Abstract
Purpose
Pseudomonas aeruginosa causes complicated and/or nosocomial UTI. These infections are usually associated with severe and multi-drug resistant P. aeruginosa isolates. As there is no study about the activity of novel antibiotics ceftazidime-avibactam (CZA) and ceftolozane-tazobactam (C/T) against P. aeruginosa isolates in Iran, we aimed to evaluate for the first time the efficacy of these agents against P. aeruginosa isolated from patients with UTI in Iran. Then, the genetic diversity of the resistant isolates was assayed.
Methods
In this study, a total of 200 P. aeruginosa isolates were collected from patients with UTI in Tehran, Iran. Disk diffusion and Minimum Inhibitory Concentration (MIC) methods were applied to determine the resistance of the isolates to CZA, C/T, and the other antibiotics. Extended-spectrum β-lactamases (ESBLs) and Metallo Beta Lactamase (MBL) production were assayed by Combination disk diffusion test (CDDT). Polymerase chain reaction (PCR) was carried out to detect the resistance genes, including beta-lactamases and carbapenemases genes. Finally, genomic analysis of the isolates was performed using the Pulse field gel electrophoresis (PFGE).
Results
Among the isolates, 16 (8%) were resistant to CZA and C/T that MIC confirmed it. The resistant isolates showed high resistance to the other classes of antibiotics. Among the resistant isolates, 31.2% and 75% were ESBL and MBL producers, respectively. The prevalence of blaOXA10, blaVIM, blaOXA48, blaOXA2, and blaCTX-M was 100%, 50%, 31.2%, 25%, and 12.5%. Furthermore, two isolates (12.5%) harbored blaPER and blaNDM genes. The resistant isolates were grouped into 14 distinct pulsotypes and two shared pulsotypes were found.
Conclusion
Ceftazidime-avibactam and ceftolozane-tazobactam showed high activity against the P. aeruginosa isolated from patients with UTI in Iran. The low rate of resistance to the antibiotics is also alarming and should be considered to avoid further spreading of the antibiotic resistance among the P. aeruginosa and the other bacteria.
Keywords: P. aeruginosa, urinary tract infection, ceftazidime-avibactam, ceftolozane-tazobactam, antibiotic resistance, PFGE
Introduction
Urinary tract infection (UTI) is one of the most common bacterial infections affecting women and men throughout their life periods.1 The most common agents responsible for UTIs are members of Enterobacteriaceae family such as Escherichia coli, Proteus mirabilis, and Klebsiella pneumoniae and Pseudomonas aeruginosa.2 P. aeruginosa is originally an environmental bacterium considered as a potential opportunistic pathogen which usually infects the hospitalized and immune-compromised patients. P. aeruginosa strains are regarded as a common cause of nosocomial UTIs, especially among patients hospitalized in the intensive care unit (ICU). These infections are associated with different forms of severe UTIs, including prostatitis, urolithiasis, and UTIs associated with urinary instruments such as indwelling catheters.2
Increasing antibiotic resistance and limits in the treatment options is a growing challenge for infections caused by P. aeruginosa strains in hospitals. Also, the infections caused by multi-drug resistant (MDR) P. aeruginosa causes significant mortality and morbidity, increase the hospitalization and healthcare costs compared with the infections caused by susceptible strains. These show the need for further researches on antibiotic resistance of P. aeruginosa strains and introduction of new antimicrobial compounds.3
Ceftazidime-avibactam (CZA) combines a third-generation cephalosporin with a novel broad-spectrum class of non-β-lactam beta-lactamase inhibitor that binds to the active site of β-lactamases.4 Avibactam has inhibitory effects against a wide range of β-lactamase enzymes from different β-lactamases classes, including class A (extended-spectrum β-lactamases (ESBLs) and KPC), class C (CMY, ACT, and FOX), and some numbers of class D such as OXA-48 β-lactamase. Furthermore, avibactam has shown a more inhibitory effect in comparison to the common β-lactamase inhibitors such as clavulanic acid, sulbactam, and tazobactam.5
Ceftolozane-tazobactam (C/T) is a mixture of a new cephalosporin merged with a β-lactam β-lactamase inhibitor called tazobactam. Tazobactam irreversibly binds to the active site of β-lactamases and protects ceftolozane from destruction by the majority of β-lactamases such as ESBLs enzymes; however, it does not improve its activity against pathogens such as P. aeruginosa.6 Unlike avibactam, the studies demonstrated that tazobactam has no ability in inhibiting AmpC β-lactamases and several carbapenemases such as OXA-48, KPC-2, and KPC-3.7
Recently, ceftazidime-avibactam and ceftolozane-tazobactam were approved by the US Food and Drug Administration (FDA) and by the European Medicine Agency (EMA) to treat infections such as complicated urinary tract infections (CUTIs), complicated intra-abdominal infections (CIAIs), and complicated hospital-acquired pneumonia (HAP) in patients with limited treatment options.8
The present study aimed, for the first time in Iran, to evaluate the efficacy of ceftazidime-avibactam and ceftolozane-tazobactam against P. aeruginosa isolates from patients with UTIs in two Iranian hospitals. It also assayed the molecular mechanisms and genetic diversity of the isolates with the capability of resistance to ceftazidime-avibactam and ceftolozane-tazobactam.
