Abstract
Atrial fibrillation (AF) is the most common arrhythmia in adults and contributes directly to adverse clinical events, ranging from ischemic stroke to heart failure and cardiovascular death. Because the incidence of AF and its attendant complications increase with age, there is a strong and growing need to develop safe, effective and widely available therapies. The following review summarizes the use of oral anticoagulants in older adults with AF, focusing on practical topics such as drug metabolism, drug- drug interactions, co-morbidities and cost considerations in a complex payer environment.
Keywords: Atrial fibrillation, novel oral anticoagulant, older adult
AGE AND ATRIAL FIBIRLLATION
Defining Old Age
There is no general agreement on the age at which a person becomes old [1]. The use of a calendar age to mark the threshold of old age assumes “equivalence” with biological age, yet it is generally accepted that these are not necessarily synonymous [1]. In many societies, old age is precipitated by a change in social role or physical status. If one considers the self-definition of “old age”, as people grow older, it seems their self-definitions of old age become decreasingly multifaceted and increasingly related to health status [2,3]. Yet others consider an adult old when one has surpassed the average life span of human beings [4].
According to a 2004 report by the Centers for Disease Control Prevention (CDC), The Merck Institute of Aging and Health, and the Gerontological Society of America (GSA), Americans are living longer due to declines in heart disease and stroke mortality, but chronic diseases such as diabetes and high blood pressure are becoming more prevalent among older adults. The proportion of those aged 65–74 reporting very good- to- excellent health rose to 42% in 1999, which is an increase of 7% since 1982. Those age 75 years and older, however, reported little improvement or a decline in health status (34% and 28% of those aged 75–84 and aged 85+, respectively, reported themselves in very good- to -excellent health) [5].
Accordingly, health care providers now identify subgroups of older adults as “younger old” (ages 65–74), “older old” (ages 75–85) and “oldest old” (ages 85 and older) [4]. The number of “younger old” increased when the first of 77 million “Baby Boomers” turned 65 years old in 2011. Presently, nearly 10,000 Americans turn 65 years old daily [5].
For the purpose of our review, we will define “older adults” as age 75 and older. Patients who are ≥ 75 years old often present a complex clinical picture with coexisting conditions and frailty [6].
Atrial Fibrillation (AF) in an Aging Population
The mean age in phase 3 clinical trials of novel oral anticoagulants (NOAC) among patients with non- valvular atrial fibrillation (AF) exceeds 70 years of age. This is not unexpected given that the incidence of AF increases with age and nearly half of all patients with AF are 75 or older [7]. In addition, stroke as a complication of AF increases with age: the attributable risk of stroke from AF is estimated at 1.5% for those aged 50 to 59 years, and 23.5% for those aged 80–89 years [8]. While older adults with AF are at higher risk of thromboembolic stroke compared to their younger counterparts, they are also less likely to be anticoagulated largely due to greater propensity for bleeding and, at times, an unfounded perception of prohibitive risk [9].
The available data show that anticoagulant and platelet-directed therapy reduce stroke risk by approximately 60% and 20%, respectively, in patients with AF [10]. Yet just over half of eligible patients receive vitamin K antagonists (VKAs) [11]. Factors that limit the use of VKAs include: their narrow therapeutic index; requirement for frequent coagulation monitoring; complex dosing; prolonged pharmacodynamic effects; and numerous food-drug and drug-drug interactions.
Accordingly, NOACs should represent an attractive alternative to VKAs in older adults with AF. To assure optimal utilization, clinicians must be familiar with: NOAC pharmacology; clinical trial methodology, including entry and exclusion criteria; and several factors specific to older patients that impact drug metabolism and clearance, toxicity and effectiveness.
DRUG METABOLISM AND EFFECTS OF AGING
Drug metabolism is altered with aging, in ways that may not be readily apparent (or easily quantitated). Inter-individual variability in response to drug effects is noticeable among older adults and far more prominent compared to young adults.
Pharmacokinetic Considerations
Bioavailablity
The bioavailability of any drug after oral administration depends upon many factors, including the fraction of the administered dose absorbed through the gastrointestinal mucosa, the fraction of the absorbed dose that passes through the gastrointestinal tract into the hepatic portal blood unmetabolized, and the hepatic first-pass availability [12]. Gut absorption typically declines and first pass metabolism slows with aging, often producing a clinically insignificant net effect on drug bioavailability compared to young adults. Bioavailability can be increased, however, if the hepatic first-pass effect of highly cleared drugs is greatly reduced, due to decreases in liver mass and perfusion with aging [13]. Accordingly, the conclusions of studies on old age and the bioavailability of orally administered drugs are variable: the absolute bioavailability of labetaolol and lidocaine, for example, nearly doubles with age, whereas no differences have been reported for metoprolol and morphine [12]. No specific effect of aging on the efflux pump, P-glycoprotein, in the gastrointestinal tract has been reported [12].
Volume of Distribution
Alterations in protein binding that occur with aging do not typically affect orally administered medications. Body fat increases and body water decreases with aging; this may affect peak plasma concentrations and other pharmacokinetic parameters following loading doses [12].
