Abstract
The case of a patient with metastatic renal cell carcinoma who exhibited the abscopal effect following treatment by anti-programmed death-1 (PD-1) antibody is presented. A 40-year-old woman was diagnosed with an 8.2-cm renal tumor without distant metastases, and radical nephrectomy was subsequently performed. Pathological examination revealed a clear cell renal cell carcinoma. At 3 months after surgery, the patient developed one lung metastasis. Following treatment with interferon and three types of tyrosine kinase inhibitors, anti-PD1 antibody (nivolumab) was started. During the treatment, para-aortic/supraclavicular lymph nodes and several lung lesions remained, although other lesions decreased markedly. The patient was subsequently treated by palliative radiotherapy to the para-aortic and supraclavicular lymph nodes for pain control. After the radiotherapy, the lung lesions previously refractory to nivolumab started to decrease, probably due to an abscopal effect. Additionally, the laboratory data and Karnofsky Performance Status improved. Histological re-examination of the primary lesion revealed heterogeneity of the immunological microenvironment, which may be associated with the heterogeneity of treatment sensitivity.
Keywords: renal cell carcinoma, immune checkpoint inhibitor, anti-PD1 antibody, abscopal effect, radiation therapy, cytotoxic T lymphocytes, cytotoxic T lymphocyte
Introduction
The introduction of the human programmed death-1 (PD-1) immune checkpoint inhibitor Nivolumab has changed the therapeutic strategy for metastatic renal cell carcinoma (mRCC). Nivolumab has shown to prolong the overall survival of mRCC patients in second line after vascular endothelial growth factor receptor tyrosine kinase inhibitors (VEGFR TKIs) failure (1). Nevertheless, the efficacy of subsequent therapies that are considered after VEGFR TKIs and immunotherapy failure is still unclear and additional therapeutic strategy is limited. The abscopal effect is a rare phenomenon that was first described over half a century ago (2), in which tumor regression occurs outside the irradiated sites through activation of the immune system. Recently, the efficacy of cancer immunotherapy combined with radiotherapy (RT) has been suggested (3). We experienced a case of a patient with mRCC who demonstrated the abscopal effect during nivolumab treatment after palliative radiotherapy. This patient had a unique treatment course after the abscopal effect. Furthermore, pathological re-examination of the primary specimen showed unique pathological findings. The unique treatment course with Nivolumab combined with RT and the appearance of abscopal effect might be related to the unique pathological findings.
Case report
A 40-year-old woman who had never been diagnosed with any other disease and malignancy presented with lumbar pain. Computed tomography (CT) showed a left renal tumor with a maximum diameter of 8.2 cm, without distant metastases. She underwent radical nephrectomy, and pathological examination showed a clear cell renal cell carcinoma (ccRCC), stage pT2aN0M0, Fuhrman grade 2. Three months after surgery, she developed two lung metastases. During the following two years, she received various systemic therapies, including interferon-α (3 months), axitinib (9 months), everolimus (3 months), and pazopanib (9 months). However, their effects were transient, and follow-up CT showed progression of lung metastases with pleural effusion and new lesions (right supraclavicular and para-aortic lymph node swellings).
Because nivolumab received government approval in Japan, it was started at 3 mg/kg intravenously every 2 weeks. After 26 cycles, most of the lung nodules had shrunk, and the pleural effusion had disappeared completely (Fig. 1). However, several lung nodules and the right supraclavicular and para-aortic lymph nodes were still growing (Fig. 2). The patient also complained of lumbar pain, probably due to nerve compression by metastatic nodes, and her Karnofsky Performance Status (KPS) deteriorated to 50. Thereafter, palliative radiotherapy (RT) was performed to the right supraclavicular and para-aortic lymph nodes (30 Gy/10 Fr and 40 Gy/20 Fr, respectively). After the RT, nivolumab was resumed. Follow-up CT showed the decrease in size of both irradiated lesions (Fig. 2), and, interestingly, the nivolumab-resistant lung nodules also appeared to be decreasing after RT (Fig. 3), probably due to the abscopal effect. The patient's laboratory data also normalized, as shown in Fig. 4, and her KPS improved from 50 to 100. Her laboratory data and KPS have remained excellent and she has been received 33 cycles of niv after RT (total 64 cycles from induction).
