Abstract
Neural tissue engineering (NTE) is a rapidly progressing field that promises to address several serious neurological conditions that are currently difficult to treat. Selecting the right scaffolding material to promote neural and non-neural cell differentiation as well as axonal growth is essential for the overall design strategy for NTE. Among the varieties of scaffolds, hydrogels have proved to be excellent candidates for culturing and differentiating cells of neural origin. Considering the intrinsic resistance of the nervous system against regeneration, hydrogels have been abundantly used in applications that involve the release of neurotrophic factors, antagonists of neural growth inhibitors and other neural growth-promoting agents. Recent developments in the field include the utilization of encapsulating hydrogels in neural cell therapy for providing localized trophic support and shielding neural cells from immune activity. In this review, we categorize and discuss the various hydrogel-based strategies that have been examined for neural-specific applications and also highlight their strengths and weaknesses. We also discuss future prospects and challenges ahead for the utilization of hydrogels in NTE.
Keywords: hydrogel, neural tissue engineering, physical guidance, chemical cues, axonal growth
1. Introduction
The mammalian nervous system is broadly divided into the central and peripheral nervous systems. The central nervous system, comprising the brain and spinal cord, is afflicted by numerous neurological conditions such as neurodegenerative disorders, demyelinating diseases and ischaemic insults. Treatment of such disorders varies in number and approach, though very few result in complete recovery or resolution. Current treatment strategies for neurological conditions broadly encompass a twofold approach, where the first step is to curtail the ongoing disease pathogenesis, followed by a more long-term strategy that aims to prevent further tissue damage and recurrence. A third approach, which is currently more in an experimental stage, attempts to replace lost neural tissue or promote existing tissue to regenerate within the nervous system. However, all of the above three approaches face significant challenges. The nervous system is well known to be protected by the blood–brain barrier, which blocks the majority of therapeutic compounds from reaching the neural tissue [1]. Secondly, neural regeneration in the central nervous system poses a unique challenge to tissue engineering strategies, mainly because the intrinsic nature of the adult nervous system is heavily stacked against neuronal differentiation and regeneration. Cell-secreted molecules such as chondroitin sulfate proteoglycans (CSPGs) [2], Nogo [3] and myelin-associated glycoprotein [4] are well known to inhibit neuronal regeneration. This growth-inhibitory environment is particularly apparent in the central nervous system, while the peripheral nervous system demonstrates comparatively better regenerative potential, mainly because of the lack of neural inhibitory factors [5]. Despite such challenges, researchers have developed various tissue engineering strategies that are specific for the brain, spinal cord and nerve, culminating in the specialized field of neural tissue engineering (NTE). The underlying principle of NTE is to develop an artificial biological environment that is conducive for neural tissue growth. NTE strategies consist of several multi-faceted approaches that aim to develop three-dimensional (3D), viable neural tissue capable of replacing damaged or diseased neural components, promote neural regeneration and restore function. Here, it must be noted that the term ‘neural tissue’ in the context of tissue engineering is slightly misleading since it appears to give an impression that it predominately deals with neurons. In fact, strategies in NTE encompass contributions from both neurons and non-neuronal glial cells [6] for developing functional neural tissue. Incidentally, during embryological development, the loss of neuronal regenerative potential, a major challenge in NTE, coincides with the development of the non-neuronal glial cells, suggesting the importance of these cells in structuring the nervous system. Hence, when designing and developing ideal tissue mimetic conditions for NTE, consideration of factors that sustain and promote neuronal, non-neuronal and extra-neural matrix has prime importance [7,8].
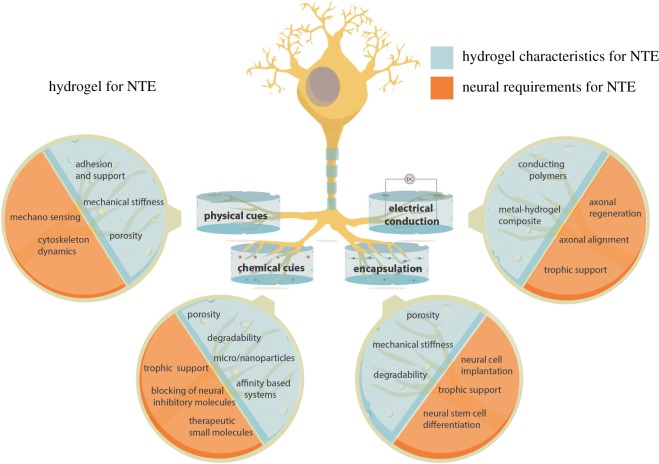
NTE uses biomaterials, cellular components and neurotrophic factors to facilitate neuronal differentiation and regeneration. Currently, tissue engineering strategies for neuronal regeneration can be classified into four categories. These include (a) incorporation of guidance cues, (b) factors to promote cell adhesion and proliferation, (c) drug delivery components, and (d) electrical conductivity of the tissue supporting matrix. Each of these components has been used in previous studies, either individually or in combination to promote neural regeneration. Among the several biomaterials available, hydrogels provide maximum flexibility and ease in modifying material characteristics to suit neural regenerative requirements (figure 1). In this review, we explore the role of hydrogels in assisting neural tissue growth with the potential for clinical applicability.
Figure 1.
Schematic overview depicting the different characteristics of hydrogel systems that can be used for neuronal growth and differentiation. (Online version in colour.)
Hydrogels are a category of polymeric materials with physical and chemical properties that make them conducive for cell growth. It is well known that tissue development within artificial regenerative environments requires scaffolding platforms that provide an overall microstructure that mimics the extracellular matrix. Hydrogels provide such a 3D micro-architecture [9] that is conducive for tissue regeneration and forms the primary basis for their use in neural regeneration (figure 2). The ease and adaptability of hydrogels can be assessed by their applicability in regenerating a range of varying tissue types such as highly dense bone to very soft tissues like liver. However, hydrogels are particularly suitable for neural tissue regeneration, as discussed below.
Figure 2.
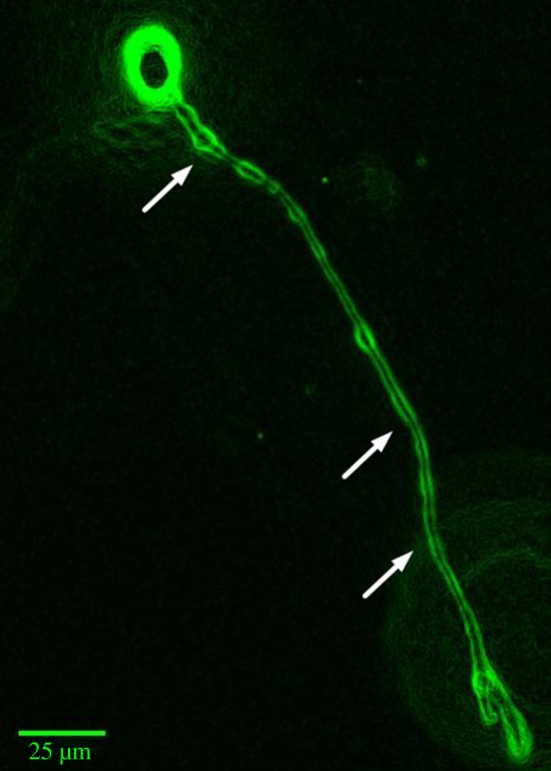
Edge and contrast-enhanced image of a calcein-stained PC-12 cell encapsulated in a collagen type I–alginate hydrogel matrix, showing an extended neurite manoeuvring through the hydrogel matrix (depicted by arrows). (Online version in colour.)
2. Physical guidance cues within hydrogel systems
A neuron comprises the cell body and its extensions, called axons and dendrites, that conduct action potentials across long distances. Axons use micro-topographical cues to sense and probe their immediate surroundings to trigger adaptive responses such as neuritic extension or retraction [10]. Such observations are clearly visible in 3D scaffolds developed using advanced lithography techniques [11,12], where neurites track along scaffold surfaces and change direction according to topography (figure 3). In addition, extracellular proteins or lipid molecules found on the surface of adjacent cells embedded within hydrogels are also used as guidance cues [13]. Structurally, it has been observed that the tip of the growing axon, termed the growth cone, adheres to the adjacent substrate, and the resulting structural rearrangement of the microtubule and F-actin proteins within the axon promotes its extension [14]. In conditions such as spinal cord injury and amyotrophic lateral sclerosis, where axons degenerate, hydrogels have been shown to promote axonal growth, repair and restoration of function to a certain extent [15,16], partly by altering the surface micro-topography that is sensed by the axons [17]. Observations such as the above suggest that axonal growth and development in a hydrogel matrix primarily depend on certain interconnected factors:
(a) mechanical stress experienced by the neural membrane
(b) neuronal cytoskeletal dynamics within the scaffold
(c) mechano-sensing ability of the neurons.
Figure 3.
(a,b) Representative scanning electron microscopy images of the 3D-PEGDA-hydrogel scaffold cultured with neuro2A cells. (c) Representative Z-stacked two-photon confocal image depicting the presence of neuro-2A cellular network (DAPI, blue; phalloidin, red; β-tubulin, green). (All images were kindly provided by Dr Angelo Accardo, TU Delft (Delft University of Technology), Department of Precision and Microsystems Engineering (PME), The Netherlands.) (Online version in colour.)
2.1. Mechanical stress
As mentioned earlier, the interaction of neurons with their environment has been shown to have several implications for morphological and functional outcomes. The mechanical stress experienced by the neuronal membrane along the interface of the hydrogel surface dictates, to a large extent, axonal growth and directionality. However, it is important to note that the mechanical stress of the membrane is also affected by intrinsic factors such as the contractile nature of the filopodia and cytoplasmic and cytoskeletal pushing forces [18]. In addition, the maintenance of mechanical tension all along the neurite helps in attachment, in establishing neuronal synapses, in regulating the diameter of the neurites and in neuronal arborization and arrangement [10]. An increase in the viscoelasticity of the axoplasm due to microtubule and actin polymerization has also been reported to contribute to the rate of axonal elongation [19]. Hydrogel systems capable of providing physical guidance cues for neural growth are listed in table 1.