Renal Aging
There have been many studies of the pharmacokinetics of renally excreted drugs with aging, although few have attempted to define the specific relationship among normal aging, renal function, and altered pharmacokinetics [12]. The incidence of hypertension increases with age as peripheral blood vessels calcify and stiffen. The presence of small vessel pathology in older adults without apparent renal disease or hypertension suggests that even in healthy older adults, renal changes may be secondary to vascular disease and altered vascular responsiveness [12]. In addition, glomerulosclerosis has been associated with aging and disease (including hypertension) [14]. Regardless of etiology, most adults demonstrate a decline in renal function over time [15]. When subjects with “possible renal or urinary tract disease” and subjects on diuretics and antihypertensives were excluded from the Baltimore Longitudinal Study of Aging, the mean decrease in creatinine clearance was 0.75 ml/min/year. However, one-third had no decrease in creatinine clearance over a 23 year period [16].
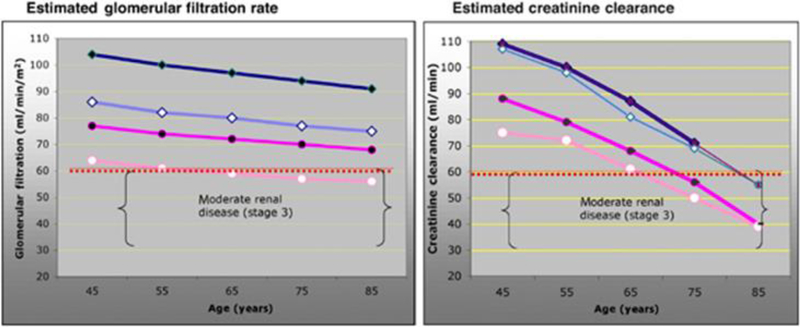
Serum creatinine does not provide an accurate assessment of renal function, making methods used to calculate function based on creatinine imperfect. In older adults, the daily production of creatinine is reduced as result of decreased muscle mass. Figure 1 demonstrates the estimated glomerular filtration rate (eGFR) using the Modification of Diet in Renal Disease (MDRD) simplified algorithm (left) and estimated creatinine clearance using the Cockcroft-Gault formula (right) for a serum creatinine of 1.0 mg/dL. Renal dysfunction in older adults (especially white women) calculated using the Cockcroft-Gault formula may be underestimated, while it may be overestimated using the MDRD formula [17,12]. As a consequence, careful examination of the safety and efficacy of renally-cleared drugs in older adults is particularly important in drug development.
Figure 1:
eGFR for Americans aged 45–85 years using the modification of diet in renal disease simplified algorithm [74] is presented in the left panel, and estimated creatinine clearance using the Cockcroft and Gault formula [75] is presented in the right panel. For calculations, mean weight and height by decade were obtained from US survey data (NHANES, http://www.cdc.gov), with serum creatinine designated as 1.0 mg/dl. Pink lines and circles represent estimates for women; blue lines and triangles are estimates for men; white, open symbols are estimates for Caucasians; and solid black symbols represent estimates for African Americans. The dotted red line indicates a GFR of 60 ml/min/1.73m2, demarcating the level below which there is at least a moderate decrease in GFR. The brackets enclose GFRs of 30–59 ml/min/m2 classified as stage 3 renal disease or moderate GFR decrease (adapted from Schwartz) [17].
Reprinted by permission from Macmillan Publishers Ltd: Clinical Pharmacology and Therapeutics, Schwartz JB (2007).
Hepatic Aging
Aging is associated with a decrease in hepatic blood flow and hepatocyte mass as well as pseudo-capillarization [12]. In general, the inter-individual variation in metabolic drug clearance by cytochrome (CYP) P450 enzymes or phase I reactions exceeds the decline caused by aging [18]. Phase II pathways (e.g., glucuronidation, acetylation, sulfation) are well preserved in older adults [13].
In addition to the enzymatic processing of drugs within the hepatocytes, various transporter proteins accomplishing the hepatic uptake of drugs and the biliary and hepatic extrusion of metabolites are indirectly involved in drug metabolism. During recent years, the importance of drug transporters for absorption, distribution, and elimination processes has been increasingly recognized. Such transport processes and the oxygen supply for phase I reactions to the hepatocytes might demonstrate age- dependencies yet to be fully delineated [13].
Epigenetic influences have potentially important implications for hepatic clearance and variability in response to drugs in older adults [19]; however, age-related changes in the liver itself may have a greater impact on overall hepatic clearance [20].
Pharmacodynamic Considerations
The effects of age on the activity and expression of many receptors have been reported. However, the impact of age on pharmacodynamic effects of drugs acting on specific receptors is less well established [12]. Altered responses to drugs in older adults could be due to several factors, including age-related changes in drug-receptor interactions, receptor-membrane interactions, post-receptor events, structural changes in organs or tissues, and altered homeostatic functions [21].
Pharmacodynamic studies have focused primarily on relatively healthy older adults, yet the pathophysiology of human diseases themselves may alter the pharmacodynamic response and therapeutic outcome [12]. Therefore, in sicker older adults, dose-response relationships may be more difficult to characterize with medications including NOAC.
Adverse Drug Reactions
When adverse drug reactions occur in older adults, they are more likely to be severe and less likely to be recognized or reported by the patient [12]. Although age-related changes in pharmacokinetics and pharmacodynamics may contribute, adverse drug reactions appear more strongly associated with comorbidity and polypharmacy [22,23]. Polypharmacy has been associated with frailty in older adults independent of medical diagnoses [24,25]. How drug metabolism is altered in the presence of frailty is not well established: frailty may produce a general impairment of conjugation pathways, and possibly reduced drug clearance through CYP3A4 [26].
