Abstract
Burnet postulated that the diversity of T-cell receptors (TCRs) allows T cells to protect against the development of cancers that display antigens with a similar, seemingly endless diversity. To test this hypothesis, we developed a strategy in which a single breeding pair of mice gives rise to four groups of sibling mice. Three of the four groups had a similar number of CD8+ T cells, but TCR diversity was either broad, significantly reduced, or absent when expressing only one type of TCR. The fourth group had no T cells. All mice shared the same housing, and therefore, their microbial environment was similar. Only slight differences in the intestinal flora were observed under these conditions. An undisturbed broad TCR repertoire was required for the rejection of inoculated cancers displaying the natural antigenic heterogeneity of primary tumors, whereas even one type of TCR was sufficient to protect against artificial cancers stably expressing cognate antigens. The three groups of mice with limited or no TCR repertoire showed an increased risk to developing primary tumors after chemical induction. However, the risk of early death or morbidity in these cohorts of mice was significantly higher than in mice with a diverse TCR repertoire, and it remains unknown whether mice with reduced TCR diversity, who died early without cancer, would have developed tumors with higher, lower, or equal probability after induction. Together, TCR diversity seems crucial to overcome the natural genetic instability of cancers and their antigenic heterogeneity, which impacts on the design of cellular therapies.
Keywords: TCR diversity, Cancer immune surveillance, Competing risk, Tumor heterogeneity, Neoantigens
INTRODUCTION
As is the case for T-cell receptors (TCRs)(1), tumor-specific antigens also have a seemingly endless diversity (2-4). These tumor-specific antigens are usually referred to as being unique because most of them are individually distinct, even for cancers of the similar histological type and when arising in genetically identical hosts. This remarkable diversity was discovered in mice using tumor cell transplantation assays and cytolytic T-cell clones (reviewed in (5)). Unique antigens result from somatic tumor-specific mutations due to non-synonymous single nucleotide substitutions (6) and are now usually referred to as neoantigens. Neoantigens are also widely found on human cancers (reviewed in (7)).
Macfarlane Burnet has noted that the diversity of unique cancer antigens seems to match the diversity of specificities of T-cell immunity (3,8). Lewis Thomas (9) and Macfarlane Burnet (10), therefore, suggested that the primary reason for development of thymus-dependent T cell–mediated immunity during the evolution of vertebrates was defense against the development of cancer. Adaptive T-cell immunity can, indeed, prevent humans from dying of virally induced cancers (11). For some virally induced cancers, such as those associated with HPV, this protection comes primarily from resistance to infection. However, a different mechanism protects humans against developing EBV-related cancers. About 95% of humans are latently EBV-infected, and the development of primary EBV-related cancers is prevented by EBV-specific T cells continuously surveilling and eliminating incipient EBV-transformed cells (11), as suggested by the immune surveillance hypothesis of Burnet and Thomas. This is the reason EBV-induced cancers are referred to as the “flag-ship of immune surveillance” (12). Although many experiments have examined T cell–mediated cancer surveillance (13,14), the question of whether TCR diversity, and also how much TCR diversity, is needed to protect against primary cancer development remains unanswered.
It is reported that T cells can carry out their function independent of their TCRs’ specificity, i.e., in a non-cognate, not antigen-specific manner (reviewed in (15)). Such non-cognate T cells can control infection, cause severe autoimmunity, kill cancer cells, control T-cell memory, and eradicate large established tumors (16) or are activated to produce IFNγ (17). IFNγ has been implicated to prevent cancer development (14,18), the outgrowth of latent cancers (19), and vascular perfusion once tumors are established (20). Thus, activation of T cells independent of their cognate antigen may occur by various mechanisms and is influenced by different conditions, e.g., the gut microbiome (21-24).
Therefore, we examined the role of TCR diversity in protecting against cancer development after cancer cell inoculation. We also induced primary cancers with 3’-methylcholanthrene (MCA) because this carcinogen had been used by most classical experiments examining the immune surveillance theory (reviewed in (5)). We conducted experiments on cancer immune surveillance in a familial full-sibling setting in which the experimental groups differed in TCR diversity but were derived from the same set of parents and kept as littermates sharing their microbial environment.
MATERIALS AND METHODS
Mice
C57BL/6 and Rag−/− (B6.129S7-Rag1tm1Mom) mice were purchased from the Jackson Laboratory. OT-IxRag−/− mice (B6.129S7-Rag1tm1MomTg(TcraTcrb)1100Mjb) were generated by crossing Rag−/− and TCR OT-I-transgenic mice (provided by Matthew Mescher, University of Minnesota, Minneapolis, MN) for at least 20 generations. 1D9xRag−/− mice (B6.129S7-Rag1tm1Mom Tg(MP71–1D9)1KsHs) were generated in our laboratory (25). Mice transgenic for TCR-2C, which is specific for the peptide SIYRYYGL (SIY) presented on Kb (26), were obtained from D. Loh (Washington University, St. Louis, MO). Mice were backcrossed for over 20 generations with C57BL/6 mice and bred with Rag−/− mice to generate 2CxRag−/− (B6.129S7-Rag1tm1Mom Tg(Tcra2C,Tcrb2C)1Dlo/KsHs). To minimize differences in genetic lineage (27), microbial flora (28), and housing conditions, we made use of littermates (29,30) to study tumor development after MCA injection (as described in the “Tumor induction” section). A single cross between a C57BL/6 and a 2CxRag−/− mouse generated the heterozygous female parent that was bred with a male Rag−/− to generate siblings that made up mice of the 4 groups in this study (Figure 1A). Upon weaning, mice were separated according to gender, ear-tagged, and genotyped according to presence of TCR-2C–expressing CD8+ T cells and B cells by flow cytometry. All mice were maintained in a specific pathogen-free (SPF)-facility at the University of Chicago. The Institutional Animal Care and Use Committee at the University of Chicago approved all animal experiments, and all experiments were performed to conform to the relevant regulatory standards.
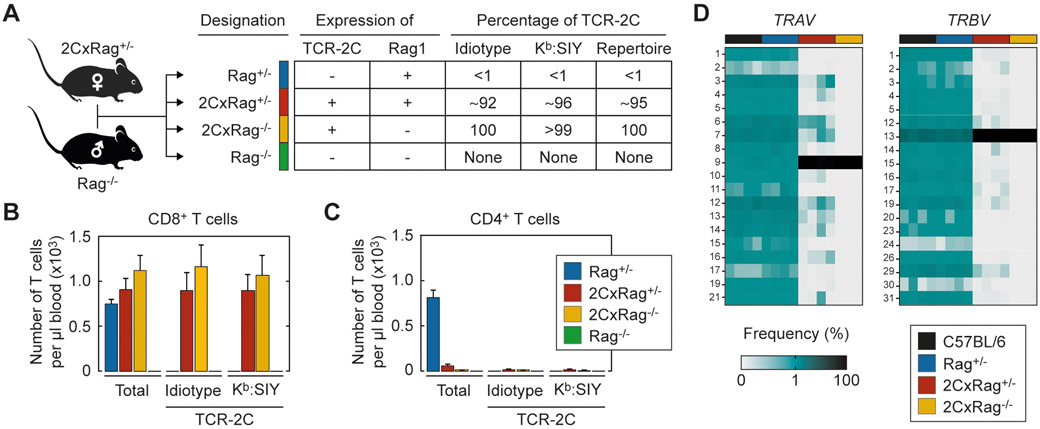
Figure 1. Derivation and description of the four experimental groups having different TCR diversities.
(A) Mice in the 4 experimental groups are all siblings generated from a C57BL/6 breeding of a female 2CxRag+/− and a male Rag−/− mouse. The alternating combination of Rag1 wild-type and knockout alleles and the transgene encoding for TCR-2C results in a varying percentage of TCR-2C-expressing T cells in the peripheral blood of mice in the 4 groups as determined by staining with a TCR-2C-specific antibody (1B2, Idiotype) or H-2Kb:SIY tetramer (see also B, C). The percentage of TCR-2C-expressing T cells in the T cell repertoire of the mice of the 4 groups was calculated based on TCR repertoire analysis (see also D). (B) CD8+ or (C) CD4+ T cells from the peripheral blood of indicated mice were analyzed in numbers and for expression of TCR-2C using 1B2 antibodies or H-2Kb:SIY tetramer. Bars show mean ± standard deviation. Rag+/− (n=9), 2CxRag+/− (n=5), 2CxRag−/− (n=9), Rag−/− (n=6). (D) TCR repertoire analysis was performed on splenic CD8+ T cells to determine the frequency of functional TRAV and TRBV genes. The sum of values in each column equals 100%. C57BL/6 (B6, n=4), Rag+/− (n=4), 2CxRag+/− (n=4), 2CxRag−/− (n=3).
