Abstract
Objectives:
Drug-coated balloons (DCB) and drug-eluting stents (DES) have significantly altered treatment paradigms for femoropopliteal lesions. We aimed to describe changes in practice patterns as a result of the infusion of these technologies into the treatment of peripheral arterial disease.
Methods:
We queried the Vascular Quality Initiative (VQI) registry from 2010–2017 for all peripheral vascular interventions (PVI) involving the superficial femoral artery (SFA) and/or the popliteal artery. Cases were divided into a PRE and a POST era with a cutoff of September 2016 when specific device identity was first recorded in VQI. For each artery, a primary treatment was identified as either plain balloon angioplasty (POBA), atherectomy, DCB, bare-metal stent (BMS), or DES. The relative distribution of primary treatments between the PRE and POST eras was evaluated, as were lesion characteristics associated with DCB and DES utilization and regional variability in the adoption of these new technologies.
Results:
Of 210,666 arteries in the dataset, 91,864 femoropopliteal arteries (across 74,842 procedures in 55,437 patients) were included. Each artery received 1.5 ± 0.6 treatments. Primary treatment utilization changed from 40% balloon angioplasty, 45% stenting, and 15% atherectomy in the PRE era to 22% POBA, 26% BMS, 8% atherectomy, 37% DCB, and 8% DES in the POST era (P < .001). 43% of arteries received a drug-containing device as a primary or adjunctive therapy and 1.3% received both a DCB and DES in the POST era. DCB utilization as the primary treatment was highest in lesions with length 10–19.9 cm (42%), TASC A-C lesions (38%), and lesions with mild to no calcification (38%). DES utilization was highest in lesions with length 20+ cm (12%), TASC D lesions (13%), and lesions with moderate to severe calcification (9%). The range of utilization across 18 regions was 12–40% for DCB and 1–14% for DES. Regional variability was greater for DES (SD 4% vs. mean 8%) than for DCB (SD 7% vs. mean 29%).
Conclusions:
There has been a rapid dissemination of DCB and DES technology in the femoropopliteal vessels, with nearly half of arteries receiving a drug-containing therapy in modern practice. DCBs are most utilized in medium-length, minimally calcified lesions, while DESs are most utilized in longer, more heavily calcified lesions. There is significant regional variability in adoption, especially with DES.
Keywords: Drug-coated balloons, drug-eluting stents, trends, femoropopliteal
Table of Contents Summary
This Vascular Quality Initiative (VQI) study of 91,864 femoropopliteal endovascular procedures found that 43% were treated recently with drug coated balloons (DCB) or drug coated stents (DES). DES use was less prevalent and more variable than DCB use.
Introduction
Peripheral arterial disease (PAD) is a prevalent manifestation of atherosclerotic disease, affecting one in five elderly Americans by the age of 80 years.1 Treatment options include both open surgical and endovascular interventions at all anatomical levels. While endovascular interventions for aortoiliac disease have shown excellent durability that rivals that of surgical bypass, femoropopliteal disease, involving either the superficial femoral artery (SFA) or the popliteal artery, continues to pose a challenge for endovascular interventions.2
In the past decade, endovascular therapy has been widely adopted as an alternative to open bypass surgery. Endovascular interventions have achieved reasonable patency rates even in the more complex TransAtlantic InterSociety (TASC) II D occlusive lesions, and are particularly attractive in those patients with critical limb ischemia who are perceived to be poor open surgical candidates.4 Percutaneous therapies continue to evolve with various options currently available, including balloon angioplasty, stents, and atherectomy devices. Although plain balloon angioplasty (POBA) and stenting have long been considered the standard of practice,2, 3 both are limited by high restenosis rates.5, 6 Moreover, stent fractures and in-stent restenosis rates7 have led to increasing caution in the use of stents in the femoropopliteal segment and the search for novel therapies which improve upon the patency limitations of balloon angioplasty while minimizing the need for permanent stent implantation. Drug-coated balloons (DCBs) and drug-eluting stents (DESs) have emerged as newer and more sophisticated endovascular therapies with multiple randomized controlled trials demonstrating their superior patency rates compared to POBA.7–9 Both DCB and DES technologies additionally show promise in improving the cost-effectiveness of femoropopliteal interventions, with literature suggesting that the higher upfront cost is offset by improvements in patency and subsequently decreased need for expensive re-interventions.10, 11
As both devices are relatively new, little is known about how these technologies are utilized in mainstream practice and how they have influenced treatment patterns in patients with infrainguinal occlusive disease. This study aims to use a large national database to study trends over time in management strategies utilized in the treatment of femoropopliteal arterial disease and to examine the specific effect of infusion of DCB and DES technology into the treatment paradigm for these patients.
