Abstract
Aim/Background
This research aims to prevent progression from mild cognitive impairment (MCI) to Alzheimer's disease. A Japanese study of leprosy patients revealed that the incidence of dementia in leprosy patients was lower than that in patients taking dapsone who had never been treated. But a similar study the following year refuted the finding of less dementia in leprosy patients taking dapsone. According to conflicting reports, Mycobacterium leprae was a factor in reducing the incidence of Alzheimer's disease. Thus, we formed a hypothesis that if dapsone is administered to patients without leprosy but with MCI and the prophylactic effect of dementia syndrome is observed over a long period of time, we can determine whether dapsone can prevent the progression of MCI to dementia syndrome. If dementia does not occur after treating inflammation in brain cells while dementia develops after a certain long-term period (usually within 2–3 years), brain cell inflammation can be demonstrated as the cause of dementia.
Methods
This is a prospective cohort research. We report on an elderly patient diagnosed with MCI from February 2008 to January 2019. The patient took dapsone 100 mg once a day from 2010 to 2015 for the treatment of MCI. Since 2016, the production of dapsone has ceased in Korea. In June 2018, the patient was diagnosed with Alzheimer's disease. The patient took Aricept for the treatment of Alzheimer's disease but complained of serious side effects. And dapsone was re-administered to the patient from November 2018.
Results
The patient recovered to MCI and improved her daily life owing to the treatment with dapsone. The drug controls the inflammatory response in the brain, irrespective of whether proteins are deposited in neurons.
Conclusions
This finding means that dementia syndrome is an inflammatory disease. This research suggests that diagnostic criteria for Alzheimer's disease should be based on the presence or absence of inflammation in neurons. Because inflammation in neurons can occur in middle age due to various causes, we can treat inflammation in neurons and prevent and treat dementia syndrome, including Alzheimer's disease.
Keywords: Activities of daily living, Alzheimer's disease, Dapsone, Dementia syndrome, Diaminodiphenyl sulfone, Brain cell inflammation, Magnetic resonance imaging, Mild cognitive impairment, Neuropsychological assessment
Introduction
Since 2011, the diagnosis of Alzheimer's disease has included amyloid beta protein or tau protein deposits in brain cells, even if dementia symptoms are absent [1]. Dementia refers to a state in which cognitive function is generally deteriorated by various causes, causing significant disruption to daily life.
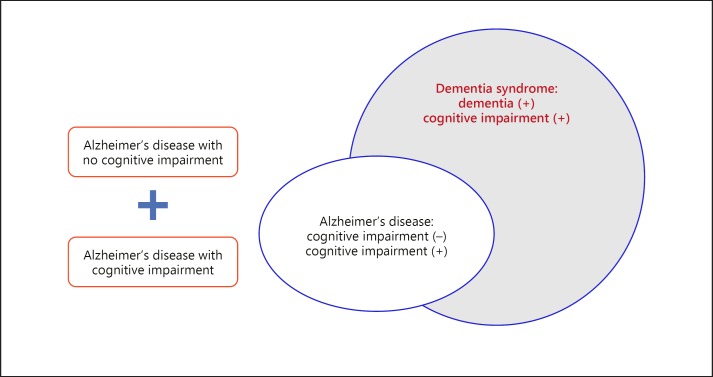
Dementia is a “syndrome” that is a set of specific symptoms with cognitive impairment. More than 70 cases of the disease that causes the clinical syndrome of dementia are classified. Alzheimer's disease belongs to dementia syndrome with cognitive impairment, but it is a degenerative brain disease in which amyloid beta protein or tau protein is deposited in brain cells even in the absence of cognitive impairment (Fig. 1).
Fig. 1.
Scope of Alzheimer's disease and dementia syndrome [1]. Alzheimer's disease belongs to dementia syndrome with cognitive impairment, but it is a degenerative brain disease in which the amyloid beta protein or tau protein is deposited in brain cells even in the absence of cognitive impairment [1]. In this study, we suggest that the diagnosis criteria and treatment plan of Alzheimer's disease should be changed. Whether inflammatory reactions are persistent is important when diagnosing and treating Alzheimer's disease.
The first hypothesis of this study is that we can prevent or cure cognitive disorders by treating brain cell inflammation in patients with dementia syndrome (Alzheimer's disease).
