Abstract
This study aimed to compare the clinicopathologic features and prognosis in patients with Xp11 translocation renal cell carcinomas (RCCs). In total, 8083 RCCs were screened at five centres from January 2007 to December 2018, including 8001 adults (≥18 years) and 82 children (<18 years). Finally, 73 adults and 17 children were identified as Xp11 translocation RCCs, accounting for 1.1% (90 of 8083) of the RCCs. However, 4 children and 1 adult were excluded because of loss to follow-up when performing the survival analysis. The proportion of paediatric and adult Xp11 translocation RCCs was 20.7% (17 of 82) and 0.9% (73 of 8001) of RCCs, respectively, and the incidence in children and adults was significantly different (P < 0.01). Lymph node positivity (LN+) most commonly occurred in children (58.8%) compared with adults (28.8%; P = 0.02), but children with LN+ showed significantly higher five-year overall survival and progression-free rates (OS: 75.0%; PFS: 64.8%) than adult patients (OS: 40.3%; PFS: 0%) (log-rank PPFS < 0.01; POS = 0.04). Multivariable analysis indicated that local lymph node metastasis was associated with both PFS (HR = 0.10; 95% CI 0.02–0.51; P = 0.01) and OS (HR = 0.11; 95% CI 0.01–0.98; P = 0.04) in adults. Adult patients with LN+ may indicate a worse prognosis than paediatric patients.
Subject terms: Cancer epigenetics, Renal cell carcinoma
Introduction
Xp11 translocation renal cell carcinomas (RCCs) are characterized by several different chromosomal translocations involving Xp11 and the formation of TFE3 fusion genes, followed by overexpression of TFE3 protein1. The first case was a 2.4-year-old child diagnosed by karyotype analysis in 19862. In 2004, Xp11 translocation RCCs were classified as a distinct entity of RCC by the World Health Organization (WHO)3. Recent reports have found that RCC associated with t(6; 11) (p2l; q12)/TFEB gene fusions is exceedingly similar to Xp11 translocation RCCs with respect to clinical characteristics, pathology, and molecular genetics4. Given that both TFE3 and TFEB pertain to the microphthalmia-associated transcription factor family, Xp11 translocation RCCs and RCC associated with t (6; 11) (p2l; q12)/TFEB gene fusions were reclassified as MiT family translocation RCCs in the 2016 WHO renal tumour classification scheme5.
TFE3 protein immunohistochemistry (IHC) combined with the fluorescence in situ hybridization (FISH) assay was an effective method for the diagnosis of Xp11 translocation RCCs6. Several clinical studies have shown that the incidence of paediatric Xp11 translocation RCCs is significantly higher than that of adults7–9. Because its histomorphology is extremely similar to clear cell renal cell carcinoma and papillary renal cell carcinoma, it is easily misdiagnosed and missed. Therefore, the actual incidence of the disease is worthy of further investigations with a large sample size. Patients with Xp11 translocation RCCs often present at advanced stages10, but paediatric patients with a higher incidence of regional lymph node positivity (LN+) seem to have a better prognosis than adults when regional lymph node involvement was completely resected11,12. In addition, the natural history of the disease is variable between children and adults based on clinical experience. Meta-analysis showed that clinical characteristics between children and adults were distinctive, including the incidence of lymphatic metastases, tumour stage and prognosis13. In view of the lack of a large sample size and longer follow-up for this relatively recent category of tumours, we grouped five urological cancer research institutes together to compare the clinicopathologic features and outcomes in patients with paediatric and adult Xp11 translocation RCCs.
The purpose of the present study was to compare the clinicopathologic features and outcomes of Xp11 translocation RCCs between children and adults by multi-institutional analysis of patients.
Materials and Methods
Patient selection
The retrospective study was conducted according to the ethical principles of the Helsinki Declaration II, and written informed consent was obtained from both adult patients and legal guardians of paediatric patients before the clinical investigations were conducted. The present study was approved by the Institutional Review Board of Nanjing Drum Tower Hospital, Jiangsu Province Hospital, Jiangsu Cancer Hospital, Zhongda Hospital Southeast University and Nanjing First Hospital. We have verified that the data collected at five institutions can be treated as independent data (Supplementary Tables S1–S3). We screened 8083 RCCs from patients consecutively treated by surgery at five centres from January 2007 until December 2018 (Table 1), including 8001 adults (≥18 years) and 82 children (<18 years). Eventually, 73 adults and 17 children who were diagnosed with Xp11 translocation RCCs were included in the analyses for whom clinical information was available, but 4 children and 1 adult were excluded from the study because of loss to follow-up when performing the survival analysis. We included patients who were identified by TFE3-IHC combined with the break-apart FISH assay performed on paraffin-embedded tissue blocks14. Patients were excluded for incomplete clinical and follow-up data, negative TFE3-IHC and break-apart FISH assays.
Table 1.
The number of cases per institution is provided.
| Institution | Included | Excluded | ||
|---|---|---|---|---|
| Adults | Children | Adults | Children | |
| Nanjing Drum Tower Hospital | 46 | 6 | 0 | 2 |
| Jiangsu Province Hospital | 14 | 1 | 1 | 2 |
| Jiangsu Cancer Hospital | 7 | 0 | 0 | 0 |
| Zhongda Hospital Southeast University | 2 | 6 | 0 | 0 |
| Nanjing First Hospital | 3 | 0 | 0 | 0 |
Evaluation of clinicopathologic data
The clinicopathologic data for patients in two groups were analysed, including sex, clinical symptoms, operation, laterality, pathological features (pT stage, local lymph node metastasis, vena cava tumour thrombosis, tumour boundary, nuclear grade and American Joint Committee On Cancer [AJCC]) stage), adjuvant therapy and clinical outcomes (stable, recurrent or dead). Symptoms were analysed when patients had one or more initial symptoms. A clear tumour boundary was defined as a complete continuous tumour pseudocapsule between the tumour mass and normal kidney tissue. The 2016 WHO/ISUP pathological nuclear grading system was referenced by the nuclear grade standards. TNM staging was based on the eighth edition issued by the AJCC staging in 2017. All of the patients were followed up until the time of death or loss to follow-up.