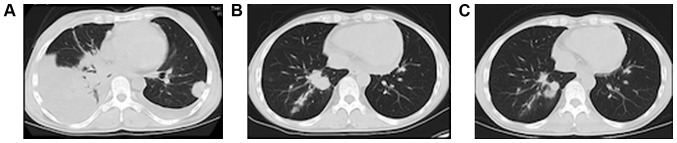
Figure 1.
Images of the lung nodules after nivolumab induction. (A) Pretreatment computed tomography reveals increases in the number and size of nodules in the lung lobes with pleural effusion. After (B) 6 and (C) 26 cycles, most lung nodules had shrunk, and the pleural effusion had completely resolved.
Figure 2.
Images of lymph nodes after nivolmab induction and radiotherapy. (A and B) Right supraclavicular lymph node (yellow arrow) was enlarged after 26 cycles of nivolumab. (C) After 4 months following radiotherapy, an objective response was observed in the right supraclavicular lymph node(yellow arrow). (D and E) Para-aortic lymph node (yellow arrow) was enlarged after 26 cycles of nivolumab. (F) After 8 months following radiotherapy, an objective response was observed in the para-aortic lymph node (yellow arrow).
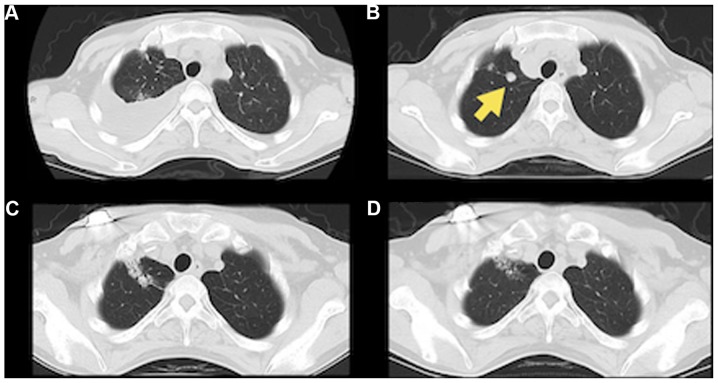
Figure 3.
Images of abscopal effect on the nivolumab-resistant lung nodules. (A and B) One nodule (yellow arrow) in the right lung had grown after nivolumab induction. After (C) 1 month (C and D) 6 months following radiotherapy to the lymph nodes, a treatment response was seen in the lung lobe.
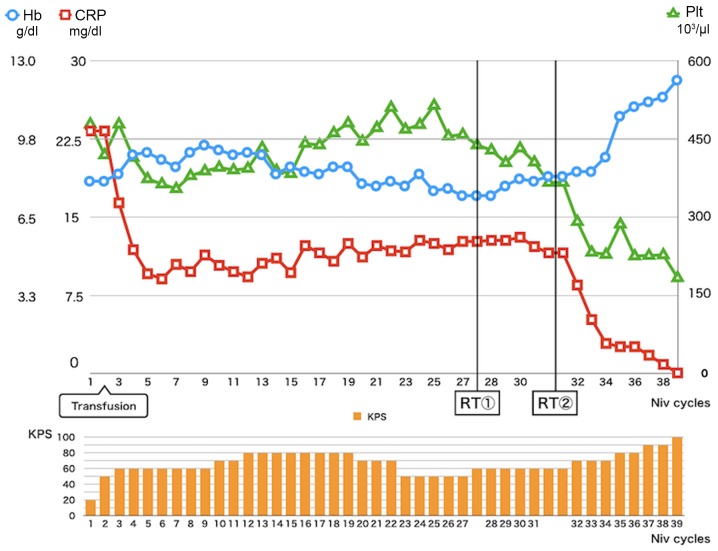
Figure 4.
Transition of laboratory data and KPS during the Following the Niv induction, CRP improved partially. At 3 months after the RT, CRP, Hb and Plt values were normalized. KPS also improved from 50 to 100. Niv, nivolumab; CRP, C-reactive protein; RT, radiotherapy; Hb, hemoglobin; Plt, platelet; KPS, Karnofsky Performance Status.