In the 1990s, a Japanese epidemiological survey study of leprosy patients aged 65 years or older revealed that the incidence of dementia in leprosy patients who had been treated with dapsone and related medications was lower than that in patients taking the drug who had never been treated [2, 3]. A neuropathological case series of leprosy patients detected the absence of senile plaques but detected the expected levels of neurofibrillary tangles compared to those in the general population [3, 4]. A similar study the following year showed no difference in the discovery of senile plaques between the patient and control groups, thus refuting the finding of less dementia in leprosy patients taking dapsone [3, 5].
According to conflicting reports by the Japanese Sanatorium for leprosy patients, Hansen's disease was a factor in reducing the incidence of Alzheimer's disease [6]. In addition, the Japanese researchers tested dapsone-containing medicines for leprosy patients, but they were unable to find the effect of removing amyloid beta protein; thus, they proposed that Mycobacterium lepraeis a major causative factor of Hansen's disease [7]. Worldwide, M. leprae is considered responsible for reducing the incidence of Alzheimer's disease based on these two papers [6, 7].
However, the study [6] by the Japanese Sanatorium for leprosy patients selected the target group from the 65–84 age category. When comparing and analyzing two traits, patients with lepromatous leprosy and those with tuberculosis, the leprosy patients in the 80–95 age group with the high incidence of dementia should be selected as the target group with which to compare the occurrence of dementia. In other words, the incidence of dementia is not relatively high in the 65–75 age groups. Importantly, dementia incidence should have been compared in the 80–95 age groups with a high incidence of dementia. Potential selection bias was identified in data processing. There were also a number of leprosy patients who stopped taking dapsone due to Hansen's inactivation. When analyzing the preventive effect of dementia, we compared the period in which leprosy patients were taking dapsone continuously with the period in which these patients stopped taking dapsone. Notably, the pharmacological effect of dapsone is to control inflammation in brain cells, not to remove amyloid beta protein [7]. M. leprae was posited as the major cause of dementia reduction by studying the amyloid beta protein removal function of dapsone, which is a null hypothesis.
The null hypothesis can be proved if dapsone is administered to patients without leprosy but with mild cognitive impairment (MCI) and the prophylactic effect of dementia syndrome is observed over a long period of time. Through long-term observation, we can determine whether dapsone can prevent the progression of MCI to dementia syndrome when brain cell inflammation is controlled. If dementia does not occur after treating inflammation in brain cells while dementia develops after a certain long-term period (a period of transition from MCI to dementia within 2–3 years), brain cell inflammation can be demonstrated as the cause of dementia.
The second hypothesis of this study is that the following represents the null hypothesis: M. leprae, which induces leprosy, is the cause of the low incidence of Alzheimer's disease in patients with Hansen's disease.
Methods
This Seoul study was a prospective cohort study of patients diagnosed with MCI from February 2008 to January 2019. Dapsone was administered to patients from 2010 to 2015 for the treatment of MCI. SNU institute of Aging in Seoul National University approved the clinical study of patients based on The Nuremberg Code (1947) and FDA guidelines ethically in accordance with the World Medical Association Declaration of Helsinki. All methods were carried out in accordance with relevant guidelines and regulations. The subjects (or their parents or guardians) gave written informed consent, and the study protocol was approved by the committee on human research of SNU Institute of Aging, Seoul National University. We administered medicines in compliance with medical and pharmacist laws under the informed consent of the patient.
Dapsone is an antibiotic similar to other sulfonamides and blocks bacterial synthesis of dihydrofolic acid via competition with p-aminobenzoic acid for the active site of dihydropteroate synthetase [8]. Dapsone is a common treatment for leprosy and is also used as an important drug in many other skin diseases [9].
Dapsone binds with hemoglobin, which decreases oxygen transport capacity, causes methemoglobinemia, and induces hemolytic anemia [10]. In addition to hemoglobin, dapsone can affect biochemical metabolism by binding to various constituents, such as human enzymes [11]. Dapsone is a substance with allosteric regulation of structure and function.
Immune cells such as leukocyte neutrophils produce hypochlorite to attack foreign bodies to defend cells when a foreign substance attacks the cells. Myeloperoxidase is a kind of oxidoreductase that catalyzes the chemical reaction of the following reaction: H2O2 + Cl− = H2O + OCl−. Dapsone binds to myeloperoxidase and regulates the production of hypochlorite, thereby reducing the inflammatory response of cells [12, 13, 14].
Myeloperoxidase is a hemoprotein. Dapsone reduces the inflammatory response of neutrophils and preserves cells by regulating the production of hypochlorous acid. The ability of dapsone to exert its anti-inflammatory and immunomodulatory effects reduces the inflammatory response by binding to myeloperoxidase contained in leukocytes.