Statistical analysis
Count data were expressed as percentages. Categorical variables were analysed using the Pearson chi-square test, while it was performed using Fisher’s exact test when the expected frequency was less than 5 in any cell. Progression-free survival (PFS) was defined from the initiation of surgery to the date of disease progression or death outcome or censored at last follow-up. Overall survival (OS) was defined as the time interval between the date of surgery and the date of death or last follow-up. The Kaplan-Meier method was used to estimate both PFS and OS, and statistical comparisons were assessed by the log-rank test. A Cox proportional hazards model was used to evaluate the predictive role of the factors that showed significance in the long-rank test. A two-side p-value of <0.05 was considered statistically significant. SPSS software version 23.0 (SPSS Inc., Chicago, IL, USA) was used for all statistical analyses. The survival curves were drawn using GraphPad Prism software version 5.0.
Results
Epidemiological characteristics
Of the 8083 patients with RCCs, 90 cases (male to female ratio of 1:1.43) with a median age of 28 years old (ranging from 3 to 71 years) were diagnosed with Xp11 translocation RCCs, including 17 children, 53 patients between 18 and 45, and 20 patients older than 45. The overall proportion of Xp11 translocation RCCs was 1.1% (90 of 8083), and the proportion of patients younger than 18 years and older than 18 years was 20.7% (17 of 82) and 0.9% (73 of 8001) of RCCs, respectively. The difference in the incidence rates between children and adults was statistically significant (P < 0.01).
Comparative clinicopathologic features and prognosis of children and adults
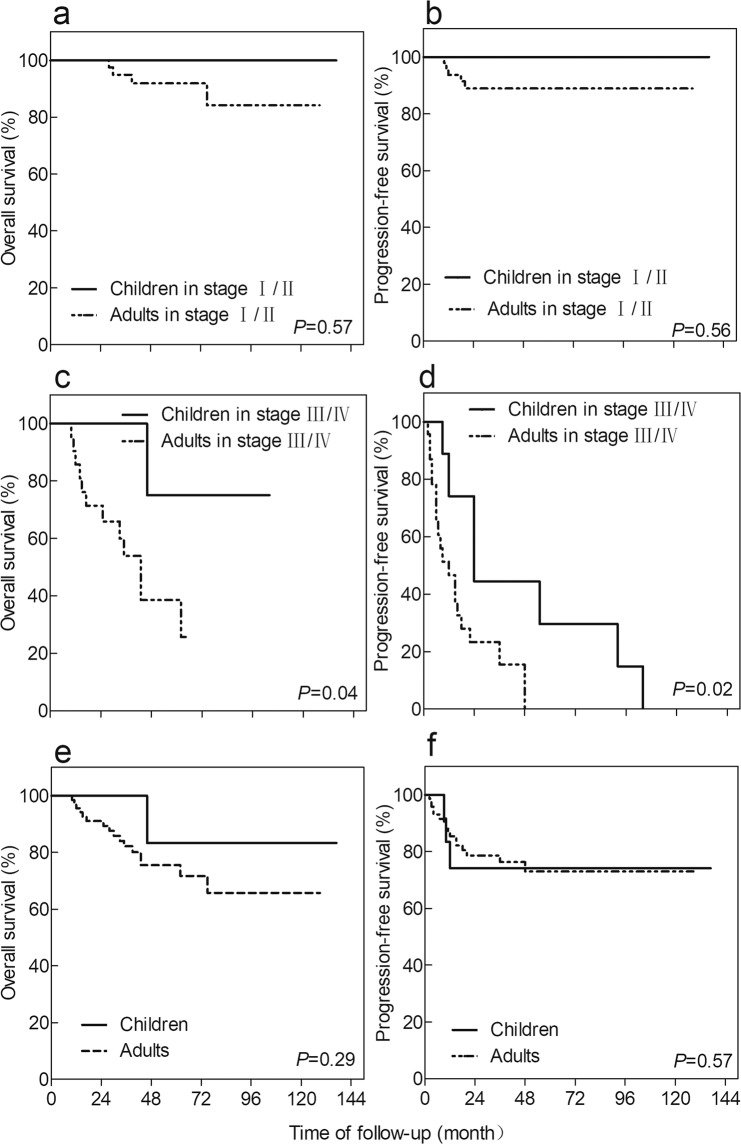
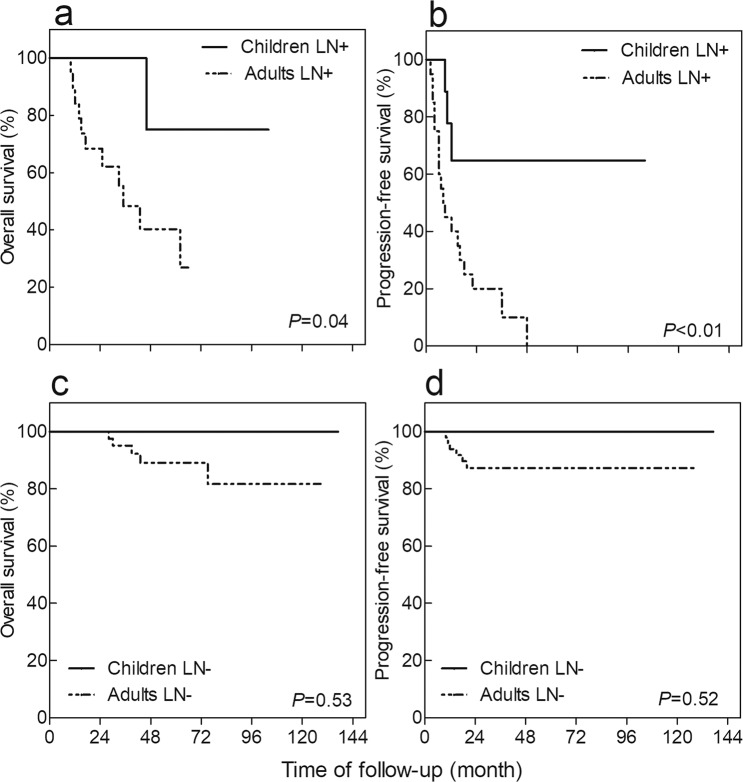
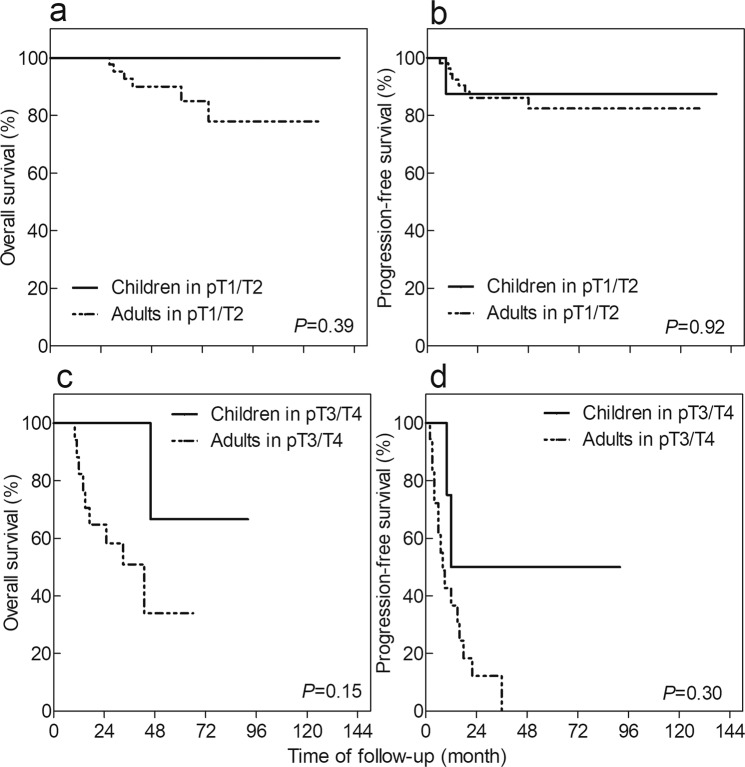
There were significant differences in the clinicopathologic characteristics and outcomes of the patients within the two groups (Table 2, Figs. 1, 2, 3). The Xp11 translocation RCCs in the adult group were discovered by routine examination (48.7%) or haematuria (28.2%), while those in the paediatric group were diagnosed with