Methods
As this study involved a de-identified data source, Institutional Review Board approval and informed consent were not required. We utilized data from the Vascular Quality Initiative (VQI) registry’s Peripheral Vascular Intervention (PVI) dataset between January 2010 and November 2017 to conduct this study. The VQI is a collaboration of regional quality groups governed by the SVS Patient Safety Organization with approximately 400 enrolled sites to date. The VQI registry stores data on vascular surgical procedures performed at each participating institution. Patient-level clinical data is collected by each institution and uploaded to VQI.12 We queried all PVI procedures involving treatment of the femoropopliteal arteries, including those in which other arterial beds were treated in the same setting. Patients undergoing open surgical bypass were excluded, as were patients in whom no treatment was performed due to technical failure.
The VQI dataset allows for entry of up to four arteries per procedure and up to three treatments per artery. The data was reformatted such that all analysis was performed with the treated artery, rather than the patient or the procedure, as the individual unit of study. Only the SFA and popliteal artery were included for study. All treatments for each artery were included. Treatments were categorized as plain balloon angioplasty (POBA), bare-metal stent (BMS) placement, drug-coated balloon (DCB) angioplasty, drug-eluting stent (DES) placement, or atherectomy. Additionally, a single “primary” treatment was identified for each procedure. As the order in which treatments were performed is not available in VQI data, the primary treatment was defined based on a hierarchy of DES > DCB > BMS > atherectomy > POBA. For example, a patient who underwent DCB angioplasty, POBA, and atherectomy would have DCB classified as the primary treatment, regardless of the order in which these treatments were documented.
Utilization was measured by the percentage of interventions which involved DCB and DES treatments. DCBs and DESs were first identifiable in the VQI dataset in September 2016, when an update to the entry form allowed for labeling of the specific device (balloon or stent) used in each treatment. Prior to this, DCB was indistinguishable from POBA and DES indistinguishable from BMS. Thus, a pre-post analysis was conducted using September 2016 as a cutoff date. Utilization of POBA, BMS, DCB, DES, and atherectomy were examined in the POST era as compared to overall utilization of balloon angioplasty, stenting, and atherectomy in the PRE era, with the caveat that DCB and DES use would not be specifically identifiable in the PRE era.
After examination of trends, the PRE group was excluded from further analysis such that all treatments were identifiable. Interventions from the POST era were then studied for factors which influenced use of DCB or DES. First, the SFA and popliteal arteries were studied as separate subgroups. Second, lesion characteristics, including the length of the treated segment, TASC classification, and subjective rating of calcification (in VQI documented as none, focal, mild, moderate, or severe), were studied for their influence on DCB or DES use. Finally, regional variation was studied by determining the overall DCB and DES utilization in each of 18 de-identified geographic practice regions.
Stata 15.1 (StataCorp, College Station, TX) was used for all statistical analysis. Time trends were initially studied visually, after which the pre/post data was evaluated by a Chi-squared test. The Chi-squared test was also used to study the effect of treated vessel and most lesion characteristics on DCB and DES use. Lesion length was first studied as a continuous variable, tested by analysis of variance (ANOVA) and pairwise t-tests, and then as a categorical variable with lesions divided into three categories (less than 10 cm, 10–19.9 cm, and greater than 20 cm) and analyzed by Chi-squared test. Regional variability was first depicted visually, then confirmed objectively. As there is no accepted standard by which to objectively measure regional variability, the presence of variability was first confirmed by Chi-squared test, after which the degree of variability was compared between treatment modalities in two ways. First, summary statistics on the regional utilization of each treatment were evaluated, using the standard deviation relative to the mean as a surrogate of the magnitude of varability. A second analysis was conducted in which each treatment was used as the outcome in a mixed-effects logistic regression model where, adjusted for treated artery and TASC grade, the variance in beta-estimates introduced by region ID was examined as an indicator of variability.