Therefore, we prepared a prospective cohort Seoul study for patients with MCI who underwent precise neuropsychiatry and MRI examinations for the elderly with short-term memory impairment in 2008; this study was performed in accordance with the Korean medical and pharmacist law and abided by research ethics. The Seoul Municipal Boramae Hospital operated by Seoul National University Hospital conducted Chest PA (online suppl. 4 [page 16], online suppl. 4–1 [data], Patient 20080129-Chest- Rx image) and MRI brain imaging (online suppl. 4 [page 17], online suppl. 4–2 [data], Patient 20080129-BRAIN MRI- Rx image) and psychiatric examination through a common protocol of the Center for Geriatric Dementia Clinical Research, Ministry of Health and Welfare (online suppl. 4 [pages 2–7]). Next, from August 18, 2010, to May 2019, the cohort was formed for 10 years. A physician who graduated from Seoul National University College of Medicine performed close observation. He worked as an information technology scientist and lived with a patient. He is the patient's son (online suppl. 5 [page 2]). On February 5, 2008, the patient underwent a mental neurological examination through the MRI brain imaging (online suppl. 4 [page 17]) and common protocol of the Center for Geriatric Dementia Clinical Research, Ministry of Health and Welfare (online suppl. 4 [page 2–7]). MRI showed that the brain had slight cerebrovascular disease and several lacunae in the right basal ganglia (online suppl. 4 [page 17]) with normal ECG (online suppl. 4 [page 12–13]). Nuclear medicine examination (online suppl. 4 [page 18]) and clinical pathology examination (online suppl. 4 [page 14–15]) were not specific. In the psychiatric examination through the common protocol, the patient was diagnosed with MCI by Prof. Jun-young Lee (online suppl. 4 [page 11]).
In this study, we hypothesized that the deterioration of MCI to Alzheimer's disease could be prevented by the inflammation control effect of dapsone. The patient was dosed once daily with 50–100 mg dapsone. The patient is the second daughter, a pharmacist graduated from Seoul National University College of Pharmacy (online suppl. 5 [page 1]). The patient took the dapsone from the Najin pharmacy. The dapsone tablets were produced by Taegeuk Pharmaceutical Company (Table 1).
Table 1.
Taegeuk dapsone tablets of Najin Pharmacy operated by Eunju Lee (online suppl. 7)
| On the day | Name | Resident registration number | Prescription issuing agency | Doses, n | Dosage | Daily dose, mg |
|---|---|---|---|---|---|---|
| 2010-08-18 18:00:05 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 50 | 50 |
| 2010-09-17 14:52:17 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 75 | 75 |
| 2010-10-19 11:04:18 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 3 | 50 | 150 |
| 2010-11-15 12:35:18 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 3 | 50 | 150 |
| 2010-12-17 15:23:29 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 75 | 75 |
| 2011-01-18 17:37:12 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 75 | 75 |
| 2011-02-21 18:47:52 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2011-03-15 18:03:59 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2011-04-14 17:59:22 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2011-05-19 11:47:19 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2011-07-06 18:16:45 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2011-08-04 17:36:40 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2011-09-02 18:02:18 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2011-09-30 19:44:04 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2011-11-04 09:37:02 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2011-12-08 13:50:17 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2012-02-17 17:59:39 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2012-04-09 18:12:42 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2012-08-14 18:09:32 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2012-09-24 19:32:00 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2012-11-02 17:22:46 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2013-01-18 19:46:07 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2013-04-13 13:10:20 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2013-07-19 20:51:30 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2013-12-21 11:30:19 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2014-03-20 18:25:23 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2014-11-15 16:39:15 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
| 2015-03-02 19:37:23 | Patient | 331209-2****** | The Korea Association of Health Promotion Clinic | 1 | 100 | 100 |
The symptoms of the patient's memory impairment disappeared after dosing. The patient continued her daily life, including taking care of grandchildren, paying bills, shopping, washing clothes, preparing meals, and managing savings. The patient visited her youngest daughter at Gyeong Hee Lee's house (online suppl. 5 [page 3]) every day.
However, after the prescription of dapsone on March 2, 2015, Taegeuk Pharmaceutical Co., which produced dapsone in Korea, stopped the production of dapsone. Since then, the Korea Food and Drug Administration has designated dapsone as an anti-retraction drug and only produced dapsone for leprosy.