haematuria (38.1%) and abdominal mass (23.8%) as the first symptoms. There was a significant difference in the distribution of clinical symptoms between children and adults (P < 0.01). Only one child underwent NSS for the primary tumour; however, 24 adult-onset patients received NSS apart from 48 cases of RN (RN: 94.1% children vs 67.1% adults; P = 0.03). only one adult patient (1.1%) had sarcomatoid tissue in the tumour, and 10 adult patients (11.1%) had coagulative tumour necrosis. Neither the sarcomatoid histology nor the coagulative necrosis has yet been found in the child patients. Vena cava tumour thrombectomy was performed on 9 cases in the adult group and only one case in the paediatric group. Paediatric patients had a higher five-year overall survival and progression-free rate (OS: 83.3%; PFS: 74.1%) than adult patients (OS: 75.5%; PFS: 63.0%), but they were not statistically significant (log-rank PPFS = 0.57; POS = 0.29). Adults showed slightly higher pT stage than children at diagnosis (pT3/T4: 26.0% adults vs 24.5% children; P = 0.99), but the prognosis of the two groups at the pT3/T4 stage had no significant difference (log-rank PPFS = 0.30; POS = 0.15). Children were more prone to LN+ compared with adults (LN+: 58.8% children vs 28.8% adults; P = 0.02), but children with LN+ showed significantly higher five-year overall survival and progression-free survival (OS: 75.0%; PFS: 64.8%) than those of adult patients (OS: 40.3%; PFS: 0%) (log-rank PPFS < 0.01; POS = 0.04). There was no significant difference in prognosis between children and adults in stage I/II (log-rank PPFS = 0.56; POS = 0.57), but the five-year overall survival and progression-free survival of adults (OS: 38.5%; PFS: 0%) in stage III/IV were lower than those in paediatric patients (OS: 75.0%; PFS: 64.8%) with the same stage (log-rank PPFS = 0.02; POS = 0.04). Among child-onset patients in stage III/IV, 2 patients received immunotherapy, and only one child had targeted treatments. However, in the adult group, 26 cases in stage I/II and 5 cases in stage III/IV received immunotherapy, and 13 cases in stage III/IV and the 5 recurrence cases received targeted therapy.
Table 2.
