Abstract
Objective
To compare progression of subclinical atherosclerosis and factors promoting it in patients with SLE and controls.
Methods
Consecutive patients with SLE and age-matched, sex-matched population controls from the SLEVIC cohort were assessed at inclusion and after 7 years with standardised data collection and carotid ultrasound. Effect of risk factors on carotid intima–media thickness (cIMT) progression was examined with adjusted linear mixed models.
Results
A total of 77 patients and 74 controls, 68% and 61% of the original cohort, completed follow-up. The patients were (mean) 47 years old, 90% were women, and controls were 51 years old, 92% women. Patients had disease duration of (mean) 11 years, mild disease activity and low severity at both assessments. Baseline cIMT did not differ between the groups. An average absolute cIMT progression was 0.009 mm/year in patients and 0.011 mm/year in controls, intergroup difference p=0.9.
Of factors at inclusion, dyslipidaemia, lower levels of high-density lipoprotein (HDL) and carotid plaque in patients and controls, and higher systolic blood pressure, total cholesterol:HDL and LDL:HDL ratios and triglycerides in patients were associated with cIMT progression. Of factors at follow-up, hypertension and blood lipids in patients and HDL in controls were significantly associated with cIMT progression. History of lupus nephritis and a higher average dose of prednisolone used since diagnosis were associated with cIMT progression in patients. Associations of risk factors with cIMT progression were stronger in presence of plaques.
Conclusion
We observed a statistically comparable progression of cIMT in patients with mild SLE and controls over 7 years, which implies that progression of subclinical atherosclerosis in some patients with SLE could follow that of the general population. Traditional cardiovascular (CV) risk factors, history of lupus nephritis and higher use of corticosteroids promote cIMT progression in SLE. Detection of carotid plaque may add to CV risk stratification.
Keywords: systemic lupus erythematosus, cardiovascular risk factors, carotid atherosclerosis
Introduction
SLE is an autoimmune systemic disease which is characterised by flares, has a significant impact on quality of life and may lead to severe accumulated damage in the long term.1–3
Atherosclerosis is an inflammatory condition, characterised by the presence of immune competent cells producing cytokines and apoptotic cells in the lesions.4 The excess cardiovascular (CV) risk in patients with SLE is well recognised.5–7 CV events are the leading cause of morbidity and mortality in SLE and prevention of progression of atherosclerosis to clinically manifest atherosclerosis is an important task. Genetic factors, traditional risk factors such as smoking, hypertension, hyperlipidaemia, diabetes mellitus and obesity, and disease factors, for example, SLE-related immune activity, accumulated disease damage and treatments contribute to vessel changes and accelerated atherosclerosis in SLE.8–13 It is unclear whether contribution of classical CV risk factors and inflammatory factors to vascular changes is different in patients with SLE in comparison with the general population. There is increasing evidence that disease control could improve the long-term outcomes; however, whether SLE treatments would arrest the excess of atherosclerosis is not established.
Therefore, we aimed to examine which factors promote and protect for atherosclerosis progression in patients with SLE and population controls, and to compare the atherosclerosis progression in patients and controls. We took advantage of the case–control population of patients with SLE and age-matched and sex-matched population controls who were prospectively followed 7 years after inclusion into the original cohort.14 Carotid intima–media thickness (cIMT) and carotid plaques were used as a surrogate measure of subclinical atherosclerosis.15 16
Patients and methods
Patients
The study sample for this 7-year follow-up analysis originated from the previously described single-centre matched control population of the SLEVIC cohort (SLE vascular impact cohort study).14 In brief, 114 patients with SLE, who fulfilled the 1982 revised criteria of the American College of Rheumatology for SLE17 and were younger than 70 years, and 122 age-matched and sex-matched controls were enrolled to the SLEVIC cohort. Seven years after inclusion, all participants were asked to participate in the follow-up investigation. Of all, 77 patients and 74 controls participated in the follow-up and were included in this prospective longitudinal analysis (online supplementary figure 1).
lupus-2019-000362supp001.pdf (64.1KB, pdf)
There were no statistically significant differences in distribution of age, traditional CV risk factors and prevalent CV events at inclusion into the original cohort between participants of the follow-up analysis (both patients and controls) and those who were lost to follow-up. Also, there was no significant difference in SLE disease characteristics between participants of the follow-up and those who dropped out from the cohort. The baseline cIMT was numerically lower in patients participating in the follow-up assessment than in those who were not followed, mean (SD) cIMT of 0.607 (0.123) versus 0.631 (0.147), p=0.4, but there was no difference between controls participating in the follow-up and those who were not followed, mean cIMT of 0.629 (0.114) versus 0.625 (0.142), p=0.9. Likewise, prevalence of carotid plaque at inclusion in patients participating in the follow-up assessment was lower than in drops-off, 53% versus 39%, p=0.16, but did not differ in controls who were followed or not, 30% versus 31%, p=0.9.
Data collection
Structured data collection was performed at inclusion and at 7-year assessment, including complete physical examination. Information was collected on CV risk factors including history of smoking (ever or never), history of hypertension, prescription of antihypertensive drugs or blood pressure ≥140/90 mm Hg at the assessments, history of diabetes mellitus, prescription of antidiabetic drugs, fasting blood glucose ≥7.0 mmol/L, history of dyslipidaemia or lipid-lowering medication prescription, low-density lipoprotein (LDL) >3.4 mmol/L, high-density lipoprotein (HDL) <1.0 mmol/L and obesity (body mass index (BMI) ≥30 kg/m2), and family history of CV disease. History of atherosclerotic CV event was recorded (acute myocardial infarction, bypass grafting or percutaneous artery intervention, ischaemic stroke, transient ischaemic attack).
For patients with SLE, history of nephritis and antiphospholipid syndrome (APS) was recorded and SLE disease activity was assessed with the Systemic Lupus Erythematosus Diseases Activity Index18 without the laboratory tests, and organ damage was measured using the Systemic Lupus International Collaborating Clinics (SLICC) damage index.19 Flares at any time during the 7-year follow-up were recorded. Flare was defined as new symptoms or worsening of symptoms related to the SLE disease requiring adaptation of disease-modifying antirheumatic drugs and/or glucocorticoids.
Carotid ultrasound
Carotid ultrasound was performed at inclusion and at 7-year follow-up assessment at the same laboratory as described in detail previously.14 The right and left carotid arteries were examined with a duplex scanner (Sequoia; Siemens Acuson, Mountain View, California, USA) using a 6 MHz linear array transducer. The far wall of the common carotid artery (CCA), 0.5 to 1.0 cm proximal to the beginning of the carotid bulb, was used for measurements of the cIMT. The cIMT was defined as the distance between the leading edge of the lumen–intima echo and the leading edge of the media–adventitia echo. The examinations were digitally stored for subsequent analyses by a computer system.20 When a plaque was observed in the region of the CCA measurements, the IMT was not measured. The mean values of the cIMT within the 10 mm long section were calculated. The mean cIMT, (cIMT right+cIMT left)/2, was calculated. The difference between repeated measurements of cIMT was 4.9% (coefficient of variation) by using the automated analysing system.
At inclusion, carotid plaque was defined as a localised intima–media thickening of >1 mm and at least a 100% increase in thickness compared with adjacent wall segments. Plaque was screened for in the common, internal and external carotid arteries.