Results
In the years studied, the VQI dataset contained 125,988 procedures among 93,200 patients with a total of 210,666 treated arteries. Of these, 91,864 (44%) femoropopliteal arterial interventions were documented, as part of 74,842 procedures in 55,437 patients. There were 58,516 SFAs (64%) and 29,683 popliteal arteries (32%) treated, with 3,665 (4%) labeled as both. Annual volume of femoropopliteal interventions increased in the VQI from 1,359 in 2010 to 19,235 in 2016 (the last complete year of available data).
Using the cutoff date of September 2016, the PRE era contained 72,192 interventions, with the remaining 19,672 cases in the POST era. Baseline characteristics stratified by primary treatment modality are shown in Table I including only patients in the POST era. All differences were statistically but not clinically significant. Overall utilization of each treatment is shown in Figure 1. Most notably, a total of 45% of arteries in the POST era were treated with either a DCB or a DES as the primary intervention (37% DCB, 8% DES). Also notable is the decline in overall stenting rates between eras, with 34% of arteries undergoing stenting in the POST era (26% BMS, 8% DES) compared to 45% in the PRE era. In the POST era, 6.3% of arteries received both a DCB and a stent, including a 1.3% utilization of both DCB and DES in the same artery. Atherectomy was used as a primary treatment in 33% and in conjunction with BMS, DCB, or DES in the remainder.
Table 1:
Baseline characteristics by treatment type. BMI: body mass index; DM: diabetes mellitus; CAD: coronary artery disease; CHF: congestive heart failure; COPD: chronic obstructive pulmonary disease; CVA: cerebrovascular accident; PAD: peripheral arterial disease.
| POBA N = 4134 | Atherectomy N = 1456 | BMS N = 4826 | DCB N = 6922 | DES N = 1541 | P-value | |
|---|---|---|---|---|---|---|
| Age | 69.9 ± 11.7 | 70.1 ± 10.7 | 69.1 ± 11.4 | 68.4 ± 11.1 | 68.8 ± 11.0 | <.001 |
| Female | 46.8% | 38.9% | 38.7% | 42.7% | 35.6% | <.001 |
| BMI (kg/m2) | 27.8 ± 6.5 | 28.3 ± 6.3 | 27.9 ± 6.3 | 28.3 ± 6.2 | 28.2 ± 5.9 | <.001 |
| Hypertension | 89.4% | 90.6% | 88.1% | 90.7% | 89.4% | <.001 |
| DM | 60.7% | 59.1% | 53.4% | 56.4% | 51.8% | <.001 |
| CAD | 37.1% | 34.6% | 33.7% | 37.6% | 41.4% | <.001 |
| CHF | 25.8% | 23.4% | 20.3% | 20.0% | 19.4% | <.001 |
| COPD | 26.1% | 21.6% | 27.4% | 26.6% | 27.9% | <.001 |
| Renal failure | ||||||
| CVA | 17.9% | 14.8% | 14.4% | 14.5% | 14.0% | <.001 |
| Prior procedure for PAD | 54.7% | 57.8% | 50.9% | 57.2% | 51.5% | <.001 |
Figure 1:
Utilization of each technology in the PRE and POST eras.
A subgroup analysis of interventions by treated vessel is shown in Figure 2. Though overall utilization patterns differed significantly between the SFA and popliteal arteries (P < .0001), DCB utilization was similar between the SFA and popliteal artery at 36% and 37%, respectively. POBA, on the other hand, was significantly more common in the popliteal artery (32%) than in the SFA (19%). Stent utilization (both BMS and DES) was lower in the popliteal artery (18% and 3%, respectively) than in the SFA (28% and 10%, respectively). Atherectomy was used as a primary treatment more commonly in the popliteal artery (10%) than in the SFA (7%).
Figure 2:
Relative utilization of each technology in the superficial femoral artery and popliteal artery.
Mean lesion lengths for each of the primary treatments is shown in Figure 3A (P < .001 for overall comparison and for all pairwise comparisons). DES was utilized in the longest lesions (mean 16.1 cm) and was utilized to treat significantly longer lesions than BMS (15.2 cm). Likewise, DCB (13.2 cm) was used to treat significantly longer lesions than POBA (11.0 cm), but shorter lesions than those treated with stents. When lesions were stratified into three length categories, utilization varied significantly between these categories (Figure 3B; P < .0001). DCBs were most commonly used in lesions 10–19.9 cm in length (43%), and less commonly in shorter (36%) or longer lesions (31%). DES utilization was highest in lesions at least 20 cm in length (12%) and lowest in those under 10 cm in length (6%).
Figure 3:
Utilization by lesion length, by mean lesion length for each technology (A) and by relative utilization based on lesion length (B).