Clinical and pathological characteristics of 90 patients.
| Characteristic | Children (17 cases) | Adults (73 cases) | P-value |
|---|---|---|---|
| No. (%) | No. (%) | ||
| Sex | 0.99 | ||
| Male | 7 (41) | 30 (41) | |
| Female | 10 (59) | 43 (59) | |
| Symptom | <0.01 | ||
| Symptomless | 4 (19) | 38 (49) | |
| Haematuria | 8 (38) | 22 (28) | |
| Abdominal mass | 5 (24) | 3 (4) | |
| Flank pain | 4 (19) | 15 (19) | |
| Laterality | 0.61 | ||
| Left | 7 (41) | 35 (47) | |
| Right | 10 (59) | 38 (53) | |
| Operation | 0.03 | ||
| RN | 16 (94) | 49 (67) | |
| NSS | 1 (6) | 24 (33) | |
| pT stage | 0.99 | ||
| T1/T2 | 13 (76) | 54 (74) | |
| T3/T4 | 4 (24) | 19 (26) | |
| Nuclear grade | 0.49 | ||
| 1/2 | 12 (71) | 45 (62) | |
| 3/4 | 5 (29) | 28 (38) | |
| AJCC stage | <0.01 | ||
| I/II | 5 (29) | 49 (67) | |
| III/IV | 12 (71) | 24 (33) | |
| Local lymph node metastasis | 0.02 | ||
| LN+ | 10 (59) | 21 (29) | |
| LN− | 7 (41) | 52 (71) | |
| Vena cava tumour thrombosis | 0.68 | ||
| Positive | 1 (6) | 9 (12) | |
| Negative | 16 (94) | 64 (88) | |
| Adjuvant therapy | <0.01 | ||
| None | 14 (82) | 23 (31) | |
| Immunotherapy | 2 (12) | 31 (43) | |
| Targeted therapy | 1 (6) | 19 (26) | |
| Tumour boundary | 0.08 | ||
| Clear | 6 (35) | 43 (59) | |
| Unclear | 11 (65) | 30 (41) |
RN, Radical nephrectomy; NSS, Nephron-sparing surgery; AJCC, American Joint Committee On Cancer; LN+, Lymph node positivity; LN−, Lymph node negativity.
Figure 1.
Kaplan-Meier method depicting overall survival (OS) and progression-free survival (PFS) both in all patients and patients grouped by AJCC stage, stratified according to the age of onset. (a) Survival analysis of OS in stage I/II patients, (b) Survival analysis of PFS in stage I/II patients, (c) Survival analysis of OS in stage III/IV patients, (d) Survival analysis of PFS in stage III/IV patients, (e) Survival analysis of OS in all patients and (f) Survival analysis of PFS in all patients.
Figure 2.
Kaplan-Meier method depicting overall survival (OS) and progression-free survival (PFS) in patients grouped by local lymph node metastasis, stratified according to the age of onset. (a) Survival analysis of OS in patients with LN+, (b) Survival analysis of PFS in patients with LN+, (c) Survival analysis of OS in patients with LN−, and (d) Survival analysis of PFS in patients with LN−.
Figure 3.
Kaplan-Meier method depicting overall survival (OS) and progression-free survival (PFS) in patients grouped by pT stage, stratified according to the age of onset. (a) Survival analysis of OS in pT1/T2 patients, (b) Survival analysis of PFS in pT1/T2 patients, (c) Survival analysis of OS in pT3/T4 patients, and (d) Survival analysis of PFS in pT3/T4 patients.
The outcomes of follow-up
Except for five patients who were lost to follow-up, the remaining cases were followed up, with a mean duration of 52 months (range: 1–137 months) for the children and 46 months (range: 4–129 months) for the adults. only one adult patient (1.1%) had sarcomatoid tissue in the tumour and died of multiple organ metastasis 24 months after surgery. Of 10 adult patients (11.1%) with coagulative tumour necrosis, 3 cases had disease progression and adverse outcome, and the remaining 7 cases stayed stable. Four cases of adult patients with pT1/pT2 and LN+ were followed up, and 3 cases had an adverse outcome. Of 3 children with disease progression, only one child died of brain metastasis 46 months after the operation, and the remaining 2 recurrence cases had local LN+, followed by the dissection of local lymph nodes. Of the 24 adults with disease progression, 16 cases died of distant metastasis with a mean follow-up time of 30 months: 8 cases in a distant lymph node, 3 cases in the liver, 2 cases in the lung, 1 case in the brain, 1 case in the bone, 1 case in the peritoneum, and the other patients were stable. Survival analysis showed that pT stage, adjuvant treatment, local lymph node metastasis, AJCC stage and vena cava tumour thrombosis influenced OS (all P < 0.05) and that tumour boundary, pT stage, adjuvant treatment, local lymph node metastasis, AJCC stage and vena cava tumour thrombosis were associated with PFS (all P < 0.05) in the adult group (Table 3). Furthermore, adjuvant therapy and pT stage were associated with PFS (all P < 0.05) in the paediatric group (Table 4). Multivariable analysis was executed to estimate the prognostic value of the clinicopathologic characteristics for PFS and OS. Due to the small sample size and favourable prognosis of the children, multivariable analysis was conducted only in the adults. When performing multivariable analysis, both adjuvant treatment and AJCC stage were excluded to obtain a more reliable result because they were closely associated with postoperative pathological stage. Multivariable analysis indicated that local lymph node metastasis was associated with both PFS (HR = 0.10; 95% CI 0.02–0.52; P = 0.01) and OS (HR = 0.11; 95% CI 0.01–0.98; P = 0.04) in adults (Table 5).
Table 3.
Survival analysis of clinicopathologic variables for overall survival (OS) and progression-free survival (PFS) in the adult group.