Statistical methods
Descriptive statistics are reported as means (SD) for continuous and percentages for categorical variables. To compare variables at baseline and follow-up assessments, one-way ANOVA, Mann-Whitney U test, χ2, Fisher’s exact test or McNemar’s test was used, as suitable.
To analyse the association of traditional risk factors with cIMT progression, linear mixed models were applied. Because of strong effect of age and sex on progression of cIMT, we examined first which risk factors were significantly associated with the changes in cIMT between the two assessments, adjusted for age and sex (model 1). Second, the factors with statistical significance p value <0.10 were further tested in the multivariate models additionally controlled for traditional CV risk factors (covariates), that is, smoking, hypertension, diabetes mellitus, dyslipidaemia, BMI and family history of CV disease (model 2). To allow different effects of independent variables and covariates over time, interaction terms by assessment visit were included in the multivariate models. Effect modification of common CV risk factors with carotid plaque was investigated with their corresponding interaction terms.
Level of statistical significance was set at α <0.05. IBM SPSS V.25 was used for the analyses.
Results
The characteristics of patients and controls who completed the follow-up and participated in this study are shown in tables 1 and 2. Data on the original SLEVIC-cohort population have been described previously.14 As shown in table 1, the patients had at inclusion lower levels of LDL-cholesterol than controls but were more likely to have higher levels of hypertension and triglycerides. Between the baseline and the 7-year follow-up visit, an increase in systolic and diastolic blood pressure, levels of total cholesterol, LDL, BMI and waist circumference were observed both in patients and controls. Reflecting disease-related dyslipidaemia, however, the patients still had lower total cholesterol (TC), LDL and HDL-cholesterol levels and higher triglycerides than controls at 7-year assessment, without significant between-group difference in other cardiometabolic and traditional CV risk factors (table 1). At both assessments, patients used more frequent antihypertensive agents than controls, and patients used also more likely aspirin than controls. In patients at inclusion, aspirin was given in 26% due to antiphospholipid antibodies (aPL) positivity, in 37% as general CV risk prevention and in 37% due to both reasons. The patients experienced CV events numerically more likely than controls at inclusion assessment, 9.1% versus 2.7%, p=0.17, and the cumulative number of events at follow-up was higher in patients, 15.6% versus 5.4%, p=0.042 (table 1). Seven patients and two controls were known with previous coronary CV event (n=4) and ischaemic cerebrovascular events (n=6).
Table 1.
Baseline and follow-up descriptive in 77 patients with SLE and 74 controls
| SLE | P value | Controls | P value | |||
| Baseline | Follow-up | Baseline | Follow-up | |||
| Age, years | 46.7 (13.6) | 54.0 (13.7) | NA | 50.5 (11.5) | 57.7 (11.7) | NA |
| Male sex % (n) | 13 (10) | NA | NA | 11 (8) | NA | NA |
| CV clinical and laboratory characteristics | ||||||
| Systolic blood pressure (mm Hg) | 127.2 (20.5) | 133.4 (22.0) | 0.011 | 121.5 (19.1) | 136.4 (22.5) | < 0.001 |
| Diastolic blood pressure (mm Hg) | 79.2 (12.7) | 84.9 (11.1) | < 0.001 | 76.5 (11.0) | 85.5 (10.3) | < 0.001 |
| Blood pressure ≥140/90 mm Hg % | 39.0* | 57.3 | 0.011 | 23.0 | 56.2 | < 0.001 |
| Total cholesterol (mmol/L) | 4.6 (1.1) | 4.8 (1.1)* | 0.048 | 4.9 (1.0) | 5.3 (1.0) | < 0.001 |
| LDL-cholesterol (mmol/L) | 2.5 (0.9)* | 2.7 (0.9)* | 0.051 | 2.8 (0.8) | 3.1 (1.0) | < 0.001 |
| HDL-cholesterol (mmol/L) | 1.6 (0.5)* | 1.6 (0.4)* | 0.9 | 1.8 (0.6) | 1.8 (0.5) | 0.6 |
| TC:HDL ratio | 3.1 (2.9) | 3.2 (2.9) | 0.3 | 3.0 (0.9) | 3.2 (0.9) | 0.021 |
| LDL:HDL ratio | 1.73 (0.82) | 1.79 (0.87) | 0.4 | 1.75 (0.75) | 1.90 (0.83) | 0.035 |
| Triglycerides (mmol/L) | 1.1 (0.5)* | 1.2 (0.6)* | 0.1 | 0.8 (0.4) | 0.9 (0.4) | 0.081 |
| BMI (kg/m2) | 24.6 (4.6) | 26.2 (5.4) | < 0.001 | 25.2 (4.2) | 26.2 (25.5) | 0.001 |
| Waist circumference (cm) | 84.6 (15.3) | 90.1 (14.3) | 0.001 | 85.5 (11.2) | 88.5 (12.2) | 0.003 |
| CRP (mg/L), median (IQR) | 1.4 (0.7–3.8) | 1.6 (0.5–4.4) | 0.7 | 1.1 (0.5–2.6) | 0.8 (0.5–2.6) | 0.8 |
| ESR (mm/h) | 22.9 (17.4)* | 21.4 (17.9)* | 0.4 | 9.8 (5.6) | 12.0 (9.4) | 0.021 |
| Serum creatinine (µmol/L) | 77.0 (26.9) | 75.1 (30.9) | 0.050 | 69.0 (11.3) | 67.2 (11.7) | 0.2 |
| Traditional CV risk factors, % | ||||||
| Smoking ever | 55.8 | 57.3 | 0.500 | 51.4 | 56.2 | 0.3 |
| Hypertension | 55.8* | 70.7 | 0.013 | 25.7 | 56.2 | < 0.001 |
| Diabetes mellitus | 2.6 | 9.3 | 0.063 | 2.7 | 4.1 | 1.0 |
| Dyslipidaemia | 33.8 | 40.5 | 0.5 | 37.8 | 54.8 | 0.002 |
| Obesity | 13.9 | 21.3 | 0.109 | 9.5 | 17.8 | 0.070 |
| Family history of CV disease | 26.0 | 31.9 | 0.3 | 32.4 | 41.7 | 0.016 |
| History of CV events % (n)† | 9.1% (7) | 15.6% (12) | 0.063 | 2.7% (2) | 5.4% (4) | 0.5 |
| Current medications % | ||||||
| Antihypertensive | 45.5* | 50.7* | 0.5 | 8.1 | 17.8 | 0.016 |
| Lipid lowering | 9.1 | 17.3 | 0.070 | 6.8 | 9.6 | 0.6 |
| ASA | 24.7* | 33.3* | 0.070 | 4.1 | 4.2 | 1.0 |
| Carotid ultrasound measurements | ||||||
| Mean cIMT (mm) | 0.607 (0.123) | 0.670 (0.125) | < 0.001 | 0.629 (0.114) | 0.701 (0.130) | < 0.001 |
| Mean cIMT absolute progression (mm) | 0.063 (0.099) | NA | 0.077 (0.093) | NA | ||
| Cartoid plaque % (n) | 39.0%(30) | – | NA | 35.1%(27) | – | NA |
Values are means (SD) unless noted otherwise; p value represents the comparisons within the groups at baseline and follow-up assessments.
*if p<0.05 for between-group differences at the same time-point assessment.
†CV events at follow-up include the events present at baseline.