Inflammation is a component of both frailty and aging that has the capacity to down-regulate drug metabolism and transporter pathways, reducing systemic clearance and possibly leading to adverse drug effects [27]. Inflammation can also increase the chance of adverse drug effects, both on- and off-target, through activation of the coagulation system: IL-6 plays a major role in synthesis and release of fibrinogen, tissue factor and factor VIII from multiple tissues [28].
Many coagulation proteins that increase with aging are also acute phase reactants, raising the possibility that age-related thrombosis could be secondary to inflammation. This seems plausible when considering that regular physical activity in older adults has been associated with reductions in fibrinogen, factor XIII and factor IX [28].
Inter-individual Variability in Drug Response among Older Adults
Chronological age is recognized as a poor predictor of variability in response to medications [20]. Variability is a likely consequence of age-associated changes in organ function and body composition, declining homeostatic reserve, and comorbid disease, all of which alter drug pharmacokinetics and pharmacodynamics [20].
Frailty is a confounding factor when considering the impact of aging on drug disposition [13]. Inflammation associated with frailty has the potential to significantly alter drug transporter and metabolizing enzyme expression and thereby contribute to variability in drug clearance (and therefore response) in frail older people [20].
PHARMALOGICAL PROFILE OF NOVEL ORAL ANTICOAGULANTS FOR STROKE PREVENTION IN NON-VALVULAR ATRIAL FIBRILLATION
Dabigatran
As the first NOAC approved for non-valvular AF in adults, dabigatran was received by the medical community with high expectations. Unlike the vitamin K antagonist (VKA) warfarin, dabigatran has a low propensity to interact with other drugs, which is critical for older adults at risk for adverse drug effects. With predominantly (80%) renal clearance and a lack of an antidote, dabigatran has not been fully embraced by practicing clinicians. To better understand the potential reasons for its relatively slow uptake in clinical practice, dabigatran’s pharmacology, performance in clinical trials, and the U.S. Food and Drug Administration (FDA) approval process will be detailed in the following section.
Dabigatran etexilate, a pro-drug, is converted to the active metabolite dabigatran which directly inhibits free thrombin (factor IIa) and clot-bound thrombin. Dabigatran does not exert direct inhibitory activity against factor Xa, trypsin, plasmin, tissue plasminogen activator, and activated protein C [29] (Table 1). Two doses of dabigatran (110 mg, 150 mg) given twice daily were studied in the RE-LY trial [30]. The 150 mg dose was superior to warfarin in reducing the primary endpoint of stroke (comprised of both ischemic and hemorrhagic stroke) and systemic embolism by 34% with no significant difference in major bleeding. The incidence of intracranial hemorrhage (ICH), a composite comprised of intraparenchymal, subarachnoid and sub-dural hemorrhage was much less common with dabigatran than with warfarin; however, major gastrointestinal bleeding was more common. By comparison, the 110 mg dose was non- inferior to warfarin in preventing ischemic stroke and systemic embolism, and was associated with a 20% relative risk reduction in major bleeding compared with warfarin.
Table 1:
Novel Oral Anticoagulant Properties and Trial Data
| Novel Oral Anticoagulant | Dabigatran Etexilate | Rivaroxaban | Apixaban |
|---|---|---|---|
| Mechanism of Action | Direct thrombin inhibitor | Competitive factor Xa inhibitor | Competitive factor Xa inhibitor |
| Manufacturer | Boehringer Ingelheim | Bayer/Johnson & Johnson | Bristol-Meyers Squibb/Pfizer |
| FDA approval | October 19, 2010 | November 4, 2011 | Pending |
| Half-life (hours) | 12–17 | 5–13 | 9–14 |
| Bioavailability | 3.5–6.5% | 70–80% | >50% |
| Hepatic clearance | 20% (glucuronidation) | 28%–35% | 75% |
| Renal clearance | 80% | 65% | 25% |
| Protein binding | 35% | 92–95% | 87% |
| P-gp inhibition/induction | Yes | Yes | yes |
| CYP3A4 inhibition/induction | No | Yes | yes |
| Food interference | Acidic milieu needed for absorption; food and drugs increasing stomach / intestine pH may reduce absorption | Absorption increased by food | Possibly no effect |
| Phase III Clinical Trial | RE-LY | ROCKET-AF | ARISTOTLE |
| Study size | n=18,113 | n=14,264 | n=18,206 |
| Age | 71.4 years (mean) | 71.2 years (mean) | 70 years (median) |
| Study design | intention to treat | on treatment | intention to treat |
| Dose(s) studied | 110 mg bid, 150 mg bid | 15 mg daily (CrCl 30–49 mL/min), 20 mg daily | 5 mg bid, 2.5 mg bid (CrCl 30–49 mL/min) |
| Time in Therapeutic Range | 67% | 57% | 66% |
| % VKA Naïve | 50% | 38% | 43% |
| CHADS2 score | CHADS ≥ 1 (mean 2.1) | CHADS ≥ 2 (mean 3.5) | CHAD ≥ 1 (mean 2.1) |
| Creatinine Clearance (ml/min) | 69 (median) | 67 (median) | >80 (41%); 50–80 (42%); 30–50 (15%); ≤30 (2%) |
According to the methods paper for RE-LY [31]:
The decision to test 2 doses of dabigatran in a large phase 3 trial is unusual. Identifying the correct dose was considered critical. The dose of 150 mg BID was tested in the phase 2 PETRO study, whereas 110 mg twice a day had not been directly tested. The choice of this lower dose was based on interpolations of phase 2 data, considerations of the peak and time course of anticoagulant effect of dabigatran, and the observation that a total daily dose of 220 mg was effective in orthopedic surgery patients for deep venous thrombosis prevention. The identification of a lower dabigatran etexilate dose [for non-valvular atrial fibrillation] that might potentially have similar efficacy to adjusted-dose warfarin with less bleeding was considered important. Having 2 doses might allow tailoring of dosing to optimize the benefit against risk.