Cells
MC57 is an MCA-induced, C57BL/6-derived fibrosarcoma [provided by Pamela Ohashi (University of Toronto), with permission of Hans Hengartner (University Hospital Zurich)]. Its transfectants MC57-SIY and MC57-mp68 were generated in our laboratory and have been described (25). The tumor 8101 originated in a UV-treated C57BL/6 mouse and was generated in our laboratory (31). 8101-bulk is a primary tumor cell culture generated from approximately 20 fragments (1–2 mm in size) of the autochthonous 8101 cancer. 8101-bulk was only minimally expanded, not cloned, or otherwise modified. The re-isolates #1-#3 were generated from fragments of tumors that grew for 50–60 days in 1D9xRag−/− mice after transplantation of 8101-bulk. The clonal mp68+ tumor cell line (8101-clone) was generated in our laboratory and has been described (31). Clones derived from 8101-bulk were generated by limiting dilution. All tumor cell lines were maintained in DMEM supplemented with 10% FCS (Gemini Bio-Products). Unless otherwise indicated, cell culture reagents were purchased from Invitrogen (Life Technologies). Before use, tumor cell lines were authenticated by sequencing and/or coculture with antigen-specific T cells (generated as described in the “TCR gene transfer and T-cell analysis” section). Plat-E packaging cells (32) were shortly passaged after thawing of the initial frozen stock to generate master cell banks. Plat-E working batches were cultured as described (33), passaged no longer than 3 months, and authenticated by cellular morphology. All cell cultures were checked for presence of mycoplasma (Lonza) every 3 months.
Tumor induction
Tumors were induced in 2–6–month-old mice by subcutaneous injection of either 25 μg of MCA dissolved in 250 μL sesame oil (both Sigma, St Louis, MO) or by transplanting 2x107 tumor cells under the shaved back skin. MCA is a carcinogen commonly used for examining immune surveillance (13,14,34-46). Mice were monitored at least three times a week for health status and tumor development. The size of lesions was measured along 3 orthogonal axes (a, b, and c) every 3 to 4 days using a caliper, and tumor volume was calculated as abc/2 (47). Mice having tumors with a volume of 250 mm3 or larger at the site of injection were considered as having developed tumors. Mice were sacrificed when moribund or when tumors reached 1.5 cm3 in volume. All mice whether sacrificed or found dead received a complete necropsy.
TCR gene transfer and T-cell analysis
1x106 Plat-E packaging cells were transiently transfected with MP71–1D9 (25), virus supernatant was harvested, and 1 mL was used for transduction of 1x106 T cells isolated from spleens of C57BL/6 or OT-IxRag−/− mice (detailed protocol in (25)). Expression of TCR-1D9 in CD8+ T cells was verified by flow cytometry using TCRvβ6- and CD8-specific antibodies (see below). To analyze presence of the mp68 ligand on the indicated tumor cells, 24-hour cocultures with 5x104 TCR-1D9-engineered T cells were done, and T-cell activation was assessed by measuring IFNγ content in supernatants by ELISA (OptEIA IFNγ ELISA Kit II, BD Biosciences), following the manufacturer’s protocol. Unmodified T cells were used as control. Ionomycin (Iono, 1 μM) and phorbol-12-myristate-13-acetate (PMA, 5 ng/mL; both Sigma Aldrich) or a mixture of CD3- and CD28-specific anti-mouse antibodies [CD3, 145–2C11 (clone), 8 μg/mL (The University of Chicago); CD28, 3751; 2.5 μg/mL (BioLegend, San Diego, CA)] were used for TCR-independent stimulation of T-cell cultures.
Flow cytometry and antibodies
Erythrocytes in 50 μL blood were lysed by ammonium chloride treatment (Gibco). 1–2 μg FcR block (anti-mouse CD16/CD32, 2.4G2, BioLegend) was added to the samples, and after 20 minutes, 1x106 cells were incubated with 1 μg of anti-mouse antibodies specific for CD3 [145–2C11, Peridinin Chlorophyll Protein Complex (PerCP); BioLegend], CD4 [GK1.5, fluorescein isothiocyanate (FITC); BioLegend], CD8 [53–6.7, phycoerythrin (PE) or allophycocyanin (APC); BioLegend], TCRvβ6 (RR4–7, PE, FITC or APC; BD Biosciences), TCR-2C [first antibody: 1B2 (48), secondary antibody: APC-labeled goat anti-mouse IgG (Invitrogen)], CD45R (RA3–6B2, PE; BioLegend) or using H-2Kb:SIY multimers [Dimer X (loaded with SIY peptide following the manufacturer’s protocol), PE; BD Biosciences]. Separation of living and dead cells was performed using SYTOX Blue Dead Cell Stain (Invitrogen). For T-cell quantification, fluorescent beads (Spherotech, Lake Forest, IL) were used according to manufacturer’s specifications. Samples were analyzed using flow cytometers LSR II or Canto II and FlowJo software (BD Biosciences).
Microbiome analyses
DNA extraction:
Fecal pellets were collected from individual mice of each of the four groups (Figure 1A). Sample collection was done from July of 2010 through September of 2010. Mice were scruffed, and fecal samples were collected directly in Eppendorf tubes labeled according to mouse number. Each pellet was suspended in a solution containing 500 μL of extraction buffer (200 mM Tris, pH 8.0; 200 mM NaCl; and 20 mM EDTA), 210 μL of 20% SDS, 500 μL of phenol/chloroform/isoamyl alcohol (25:24:1), and 500 μL of 0.1-mm diameter zirconia/silica beads (BioSpec Products, Bartlesville, OK). Microbial cells were lysed by mechanical disruption with a bead beater (BioSpec Products) for 2 minutes, after which, two rounds of phenol/chloroform/isoamyl alcohol extraction were performed. DNA was precipitated with 100% ethanol and resuspended in 50 μL of nuclease-free water. DNA was subjected to additional purification with the QIAamp DNA Mini Kit (Qiagen, Germany).
PCR amplification and sequencing:
For each specimen, 3 replicate 25 μL PCRs were performed, each containing 5 ng of purified DNA, 0.2 mM dNTPs, 1.5 mM MgCl2, 1.25 U Platinum Taq DNA polymerase, 2.5 μL of 10x PCR buffer (IDT), and 0.2 μM each of broad-range bacterial forward and reverse primers as described previously (49) flanking the V1-V3 variable region. The primers were modified to include adaptor sequences required for 454 sequencing, with the addition of a unique 6–8 base barcode in the reverse primer. The forward primer (5'-cct atc ccc tgt gtg cct tgg cag tct cag agt ttg atc ctg gct cag-3') consisted of the 454 Lib-L primer B (underline) and the broad-range universal bacterial primer 8F (italics); the reverse primer (5′-cca tct cat ccc tgc gtg tct ccg act cag nnn nnn nat tac cgc ggc tgc tgg-3′) consisted of the 454 Lib-L primer A (underline), barcode (n), and the broad-range primer 534R (italics). The cycling conditions used were: 94 °C for 3 minutes, followed by 25 cycles of 94 °C for 30 seconds, 56 °C for 30 seconds, and 72 °C for 1 min. The three replicate PCR products were pooled and subsequently purified using the Qiaquick PCR Purification Kit (Qiagen). The purified PCR products were sequenced unidirectionally on a 454 GS FLX Titanium pyrosequencing platform following the Roche 454 recommended procedures.
Analysis:
Sequence data were compiled and processed using Mothur version 1.20 (50). Sequence data were screened and filtered for quality, then aligned to the full-length 16S rRNA gene using the SILVA reference alignment as the template (51). Sequences were grouped into operational taxonomic units of 97% similarity. Phylogenetic classification to genus level was performed for each 16S sequence using the Ribosomal Database Program naive Bayesian classification scheme (52). UniFrac (53) distances were calculated using the phyloseq (54) R package using R version 3.2.1. Relative abundance and PCoA plots were made using ggplot2 (55). Sequences were deposited into the SRA under BioProject number: PRJNA590799.
TCR Sequencing analysis
TCR genes were sequenced following a modified, previously described method (56,57). Briefly, splenic CD8+ T cells from either C57BL/6, Rag+/−, 2CxRag+/−, or 2CxRag−/− mice were isolated (CD8a+ T-cell isolation Kit, Miltenyi Biotec, Germany), and total RNA was extracted (RNeasy Plus Mini Kit, Qiagen). Synthesis of cDNA with a common 5’-RACE adapter was performed from total RNA (43-300 ng) by using SMART library construction kit (TaKaRa Bio, CA). PCR amplification of TCRα and β regions from the cDNAs was done by using a forward primer for the SMART adapter and reverse primers corresponding to the constant region of the TCRs (Trac: 5’-tcg tcg gca gcg tca gat gtg tat aag aga cag gca cat tga ttt ggg agt c-3’, Trbc: 5’-tcg tcg gca gcg tca gat gtg tat aag aga cag ggg tag cct ttt gtt tgt ttg-3’; Illumina adapter sequence, underlined). For Illumina sequencing, the Illumina index adapter with barcode was added by using the Nextera XT Index kit (Illumina, CA), and the prepared libraries were sequenced by 300 bp paired-end reads on an Illumina MiSeq platform using MiSeq Reagent v3 600-cycles Kit (Illumina). Obtained sequence reads were analyzed using Tcrip software (57), and based on our analysis of CD8+ T cells from 2CxRag−/− mice, we set five identical reads as the threshold to exclude false diversity due to inherent sequencing errors (Supplementary Table S1). Due to minor deviations when determining gene alleles and CDR3 sequences of TCR-2C in about 4% of sequencing reads, TCR diversity was determined based on frequency of TRAV and TRBV genes. To detect the somatic mutation in p68, genomic DNA was isolated from indicated 8101 tumor cells (Invisorb Spin Tissue Mini Kit, Invitek Molecular, Germany), and a 425 bp fragment of the p68 gene was amplified by PCR [Phusion, Fisher Scientific, NH, primers: 5’-ggg gat ccg cca tga agg acg atc gtc gtg aca g-3’ (sense), 5’-aga ata ccc tgt tgg cat gg-3’ (antisense)]. The PCR fragment included base 1812 of the p68 gene that is affected by a somatic mutation in 8101 and causative for expression of mp68. The PCR fragments were analyzed by Sanger sequencing using given oligonucleotides as primers (Eurofins Genomics, Germany).