In May 2018, the patient worsened and developed memory problems and dementia syndrome. The patient underwent MRI examination at Dobong General Hospital on June 23, 2018, and was diagnosed based on radiological findings (online suppl. 6 [page 1], online suppl. 6–1 [data], Patient 20180623-MRI-Rx image). On June 27, 2018, Hyeong-gil Lee, a neurologist at the Kyunghee Neulfureun Geriatric Hospital, diagnosed the patient with Alzheimer's disease (online suppl. 6 [page 2]).
For Alzheimer's disease treatment, this patient took Aricept, one of the four currently available Alzheimer's drugs in May 2019, according to a prescription from Hyeong-gil Lee, a neurologist at Kyunghee Neulfureun Geriatric Hospital. However, the patient experienced serious adverse drug reactions (Table 2).
Table 2.
Aricept's side effects (patient's symptoms are italicized)
| Body organs | 1% or more (frequently) | 0.1–1% (sometimes present) |
| Whole body | Abdominal pain, asthenia, fungal infections, influenza-like syndrome | Allergies, soft tissue salts, boredom, sepsis, facial edema, hernia |
| Cardiovascular system | Hypotension, bradycardia, ECG abnormalities, heart failure | Myocardial infarction, angina pectoris, atrial fibrillation, congestive heart failure, peripheral vascular disease, supraventricular outflow tract, ventricular excess contraction, cardiac hypertrophy |
| Digestive system | Constipation, gastroenteritis, perineal incontinence, indigestion | γ-GT increase, gastritis, dysphagia, periodontitis, gastric ulcer, periodontal abscess, abdominal bloating, liver dysfunction, trim, esophagitis, anal bleeding |
| Endocrine system | Diabetes | |
| Blood and lymphatic system | Anemia | Leukocytosis |
| Metabolism and nutrition | Weight loss, peripheral edema, edema, increased LDH, increased alkaline phosphatase | Increased AST, elevated ALT, increased blood pressure, hypercholesterolemia, hypokalemia, hypoglycemia, weight gain, bilirubinemia, BUN increase, B12 deficiency anemia, cachexia, creatinine increase, gout, hypotonia, hypoproteinemia, iron deficiency anemia |
| Musculoskeletal system | Arthritis | Arthrosis, bone fracture, joint pain, leg cramps, osteoporosis, myalgia |
| Nervous system | Irritability, anxiety, progression, convulsions, mental disorder of immigration, gait abnormality | Apathy, dizziness, delusion, dream more, cerebrovascular or more, increased salivation, ataxia, more than ecstatic, vasodilation, cerebral hemorrhage, cerebral infarction, cerebral ischemia, outside dementia, pyramidal symptoms, seizures convulsions, hemiplegia, hypertension, exercise reduction |
| Respiratory machine | Pharyngitis, pneumonia, increased cough, bronchitis | Difficulty breathing, rhinitis, asthma |
| Skin and skin attachment machine | Rash, skin ulcer, itching | Psoriasis, skin discoloration, shingles, dry skin, sweating, urticaria, bullous rash |
| Special sensory machine | Conjunctivitis, glaucoma, visual anomaly, ear pain, tear abnormalities | |
| Genitourinary system | Urinary tract infection, cystitis, hematuria, diabetes | Vaginitis, dysuria, urinary frequency, proteinuria |
The patient received medical care from Prof. Kyung-min Lee of the Neurology Department of Seoul National University Hospital on October 11, 2018 (online suppl. 2 [page 1]). On November 6, 2018, a test was conducted to determine whether a cognitive disorder was present to determine the presence or absence of cognitive impairment (online suppl. 1 [pages 1–2]). The duration of the test was 1.5 h. Prof. Kyung-min Lee reduced the dose of Aricept to 15 days 1/2 T but after 15 days, increased the dose to 1 T. The same side effects occurred in the patient after mid-November 2018.
The patient's son (as a physician) prescribed dapsone to the patient, who had taken the drug in the MCI state. Domestic production was discontinued, but the Korea Orphan & Essential Drug Center designated dapsone as an essential medicinal product and imported it from Riemser Pharma GmbH (Germany). The Korea Orphan and Essential Drug Center supplied DAPSON-Fatol (50 mg dapsone). Dr. Jong-hoon Lee administered 50 mg twice daily to the patient. He purchased 100 tablets on November 28, 2018; 100 tablets on December 27, 2018; and 300 tablets on April 2, 2019, from the Korea Orphan and Essential Drug Center (online suppl. 3).