To date, the FDA has approved two dosing regimens, a 150 mg twice-daily regimen for those with normal, mildly impaired and moderately impaired kidney function (CrCl 30 to 50 ml/min) and a 75 mg twice-daily regimen for those with severely impaired kidney function (CrCl 15 to 30 ml/min). No age or weight-based recommendations have been made.
A rationale for the FDA’s disapproval of the 110 mg dose appeared in the New England Journal of Medicine [32]:
Among the 40% of patients in RE-LY who were 75 years of age or older (n = 7238), the rate of stroke or systemic embolism was lower with 150 mg of dabigatran (1.4 per 100 patient years) than with 110 mg (1.9 per 100 patient-years), but the rate of major bleeding was higher (5.1 vs. 4.4 per 100 patient-years). If stroke or systemic embolism and major hemorrhage were considered equally undesirable, these rates would indicate similar benefit–risk assessments for the two doses. Most people would agree, however, that the irreversible effects of strokes and systemic emboli have greater clinical significance than nonfatal bleeding.
Subsequent sub-group analyses by age support this position for non-central nervous system major bleeding, but not for intracranial hemorrhage [33]. It is of interest that the FDA’s approval of 75 mg twice daily dosing in patients with severe renal insufficiency was based entirely on small pharmacokinetic studies [34] and simulations [35] including a population pharmacokinetic analysis performed by Liesenfeld et al. using RE-LY data [36]. They analyzed 27,706 plasma concentrates from 9,522 patients who received 110 mg or 150 mg dabigatran using non-linear mixed-effects modeling; of those studied 3,505 had severe renal insufficiency. Simulations included a 6-hour delay in dosing (e.g., dose 1 at 0:00, dose 2 at 18:00, dose 3 at 24:00 hours). Statistically significant factors (CrCl, age, sex, heart failure, South Asian race, body weight and hemoglobin), except for renal function status, showed only small- to- moderate effects on the apparent clearance of dabigatran (<26% change in exposure at steady state).
Renal function in RE-LY was approximated using estimated creatinine clearance (CrCl), not estimated glomerular filtration rate (eGFR). In essence, any effect of age on dabigatran clearance might have been taken into account by using the formula for CrCl. Interestingly, curves of renal function calculated by CrCl and eGFR (Figure 1) seem to deviate most dramatically for white women ≥ 75 years of age, a common demographic for AF with a recognized unmet need for anticoagulation. Moreover, the majority of patients in RE-LY were classified as having mild chronic kidney disease, highlighting the relevance of accurately accounting for age in the determination of renal insufficiency.
One must recognize that age lies on a continuum, as does renal dysfunction. Therefore, two disparate doses (one half of the other) may seem restrictive to the prescribing clinician treating a patient with ‘borderline’ or fluctuating renal function. As a result, dabigatran has been prescribed at a dose of 150 mg q12h every other day and 150 mg q day every other day; other possible dosing regimens are likely prescribed in daily clinical practice. This practice pattern is reminiscent of dosing VKA to a low target International Normalized Ratio (INR) in patients believed or known to be at risk for bleeding. Given the non-linear relationship between renal dysfunction and drug exposure, and varying peaks and troughs with regimens similar to those described, ‘mix and match dosing’ based upon most recent measurement of patient’s renal function may ultimately carry greater risk than likelihood of benefit.
According to Health Canada’s Health Products and Food Branch (HPFB) prescribing information, patients aged 80 years and older with AF should be treated with dabigatran 110 mg BID for prevention of thromboembolic events [37]. This recommendation is based, in part, on the pharmacokinetic study showing exposure to dabigatran increased 1.7 to 2-fold in 36 participants ≥ 65 years (compared with young participants in earlier studies); this finding was attributed to slightly decreased renal function of older patients (CrCl 77 ± 13.4 mL/min) [38]. In patients with increased risk of bleeding (including those with moderate renal impairment; using concomitant medications that inhibit P-glycoprotein; receiving aspirin, NSAIDS, or clopidogrel; with concurrent disease; or undergoing procedures with special hemorrhagic risk), Canadian prescribing information also recommends 110 mg BID. Lastly, according to the European Medicines Agency (EMA) [39] and Health Canada’s HPBP prescribing information, dabigatran is not recommended for patients with liver enzymes >2 times the upper limit of normal.
While it may be difficult to completely reconcile different prescribing patterns in North America, it is easy to understand how existing evidence fails to provide completely clear guidance on how to optimally prescribe dabigatran to an older adult with AF.