ASA, low-dose aspirin; BMI, body mass index; cIMT, carotid intima–media thickness; CRP, C reactive protein; CV, cardiovascular; ESR, erythrocyte sedimentation rate; HDL, high-density lipoprotein; LDL, low-density lipoprotein; NA, not available; TC, total cholesterol.
Table 2.
Disease characteristics of 77 patients with SLE
| Baseline | Follow-up | P value | |
| Disease duration, years | 11.4 (8.9) | 18.7 (9.0) | NA |
| History of nephritis % | 35.6 | 45.8 | 0.5 |
| Flare during follow-up % | NA | 69.3 | NA |
| SLEDAI | 3.0 (4.1) | 1.6 (2.7) | 0.008 |
| SLICC | 1.1 (1.5) | 2.1 (1.4) | < 0.001 |
| History of APS % | NA | 24.6 | – |
| aPL antibodies % | 44.9 | NA | – |
| Current SLE medications % | |||
| Prednisolone | 59.7 | 53.3 | 0.3 |
| Current dose, mg/day | 6.3 (4.5) | 6.1 (3.5) | 0.9 |
| HCQ | 51.9 | 43.2 | 0.3 |
| AZA | 19.5 | 10.7 | 0.1 |
| MTX | 10.4 | 6.7 | 0.7 |
| MMF | 7.8 | 17.3 | 0.065 |
| No DMARDs | 10.4 | 25.3 | 0.003 |
| History of SLE medications (ever) % | |||
| Prednisone | 92.2 | 97.4 | 0.3 |
| Cumulative duration, months | 67.5 (67.8) | 117.3 (97.2) | NA |
| Cumulative dose, g | 16.7 (15.0) | 26.7 (21.7) | NA |
| Average dose, mg/day | 4.1 (3.3) | 4.2 (4.2) | 0.3 |
| HCQ | 87.0 | 93.5 | 0.063 |
| Cyclophosphamide and/or rituximab | 16.9 | 35.6 | < 0.001 |
Values are means (SD) unless noted otherwise; p value represents the comparisons between baseline and follow-up assessments.
aPL, antiphospholipid antibodies; APS, antiphospholipid syndrome; AZA, azathioprine; DMARDs, disease-modifying antirheumatic drugs; HCQ, hydroxychloroquine; MMF, mycophenolate mofetil; MTX, methotrexate; NA, not available; SLEDAI, Systemic Lupus Erythematosus Disease Activity Index (without laboratory tests); SLICC, Systemic Lupus International Collaborating Clinics (damage index).
The disease characteristics of the patients in this analysis are shown in table 2. The patients had mostly long-standing disease of low disease activity and low severity at both assessments. Two-thirds of patients experienced flares during follow-up, and almost half of the patients had ever history of lupus nephritis. Ever use of prednisolone and hydroxychloroquine was common.
Progression of cIMT in patients and controls
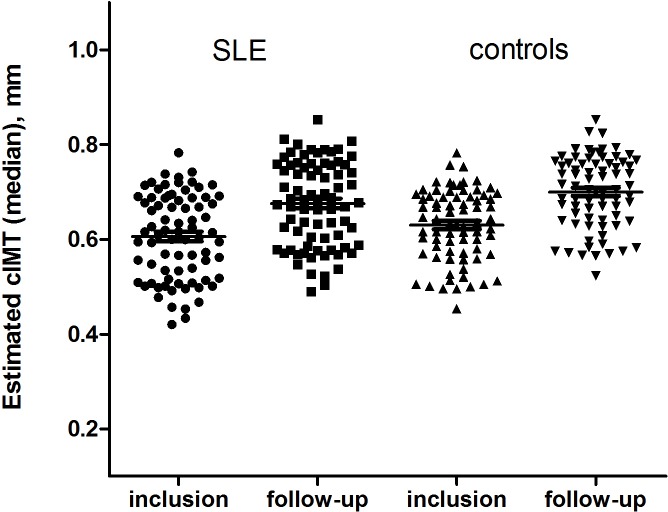
There was no difference in progression of cIMT between the patients and controls. The mean cIMT increased significantly between the two assessments in both groups, with an averaged absolute progression of 0.009 mm per year in patients and 0.011 mm per year in controls (table 2), between-group difference p=0.867, age and sex adjusted (figure 1).
Figure 1.
Progression of carotid intima–media thickness (cIMT) over 7-year follow-up in patients with SLE and controls. Presented are predicted medians of cIMT estimated with linear mixed model adjusted for age and sex in patients and controls, per group.
Association of common risk factors with progression of cIMT over 7 years in patients and controls
To investigate whether and which common risk factors at inclusion affected cIMT progression, a linear mixed model was used. The results are presented in table 3.
Table 3.
Association of common risk factors at inclusion and follow-up with carotid intima–media thickness (cIMT) over 7 years in patients with SLE and controls
| Effect of risk factors at inclusion | Effect of risk factors at follow-up | |||
| Beta coefficient (95% CI) | P value | Beta coefficient (95% CI) | P value | |
| Systolic blood pressure | ||||
| SLE Model 1 | 0.0014 (0.0004 to 0.0025) | 0.008* | 0.0006 (−0.0005 to 0.0016) | 0.3 |
| Model 2 | 0.0015 (0.0003 to 0.0026) | 0.013† | NS | |
| Controls Model 1 | 0.0011 (−0.001 to 0.0022) | 0.052 | 0.0007 (−0.0003 to 0.0017) | |
| Model 2 | 0.0010 (−0.0002 to 0.0021) | 0.102 | NS | 0.1 |
| Diastolic blood pressure | ||||
| SLE Model 1 | 0.0016 (0.0001 to 0.0031) | 0.047 | 0.0005 (−0.0013 to 0.0024) | 0.6 |
| Model 2 | 0.0016 (0.0 to 0.0033) | 0.061 | NS | |
| Controls Model 1 | 0.0001 (−0.002 to 0.0019) | 0.9 | −0.0012 (−0.0032 to 0.0008) | 0.3 |
| Total cholesterol | ||||
| SLE Model 1 | −0.0018 (−0.0203 to 0.0167) | 0.8 | −0.0059 (−0.0248 to 0.0129) | 0.5 |
| Controls Model 1 | −0.0089 (−0.0295 to 0.0117) | 0.4 | 0.0191 (−0.0027 to 0.0409) | 0.085 |
| Model 2 | NS | 0.0236 (0.0002 to 0.0475) | 0.052 | |
| LDL-cholesterol | ||||
| SLE Model 1 | 0.0060 (−0.0170 to 0.0293) | 0.6 | 0.0042 (−0.0187 to 0.0270) | 0.7 |
| Controls Model 1 | 0.0001 (−0.0235 to 0.0253) | 0.9 | 0.0117 (−0.0101 to 0.0335) | 0.3 |
| HDL-cholesterol | ||||
| SLE Model 1 | −0.0573 (−0.9889 to −0.0158) | 0.007* | −0.0850 (−0.1292 to −0.0408) | <0.001* |
| Model 2 | −0.0598 (−0.1026 to −0.0171) | 0.007† | −0.0922 (−0.1421 to −0.0424) | <0.001† |
| Controls Model 1 | −0.0401 (−0.0729 to −0.0075) | 0.017* | −0.0481 (−0.0921 to −0.0041) | 0.033* |
| Model 2 | −0.0359 (−0.0707 to −0.0011) | 0.043 | −0.0573 (−0.1042 to −0.0103) | 0.018† |
| TC:HDL ratio | ||||
| SLE Model 1 | 0.0272 (0.0094 to 0.0450) | 0.003* | 0.0245 (0.0056 to 0.0434) | 0.012* |
| Model 2 | 0.0312 (0.0140 to 0.0483) | 0.001† | 0.0277 (0.0080 to 0.0473) | 0.006† |
| Controls Model 1 | 0.0156 (−0.0067 to 0.0378) | 0.2 | 0.0090 (−0.0129 to 0.0310) | 0.4 |