Dapsone had no side effects, and the patient's dementia syndrome improved significantly. The patient became able to live everyday life. On December 20, 2018, the patient received an outpatient medical examination by Dr. Soh-hyun Ahn, a neurologist at Seoul National University Hospital, for an objective examination. Dr. Jong-hoon Lee explained to Dr. Soh-hyun Ahn that the patient stopped taking Aricept due to severe side effects and that he administered dapsone; thereafter, the side effects caused by Aricept were eliminated, and the patient became able to live everyday life. On January 14, 2019 Dr. Soh-hyun Ahn proposed to re-examine the test for cognitive impairment and compare the results (online suppl. 2 [page 2]).
On January 18, 2019, the patient received outpatient medical care from Prof. Kyung-min Lee of the Department of Neurology at Seoul National University Hospital (online suppl. 2 [page 3]) and received a second test to determine the presence or absence of cognitive impairment (online suppl. 1 [page 3]). The duration of this test was also 1.5 h. Patient received a medical record, a report on the examination for the presence of cognitive impairment, etc., and a medical certificate for the current patient status from Seoul National University Hospital (online suppl. 1 [5 page]).
Results
The patient was examined twice by the Neurology Department of Seoul National University Hospital for cognitive impairment (online suppl. 1 (pages 1–4); for all online suppl. material, see www.karger.com/doi/10.1159/000504880). On November 6, 2018, the patient was taking Aricept (online suppl. 2), and on January 14, 2019, the patient was taking dapsone (online suppl. 3). Since there were no side effects after taking dapsone, the patient was able to take it steadily, and the results were comparable. The results of the examination are shown in the comparison table (Table 3).
Table 3.
The comparison table of the neurological examination at Seoul National University Hospital
| Test results 6.11.2018 | Test results 14.1.2019 | Commentary | Reference | Note | |
|---|---|---|---|---|---|
| Mini-Mental State Examination (MMSE) |
(18)/30 | (17)/30 | Cognitive disorder Improving, No difference | 27–30: Normal 25–26: Suspected 24 or less: Cognitive impairment |
Because of the high correlation between the education level and the score, clinical consideration of cognitive impairment is needed |
| Clinical Dementia Rating (CDR) | 1.0 | 1.0 | Dementia status Improving, No difference |
0: Normal 0.5: Questionable 1: Mild 2: Moderate 3: Severe |
For CDR 0.5, please retest after 6 months |
| Frontal Assessment Battery (FAB) | (16)/18 | (14)/18 | Frontal dysfunction Improving, No difference |
17-18: Normal 16 or less: Frontal dysfunction 12 or less: FTD estimation (sensitivity 79%) |
Overall evaluation items (Item 6) Conceptualization, cognitive flexibility, continuity of behavior, conflicting instruction, inhibition control |
| Neuropsychiatric Inventory Questionnaire (NPI-Q) | Total score: 30 points Reported abnormal behavior: delusions, abnormal movements | Total score: 0 points Abnormal behavior reported: None | Abnormal behavior Completely gone | The larger the score, handicapped Enumerate the top 5 items with severe disabilities | All survey items (section 12) delusions, hallucination, stubbornness, depression, anxiety, mania, apathy, disinhibition, nervousness, ataxia, night-time behavior, appetite |
| Instrumental Activities of Daily Living (K-IADL) | 0.6 | 0.3 | Everyday life activities became possible | Normal: Less than 0.43 Score range: 0–3 The higher the score, handicapped |
Full survey items (section 11) buy in the market, transportation, money management, housework, food preparation, phone, taking medicine, recent memories, hobbies, watching TV, house repair |
| Geriatric Depression Scale (GDS) | 3 | 2 | Depression, normal | Score Scope: 0–30 Normal: 0–17 points Depression: 18 points or more 18~20 points (reliability 66.67%) 21 points or more (reliability 85%) | If you have a score of 21 or more, consider antidepressants |
| Geriatric Stress Scale (GSS) | 0 | 2 | Stress, normal | Score range: 0–48 points | |
| Lewy body composite risk score | 0 | 0 | Low body, none | ||
| Conclusion | Acute cognitive impairment | Attention disorder | |||
| Recommendations | Memory and attention deficits were observed | Attention disorder and memory impairment are observed This is an improvement over the previous test (6.11.2018) | |||
The patient's memory and cognitive function were measured; there was no significant difference in these factors between Aricept and dapsone. However, with dapsone, the NPI-Q and K-IADL items were normalized, abnormal behavior disappeared, and the patient returned to daily life. Additionally, the patient's personality was restored (Fig. 2).