Figure 4 shows treatment utilization by TASC classification of the index lesion. Stents were used most readily in TASC D lesions, with both BMS (33%) and DES (13%) having their highest utilization in this group. DCB use was fairly consistent at 38–39% for TASC A, B and C lesions and was lowest in TASC D lesions (30%). Most notably, with increasing TASC classification there was a decreasing reliance on POBA alone as the primary treatment. POBA utilization decreased progressively from 26% in TASC A lesions to 15% in TASC D lesions.
Figure 4:
Utilization by TASC classification.
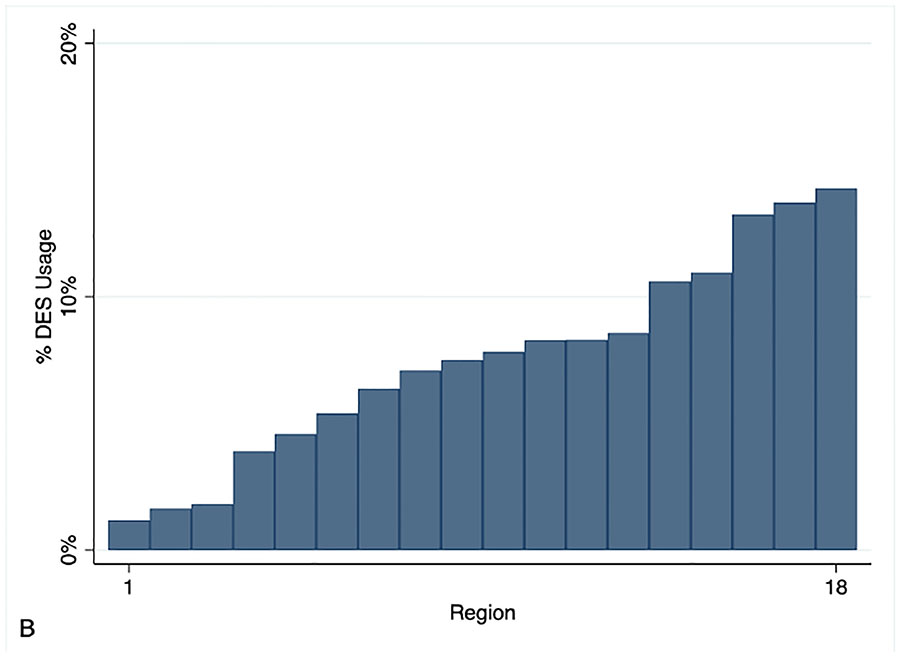
Among 18 de-identified geographic regions in the VQI dataset, regional utilization of each treatment modality is shown in Figure 5 and shows greater variation in use of DES. This was quantitatively studied in two methods as noted above. Relative to each treatment’s mean utilization, standard deviation was highest with atherectomy (SD 69% of mean), followed by DES (54%), POBA (30%), DCB (26%), and BMS (17%), indicating that regional variability was highest with atherectomy and lowest with BMS utilization. In the second method, mixed-effects logistic regression models were constructed to derive the variance in beta-estimates imposed by regional variability (higher variance indicates higher variability in utilization across regions). The variances by this method followed a similar pattern: highest with atherectomy (1.18), followed by DES (1.07), DCB (0.22), POBA (0.13), and BMS (0.07). Both methods indicated that regional variability is markedly higher for DES utilization than for DCB utilization.
Figure 5:
Regional utilization of DCB and DES technologies. Each de-identified region is shown as a separate bar.
Discussion
Utilizing the VQI registry from 2010 to 2017, this study describes changes in treatment patterns of femoropopliteal arterial disease. Since initial approval of DESs in 2012 and DCBs in 2014, use of DCB and DES technology has undergone rapid adoption in contemporary practice. While DCBs are similarly utilized in the SFA and popliteal artery, rates of overall stent use, including DES, are lower in the popliteal artery. DCB utilization as primary treatment is highest in medium-length, TASC A-C, and minimally calcified lesions. In contrast, DES use is more common in longer and more heavily calcified TASC D lesions. Regional variability in rates of adoption appears to be significant, specifically with DES.