| LDL:HDL ratio | ||||
| SLE Model 1 | 0.0336 (0.0105 to 0.0567) | 0.005* | 0.0277 (0.0040 to 0.0514) | 0.023* |
| Model 2 | 0.0402 (0.0179 to 0.0625) | 0.001† | 0.0316 (0.0072 to 0.0560) | 0.012† |
| Controls Model 1 | 0.0169 (−0.0095 to 0.0434) | 0.2 | 0.0079 (−0.0166 to 0.0325) | 0.5 |
| Triglycerides | ||||
| SLE Model 1 | 0.0431 (0.0067 to 0.0797) | 0.021* | 0.0276 (−0.0049 to 0.0600) | 0.095 |
| Model 2 | 0.0400 (0.0033 to 0.0767) | 0.033† | 0.0318 (−0.0043 to 0.0679) | 0.083 |
| Controls Model 1 | −0.0221 (−0.0692 to 0.0251) | 0.4 | 0.0211 (−0.0268 to 0.0689) | 0.4 |
| BMI | ||||
| SLE Model 1 | 0.0041 (−0.0003 to 0.0086) | 0.064 | 0.0023 (−0.0014 to 0.0060) | 0.2 |
| Model 2 | 0.0029 (−0.0018 to 0.0077) | 0.2 | NS | |
| Controls Model 1 | 0.0022 (−0.0026 to 0.0070) | 0.4 | 0.0008 (−0.0038 to 0.0055) | 0.7 |
| Smoking ever vs never | ||||
| SLE Model 1 | 0.0056 (−0.0351 to 0.0463) | 0.8 | 0.0092 (−0.0322 to 0.0507) | 0.7 |
| Controls Model 1 | 0.0127 (−0.0275 to 0.0259) | 0.5 | 0.0125 (−0.0290 to 0.0539) | 0.6 |
| Hypertension | ||||
| SLE Model 1 | 0.0527 (0.0125 to 0.0928) | 0.011* | 0.0452 (−0.0004 to 0.0909) | 0.052 |
| Model 2 | 0.0404 (−0.0030 to 0.0838) | 0.067 | 0.0579 (0.0092 to 0.1066) | 0.020† |
| Controls Model 1 | 0.0083 (−0.0403 to 0.0570) | 0.7 | 0.0027 (−0.0406 to 0.0460) | 0.9 |
| Diabetes mellitus | ||||
| SLE Model 1 | 0.0735 (−0.0502 to 0.1972) | 0.2 | 0.0089 (−0.0622 to 0.0801) | 0.8 |
| Controls Model 1 | 0.0137 (−0.1111 to 0.1385) | 0.8 | 0.0138 (−0.0893 to 0.1170) | 0.8 |
| Dyslipidaemia | ||||
| SLE Model 1 | 0.0362 (−0.0050 to 0.0773) | 0.084 | 0.0624 (0.0201 to 0.1048) | 0.004 |
| Model 2 | 0.0506 (0.0084 to 0.0928) | 0.019† | 0.0788 (0.0328 to 0.1249) | 0.001† |
| Controls Model 1 | 0.0501 (0.0010 to 0.0903) | 0.015* | 0.0228 (−0.0186 to 0.0643) | 0.3 |
| Model 2 | 0.0536 (0.0125 to 0.0947) | 0.011† | NS | |
| Obesity | ||||
| SLE Model 1 | 0.0368 (−0.0209 to 0.0945) | 0.2 | 0.0379 (−0.0102 to 0.0859) | 0.1 |
| Controls Model 1 | 0.0404 (−0.0271 to 0.1079) | 0.2 | 0.0181 (−0.0351 to 0.0712) | 0.5 |
| Family history of CV disease | ||||
| SLE Model 1 | 0.0017 (−0.0428 to 0.0462) | 0.9 | 0.0040 (−0.0392 to 0.0471) | 0.9 |
| Controls Model 1 | 0.0281 (−0.0155 to 0.0718) | 0.2 | 0.0383 (−0.0076 to 0.0774) | 0.068 |
| Model 2 | NS | 0.0368 (−0.0005 to 0.0783) | 0.081† | |
| Carotid plaque at inclusion | ||||
| SLE Model 1 | 0.0949 (0.0588 to 0.1331) | <0.001 | – | – |
| Model 2 | 0.0756 (0.0353 to 0.1158) | <0.001† | ||
| Controls Model 1 | 0.0583 (0.0116 to 0.1050) | 0.015 | ||
| Model 2 | 0.0667 (0.0221 to 0.1114) | 0.004† | ||
Presented results of beta coefficients with 95% CI are based on mixed linear regression models with two measurements of mean cIMT overtime as response.
Model 1 was adjusted for age and sex.
Model 2 was run only for the variables with p value <0.1 in model 1, and included, in addition to age and sex, traditional CV risk factors as ever smoking, hypertension (or systolic/diastolic blood pressure if indicated by analysis), diabetes mellitus, dyslipidaemia (or blood lipids if indicated by analysis), BMI, family history of CV disease and interaction terms of tested independent variables by assessment visit.
Results indicated as NS if p value >0.10 in model 2.
*P value <0.05 withal by additional adjustment for medication prescription at inclusion or follow-up, if indicated.
†P value <0.05 withal by additional adjustment for history of atherosclerotic CV event.
BMI, body mass index; CV, cardiovascular; HDL, high-density lipoprotein; LDL, low-density lipoprotein; NS, not significant; TC, total cholesterol.
In patients, a significant association was identified between cIMT progression and higher systolic and diastolic blood pressure, lower levels of HDL, higher TC:HDL and LDL:HDL ratios, higher triglycerides, dyslipidaemia and carotid plaque at inclusion. The significant association between these factors, with exception for diastolic blood pressure, remained in multivariate analyses after additional adjustment for other traditional risk factors, prescription of antihypertensive, lipid-lowering drugs and prevalent CV events at inclusion.
In the control group, cIMT progression associated with lower HDL, dyslipidaemia and carotid plaque at inclusion, independently of traditional CV risk factors. Controlling for medication prescription and prevalent CV event did not change the association.
To investigate the effect of accumulated burden of risk factors on cIMT progression, we analysed whether factors measured at 7-year assessment were associated with progression of cIMT. In patients, independent effect of hypertension, lower HDL, higher TC:HDL ratio and LDL:HDL ratio at follow-up was confirmed. In the control group, lower levels of HDL were independent of other traditional CV risk factors associated with cIMT progression.
Of all examined risk factors, the strongest association with cIMT progression was shown for lower HDL at both assessments in patients and controls. Moreover, rate of cIMT progression depended on HDL levels at inclusion, beta coefficient (95% CI) β=−0.0358 (−0.0082 to −0.0634), p=0.011, difference between patients and controls was not significant, p=0.6.