Materials and Methods
Isolation and Identification of P. aeruginosa Isolates
In this study, during the period from March to October 2018, 200 P. aeruginosa isolates were collected from outpatients (n=100) and inpatients (n=100) in two general hospitals (A and B) in Tehran, Iran. These isolates were collected from urine specimens of patients with UTI symptoms. After transporting the samples to the Department of Molecular Biology, Pasteur Institute of Iran, the isolates were again inoculated into MacConkey agar medium and pure colonies were identified according to the routine identification tests for P. aeruginosa including Gram staining and biochemical tests such as oxidase, catalase, oxidative-fermentative test, growth on media TSI, SIM, cetrimide agar, and growth at 42°C. Then, confirmed isolates were preserved in Trypticase soy broth media (TSB) containing 20% glycerol and stored at −70°C until further use.9 No ethical approval was obtained for collecting the clinical isolates since they were collected during routine bacteriological analysis in the involved hospitals.
Susceptibility Tests Including Disc Diffusion and MIC
At first, the susceptibility pattern of P. aeruginosa isolates to ceftazidime-avibactam (30µg/20µg) and ceftolozane-tazobactam (30µg/10µg) (MAST Co. UK) was assayed using disk diffusion method (Kirby-Bauer). Then, the susceptibility patterns of the ceftazidime-avibactam and ceftolozane-tazobactam resistant isolates to the other antibiotics, including imipenem (10μg), ertapenem (10μg), cefotaxime (30μg), cefoxitin (30µg), ceftazidime (30µg), cefepime (30µg), amikacin (30μg), gentamicin (10μg), ciprofloxacin (5μg), nitrofurantoin (300µg), levofloxacin (10μg), aztreonam (10μg), and fosfomycin (200µg) (MAST Co, UK), were evaluated.10 For performing disk diffusion method, isolates were cultured on Muller-Hinton agar plates (Merck, Germany) and the other procedures were performed according to the CLSI recommendations.11 P. aeruginosa ATCC 27853 was used as a control strain.
After disk diffusion, Minimum Inhibitory Concentration (MIC) of the resistant isolates to both CZA and C/T in disk diffusion was measured. MIC was assessed using the microdilution broth method, according to the recommendations of CLSI.11 Then, MIC of selected antibiotics including imipenem, ceftazidime, amikacin, aztreonam, and ciprofloxacin was evaluated against the resistant isolates to both CZA and C/T. In this assay, E. coli ATCC 25922 and P. aeruginosa ATCC 27853 were used as control strains.
Phenotypic Detection of ESBL Producer Isolates
All of the P. aeruginosa isolates that were resistant to ceftazidime-avibactam and ceftolozane-tazobactam by disc diffusion were selected for confirmation of ESBL production by Combination disk diffusion test (CDDT). CDDT as the confirmatory test of disc diffusion was performed using ceftazidime (CAZ) or cefotaxime (CEF) discs alone and discs with these agents plus clavulanate (CAZ/CL and CEF/CL, respectively). An isolate was defined as ESBL-positive when a difference of at least 5 mm in the inhibition zones of CAZ/CL versus CAZ or CEF/CL versus CEF was obtained. E. coli ATCC 25922 and Klebsiella pneumoniae ATCC 700603 were used as non-ESBL and ESBL-producing strains, respectively.12
Metallo Beta-Lactamase (MBL) Phenotypic Test
Combination disk diffusion test (CDDT) was also used for phenotypic detection of Metallo Beta-Lactamase (MBL) producing isolates. In brief, 5 μL of 0.5 M EDTA (935µg) plus 10μg of imipenem were placed on the Muller Hinton agar plates which were inoculated with P. aeruginosa. After 18–24 h of incubation at 37°C, an isolate was considered as an MBL producer, if the growth inhibition zone was increased 7 mm or more in comparison with imipenem disk alone.13
Extraction of DNA and Molecular Analysis
DNA was extracted from the cultured P. aeruginosa isolates using a DNA extraction kit (Roche, Germany) with regard to the kit protocols. Thereafter, the quality and quantity of the extracted products were checked by electrophoresis on agarose gel (Sigma, USA) and spectrophotometry (OD260/280 nm) to confirm the quality of products for the next stage.
Polymerase chain reaction (PCR) was carried out to detect the presence of resistance genes including beta-lactamases (blaPER, blaCTX-M, blaGES, blaOXA2, and blaOXA10) and carbapenemases genes (blaIMP, blaVIM, blaNDM, blaSPM, and blaOXA48) among ceftazidime-avibactam and ceftolozane-tazobactam resistant isolates on a thermal cycler (Eppendorf, Germany). The primer sequences used in this study and the PCR conditions are detailed in Table 1. Depend on the molecular size of PCR products, separation of the products was carried out on 1% to 2% agarose gel stained with ethidium bromide and visualized under UV light in a UV gel documentation box.
Table 1.