Rivaroxaban
Rivaroxaban was approved for the treatment of non-valvular AF on November 4, 2011, approximately a year after dabigatran received FDA approval on October 19, 2010. Rivaroxaban has a dual mode of elimination; one-third is eliminated unchanged by the kidneys and two-thirds is metabolized by the liver. Approximately half of the fraction metabolized by the liver is eliminated through the hepatobiliary route; the other half is eliminated renally. As a NOAC with predominantly hepatic metabolism and once daily dosing, rivaroxaban was expected to overcome dabigatran’s shortcomings related to dosing and renal clearance in older adults. To understand why rivaroxaban might not always be an optimal choice for older adults, its pharmacology and clinical trials will be summarized (Table 1).
Rivaroxaban is a direct, specific and competitive active site factor Xa inhibitor [40]. In ROCKET-AF [41], 20 mg of daily rivaroxaban (and 15 mg in patients with moderate renal impairment defined as a CrCl of 30–49 mL/min) was non-inferior to warfarin for the prevention of stroke or non-CNS systemic embolism. Overall, there was no significant difference in major bleeding compared to warfarin; however, in the rivaroxaban group, intracranial and fatal bleeding occurred less frequently, and non-fatal gastrointestinal bleeding occurred more frequently. There was no evidence of heterogeneity in treatment effect across dosing groups [42]. Dosing was based on simulation and prior study of rivaroxaban in deep venous thrombosis [43].
Despite its predominantly hepatic metabolism, rivaroxaban has not been tested in, or approved for, patients with severe renal impairment. Within the liver, rivaroxaban is metabolized primary via cytochrome P450 CYP3A4/5 and to a lesser extent CYP2J2; P-glycoprotein transporters are also involved. With a systemic clearance of approximately 10L/h, rivaroxaban can be classified as a low- clearance drug, making it less likely to be susceptible to altered metabolism due to hepatic aging (from diminished hepatic blood flow).
The maximum concentration (Cmax) of rivaroxaban is unaffected by age; however, exposure is 41% higher in subjects > 75 years of age compared with those age 18 to 45 years [44]. Elimination occurs with mean terminal half-life of 5 to 9 hours in young individuals, and 11 to 13 hours in the elderly [45]. These findings are attributed to decreased renal function in the elderly [45,44]. Kubitza et al. performed a dose- escalation study of 48 healthy elderly subjects, age 60 to 76 years; they found no significant impact of age or gender on rivaroxaban exposure [45]. Simulations of data taken from two phase IIb studies of patients undergoing major orthopedic surgery indicate that only in case of 90-year-old patient with lean body mass of 30 kg would predicted rivaroxaban plasma concentration exceed a 90% confidence interval of the average (mean) younger, heavier patient [46]. Nevertheless, physiologically-based pharmacokinetic simulations suggest that drug-drug-disease interactions with rivaroxaban are possible, which could significantly increase rivaroxaban exposure and increase bleeding risk. Depending on severity of renal impairment, rivaroxaban exposure with the P-gp inhibitor erythromycin increases 1.9 to 2.6- fold in subjects 20–45 years and 2.5 to 3.0- fold in subjects 55–65 years [47].
Preliminary ROCKET-AF sub-group analyses show a greater benefit in stroke reduction in those ≥ 75 years (HR 0.67, 95% CI 0.51–0.88, in on-treatment analysis of patients ≥ 75 years vs. HR 0.91, 95% CI 0.70–1.19, <75 years) [48]. Though confidence intervals span 1.0, a relative risk reduction in ischemic stroke relative to warfarin in patients ≥ 75 years was also seen (HR 0.88, 95% CI 0.67–1.66); this advantage was not apparent in patients <75 (HR 1.10, 95% CI 0.84–1.44) [48].
Major bleeding was greater in those ≥ 75 years compared to the younger group (4.86 vs. 2.69 events per 100 patient years in those ≥ 75 years vs. <75 years, respectively; however, unlike dabigatran there was not an age-by-bleeding interaction); a trend toward more major bleeding in those ≥ 75 years versus warfarin was also seen (4.86 vs. 4.40 events per 100 patient years in those ≥ 75 years on rivaroxaban vs. warfarin, respectively) [48]. Hemorrhagic stroke risk was lower versus warfarin regardless of age (0.41 vs. 0.20 events per 100 patient years in those < 75 years on warfarin and rivaroxaban, respectively; 0.49 vs. 0.34 events per 100 patient years in those ≥ 75 years on warfarin and rivaroxaban, respectively). The Hazard Ratio for major bleeding on rivaroxaban vs. warfarin was 1.11 (95% CI 0.92–1.34) in patients ≥ 75 years vs. 0.96 (95% CI 0.78–1.19) in patients < 75 years [48].
Like dabigatran, rivaroxaban does not penetrate the blood-brain barrier [49] which could explain, at least in part, the lower rate of ICH compared with warfarin which is known to cross. ICH rates appeared lower regardless of stroke history [50]. Further study is warranted to determine what mechanism explains this observation with NOACs, and whether age plays an important role.
Apixaban
Apixaban is the third promising NOAC studied in phase 3 clinical trials for non-valvular AF. With EMA approval for non-valvular AF on November 20, 2012, followed shortly thereafter by Health Canada, FDA approval for non-valvular AF occurred on Friday, December 28, 2012. With very few drug interactions identified and mixed renal-hepatic clearance, it may be a particularly attractive treatment option for older adults (Table 1).