High restenosis rates of standard balloon angioplasty and concern for in-stent restenosis and stent kinking and fracture5–7 highlight the need for improved endovascular technology in the femoropopliteal space. Several small-scale randomized controlled trials have demonstrated the superior long-term patency of paclitaxel-based technologies.7–9 In the IN.PACT SFA trial, patency rates remained significantly higher among patients with femoropopliteal lesions treated with DCBs as compared with POBA even after three years (69.5% vs. 45.1%).9, 13 The Zilver PTX randomized trial has similarly demonstrated superiority of DES over percutaneous balloon angioplasty with significantly higher patency rates and >40% relative risk reduction for restenosis maintained over 5 years.8, 14
Our results show rapid adoption rates of drug-containing therapy in the United States, with almost half of diseased femoropopliteal vessels receiving either a DCB or DES as primary therapy. DCBs have particularly gained widespread momentum in treating femoropopliteal lesions and have been used in Europe since 2007.15 They can deliver antiproliferative drugs to diseased vessels and offer improved patency rates without the need for a permanent stent. In recent years, DCBs were most commonly used as primary treatment, exceeding rates of classic POBA or the use of BMS or DES technologies.
Although primary utilization rates are trending upward, DESs appear to be used less commonly than DCBs despite results demonstrating their favorable long-term patency.7, 14 We attribute this to a growing trend favoring selective stenting and avoidance of stent placement where possible. The dynamic physical forces to which the femoropopliteal segment is subjected to during movement16 can increase the risk of stent fracture,17 which still remains challenging to treat.18 While high rates of stent fractures were observed (18% at 6 months) in the earlier SIROCCO trials comparing sirolimus-eluting stent to BMS,19 much lower rates were reported in the more recent Zilver PTX trial (0.9% at 12 months). This is likely attributable partly to newer, more flexible stent designs and partly to treatment of shorter lesions.20 With further advances in stent technology, it is possible that improvements in stent fracture, kinking, and restenosis rates could lead to a resurgence in the willingness to primarily stent longer or more severe lesions or those extending across the flexion zone of the knee joint.
Long femoropopliteal lesions still present a challenge in the world of endovascular therapy. There appears to be a greater use of DCBs and DESs in longer lesions than standard POBA and BMS. Specifically, DES utilization was highest in heavily calcified, TASC D lesions with lengths greater than 20 cm and DCBs were most commonly used in lesions 10–19.9 cm in length. DCBs, as with POBA, appear not to be commonly used in heavily calcified lesions, likely because of suboptimal post-angioplasty results which necessitate stent placement. Moreover, calcification can serve as a barrier for optimal drug delivery and absorption in DCBs, interfering with their ability to suppress restenosis and leading to earlier clinical failure.21, 22
It is notable that 6.3% of arteries in recent years were treated with both a DCB and a stent, including some cases of DCB and DES use. Such treatment strategies add to the cost of treatment significantly. Atherectomy similarly increases the cost of treatment, especially when combined with DCB and DES use. Further research is warranted into the role for these combination therapies and the circumstances under which they offer true clinical benefit and cost-effectiveness.
There was a high degree of regional variability in the different treatment modalities utilized. DES has particularly demonstrated great variation across regions. Further investigation into the reasons for this variability is outside the scope of a study utilizing the VQI database, as region identity is unknown. Different regions may contain different patient populations, with a unique mix of clinical and anatomic factors which lend themselves to one treatment paradigm over another. In addition, as a range of specialists (including but not limited to vascular surgeons, interventional cardiologists, and interventional radiologists) offer endovascular lower extremity interventions, each region may contain a different provider mix with differing treatment bias. Differences in marketing pressure and relative reimbursement of each of the various treatments may also contribute to the observed differences. Drug-containing therapies seem to be cost-effective when longer patency is taken into account, and a shift to DCB use as a primary strategy is estimated to lead to an annual cost reduction of $250 million to Medicare.23 Higher upfront costs, however, may create a barrier to adoption in some regions and in specific institutions within them.
There are several limitations to this study. It is retrospective with all the inherent selection biases related to such study design. VQI data is self-reported by each institution, so definitions, accuracy, and completeness of data may vary across centers. Data may also not be entirely reflective of nationwide trends as it is limited to only the centers participating in VQI. As an example, physician-owned angiography suites, which are known to be especially cost-conscious due to current reimbursement models, are likely under-represented in VQI. Another drawback is the inability to identify the specific treatment algorithms for each case, as treatments could have been entered in any order. In particular, when both balloon angioplasty and stenting were documented, it was not possible to identify which stents were placed primarily and which were bailout stents for residual stenosis or dissection. Finally, as previously mentioned, DCB and DES utilization prior to September 2016 was not able to be identified. Despite these limitations, the VQI is currently the only registry in the world collecting specific device information including linkage to the global universal device identifier, and this study is the first to leverage such information to study changes in management strategies used in the treatment of femoropopliteal arterial disease and to examine the infusion of drug-containing technologies into lower extremity interventions.