The List of Primer Sequences Used for PCR Conditions
| Target | Primer Sequence (5′–3′) | Size (bp) | Annealing Temp (°C) | References |
|---|---|---|---|---|
| blaVIM | GATGGTGTTTGGTCGCATA CGAATGCGCAGCACCAG |
390 | 61° | [14] |
| blaIMP | GGAATAGAGTGGCTTAAYTCTC GGTTTAAYAAAACAACCACC |
232 | 55° | [14] |
| blaNDM | GGTTTGGCGATCTGGTTTTC CGGAATGGCTCATCACGATC |
621 | 56° | [14] |
| blaSPM | CCTACAATCTAACGGCGACC TCGCCGTGTCCAGGTATAAC |
674 | 60° | [15] |
| blaOXA48 | GCGTGGTTAAGGATGAACAC CATCAAGTTCAACCCAACCG |
438 | 55° | [14 |
| blaOXA2 | AAGAAACGCTACTCGCCTGC CCACTCAACCCATCCTACCC |
478 | 56° | [16] |
| blaOXA10 | CCGAAGCCGTCAATGGTG CCAACCCACCATGCGACA |
571 | 61° | [12] |
| blaCTX-M | CGCTTTGCGATGTGCAG ACCGCGATATCGTTGGT |
550 | 51° | [17] |
| blaPER | ATGAATGGTCATTATAAAAGC AATTTGGGCTTAGGGCAGAA |
580 | 43° | [18] |
| blaGES | ATGCGCTTCATTCACGCAC CTATTTGTCCGTGCTCAGG |
846 | 56° | [19] |
PFGE Analysis
Genomic analysis of resistant isolates to CZA and C/T was performed using the Pulse field gel electrophoresis (PFGE). Briefly, P. aeruginosa isolates were grown in Luria Bertani (LB) broth overnight at 37°C to reach the late exponential phase. Bacterial suspensions were adjusted to an optical density of 0.8–1 at 620 nm and mixed with low melting agarose (Sigma, USA) to make the agarose plugs. Plugs were then incubated in a buffer containing 0.5 M EDTA and 1% N-laurylsarcosine and incubated at 37°C. Agarose plugs were washed and digested with 10 U of SpeI restriction enzyme (Fermentas, Lithuania). Digested plugs were loaded on the agarose gels and electrophoresis was carried out in a CHEF-DR2 cell system (BioRad, USA) using a linear increase of pulse intervals for 20 h. Finally, the PFGE gels were stained with ethidium bromide and visualized using a gel documentation system.20
Statistical Analysis
Statistical data analysis was performed using the SPSS software version 19.0 for Windows (IBM, Chicago, USA). Chi-square and two-tailed Fisher ʼs exact were among the used tests to assess and compare the relationships between the results in the isolates. P-value <0.05 was considered as significant for all statistical accounts.
Results
Bacterial Strains and Antibiotic Susceptibility results
In this study, a total of 200 P. aeruginosa clinical isolates were collected from the urine specimens of patients with UTI. Based on the susceptibility test results, 16 isolates (8%) were resistant to both ceftazidime-avibactam and ceftolozane-tazobactam. Among the resistant isolates, 11 (68.7%) were isolated from men and 5 (31.3%) from women with the age range 10 to 80 years and mean age of 31.63 ± 15.39 years old. Furthermore, of the 16 resistant isolates, 10 isolates were collected from hospital A and 6 from hospital B. The resistant isolates were recovered from inpatients (ICU: 5, Internal ward: 4, Neurology: 1, and Digestive ward: 1 isolate), and outpatients (5 isolates).
The resistance rates of the 16 isolates to other antibiotics are shown in Table 2. In accordance with disk diffusion results, the most resistance among the ceftazidime-avibactam and ceftolozane-tazobactam resistant isolates belonged to cefoxitin (100%), cefotaxime (100%), aztreonam (100%), and nitrofurantoin (100%). In addition, the lowest resistance belonged to fosfomycin (62.5%) (Table 2). The antibiotic resistance patterns of the 16 resistant isolates are mentioned in Table 3.
Table 2.
Resistance Patterns of Ceftazidime-Avibactam and Ceftolozane-Tazobactam Resistant P. aeruginosa Isolates
| Antibiotics | R | I | S | Resistant (%) | Sensitive (%) |
|---|---|---|---|---|---|
| Imipenem | 14 | 0 | 2 | 87.5 | 12.5 |
| Ertapenem | 14 | 0 | 2 | 87.5 | 12.5 |
| Gentamicin | 15 | 0 | 1 | 93.8 | 6.2 |
| Amikacin | 12 | 3 | 1 | 75 | 6.2 |
| Cefotaxime | 16 | 0 | 0 | 100 | 0 |
| Ceftazidime | 13 | 3 | 0 | 81.2 | 0 |
| Cefoxitin | 16 | 0 | 0 | 100 | 0 |
| Aztreonam | 16 | 0 | 0 | 100 | 0 |
| Cefepime | 15 | 0 | 1 | 93.8 | 6.2 |
| Fosfomycin | 10 | 0 | 6 | 62.5 | 37.5 |
| Nitrofurantoin | 16 | 0 | 0 | 100 | 0 |
| Ciprofloxacin | 15 | 0 | 1 | 93.8 | 6.2 |
| Levofloxacin | 15 | 0 | 1 | 93.8 | 6.2 |
Abbreviations: R, resistant; I, intermediate; S, sensitive.
Table 3.
Antibiotic Resistance Patterns of the 16 Resistant Isolates to CZA and C/T
| Patterns | Antibiotics | Isolate No. |
|---|---|---|
| AP1 | IMI, ETP, GM, AK, CTX, CAZ, FOT, ATM, CPM, NI, CRO, LEV | 1, 2, 3, 9, 11, 13 |
| AP2 | IMI, ETP, GM, AK, CTX, CAZ, FOT, ATM, CPM, FOX, NI, CRO, LEV | 4, 6, 10, 16 |
| AP3 | IMI, ETP, GM, CTX, CAZ, FOT, ATM, FOX, CPM, NI, CRO, LEV | 5, 7 |
| AP4 | CTX, FOX, ATM, FOT, NI | 8 |
| AP5 | GM, CTX, CAZ, FOX, ATM, CPM, FOT, NI, CRO, LEV | 12 |
| AP6 | IMI, ETP, GM, AK, CTX, FOX, ATM, CPM, FOT, NI, CRO, LEV | 14, 15 |
Abbreviations: IMI, imipenem; CAZ, ceftazidime; AK, amikacin; ATM, aztreonam; CRO, ciprofloxacin; GM, gentamicin; FOT, fosfomycin; NI, nitrofurantoin; FOX, cefoxitin; ETP, ertapenem; CPM, cefepime; CTX, cefotaxime; LEV, levofloxacin; AP, antibiotic pattern.