To examine whether associations between common risk factors and progression of cIMT progression were different in patients with SLE and in controls, group (SLE vs controls) as cofactor was included in the model adjusted for age and sex. These analyses did not show any statistically significant between-group differences.
SLE disease-related factors in relation to progression of cIMT over 7 years
In multivariate analyses in patients, history of lupus nephritis at inclusion and follow-up was associated with cIMT progression independently of age, sex, traditional CV risk factors and history of atherosclerotic disease. Disease measures, having flares during follow-up, history of APS or positivity for aPL antibodies were not associated with cIMT progression in analyses adjusted for age and sex (table 4).
Table 4.
Carotid intima–media thickness over 7 years in association with disease factors at inclusion and follow-up in patients with SLE
| Effect of risk factors at inclusion | Effect of risk factors at follow-up | |||
| Beta coefficient (95% CI) | P value | Beta coefficient (95% CI) | P value | |
| Disease duration | ||||
| Model 1 | 0.0013 (−0.0007 to 0.0032) | 0.2 | 0.0011 (−0.0011 to 00032) | 0.3 |
| History of lupus nephritis | ||||
| Model 1 | 0.0413 (0.0053 to 0.0744) | 0.025 | 0.0506 (0.0109 to 0.0902) | 0.013 |
| Model 2 | 0.0482 (0.0128 to 0.0837) | 0.008* | 0.0573 (0.0190 to 0.0955) | 0.004* |
| Flare during follow-up | ||||
| Model 1 | - | 0.0046 (−0.0376 to 0.0469) | 0.8 | |
| SLEDAI | ||||
| Model 1 | 0.0017 (−0.0032 to 0.0065) | 0.5 | 0.0059 (−0.0013 to 0.0131) | 0.1 |
| SLICC | ||||
| Model 1 | 0.0042 (−0.0059 to 0.0142) | 0.4 | 0.0071 (−0.0028 to 0.0170) | 0.2 |
| History of APS | ||||
| Model 1 | NA | 0.0261 (−0.0205 to 0.0726) | 0.3 | |
| aPL antibodies | ||||
| Model 1 | 0.0050 (−0.0303 to 0.0402) | 0.8 | NA | |
| Prednisolone average dose | ||||
| Model 1 | 0.0590 (0.0193 to 0.0987) | 0.004 | 0.0160 (−0.1403 to 0.1723) | 0.8 |
| Model 2 | 0.0575 (0.0202 to 0.0948) | 0.003* | NS | |
| HCQ use | ||||
| Model 1 | −0.0186 (−0.0680 to 0.0308) | 0.5 | −0.0310 (−0493 to 0.1113) | 0.4 |
| DMARDs use | ||||
| Model 1 | 0.0022 (−0.0334 to 0.0380) | 0.9 | 0.0009 (−0.0385 to 0.0403) | 0.9 |
| Cyclophosphamide and/or rituximab ever use | ||||
| Model 1 | 0.0056 (−0.0385 to 0.0498) | 0.8 | 0.0018 (−0.0400 to 0.0436) | 0.9 |
Model 1 was adjusted for age and sex.
Model 2 included, in addition to age and sex, variables with p value <0.10 in model 1 and CV risk factors as ever smoking, systolic blood pressure, diabetes mellitus, HDL, BMI, family history of CV disease and interaction terms of tested independent variables by assessment visit.
Model 2 was run only for the variables with p value <0.1 in model 1 and included, in addition to age and sex, traditional CV risk factors as ever smoking, hypertension, diabetes mellitus, dyslipidaemia, BMI, family history of CV disease and interaction terms of tested independent variables by assessment visit.
NS if p value >0.10 in model 2.
DMARDs use: azathioprine, methotrexate, mycophenolate mofetil, ciclosporin.
*P value <0.05 withal by additional adjustment for history of atherosclerotic CV event.
aPL, anti-phospholipid antibodies; APS, anti-phospholipid syndrome; BMI, body mass index; CV, cardiovascular; DMARDs, disease-modifying antirheumatic drugs; HCQ, hydroxychloroquine; HDL, high-density lipoprotein; NA, not available; NS, not significant; SLEDAI, Systemic Lupus Erythematosus Disease Activity Index (without laboratory tests); SLICC, Systemic Lupus International Collaborating Clinics (damage index).
Analysing treatment effect, a higher average dose of prednisolone used since diagnosis was associated with a higher cIMT overtime in multivariate analyses. Use of immune-modulating therapies and hydroxychloroquine were not associated with cIMT progression in our study (table 4), but history of these therapies was about 90% at inclusion.
Effect measure modification with carotid plaque and drugs
To investigate whether the effect of identified risk factors on cIMT progression was different if carotid plaque was present at inclusion, an interaction term of these risk factors×plaque was added to the models. Because of the potential for type I error due to multiple tests and low sample size in the subgroups after stratification, findings for modification effect of carotid plaque should be interpreted as exploratory.
We found a significant interaction by carotid plaque in patients but not in controls for systolic and diastolic blood pressure, levels of HDL, TC:HDL and LDL:HDL ratios, triglycerides, defining with hypertension and dyslipidaemia, all p values <0.05 adjusted for age and sex. Association of these factors with progression of cIMT was stronger if plaques were present than if they were not present at inclusion into the study.
A positive additive interaction by carotid plaque for the association with progression of cIMT was observed for history of lupus nephritis at inclusion or follow-up, and average dose of prednisolone used before inclusion, adjusted for age and sex.
Because use of antimalarials is supposed to associate with improved CV prognosis, we investigated whether use of antimalarials could modify the association between traditional CV risk factors and cIMT progression. When including interaction product term of studied risk factors×ever use of antimalarials, no significant interactions were found. However, negative finding herewith should be interpreted with caution because use of antimalarials in our cohort was about 90%.
Discussion
In this 7-year surveillance study, we did not observe any statistically significant difference in progression of subclinical atherosclerosis in patients with mild SLE and population controls. This finding is important and encouraging because it implies that in patients with comparable characteristics as in the studied population, such as mild SLE disease, low disease damage, frequent use of antimalarials and CV preventive medications, progression of cIMT could follow the rate of progression of the general population. Nevertheless, traditional risk factors and detection of carotid plaques are important, and these factors were indeed associated with the progression of cIMT both in patients and controls in this study. In patients, further, history of lupus nephritis and use of glucocorticoids were associated with cIMT progression. The effect of the risk factors in patients was augmented in the presence of carotid plaque, suggesting that detection of carotid plaque could add for stratifying CV risk in patients.