Conclusions
There has been a rapid dissemination of DCB and DES technology in the treatment of femoropopliteal disease, with nearly half of arteries receiving a drug-containing therapy in current practice. DCBs are mostly utilized in medium-length, minimally calcified lesions, while DESs are most used in longer, more complex, heavily calcified lesions. Regional variability in rates of adoption appears to be significant, especially with DES.
ARTICLE HIGHLIGHTS.
Type of Research:
retrospective analysis of prospectively collected Vascular Quality Initiative (VQI) data
Key Findings:
In this study of 91,864 femoropopliteal endovascular procedures 43% were treated recently with drug coated balloons (DCB) or drug coated stents (DES). DES use was less prevalent and more variable than DCB use.
Take home Message:
DCB and DES technologies have rapidly disseminated in the femoropopliteal space. Both, but especially DESs, are used in varying rates across the nation.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
This work was presented as a plenary presentation at the 32nd Annual Meeting of the Eastern Vascular Society on September 7, 2018 in Washington, DC.
References
- 1.Criqui MH, Aboyans V. Epidemiology of peripheral artery disease. Circ Res. 2015;116(9):1509–26. [DOI] [PubMed] [Google Scholar]
- 2.Conte MS, Pomposelli FB. Society for Vascular Surgery Practice guidelines for atherosclerotic occlusive disease of the lower extremities management of asymptomatic disease and claudication. Introduction. J Vasc Surg. 2015;61(3 Suppl):1S. [DOI] [PubMed] [Google Scholar]
- 3.Norgren L, Hiatt WR, Dormandy JA, Nehler MR, Harris KA, Fowkes FG, et al. Inter-Society Consensus for the Management of Peripheral Arterial Disease (TASC II). J Vasc Surg. 2007;45 Suppl S:S5–67. [DOI] [PubMed] [Google Scholar]
- 4.Baril DT, Chaer RA, Rhee RY, Makaroun MS, Marone LK. Endovascular interventions for TASC II D femoropopliteal lesions. J Vasc Surg. 2010;51(6):1406–12. [DOI] [PubMed] [Google Scholar]
- 5.Krankenberg H, Schluter M, Steinkamp HJ, Burgelin K, Scheinert D, Schulte KL, et al. Nitinol stent implantation versus percutaneous transluminal angioplasty in superficial femoral artery lesions up to 10 cm in length: the femoral artery stenting trial (FAST). Circulation. 2007;116(3):285–92. [DOI] [PubMed] [Google Scholar]
- 6.Laird JR, Katzen BT, Scheinert D, Lammer J, Carpenter J, Buchbinder M, et al. Nitinol stent implantation versus balloon angioplasty for lesions in the superficial femoral artery and proximal popliteal artery: twelve-month results from the RESILIENT randomized trial. Circ Cardiovasc Interv. 2010;3(3):267–76. [DOI] [PubMed] [Google Scholar]
- 7.Scheinert D, Duda S, Zeller T, Krankenberg H, Ricke J, Bosiers M, et al. The LEVANT I (Lutonix paclitaxel-coated balloon for the prevention of femoropopliteal restenosis) trial for femoropopliteal revascularization: first-in-human randomized trial of low-dose drug-coated balloon versus uncoated balloon angioplasty. JACC Cardiovasc Interv. 2014;7(1):10–9. [DOI] [PubMed] [Google Scholar]
- 8.Dake MD, Ansel GM, Jaff MR, Ohki T, Saxon RR, Smouse HB, et al. Paclitaxel-eluting stents show superiority to balloon angioplasty and bare metal stents in femoropopliteal disease: twelve-month Zilver PTX randomized study results. Circ Cardiovasc Interv. 2011;4(5):495–504. [DOI] [PubMed] [Google Scholar]
- 9.Tepe G, Laird J, Schneider P, Brodmann M, Krishnan P, Micari A, et al. Drug-coated balloon versus standard percutaneous transluminal angioplasty for the treatment of superficial femoral and popliteal peripheral artery disease: 12-month results from the IN.PACT SFA randomized trial. Circulation. 2015;131(5):495–502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Salisbury AC, Li H, Vilain KR, Jaff MR, Schneider PA, Laird JR, et al. Cost-Effectiveness of Endovascular Femoropopliteal Intervention Using Drug-Coated Balloons Versus Standard Percutaneous Transluminal Angioplasty: Results From the IN.PACT SFA II Trial. JACC Cardiovasc Interv. 2016;9(22):2343–52. [DOI] [PubMed] [Google Scholar]
- 11.Sridharan ND, Boitet A, Smith K, Noorbakhsh K, Avgerinos E, Eslami MH, et al. Cost-effectiveness analysis of drug-coated therapies in the superficial femoral artery. J Vasc Surg. 2018;67(1):343–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.“Overview & Benefits.” Vascular Study Group of New England. Available at: https://www.vqi.org/overview-benefits/. [January 14, 2018].