Statistical analyses
Comparison of the number of mice with or without tumor growth was performed using Fisher’s exact test. The distribution of time to tumor development was estimated using the Kaplan-Meier method (58). Mice that were alive and tumor-free at the end of the experiment were censored. However, mice were also at risk of dying from non-cancer causes, and in the Kaplan-Meier analyses, these mice were censored at the time of death. Comparisons of time to tumor development between groups with different TCR diversities were performed using the logrank test (59). To examine the effects of TCR diversity on non-cancer mortality, competing risks analyses were performed, treating tumor development and death from non-cancer causes prior to tumor development as competing risks. Cumulative incidence curves (60) were generated and group comparisons performed using a Wald-type test, and for the Kaplan-Meier analyses, these mice were censored at the time of death.
RESULTS
Siblings from the same parental mice differ only in TCR diversity
The aim of the study was to investigate whether CD8+ T cells contribute to immune surveillance of tumor development, and how much this depends on a diverse TCR repertoire. To address this, we followed a strategy in which the experimental groups differ in their TCR diversity without additional parameters such as genetic bias or breeding conditions affecting the analysis. We established a C57BL/6 breeding in which the genetic information for the transgenic TCR-2C (specific for the peptide SIY presented on H-2Kb (Kb:SIY)) and the knockout of the recombination-activating gene 1 (Rag1) are passed on differently to the offspring (Figure 1A). Progeny resulted in full siblings that fell into one of the four experimental groups: Rag+/−, 2CxRag+/−, 2CxRag−/−, and Rag−/−. Mice of the Rag+/− group expressed a single copy of the Rag1 gene, sufficient for providing the animals with a broad TCR repertoire. In this group CD8+ (Figure 1B) and CD4+ T cells (Figure 1C) were similar in numbers, but less than 1% bound the monoclonal idiotype-specific antibody 1B2 that specifically detects expression of TCR-2C or H-2Kb:SIY multimers. To estimate the overall diversity of TCRs in these mice, the frequency of variable gene segments in TCRα and TCRβ chains in splenic CD8+ T cells was determined by TCR repertoire analysis. In line with our cytometric analysis, the number of TCR-2C sequences in Rag+/− mice was not significantly above background (Supplementary Table S1), and the TCRαβ chains encoded for a variety of different TRAV and TRBV genes (Figure 1D). The TCR diversity in CD8+ T cells of C57BL/6 mice used as controls was almost identical. Mice of the 2CxRag+/− group expressed the TCR-2C transgene and generated only a limited TCR repertoire. About 92% of their CD8+ T cells expressed TCR-2C and approximately 96% bound Kb:SIY multimers (Figure 1B). As a result, almost all sequences from the TCR repertoire analysis were identical to TCR-2C, and TCR diversity decreased significantly compared to the Rag+/− group (Figure 1D). This narrowed TCR repertoire is due to the preferential selection of T cells with the pre-arranged TCR-2C during thymic development. However, expression of Rag1 allows some T cells to arrange TCRs and to be selected with V(D)J combinations different from TCR-2C, as indicated by expression of other TRAV and TRBV genes (Figure 1D). Mice of the 2CxRag−/− group expressed only TCR-2C, as these animals have no functional copy of the Rag1 gene. Thus, almost all CD8+ T cells (>99%) could be stained with the 1B2 antibody and bound the Kb:SIY multimer. The V(D)J combinations of TCR-2C were the only ones detected above the threshold we set to accurately separate diverse TCR chains due to somatic recombination from false diversity due to sequencing errors (Figure 1D). The decrease in the TCR diversity in the 2CxRag+/− and the 2CxRag−/− groups was not associated with a decrease in the total number of CD8+ T cells in peripheral blood (Figure 1B). However, both the absolute and relative numbers of CD4+ T cells were substantially reduced in these groups compared to CD4+ T cells of Rag+/− mice having a broad TCR repertoire (Figure 1C). Mice of the Rag−/− group did not express the TCR-2C transgene nor did they generate TCRs via somatic recombination because they lacked functional copies of Rag1. Consequently, no T cells (and, thus, TCRs) were detectable in peripheral blood and spleen, which did not allow for analyzing TCR diversity.
Rejection of regressor tumors depends on TCR diversity and expression of cognate antigen
We first tested whether T cells from mice in the different groups could protect the animals from tumor development after transplantation of variants of the fibrosarcoma cell line MC57 (induced by MCA in a C57BL/6 mouse). In this setting, the inflammatory conditions after subcutaneous transplantation promote priming of MC57-specific T cells, typically leading to cancer elimination. Consistent with other tumor models (61), MC57 has a regressor phenotype when inoculated as cancer cell suspension (62), i.e., the diverse repertoire of T cells in immune-competent animals is sufficient to eliminate the cells after inoculation. Thus, no tumors developed in any of the animals of the Rag+/− group, and transplanted MC57 cells that were either unmodified or expressed SIY or an antigen irrelevant in this setting (mp68, resulting from a point mutation in the RNA helicase gene p68, (31)) were rejected (Table 1). Transplantation of the same cells into animals of the 2CxRag+/− group showed that a reduced TCR diversity was no longer sufficient to ensure rejection of every MC57 variant, as tumors grew in each mouse injected with MC57 or MC57-mp68. However, T cells in 2CxRag+/− animals were able to eliminate transplanted MC57 cells if they expressed the antigen SIY that is recognized by TCR-2C (Table 1). TCR-2C alone was sufficient for this effect because 2CxRag−/− animal also did not develop tumors after transplantation of MC57-SIY cancer cells. Under these conditions, CD4+ T cells were not needed because severe reduction or absence of CD4+ T cells in 2CxRag+/− or 2CxRag−/− animals (Figure 1C) did not interfere with this rejection (Table 1). The observed tumor rejection was dependent on T cells, and the MC57 cells used had comparable tumorigenicity, as demonstrated by transplantation into Rag−/− mice, all of which developed tumors after the same latency (Table 1). Tumor rejection in TCR-transgenic mice was also antigen-dependent, demonstrated by transplantation of the MC57 variants into 1D9xRag−/− mice. Although not derived from the breeding described above, these mice were kept under identical housing conditions. Due to the lack of both functional copies of the Rag1 gene, CD8+ T cells of the animals expressed only TCR-1D9, which has specificity for the H-2Kb-presented antigen mp68 (25). Accordingly, MC57-mp68 cells were rejected in 1D9xRag−/− mice, whereas unmodified and SIY-expressing MC57 cells grew into tumors (Table 1). 1D9xRag−/− mice and 2CxRag−/− mice exclusively expressed their transgenic TCR (Figure 1D, (25)) and had higher absolute numbers of CD8+ T cells than Rag+/− mice having a fully diverse TCR repertoire (Figure 1D). Nevertheless, 1D9xRag−/− and 2CxRag−/− mice selectively and completely prevented outgrowth of MC57 cancer cells with the cognate target yet had no detectable effect in preventing outgrowth of identical MC57 cancer cells, except for those expressing non-cognate targets. Thus, no evidence for bystander killing was seen.
Table 1:
A single type of TCR prevents tumor development if cognate antigen is expressed by all cancer cells, whereas TCR diversity, as determined in Figure 1D, is required to eliminate cancers displaying antigenic heterogeneity.
| Group | Specificity of transgenic TCR |
TCRtg T cells in repertoire |
Tumor growth after cancer cell inoculation* | ||||
|---|---|---|---|---|---|---|---|
| MC57 | MC57- SIY |
MC57- mp68 |
8101- bulk |
8101- clone |
|||
| Rag+/− | – | – | 0/6 | 0/5 | 0/8 | 0/8 | 0/5 |
| 2CxRag+/− | Anti-SIY | ~95% | 5/5a | 0/8 | 5/5c | 10/13d | ND |
| 2CxRag−/− | Anti-SIY | 100% | 5/5 a | 0/5 | 4/4#,a | 5/5 | ND |
| Rag−/− | – | – | 5/5 a | 5/5b | 5/5c | 5/5 | 5/5b |
| 1D9xRag−/− | Anti-mp68 | 100% | 5/5 a | 5/5 b | 0/8 | 7/7§ | 0/15¶ |
2-6-month-old mice were inoculated subcutaneously with the indicated cancer cells. Mice listed as tumor-free had no visible tumor at the injection site 3 weeks after subcutaneous inoculation. Experiments pooled from 2-3 independent experiments.