In this 7-year follow-up carotid examination, cIMT progression was not different between patients with SLE with mild SLE disease and controls. The absence of significant difference in cIMT progression between SLE and controls suggests that vascular ageing in SLE may follow a pace of ‘normal’ vascular ageing. Taking into consideration the high burden of CV risk factors in patients with SLE, this finding is of major importance because it suggests that excess progression of cIMT in SLE could be halted and argues for the concept of possibility to change the disease prognosis in SLE by applying improved monitoring, therapeutic and preventive strategies.21 It should be emphasised that the rate of antimalarial use in this cohort (about 90%) compares favourably with many other published cohort studies (35%–70%),22–25 which could account for some of the lower cIMT progression in the patients with SLE in this study. Further, frequent usage of preventive medication with antihypertensives, statins and low-dose aspirin may also contribute to favourable evolution of cIMT in these patients. Although disease activity was assessed only at two assessments per protocol of the study, the low SLICC score of the accumulated disease damage at the second assessment and increasing use of preventive medications imply accurate monitoring of the SLE disease and CV risk factors through the follow-up. Optimal management of traditional risk factors together with maintenance of low disease activity and remission could further be reflected in reduced morbidity and mortality as well as a better health-related quality of life.26–28 The incidence of CV events in SLE in the modern era has declined, which supports the suggested favourable effects of active management of classic CV risk factors and better control of SLE-disease activity.29
In line with other reports,23 30 we observed an average absolute progression of mean cIMT of 0.009 mm/year in SLE and 0.011 mm/year in controls. Earlier reported values of cIMT progression in patients with SLE is broad, from 0.001 to 0.040 mm/year, which presumably relates to different SLE-population settings, for example, age at inclusion, duration of disease and follow-up.31 32 In the present patients, the prevalence of carotid plaque was 39%, which is in range of earlier observations of 17% to 40% in other SLE cohorts.15 16 23 The here observed prevalence of carotid plaque of 31% in controls is in 15%–55% range reported in the general population.30 33 34 In contrast, the recent meta-analysis has concluded that patients with SLE, compared with healthy individuals, have a significantly increased cIMT of 0.08 mm (95% CI 0.06 to 0.09) and twofold higher odds for having carotid plaque (OR 2.01, 95% CI 1.63 to 2.47), but included studies were markedly heterogeneous.15
It is interesting to note that cIMT progression in this cohort was similar between the patients and the control population, yet the number of CV events during the 7-year follow-up was higher in the patients with SLE. In a study of 200 patients with SLE who were recruited from the clinics in England and had a baseline clinical and CV risk assessment including carotid ultrasound measures, neither presence of carotid plaque nor cIMT at baseline predicted future events after a median of 6 years, but higher triglycerides, ‘ever’ exposure to cyclophosphamide and the damage index independently predicted CV disease events.22 Possible explanations for this could be that in SLE, risk factors for the initiation of atherosclerosis may be different to those important for progression and severity of atherosclerosis. It has been further suggested that the atherosclerotic plaques in SLE could be more vulnerable,14 a feature that is associated with a risk of occlusive events irrespective of the size of the plaque.
In this study, association of several risk factors with progression of subclinical atherosclerosis in SLE was proved. We reported here a significant association of several traditional CV risk factors with progression of cIMT in SLE. The relative importance of common atherosclerotic risk factors has been suggested to differ over time in SLE. Disease-related factors have been suggested to be more important for CV risk during early stages of SLE, while traditional CV risk factors, partially related to corticosteroid treatment, play a more significant role later in disease course.35 In the recent study, this view was, however, challenged by demonstrating that traditional CV risk factors, such as older age at diagnosis, hypertension, hypercholesterolemia, family history of CV disease and smoking, were associated with early development of myocardial infarction prior to the SLE diagnosis and within the first 2 years after diagnosis of SLE disease.36 In the present study, both common CV risk factors (as dyslipidaemia and hypertension) and disease-related factors (lupus nephritis and use of glucocorticoids) were found to promote progression of subclinical atherosclerosis in patients with long-standing disease (mean disease duration of 11 years). This argues for importance of recognition and surveillance of all CV risk factors through the whole disease course in patients with SLE.
Our findings support importance of lower levels of HDL and higher ratio of LDL for progression of subclinical atherosclerosis. The finding that levels of HDL were lower in SLE than in controls and that HDL levels were negatively associated with progression rate of cIMT could be related to protective properties of HDL, which includes an anti-inflammatory effect in autoimmunity and atherosclerosis.37–39 It has been reported that oxidised LDL, as determined by exposure of phosphorylcholine in LDL, is increased in SLE and associated with CV events.40 HDL could counteract such proinflammatory phospholipids and, thus, low levels of HDL in SLE may predispose for vessel inflammation and atherosclerotic changes.41 The factors protecting atherosclerotic progression in SLE deserve further studies.
Of disease-specific factors, history of lupus nephritis and a higher dose of prednisolone used since diagnosis influenced cIMT progression, independently of traditional CV risk factors. In line with our findings, cumulative corticosteroid dose has been reported to associate with progression of subclinical atherosclerosis, development of carotid plaque and progression of coronary artery calcium in SLE.42 43 In the recent case–control study, a twofold higher frequency of subclinical atherosclerosis defined with carotid plaque has been reported in patients with lupus nephritis compared with those without nephritis and to matched controls.44
An important finding in the present study is that presence of carotid plaque at inclusion modified the effect of traditional and SLE-specific factors on cIMT progression. The effects of these factors were stronger in presence of carotid plaque than in absence of plaque. This finding supports the use of carotid ultrasound examination, additive to traditional CV risk factors, in assessment of CV risk. It has been reported that the addition of cIMT measurement and presence of carotid plaque to the Framingham risk score improves net reclassification index for predicting CV events in the general population.45 In the cross-sectional study in patients with SLE, only 6% of patients fulfilled the definitions for high or very high risk according to the SCORE risk algorithms, but as many as 32% of patients were reclassified into a very high-risk category after ultrasound assessment.46
The present findings add to the data on importance of serial ultrasound measurements in assessing CV risk factors in SLE.47 Assessment of carotid plaque may predict the main outcome of CV events more accurate than progression of cIMT, but to facilitate the prospective studies in larger patient populations, standardisation of ultrasound assessment is needed. Significant discrepancies between studies largely depend on lack of such standardisation.48 Strict definition of carotid plaque in our study (a localised thickness of IMT >1 mm) could bias estimation of plaque prevalence due to difficulties to differentiate small plaques. In addition, because of prolonged follow-up, the two ultrasound assessments at the inclusion and follow-up were not performed by the same sonographer which could influence definition of plaque prevalence between the assessments. In this analysis, therefore, we were not able to assess formation of new plaque or plaque index. In our previous study in the SLEVIC cohort, we reported that atherosclerotic plaques but not cIMT was increased in SLE as compared with controls, and also that vulnerable, echolucent plaques were increased in SLE.14 However, data on echolucency were not available at this follow-up assessment. Nevertheless, our findings add to the insights into importance of detection of carotid plaque to identify patients at a higher risk for cIMT progression in the presence of traditional and SLE-specific risk factors.
The main strength of our study is a control group assessed for the presence of risk factors and carotid subclinical atherosclerosis per study protocol along with the participating patients. We, though, acknowledge several limitations which should be considered when interpreting the findings. The sample size in the current study of 77 patients with SLE and 74 controls is relatively small, which may have influenced the ability to detect a significant difference in IMT progression between SLE and controls. Because of deaths and drop-outs (in total 32% of patients and 39% of controls of the original cohort could not be followed, of all reasons for lost to follow-up, because of deaths in 8/34 patients of the original cohort and in 1/48 controls) progression of subclinical atherosclerosis could not be assessed in the whole original cohort at the 7-year visit. A comparable retention rate in the follow-up analysis of CV risk assessment and outcomes after 6 years, 62%, has been reported for the patients with SLE of a mean disease duration of 12 years.22 The presented results may not be applicable to all SLE populations, or patients with more severe disease, and should be interpreted in the context of patients, representative for herewith studied population.