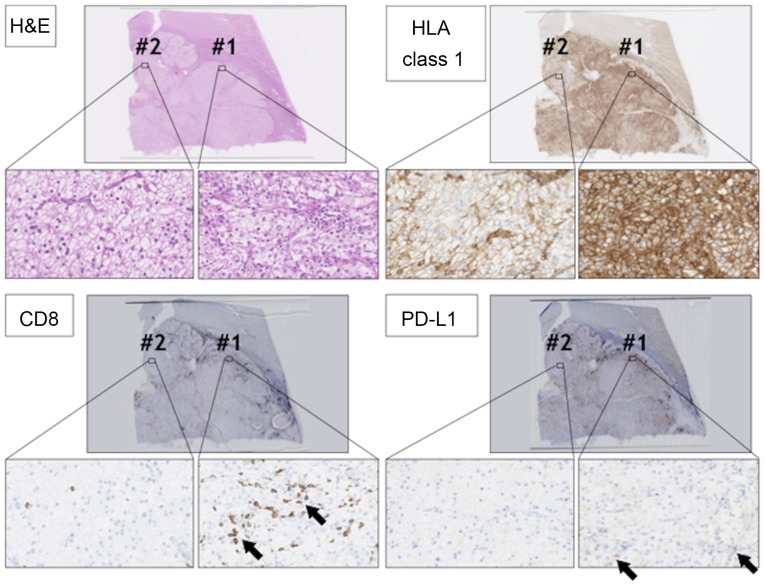
Repeat pathological examination of the tumor specimen was performed, including immunohistochemical (IHC) staining. IHC staining using anti-human leukocyte antigen (HLA) class 1 (clone: EMR8-5) showed heterogeneity of the tumor with two staining patterns (components #1 and #2) in the RCC lesion (Fig. 5). In component #1, RCC cells showed strong membrane-positive staining for anti-HLA class 1 antibody. On the other hand, in component #2, RCC cells showed relatively weak HLA class 1 staining. Infiltration of CD8-positive cytotoxic T lymphocytes (CTLs) also showed heterogeneity. There were many CTLs in component #1, whereas CD8-positive CTLs were few in component #2, suggesting that component #1 RCC was an inflamed tumor, and component #2 RCC was an immune desert tumor (3). Programmed death-ligand 1 (PD-L1) staining using anti-PD-L1 antibody (clone E1L3N) showed that PD-L1 was expressed in component #1 RCC cells, but not in component #2 RCC cells.
Figure 5.
Histological analysis of the primary RCC lesion. RCC sections were stained with H&E, anti-CD8 antibody (arrows), anti-HLA class 1 antibody, and anti-PD-L1 antibody (arrows). Magnification, ×2 or ×400. There were two staining patterns in RCC lesions #1 and #2. RCC, renal cell carcinoma; H&E, hematoxylin and eosin; HLA, human leukocyte antigen; PD-L1, programmed cell death ligand 1.
Discussion
Several prognostic markers have been reported in RCCs (4,5), and high C-reactive protein (CRP), low hemoglobin, and thrombocythemia were also reported to be related to poorer prognosis in RCCs (6,7). In the present case, the patient showed a relatively good response to nivolumab, and CRP improved partially, whereas anemia and thrombocythemia did not improve with PD1 blockade, even after most of the metastatic RCC lesions had shrunk. Interestingly, after the RT to the metastatic nodes, the nivolumab-resistant lung lesion shrank, probably due to the abscopal effect, and the patient's laboratory data also normalized. High CRP has also been shown to be related to poorer prognosis in melanoma patients treated by cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) blockade (8). CRP is a product of IL-6 stimulation, and so high-CRP level indicates type 2 helper dominancy in the patient. Surprisingly, laboratory data normalization including CRP indicates the improvement of systematic type 2 helper dominancy that might help to induce new cytotoxic T lymphocytes.
Histological re-examination showed the heterogeneity of the primary RCC lesions in this case, with two components, #1 and #2. In component #1, ccRCC cells strongly expressed HLA class 1 molecule, and many infiltrating CD8+ CTLs were observed in the intra-tumoral region even before anti-PD1 treatment, indicating that the ccRCC cells in component #1 were immunogenic.
The ccRCC cells in component #1 expressed PD-L1, indicating that inhibitory receptor ligand PD-L1 was essential for immunological escape for component #1 ccRCC cells. On the other hand, component #2 ccRCC cells expressed lower levels of HLA class 1 and did not express PD-L1. CD8+ CTL infiltration was small in component #2 of the ccRCC, indicating that ccRCC cells in component #2 might be less immunogenic, and PD-L1 expression was not necessary for immunological escape. After the abscopal effect, almost all lesions shrank, suggesting that PD1 blockade therapy also became effective in the less immunogenic component, component #2. At this moment, we do not know the mechanisms for how RT transformed ccRCC cells to become sensitive to PD1 blockade; however, RT is known to induce anti-tumor immunity by releasing tumor-associated antigens, over expression of MHC class 1, and release danger signal molecules including High mobility group box 1 protein (HMGB1) and adenosine triphosphate (ATP) (9,10). Furthermore, normalization of CRP highly suggest that type 2 helper dominancy was improved to type 1 helper dominancy in this case as described above. These improvements of immunological environment might provoke a different anti-component #2 immunological reaction. This case report has some limitations. we could not perform genetical analysis such as Whole exome sequence in this case, because only limited volume of formalin-fixed paraffin-embedded specimen samples are available in this case.