Table 1.
Hydrogel systems capable of providing physical guidance cues for neural growth. iPSC, induced pluripotent stem cell.
| no. | hydrogel material | cell type | hydrogel property | observation | reference |
|---|---|---|---|---|---|
| 1 | alginate–calcium 3D scaffolds | neural stem cells | elastic modulus (alteration in cross-linking density) | hydrogel with modulus of 180 Pa showed a 20-fold increase in β-III tubulin expression | [20] |
| 2 | polyacrylamide with laminin | retinal ganglion cell axons | shear modulus (0.1 kPa and 1 kPa) | axons grow towards the soft substrate | [21] |
| 3 | polyacrylamide gel bi-functionalized with poly-lysine and laminin motif IKVAV | embryonic and adult neural progenitor cells | gel stiffness (2 kPa and 0.2 kPa) | increased adhesion, maturation and neurogenesis | [22] |
| 4 | polyacrylamide gel with PDL coating | hippocampal neurons | substrate stiffness altered | stiffer substrates suppressed neuritogenesis | [23] |
| 5 | PMMA-coated silicon wafers | adult sympathetic and sensory ganglia | the presence of multiple ridges/grooves in the hydrogel matrix | axon projections guided towards hydrogel ridges | [24] |
| 6 | polyacrylamide or fibrin gels | cortical neurons and astrocytes | substrate stiffness altered | neural sprouting observed on softer gels, astrocyte attachment on stiffer gels | [25] |
| 7 | DNA strands covalently linked to polyacrylamide hydrogel | neurons and glia | substrate stiffness altered | neural sprouting observed on softer substrates | [26] |
| 8 | polyacrylamide hydrogel with (GAG) binding peptide | human pluripotent stem cells | substrate stiffness altered | softer substrates enhanced neuronal differentiation | [27] |
| 9 | methacrylate-modified hyaluronic acid (multi-layer) | human iPSC-derived neural progenitor cells | 3D culture system and varying elastic moduli | neuronal differentiation enhanced in soft culture systems | [28] |
In addition to the inherent mechanical forces acting within the neuron, those cultured on hydrogel matrices experience forces that are intrinsic to the scaffold material. One such hydrogel property is substrate stiffness: it has been observed that cortical neurons cultured on synthetic and naturally occurring hydrogel materials such as polyacrylamide gels and fibrin, respectively, have superior cell survival and neuritic extension when the hydrogel elastic modulus is closer to that of the softer extracellular matrix [25]. While the F-actin polymerization mechanism remains unchanged in neurons cultured on hard and soft hydrogels, the softer gels tend to induce increased neuronal sprouting compared with harder gels. Interestingly, astrocytes demonstrate adhesion properties that seem to be in stark contrast to those of neurons, as they appear to do much better on stiffer substrates [25]. Similar observations have been noted in hydrogels containing varying concentrations of alginate and calcium that have been developed into 3D cell-encapsulating scaffolds. Neural stem cells cultured on alginate–calcium 3D hydrogels with relatively lower stiffness (Young's modulus of approx. 180 Pa) showed a (approx. 20-fold) relative increase in expression of neuronal marker (β-III tubulin) [20], pointing towards a clear correlation between neural differentiation and hydrogel stiffness. Similarly, neurons cultured on DNA-cross-linked hydrogels with reduced mechanical stiffness exhibit enhanced axonal length and an increase in expression of focal adhesion kinase [26], a protein that is known to sense mechanical stress [29].
Another curious observation that may in part be related to the mechanical stress experienced by the axonal membrane is the propensity of axons to follow ridges on scaffolds. Axonal growth of adult neurons cultured on synthetic hydrogel (polymethyl-methacrylate; PMMA) with nano-printed patterns has been found to be dependent on topographical surface patterns where a distinct preference to adhere and extend on hydrogel ridge edges and elevations was observed, though the actual mechanism and the reason for such a preference remains unclear [24].
Axonal tips have also been shown to advance faster on softer substrates, and have been specifically found to move away from harder areas on a scaffold, depending on the local stiffness gradient. It has been increasingly found that, as the stiffness of the hydrogel scaffold comes closer to the stiffness of the local tissue matrix for the specific neural cell type, neural cells tend to do better in terms of growth and function. There can be multiple underlying reasons for this observation, but, specifically, piezo 1, a mechano-sensitive ion channel, has received considerable attention [21]. External traction forces exert their effect on the lever-like mechano-gating mechanism of the piezo 1 channel that elicits the entry of calcium, a known modulator of differentiation and axonal growth [30]. This will be discussed in more detail in §2.3.
2.2. Neuronal cytoskeletal dynamics within the hydrogel scaffold
Developing or regenerating neurons project extensions (neurites) that later differentiate into dendrites or axons. Actin, microtubules and neurofilament proteins form the major structural components of such neuronal projections. Actin filaments are thin, flexible, two-stranded lattice structures, consisting of the small bi-lobed protein called actin, that are found just underneath the neurite membrane. Actin provides membrane stability and acts as a supporting platform for the movement of cargo over short distances. Neurite growth is a function of dynamic shortening and lengthening of the actin filament, primarily controlled by the dynamic actin polymerization mechanism [31]. Branch retraction and collateral branching are some of the neurite properties that are heavily controlled by actin filament dynamics, aided by the actin-binding cytoplasmic proteins. Microtubules, unlike actin filaments, are hollow, tubular structures present within axons; their flexural rigidity is far higher than that of the actin filament, and they are capable of resisting compressive forces. Stiffness and mechanical rigidity of the axon depend on the cross-linking of actin and microtubules [14,32]. During neuritic advancement, the dynamic polymerization–depolymerization mechanism generates cyto-mechanical forces that regulate the dynamic assembly and organization of the actin–microtubule cytoskeleton [33]. Furthermore, the adhesion of filamentous actin cytoskeleton to the neural membrane through numerous adaptors and signalling proteins acts as a ‘molecular clutch’, thereby redirecting the force of actin polymerization towards the leading edge and resulting in growth and guidance of the axon [34].
In addition to the growing actin network (pushing force), the leading edge is tightly regulated by myosin II, a motor protein recently implicated in the severing of actin bundles, causing retrograde flow of the actin network [35]. It is also interesting to observe that filamentous actin tended to exhibit a larger circumferential meshwork and larger growth cone neuritic tips when neuronal cells were cultured on a hydrogel surface whose Young's modulus (1.7 ± 0.3 × 102 Pa) was comparable to that of brain tissue [36], while the average length of F-actin bundles was found to be significantly reduced in cells cultured on softer hydrogels [23]. Thus, it is important to note that physical cues such as stiffness of the hydrogel induce a change in the cytoskeletal arrangement of F-actin, which may, in turn, regulate neuritogenesis. The cytoskeletal rearrangement is responsive to various extracellular cues, including spatial and temporal changes in trophic signal concentration gradient, physical cues and the presence of other cells in the vicinity [37,38].
Adhesion of neurites to the functionalized surface of hydrogel substrate via proteins such as integrins, cadherins and other cell-specific adhesion molecules is known to affect filamentous actin dynamics [18]. One of the most likely mechanisms for the substrate–actin interaction could be the recruitment of scaffolding and signalling proteins such as focal adhesion kinases, talin, paxillin and A-actinin within the neural cells [14]. The modulation of neuro-cytoskeletal structure and its effects on neuronal stem cell differentiation within hydrogel matrices have also been recently studied. Neuronal stem cells encapsulated in polyacrylamide hydrogel functionalized with a glycosaminoglycan (GAG) binding peptide demonstrated good substratum-induced differentiation [27]. Conversely, highly compliant hydrogels have been observed to reduce neuronal differentiation, possibly because of lower filamentous actin polymerization [27].
Alternatively, modifying F-actin polymerization and stress fibre formation in neural cells by directly modulating F-actin regulatory proteins such as Yes-associated protein (YAP) and transcription co-activator with PZD-binding domain (TAZ) are among the strategies that have been incorporated into hydrogel-based neural cultures [27].
In a recent study, embryonic and adult-derived neuronal progenitor cells (aNPCs) have also been observed to adhere and undergo differentiation within a hydrogel bi-functionalized with poly-lysine (PL) and laminin motif IKVAV (isoleucine–lysine–valine–alanine–valine) with varying stiffness. IKVAV and PL functionalization, with an overall hydrogel stiffness of 2 kPa, exhibited a more conducive environment for the attachment of cortical progenitors and showed a marked increase in β1-integrin expression, which is crucial for neurite growth [22].
Additionally, in many of the studies designed to identify neuronal cytoskeletal determinants, it is important to recognize that 3D culture systems provide a more tissue-mimetic architecture and elicit responses that are more physiologically relevant than conventional two-dimensional (2D) culture systems. Cytoskeletal forces that govern the movement of neurites in a 2D system are drastically different from the forces experienced in a 3D culture system that leads to neuronal migration and maturation [39]. This is possibly due to differences in cellular adhesion, surface topography and ability to incorporate soluble factors into a 3D culture system. Additionally, in a 3D system, incorporation of soluble factors such as nutrients or growth factors results in dynamic spatial gradients that influence cellular migration, interaction and differentiation, which are not observed in a homogeneously distributed 2D system. The planar environment of 2D systems confines the cells and offers negligible resistance to migration; also, only segments of the cellular membrane interact with the extracellular matrix, leading to an unnatural and polarized integrin binding that affects the phenotype and cytoskeletal arrangement of the cell [39].