The results of MIC also confirmed the resistance of the tested isolates in disk diffusion. According to the results, MIC range of 16 tested isolates for ceftazidime-avibactam, ceftolozane-tazobactam, imipenem, ceftazidime, amikacin, aztreonam, and ciprofloxacin was 16->32, 16->32, 1->64, 16->256, 16->256, 32->128, and 1–64 µg/mL, respectively. In addition, MICs for each tested isolate are shown in Table 4.
Table 4.
Distribution of the Resistance Genes Among the Isolates and Their Relationships with Production of ESBL and MBL
| Isolate | MBL | ESBL | MIC (µg/mL) | Gene (s) | IP/OP | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CZA | C/T | IMI | CAZ | AK | ATM | CRO | ||||||
| 1 | + | – | 16 | 16 | 64 | >256 | 256 | 32 | 16 | OXA2, OXA10 | IP | |
| 2 | – | – | 16 | 32 | 32 | >256 | 64 | 64 | 16 | OXA2, OXA10, VIM | OP | |
| 3 | + | + | >32 | >32 | 64 | >256 | 64 | >128 | 4 | PER, OXA2, OXA10, VIM, OXA48 | IP | |
| 4 | + | – | 16 | 16 | >64 | 64 | >256 | 64 | 8 | OXA10, VIM | IP | |
| 5 | + | – | 32 | 32 | >64 | >256 | 32 | >128 | 64 | OXA10, VIM | OP | |
| 6 | – | – | 16 | 16 | 64 | 128 | 64 | 128 | 8 | OXA2, OXA10 | OP | |
| 7 | + | + | >32 | 16 | 64 | >256 | 32 | 64 | 64 | PER, OXA10 | IP | |
| 8 | – | – | 16 | 16 | 2 | 16 | 16 | 64 | 1 | OXA10 | IP | |
| 9 | + | – | 32 | 32 | 64 | >256 | 128 | >128 | 16 | OXA10, OXA48 | IP | |
| 10 | + | – | 16 | 16 | 32 | >256 | 64 | 32 | 16 | OXA10, OXA48 | IP | |
| 11 | + | – | 32 | 32 | 8 | 64 | 128 | 64 | 8 | OXA10, VIM, NDM, OXA48 | OP | |
| 12 | – | – | 16 | 16 | 1 | >256 | 32 | >128 | 16 | OXA10 | IP | |
| 13 | + | + | 32 | 32 | 32 | >256 | 64 | 32 | 32 | OXA10, CTXM | OP | |
| 14 | + | + | >32 | >32 | >64 | 16 | >256 | 32 | 64 | OXA10, CTXM, VIM, NDM, OXA48 | IP | |
| 15 | + | – | 32 | 16 | >64 | 16 | >256 | 64 | 64 | OXA10, VIM | IP | |
| 16 | + | + | 32 | 32 | >64 | >256 | >256 | 128 | 64 | OXA10, VIM | OP | |
Abbreviations: CZA, ceftazidime-avibactam; C/T, ceftolozan-tazobactam; IMI, Imipenem; CAZ, ceftazidime; AK, amikacin; ATM, aztreonam; CRO, ciprofloxacin; MBL, metallo beta lactamase; ESBL, extended-spectrum β-lactamase; MIC, minimum inhibitory concentration; IP, inpatient; OP, outpatient.
Phenotypic Detection of ESBL by CDDT
The ESBL phenotypic screening by CDDT showed that among 16 ceftazidime-avibactam and ceftolozane-tazobactam resistant isolates, 5 isolates (31.2%) were ESBL producers. Among the ESBL producers, 3 (60%) belonged to inpatients and 2 (40%) to outpatients. The rate of ESBL-producing isolates among different wards was Internal ward (2 isolates) and ICU (1 isolate). It was also observed that 4 (80%) of ESBL-positive isolates were collected from men and 1 (20%) from women. Furthermore, the ESBL-positive isolates were recovered from different age group, including 20–40 (3 isolates), 41–60 (1 isolate), and 61–80 (1 isolate). In addition, two isolates with the ability of producing ESBLs were collected from outpatients referred to hospital A.
Phenotypic Detection of MBLs Using CDDT
In the present study, of 16 ceftazidime-avibactam and ceftolozane-tazobactam resistant isolates, 12 (75%) isolates were MBL producers as determined by CDDT. The majority of the MBL producing isolates were resistant to the examined antibiotics. Distribution of the 12 MBL producer isolates according to the hospital wards showed that 41.7% (n=5) of these isolates were collected from ICU, 25% (n=3) from the internal ward and 33.3% (n=4) isolates from outpatient. In the present study, the rate of MBL producer’s isolates was higher at hospital A (58.3%) compared to hospital B (41.7%). In addition, 8 (66.6%) of MBL producer isolates were recovered from men and 4 (33.4%) from women.
Genomic Analysis
The results of PCR assay for 16 ceftazidime-avibactam and ceftolozane-tazobactam resistant isolates showed that all isolates (100%) contained blaOXA10. Furthermore, the results of PCR showed that eight resistant isolates (50%) harbored blaVIM, five isolates (31.2%) harbored blaOXA48, four isolates (25%) harbored blaOXA2, two isolates (12.5%) contained blaCTX-M, two isolates (12.5%) harbored blaPER, and two isolates (12.5%) contained blaNDM, whereas none of the resistant isolates were positive for blaGES, blaIMP, and blaSPM genes. Distribution of the resistance genes among each isolate and their relationships with the production of ESBL and MBL are shown in Table 4. Distribution of the resistance genes among the isolates showed that the isolates containing the resistance genes were collected at a higher rate from male than female patients. Furthermore, the majority of the isolates were among the MBL producer isolates.