| Variable | One-year | Five-year | One-year | Five-year | P-value | |
|---|---|---|---|---|---|---|
| OS | OS | PFS | PFS | Log-rank test | ||
| % (SE) | % (SE) | % (SE) | % (SE) | OS | PFS | |
| Sex | 0.29 | 0.17 | ||||
| Male | 89.4 (0.06) | 71.3 (0.10) | 69.2 (0.09) | 58.0 (0.09) | ||
| Female | 100 | 78.5 (0.07) | 85.5 (0.06) | 67.3 (0.08) | ||
| Symptom | —* | —* | ||||
| Symptomless | 97.3 (0.03) | 78.5 (0.08) | 83.9 (0.06) | 68.2 (0.08) | ||
| Haematuria | 90.0 (0.07) | 78.5 (0.10) | 72.0 (0.10) | 59.7 (0.11) | ||
| Abdominal mass | —* | — | — | — | ||
| Flank pain | 100 | 51.4 (0.19) | 72.7 (0.13) | 51.9 (0.16) | ||
| Laterality | 0.80 | 0.78 | ||||
| Left | 93.6 (0.04) | 71.8 (0.10) | 78.5 (0.07) | 67.9 (0.09) | ||
| Right | 97.3 (0.03) | 78.2 (0.07) | 78.9 (0.07) | 60.2 (0.09) | ||
| Operation | 0.78 | 0.16 | ||||
| RN | 95.6 (0.03) | 75.9 (0.07) | 74.3 (0.06) | 57.5 (0.08) | ||
| NSS | 95.8 (0.04) | 72.6 (0.13) | 87.5 (0.07) | 75.4 (0.10) | ||
| pT stage | <0.01 | <0.01 | ||||
| T1/T2 | 100 | 90.0 (0.05) | 92.4 (0.04) | 82.4 (0.06) | ||
| T3/T4 | 82.4 (0.09) | 34.0 (0.13) | 36.7 (0.12) | 0 | ||
| Nuclear grade | 0.64 | 0.50 | ||||
| 1/2 | 95.3 (0.03) | 75.6 (0.07) | 79.7 (0.06) | 67.1 (0.07) | ||
| 3/4 | 96.0 (0.04) | 74.6 (0.10) | 77.2 (0.08) | 53.6 (0.13) | ||
| AJCC stage | <0.01 | <0.01 | ||||
| I/II | 100 | 91.9 (0.05) | 93.7 (0.04) | 88.9 (0.05) | ||
| III/IV | 85.7 (0.08) | 38.5 (0.12) | 46.6 (0.11) | 0 | ||
| Local lymph node metastasis | <0.01 | <0.01 | ||||
| LN+ | 84.2 (0.08) | 40.3 (0.13) | 40.0 (0.11) | 0 | ||
| LN− | 100 | 89.1 (0.05) | 93.9 (0.03) | 87.3 (0.05) | ||
| Vena cava tumour thrombosis | <0.01 | <0.01 | ||||
| Positive | 88.9 (0.11) | 22.2 (0.14) | 11.1 (0.11) | 0 | ||
| Negative | 96.6 (0.02) | 85.1 (0.05) | 88.6 (0.04) | 72.8 (0.06) | ||
| Adjuvant therapy | <0.01 | <0.01 | ||||
| None | 95.7 (0.04) | 87.7 (0.09) | 95.7 (0.04) | 85.2 (0.08) | ||
| Immunotherapy | 100 | 90.5 (0.07) | 86.4 (0.06) | 82.7 (0.07) | ||
| Targeted therapy | 88.9 (0.07) | 43.8 (0.12) | 44.4 (0.11) | 11.1 (0.07) | ||
| Tumour boundary | 0.35 | 0.02 | ||||
| Clear | 97.6 (0.02) | 80.0 (0.08) | 88.0 (0.05) | 71.1 (0.08) | ||
| Unclear | 92.6 (0.05) | 68.6 (0.09) | 65.2 (0.09) | 50.5 (0.10) | ||
RN, Radical nephrectomy; NSS, Nephron-sparing surgery; AJCC, American Joint Committee On Cancer; LN+, Lymph node positivity; LN−, Lymph node negativity; SE, Standard error; *No data or insufficient data for statistical analysis.
Table 4.
Survival analysis of clinicopathologic variables for overall survival (OS) and progression-free survival (PFS) in the paediatric group.
| Variable | One-year | Five-year | One-year | Five-year | P-value | |
|---|---|---|---|---|---|---|
| OS | OS | PFS | PFS | Log-rank test | ||
| % (SE) | % (SE) | % (SE) | % (SE) | OS | PFS | |
| Sex | 0.32 | 0.36 | ||||
| Male | 100 | 66.7 (0.27) | 60 (0.22) | 60 (0.22) | ||
| Female | 100 | 100 | 85.7 (0.13) | 85.7 (0.13) | ||
| Symptom | —* | —* | ||||
| Symptomless | 100 | — | 100 | — | ||
| Haematuria | 100 | 75.0 (0.22) | 80 (0.18) | 80 (0.18) | ||
| Abdominal mass | —* | — | — | — | ||
| Flank pain | 100 | — | 100 | — | ||
| Laterality | 0.16 | 0.13 | ||||
| Left | 100 | 100 | 100 | 100 | ||
| Right | 100 | 50.0 (0.35) | 57.1 (0.19) | 57.1 (0.19) | ||
| Operation | — | — | ||||
| RN | 100 | 83.3 (0.15) | 74.1 (0.13) | 74.1 (0.13) | ||
| NSS | — | — | — | — | ||
| pT stage | 0.32 | 0.24 | ||||
| T1/T2 | 100 | 100 | 87.5 (0.12) | 87.5 (0.12) | ||
| T3/T4 | 100 | 66.7 (0.27) | 50.0 (0.25) | 50.0 (0.25) | ||
| Nuclear grade | 0.48 | 0.73 | ||||
| 1/2 | 100 | 75.0 (0.22) | 76.2 (0.15) | 76.2 (0.15) | ||
| 3/4 | 100 | 100 | 66.7 (0.27) | 66.7 (0.27) | ||
| AJCC stage | 0.48 | 0.27 | ||||
| I/II | 100 | 100 | 100 | 100 | ||
| III/IV | 100 | 75.0 (0.22) | 64.8 (0.17) | 64.8 (0.17) | ||
| Local lymph node metastasis | 0.48 | 0.27 | ||||
| LN+ | 100 | 75.0 (0.22) | 64.8 (0.17) | 64.8 (0.17) | ||
| LN− | 100 | 100 | 100 | 100 | ||
| Vena cava tumour thrombosis | 0.66 | 0.58 | ||||
| Positive | 100 | 100 | 100 | 100 | ||
| Negative | 100 | 80.0 (0.18) | 71.6 (0.14) | 71.6 (0.14) | ||
| Adjuvant therapy | — | — | ||||
| None | 100 | 83.3 (0.15) | 88.9 (0.11) | 88.9 (0.11) | ||
| Immunotherapy | — | — | — | — | ||
| Targeted therapy | — | — | — | — | ||
| Tumour boundary | 0.32 | 0.21 | ||||
| Clear | 100 | 100 | 100 | 100 | ||
| Unclear | 100 | 66.7 (0.27) | 62.5 (0.17) | 62.5 (0.17) | ||
RN, Radical nephrectomy; NSS, Nephron-sparing surgery; AJCC, American Joint Committee On Cancer; LN+, Lymph node positivity; LN−, Lymph node negativity; SE, Standard error; *No data or insufficient data for statistical analysis.