- 13.Schneider PA, Laird JR, Tepe G, Brodmann M, Zeller T, Scheinert D, et al. Treatment Effect of Drug-Coated Balloons Is Durable to 3 Years in the Femoropopliteal Arteries: Long- Term Results of the IN.PACT SFA Randomized Trial. Circ Cardiovasc Interv. 2018;11(1):e005891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dake MD, Ansel GM, Jaff MR, Ohki T, Saxon RR, Smouse HB, et al. Durable Clinical Effectiveness With Paclitaxel-Eluting Stents in the Femoropopliteal Artery: 5-Year Results of the Zilver PTX Randomized Trial. Circulation. 2016;133(15):1472–83; discussion 83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Scheinert D, Schmidt A, Zeller T, Muller-Hulsbeck S, Sixt S, Schroder H, et al. German Center Subanalysis of the LEVANT 2 Global Randomized Study of the Lutonix Drug-Coated Balloon in the Treatment of Femoropopliteal Occlusive Disease. J Endovasc Ther. 2016;23(3):409–16. [DOI] [PubMed] [Google Scholar]
- 16.Klein AJ, Chen SJ, Messenger JC, Hansgen AR, Plomondon ME, Carroll JD, et al. Quantitative assessment of the conformational change in the femoropopliteal artery with leg movement. Catheter Cardiovasc Interv. 2009;74(5):787–98. [DOI] [PubMed] [Google Scholar]
- 17.Scheinert D, Scheinert S, Sax J, Piorkowski C, Braunlich S, Ulrich M, et al. Prevalence and clinical impact of stent fractures after femoropopliteal stenting. J Am Coll Cardiol. 2005;45(2):312–5. [DOI] [PubMed] [Google Scholar]
- 18.Laird JR, Yeo KK. The treatment of femoropopliteal in-stent restenosis: back to the future. J Am Coll Cardiol. 2012;59(1):24–5. [DOI] [PubMed] [Google Scholar]
- 19.Duda SH, Bosiers M, Lammer J, Scheinert D, Zeller T, Oliva V, et al. Drug-eluting and bare nitinol stents for the treatment of atherosclerotic lesions in the superficial femoral artery: long-term results from the SIROCCO trial. J Endovasc Ther. 2006;13(6):701–10. [DOI] [PubMed] [Google Scholar]
- 20.Litsky J, Chanda A, Stilp E, Lansky A, Mena C. Critical evaluation of stents in the peripheral arterial disease of the superficial femoral artery - focus on the paclitaxel eluting stent. Med Devices (Auckl). 2014;7:149–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Schmidt A, Piorkowski M, Gorner H, Steiner S, Bausback Y, Scheinert S, et al. Drug- Coated Balloons for Complex Femoropopliteal Lesions: 2-Year Results of a Real-World Registry. JACC Cardiovasc Interv. 2016;9(7):715–24. [DOI] [PubMed] [Google Scholar]
- 22.Fanelli F, Cannavale A, Gazzetti M, Lucatelli P, Wlderk A, Cirelli C, et al. Calcium burden assessment and impact on drug-eluting balloons in peripheral arterial disease. Cardiovasc Intervent Radiol. 2014;37(4):898–907. [DOI] [PubMed] [Google Scholar]
- 23.Cortese B, Granada JF, Scheller B, Schneider PA, Tepe G, Scheinert D, et al. Drug-coated balloon treatment for lower extremity vascular disease intervention: an international positioning document. Eur Heart J. 2016;37(14):1096–103. [DOI] [PubMed] [Google Scholar]