Apixaban, like rivaroxaban, is a direct, selective and competitive active site factor Xa inhibitor, but has a lower Ki (inhibitory constant) than rivaroxaban, indicating it has a higher potency; this is likely due to a better complementary “fit” for apixaban in the FXa protein’s active site. Whether this property will impact drug reversibility is unknown. Apixaban is metabolized by the liver (primarily by CYP3A4, CYP3A5 and sulfotransferases 1A1), kidneys and intestine, and excreted through the hepatobiliary system (75%) and kidneys (25%).
In ARISTOTLE [51], apixaban was associated with a 31% relative risk reduction in major bleeding (including a 48% relative risk reduction in hemorrhagic stroke), a 21% relative risk reduction in the combined endpoint of stroke or systemic embolism, and an 11% relative risk reduction in mortality relative to warfarin. Similar to AVERROES [52], 2.5 mg twice daily rather than 5 mg twice daily was given to patients with at least two of the following: age ≥ 80 years; weight ≤ 60 kg; serum Cr ≥ 1.5 mg/dL. Per protocol, apixaban dosing was based primarily on the phase 2 dose-ranging study in venous thromboembolism prevention [53], the study in deep venous thrombosis treatment [43], and “experiences with other anticoagulants studied in this patient population.” Outcomes for the sub-group receiving the lower dose of apixiban have not been published to date.
In a sub-study of AVERROES, moderate renal impairment (CrCl 30–49 mL/min) was found to be an independent predictor of stroke in AF patients. Among these patients, 2.5 mg twice daily apixaban significantly reduced the incidence of stroke relative to aspirin without increasing the risk for major hemorrhage. An ARISTOTLE sub-study supported these results: both the annual rates of stroke or systemic embolism and ischemic strokes were more than doubled in patients with moderate/severe renal impairment when compared with patients with normal renal function. With a consistent relative reduction in the rate of stroke or systemic embolism, apixaban provided the largest absolute benefits in patients with renal impairment [54]. Renal function was determined using two creatinine-based estimates of GFR namely, Cockcroft-Gault and Chronic Kidney Disease Epidemiology Collaboration, and using cystatin C, possibly a more reliable marker of renal function than serum creatinine [54]. Of the three formulas, only the one using cystatin C excludes age. Cystatin serum levels have been shown to depend upon body composition though which may affect cystatin-based measurement of GFR in those who are frail [55].
As an agent with primarily hepatic metabolism, apixaban may ultimately outperform dabigatran in patients with renal dysfunction. Whether apixaban will outperform dabigatran in older adults is less clear. Like rivaroxban and dabigatran (110 mg), apixaban did not show a significant overall reduction in ischemic stroke compared to warfarin. In an adjusted indirect comparison, apixaban did lower the risk of overall major and gastrointestinal bleeding versus dabigatran and rivaroxaban [56]. Additionally, with multiple clearance pathways, apixaban carries a comparatively modest potential for drug-drug interactions [57].
Several months prior to FDA approval, the American Heart Association / American Stroke Association incorporated apixaban into its AF management guidelines [58].
BLEEDING RISK IN OLDER ADULTS WITH AF
The complexity of atherothrombosis in older adults is heightened by a concomitant bleeding tendency—conceivably an age-related vasculopathy involving small, hemostasis-maintaining vessels that is characterized by impaired vascular healing, loss of anatomic vasoreactivity, and immune incompetence [59]. Apart from age-associated remodeling of the vascular wall, which includes luminal enlargement, intimal and medial thickening, and increased vascular stiffness, endothelial function declines with age [60].
In the review by Puca et al., the authors report endothelial dysfunction appears to be the hallmark of vascular damage in advancing age [61]. Many functions of the vascular endothelium are modulated by nitric oxide which is able to: induce smooth muscle relaxation; inhibit platelet aggregation and leukocyte adhesion to endothelial cells; and preserve endothelial progenitor cell function. Oxidative stress reduces nitric oxide bioavailability, contributing to endothelial dysfunction and vascular aging [61]. How vessel wall integrity and repair processes change with aging is a fundamental question in vascular biology that is under active investigation.
WHEN DOES THE RISK OF COMPLICATIONS FROM NOVEL ORAL ANTICOAGULANT THERAPY OUTWEIGH ITS BENEFIT?
Overestimation of the risk of bleeding by physicians is a key barrier to oral anticoagulant prescription, particularly among elderly patients [62]. The concept of “net clinical benefit” has been used to quantify the balance between risk of ischemic stroke and risk of ICH for oral anticoagulant therapy in the setting of non-valvular AF. Modeling of NOAC has shown that each agent offers net clinical benefit over warfarin therapy in patients with a CHADS2 score ≥1 or CHA2DS2-VASc score ≥2, regardless of bleeding risk [63]. By definition, net clinical benefit excludes major gastrointestinal and nuisance bleeding, both of which may be important aspects of older adults’ quality of life. Nonetheless, the analysis incorporated CHADS2, CHA2DS2-VASc and HAS-BLED scoring systems, highlighting the inherent trade-off between thromboembolism and bleeding. Many risk factors for bleeding in older adults are also risk factors for stroke (e.g., prior stroke, kidney disease, malignancy), some of which are incorporated into these scoring systems. When older adults prioritize quality of life over longevity, the likelihood of surviving a severe stroke must be considered in relation to the likelihood of major gastrointestinal and/or nuisance bleeding. Further investigation, development and validation of scoring systems that take all factors into consideration are needed.