Tumor-free mice were kept for 100 days after inoculation to monitor for relapse.
An additional mouse was found dead 9 days after injection before a tumor developed.
Data detailed in Figure 2.
An additional mouse developed a tumor but was excluded because it also developed leukemia. The resected tumor was still recognized by 1D9 T cells.
p=0.001 vs. Rag+/−.
p=0.008 vs. Rag+/−.
p=0.001 vs. Rag+/−.
p=0.001 vs. Rag+/−. TCRtg: TCR-transgenic; ND: not done.
TCR diversity is required for rejection of cancers with heterogeneous antigen expression
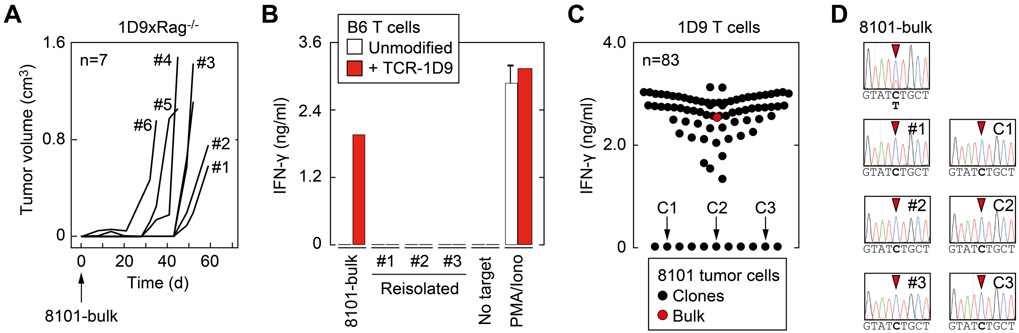
To investigate whether a diverse TCR repertoire was also effective in eliminating heterogeneous cancer cell populations after inoculation, we continued our work with the C57BL/6-derived cell line 8101-bulk. The cell line was derived from the UV-induced tumor 8101 (31) and harbors the immunodominant neoantigen mp68. In contrast to MC57, 8101-bulk was not cloned, passaged in vivo, or expanded extensively in cell culture. However, 8101-bulk cells also show a regressor phenotype and were rejected by immune-competent Rag+/− mice after transplantation (Table 1). A broad TCR diversity was required for this rejection because 8101-bulk cancer cells developed into tumors in 10 out of 13 2CxRag+/− mice, which have a reduced TCR diversity compared to Rag+/− animals. Consequently, 8101-bulk tumors grew in all 2CxRag−/− and Rag−/− mice, which either generated only SIY-specific CD8+ T cells expressing TCR-2C or no T cells, respectively (Table 1). Although T cells in most mice prevented growth of 8101-bulk tumors until 1 month after inoculation, a longer observation time revealed that tumors eventually grew in all 1D9xRag−/− animals (Figure 2A, Table 1). These tumors consisted of variants that no longer expressed the mp68 ligand (Figure 2B, Supplementary Figure S1). This observation is consistent with our previous experiments showing that despite abundant expression of mp68, 8101-bulk cancer cells also escape adoptive therapy using TCR-1D9-expressing T cells as mp68-negative variants (25). These results suggested that mp68-negative variants might already be contained in 8101-bulk. To test this, we generated tumor cell clones from 8101-bulk and analyzed whether each clone could stimulate TCR-1D9-expressing T cells. Eleven of the 83 analyzed clones generated from 8101-bulk failed to activate mp68-specific T cells (Figure 2C), suggesting that about 13% of 8101-bulk tumor cells were prone to escape mp68-specific immune surveillance. Absence of mp68 expression in re-isolated 8101 variants and in representative tumor cell clones was confirmed by sequencing (Figure 2D). Thus, even after transplantation of only small percentages of antigen-negative tumor cells, TCR-1D9-expressing T cells failed to reject these variant cancer cells as bystanders. Subsequently, using an mp68-positive tumor cell clone isolated from 8101 (8101-clone), we demonstrated that a homogeneous, non-manipulated tumor cell population was rejected by T cells in 1D9xRag−/− mice (Table 1). Together, the experiments suggested that CD8+ T cell–mediated immune surveillance was antigen-dependent and, therefore, required a wide variety of TCRs to detect potential rejection antigens on tumor cells.
Figure 2. Rejection of transplanted cancer cells by CD8+ T cells is antigen-dependent and not supported by bystander elimination.
(A) 8101-bulk cancer cells were subcutaneously injected into 1D9xRag−/− mice (n=7), and tumor growth was monitored. Progressing tumors were resected from indicated mice approximately 2 months after transplantation, and cancer cells were cultured for further experiments. (B) IFNγ secretion of TCR-1D9-expressing T cells after coculture with 8101-bulk cancer cells and re-isolates from 1D9xRag−/− mice (see panel A). Unmodified C57BL/6-derived T cells (B6) and PMA and ionomycin (Iono) for TCR-independent stimulation of T cells were used as controls. Data are means of duplicates ± standard error and representative of two independent experiments. (C) Single clones of 8101-bulk were generated by limiting dilution (n=83) and analyzed for presence of the mp68 ligand by incubation with TCR-1D9-expressing T cells. Recognition of mp68 was assessed by IFNγ secretion. Data are means of duplicates. Absence of the mp68 ligand in 11 clones was verified in a second experiment. (D) Electropherogram of sequencing analysis of p68 DNA isolated from 8101-bulk, tumor re-isolates from 1D9xRag−/− mice (#1-3, see panel A) and 3 representative mp68 ligand–negative clones (C1-3, see panel C). Exchange of C>T nucleotides in codon 551 is indicated with the red arrowhead.
Compromised TCR diversity increased the risk to develop cancer
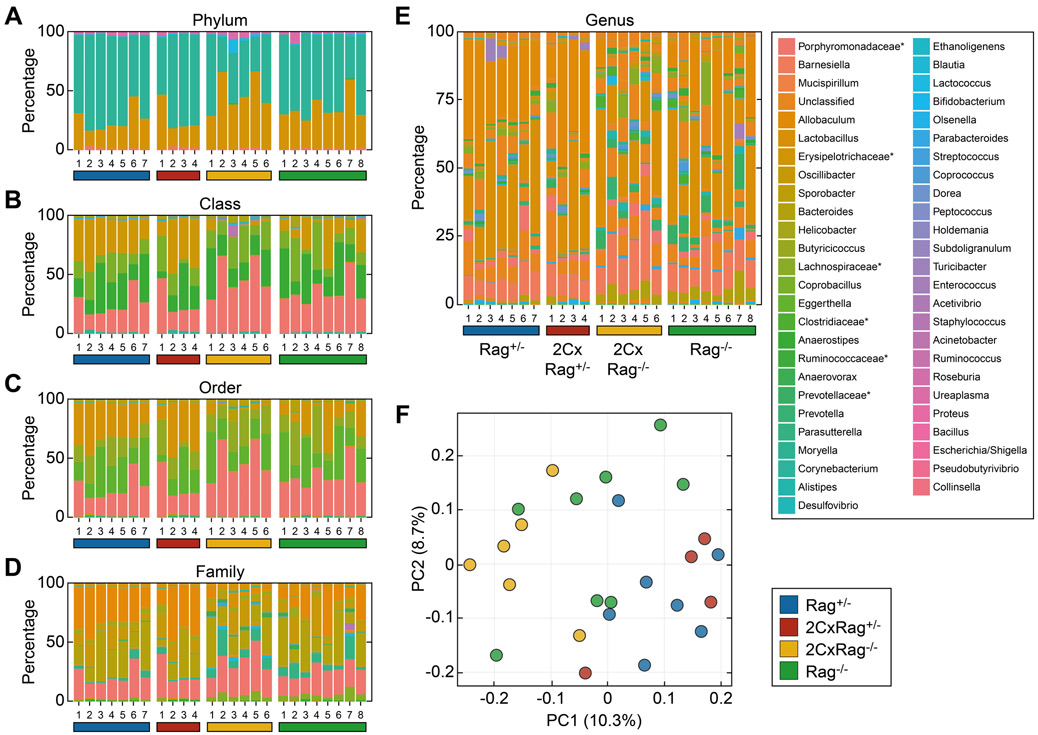
In contrast to the transplantation of cancer cells, the sporadic development of tumors is more complex and, therefore, difficult to replicate in mice. In humans, this process usually takes months or even years, and only during the last step of cancer development are tumors diagnosed clinically. However, the process can be simulated by subcutaneous injection of the carcinogen MCA, which initiates cancer development at the site of injection and, thus, allows monitoring of tumor growth. We knew from previous experiments that differences in the intestinal flora can influence tumor induction by carcinogens (28). Although commensal bacteria are mainly transferred to offspring by maternal transmission (63), mice with either broad or reduced TCR diversity may subsequently develop a different intestinal flora when kept in separate cages. To minimize potential variations in the microbiota, we housed at least one mouse from each group together in a cage. Thus, siblings were separated only by gender, not by genotype, and kept as littermates in the same cage. To determine the composition of the intestinal microbiota in these co-housed animals, we isolated fecal pellets from individual mice of the four groups and sequenced 16S ribosomal RNA to classify commensal bacteria (Figure 3A-E, legends in Supplementary Figure S2). Unweighted UniFrac analysis was used to compare the relative abundance of 51 genera. Differences between the four genetic groups were detected across at least one of the categories despite mice being kept in the same cage (Figure 3F). Although differences among the groups remained small, mice in the groups having a diverse TCR repertoire (Rag+/−) or with limited TCR diversity (2CxRag+/−) were more similar to each other than mice of the 2CxRag−/− and Rag−/− group, which either had T cells with only one type of TCR or lacked T cells.