Table 5.
Multivariable analysis of overall survival and progression-free survival in adult patients.
| Variable | Progression-free survival | Overall survival | ||||||
|---|---|---|---|---|---|---|---|---|
| HR | 95% CI | P-value | HR | 95% CI | P-value | |||
| Lower | Upper | Lower | Upper | |||||
| Sex | 1.90 | 0.66 | 5.47 | 0.24 | 1.48 | 0.44 | 4.99 | 0.52 |
| Operation | 1.90 | 0.58 | 5.40 | 0.32 | 0.99 | 0.26 | 3.78 | 0.99 |
| Nuclear grade | 0.80 | 0.28 | 2.29 | 0.67 | 0.67 | 0.17 | 2.63 | 0.57 |
| pT stage | 1.55 | 0.30 | 7.99 | 0.60 | 1.04 | 0.12 | 9.08 | 0.97 |
| Local lymph node metastasis | 0.10 | 0.02 | 0.51 | 0.01 | 0.11 | 0.01 | 0.98 | 0.04 |
| Vena cava tumour thrombosis | 0.43 | 0.13 | 1.43 | 0.17 | 0.52 | 0.11 | 2.44 | 0.41 |
| Tumour boundary | 1.45 | 0.56 | 3.79 | 0.45 | 0.78 | 0.25 | 2.46 | 0.68 |
CI, Confidence interval; HR, Hazard ratio.
Discussion
Xp11 translocation RCCs were classified as MiT family translocation carcinomas with an incidence among diagnosed RCCs of one-third in paediatric patients and approximately 0.2–5.0% in adults8,9,15. The incidence of adults in previous studies7,16 was broadly consistent with ours, while the incidence of children was significantly underestimated in our multi-centre study, which could be caused by insufficient sample size. Although children diagnosed with Xp11 translocation RCCs exhibit a high incidence compared to adults, adult Xp11 translocation RCCs could still outnumber paediatric Xp11 translocation RCCs in that RCCs are more common in adults. In addition, our study inferred that the incidence of female Xp11 translocation RCCs in the adult group was significantly higher than that in the male group, as it is a rare malignant tumour in the urinary system with a higher incidence in females. Meta-analysis confirmed that females were more predominant than males in adult patients, while in children, no sex-related predominance was observed13. However, further exploration is still essential to understand why there is an age-related difference in incidence and a high incidence in adult female patients.
The clinical manifestations of Xp11 translocation RCCs are basically similar to other types of RCCs. Statistical results indicate that haematuria is the most common symptom in the Xp11 translocation RCCs, which is consistent with the findings of several clinical studies8,17. Over the past decades, the methods of diagnosing Xp11 translocation RCCs have been gradually appreciated and deeply studied. Recent studies have confirmed that TFE3-IHC combined with the FISH assay is an effective method for the diagnosis of Xp11 translocation RCCs, which could improve the specificity and may reduce false-positive results6,16. Therefore, all eligible patients were diagnosed by TFE3-IHC combined with the TFE3 break-apart FISH assay in the present research. Interestingly, it has been confirmed that dual-fusion FISH probes were useful for identifying ASPL-TFE3 and PRCC-TFE3 RCC18,19, and other probes are worthy of designing to diagnose the remaining known subtypes of Xp11 translocation RCCs.
Considering that Xp11 translocation RCCs are obviously more invasive and prone to lymph node and distant metastasis in adults, taking the most appropriate clinical therapy is of great significance. To date, although there is no agreement on clinical treatments for Xp11 translocation RCCs, the recommendation for all surgeries is RN if possible. As shown in our study, RN was performed on the majority of patients, especially in the paediatric group. We then inferred from the statistical results that 10 of 13 cases in the paediatric group who received RN showed no evidence of progression with a mean follow-up time of 57 months. Similarly, good outcomes have been reported in 8 of 9 cases of Xp11 translocation RCCs, which were confirmed by comparative genomic hybridization 3–10 years after RN20. However, with the development of laparoscopic technology and the application of the Da Vinci robotic surgery system, NSS is popularly recommended in solitary renal tumours with diameters of less than 4 cm21. Cheng et al. found that treating small Xp11 translocation RCCs (≤4 cm) with intact pseudocapsules by receiving NSS produced a favourable treatment outcome, which provided a theoretical basis for NSS treatment22. In addition, Gorin et al. reported that 4 patients with T1a Xp11 translocation RCCs who received partial nephrectomy for an incidentally detected small renal mass (mean imaging diameter: 2.6 cm) were alive without evidence of disease during a mean follow-up of 37 months23. Moreover, Liu et al. reported that 9 children with Xp11 translocation RCCs who underwent NSS were alive without postoperative recurrence and metastasis for the mean follow-up period of 50.1 months and concluded that NSS is safe and feasible for Xp11 translocation RCCs sized < 4–7 cm in diameter and located in one pole24. Our statistical results implied that NSS was performed for the treatment of 24 Xp11 translocation RCCs, and most patients had a favourable prognosis. Although many recent studies and guidelines have extended NSS to RCCs, most of these subjects are CCRCC or other common subtypes of RCC21,25. Considering that Xp11 translocation RCCs are more aggressive, more clinical studies with large sample sizes in multiple centres are needed to determine whether NSS is feasible and safe for Xp11 translocation RCCs.