PRACTICAL ISSUES: NOAC IN OLDER ADULTS
NOAC have been shown to be non-inferior to warfarin for stroke prevention in non-valvular AF, and significantly decrease ICH compared to warfarin. In older adults, decreased hemorrhagic stroke risk is particularly important. Yet in this $10 billion industry, adoption of NOAC in younger and older adults alike has been more modest than anticipated. Some have suggested that a lack of reversibility is, at least in part, responsible. Additional concerns among older adults may also play a part.
Age, creatinine and weight may poorly reflect underlying practical concerns such as comorbidity, renal insufficiency and frailty. Although no universally accepted definition of frailty exists, most agree that the “frailty phenotype” is highlighted by decreased physiologic reserve and diminished resilience. The pathophysiology of frailty appears to be rooted in the dysregulation of immune, metabolic, vascular, inflammatory and neuroendocrine systems. Increased inflammatory markers (CRP, IL-6, TNF-α) have been associated with frailty in numerous studies [64–66].
No studies of frailty and NOAC have been published to date. One study of frailty and warfarin use underscored several practical limitations for the use of NOAC. Using the Edmonton Frail Scale, Perera et al. found that frail older inpatients with AF were significantly less likely to receive warfarin than non-frail patients upon hospital admission and discharge [67]. During hospitalization, the proportion of frail patients prescribed warfarin decreased by 10.7% and the proportion of non-frail patients prescribed warfarin increased by 6.3%. In this study, the association between frailty and prescription of warfarin was consistent across three distinct clinical services: geriatric medicine, general medicine and cardiology. The study was not powered to detect differences in outcomes between frail and non-frail patients, though the frail appeared more vulnerable to worse clinical outcomes with and without antithrombotic therapies.
Besides frailty, certain factors that limit use of warfarin may similarly restrict the use of NOAC in older adults: cognitive impairment; terminal illness; and patient values and preferences. In a systematic review by Pugh et al., fall risk was noted to be a disproportionate barrier to warfarin prescription [68]. Frequent falls should not deter one from considering a NOAC unless the risk of morbidity secondary to falls outweighs concern of stroke-related morbidity and mortality.
Table 2 offers a practical guide to prescribing NOAC. Given that rivaroxaban is approved for use in the prevention of venous thromboembolism, it could be the novel agent of choice for an older adult at high risk of venous thromboembolism (e.g., certain active malignancies) and non-valvular AF. Further, rivaroxaban was recently approved by the FDA for the treatment of deep vein thrombosis and pulmonary embolism; however, one must be cognizant of dosing differences if treating for both conditions.
Table 2:
Suggested NOAC by patient condition
| CONDITION | Suggested NOAC | RATIONALE |
|---|---|---|
| Kidney Disease: | ||
| Mild | rivaroxaban, apixaban (or dabigatran) | See discussion under ‘Dabigatran Etexilate’ |
| Moderate | rivaroxaban, apixaban | |
| Severe | none (use warfarin instead) | |
| History of: | ||
| Dyspepsia | rivaroxaban or apixaban | Dabigatran not well tolerated |
| Cerebrovascular disease | rivaroxaban, dabigatran | See discussion under ‘Practical Issues’ |
| Myocardial infarction (MI) | rivaroxaban, or possibly apixaban | MI rate numerically higher with dabigatran in RE- LY; no increase in fatal bleeding with rivaroxaban in recent ACS [76]; APPRAISE-2 terminatedearly secondary to increased bleeding with apixaban after acute coronary syndrome [77] |
| Mild cognitive impairment | case-by-case basis | See discussion under ‘Practical Issues’ |
| Frequent falls | dabigatran, rivaroxaban or apixaban | Unless risk of morbidity secondary to falls outweighs concern of stroke-related morbidity and mortality, NOACs suggested [68,9] |
| Remote major bleeding | apixaban, none | 31% risk reduction in major bleeding with apixaban vs. warfarin [51]; nonetheless, apixaban increases bleeding risk relative to placebo |
| Intracranial hemorrhage | None | NOACs decrease intracranial hemorrhage relative to warfarin, not placebo |
| Deep venous thrombosis | rivaroxaban | Rivaroxaban FDA approved for treatment of deep venous thrombosis (not AF dose) [78]; other agents under review in USA |
| Pulmonary embolism | rivaroxaban | Rivaroxaban FDA approved for treatment of pulmonary embolism (not AF dose) [78]; other agents under review in USA |
| Venous thromboembolism risk | Rivaroxaban (or apixaban) | Rivaroxaban FDA approved to reduce risk of recurrence of venous thromboembolism (not AF dose) [78]; other agents under review in USA |
| Ischemic stroke risk | rivaroxaban, dabigatran (or apixaban) | See discussion under ‘Practical Issues’ |
| Low body weight | rivaroxaban, apixaban | See ‘Dabigatran Etexilate’ and ‘Practical Issues’ sections; apixaban dose-adjusted for low body weight |
| Frail | rivaroxaban, apixaban | May be more prone to fluctuations in renal function affecting bleeding risk with dabigatran |
| Needs to use pill box | rivaroxaban, apixaban | Dabigatran cannot be placed in a pill box |
| Needs once daily dosing | rivaroxaban | Dabigatran and apixaban are dosed twice daily |
| Chronic NSAID use | reconsider chronic NSAID use before prescribing NOAC | Rivaroxban and apixaban not well-studied with chronic NSAID use; chronic NSAID use with dabigatran increases bleeding risk by 50% [70] |
Rivaroxaban may also be the best option for a NOAC in an older adult with mild cognitive impairment, given that it is the only agent studied as once daily dosing and can be placed in a pill box (unlike dabigatran). Apixaban and dabigatran are alternatives to consider in those who can reliably take medication twice daily. Assistance with dosing from family members and/or skilled care providers may be necessary regardless of agent selected. In older adults who are thin or frail, or with history of renal insufficiency, dabigatran should be used with extreme caution, and possibly even avoided; rivaroxaban and apixaban may be safer alternatives.