2.3. Mechano-sensing by neurons
Development of the nervous system encompasses complex spatio-temporal signalling patterns that regulate axonal growth and synapse formation. Interestingly, alteration of physical cues in the neuronal environment such as surface topography, rigidity/stiffness, anisotropy, wettability and surface charge can introduce dynamic changes in cellular development and response. Neurons convert physical forces into biochemical signals owing to the presence of specialized channels called mechanically activated (MA) ion channels. Acid-sensing ion channels and transient receptor potential ion channels, particularly TRPV4, potassium ion channels with ‘two-pore domains’ such as the TWIK-1-related K+ channel (TREK-1), TREK-2, TWIK-related arachidonic acid-stimulated K+ channel and Piezo ion channels, are among the few such ion channels associated with mechano-sensing [40]. Previous studies have demonstrated that exertion of mechanical stress on neural cells cultured within polyacrylamide hydrogels induces growth cone collapse, retraction of neuritis and alteration of the cytoskeleton. These effects have been primarily attributed to the sharp increase in intracellular calcium within the cell soma and growth cones, secondary to activation of mechanical stretch receptors [10]. However, there is very little information in the literature regarding the role of neuronal mechano-sensitive ion channels within a larger hydrogel platform, and further studies are needed to shed light on this important topic.
3. Controlled release of biomolecules from hydrogel systems
In addition to providing mechanical support and physical cues for regenerating neurons, hydrogels can be further used to release exogenous biomolecules in a controlled manner by incorporation of these agents into the hydrogel matrix. In fact, hydrogels have been widely used for the incorporation of drugs, small molecules, extracellular matrix proteins and even secretory cells for the purpose of neural regeneration. The fundamental principle behind the release of loaded molecules in any hydrogel system stems from its degradative property. Overall, hydrogel degradation is primarily the function of its surface erosion (degradation of the overall polymeric network architecture) and bulk erosion (cleavage of individual bonds within the polymer) properties [41]. Hydrogel degradation can be controlled by changing the polymeric content, molecular weight, pore density and degree of cross-linking, which in turn affects the release profile of loaded substances. Control over hydrogel degradation plays a crucial role in the design and development of strategies for NTE.
A list of hydrogel systems used for the delivery of various biomolecules is shown in table 2. Thus, by incorporating physical cues and biomolecules, hydrogels can be designed to provide a more synergistic environment that allows better development of functional neural tissue.
Table 2.
Hydrogels for the delivery of therapeutic molecules for NTE.
| no. | hydrogel | drug/factor | injury model | mode of delivery | outcome | reference |
|---|---|---|---|---|---|---|
| 1 | PLA-b-PEG-b-PLA | NT3 | spinal cord injury (T8 segment) | injection at the lesion cavity | enhanced axonal growth | [42] |
| 2 | poly-L-ornithine-coated alginate | BDNF-secreting fibroblast cells | spinal cord injury (C4 segment) | graft placement | functional recovery of forelimb and hindlimb | [43] |
| 3 | fibrin conjugated with heparin | NGF, BDNF or NT3 | in vitro study | in vitro | prolonged release of trophic factors; neurite extensions observed | [44] |
| 4 | fibrin conjugated with heparin | NT3 | spinal cord injury (T9 segment) | graft implants | increase in neural fibre density | [45] |
| 5 | hyaluronan and methylcellulose | EGF or PEG-EGF | stroke | epi-cortical graft implant | enhanced proliferation of neural stem/progenitor cells | [46] |
| 6 | fibrin | chondroitinase ABC | spinal cord injury (C4 segment) | graft implant adjacent to the lesion | decrease in glycosaminoglycan | [47] |
| 7 | fibrin | GDNF | — | in vitro | enhanced neurite extensions | [48] |
| 8 | fibrin | heparin-immobilized NGF | sciatic nerve transection | conduit implant | axon regeneration observed | [49] |
| 9 | fibrin | NT3 | spinal cord injury (T9 segment) | graft implant | neuronal fibre sprouting but no functional recovery | [50] |
| 10 | hyaluronic acid | Nogo-66 receptor antibody | — | in vitro | pH-dependent release of antibody | [51] |
| 11 | hyaluronic–poly-L-lysine | anti-NgR | spinal cord injury (T8–T9 segment) | implant | axon extensions towards HA-PLL/anti-NgR hydrogel | [52] |
3.1. Release of neurotrophic factors
Neurotrophic factors are a group of biomolecules that have received special attention in NTE. It is well known that neurotrophic factors are essential for regulating and promoting growth, proliferation and overall survival of neural cells. Some well-known neurotrophic factors include nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF), neurotrophin-3 or -4 (NT3/NT4), glial cell line-derived neurotrophic factor (GDNF) and ciliary neurotrophic factor (CNTF). Tyrosine receptor kinase (Trk A, Trk B, Trk C) and p75 are some common neurotrophic receptors that have been targeted in NTE. In neuronal injury, neurotrophic factors such as NGF and BDNF along with their receptors have shown increased expression [53], while trophic factors such as NT3 and Trk B are downregulated [54]. Such alterations in trophic factors and their receptor expression have severe detrimental effects during axonal regeneration. A viable strategy that has been used in NTE is the exogenous provision of growth factors to enhance recovery and regeneration of injured axons. Direct delivery of neurotrophic factors has proved to be difficult, considering the large molecular size of the trophic factors, relatively short duration of activity and systemic side effects such as diffuse myalgia and hyperalgesia [55–57].
Hydrogels, with their ease of synthesis and ability to deliver loaded biomolecules, provide a strong platform for supplying neurotrophic factors to the region of interest. However, the delivery of neurotrophic factors to facilitate neuronal regeneration comes with its own set of challenges. Regenerating neural cells require trophic support in specific concentrations and at particular phases of their growth, making it imperative that the hydrogel system is designed to suit such fundamental requirements. Despite such challenges, based on the rate of degradation of cross-linking polymers, the nature of pores and other physical characteristics of the hydrogel, the incorporated neurotrophic factors can be temporally released with control over concentration, duration and rate of release. For instance, the commonly used poly-lactic acid (PLA)–polyethylene glycol (PEG) hydrogel formulations, by a series of chemical modifications, have been shown to release neurotrophin-3 (NT3) in a manner in which the initial burst release followed by a prolonged release contributed positively towards sustained neural growth [58]. It has also been observed that, by simply decreasing the hydrogel pore size and density, the initial NT3 burst release could also be concomitantly decreased, thus allowing for flexibility in delivering neurotrophic factors in the desired concentration and duration. Moreover, such NT3-loaded hydrogels have also been successfully administered at the site of spinal cord lesions in animal models and improvements in axonal regeneration have been observed [42].
Even though the physical and chemical characteristics of the hydrogel are important in determining the duration and the delivery dosage of the loaded biomolecules, it has also been observed that some trophic factors tend to interact with the hydrogel material and affect their release profile. For example, collagen hydrogels designed for releasing epidermal and basic fibroblast growth factors (EGF and FGF-2) showed a differential release profile, with prolonged release of FGF-2 in comparison with EGF [59]. Such observations are attributed to strong ionic interactions between charged groups present between collagen and FGF-2, which ultimately prolong its release in the gel matrix. Similarly, affinity-based hydrogels are a group of specially modified systems that release the incorporated biomolecule only after cleavage of the bond tethering the biomolecule to the hydrogel. Several neurotrophic factors have been incorporated into the hydrogel matrices using the affinity-based system. Negatively charged sulfate groups present on heparin have been extensively used to immobilize various neurotrophic growth factors such as NGF, GDNF and NT3 [44,45,48,60]. Fibrin is a widely used polypeptide that has been used with heparin to aid in biomolecule delivery [61]. A common strategy is to cross-link heparin to the fibrin matrix using a bi-domain peptide, where one end of the domain cross-links within the fibrin and the other end interacts with heparin [62]. Incorporated growth factors non-covalently interact with the negatively charged groups of free heparin, reducing the diffusivity of the trophic factors from the hydrogel and prolonging their release. Similar to fibrin, collagen is another polypeptide widely used as a wound dressing, as a filling material and as a drug delivery reservoir. Applications of injectable collagen hydrogels have been explored for the delivery of trophic factors such as platelet-derived growth factor and insulin-like growth factor I in injury models of the peripheral nerve [63].
Other innovative methods for controlling biomolecule release include the incorporation of trophic factor-loaded micro/nanoparticles within the hydrogel matrix. Studies using hyaluronan and methylcellulose (HAMC) hydrogel incorporated with polymeric PLGA nanoparticles loaded with NT3 have demonstrated slow release of NT3 [64]. The prolonged release from such dual composite systems can be attributed to hydrophobic interactions between methylcellulose and the PLGA nanoparticles forming a diffusive barrier that hinders and slows payload release. Such dual composite systems have also been tested under in vitro conditions using sensory neuronal cultures, where enhanced neuritic extensions have been observed for as long as 28 days [64]. Similarly, altering the composition of BDNF and GDNF containing PLGA nanoparticles within a PEG hydrogel system has been shown to produce a differential release profile of the loaded neurotrophic factors. This system offered a slow-release component that provided BDNF for 56 days, and a relatively faster component that released GDNF within 28 days [65]. Such types of delivery systems provide temporal control for releasing multiple biomolecules from a single hydrogel matrix which can be used to provide the complex cues necessary for neural regeneration.
3.2. Release of factors that block neural regeneration inhibitors
A significant challenge in neural tissue repair is the non-permissive environment created at the site of injury due to the release of various inhibitory molecules. Glial scar, which is composed of astrocytes and elements of connective tissue, is one of the major impediments to axonal regeneration. The inhibitory molecules produced from activated glia within the scar such as CSPGs [66], tenascin [67] or ephrin-B2 [68] greatly impede the regeneration process.