Compared with traditional types of RCC, patients with Xp11 translocation RCCs often presented at an advanced stage and demonstrated an invasive clinical course and poor prognosis10. Moreover, the clinical behaviour of Xp11 translocation RCCs that occurred in adults was more aggressive26. However, Song et al. reported that progression-free survival was observed in 13 of 15 children after surgery and concluded that paediatric patients with LN+ had a good prognosis following surgery alone27. The results from our research confirmed that the prognosis of adults in higher AJCC stage was poorer compared with that of children. A recent clinical study suggested that cancer-specific survival differed significantly between adult patients with positive TFE3 rearrangement RCCs and adult patients with negative TFE3 rearrangement PRCC, which implied that adult RCCs with TFE3 rearrangement may be a clinically aggressive tumour9. It has been presented in our study that LN+ is associated with an increased risk of disease progression and death in adults. Therefore, adult patients diagnosed with Xp11 translocation RCCs with higher AJCC stage and LN+ need more active follow-up and treatments after surgery. The significant difference in the prognosis between children and adults deserves our in-depth study. Basic research may be able to find a reasonable explanation at the genetic level.
There are some limitations to our study. First, although our study is the first multi-institutional clinical design in China concerning Xp11 translocation RCCs, the sample size was still not sufficiently large due to the low incidence of this rare disease and potential missed diagnosis, and the follow-up time for patients receiving NSS was relatively short. Long-term follow-up is essential to estimating the therapeutic efficacy of NSS. Second, the multi-institutional study is advantageous for reducing biases caused by single centre studies, while it is also subject to heterogeneity in data collection and follow-up. Third, we have to note that we did not finish identifying the gene-fusion partners for all patients, and further study on genetic fusion types using fusion probes is in progress in our centres.
Conclusions
We have demonstrated that Xp11 translocation RCCs are rare entities of RCC and differ in age-related incidence. The results from survival analysis suggest that adult patients with LN+ have higher progression and poorer prognosis compared with those of children. Local lymph node metastasis could be associated with an increased risk of disease progression and death outcome in adults, and adult patients diagnosed with Xp11 translocation RCCs with higher AJCC stage and LN+ need more active follow-up and treatments after surgery.
Supplementary information
Acknowledgements
The authors would like to thank the members of the research group for useful discussions. This research was supported by the National Natural Science Foundation of China (ID: 81572512), the Nanjing SCI-TECH Development project (ID: 201803025), and the Nanjing Medical Science and Technique Development Foundation (QRX17049).
Author contributions
Conception and design: Wenliang Ma, Ning Liu, Xiaozhi Zhao and Xiaogong Li; Acquisition of data: Wenyuan Zhuang, Weijian Li, Lihua Zhang, Ruipeng Jia and Linfeng Xu; Analysis and interpretation of data: Feng Qu, Jing Sun, Wei Xu, Gutian Zhang and Hongqian Guo; Manuscript drafing or revising: Wenliang Ma, Dongmei Li and Weidong Gan.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
is available for this paper at 10.1038/s41598-020-59162-5.
References
- 1.Wang XT, Xia QY, Zhou XJ, Rao Q. Xp11 Translocation Renal Cell Carcinoma and the Mesenchymal Counterparts: An Evolving Concept with Novel Insights on Clinicopathologic Features, Prognosis, Treatment, and Classification. Crit. Rev. oncogenesis. 2017;22:481–497. doi: 10.1615/CritRevOncog.2017020558. [DOI] [PubMed] [Google Scholar]
- 2.de Jong B, Molenaar IM, Leeuw JA, Idenberg VJ, Oosterhuis JW. Cytogenetics of a renal adenocarcinoma in a 2-year-old child. Cancer Genet. cytogenetics. 1986;21:165–169. doi: 10.1016/0165-4608(86)90042-7. [DOI] [PubMed] [Google Scholar]
- 3.Lopez-Beltran A, Scarpelli M, Montironi R, Kirkali Z. 2004 WHO classification of the renal tumors of the adults. Eur. Urol. 2006;49:798–805. doi: 10.1016/j.eururo.2005.11.035. [DOI] [PubMed] [Google Scholar]
- 4.Xia Q, et al. Renal cell carcinoma with t(6;11)(p21.2;q13)/MALAT1-TFEB fusion: a clinical and pathological analysis. Zhonghua bing. li xue za zhi = Chin. J. Pathol. 2015;44:895–899. [PubMed] [Google Scholar]
- 5.McGuire S. World Cancer Report 2014. Geneva, Switzerland: World Health Organization, International Agency for Research on Cancer, WHO Press, 2015. Adv. Nutr. 2016;7:418–419. doi: 10.3945/an.116.012211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rao Q, et al. TFE3 break-apart FISH has a higher sensitivity for Xp11.2 translocation-associated renal cell carcinoma compared with TFE3 or cathepsin K immunohistochemical staining alone: expanding the morphologic spectrum. Am. J. surgical Pathol. 2013;37:804–815. doi: 10.1097/PAS.0b013e31827e17cb. [DOI] [PubMed] [Google Scholar]