Conclusion
Altogether, we observed a similar progression of cIMT in mild SLE disease and population controls over 7 years, which suggests that excess of CV risk in some patients with SLE could be arrest. Our findings suggest the importance of management of CV risk factors and limited usage of corticosteroids, along with management of SLE disease, and should encourage clinicians to treat modifiable CV risk factors also in patients with mild disease, to improve outcomes in patients with SLE.
Acknowledgments
We acknowledge invaluable contribution from Tomas Jogestrand, Margareta Wörnert and Eva Waldheim.
Footnotes
Contributors: IH, JF and SA designed the study. SA was responsible for acquisition of the data, analysis, interpretation of the data and drafting the manuscript. TG was responsible for the carotid ultrasound investigations and LL performed it. All authors participated in interpretation of the data and approved the final manuscript.
Funding: This study was supported by The Swedish Rheumatism Association, The Swedish Heart Lung foundation, King Gustav V’s 80-year fund, 6th Framework Program of the European Union (grant LSHM-CT-2006-037227 CVDIMMUNE) with JF as co-ordinator.
Competing interests: None declared.
Patient consent for publication: Obtained.
Ethics approval: The study was approved by the local ethics committee and was performed in accordance with the declaration of Helsinki.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement: No additional data are available.
References
- 1. Tsokos GC. Systemic lupus erythematosus. N Engl J Med 2011;365:2110–21. 10.1056/NEJMra1100359 [DOI] [PubMed] [Google Scholar]
- 2. Olesińska M, Saletra A. Quality of life in systemic lupus erythematosus and its measurement. R 2018;56:45–54. 10.5114/reum.2018.74750 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Carter EE, Barr SG, Clarke AE. The global burden of SLE: prevalence, health disparities and socioeconomic impact. Nat Rev Rheumatol 2016;12:605–20. 10.1038/nrrheum.2016.137 [DOI] [PubMed] [Google Scholar]
- 4. Frostegård J, Ulfgren A-K, Nyberg P, et al. Cytokine expression in advanced human atherosclerotic plaques: dominance of pro-inflammatory (Th1) and macrophage-stimulating cytokines. Atherosclerosis 1999;145:33–43. 10.1016/S0021-9150(99)00011-8 [DOI] [PubMed] [Google Scholar]
- 5. Singh RR, Yen EY. SLE mortality remains disproportionately high, despite improvements over the last decade. Lupus 2018;27:1577–81. 10.1177/0961203318786436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Yen EY, Shaheen M, JMP W, et al. 46-Year trends in systemic lupus erythematosus mortality in the United States, 1968 to 2013: a nationwide population-based study. Ann Intern Med 2017;167:777–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Schoenfeld SR, Kasturi S, Costenbader KH. The epidemiology of atherosclerotic cardiovascular disease among patients with SLE: a systematic review. Semin Arthritis Rheum 2013;43:77–95. 10.1016/j.semarthrit.2012.12.002 [DOI] [PubMed] [Google Scholar]
- 8. Giannelou M, Nezos A, Fragkioudaki S, et al. Contribution of MTHFR gene variants in lupus related subclinical atherosclerosis. Clin Immunol 2018;193:110–7. 10.1016/j.clim.2018.02.014 [DOI] [PubMed] [Google Scholar]
- 9. Clancy R, El Bannoudi H, Rasmussen SE, et al. Human low-affinity IgG receptor FcγRIIA polymorphism H131R associates with subclinical atherosclerosis and increased platelet activity in systemic lupus erythematosus. J Thromb Haemost 2019;17:532–7. 10.1111/jth.14385 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Frostegård J, Hellström C, Nilsson P, et al. Autoantibody profiling reveals four protein candidate autoantigens associated with systemic lupus erythematosus. Lupus 2018;27:1670–8. 10.1177/0961203318788153 [DOI] [PubMed] [Google Scholar]
- 11. Kao AH, Lertratanakul A, Elliott JR, et al. Relation of carotid intima–media thickness and plaque with incident cardiovascular events in women with systemic lupus erythematosus. Am J Cardiol 2013;112:1025–32. 10.1016/j.amjcard.2013.05.040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Di Minno MND, Emmi G, Ambrosino P, et al. Subclinical atherosclerosis in asymptomatic carriers of persistent antiphospholipid antibodies positivity: a cross-sectional study. Int J Cardiol 2019;274:1–6. 10.1016/j.ijcard.2018.06.010 [DOI] [PubMed] [Google Scholar]
- 13. Leonard D, Svenungsson E, Dahlqvist J, et al. Novel gene variants associated with cardiovascular disease in systemic lupus erythematosus and rheumatoid arthritis. Ann Rheum Dis 2018;77:1063–9. 10.1136/annrheumdis-2017-212614 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Anania C, Gustafsson T, Hua X, et al. Increased prevalence of vulnerable atherosclerotic plaques and low levels of natural IgM antibodies against phosphorylcholine in patients with systemic lupus erythematosus. Arthritis Res Ther 2010;12:R214 10.1186/ar3193 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Henrot P, Foret J, Barnetche T, et al. Assessment of subclinical atherosclerosis in systemic lupus erythematosus: a systematic review and meta-analysis. Joint Bone Spine 2018;85:155–63. 10.1016/j.jbspin.2017.12.009 [DOI] [PubMed] [Google Scholar]
- 16. Wu G-C, Liu H-R, Leng R-X, et al. Subclinical atherosclerosis in patients with systemic lupus erythematosus: a systemic review and meta-analysis. Autoimmun Rev 2016;15:22–37. 10.1016/j.autrev.2015.10.002 [DOI] [PubMed] [Google Scholar]
- 17. Hochberg MC. Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 1997;40:1725 10.1002/art.1780400928 [DOI] [PubMed] [Google Scholar]
- 18. Bombardier C, Gladman DD, Urowitz MB, et al. Derivation of the SLEDAI. A disease activity index for lupus patients. The Committee on Prognosis Studies in SLE. Arthritis Rheum 1992;35:630–40. 10.1002/art.1780350606 [DOI] [PubMed] [Google Scholar]
- 19. Gladman D, Ginzler E, Goldsmith C, et al. The development and initial validation of the Systemic Lupus International Collaborating Clinics/American College of Rheumatology damage index for systemic lupus erythematosus. Arthritis Rheum 1996;39:363–9. 10.1002/art.1780390303 [DOI] [PubMed] [Google Scholar]
- 20. Wendelhag I, Liang Q, Gustavsson T, et al. A new automated computerized analyzing system simplifies readings and reduces the variability in ultrasound measurement of intima–media thickness. Stroke 1997;28:2195–200. 10.1161/01.STR.28.11.2195 [DOI] [PubMed] [Google Scholar]
- 21. Fanouriakis A, Kostopoulou M, Alunno A, et al. Update of the EULAR recommendations for the management of systemic lupus erythematosus. Ann Rheum Dis 2019;2019. [DOI] [PubMed] [Google Scholar]