To counter this intrinsic resistance of neural scar tissue to axonal growth, strategies have been adopted where molecules that neutralize these inhibitory factors have been incorporated into hydrogels for local release in the scar areas. Chondroitinase ABC (ChABC), a bacterial enzyme that degrades GAG chains on CSPGs, has been well reported to facilitate the growth of axons in nerve injury models [69]. In one such study, fibrin gels loaded with ChABC, implanted proximal to the site of the spinal cord lesion, demonstrated a decrease in GAG level at the site of the lesion [47]. Since ChABC has a high rate of degradability and poor thermal stability, various strategies have been developed to improve its functionality. An affinity-based system using the recombinant fusion protein of ChABC–Src homology domain 3 (SH3), incorporated in a methylcellulose hydrogel modified with SH3 binding peptide, provided a platform for tunable release [70]. In another study, a combinatorial approach was attempted, where NT3 and thermally stabilized ChABC in agarose hydrogel were tested in a spinal cord injury model, with results demonstrating enhanced axonal regeneration capacity and improvement in locomotor function [71]. Similarly, Nogo, a known myelin-derived axonal outgrowth inhibitor, and its axonal receptor have been targeted for promoting axonal regeneration. Antibodies against Nogo-66, which is a 66-residue domain of Nogo that is expressed on the surface of oligodendrocytes, were conjugated to hyaluronic acid (HA) hydrogel to yield a prolonged release of the antibody [51]. Similarly, incorporation of the antibody against the Nogo receptor (NgR) in HA hydrogel was found to enhance the growth of hippocampal neurons in vitro [72] and to promote functional recovery in an animal model of stroke [73].
3.3. Release of drug molecules from hydrogel
Injectable hydrogel systems have been quite extensively used to deliver therapeutic molecules in various neurological disorders, but particularly so for treating tumours arising in the nervous system, such as glioblastoma multiforme (GBM), a fast-growing aggressive tumour with high mortality rates. The overall rationale is to maintain a therapeutically relevant concentration of the drug in the local area, thereby serving two purposes: preventing tumour recurrence and reducing systemic toxicity. However, the key feature is to have a drug delivery system that can release the anti-cancer drug for prolonged periods of time. Polyethylene glycol-dimethacrylate (PEG-DMA) injectable hydrogel incorporated with temozolomide, a commonly used chemotherapeutic agent against GBM, has been shown to release the drug for extended durations of time [74]. Similarly, hydrogels have also been used to release neurotransmitters in animal models mimicking Parkinson's disease. Hydrogel composite comprising of dextran dialdehyde and gelatin, loaded with dopamine, when delivered into the striatum region of a dopamine-deficient Parkinson's mouse model, resulted in restoration of motor activity [75].
Alternatively, strategies that do not involve direct administration of dopamine-loaded hydrogels into the region of interest include attempts to breach the blood–brain barrier. Intraperitoneally administered polyvinylpyrrolidone-poly (acrylic acid) (PVP/PAAc) nano-gels loaded with dopamine have been found to partially cross the blood–brain barrier and release the neurotransmitter within the brain parenchyma [76]. In addition to the examples cited above, drug-incorporated hydrogels have also been tested on several experimental models of stroke [77], Alzheimer's disease [78] and seizure [79].
Be it the release of drug molecules, blockers of regenerative inhibitors or neurotrophic factors, the fundamental principle behind release of loaded substances from the hydrogel depends on its degradation. For instance, in spinal cord injury models, it has been observed that axonal regeneration occurs at a rate of 1–2 mm per day [80], and most studies have routinely shown that recovery of spinal cord function occurs within the first three weeks in rodent models [81]. Any hydrogels being tested for spinal cord injury would likely consider these time lines for release of therapeutic molecules. However, from a translational point of view, this scenario may be different, since the nature of the spinal cord injury (crush, contusion, section, etc.) would dictate the rate of regeneration, and the hydrogel may have to be customized based on the extent and type of the injury. Thus, consideration has to be given to such biological variability for specific applications and the hydrogel matrix needs to be specifically formulated.
3.4. Cells encapsulated in hydrogels
Cellular transplantation, a process where cells cultured in vitro are implanted into an injured or diseased area, is gaining much recognition for its application in NTE. The overall idea is to introduce specialized cells that can either produce therapeutic molecules to aid neural regeneration or simply replace the injured or dead cells by integrating with the surrounding tissue. Encapsulation of such implanted cells has been shown to enhance functional outcome, particularly if the implanted cells are protected from the immune response. Several natural and synthetically derived hydrogels have been used to encapsulate cells, and it has been observed that the encapsulated cells are directly affected by the degree of cross-linking and degradability of the hydrogel [82]. For instance, cross-linked alginate hydrogel coated with poly-L-ornithine has been used to encapsulate and form a size exclusion barrier around non-autologous fibroblast cells, for facilitating cell secretion. It was observed that the alginate gel allowed diffusion of cell-secreted neurotrophic growth factors, but prevented the infiltration of immune cells [43]. In a similar study using the same concept, BDNF-secreting non-autologous fibroblast cells encapsulated in alginate beads showed good results when implanted in the dorsolateral funiculus of the spinal cord. The implanted cells–hydrogel complex induced recovery of limb motor strength in spinal cord-injured rats, along with neuritic sprouting in the injured axons. Interestingly, there was marked absence of immune reactivity against the implanted cells within the alginate beads [43].
In recent years, a large body of research has strongly pointed towards the feasibility of using hydrogel systems to encapsulate and stimulate neural progenitor cells to naturally undergo differentiation into various cell types. In a recent study, it was observed that 4-dibenzocyclooctynol (DIBO)-functionalized PEG hydrogel tethered with laminin and interferon-γ strongly facilitated the differentiation of encapsulated neural stem cells to functional neurons [83]. The DIBO-PEG hydrogel with a shear modulus measuring around 0.7 kPa, which housed encapsulated stem cells, facilitated a marked increase in neuronal differentiation, suggesting that strategies that combine techniques such as encapsulation and provision of trophic support appear to have better outcomes [83]. Interestingly, it has also been observed that different types of neural progenitor cells behave differently within hydrogel matrices. A well-documented observation is that aNPCs cultured in HA and PEG-based hydrogel show strong differentiation potential, compared with fetal-derived neuronal progenitor cells [84].
While there are several ongoing studies investigating the regulatory and signalling pathways that are instrumental in dictating how neuronal differentiation occurs in 3D matrices in vitro, attempts to incorporate stem cell-encapsulated hydrogels in animal models of neurological disorders have yielded some promising results. An amyloid-inspired peptide hydrogel that uses α-synuclein protein to form a nano-fibrous network was found to promote differentiation of mesenchymal stem cells into a neuronal lineage when implanted into the substantia nigra and striatal regions of the basal ganglia in animal models of Parkinson's disease [85].
In addition to the incorporation of simple peptides in hydrogels, there have been recent attempts to use certain self-assembling peptides (SAPs) in the development of specialized hybrid hydrogels. SAPs are small peptide molecules that can self-assemble, form nano-fibrous scaffolds, incorporate bioactive sequences and be adapted for external stimulus-triggered responses (pH, ionic gradient, hydrophobicity, temperature) [86]. The self-assembling nature of these peptides has been attributed to the various non-covalent interactions, such as hydrophobic, π–π, hydrogen bonding and electrostatic interactions, which can be modified to yield gels with the required properties [87]. SAP hydrogels consisting of peptides arginine–alanine–aspartate (RAD)16-I and RAD16-II were found to undergo molecular self-assembly to form ionic β sheets, and were used as substrates for neuritic growth and synapse formation in vitro [88]. Neuritic cells incorporated in the above SAP hydrogels demonstrated excellent growth and synapse formation [88], and also exhibited minimal inflammatory response when implanted in vivo. Similarly, nano-fibrous scaffolds formulated with RADA16 peptide, with appended motifs from naturally occurring cellular proteins, demonstrated the differentiation ability of encapsulated neural stem cells, along with upregulation of genes responsible for neuronal growth [89]. However, RADA16-I SAP hydrogel functionalized with BMHP1 (bone-homing peptide) showed only moderate gains in motor recovery when implanted in animal models of spinal cord injury [90]. Furthermore, SAP hydrogels have also been recently used in the delivery of neural progenitor cells to the brain, where N-fluorenylmethyloxycarbonyl self-assembling peptides (Fmoc- SAPs) appended with laminin and fibronectin were used to deliver cortical neural progenitor cells to the mouse brain [91]. Results from this study showed neuritic extensions from the cortical neural progenitor cells into the brain parenchyma and displayed limited glial scarring [91]. These results indicate that SAP hydrogel systems have generally exhibited favourable outcomes for neural regeneration, but there are several questions related to immune response towards peptides, functional and behavioural changes in response to SAP hydrogel incorporation in the brain and long-term tissue effects that need to be addressed.
4. Electrically conductive hydrogels for neural tissue engineering
It has been increasingly found that electrical stimulation of injured neural tissue leads to profound cellular changes in terms of regeneration and repair. Experiments in both in vitro and in vivo models have demonstrated that a depolarizing current applied in the vicinity of injured axons has significant effects on axonal sprouting and regeneration [92]. The mechanism of how electrical stimulation facilitates neuronal regeneration is not completely clear, but several hypotheses, including the role of voltage-dependent calcium channels [93], changes in local electrical field of extracellular matrix molecules [94] and electrical stimulation-dependent modulation of neurotrophic factor release, have been proposed [95].