In summary, this case had a unique treatment course after an abscopal effect by RT. PD1 blockade combined with RT might be an option for RCC patients in whom PD1 blockade monotherapy is ineffective.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
Authors' contributions
KH, TAo, NT, MM and KM designed the study, collected and analysed the clinical data. KH and YH wrote the manuscript. YH, HM and TT collected and analysed the pathological data. TAb and NS contributed to the conception and design of the study, and revised the manuscript.
Ethics approval and consent to participate
Ethical approval was granted by The Kushiro City General Hospital (approval no. 2019-2).
Patient consent for publication
Informed consent was obtained from the patient, including for the publication of this report.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Mole RH. Whole body irradiation; radiobiology or medicine? Br J Radiol. 1953;26:234–241. doi: 10.1259/0007-1285-26-305-234. [DOI] [PubMed] [Google Scholar]
- 2.Formenti SC, Demaria S. Combining radiotherapy and cancer immunotherapy: A paradigm shift. J Natl Cancer Inst. 2013;105:256–265. doi: 10.1093/jnci/djs629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Galluzzi L, Chan TA, Kroemer G, Wolchok JD, López-Soto A. The hallmarks of successful anticancer immunotherapy. Sci Transl Med. 2018;10:eaat7807. doi: 10.1126/scitranslmed.aat7807. [DOI] [PubMed] [Google Scholar]
- 4.Sun M, Shariat SF, Cheng C, Ficarra V, Murai M, Oudard S, Pantuck AJ, Zigeuner R, Karakiewicz PI. Prognostic factors and predictive models in renal cell carcinoma: A contemporary review. Eur Urol. 2011;60:644–661. doi: 10.1016/j.eururo.2011.06.041. [DOI] [PubMed] [Google Scholar]
- 5.Shinohara N, Abe T. Prognostic factors and risk classifications for patients with metastatic renal cell carcinoma. Int J Urol. 2015;22:888–897. doi: 10.1111/iju.12858. [DOI] [PubMed] [Google Scholar]
- 6.Xia L, Hu G, Guzzo TJ. Prognostic significance of preoperative anemia in patients undergoing surgery for renal cell carcinoma: A meta-analysis. Anticancer Res. 2017;37:3175–3181. doi: 10.21873/anticanres.11677. [DOI] [PubMed] [Google Scholar]
- 7.Bensalah K, Leray E, Fergelot P, Rioux-Leclercq N, Tostain J, Guillé F, Patard JJ. Prognostic value of thrombocytosis in renal cell carcinoma. J Urol. 2006;175:859–863. doi: 10.1016/S0022-5347(05)00526-4. [DOI] [PubMed] [Google Scholar]
- 8.Wilgenhof S, Du Four S, Vandenbroucke F, Everaert H, Salmon I, Liénard D, Marmol VD, Neyns B. Single-center experience with ipilimumab in an expanded access program for patients with pretreated advanced melanoma. J Immunother. 2013;36:215–222. doi: 10.1097/CJI.0b013e31828eed39. [DOI] [PubMed] [Google Scholar]
- 9.Lugade AA, Moran JP, Gerber SA, Rose RC, Frelinger JG, Lord EM. Local radiation therapy of B16 melanoma tumors increases the generation of tumor antigen-specific effector cells that traffic to the tumor. J Immunol. 2005;174:7516–7523. doi: 10.4049/jimmunol.174.12.7516. [DOI] [PubMed] [Google Scholar]
- 10.Gameiro SR, Jammeh ML, Wattenberg MM, Tsang KY, Ferrone S, Hodge JW. Radiation-induced immunogenic modulation of tumor enhances antigen processing and calreticulin exposure, resulting in enhanced T-cell killing. Oncotarget. 2014;5:403–416. doi: 10.18632/oncotarget.1719. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.