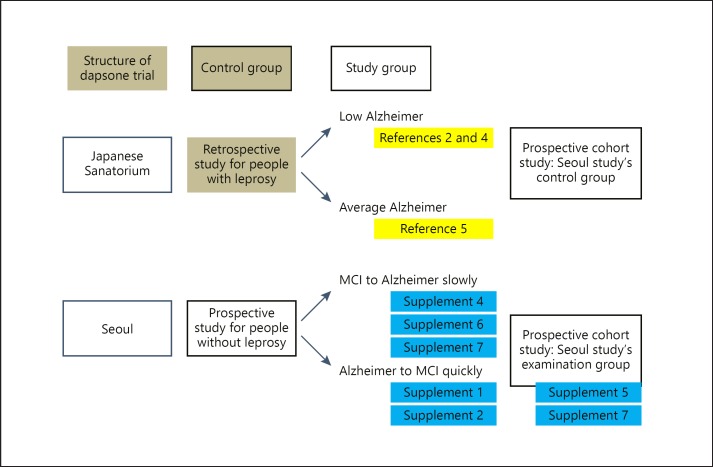
Fig. 2.
Structure and result of prospective cohort. Japanese Sanatorium Study: In the 1990s, a Japanese epidemiological survey study of leprosy patients aged 65 years or older revealed the incidence of dementia in leprosy patients [2]. A neuropathological case series of leprosy patients detected the absence of senile plaques [4]. But a similar study the following year showed no difference in the discovery of senile plaques between the patient and control groups [5]. Seoul study: a patient diagnosed with MCI through MRI and mental neurological examination in 2008 continued to take dapsone regularly until 2016 and lived a normal routine (online suppl. 4, 5, 6). However, after discontinuation, the patient was diagnosed with Alzheimer's disease in 2018, but lives with MCI as of November 2019 after taking dapsone (online suppl. 1, 2).
Our observations suggest that reducing inflammation to treat dementia syndrome, including Alzheimer's disease, is important. In addition, the cause of dementia syndrome, including Alzheimer's disease, is due to the inflammatory reaction. Moreover, we can deduce that amyloid beta protein and tau protein accumulate if the inflammatory reaction is not treated. Therefore, the onset of Alzheimer's disease is due to an untreated inflammatory response. Now, we need to find a trigger that determines that DNA and RNA produce amyloid beta proteins in response to an inflammatory response in neurons.
Discussion
Hemoglobin is a human constituent used in studies on structure and function, such as changes in hemoglobin oxygen transport ability and changes in the structure of hemoglobin in the blood. Dapsone binds to hemoglobin and causes cyanosis [10]. When dapsone binds to the molecules and structures of myeloperoxidase contained in leukocytes, it exerts anti-inflammatory and immunomodulatory effects [12, 13, 14]. These findings mean that dapsone can bind to various constituents of the body, such as various enzymes, and affect cell physiology [10]. Considering the structure of dapsone and the allosteric regulation of the molecular unit, dapsone can be presumed to preserve cells by decreasing the inflammatory response of neutrophils and can regulate the production of hypochlorous acid. This response is associated with myeloperoxidase, a kind of reductase enzyme, and has the effect of reducing inflammatory reactions. Myeloperoxidase is also a hemoprotein [15]. Dapsone inhibits myeloperoxidase, a mechanism that protects neurons. Moreover, dapsone is effective in Alzheimer's disease or stroke, a neurodegenerative disease caused by inflammation [16, 17, 18].
As a result, patients with Alzheimer's disease were able to return to their normal lives when dapsone stopped the inflammation and protected the brain cells. These findings suggest that the cause of dementia syndrome, including Alzheimer's disease, is probably due to an inflammatory response. The NPI-Q and K-IADL items in the test for the presence of cognitive impairment can be checked by examining whether the patient has returned to his/her daily life.
Inflammatory reactions are intensified by various causes in brain cells, and these cells are attacked by hypochlorite. Various foreign substances and intracellular components accumulate and cause brain cells to die. Here, dapsone was used as a prophylactic material for stopping neuronal death. One experiment demonstrated the function of dapsone in neurons in mice that experienced neuron injury by temporary cerebral ischemia followed by cerebral blood flow recovery after dapsone was administered [19].