It is interesting to note that much of the research pertaining to electrical stimulation of neural tissue has been focused on peripheral nerve and spinal cord repair. Traditionally, in peripheral nerve defect(s) and spinal cord injury models, nerve grafts or synthetic nerve conduits have been used to bridge gaps between severed or damaged neural tissue. Nerve conduits are made up of either biological or synthetic polymers such as collagen and gelatin or polyethylene glycol and polyvinyl alcohol polymers, respectively [96]. To facilitate the flow of ions through the supporting matrix, various conducting materials have been tried, and such systems have been termed conducting hydrogels. At this juncture, it is important to understand the nature of electrical interaction between tissues and external conducting materials. Tissues use mobile ions as charge carriers, while traditional electrical conductors such as metallic electrodes solely depend on mobile electrons. Upon electrical stimulation of an electrode, current is transduced to the tissue via Faradic and capacitance charge transfer [97]. Faradic currents are generated through electrochemical reactions (oxidation or reduction) at the electrode–tissue interface, which over time can be detrimental to tissue, while capacitance currents are generated through simple charge separation at the electrode–tissue interface [97]. Since conducting hydrogels have both mobile electrons (from conducting polymer chains) and ions (from the aqueous phase) and exhibit higher volumetric capacitance, they offer tremendous opportunity for interfacing material–neural interactions.
The electro-conductive hydrogel usually comprises two varieties of polymers, namely a conducting and a non-conducting polymer. The latter provides 3D aqueous gel properties and acts as a structural support for the growth of cells, while the conducting polymer is responsible for imparting electrical properties. Synthesis of this hybrid network can be carried out in two ways: (i) electro- or chemical polymerization of the conducting monomer in the prefabricated hydrogel or (ii) combining the conducting polymer monomers followed by an electrochemical polymerization process [98]. In addition to conducting polymers, hydrogels have been supplemented with metallic components to increase their conducting property. A micro-patterned silver nanowire–PEG hydrogel composite was used to provide both electrical and physical cues to facilitate directional and enhanced growth of neurites [99].
Neural stem cells encapsulated within the hydrogel system and subjected to an electrical potential in the range of 5–20 V (single pulse every 2 h) for a period of 6 days showed differentiated cells that were positive for neuronal markers, with enhanced neuritic projections [99]. Similarly, the semiconducting property of carbon nanotubes (CNTs) was used in a PEG–multi-walled CNT nanocomposite hydrogel, where electrical stimulation (30 V m−1 for 2 h) and alteration of mechanical properties of the hydrogel showed an enhanced differentiation status of the encapsulated neuron-like cells (PC-12 cells). Curiously, clustering of PC-12 cells and alignment of neurites in the direction of the applied current were also observed in the hydrogel system with no detrimental effects on cellular health [100]. In a similar study, electrical stimulation of DRG neurons cultured in a composite collagen-based hydrogel with embedded single-walled CNTs showed an increase in neuritic differentiation [101]. In parallel, there have been a few studies that have attempted to examine the functionality of conducting hydrogels in animal models. A conducting hydrogel comprising tannic acid, poly-pyrrole and FeCl3 showed moderate benefit when administered locally in a spinal cord injury animal model [102].
In addition to neurons, the effect of electrical stimulation has also been observed in glial cells cultured within hydrogel matrices. Schwann cells cultured within chitosan–poly-pyrrole composite hydrogels in the presence of an electrical field for 4 h were found to express increased mRNA and protein levels of NGF and BDNF. Increased secretion of neurotrophins by electrically stimulated glial cells cultured in a hydrogel matrix raises the possibility of using this strategy for facilitating neuronal survival and regeneration in neural injury conditions [103].
Another important development in this field has been the recent interest in neuromodulatory devices. Hydrogel has been used in fabricating electrodes and circuits that could be implanted in the brain or spinal cord. Alginate hydrogel in combination with poly-pyrrole or poly(3,4-ethylenedioxythiophene) has been used as a neural electrode coating, for reducing electrode impedance and facilitating better neural recording [104,105]. In addition, conducting hydrogels made of poly(3,4-ethylenedioxythioephene)–poly(styrenesulfonate) have also been used to develop ‘stretchable electrode’ systems with enhanced charge storage capacity for use in prolonged nerve stimulation protocols [106].
While the outlook seems promising for conducting hydrogels, challenges related to tissue toxicity in response to the conducting components, the difference in signal transduction due to hydrogel degradation, signal decay and long-term sustenance of these gels within the hostile in vivo environment need to be better addressed.
5. Future directions and challenges
It is interesting to note that, to date, in terms of approval from the US Food and Drug Administration, only nerve conduits for peripheral nerve repair have been moderately successful, while translational products addressing more complex neurological issues are extremely minimal. One of the main reasons for the dearth of tissue-engineered products in the central nervous system is the immense complexity of the nervous system. Even though several hydrogel systems have been developed to facilitate neuritic growth and regeneration, the outcomes, though promising, still appear to be a long way from translation. The central problem lies in the restoration of functional connectivity between various axons, neural circuits and non-neuronal cells, which is notoriously challenging because of the various inhibitory moieties present within the nervous system. Progress in understanding these fundamental principles of neural regenerative biology would immensely assist tissue engineering scientists to develop specialized scaffold systems that can more closely mimic the physiological requirement of neural regeneration. Secondly, from a translational point of view, it may also be important to consider strategies that enhance the pro-regenerational environment within the patient; this may significantly increase the probability of success of tissue engineered neural constructs. Thirdly, looking at the complexities of the nervous system, it appears that a successful construct would incorporate multiple tissue engineering components such as optimal scaffold stiffness, relevant trophic signals with appropriate spatio-temporal release patterns, electro-conductive properties and local immunomodulatory properties. Finally, from a broader perspective it is important to understand the evolutionary pressures that may have resulted in human neural tissue adopting an overall rigid regenerative environment. As we go further in trying to facilitate and replace neural tissue, especially in core-brain areas, concerns regarding the effects of engineered neural tissue on higher mental functions such as memory, personality and behaviour will arise. Nonetheless, these are aspects that need to be considered and addressed as the field progresses.
6. Conclusion
NTE is an emerging field in bioengineering that attempts to craft materials capable of artificially mimicking a biological environment that is conducive for neural tissue growth. The main focus is to develop 3D, viable neural tissue capable of replacing damaged or diseased neural components, promote neural regeneration and restore function. In a broad sense, hydrogel matrices exhibit properties ideally suited for neuronal growth and regeneration, such as the ability to act as a platform capable of providing physical, chemical and electrical cues either separately or in a synergistic manner. However, there are still several questions that need to be answered before hydrogels can be used for mainstream NTE.
Acknowledgements
We are very thankful to Dr Angelo Accardo (A.Accardo@tudelft.nl), Delft University of Technology, The Netherlands, for the 3D hydrogel scaffold image. We also thank Dr Shivanee Shah for her editorial support and valuable suggestions. We thank Ms Amata Sara Boban for the PC-12 cell images.
Data accessibility
This article has no additional data.
Authors' contributions
P.M. and G.R. compiled the references and wrote the review. S.S. initiated the review idea and wrote and edited the work.
Competing interests
We declare we have no competing interests.
Funding
P.M. and G.R. received support from the CSIR fellowship (Department of Science and Technology, Government of India), while S.S. received support from grant no. BT/PR24515/MED/30/1926/2017, Department of Biotechnology, Government of India.
