schneider LS (2007) Efficacy and adverse effects of cholinesterase inhibitors and memantine in vascular dementia: a meta-analysis of randomised controlled trials. Lancet Neurol. 6:782–792. 10.1016/S1474-4422(07)70195-3 [DOI] [PubMed] [Google Scholar]
- 7.Berthier ML, Green C, Higueras C, Fernández I, Hinojosa J, et al. (2006) A randomized, placebo-controlled study of donepezil in post stroke aphasia. Neurology. 67:1687–1689. 10.1212/01.wnl.0000242626.69666.e2 [DOI] [PubMed] [Google Scholar]
- 8.Whyte EM, Lenze EJ, Butters M, Skidmore E, Koenig K, et al. (2008) An Open-Label Pilot Study of Acetylcholinesterase Inhibitors to Promote Functional Recovery in Elderly Cognitively Impaired Stroke Patients. Cerebrovasc Dis. 26:317–321. 10.1159/000149580 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Black S, Román GC, Geldmacher DS, Salloway S, Hecker J, et al. (2003) Efficacy and Tolerability of Donepezil in Vascular Dementia Positive Results of a 24-Week, Multicenter, International, Randomized, Placebo-Controlled Clinical Trial. Stroke. 34:2323–2330. 10.1161/01.STR.0000091396.95360.E1 [DOI] [PubMed] [Google Scholar]
- 10.Chang WH, Park YH, Ohn SH, Park CH, Lee PK, et al. (2011) Neural correlates of donepezil-induced cognitive improvement in patients with right hemisphere stroke: A pilot study. Neuropsychol Rehabil. 21:502–514. 10.1080/09602011.2011.582708 [DOI] [PubMed] [Google Scholar]
- 11.Moretti R, Torre P, Antonello RM, Cazzato G, Pizzolato G (2008) Different Responses to Rivastigmine in Subcortical Vascular Dementia and Multi-Infarct Dementia. Am J Alzheimers Dis Other Demen. 23:167–176. 10.1177/1533317507312558 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Narasimhalu K, Effendy S, Sim CH, Lee JM, Chen I, et al. (2010) A randomized controlled trial of rivastigmine in patients with cognitive impairment no dementia because of cerebrovascular disease. Acta Neurol Scand. 121:217–224. 10.1111/j.1600-0404.2009.01263.x [DOI] [PubMed] [Google Scholar]
- 13.Pratt RD (2002) Patient populations in clinical trials of the efficacy and tolerability of donepezil in patients with vascular dementia. J Neurol Sci. 203–204:57–65. 10.1016/s0022-510x(02)00266-6 [DOI] [PubMed] [Google Scholar]
- 14.Román GC, Salloway S, Black SE, Royall DR, Decarli C, at al. (2010) Randomized, Placebo-Controlled, Clinical Trial of Donepezil in Vascular Dementia Differential Effects by Hippocampal Size. Stroke. 41:1213–1221. 10.1161/STROKEAHA.109.570077 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wilkinson D, Doody R, Helme R, Taubman K, Mintzer J, et al. (2003) Donepezil in vascular dementia A randomized, placebo-controlled study. Neurology. 61:479–486. 10.1212/01.wnl.0000078943.50032.fc [DOI] [PubMed] [Google Scholar]
- 16.Rockwood K, Howlett SE, Hoffman D, Schindler R, Mitnitski A (2007) Clinical meaningfulness of Alzheimer’s Disease Assessment Scale–Cognitive subscale change in relation to goal attainment in patients on cholinesterase inhibitors. Alzheimers Dement. 13:1098–1106. 10.1016/j.jalz.2017.02.005 [DOI] [PubMed] [Google Scholar]
- 17.Román GC, Kalaria RN (2006) Vascular determinants of cholinergic deficits in Alzheimer disease and vascular dementia. Neurobiol Aging. 27:1769–1785. 10.1016/j.neurobiolaging.2005.10.004 [DOI] [PubMed] [Google Scholar]
- 18.Masanic CA, Bayley MT, VanReekum R, Simard M (2001) Open-label study of donepezil in traumatic brain injury. Arch Phys Med Rehabil. 82:896–901. 10.1053/apmr.2001.23833 [DOI] [PubMed] [Google Scholar]
- 19.Sarter M, Hasselmo ME, Bruno JP, Givens B (2005) Unraveling the attentional functions of cortical cholinergic inputs: Interactions between signal-driven and cognitive modulation of signal detection. Brain Res Brain Res Rev. 48:98–111. 10.1016/j.brainresrev.2004.08.006 [DOI] [PubMed] [Google Scholar]
- 20.Conner JM, Chiba AA, Tuszynski MH (2005) The basal forebrain cholinergic system is essential for cortical plasticity and functional recovery following brain injury. Neuron. 46:173–179. 10.1016/j.neuron.2005.03.003 [DOI] [PubMed] [Google Scholar]
- 21.Wang L, Conner JM, Nagahara AH, Tuszynski MH (2016) Rehabilitation drives enhancement of neuronal structure in functionally relevant neuronal subsets. Proc Natl Acad Sci U S A. 113:2750–2755. 10.1073/pnas.1514682113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nys GM, van Zandvoort MJ, de Kort PL, Jansen BP, Kappelle LJ, et al. (2005) Restrictions of the Mini-Mental State Examination in acute stroke. Arch Clin Neuropsychol. 20:623–629. 10.1016/j.acn.2005.04.001 [DOI] [PubMed] [Google Scholar]
- 23.Koyama T, Matsumoto K, Okuno T, Domen K(2005) A new method for predicting functional recovery of stroke patients with hemiplegia: logarithmic modelling. Clin Rehabil. 19:779–789. 10.1191/0269215505cr876oa [DOI] [PubMed] [Google Scholar]
- 24.Barrett KM, Brott TG, Brown RD Jr, Carter RE, Geske JR, et al. (2011) Enhancing recovery after acute ischemic stroke with donepezil as an adjuvant therapy to standard medical care: results of a phase IIA clinical trial. J Stroke Cerebrovasc Dis. 20:177–182. 10.1016/j.jstrokecerebrovasdis.2010.12.009 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOC)
(DOCX)
Data Availability Statement
All relevant data are within the Supporting Information file.