Figure 3. Mice in the four experimental groups sharing their microbial environment have a similar but not identical bacterial flora.
Intestinal bacteria in fecal pellets isolated from mice of the 4 experimental groups with different TCR diversity were determined by sequencing. Samples from 4-8 mice of each of the 4 groups were analyzed. The stacked bar plots show the relative abundances at the (A) phylum, (B) class, (C) order, (D) family, and (E) genus level by cluster of related operational taxonomic units (crOTU) community type. Legends for panels A-D are provided in Supplementary Figure S2. *Could not be confidentially classified to the family level. (F) Principal coordinate analysis (PCoA) of UniFrac distances between samples of the indicated experimental groups.
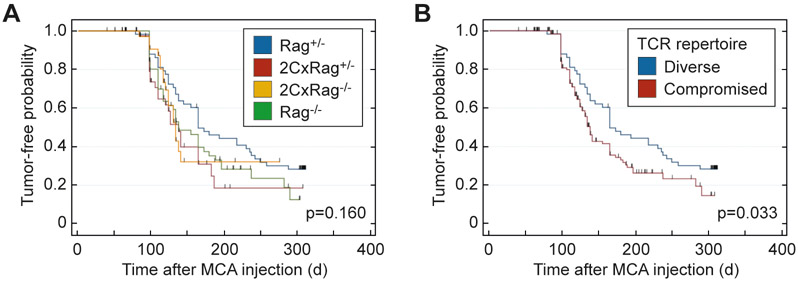
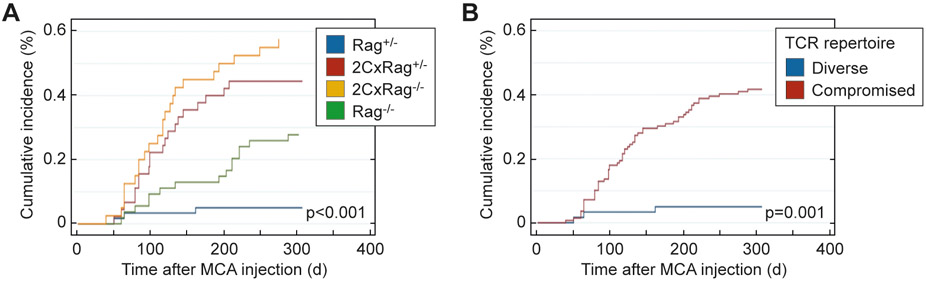
We injected 25 μg of MCA into 40-60 animals of the four experimental groups described above (Figure 1A). The results obtained under these defined conditions suggested that mice with reduced or no TCR diversity or no T cells (2CxRag+/−, 2CxRag−/−, and Rag−/−) developed MCA-induced tumors somewhat faster than Rag+/− mice that have a diverse TCR repertoire (Figure 4A). Although the time to onset of tumor development was not statistically significantly different when comparing the four different groups individually (p=0.16), after pooling mice of the three groups with reduced TCR diversity, the difference reached statistical significance (p=0.033)(Figure 4B).
Figure 4. MCA-induced cancer development in mice varying in TCR diversity but sharing the same microbial environment.
Mice of the experimental groups showing different TCR diversity were subcutaneously injected with a single dose of 25 μg MCA and observed for tumor development for approximately 300 days. Kaplan-Meier plots indicate the proportion of tumor-free mice in (A) the 4 experimental groups [Rag+/− (n=60), 2CxRag+/− (n=45), 2CxRag−/− (n=40), Rag−/− (n=54)] and (B) the groups pooled according to diverse (n=60) vs. compromised TCR repertoire (2CxRag+/−, 2CxRag−/−, Rag−/−, n=139). Tic markers prior to the end of the experiment indicate mice censored due to death from causes other than MCA-induced cancer. Logrank test was used to compare groups in A and B. P≤0.05 indicates statistical significance.
Loss of TCR diversity can increase the risk of early death from causes other than cancer
In the experiment shown in Figure 4, mice were injected with MCA when they were 3-4 months old, and the induction of primary cancers took many months to develop. Mice from each of the three groups with compromised TCR diversity (2CxRag+/−, 2CxRag−/−, and Rag−/−) had a much higher risk of dying from causes other than MCA-induced cancer compared to Rag+/− mice with a high TCR diversity (Figure 5A). Death from lymphomas or thymomas was only one of the causes because many of the immune-compromised mice died for unknown reasons. The cumulative incidence curves after pooling the immune-compromised groups are shown in Figure 5B. The competing risk of the groups of mice with compromised TCR diversity began to rise ~50 days after the injection of MCA when the mice were 150-180 days old. Although we found a difference in time to tumor development after MCA injection when comparing mice with a reduced TCR diversity to animals with broad TCR diversity (Figure 4B), this difference remains marginal compared to the risk of early death in the two cohorts (Figure 5B).
Figure 5. Death from causes other than MCA-induced cancer in mice varying in TCR diversity.
(A) Kaplan-Meier curves show cumulative incidence of deaths unrelated to MCA-induced cancer for the 4 groups with different TCR diversities in the experiment detailed in Figure 4A [Rag+/− (n=60), 2CxRag+/− (n=45), 2CxRag−/− (n=40), Rag−/− (n=54)]. (B) The cumulative incidence of deaths unrelated to MCA-induced cancer in groups pooled according to diverse vs. compromised TCR repertoire is shown for the experiment shown in Figure 4B [Rag+/− (n=60); 2CxRag+/−, 2CxRag−/−, Rag−/− (n=139)]. Wald test was used to compare groups in A and B. P≤0.05 indicates statistical significance.
DISCUSSION
In this study, we not only analyzed directly the presence or absence of T cells but also the relative influence of TCR diversity on (i) the development of tumors and (ii) the competing risks caused by loss of TCR diversity or T cells. Tumors were induced either by cancer cell inoculation or by injecting the chemical carcinogen MCA. These two types of animal models used in our study were selected to at least approximate two stages of cancer development in humans: (i) the elimination of residual or incipient but fully developed cancer cells and (ii) the elimination of carcinogen-initiated and/or premalignant cells before they become malignant.
In the first model, mice were 2-6 months old at the start of the experiment that was terminated 100 days later. During the short duration of the experiment mice had virtually no other risk than death from cancer. These experiments indicated that lack of TCR diversity substantially increased the risk of developing tumors after inoculating cancer displaying antigenic heterogeneity. When using the cancer model 8101, all potential T-cell targets were naturally expressed as autochthonous neoantigens, and the heterogeneity was not manipulated since the bulk cancer cell population used was a minimally expanded culture derived from tumor fragments isolated from the primary tumor 8101. Detailed genetic analysis of the primary 8101 cancer reveals that the neoantigen mp68 was expressed in every analyzed branch (fragment) of the tumor and is therefore considered a trunk mutation (25). However, analysis of cancer cell clones from the 8101-bulk population revealed that approximately 13% of the cancer cells in the population lacked the immunodominant mp68 ligand. Nevertheless, full diversity sufficed to eliminate these variants and eradicated the inoculum. A reduced TCR repertoire caused a major increase in the rate of relapse, but the remaining TCR diversity in the 2CxRag+/− group still allowed some of the mice to reject the heterogeneous cancer cell population. The loss of resistance to transplanted cancer cells was not the result of reduced numbers of CD8+ T cells but loss of the TCR diversity, because the 2CxRag+/− mice showed no decrease in the absolute number of CD8+ T cells. Also, these CD8+ T cells were fully functional, even when expressing only a single TCR specificity and did not need help by CD4+ T cells. The T cells rejected inoculated cancer cells as long as the cognate neoantigen was expressed on all cancer cells either after transfection (MC57-SIY, MC57-mp68) or when using a clone of the heterogeneous 8101 cancer cell population (8101-clone). In the absence of cognate antigen ligand on the surface of the cancer cells, CD8+ T cells, though normal in numbers, had no effect on “bystanders”. Thus, the presence of CD8+ T cells in same absolute numbers as in Rag+/− mice but with an irrelevant TCR did not prolong the time interval when mice reached a tumor volume that required sacrifice.