References
- 1.Abbott NJ. 2013. Blood-brain barrier structure and function and the challenges for CNS drug delivery. J. Inherit. Metab. Dis. 36, 437–449. ( 10.1007/s10545-013-9608-0) [DOI] [PubMed] [Google Scholar]
- 2.Silver DJ, Silver J. 2014. Contributions of chondroitin sulfate proteoglycans to neurodevelopment, injury, and cancer. Curr. Opin. Neurobiol. 1, 171–178. ( 10.1016/j.conb.2014.03.016.) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Buchli AD, Schwab ME. 2005. Inhibition of Nogo: a key strategy to increase regeneration, plasticity and functional recovery of the lesioned central nervous system. Ann. Med. 37, 556–567. ( 10.1080/07853890500407520) [DOI] [PubMed] [Google Scholar]
- 4.McKerracher L, Rosen KM. 2015. MAG, myelin and overcoming growth inhibition in the CNS. Front. Mol. Neurosci. 8, 51 ( 10.3389/fnmol.2015.00051) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chan KM, Gordon T, Zochodne DW, Power HA. 2014. Improving peripheral nerve regeneration: from molecular mechanisms to potential therapeutic targets. Exp. Neurol. 261, 826–835. ( 10.1016/j.expneurol.2014.09.006) [DOI] [PubMed] [Google Scholar]
- 6.Madhusudanan P, Reade S, Shankarappa SA. 2017. Neuroglia as targets for drug delivery systems: a review. Nanomed. Nanotechnol. Biol. Med. 13, 667–679. ( 10.1016/j.nano.2016.08.013) [DOI] [PubMed] [Google Scholar]
- 7.Gopalakrishnan A, Shankarappa SA, Rajanikant GK. 2018. Hydrogel scaffolds: towards restitution of ischemic stroke-injured brain. Transl. Stroke Res. 10, 1–18. ( 10.1007/s12975-018-0655-6) [DOI] [PubMed] [Google Scholar]
- 8.Carballo-Molina OA, Velasco I. 2015. Hydrogels as scaffolds and delivery systems to enhance axonal regeneration after injuries. Front. Cell Neurosci. 9, 13 ( 10.3389/fncel.2015.00013) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Accardo A, Blatché MC, Courson R, Loubinoux I, Vieu C, Malaquin L. 2018. Two-photon lithography and microscopy of 3D hydrogel scaffolds for neuronal cell growth. Biomed. Phys. Eng. Express. 4, 27009 ( 10.1088/2057-1976/aaab93) [DOI] [Google Scholar]
- 10.Franze K, et al. 2009. Neurite branch retraction is caused by a threshold-dependent mechanical impact. Biophys. J. 97, 1883–1890. ( 10.1016/j.bpj.2009.07.033) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Accardo A, Blatché MC, Courson R, Loubinoux I, Thibault C, Malaquin L, Vieu C. 2017. Multiphoton direct laser writing and 3D imaging of polymeric freestanding architectures for cell colonization. Small 13, 1700621 ( 10.1002/smll.201700621) [DOI] [PubMed] [Google Scholar]
- 12.Accardo A, Blatché M-C, Courson R, Loubinoux I, Vieu C, Malaquin L. 2018. Direct laser fabrication of free-standing PEGDA-hydrogel scaffolds for neuronal cell growth. Mater. Today. 21, 315–316. ( 10.1016/j.mattod.2018.02.004) [DOI] [Google Scholar]
- 13.Barros CS, Franco SJ, Müller U. 2011. Extracellular matrix: functions in the nervous system. Cold Spring Harb. Perspect. Biol. 3, a005108 ( 10.1101/cshperspect.a005108) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dent EW, Gupton SL, Gertler FB. 2011. The growth cone cytoskeleton in axon outgrowth and guidance. Cold Spring Harb. Perspect. Biol. 3, a001800 ( 10.1038/nmeth1204-190a) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Woerly S, Van Doan D, Evans-Martin F, Paramore CG, Peduzzi JD. 2001. Spinal cord reconstruction using NeuroGelTM implants and functional recovery after chronic injury. J. Neurosci. Res. 66, 1187–1197. ( 10.1002/jnr.1255) [DOI] [PubMed] [Google Scholar]
- 16.Uzel SGM, Platt RJ, Subramanian V, Pearl TM, Rowlands CJ, Chan V, Boyer LA, So PTC, Kamm RD. 2016. Microfluidic device for the formation of optically excitable, three-dimensional, compartmentalized motor units. Sci. Adv. 2, e1501429 ( 10.1126/sciadv.1501429) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dillon GP, Yu X, Sridharan A, Ranieri JP, Bellamkonda RV. 1998. The influence of physical structure and charge on neurite extension in a 3D hydrogel scaffold. J. Biomater. Sci. Polym. Ed. 9, 1049–1069. ( 10.1163/156856298X00325) [DOI] [PubMed] [Google Scholar]
- 18.Kerstein PC, Nichol RH IV, Gomez TM. 2015. Mechanochemical regulation of growth cone motility. Front. Cell Neurosci. 9, 244 ( 10.3389/fncel.2015.00244) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lu Y, et al. 2006. Viscoelastic properties of individual glial cells and neurons in the CNS. Proc. Natl Acad. Sci. USA 103, 17 759–17 764. ( 10.1073/pnas.0606150103) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Banerjee A, Arha M, Choudhary S, Ashton RS, Bhatia SR, Schaffer DV, Kane RS. 2009. The influence of hydrogel modulus on the proliferation and differentiation of encapsulated neural stem cells. Biomaterials 30, 4695–4699. ( 10.1016/j.biomaterials.2009.05.050) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Koser DE, et al. 2016. Mechanosensing is critical for axon growth in the developing brain. Nat. Neurosci. 19, 1592–1598. ( 10.1038/nn.4394) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Farrukh A, Ortega F, Fan W, Marichal N, Paez JI, Berninger B, Campo AD, Salierno MJ. 2017. Bifunctional hydrogels containing the laminin motif IKVAV promote neurogenesis. Stem Cell Rep. 9, 1432–1440. ( 10.1016/j.stemcr.2017.09.002) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tanaka A, Fujii Y, Kasai N, Okajima T, Nakashima H. 2018. Regulation of neuritogenesis in hippocampal neurons using stiffness of extracellular microenvironment. PLoS ONE 13, 1–16. ( 10.1371/journal.pone.0191928) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Johansson F, Carlberg P, Danielsen N, Montelius L, Kanje M. 2006. Axonal outgrowth on nano-imprinted patterns. Biomaterials 27, 1251–1258. ( 10.1016/j.biomaterials.2005.07.047) [DOI] [PubMed] [Google Scholar]
- 25.Georges PC, Miller WJ, Meaney DF, Sawyer ES, Janmey PA. 2006. Matrices with compliance comparable to that of brain tissue select neuronal over glial growth in mixed cortical cultures. Biophys. J. 90, 3012–3018. ( 10.1529/biophysj.105.073114) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jiang FX, Yurke B, Schloss RS, Firestein BL, Langrana NA. 2010. The relationship between fibroblast growth and the dynamic stiffnesses of a DNA crosslinked hydrogel. Biomaterials 31, 1199–1212. ( 10.1016/j.biomaterials.2009.10.050) [DOI] [PubMed] [Google Scholar]
- 27.Musah S, et al. 2014. Substratum-induced differentiation of human pluripotent stem cells reveals the coactivator YAP is a potent regulator of neuronal specification. Proc. Natl Acad. Sci. USA 111, 13 805–13 810. ( 10.1073/pnas.1415330111) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhang Z-N, et al. 2016. Layered hydrogels accelerate iPSC-derived neuronal maturation and reveal migration defects caused by MeCP2 dysfunction. Proc. Natl Acad. Sci. USA 113, 3185–3190. ( 10.1073/pnas.1521255113) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chen WH, Cheng SJ, Tzen JTC, Cheng CM, Lin YW. 2013. Probing relevant molecules in modulating the neurite outgrowth of hippocampal neurons on substrates of different stiffness. PLoS ONE 8, e83394 ( 10.1371/journal.pone.0083394) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pathak MM, et al. 2014. Stretch-activated ion channel Piezo1 directs lineage choice in human neural stem cells. Proc. Natl Acad. Sci. USA 111, 16 148–16 153. ( 10.1073/pnas.1409802111) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gomez TM, Letourneau PC. 2014. Actin dynamics in growth cone motility and navigation. J. Neurochem. 129, 221–234. ( 10.1111/jnc.12506) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mitchison TJ, Cramer LP. 1996. Actin-based cell motility and cell locomotion. Cell 84, 371–379. ( 10.1016/S0092-8674(00)81281-7) [DOI] [PubMed] [Google Scholar]
- 33.Mogilner A, Oster G. 2003. Polymer motors: pushing out the front and pulling up the back. Curr. Biol. 13, R721–R733. ( 10.1016/S0960-9822(03)00652-3) [DOI] [PubMed] [Google Scholar]
- 34.Insall HR, Machesky M. 2009. Actin dynamics at the leading edge: from simple machinery to complex networks. Dev. Cell. 17, 310–322. ( 10.1016/j.devcel.2009.08.012.) [DOI] [PubMed] [Google Scholar]
- 35.Medeiros NA, Burnette DT, Forscher P. 2006. Myosin II functions in actin-bundle turnover in neuronal growth cones. Nat. Cell Biol. 8, 215–226. ( 10.1038/ncb1367) [DOI] [PubMed] [Google Scholar]
- 36.Elkin BS, Azeloglu EU, Costa KD, Morrison B III. 2007. Mechanical heterogeneity of the rat hippocampus measured by atomic force microscope indentation. J. Neurotrauma 24, 812–822. ( 10.1089/neu.2006.0169) [DOI] [PubMed] [Google Scholar]
- 37.Fletcher DA, Mullins RD. 2010. Cell mechanics and the cytoskeleton. Nature 463, 485–492. ( 10.1038/nature08908.Cell) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chierico L, Joseph AS, Lewis AL, Battaglia G. 2014. Live cell imaging of membrane/cytoskeleton interactions and membrane topology. Sci. Rep. 4, 6056 ( 10.1038/srep06056) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Duval K, Grover H, Han L-H, Mou Y, Pegoraro AF, Fredberg J, Chen Z. 2017. Modeling physiological events in 2D vs. 3D cell culture. Physiology 32, 266–277. ( 10.1152/physiol.00036.2016) [DOI] [PMC free article] [PubMed] [Google Scholar] [Research Misconduct Found]