PFGE Results
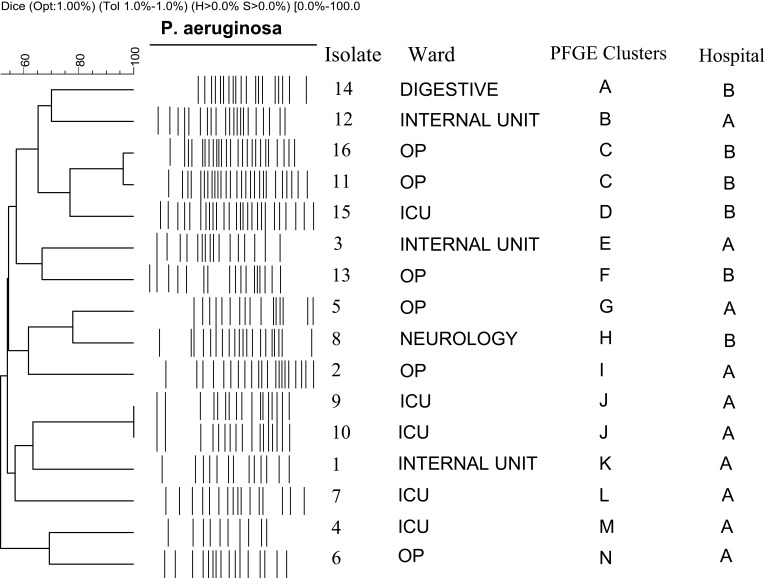
The ceftazidime-avibactam and ceftolozane-tazobactam resistant isolates from different patients were typed using PFGE. According to the genetic relatedness (defined as 85% band identity), they were grouped into 14 distinct pulsotypes (designated as pattern types A-N; Figure 1). When we compared the isolates in Figure 1, two shared pulsotypes were found. The isolates related to pulsotype C were included two isolates with different phenotypically antibiotic resistance pattern but were closely related and both produced the MBL enzyme. One isolate related to pulsotype C had 2 different carbapenemase genes, including blaNDM and blaOXA48 that none of them were presented in the other isolate. Furthermore, two isolates in pulsotype J showed different antibiotic resistance patterns that phenotypically were negative for ESBL production. In addition, both blaOXA10 and blaOXA48 resistance genes were detected in two isolates in pulsotype J.
Figure 1.
Genomic analysis of the isolates using PFGE. Dendrogram was constructed based on UPGMA by using Dice coefficient with a 1.0% band position tolerance. The scale above the dendrogram shows percentage of similarity and the dotted line indicates 85% similarity.
Abbreviations: OP, outpatient; ICU, intensive care unit.
Discussion
From the past, the treatment of bacterial infections such as UTIs relied on the prescription of antibiotics from different classes. Today, because of the increasing antibiotic resistance among the causative pathogens, there are limited options for the treatment of these infections that some of them are among the uncommon or harmful antibiotics. Furthermore, the inadequacies in clinical data more complicated the challenge for proper antimicrobial treatment of these infections.21,22
Ceftazidime-avibactam and ceftolozane-tazobactam were released into the market on about 2015 as novel antibiotics to treat severe gram-negative infections such as MDR P. aeruginosa. In this regard, the results from several general investigations in the USA have shown that ceftazidime-avibactam has maintained its activity since its FDA approval in 2015. In contrast, the efficacy of other beta-lactam antibiotics, such as piperacillin-tazobactam and meropenem has decreased.23–25
To the best of our knowledge, this study is the first report of the efficacy of CZA and C/T against P. aeruginosa isolated from UTI patients in Iran. The results could provide valuable information for clinicians and represent important data in the development of antibiotic therapy of UTI, especially about complicated UTI in Iran. In the present study, both CZA and C/T inhibited 92% of P. aeruginosa isolates. Thus, the results of this investigation could confirm the high activity of these compounds against P. aeruginosa isolated from UTI patients in Iran. Similar to our findings, in several studies conducted in the USA, CZA and C/T retained high activity against P. aeruginosa isolates that were resistant to routine antibiotics such as ceftazidime, cefepime, meropenem, and piperacillin-tazobactam.23,25 Furthermore, the high activity of CZA and C/T is reported against P. aeruginosa collected from UTI patients and other complicated infections in Asia-Pacific region, Qatar, Israel, UK, Spain, and some of the other parts of Europe.26–29
In line with our expectations, it was also found that the resistant isolates to CZA and C/T showed high resistance to the other tested antibiotics from different classes. Among the tested compounds, fosfomycin showed the best in vitro activity against the CZA and C/T resistant isolates and may be an alternative to treat high resistant UTIs. Unlike this finding, Sader et al23 demonstrated that in addition to the CZA and C/T, colistin showed the best activity against the tested isolates.
In accordance with one study in the USA, the susceptibility rates exhibited by both CZA and C/T in the present study were similar,23 whereas a higher activity of C/T is shown as compared to CZA in the other studies. In this regard, Horn et al26 observed that among ceftazidime and piperacillin-tazobactam resistant P. aeruginosa isolates, 88% and 66% isolates were sensitive to C/T and CZA, respectively. In the other study, Buehrle et al30 reported that C/T was more active than CZA against 38 meropenem-resistant P. aeruginosa isolates (67% vs 33%). In a recent study by Humphries et al,31 ceftazidime resistant P. aeruginosa isolates were shown to be 62% and 46% susceptible to C/T and CZA, respectively, and resistant isolates to 5 tested antibiotics were 57% and 28% susceptible to C/T and CZA, respectively.
In accordance with our findings, the in vitro results from several studies showed that MIC for CZA was lower than the MIC obtained for ceftazidime alone that demonstrated the increased susceptibility of P. aeruginosa isolates to CZA compared to the ceftazidime alone.32–34 Livermore et al35 also evaluated the activity of C/T against a wide range of P. aeruginosa and found that MICs of C/T were two to eight folds lower than those for ceftazidime alone.