In the second model, the induction of primary tumors by injection of MCA required much longer time and experiments continued until animals had died or needed to be sacrificed. In comparison to mice with a broad TCR repertoire (Rag+/−), none of the groups with reduced TCR diversity showed a statistically significant increased risk of developing a primary tumor in response to MCA. However, a statistically significant difference was detected when pooling the groups of mice with reduced TCR diversity. It could be argued that the development of primary tumors in each of the TCR-deficient groups would be significantly different if we had increased the group sample sizes even further. Similarly, using a lower dose of carcinogen might have provided more time for T cells to become effective. However, none of these changes in the experimental design would have resolved the problem of major differences in competing risks between the immune-competent and -compromised groups that were also observed.
Deaths from unrelated causes are inherent in long-term experiments and may affect the results of such studies. The Kaplan-Meier survival estimate and accompanying logrank test were used to correct for competing risks by censoring (64). However, the following assumptions should be met for such censoring (65): (#1) the censoring pattern must be similar in amount in all groups tested and (#2) the conditions that caused early death in the immune-compromised groups must have no influence on the measured event, in our case, primary tumor development. Assumption #1 was not justified in our studies when comparing the Rag+/− group with the three groups of mice with reduced TCR diversity. Also, #2 is an unverifiable assumption. We do not know whether the mechanism that caused death without tumor development preferentially killed mice that would have developed cancer with a higher, lower, or equal risk had they not died. This uncertainty applies to each of the immune-compromised groups and raises the additional question of whether these cohorts can be pooled at all for statistical analysis. With the competing risk of both parameters, it remains difficult to discern the extent to which CD8+ T cells, whose broad TCR diversity is sufficient to eliminate cancer cells after transplantation, can arrest or slow the development of primary tumors. Certainly CD8+ T cell function should be improved by synergy with CD4+ T cells present in the mice with a diverse repertoire (66).
It is somewhat surprising that the increased risk of mice in the immune-compromised groups to die early, even without tumor development, has not been discussed and reported in earlier studies given the extensive use of such mice in many types of experiments examining immune surveillance of cancer development (13,14,19,67). One could assume that subclinical infections may be associated with the increased incidence of death from other causes than cancers in the severely immunodeficient mice. However, common mouse pathogens were unlikely responsible for the observed effects in immune-compromised mice because the mice were kept in the SPF facility of the University of Chicago that routinely controls for pathogens. Nevertheless, exclusion of pathogens never means the flora is fully defined. It must be expected that additional pathogens will continue to be discovered and that most of these previously unknown agents will not result in clinical disease, but could still affect experimental results (68). Only when laboratory animals are completely free of pathogens, i.e., germfree, carcinogenesis experiments will not be influenced by flora. Germfree animals have been shown to differ in their response to chemical carcinogens (28), but we are not aware of experiments comparing MCA-induced tumor development in germfree mice differing in their TCR diversity. Also, in real life, an immune system with decreased TCR diversity must deal with cancer in the face of natural pathogens and will naturally have competing risks that are far greater for immunodeficient individuals. Therefore, all experimental or clinical data generated must be interpreted with these reservations in mind. The major reason why all the groups in our study were siblings derived from a single genetically defined set of parents in a single colony was to minimize differences in genetic background and environmental factors including microbial ones.
By contrast, there were no unrelated deaths during the brief observation period needed for obtaining the results in studying the influence of TCR diversity on tumor development after cancer cell inoculation. Within 50 days after cancer cell inoculation, either lethal tumors developed or the inoculum was rejected. No mortality due to unrelated causes was noticed during this short observation period, and loss of TCR diversity clearly increased the risk of tumor development following cancer cell inoculation. Nonetheless, criticism of the inoculation experiments is justified in that antigen-specific T cells are helped in the detection and elimination of fully malignant cancers by the artificially acute inflammation at the site of cancer cell inoculation (69). However, in our study this artificial inflammatory response was insufficient to overcome the need for TCR diversity to reject an antigenically diverse cancer cell population.
Microbiota may amplify or mitigate primary cancer induction by radiation (70) and chemical carcinogens (28), as well as influence antitumor immunity and the therapeutic response to checkpoint blockade in patients (21-24). In contrast to earlier studies comparing effects of the immune system on primary tumor induction, all four groups of mice in our experiments were derived from one set of parents and shared their microbial environment as much as possible. We used UniFrac analysis, which measures the distance between microbial communities (based on the lineages they contain), to analyze the relatedness of the microbial communities in our four groups. Although this method is designed to emphasize the differences rather than similarities between populations, we found that the differences between the microbial communities of the four experimental groups were small. This may not be surprising as mice are coprophages and thereby stabilize their gut microbiome. From other studies on cohabiting family members sharing their environment (71), it is expected that very small differences based on genetic difference between the groups will remain. Thus, cohabitation made the microbiome more similar, but cohabitation did not suffice to erase all differences between the groups. This is very much what we observed. The 2CxRag−/− and the Rag−/− groups lack IgA and cohabitation may not have completely erased the effects that are caused by the lack of secretory IgA (72,73) in shaping the composition of the intestinal microbiota and altering immune response toward the microbiome. Thus, even when using littermates and co-housing of the different immune-deficient mice, it cannot be fully excluded that minor differences in the microbiome affected the experimental outcome of carcinogenesis but sharing the microbiome should have reduced the artifacts due to differences. However, we cannot exclude that a given virus or bacterium, even when not considered a pathogen, differentially influenced the immune response in our four genetic groups. An example could be murine norovirus (74) that neither causes lethality nor histological lesions in immunocompetent or Rag-deficient mice but is the most commonly known virus in laboratory mice and was present in our colonies (75).
Studies determining whether TCR diversity in immune-competent patients leads to a selection of neoepitopes predicted to be immunogenic in human cancers are either lost or preserved are not yet consistent, so there is no clarity as to whether TCR diversity in immune-competent patients leads to such a selection (76,77). Clearly, however, all cancers in humans or mice harbor a unique set of cancer-specific mutations causing neoantigens and most of them are caused by point mutations (6,31,78-81). A study using a mosaic composite of EL4 cancer cells endowed with point mutations known to be immunogenic when expressed by a cancer cell population (82) shows that rejection of the inoculum requires a relatively large fraction of cancer cells to express an immunogenic mutation to cause rejection. Thus, if the fraction in the population is small, cancer cells escape without evidence for a response or immune selection for loss of the antigen. Indeed, our experiments with the 8101-bulk cell line and the clones established from it showed that the majority of the heterogeneous tumor cell population expressed the neoantigen mp68. Nevertheless, antigenic heterogeneity was present because individual clones lacked the mp68 ligand. Thus, high TCR diversity that allowed immune responses to other remaining antigens was essential to destroy all variants and prevent outgrowth that frequently occurred with a reduced TCR diversity and regularly occurred when all TCRs were only specific for mp68. The main question is whether, and to what extent after loss of an immunodominant antigen, further cancer-specific antigens remain, which can be used as targets for eliminating cancer with help of mutation-specific T cells. Our results suggest that TCR diversity is critical to overcome the natural genetic instability of cancers and their antigenic heterogeneity.
Supplementary Material
ACKNOWLEDGMENTS
This work was supported by National Institutes of Health grants (R01-CA22677, R01-CA37156, P01-CA74182), the University of Chicago Cancer Center (CA-14599), Harriet and Allan Wulfstat, the Gerald O. Mann Foundation, the Cancer Research Foundation to HS and the Deutsche Forschungsgemeinschaft, the German Cancer Consortium (DKTK) and the Berliner Krebsgesellschaft e.V. to ML. KK was supported by The Japan Agency for Medical Research and Development and by the Japan Society for the Promotion of Science. We thank Kimberley Borutta for excellent technical support and Avrion Mitchison for his encouragement to analyze the influence of TCR diversity.
This work was supported by the National Institute of Health (R01-CA37516, R01-CA22677, P01-CA74182), The University of Chicago Cancer Center (CA-14599) to HS and by the Deutsche Forschungsgemeinschaft, the German Cancer Consortium (DKTK), and the Berliner Krebsgesellschaft e.V. to ML.
Footnotes
The authors have no conflicting financial interests.