Therefore, in this study, we suggest that the diagnostic criteria and treatment plan of Alzheimer's disease should be changed. Whether inflammatory reactions are persistent is important when diagnosing and treating Alzheimer's disease. There have been reports that most epidemiological studies are ineffective in examining a variety of issues, such as the effects of NSAIDs, MCI, and Alzheimer's disease [20]. Other results are expected if the evaluation of the effects of dapsone and MCI or Alzheimer's disease is based on NPI-Q and K-IADL items.
In the future, we will be able to prevent the development of a broad spectrum of dementia syndrome [21].
Statement of Ethics
This research was conducted ethically in accordance with the World Medical Association Declaration of Helsinki.
Disclosure Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The authors have no conflicts of interest to declare.
Funding Sources
No funding.
Author Contributions
J. Lee designed this research, performed the prospective cohort study, and wrote the manuscript. S. Choi analyzed the medical record data of dapsone and patient's mild cognitive impairment and Alzheimer's disease. C.J. Lee analyzed the medical data of dapsone and mild cognitive impairment and Alzheimer's disease. S. Oh analyzed the data of dapsone and inspected this research.
Supplementary Material
Supplementary data
Supplementary data
Supplementary data
Supplementary data
Supplementary data
Supplementary data
Supplementary data
Supplementary data
Supplementary data
Supplementary data
Acknowledgement
Prof. Chang-Soon Koh (1932–2012) who participated as a researcher who took dapsone as a joint researcher since 2010. He was a presidential physician. He reported that dapsone could also be used in his Parkinson's disease.
We would like to express our special thanks to the medical doctors who treated the patient of this research. Prof. June-young Lee, Department of Psychiatry, Seoul Municipal Boramae Hospital operated by Seoul National University Hospital; Prof. Kyung-min Lee, Department of Neurology, Seoul National University Hospital; Dr. Soh-hyun Ahn, Department of Neurology, Seoul National University Hospital; Dr. Hyeong-gil Lee, a neurologist at Kyunghee Neulfureun Geriatric Hospital. The doctors did not know this research at all. The doctors have done sincere medical care. And thanks to Eunju Lee, pharmacist. She did her best to take care of the patient.
References
- 1.McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR, Jr, Kawas CH, et al. The diagnosis of dementia due to Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011 May;7((3)):263–9. doi: 10.1016/j.jalz.2011.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.McGeer PL, Harada N, Kimura H, McGeer EG, Schulzer M. Prevalence of dementia amongst elderly Japanese with leprosy: apparent effect of chronic drug therapy. Dement Geriatr Cogn Disord. 1992;3((3)):146–9. [Google Scholar]
- 3.Appleby BS, Nacopoulos D, Milano N, Zhong K, Cummings JL. A review: treatment of Alzheimer's disease discovered in repurposed agents. Dement Geriatr Cogn Disord. 2013;35((1-2)):1–22. doi: 10.1159/000345791. [DOI] [PubMed] [Google Scholar]
- 4.Namba Y, Kawatsu K, Izumi S, Ueki A, Ikeda K. Neurofibrillary tangles and senile plaques in brain of elderly leprosy patients. Lancet. 1992 Oct;340((8825)):978. doi: 10.1016/0140-6736(92)92870-l. [DOI] [PubMed] [Google Scholar]
- 5.Kimura T, Goto M. Existence of senile plaques in the brains of elderly leprosy patients. Lancet. 1993 Nov;342((8883)):1364. doi: 10.1016/0140-6736(93)92274-w. [DOI] [PubMed] [Google Scholar]
- 6.Goto M, Kimura T, Hagio S, Ueda K, Kitajima S, Tokunaga H, et al. Neuropathological analysis of dementia in a Japanese leprosarium. Dementia. 1995 May-Jun;6((3)):157–61. doi: 10.1159/000106939. [DOI] [PubMed] [Google Scholar]