Different mechanisms were reported for the resistance to CZA and C/T among the P. aeruginosa strains. The results of Mendes et al36 identified that the resistance mechanism of P. aeruginosa isolates to CZA was related to the overexpression of AmpC, blaOXA10, blaPER-1, or their mixture. Because of the presence of blaOXA10 in all resistant isolates in the present study, it is likely that the presence of blaOXA10 gene has been one of the resistance mechanisms of the isolates. In the present study, the majority of resistant isolates to CZA and C/T with MIC=32 or >32 µg/mL produced both ESBL and MBL enzymes phenotypically that suggests there is a relationship between the production of the enzymes and resistance to CZA and C/T. The production of carbapenemases blaVIM and blaNDM among our isolates (50% and 12.5%) could be another resistance mechanism.37 The results from the other studies also indicated that the resistant isolates to CZA usually employ several resistance mechanisms such as loss of OprD, overexpression of AmpC, MexCD-OprJ, MexAB-OprM, MexXY-OprM, and decreased permeability to these agents38,39 which were not evaluated in this study.
The PFGE analysis indicated that there was a significant genomic diversity among the resistant P. aeruginosa isolates to CZA and C/T. There were only two identical pulsotypes (identity more than 85%) in the tested isolates. Similar to one study in Turkey,40 we found that the isolates in shared pulsotypes (pulsotypes C and J) did not have similar antimicrobial resistance patterns. We followed the hospital records of the patients in pulsotypes C and J and found that both patients in pulsotype J (patients 9, a 49 years old man, and patient 10, a 25 years old woman) had been hospitalized in same ward (ICU) of one hospital (hospital A) on the same dates. Thus, it is likely that the hospitalization in one ward resulted in the cross-transmission of the resistant isolates between the two patients by the health-care personnel or other routes. The patients in pulsotypes C (patients 11, a 72 years old man, and patient 16, a 74 years old woman) had been referred as outpatients to hospital B on different dates to examine UTI by the laboratory tests. We found that the two patients had never been hospitalized in the past in the hospital and there was no contact between the two patients. Thus, according to the limited clinical information of the patients, the transmission route of the pulsotype between the two patients is unclear. This isolate could be transferred from the un-cleaned outpatient waiting room in the hospital by physical contact or other routes. Such studies demonstrated that improvements in the hospitals especially in special wards such as ICU, outpatient waiting rooms, and health care personnel are required to prevent or decrease the chance of transmitting resistant isolates in the hospitals and environments.
Our study has several limitations. First, as the study was performed at two big hospitals in Tehran, Iran, there were limitations in obtaining all detailed clinical information of patients. Second, we could not follow the patients, especially the patients referred to the mentioned hospitals as outpatients, for a long time. This could be especially important about the analysis of the results obtained from identical pulsotypes in PFGE.
Conclusion
Ceftazidime-avibactam and ceftolozane-tazobactam showed high in vitro activity against a collection of P. aeruginosa isolated from patients with UTI in 2 Iranian hospitals. Thus, these agents could be valuable treatment options for UTIs caused by P. aeruginosa, especially isolates that are resistant to most antibiotics currently available. On the other hand, Iran is one of the countries in which the new antibiotics are used in limited medical centers; thus, the low rate of resistance to the antibiotics is alarming for hygienic systems and also will fail antibiotic stewardships. This should be considered to avoid further spreading of the antibiotic resistance among the P. aeruginosa and other bacteria in medical centers.
Acknowledgments
The study was supported by Pasteur Institute of Iran.
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Flores-Mireles AL, Walker JN, Caparon M, Hultgren SJ. Urinary tract infections: epidemiology, mechanisms of infection and treatment options. Nat Rev Microbiol. 2015;13(5):269–284. doi: 10.1038/nrmicro3432 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Streeter K, Katouli M. Pseudomonas aeruginosa: a review of their pathogenesis and prevalence in clinical settings and the environment. Infect Epidemiol Med. 2016;2:25–32. doi: 10.18869/modares.iem.2.1.25 [DOI] [Google Scholar]
- 3.Nguyen L, Garcia J, Gruenberg K, MacDougall C. Multidrug-resistant pseudomonas infections: hard to treat, but hope on the horizon? Curr Infect Dis Rep. 2018;20(8):1–10. doi: 10.1007/s11908-018-0629-6 [DOI] [PubMed] [Google Scholar]