- 40.Ranade SS, Syeda R, Patapoutian A. 2015. Mechanically activated ion channels. Neuron 87, 1162–1179. ( 10.1016/j.neuron.2015.08.032.Mechanically) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Madl CM, Katz LM, Heilshorn SC. 2018. Tuning bulk hydrogel degradation by simultaneous control of proteolytic cleavage kinetics and hydrogel network architecture. ACS Macro Lett. 7, 1302–1307. ( 10.1021/acsmacrolett.8b00664) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Piantino J, Burdick JA, Goldberg D, Langer R, Benowitz LI. 2006. An injectable, biodegradable hydrogel for trophic factor delivery enhances axonal rewiring and improves performance after spinal cord injury. Exp. Neurol. 201, 359–367. ( 10.1016/j.expneurol.2006.04.020) [DOI] [PubMed] [Google Scholar]
- 43.Tobias CA, et al. 2005. Alginate encapsulated BDNF-producing fibroblast grafts permit recovery of function after spinal cord injury in the absence of immune suppression. J. Neurotrauma 22, 138–156. ( 10.1089/neu.2005.22.138) [DOI] [PubMed] [Google Scholar]
- 44.Sakiyama-Elbert SE, Hubbell JA. 2000. Controlled release of nerve growth factor from a heparin-containing fibrin-based cell ingrowth matrix. J. Control. Release 69, 149–158. ( 10.1016/S0168-3659(00)00296-0) [DOI] [PubMed] [Google Scholar]
- 45.Johnson PJ, Parker SR, Sakiyama-Elbert SE. 2009. Controlled release of neurotrophin-3 from fibrin-based tissue engineering scaffolds enhances neural fiber sprouting following subacute spinal cord injury. Biotechnol. Bioeng. 104, 1207–1214. ( 10.1002/bit.22476) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Cooke MJ, Wang Y, Morshead CM, Shoichet MS. 2011. Controlled epi-cortical delivery of epidermal growth factor for the stimulation of endogenous neural stem cell proliferation in stroke-injured brain. Biomaterials 32, 5688–5697. ( 10.1016/j.biomaterials.2011.04.032) [DOI] [PubMed] [Google Scholar]
- 47.Hyatt AJT, Wang D, Kwok JC, Fawcett JW, Martin KR. 2010. Controlled release of chondroitinase ABC from fibrin gel reduces the level of inhibitory glycosaminoglycan chains in lesioned spinal cord. J. Control. Release 147, 24–29. ( 10.1016/j.jconrel.2010.06.026) [DOI] [PubMed] [Google Scholar]
- 48.Wood MD, Borschel GH, Sakiyama-Elbert SE. 2009. Controlled release of glial-derived neurotrophic factor from fibrin matrices containing an affinity-based delivery system. J. Biomed. Mater. Res. Part A 89, 909–918. ( 10.1002/jbm.a.32043) [DOI] [PubMed] [Google Scholar]
- 49.Lee AC, Yu VM, Lowe JB, Brenner MJ, Hunter DA, Mackinnon SE, Sakiyama-Elbert SE. 2003. Controlled release of nerve growth factor enhances sciatic nerve regeneration. Exp. Neurol. 184, 295–303. ( 10.1016/S0014-4886(03)00258-9) [DOI] [PubMed] [Google Scholar]
- 50.Taylor SJ, Rosenzweig ES, McDonald JW, Sakiyama-Elbert SE. 2006. Delivery of neurotrophin-3 from fibrin enhances neuronal fiber sprouting after spinal cord injury. J. Control. Release 113, 226–235. ( 10.1016/j.jconrel.2006.05.005) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Tian WM, Zhang CL, Hou SP, Yu X, Cui FZ, Xu QY, Sheng SL, Cui H, Li HD. 2005. Hyaluronic acid hydrogel as Nogo-66 receptor antibody delivery system for the repairing of injured rat brain: in vitro. J. Control. Release 102, 13–22. ( 10.1016/j.jconrel.2004.09.025) [DOI] [PubMed] [Google Scholar]
- 52.Wei YT, He Y, Xu CL, Wang Y, Liu BF, Wang XM, Sun X-D, Cui F-Z, Xu G. 2010. Hyaluronic acid hydrogel modified with nogo-66 receptor antibody and poly-L-lysine to promote axon regrowth after spinal cord injury. J. Biomed. Mater. Res. Part B Appl. Biomater. 95, 110–117. ( 10.1002/jbm.b.31689) [DOI] [PubMed] [Google Scholar]
- 53.Meyer M, Matsuoka I, Wetmore C, Olson L, Thoenen H. 1992. Enhanced synthesis of brain-derived neurotrophic factor in the lesioned peripheral nerve: different mechanisms are responsible for the regulation of BDNF and NGF mRNA. J. Cell Biol. 119, 45–54. ( 10.1083/jcb.119.1.45) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Funakoshi H, Frisén J, Barbany G, Timmusk T, Zachrisson O, Verge VMK, Persson H. 1993. Differential expression of mRNAs for neurotrophins and their receptors after axotomy of the sciatic nerve. J. Cell Biol. 123, 455–465. ( 10.1083/jcb.123.2.455) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Babensee JE, McIntire LV, Mikos AG. 2000. Growth factor delivery for tissue engineering. Pharm. Res. 17, 497–504. ( 10.1023/A:1007502828372) [DOI] [PubMed] [Google Scholar]
- 56.Thorne RG, Frey WH II. 2001. Delivery of neurotrophic factors to the central nervous system. Clin. Pharmacokinet. 40, 907–946. ( 10.2165/00003088-200140120-00003) [DOI] [PubMed] [Google Scholar]
- 57.Petty BG, Cornblath DR, Adornato BT, Chaudhry V, Flexner C, Wachsman M, Sinicropi D, Burton LE, Peroutka SJ. 1994. The effect of systemically administered recombinant human nerve growth factor in healthy human subjects. Ann. Neurol. 36, 244–246. ( 10.1002/ana.410360221) [DOI] [PubMed] [Google Scholar]
- 58.Burdick JA, Ward M, Liang E, Young MJ, Langer R. 2006. Stimulation of neurite outgrowth by neurotrophins delivered from degradable hydrogels. Biomaterials 27, 452–459. ( 10.1016/j.biomaterials.2005.06.034) [DOI] [PubMed] [Google Scholar]
- 59.Jimenez MC, Tator CH, Shoichet MS. 2005. Injectable intrathecal delivery system for localized administration of EGF and FGF-2 to the injured rat spinal cord. Exp. Neurol. 194, 106–119. ( 10.1016/j.expneurol.2005.01.030) [DOI] [PubMed] [Google Scholar]
- 60.Taylor SJ, McDonald JW, Sakiyama-Elbert SE. 2004. Controlled release of neurotrophin-3 from fibrin gels for spinal cord injury. J. Control. Release 98, 281–294. ( 10.1016/j.jconrel.2004.05.003) [DOI] [PubMed] [Google Scholar]
- 61.Breen A, O'Brien T, Pandit A. 2009. Fibrin as a delivery system for therapeutic drugs and biomolecules. Tissue Eng. Part B. 15, 201–214. ( 10.1089/ten.teb.2008.0527) [DOI] [PubMed] [Google Scholar]
- 62.Sakiyama SE, Schense JC, Hubbell JA. 2018. Incorporation of heparin-binding peptides into fibrin gels enhances neurite extension: an example of designer matrices in tissue engineering. FASEB J. 13, 2214–2224. ( 10.1096/fasebj.13.15.2214) [DOI] [PubMed] [Google Scholar]
- 63.Wells MR, Kraus K, Batter DK, Blunt DG, Weremowitz J, Lynch SE, Antoniades HN, Hansson HA. 1997. Gel matrix vehicles for growth factor application in nerve gap injuries repaired with tubes: a comparison of biomatrix collagen, and methylcellulose. Exp. Neurol. 146, 395–402. ( 10.1006/exnr.1997.6543) [DOI] [PubMed] [Google Scholar]
- 64.Stanwick JC, Baumann MD, Shoichet MS. 2012. Enhanced neurotrophin-3 bioactivity and release from a nanoparticle-loaded composite hydrogel. J. Control. Release 160, 666–675. ( 10.1016/j.jconrel.2012.03.024) [DOI] [PubMed] [Google Scholar]
- 65.Lampe KJ, Kern DS, Mahoney MJ, Bjugstad KB. 2011. The administration of BDNF and GDNF to the brain via PLGA microparticles patterned within a degradable PEG-based hydrogel: protein distribution and the glial response. J. Biomed. Mater. Res. Part A 96 A, 595–607. ( 10.1002/jbm.a.33011) [DOI] [PubMed] [Google Scholar]
- 66.McKeon RJ, Hoke A, Silver J. 1995. Injury induced proteoglycans inhibit the potential for laminin-mediated axon growth on astrocytic scars. Exp. Neurol. 136, 32–43. ( 10.1006/exnr.1995.1081) [DOI] [PubMed] [Google Scholar]
- 67.Apostolova I, Irintchev A, Schachner M. 2006. Tenascin-R restricts posttraumatic remodeling of motoneuron innervation and functional recovery after spinal cord injury in adult mice. J. Neurosci. 26, 7849–7859. ( 10.1523/JNEUROSCI.1526-06.2006) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Bundesen LQ, Scheel TA, Bregman BS, Kromer LF. 2003. Ephrin-B2 and EphB2 regulation of astrocyte-meningeal fibroblast interactions in response to spinal cord lesions in adult rats. J. Neurosci. 23, 7789–7800. ( 10.1523/JNEUROSCI.23-21-07789.2003) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Muir E, et al. 2017. Trafficking and processing of bacterial proteins by mammalian cells: insights from chondroitinase ABC. PLoS ONE 12, e0186759 ( 10.1371/journal.pone.0186759) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Pakulska MM, Vulic K, Shoichet MS. 2013. Affinity-based release of chondroitinase ABC from a modified methylcellulose hydrogel. J. Control. Release 171, 11–16. ( 10.1016/j.jconrel.2013.06.029) [DOI] [PubMed] [Google Scholar]
- 71.Lee H, McKeon RJ, Bellamkonda RV. 2010. Sustained delivery of thermostabilized chABC enhances axonal sprouting and functional recovery after spinal cord injury. Proc. Natl Acad. Sci. USA 107, 3340–3345. ( 10.1073/pnas.0905437106) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Wei YT, et al. 2009. Hyaluronic acid hydrogel modified with nogo-66 receptor antibody and poly(L-lysine) enhancement of adherence and survival of primary hippocampal neurons. J. Bioact. Compat. Polym. 24, 205–219. ( 10.1177/0883911509102266) [DOI] [Google Scholar]