REFERENCES
- 1.Lythe G, Callard RE, Hoare RL, Molina-Paris C. How many TCR clonotypes does a body maintain? J Theor Biol 2016;389:214–24 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Basombrio MA, Prehn RT. Studies on the basis for diversity and time of appearance of antigens in chemically induced tumors. Natl Cancer Inst Monogr 1972;35:117–24 [PubMed] [Google Scholar]
- 3.Burnet FM. A certain symmetry: histocompatibility antigens compared with immunocyte receptors. Nature 1970;226(5241):123–6 [DOI] [PubMed] [Google Scholar]
- 4.Embleton MJ, Heidelberger C. Antigenicity of clones of mouse prostate cells transformed in vitro. Int J Cancer 1972;9(1):8–18 [DOI] [PubMed] [Google Scholar]
- 5.Schreiber H. Cancer Immunology In: William EP, editor. Fundamental Immunology. Seventh ed. Philadelphia: Lippincott Williams & Wilkins; 2012. p 1200–34. [Google Scholar]
- 6.Monach PA, Meredith SC, Siegel CT, Schreiber H. A unique tumor antigen produced by a single amino acid substitution. Immunity 1995;2(1):45–59 [DOI] [PubMed] [Google Scholar]
- 7.Tran E, Robbins PF, Rosenberg SA. ‘Final common pathway’ of human cancer immunotherapy: targeting random somatic mutations. Nat Immunol 2017;18(3):255–62 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Burnet FM. The concept of immunological surveillance. Prog Exp Tumor Res 1970;13:1–27 [DOI] [PubMed] [Google Scholar]
- 9.Thomas L. Discussion of cellular and humoral aspects of the hypersensative states In: Lawrence HS, editor. A Symposium Held at the New York Academy of Medicine. New York: Hoeber & Harper; 1959. p 529–32. [Google Scholar]
- 10.Burnet M Immunological Factors in the Process of Carcinogenesis. Br Med Bull 1964;20:154–8 [DOI] [PubMed] [Google Scholar]
- 11.Klein G Immunological surveillance against neoplasia. Harvey Lect 1973(69):71–102 [PubMed] [Google Scholar]
- 12.Klein G, Klein E. Immunology. Sinking surveillance’s flagship. Nature 1998;395(6701):441, 3–4 [DOI] [PubMed] [Google Scholar]
- 13.Stutman O. Tumor development after 3-methylcholanthrene in immunologically deficient athymic-nude mice. Science 1974;183(4124):534–6 [DOI] [PubMed] [Google Scholar]
- 14.Shankaran V, Ikeda H, Bruce AT, White JM, Swanson PE, Old LJ, et al. IFNgamma and lymphocytes prevent primary tumour development and shape tumour immunogenicity. Nature 2001;410(6832):1107–11 [DOI] [PubMed] [Google Scholar]
- 15.Berg RE, Forman J. The role of CD8 T cells in innate immunity and in antigen non-specific protection. Curr Opin Immunol 2006;18(3):338–43 [DOI] [PubMed] [Google Scholar]
- 16.Liu RB, Engels B, Schreiber K, Ciszewski C, Schietinger A, Schreiber H, et al. IL-15 in tumor microenvironment causes rejection of large established tumors by T cells in a noncognate T cell receptor-dependent manner. Proc Natl Acad Sci U S A 2013;110(20):8158–63 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gilbertson B, Germano S, Steele P, Turner S, Fazekas de St Groth B, Cheers C. Bystander activation of CD8+ T lymphocytes during experimental mycobacterial infection. Infect Immun 2004;72(12):6884–91 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Qin Z, Kim HJ, Hemme J, Blankenstein T. Inhibition of methylcholanthrene-induced carcinogenesis by an interferon gamma receptor-dependent foreign body reaction. J Exp Med 2002;195(11):1479–90 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Koebel CM, Vermi W, Swann JB, Zerafa N, Rodig SJ, Old LJ, et al. Adaptive immunity maintains occult cancer in an equilibrium state. Nature 2007;450(7171):903–7 [DOI] [PubMed] [Google Scholar]
- 20.Kammertoens T, Friese C, Arina A, Idel C, Briesemeister D, Rothe M, et al. Tumour ischaemia by interferon-gamma resembles physiological blood vessel regression. Nature 2017;545(7652):98–102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gopalakrishnan V, Spencer CN, Nezi L, Reuben A, Andrews MC, Karpinets TV, et al. Gut microbiome modulates response to anti-PD-1 immunotherapy in melanoma patients. Science 2018;359(6371):97–103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Matson V, Fessler J, Bao R, Chongsuwat T, Zha Y, Alegre ML, et al. The commensal microbiome is associated with anti-PD-1 efficacy in metastatic melanoma patients. Science 2018;359(6371):104–8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Routy B, Le Chatelier E, Derosa L, Duong CPM, Alou MT, Daillere R, et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science 2018;359(6371):91–7 [DOI] [PubMed] [Google Scholar]
- 24.Sivan A, Corrales L, Hubert N, Williams JB, Aquino-Michaels K, Earley ZM, et al. Commensal Bifidobacterium promotes antitumor immunity and facilitates anti-PD-L1 efficacy. Science 2015;350(6264):1084–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Leisegang M, Engels B, Schreiber K, Yew PY, Kiyotani K, Idel C, et al. Eradication of Large Solid Tumors by Gene Therapy with a T-Cell Receptor Targeting a Single Cancer-Specific Point Mutation. Clin Cancer Res 2016;22(11):2734–43 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Udaka K, Wiesmuller KH, Kienle S, Jung G, Walden P. Self-MHC-restricted peptides recognized by an alloreactive T lymphocyte clone. J Immunol 1996;157(2):670–8 [PubMed] [Google Scholar]
- 27.Festing MF. Genetically Defined Strains in Drug Development and Toxicity Testing. Methods Mol Biol 2016;1438:1–17 [DOI] [PubMed] [Google Scholar]
- 28.Schreiber H, Nettesheim P, Lijinsky W, Richter CB, Walburg HE Jr. Induction of lung cancer in germfree, specific-pathogen-free, and infected rats by N-nitrosoheptamethyleneimine: enhancement by respiratory infection. J Natl Cancer Inst 1972;49(4):1107–14 [PubMed] [Google Scholar]
- 29.Briesemeister D, Friese C, Isern CC, Dietz E, Blankenstein T, Thoene-Reineke C, et al. Differences in serum cytokine levels between wild type mice and mice with a targeted mutation suggests necessity of using control littermates. Cytokine 2012;60(3):626–33 [DOI] [PubMed] [Google Scholar]
- 30.Holmdahl R, Malissen B. The need for littermate controls. Eur J Immunol 2012;42(1):45–7 [DOI] [PubMed] [Google Scholar]
- 31.Dubey P, Hendrickson RC, Meredith SC, Siegel CT, Shabanowitz J, Skipper JC, et al. The immunodominant antigen of an ultraviolet-induced regressor tumor is generated by a somatic point mutation in the DEAD box helicase p68. J Exp Med 1997;185(4):695–705 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Morita S, Kojima T, Kitamura T. Plat-E: an efficient and stable system for transient packaging of retroviruses. Gene Ther 2000;7(12):1063–6 [DOI] [PubMed] [Google Scholar]
- 33.Leisegang M, Kammertoens T, Uckert W, Blankenstein T. Targeting human melanoma neoantigens by T cell receptor gene therapy. J Clin Invest 2016;126(3):854–8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Outzen HC, Custer RP, Eaton GJ, Prehn RT. Spontaneous and induced tumor incidence in germfree “nude” mice. J Reticuloendothel Soc 1975;17(1):1–9 [PubMed] [Google Scholar]
- 35.Stutman O Chemical carcinogenesis in nude mice: comparison between nude mice from homozygous matings and heterozygous matings and effect of age and carcinogen dose. J Natl Cancer Inst 1979;62(2):353–8 [PubMed] [Google Scholar]
- 36.Svane IM, Engel AM, Nielsen MB, Ljunggren HG, Rygaard J, Werdelin O. Chemically induced sarcomas from nude mice are more immunogenic than similar sarcomas from congenic normal mice. Eur J Immunol 1996;26(8):1844–50 [DOI] [PubMed] [Google Scholar]
- 37.van den Broek ME, Kagi D, Ossendorp F, Toes R, Vamvakas S, Lutz WK, et al. Decreased tumor surveillance in perforin-deficient mice. J Exp Med 1996;184(5):1781–90 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Engel AM, Svane IM, Rygaard J, Werdelin O. MCA sarcomas induced in scid mice are more immunogenic than MCA sarcomas induced in congenic, immunocompetent mice. Scand J Immunol 1997;45(5):463–70 [DOI] [PubMed] [Google Scholar]
- 39.Kaplan DH, Shankaran V, Dighe AS, Stockert E, Aguet M, Old LJ, et al. Demonstration of an interferon gamma-dependent tumor surveillance system in immunocompetent mice. Proc Natl Acad Sci U S A 1998;95(13):7556–61 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Smyth MJ, Thia KY, Street SE, Cretney E, Trapani JA, Taniguchi M, et al. Differential tumor surveillance by natural killer (NK) and NKT cells. J Exp Med 2000;191(4):661–8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Girardi M, Oppenheim DE, Steele CR, Lewis JM, Glusac E, Filler R, et al. Regulation of cutaneous malignancy by gammadelta T cells. Science 2001;294(5542):605–9 [DOI] [PubMed] [Google Scholar]
- 42.Smyth MJ, Crowe NY, Godfrey DI. NK cells and NKT cells collaborate in host protection from methylcholanthrene-induced fibrosarcoma. Int Immunol 2001;13(4):459–63 [DOI] [PubMed] [Google Scholar]