- 7.Geller JI, et al. Characterization of adolescent and pediatric renal cell carcinoma: A report from the Children’s Oncology Group study AREN03B2. Cancer. 2015;121:2457–2464. doi: 10.1002/cncr.29368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ramphal R, Pappo A, Zielenska M, Grant R, Ngan BY. Pediatric renal cell carcinoma: clinical, pathologic, and molecular abnormalities associated with the members of the mit transcription factor family. Am. J. Clin. Pathol. 2006;126:349–364. doi: 10.1309/98ye9e442ar7lx2x. [DOI] [PubMed] [Google Scholar]
- 9.Sukov WR, et al. TFE3 rearrangements in adult renal cell carcinoma: clinical and pathologic features with outcome in a large series of consecutively treated patients. Am. J. surgical Pathol. 2012;36:663–670. doi: 10.1097/PAS.0b013e31824dd972. [DOI] [PubMed] [Google Scholar]
- 10.Mir MC, et al. Altered transcription factor E3 expression in unclassified adult renal cell carcinoma indicates adverse pathological features and poor outcome. BJU Int. 2011;108:E71–76. doi: 10.1111/j.1464-410X.2010.09818.x. [DOI] [PubMed] [Google Scholar]
- 11.Geller JI, et al. Translocation renal cell carcinoma: lack of negative impact due to lymph node spread. Cancer. 2008;112:1607–1616. doi: 10.1002/cncr.23331. [DOI] [PubMed] [Google Scholar]
- 12.Geller JI, Dome JS. Local lymph node involvement does not predict poor outcome in pediatric renal cell carcinoma. Cancer. 2004;101:1575–1583. doi: 10.1002/cncr.20548. [DOI] [PubMed] [Google Scholar]
- 13.Cheng X, Gan W, Zhang G, Li X, Guo H. Clinical characteristics of XP11.2 translocation/TFE3 gene fusion renal cell carcinoma: a systematic review and meta-analysis of observational studies. BMC Urol. 2016;16:40. doi: 10.1186/s12894-016-0154-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chen X, et al. Application of polyclonal break-apart probes in the diagnosis of Xp11.2 translocation renal cell carcinoma. Zhonghua yi xue za zhi. 2014;94:3675–3677. [PubMed] [Google Scholar]
- 15.Kuroda N, et al. Review of renal carcinoma associated with Xp11.2 translocations/TFE3 gene fusions with focus on pathobiological aspect. Histology histopathology. 2012;27:133–140. doi: 10.14670/hh-27.133. [DOI] [PubMed] [Google Scholar]
- 16.Hirobe M, et al. Clinicopathological characteristics of Xp11.2 translocation renal cell carcinoma in adolescents and adults: Diagnosis using immunostaining of transcription factor E3 and fluorescence in situ hybridization analysis. Int. J. urology: Off. J. Japanese Urological Assoc. 2016;23:140–145. doi: 10.1111/iju.13007. [DOI] [PubMed] [Google Scholar]
- 17.Argani P, et al. Xp11 translocation renal cell carcinoma in adults: expanded clinical, pathologic, and genetic spectrum. Am. J. surgical Pathol. 2007;31:1149–1160. doi: 10.1097/PAS.0b013e318031ffff. [DOI] [PubMed] [Google Scholar]
- 18.Chen X, et al. Newly designed break-apart and ASPL-TFE3 dual-fusion FISH assay are useful in diagnosing Xp11.2 translocation renal cell carcinoma and ASPL-TFE3 renal cell carcinoma: a STARD-compliant article. Med. 2015;94:e873. doi: 10.1097/md.0000000000000873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Xiong Lei, Chen Xiancheng, Liu Ning, Wang Zhen, Miao Baolei, Gan Weidong, Li Dongmei, Guo Hongqian. PRCC-TFE3 dual-fusion FISH assay: A new method for identifying PRCC-TFE3 renal cell carcinoma in paraffin-embedded tissue. PLOS ONE. 2017;12(9):e0185337. doi: 10.1371/journal.pone.0185337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zou H, et al. Xp11 translocation renal cell carcinoma in adults: a clinicopathological and comparative genomic hybridization study. Int. J. Clin. Exp. Pathol. 2014;7:236–245. [PMC free article] [PubMed] [Google Scholar]
- 21.Fergany AF, Hafez KS, Novick AC. Long-term results of nephron sparing surgery for localized renal cell carcinoma: 10-year followup. J. Urol. 2000;163:442–445. doi: 10.1016/S0022-5347(05)67896-2. [DOI] [PubMed] [Google Scholar]
- 22.Cheng X, et al. Pseudocapsule of renal cell carcinoma associated with Xp11.2 translocation/TFE3 gene fusion: a clue for tumor enucleation? Int. J. Clin. Exp. Pathol. 2015;8:5403–5410. [PMC free article] [PubMed] [Google Scholar]
- 23.Gorin MA, Ball MW, Pierorazio PM, Argani P, Allaf ME. Partial nephrectomy for the treatment of translocation renal cell carcinoma. Clin. Genitourin. cancer. 2015;13:e199–201. doi: 10.1016/j.clgc.2014.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Liu C, Zhang W, Song H. Nephron-sparing surgery in the treatment of pediatric renal cell carcinoma associated with Xp11.2 translocation/TFE3 gene fusions. J. pediatric Surg. 2017;52:1492–1495. doi: 10.1016/j.jpedsurg.2017.03.052. [DOI] [PubMed] [Google Scholar]
- 25.Mir MC, et al. Partial Nephrectomy Versus Radical Nephrectomy for Clinical T1b and T2 Renal Tumors: A Systematic Review and Meta-analysis of Comparative Studies. Eur. Urol. 2017;71:606–617. doi: 10.1016/j.eururo.2016.08.060. [DOI] [PubMed] [Google Scholar]
- 26.Meyer PN, Clark JI, Flanigan RC, Picken MM. Xp11.2 translocation renal cell carcinoma with very aggressive course in five adults. Am. J. Clin. Pathol. 2007;128:70–79. doi: 10.1309/lr5g1vmxpy3g0cuk. [DOI] [PubMed] [Google Scholar]
- 27.Song HC, et al. Biological characteristics of pediatric renal cell carcinoma associated with Xp11.2 translocations/TFE3 gene fusions. J. pediatric Surg. 2014;49:539–542. doi: 10.1016/j.jpedsurg.2013.10.005. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.