- 73.Ma J, Tian WM, Hou SP, Xu QY, Spector M, Cui FZ. 2007. An experimental test of stroke recovery by implanting a hyaluronic acid hydrogel carrying a Nogo receptor antibody in a rat model. Biomed. Mater. 2, 233–240. ( 10.1088/1748-6041/2/4/005) [DOI] [PubMed] [Google Scholar]
- 74.Fourniols T, Randolph LD, Staub A, Vanvarenberg K, Leprince JG, Préat V, des Rieux A, Danhier F. 2015. Temozolomide-loaded photopolymerizable PEG-DMA-based hydrogel for the treatment of glioblastoma. J. Control. Release 210, 95–104. ( 10.1016/j.jconrel.2015.05.272) [DOI] [PubMed] [Google Scholar]
- 75.Senthilkumar KS, Saravanan KS, Chandra G, Sindhu KM, Jayakrishnan A, Mohanakumar KP. 2007. Unilateral implantation of dopamine-loaded biodegradable hydrogel in the striatum attenuates motor abnormalities in the 6-hydroxydopamine model of hemi-parkinsonism. Behav. Brain Res. 184, 11–18. ( 10.1016/j.bbr.2007.06.025) [DOI] [PubMed] [Google Scholar]
- 76.Rashed ER, Abd El-Rehim HA, El-Ghazaly MA. 2015. Potential efficacy of dopamine loaded-PVP/PAA nanogel in experimental models of Parkinsonism: possible disease modifying activity. J. Biomed. Mater. Res. Part A 103, 1713–1720. ( 10.1002/jbm.a.35312) [DOI] [PubMed] [Google Scholar]
- 77.Caicco MJ, Cooke MJ, Wang Y, Tuladhar A, Morshead CM, Shoichet MS. 2013. A hydrogel composite system for sustained epi-cortical delivery of cyclosporin A to the brain for treatment of stroke. J. Control. Release 166, 197–202. ( 10.1016/j.jconrel.2013.01.002) [DOI] [PubMed] [Google Scholar]
- 78.Adak A, Das G, Barman S, Mohapatra S, Bhunia D, Jana B, Ghosh S. 2017. Biodegradable neuro-compatible peptide hydrogel promotes neurite outgrowth, shows significant neuroprotection, and delivers anti-Alzheimer drug. ACS Appl. Mater. Interfaces 9, 5067–5076. ( 10.1021/acsami.6b12114) [DOI] [PubMed] [Google Scholar]
- 79.Hsiao MH, Larsson M, Larsson A, Evenbratt H, Chen YY, Chen YY, Liu DM. 2012. Design and characterization of a novel amphiphilic chitosan nanocapsule-based thermo-gelling biogel with sustained in vivo release of the hydrophilic anti-epilepsy drug ethosuximide. J. Control. Release 161, 942–948. ( 10.1016/j.jconrel.2012.05.038) [DOI] [PubMed] [Google Scholar]
- 80.Grinsell D, Keating CP. 2014. Peripheral nerve reconstruction after injury: a review of clinical and experimental therapies. BioMed Res. Int. 2014, 698256 ( 10.1155/2014/698256) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Brown KM, Wolfe BB, Wrathall JR. 2005. Rapid functional recovery after spinal cord injury in young rats. J. Neurotrauma 22, 559–574. ( 10.1089/neu.2005.22.559) [DOI] [PubMed] [Google Scholar]
- 82.Nicodemus GD, Bryant SJ. 2008. Cell encapsulation in biodegradable hydrogels for tissue engineering applications. Tissue Eng. Part B Rev. 14, 149–165. ( 10.1089/ten.teb.2007.0332) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Li H, Zheng J, Wang H, Becker ML, Leipzig ND. 2018. Neural stem cell encapsulation and differentiation in strain promoted crosslinked polyethylene glycol-based hydrogels. J. Biomater. Appl. 32, 1222–1230. ( 10.1177/0885328218755711) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Aurand ER, Wagner JL, Shandas R, Bjugstad KB. 2014. Hydrogel formulation determines cell fate of fetal and adult neural progenitor cells. Stem Cell Res. 12, 11–23. ( 10.1016/j.scr.2013.09.013) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Das S, et al. 2016. Implantable amyloid hydrogels for promoting stem cell differentiation to neurons. NPG Asia Mater. 8, e304 ( 10.1038/am.2016.116) [DOI] [Google Scholar]
- 86.Sun L, Zheng C, Webster TJ. 2017. Self-assembled peptide nanomaterials for biomedical applications: promises and pitfalls. Int. J. Nanomed. 12, 73–86. ( 10.2147/IJN.S117501) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Nisbet DR, Williams RJ. 2012. Self-assembled peptides: characterisation and in vivo response. Biointerphases 7, 1–14. ( 10.1007/s13758-011-0002-x) [DOI] [PubMed] [Google Scholar]
- 88.Holmes TC, de Lacalle S, Su X, Liu G, Rich A, Zhang S. 2000. Extensive neurite outgrowth and active synapse formation on self-assembling peptide scaffolds. Proc. Natl Acad. Sci. USA 97, 6728–6733. ( 10.1073/pnas.97.12.6728) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Gelain F, Bottai D, Vescovi A, Zhang S. 2006. Designer self-assembling peptide nanofiber scaffolds for adult mouse neural stem cell 3-dimensional cultures. PLoS ONE 1, e0000119 ( 10.1371/journal.pone.0000119) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Cigognini D, Satta A, Colleoni B, Silva D, Donegà M, Antonini S, Gelain F. 2011. Evaluation of early and late effects into the acute spinal cord injury of an injectable functionalized self-assembling scaffold. PLoS ONE 6, e0019782 ( 10.1371/journal.pone.0019782) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Rodriguez AL, Wang TY, Bruggeman KF, Horgan CC, Li R, Williams RJ, Parish CL, Nisbet DR. 2014. In vivo assessment of grafted cortical neural progenitor cells and host response to functionalized self-assembling peptide hydrogels and the implications for tissue repair. J. Biomed. Mater. Res. B Appl. Biomater. 2, 7771–7778. ( 10.1039/c4tb01391c) [DOI] [PubMed] [Google Scholar]
- 92.Goganau I, Sandner B, Weidner N, Fouad K, Blesch A. 2018. Depolarization and electrical stimulation enhance in vitro and in vivo sensory axon growth after spinal cord injury. Exp. Neurol. 300, 247–258. ( 10.1016/j.expneurol.2017.11.011) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.André S, Boukhaddaoui H, Campo B, Al-Jumaily M, Mayeux V, Greuet D, Valmier J, Scamps F. 2003. Axotomy-induced expression of calcium-activated chloride current in subpopulations of mouse dorsal root ganglion neurons. J. Neurophysiol. 90, 3764–3773. ( 10.1152/jn.00449.2003) [DOI] [PubMed] [Google Scholar]
- 94.Nguyen HT, Wei C, Chow JK, Nguy L, Nguyen HK, Schmidt CE. 2013. Electric field stimulation through a substrate influences Schwann cell and extracellular matrix structure. J. Neural Eng. 10, 046011 ( 10.1088/1741-2560/10/4/046011) [DOI] [PubMed] [Google Scholar]
- 95.Gordon T. 2009. The role of neurotrophic factors in nerve regeneration. Neurosurg. Focus. 26, E3 ( 10.3171/FOC.2009.26.2.E3) [DOI] [PubMed] [Google Scholar]
- 96.Arslantunali D, Dursun T, Yucel D, Hasirci N, Hasirci V. 2014. Peripheral nerve conduits: technology update. Med. Devices Evid. Res. 7, 405–424. ( 10.2147/MDER.S59124) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Peckham PH, Ackermann DM, Moss CW. 2013. The role of biomaterials in stimulating bioelectrodes. In Biomaterials science: an introduction to materials, 3rd edn (eds Ratner BD, Hoffman AS, Schoen FJ, Lemons JE), pp. 981–993. Oxford, UK: Elsevier. [Google Scholar]
- 98.Guiseppi-Elie A. 2010. Electroconductive hydrogels: synthesis, characterization and biomedical applications. Biomaterials 31, 2701–2716. ( 10.1016/j.biomaterials.2009.12.052) [DOI] [PubMed] [Google Scholar]
- 99.Lee JM, Moon JY, Kim TH, Lee SW, Ahrberg CD, Chung BG. 2018. Conductive hydrogel/nanowire micropattern-based sensor for neural stem cell differentiation. Sens. Actuators B Chem. 258, 1042–1050. ( 10.1016/j.snb.2017.11.151) [DOI] [Google Scholar]
- 100.Imaninezhad M, Pemberton K, Xu F, Kalinowski K, Bera R, Zustiak SP. 2018. Directed and enhanced neurite outgrowth following exogenous electrical stimulation on carbon nanotube-hydrogel composites. J. Neural Eng. 29, 7 ( 10.1088/1741-2552/aad65b) [DOI] [PubMed] [Google Scholar]
- 101.Koppes AN, et al. 2016. Robust neurite extension following exogenous electrical stimulation within single walled carbon nanotube-composite hydrogels. Acta Biomater. 39, 34–43. ( 10.1016/j.actbio.2016.05.014) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Zhou L, et al. 2018. Soft conducting polymer hydrogels cross-linked and doped by tannic acid for spinal cord injury repair. ACS Nano. 12, 10 957–10 967. ( 10.1021/acsnano.8b04609) [DOI] [PubMed] [Google Scholar]
- 103.Huang J, Hu X, Lu L, Ye Z, Zhang Q, Luo Z. 2009. Electrical regulation of Schwann cells using conductive polypyrrole/chitosan polymers. J. Biomed. Mater. Res. Part A 34, 1799–1809. ( 10.1016/j.biomaterials.2012.11.042) [DOI] [PubMed] [Google Scholar]
- 104.Kim DH, Abidian M, Martin DC. 2004. Conducting polymers grown in hydrogel scaffolds coated on neural prosthetic devices. J. Biomed. Mater. Res. Part A 71, 577–585. ( 10.1002/jbm.a.30124) [DOI] [PubMed] [Google Scholar]
- 105.Kim DH, Wiler JA, Anderson DJ, Kipke DR, Martin DC. 2010. Conducting polymers on hydrogel-coated neural electrode provide sensitive neural recordings in auditory cortex. Acta Biomater. 6, 57–62. ( 10.1016/j.actbio.2009.07.034) [DOI] [PubMed] [Google Scholar]
- 106.Liu Y, et al. 2019. Soft and elastic hydrogel-based microelectronics for localized low-voltage neuromodulation. Nat. Biomed. Eng. 3, 58–68. ( 10.1038/s41551-018-0335-6) [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
This article has no additional data.