- 4.Mikhail S, Singh NB, Kebriaei R, et al. Evaluation of the synergy of ceftazidime-avibactam in combination with meropenem, amikacin, aztreonam, colistin, or fosfomycin against well-characterized multidrug-resistant Klebsiella pneumoniae and Pseudomonas aeruginosa. Antimicrob Agents Chemother. 2019;63(8):e00779–00719. doi: 10.1128/AAC.00779-19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Karlowsky JA, Kazmierczak KM, Bouchillon SK, de Jonge BLM, Stone GG, Sahm DF. Activity of ceftazidime-avibactam against clinical isolates of enterobacteriaceae and Pseudomonas aeruginosa collected in Latin American countries: results from the INFORM global surveillance program, 2012 to 2015. Antimicrob Agents Chemother. 2019;63(4):e01814–e01818. doi: 10.1128/AAC.01814-18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Shortridge D, Pfaller MA, Arends SR, Raddatz J, DePestel DD, Flamm RK. Comparison of the in vitro susceptibility of ceftolozane-tazobactam with the cumulative susceptibility rates of standard antibiotic combinations when tested against Pseudomonas aeruginosa from ICU patients with bloodstream infections or pneumonia. Open Forum Infect Dis. 2019;2:1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.García-Fernández S, García-Castillo M, Bou G, et al. Activity of ceftolozane/tazobactam against Pseudomonas aeruginosa and Enterobacterales isolates recovered from intensive care unit patients in Spain: the SUPERIOR multicentre study. Int J Antimicrob Agents. 2019;53(5):682–688. doi: 10.1016/j.ijantimicag.2019.02.004 [DOI] [PubMed] [Google Scholar]
- 8.Shortridge D, Castanheira M, Pfaller MA, Flamm RK. Ceftolozane-tazobactam activity against Pseudomonas aeruginosa clinical isolates from US hospitals: report from the PACTS antimicrobial surveillance program, 2012 to 2015. Antimicrob Agents Chemother. 2017;61(7):e00465–e00417. doi: 10.1128/AAC.00465-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Karami P, Mohajeri P, Mashouf RY, et al. Molecular characterization of clinical and environmental Pseudomonas aeruginosa isolated in a burn center. Saudi J Biol Sci. 2019;26(7):1731–1736. doi: 10.1016/j.sjbs.2018.07.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Alatoom A, Elsayed H, Lawlor K, et al. Comparison of antimicrobial activity between ceftolozane–tazobactam and ceftazidime–avibactam against multidrug-resistant isolates of Escherichia coli, Klebsiella pneumoniae, and Pseudomonas aeruginosa. Int J Infect Dis. 2017;62:39–43. doi: 10.1016/j.ijid.2017.06.007 [DOI] [PubMed] [Google Scholar]
- 11.CLSI. M100 Performance Standards for Antimicrobial. Clinical and Laboratory Standas Institute 950 West Valley Road, Suite 2500; 2018. [Google Scholar]
- 12.Laudy AE, Róg P, Smolińska-Król K, et al. Prevalence of ESBL-producing Pseudomonas aeruginosa isolates in Warsaw, Poland, detected by various phenotypic and genotypic methods. PLoS One. 2017;12(6):e0180121. doi: 10.1371/journal.pone.0180121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sachdeva R, Sharma B, Sharma R. Evaluation of different phenotypic tests for detection of metallo-β-lactamases in imipenem-resistant Pseudomonas aeruginosa. J Lab Physicians. 2017;9(4):249. doi: 10.4103/JLP.JLP_118_16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Daoud Z, Salem Sokhn E, Masri K, Matar GM, Doron S. Escherichia coli isolated from urinary tract infections of lebanese patients between 2005 and 2012: epidemiology and profiles of resistance. Front Med (Lausanne). 2015;2:26. doi: 10.3389/fmed.2015.00061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Franco MRG, Caiaffa-Filho HH, Burattini MN, Rossi F. Metallo-beta-lactamases among imipenem-resistant Pseudomonas aeruginosa in a Brazilian university hospital. Clinics. 2010;65(9):825–829. doi: 10.1590/S1807-59322010000900002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bert F, Branger C, Lambert-Zechovsky N. Identification of PSE and OXA β-lactamase genes in Pseudomonas aeruginosa using PCR–restriction fragment length polymorphism. J Antimicrob Chemother. 2002;50(1):11–18. doi: 10.1093/jac/dkf069 [DOI] [PubMed] [Google Scholar]
- 17.Lee S, Park Y-J, Kim M, et al. Prevalence of Ambler class A and D β-lactamases among clinical isolates of Pseudomonas aeruginosa in Korea. J Antimicrob Chemother. 2005;56(1):122–127. doi: 10.1093/jac/dki160 [DOI] [PubMed] [Google Scholar]
- 18.Shakibaie MR, Shahcheraghi F, Hashemi A, Adeli NS. Detection of TEM, SHV and PER type extended-spectrum ß-lactamase genes among clinical strains of Pseudomonas aeruginosa isolated from burnt patients at Shafa-Hospital, Kerman, Iran. Iran J Basic Med Sci. 2008;11(2):104–111. [Google Scholar]
- 19.Poirel L, Naas T, Nicolas D, et al. Characterization of VIM-2, a carbapenem-hydrolyzing metallo-β-lactamase and its plasmid-and integron-borne gene from a Pseudomonas aeruginosa clinical isolate in France. Antimicrob Agents Chemother. 2000;44(4):891–897. doi: 10.1128/AAC.44.4.891-897.2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Omeroglu EE. Determination of the genetic diversity of different bioluminescent bacteria by pulsed-field gel electrophoresis (PFGE). Jundishapur J Microbiol. 2015;8:7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Walker E, Lyman A, Gupta K, Mahoney MV, Snyder GM, Hirsch EB. Clinical management of an increasing threat: outpatient urinary tract infections due to multidrug-resistant uropathogens. Clin Infect Dis. 2016;63(7):960–965. doi: 10.1093/cid/ciw396 [DOI] [PubMed] [Google Scholar]