- 43.Street SE, Cretney E, Smyth MJ. Perforin and interferon-gamma activities independently control tumor initiation, growth, and metastasis. Blood 2001;97(1):192–7 [DOI] [PubMed] [Google Scholar]
- 44.Qin Z, Blankenstein T. A cancer immunosurveillance controversy. Nat Immunol 2004;5(1):3–4 [DOI] [PubMed] [Google Scholar]
- 45.Smyth MJ. Clarification of data used in three studies on MCA-induction of sarcoma in mice. Blood 2008;111(8):4419. [DOI] [PubMed] [Google Scholar]
- 46.Kammertoens T, Qin Z, Briesemeister D, Bendelac A, Blankenstein T. B-cells and IL-4 promote methylcholanthrene-induced carcinogenesis but there is no evidence for a role of T/NKT-cells and their effector molecules (Fas-ligand, TNF-alpha, perforin). Int J Cancer 2012;131(7):1499–508 [DOI] [PubMed] [Google Scholar]
- 47.Dethlefsen LA, Prewitt JM, Mendelsohn ML. Analysis of tumor growth curves. J Natl Cancer Inst 1968;40(2):389–405 [DOI] [PubMed] [Google Scholar]
- 48.Kranz DM, Tonegawa S, Eisen HN. Attachment of an anti-receptor antibody to non-target cells renders them susceptible to lysis by a clone of cytotoxic T lymphocytes. Proc Natl Acad Sci U S A 1984;81(24):7922–6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ubeda C, Taur Y, Jenq RR, Equinda MJ, Son T, Samstein M, et al. Vancomycin-resistant Enterococcus domination of intestinal microbiota is enabled by antibiotic treatment in mice and precedes bloodstream invasion in humans. J Clin Invest 2010;120(12):4332–41 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nossa CW, Oberdorf WE, Yang L, Aas JA, Paster BJ, Desantis TZ, et al. Design of 16S rRNA gene primers for 454 pyrosequencing of the human foregut microbiome. World J Gastroenterol 2010;16(33):4135–44 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Pruesse E, Quast C, Knittel K, Fuchs BM, Ludwig W, Peplies J, et al. SILVA: a comprehensive online resource for quality checked and aligned ribosomal RNA sequence data compatible with ARB. Nucleic Acids Res 2007;35(21):7188–96 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Wang Q, Garrity GM, Tiedje JM, Cole JR. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 2007;73(16):5261–7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lozupone C, Knight R. UniFrac: a new phylogenetic method for comparing microbial communities. Appl Environ Microbiol 2005;71(12):8228–35 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.McMurdie PJ, Holmes S. phyloseq: an R package for reproducible interactive analysis and graphics of microbiome census data. PLoS One 2013;8(4):e61217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wickham H ggplot2: Elegant Graphics for Data Analysis. Basel: Springer International Publishing; 2016. [Google Scholar]
- 56.Choudhury NJ, Kiyotani K, Yap KL, Campanile A, Antic T, Yew PY, et al. Low T-cell Receptor Diversity, High Somatic Mutation Burden, and High Neoantigen Load as Predictors of Clinical Outcome in Muscle-invasive Bladder Cancer. Eur Urol Focus 2016;2(4):445–52 [DOI] [PubMed] [Google Scholar]
- 57.Fang H, Yamaguchi R, Liu X, Daigo Y, Yew PY, Tanikawa C, et al. Quantitative T cell repertoire analysis by deep cDNA sequencing of T cell receptor alpha and beta chains using next-generation sequencing (NGS). Oncoimmunology 2014;3(12):e968467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kaplan EL, Meier P. Nonparametric-Estimation from Incomplete Observations. J Am Stat Assoc 1958;53(282):457–81 [Google Scholar]
- 59.Kalbfleisch JD, Prentice RL. The statistical analysis of failure time data. 2nd ed. Hoboken (NJ): John Wiley & Sons; 2002. [Google Scholar]
- 60.Gooley TA, Leisenring W, Crowley J, Storer BE. Estimation of failure probabilities in the presence of competing risks: new representations of old estimators. Stat Med 1999;18(6):695–706 [DOI] [PubMed] [Google Scholar]
- 61.Singh S, Ross SR, Acena M, Rowley DA, Schreiber H. Stroma is critical for preventing or permitting immunological destruction of antigenic cancer cells. J Exp Med 1992;175(1):139–46 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ochsenbein AF, Klenerman P, Karrer U, Ludewig B, Pericin M, Hengartner H, et al. Immune surveillance against a solid tumor fails because of immunological ignorance. Proc Natl Acad Sci U S A 1999;96(5):2233–8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ubeda C, Lipuma L, Gobourne A, Viale A, Leiner I, Equinda M, et al. Familial transmission rather than defective innate immunity shapes the distinct intestinal microbiota of TLR-deficient mice. J Exp Med 2012;209(8):1445–56 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Hoel DG, Walburg HE Jr. Statistical analysis of survival experiments. J Natl Cancer Inst 1972;49(2):361–72 [PubMed] [Google Scholar]
- 65.Sato F, Noda K, Kato H, Murakami M, Endoh D, Hashimoto N. An analysis of experimental radiation carcinogenesis with model setting for competing risks. J Radiat Res 1990;31(2):147–55 [DOI] [PubMed] [Google Scholar]
- 66.Schietinger A, Philip M, Liu RB, Schreiber K, Schreiber H. Bystander killing of cancer requires the cooperation of CD4(+) and CD8(+) T cells during the effector phase. J Exp Med 2010;207(11):2469–77 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Rogers LM, Olivier AK, Meyerholz DK, Dupuy AJ. Adaptive immunity does not strongly suppress spontaneous tumors in a Sleeping Beauty model of cancer. J Immunol 2013;190(8):4393–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Baker DG. Natural pathogens of laboratory mice, rats, and rabbits and their effects on research. Clin Microbiol Rev 1998;11(2):231–66 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Schreiber K, Rowley DA, Riethmuller G, Schreiber H. Cancer immunotherapy and preclinical studies: why we are not wasting our time with animal experiments. Hematol Oncol Clin North Am 2006;20(3):567–84 [DOI] [PubMed] [Google Scholar]
- 70.Walburg HE Jr., Cosgrove GE, Upton AC. Influence of microbial environment on development of myeloid leukemia in x-irradiated RFM mice. Int J Cancer 1968;3(1):150–4 [DOI] [PubMed] [Google Scholar]
- 71.Song SJ, Lauber C, Costello EK, Lozupone CA, Humphrey G, Berg-Lyons D, et al. Cohabiting family members share microbiota with one another and with their dogs. Elife 2013;2:e00458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Pabst O, Cerovic V, Hornef M. Secretory IgA in the Coordination of Establishment and Maintenance of the Microbiota. Trends Immunol 2016;37(5):287–96 [DOI] [PubMed] [Google Scholar]
- 73.Shulzhenko N, Morgun A, Hsiao W, Battle M, Yao M, Gavrilova O, et al. Crosstalk between B lymphocytes, microbiota and the intestinal epithelium governs immunity versus metabolism in the gut. Nat Med 2011;17(12):1585–93 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Karst SM, Wobus CE, Lay M, Davidson J, Virgin HWt. STAT1-dependent innate immunity to a Norwalk-like virus. Science 2003;299(5612):1575–8 [DOI] [PubMed] [Google Scholar]
- 75.Hsu CC, Piotrowski SL, Meeker SM, Smith KD, Maggio-Price L, Treuting PM. Histologic Lesions Induced by Murine Norovirus Infection in Laboratory Mice. Vet Pathol 2016;53(4):754–63 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Rooney MS, Shukla SA, Wu CJ, Getz G, Hacohen N. Molecular and genetic properties of tumors associated with local immune cytolytic activity. Cell 2015;160(1–2):48–61 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Van den Eynden J, Jimenez-Sanchez A, Miller ML, Larsson E. Lack of detectable neoantigen depletion signals in the untreated cancer genome. Nat Genet 2019;51(12):1741–8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Coulie PG, Lehmann F, Lethe B, Herman J, Lurquin C, Andrawiss M, et al. A mutated intron sequence codes for an antigenic peptide recognized by cytolytic T lymphocytes on a human melanoma. Proc Natl Acad Sci U S A 1995;92(17):7976–80 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Matsushita H, Vesely MD, Koboldt DC, Rickert CG, Uppaluri R, Magrini VJ, et al. Cancer exome analysis reveals a T-cell-dependent mechanism of cancer immunoediting. Nature 2012;482(7385):400–4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Vogelstein B, Papadopoulos N, Velculescu VE, Zhou S, Diaz LA Jr., Kinzler KW. Cancer genome landscapes. Science 2013;339(6127):1546–58 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Wolfel T, Hauer M, Schneider J, Serrano M, Wolfel C, Klehmann-Hieb E, et al. A p16INK4a-insensitive CDK4 mutant targeted by cytolytic T lymphocytes in a human melanoma. Science 1995;269(5228):1281–4 [DOI] [PubMed] [Google Scholar]
- 82.Gejman RS, Chang AY, Jones HF, DiKun K, Hakimi AA, Schietinger A, et al. Rejection of immunogenic tumor clones is limited by clonal fraction. Elife 2018;7 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.