- 22. Haque S, Skeoch S, Rakieh C, et al. Progression of subclinical and clinical cardiovascular disease in a UK SLE cohort: the role of classic and SLE-related factors. Lupus Sci Med 2018;5:e000267 10.1136/lupus-2018-000267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Thompson T, Sutton-Tyrrell K, Wildman RP, et al. Progression of carotid intima–media thickness and plaque in women with systemic lupus erythematosus. Arthritis Rheum 2008;58:835–42. 10.1002/art.23196 [DOI] [PubMed] [Google Scholar]
- 24. Zhang C-Y, Lu L-J, Li F-H, et al. Evaluation of risk factors that contribute to high prevalence of premature atherosclerosis in Chinese premenopausal systemic lupus erythematosus patients. J Clin Rheumatol 2009;15:111–6. 10.1097/RHU.0b013e31819d8489 [DOI] [PubMed] [Google Scholar]
- 25. Tektonidou MG, Kravvariti E, Konstantonis G, et al. Subclinical atherosclerosis in systemic lupus erythematosus: comparable risk with diabetes mellitus and rheumatoid arthritis. Autoimmun Rev 2017;16:308–12. 10.1016/j.autrev.2017.01.009 [DOI] [PubMed] [Google Scholar]
- 26. Zen M, Iaccarino L, Gatto M, et al. The effect of different durations of remission on damage accrual: results from a prospective monocentric cohort of Caucasian patients. Ann Rheum Dis 2017;76:562–5. 10.1136/annrheumdis-2016-210154 [DOI] [PubMed] [Google Scholar]
- 27. Sharma C, Raymond W, Eilertsen G, et al. Achieving Lupus Low Disease Activity State (LLDAS-50) is associated with both reduced damage accrual and mortality in patients with systemic lupus erythematosus. Arthritis Care Res 2019. 10.1002/acr.23867 [DOI] [PubMed] [Google Scholar]
- 28. van Vollenhoven RF, Mosca M, Bertsias G, et al. Treat-to-target in systemic lupus erythematosus: recommendations from an international task force. Ann Rheum Dis 2014;73:958–67. 10.1136/annrheumdis-2013-205139 [DOI] [PubMed] [Google Scholar]
- 29. Urowitz MB, Su J, Gladman DD. Atherosclerotic vascular events in systemic lupus erythematosus: an evolving story. J Rheumatol 2019:jrheum.180986 10.3899/jrheum.180986 [DOI] [PubMed] [Google Scholar]
- 30. Tattersall MC, Gassett A, Korcarz CE, et al. Predictors of carotid thickness and plaque progression during a decade: the multi-ethnic study of atherosclerosis. Stroke 2014;45:3257–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. de Leeuw K, Smit AJ, de Groot E, et al. Longitudinal study on premature atherosclerosis in patients with systemic lupus erythematosus. Atherosclerosis 2009;206:546–50. 10.1016/j.atherosclerosis.2009.03.018 [DOI] [PubMed] [Google Scholar]
- 32. Roman MJ, Crow MK, Lockshin MD, et al. Rate and determinants of progression of atherosclerosis in systemic lupus erythematosus. Arthritis Rheum 2007;56:3412–9. 10.1002/art.22924 [DOI] [PubMed] [Google Scholar]
- 33. Sutton-Tyrrell K, Lassila HC, Meilahn E, et al. Carotid atherosclerosis in premenopausal and postmenopausal women and its association with risk factors measured after menopause. Stroke 1998;29:1116–21. 10.1161/01.STR.29.6.1116 [DOI] [PubMed] [Google Scholar]
- 34. Fernandez-Friera L, Penalvo JL, Fernandez-Ortiz A, et al. Prevalence, vascular distribution, and multiterritorial extent of subclinical atherosclerosis in a middle-aged cohort: the PESA (Progression of Early Subclinical Atherosclerosis) study. Circulation 2015;131:2104–13. [DOI] [PubMed] [Google Scholar]
- 35. Tselios K, Gladman DD, Su J, et al. Evolution of risk factors for atherosclerotic cardiovascular events in systemic lupus erythematosus: a longterm prospective study. J Rheumatol 2017;44:1841–9. 10.3899/jrheum.161121 [DOI] [PubMed] [Google Scholar]
- 36. Urowitz MB, Gladman DD, Anderson NM, et al. Cardiovascular events prior to or early after diagnosis of systemic lupus erythematosus in the systemic lupus international collaborating clinics cohort. Lupus Sci Med 2016;3:e000143 10.1136/lupus-2015-000143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Thacker SG, Zarzour A, Chen Y, et al. High-density lipoprotein reduces inflammation from cholesterol crystals by inhibiting inflammasome activation. Immunology 2016;149:306–19. 10.1111/imm.12638 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Zhong Q, Zhao S, Yu B, et al. High-density lipoprotein increases the uptake of oxidized low density lipoprotein via PPARγ/CD36 pathway in inflammatory adipocytes. Int J Biol Sci 2015;11:256–65. 10.7150/ijbs.10258 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Chen N, Frishman WH. High-density lipoprotein infusion therapy and atherosclerosis: current research and future directions. Cardiol Rev 2016;24:298–302. 10.1097/CRD.0000000000000111 [DOI] [PubMed] [Google Scholar]
- 40. Frostegård J, Svenungsson E, Wu R, et al. Lipid peroxidation is enhanced in patients with systemic lupus erythematosus and is associated with arterial and renal disease manifestations. Arthritis Rheum 2005;52:192–200. 10.1002/art.20780 [DOI] [PubMed] [Google Scholar]
- 41. Navab M, Berliner JA, Subbanagounder G, et al. HDL and the inflammatory response induced by LDL-derived oxidized phospholipids. Arterioscler Thromb Vasc Biol 2001;21:481–8. 10.1161/01.ATV.21.4.481 [DOI] [PubMed] [Google Scholar]
- 42. Lertratanakul A, Wu P, Dyer AR, et al. Risk factors in the progression of subclinical atherosclerosis in women with systemic lupus erythematosus. Arthritis Care Res 2014;66:1177–85. 10.1002/acr.22271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Kravvariti E, Konstantonis G, Sfikakis PP, et al. Progression of subclinical atherosclerosis in systemic lupus erythematosus versus rheumatoid arthritis: the impact of low disease activity. Rheumatology 2018;57:2158–66. 10.1093/rheumatology/key233 [DOI] [PubMed] [Google Scholar]
- 44. Gustafsson JT, Herlitz Lindberg M, Gunnarsson I, et al. Excess atherosclerosis in systemic lupus erythematosus,—A matter of renal involvement: Case control study of 281 SLE patients and 281 individually matched population controls. PLoS One 2017;12:e0174572 10.1371/journal.pone.0174572 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Polak JF, Pencina MJ, Pencina KM, et al. Carotid-wall intima–media thickness and cardiovascular events. N Engl J Med 2011;365:213–21. 10.1056/NEJMoa1012592 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Quevedo-Abeledo JC, Rua-Figueroa I, Sanchez-Perez H, et al. Disease damage influences cardiovascular risk reclassification based on carotid ultrasound in patients with systemic lupus erythematosus. J Rheumatol 2019. [DOI] [PubMed] [Google Scholar]
- 47. Tselios K, Sheane BJ, Gladman DD, et al. Optimal monitoring for coronary heart disease risk in patients with systemic lupus erythematosus: a systematic review. J Rheumatol 2016;43:54–65. 10.3899/jrheum.150460 [DOI] [PubMed] [Google Scholar]
- 48. Lorenz MW, Markus HS, Bots ML, et al. Prediction of clinical cardiovascular events with carotid intima-media thickness: a systematic review and meta-analysis. Circulation 2007;115:459–67. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
lupus-2019-000362supp001.pdf (64.1KB, pdf)