- 22.Sanchez GV, Babiker A, Master RN, Luu T, Mathur A, Bordon J. Antibiotic resistance among urinary isolates from female outpatients in the United States in 2003 and 2012. Antimicrob Agents Chemother. 2016;60(5):2680–2683. doi: 10.1128/AAC.02897-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sader HS, Flamm RK, Carvalhaes CG, Castanheira M. Antimicrobial susceptibility of Pseudomonas aeruginosa to Ceftazidime-avibactam, ceftolozane-tazobactam, piperacillin-tazobactam, and meropenem stratified by U.S. Census divisions: results from the 2017 INFORM program. Antimicrob Agents Chemother. 2018;62(12):e01587–18. doi: 10.1128/AAC.01587-18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Castanheira M, Duncan LR, Mendes RE, Sader HS, Shortridge D. Activity of ceftolozane-tazobactam against Pseudomonas aeruginosa and enterobacteriaceae isolates collected from respiratory tract specimens of hospitalized patients in the United States during 2013 to 2015. Antimicrob Agents Chemother. 2018;62(3):e02125–17. doi: 10.1128/AAC.02125-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sader HS, Huband MD, Castanheira M, Flamm RK. Pseudomonas aeruginosa antimicrobial susceptibility results from four years (2012 to 2015) of the international network for optimal resistance monitoring program in the United States. Antimicrob Agents Chemother. 2017;61(3):e02252–16. doi: 10.1128/AAC.02252-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Van Horn K, Sneed D, Goldstein E. Comparative “real world” in vitro activity of two new antimicrobials (ceftolozane-tazobactam and ceftazidime-avibactam) against ceftazidime non-susceptible Pseudomonas aeruginosa and resistant Enterobacteriaceae from california long term acute care hospitals. Clin Microbiol Infect Dis. 2018;3(1):1–4. [Google Scholar]
- 27.Tato M, Garcia-Castillo M, Bofarull AM, Canton R. In vitro activity of ceftolozane/tazobactam against clinical isolates of Pseudomonas aeruginosa and Enterobacteriaceae recovered in Spanish medical centres: results of the CENIT study. Int J Antimicrob Agents. 2015;46(5):502–510. doi: 10.1016/j.ijantimicag.2015.07.004 [DOI] [PubMed] [Google Scholar]
- 28.Sutherland C, Nicolau D. 415In vitro potency of ceftolozane/tazobactam against Pseudomonas aeruginosa displaying multidrug resistance. Open Forum Infect Dis. 2014;S159. doi: 10.1093/ofid/ofu052.281 [DOI] [Google Scholar]
- 29.Sader HS, Castanheira M, Flamm RK. Antimicrobial activity of ceftazidime-avibactam against gram-negative bacteria isolated from patients hospitalized with pneumonia in U.S. Medical centers, 2011 to 2015. Antimicrob Agents Chemother. 2017;61(4):e02083–16. doi: 10.1128/AAC.02083-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Buehrle DJ, Shields RK, Chen L, et al. Evaluation of the activity of ceftazidime-avibactam and ceftolozane-tazobactam against meropenem-resistant Pseudomonas aeruginosa Isolates. Antimicrob Agents Chemother. 2016;60(5):3227–3231. doi: 10.1128/AAC.02969-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Humphries RM, Hindler JA, Wong-Beringer A, Miller SA. Activity of ceftolozane-tazobactam and ceftazidime-avibactam against beta-lactam-resistant Pseudomonas aeruginosa isolates. Antimicrob Agents Chemother. 2017;61(12):e01858–01817. doi: 10.1128/AAC.01858-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Stone GG, Newell P, Bradford PA. In vitro activity of ceftazidime-avibactam against isolates from patients in a Phase 3 clinical trial for treatment of complicated intra-abdominal infections. Antimicrob Agents Chemother. 2018;62(7):e02584–17. doi: 10.1128/AAC.02584-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sader HS, Castanheira M, Flamm RK, Huband MD, Jones RN. Ceftazidime-avibactam activity against aerobic gram negative organisms isolated from intra-abdominal infections in United States Hospitals, 2012-2014. Surg Infect (Larchmt). 2016;17(4):473–478. doi: 10.1089/sur.2016.036 [DOI] [PubMed] [Google Scholar]
- 34.Stone GG, Bradford PA, Yates K, Newell P. In vitro activity of ceftazidime/avibactam against urinary isolates from patients in a Phase 3 clinical trial programme for the treatment of complicated urinary tract infections. J Antimicrob Chemother. 2017;72(5):1396–1399. doi: 10.1093/jac/dkw561 [DOI] [PubMed] [Google Scholar]
- 35.Livermore DM, Mushtaq S, Meunier D, et al. Activity of ceftolozane/tazobactam against surveillance and ‘problem’Enterobacteriaceae, Pseudomonas aeruginosa and non-fermenters from the British Isles. J Antimicrob Chemother. 2017;72(8):2278–2289. doi: 10.1093/jac/dkx136 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Mendes RE, Castanheira M, Woosley LN, Stone GG, Bradford PA, Flamm RK. Molecular beta-lactamase characterization of aerobic gram-negative pathogens recovered from patients enrolled in the ceftazidime-avibactam Phase 3 trials for complicated intra-abdominal infections, with efficacies analyzed against susceptible and resistant subsets. Antimicrob Agents Chemother. 2017;61(6):e02447–16. doi: 10.1128/AAC.02447-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Van Duin D, Bonomo RA. Ceftazidime/Avibactam and Ceftolozane/Tazobactam: second-generation beta-Lactam/beta-Lactamase Inhibitor Combinations. Clin Infect Dis. 2016;63(2):234–241. doi: 10.1093/cid/ciw243 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mazuski JE, Gasink LB, Armstrong J, et al. Efficacy and safety of ceftazidime-avibactam plus metronidazole versus meropenem in the treatment of complicated intra-abdominal infection: results from a randomized, controlled, double-blind, Phase 3 program. Clin Infect Dis. 2016;62(11):1380–1389. doi: 10.1093/cid/ciw133 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Castanheira M, Doyle T, Davis A, Mendes R, Sader H. Intrinsic resistance mechanisms detected among ceftazidime-avibactam-susceptible and-resistant Pseudomonas aeruginosa isolates collected from United States Hospitals (2015), Sunday-166. ASM Microbe. 2017;1. [Google Scholar]
- 40.Okur DS, Yuruyen C, Gungor O, et al. Genotypic characterization of Pseudomonas aeruginosa isolates from Turkish children with cystic fibrosis. Infect Drug Resist. 2019;12:675–685. doi: 10.2147/IDR.S183151 [DOI] [PMC free article] [PubMed] [Google Scholar]