- 7.Endoh M, Kunishita T, Tabira T. No effect of anti-leprosy drugs in the prevention of Alzheimer's disease and beta-amyloid neurotoxicity. J Neurol Sci. 1999 May;165((1)):28–30. doi: 10.1016/s0022-510x(99)00057-x. [DOI] [PubMed] [Google Scholar]
- 8.Coleman MD. Dapsone: modes of action, toxicity and possible strategies for increasing patient tolerance. Br J Dermatol. 1993 Nov;129((5)):507–13. doi: 10.1111/j.1365-2133.1993.tb00476.x. [DOI] [PubMed] [Google Scholar]
- 9.Zhu YI, Stiller MJ. Dapsone and sulfones in dermatology: overview and update. J Am Acad Dermatol. 2001 Sep;45((3)):420–34. doi: 10.1067/mjd.2001.114733. [DOI] [PubMed] [Google Scholar]
- 10.Jollow DJ, Bradshaw TP, McMillan DC. Dapsone-induced hemolytic anemia. Drug Metab Rev. 1995;27((1-2)):107–24. doi: 10.3109/03602539509029818. [DOI] [PubMed] [Google Scholar]
- 11.Wozel G, Blasum C. Dapsone in dermatology and beyond. Arch Dermatol Res. 2014 Mar;306((2)):103–24. doi: 10.1007/s00403-013-1409-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bozeman PM, Learn DB, Thomas EL. Inhibition of the human leukocyte enzymes myeloperoxidase and eosinophil peroxidase by dapsone. Biochem Pharmacol. 1992 Aug;44((3)):553–63. doi: 10.1016/0006-2952(92)90449-s. [DOI] [PubMed] [Google Scholar]
- 13.van Zyl JM, Basson K, Kriegler A, van der Walt BJ. Mechanisms by which clofazimine and dapsone inhibit the myeloperoxidase system. A possible correlation with their anti-inflammatory properties. Biochem Pharmacol. 1991 Jul;42((3)):599–608. doi: 10.1016/0006-2952(91)90323-w. [DOI] [PubMed] [Google Scholar]
- 14.Uetrecht JP, Shear NH, Zahid N. N-chlorination of sulfamethoxazole and dapsone by the myeloperoxidase system. Drug Metab Dispos. 1993 Sep-Oct;21((5)):830–4. [PubMed] [Google Scholar]
- 15.Podrez EA, Abu-Soud HM, Hazen SL. Myeloperoxidase-generated oxidants and atherosclerosis. Free Radic Biol Med. 2000 Jun;28((12)):1717–25. doi: 10.1016/s0891-5849(00)00229-x. [DOI] [PubMed] [Google Scholar]
- 16.McGeer PL, Schulzer M, McGeer EG. Arthritis and anti-inflammatory agents as possible protective factors for Alzheimer's disease: a review of 17 epidemiologic studies. Neurology. 1996 Aug;47((2)):425–32. doi: 10.1212/wnl.47.2.425. [DOI] [PubMed] [Google Scholar]
- 17.Diaz-Ruiz A, Mendez-Armenta M, Galván-Arzate S, Manjarrez J, Nava-Ruiz C, Santander I, et al. Antioxidant, anticonvulsive and neuroprotective effects of dapsone and phenobarbital against kainic acid-induced damage in rats. Neurochem Res. 2013 Sep;38((9)):1819–27. doi: 10.1007/s11064-013-1087-z. [DOI] [PubMed] [Google Scholar]
- 18.Zhou T, Zhao L, Zhan R, He Q, Tong Y, Tian X, et al. Blood-brain barrier dysfunction in mice induced by lipopolysaccharide is attenuated by dapsone. Biochem Biophys Res Commun. 2014 Oct;453((3)):419–24. doi: 10.1016/j.bbrc.2014.09.093. [DOI] [PubMed] [Google Scholar]
- 19.Diaz-Ruiz A, Roldan-Valadez E, Ortiz-Plata A, Mondragón-Lozano R, Heras-Romero Y, Mendez-Armenta M, et al. Dapsone improves functional deficit and diminishes brain damage evaluated by 3-Tesla magnetic resonance image after transient cerebral ischemia and reperfusion in rats. Brain Res. 2016 Sep;1646:384–92. doi: 10.1016/j.brainres.2016.06.023. [DOI] [PubMed] [Google Scholar]
- 20.Imbimbo BP, Solfrizzi V, Panza F. Are NSAIDs useful to treat Alzheimer's disease or mild cognitive impairment? Front Aging Neurosci. 2010 May;2:19. doi: 10.3389/fnagi.2010.00019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jiang HY, Xu LL, Shao L, Xia RM, Yu ZH, Ling ZX, et al. Maternal infection during pregnancy and risk of autism spectrum disorders: A systematic review and meta-analysis. Brain Behav Immun. 2016 Nov;58:165–72. doi: 10.1016/j.bbi.2016.06.005. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary data
Supplementary data
Supplementary data
Supplementary data
Supplementary data
Supplementary data
Supplementary data
Supplementary data
Supplementary data
Supplementary data