Given that ROCKET-AF included a high proportion of patients with history of prior stroke, rivaroxaban may be considered safe in patients with history of prior stroke. Preliminary ROCKET-AF sub-group analysis shows that those ≥ 75 years in ROCKET-AF had lower incidence of prior stroke compared to those ˂ 75 years (42% vs. 65% in patients ≥ 75 years vs. < 75 years, respectively), but that the incidence of ischemic stroke in ROCKET-AF was lower among those ≥ 75 years versus warfarin (see rivaroxaban discussion above) [48]. Regardless, dabigatran is the only NOAC that achieved an overall reduction in ischemic stroke compared to warfarin. Ischemic stroke was not a primary outcome in any of these trials, making it difficult to draw any firm conclusions.
Next, inflammatory osteoarthritis is common in older adults, and over-the-counter NSAIDs are a mainstay of therapy. While short-term perioperative use of NSAIDs has not been shown to increase the risk of bleeding when given concomitantly with dabigatran [69], the chronic use of NSAIDs increases the bleeding risk by 50% [70]. The safety of long-term use of concomitant NSAIDs with rivaroxaban or apixaban has not been studied.
Finally, dabigatran may be less well-tolerated in patients with history of dyspepsia. Unlike rivaroxaban and apixaban, dabigatran is acid sensitive and has a tartaric acid core to improve bioavailability. Higher rates of dyspepsia observed with dabigatran (versus warfarin) may be related to its formulation.
Even when one or more factors favor prescribing a NOAC to older adults, practical concerns may ultimately prevail in some patients. The cost of NOACs is a major consideration in the current healthcare environment. Presently, dabigatran and rivaroxaban cost out-of-pocket consumers approximately $3,500 yearly. At least half of 2010 Medicare beneficiaries were living below 200% of the federal poverty level (<$11,000 for an individual) [71], making affordability of NOAC largely dependent on insurance coverage.
As of September 2012, 63% of Medicare beneficiaries (or 32 million) were enrolled in Medicare Part D plans, with traditional plans charging 25% coinsurance to the initial coverage limit of ˂$3,000. Given that the 2013 Medicare Part D coverage gap spans $2,970-$6,955 of total drug costs, NOAC prescription alone would place most beneficiaries in “the gap”.
Under The Patient Protection and Affordable Care Act, pharmaceutical manufacturers began providing a 50% discount on brand name prescriptions filled in the Medicare Part D coverage gap in 2011; in addition beginning in 2013, federal subsidies will account for 25% of the brand name drug cost by 2020 [72].
Of the 30% of Part D plans offering gap coverage in 2013, approximately half of these will limit gap coverage to generics only. Among plans offering gap coverage for brand-name drugs, only 10–65% of medications on formulary will be eligible for some coverage (note: this is more than double 2012) [73]. Part D enrollment is highly concentrated, with five firms –UnitedHealth, Humana, CVS Caremark, Coventry Health Care, and Express Scripts– accounting for 59% of enrollees in 2012. Clearly, Medicare Part D NOAC payment structure could heavily influence prescriber patterns of these agents in the future.
CONCLUSION
Older adults have a high prevalence of AF, and they are more likely than young adults to experience AF- related stroke. In addition, older adults have a greater incidence of comorbid conditions, and both polypharmacy and adverse drug effects are more common. Despite their high and well-known risk, older adults receive thromboprophylaxis far less often than their younger counterparts. Lack of need for routine coagulation monitoring, few drug-drug interactions and excellent performance in phase 3 clinical trials favorably position NOACs for use in older adults with non-valvular AF. With careful recognition of risks and benefits of NOACs, older adults can receive thromboprophylaxis to a greater extent than in the past.
To optimize drug selection, older adults need be carefully assessed for likelihood of stroke and bleeding-related complications of NOAC. We believe that initial and at least quarterly evaluation of renal function and face-to-face assessment of comorbidities, concurrent medications, and signs or symptoms of bleeding is vital to quality patient care. Fall risk must be carefully assessed and minimized. Cognitive impairment should be assessed and treated whenever possible. The potential cost burden of NOAC in older adults warrants consideration on a case-by-case basis. Prescribing physicians may need to consult with other providers, pharmacists and healthcare administrators to coordinate care and assure safe prescribing of NOACs.
A commonsense approach to prescribing NOAC in older adults will ultimately provide clinicians and researchers with valuable experience that may be relied upon to guide future therapeutic decision-making. Until clinical registries can provide us with experiential data, providers must rely on understanding the available drugs, their pharmacologic properties and how each was studied in phase 3 clinical trials. NOACs represent an advance in the management of older adults with AF when prescribed to the right patient at the right dose in one whom the benefit of treatment outweighs potential risk.
DISCLOSURES / ACKNOWLEDGEMENTS
Dr. Becker receives research support from Johnson & Johnson and AstraZeneca. He serves as a science advisory to Bayer, Merck, Boehringer-Ingelheim and Daiichi-Sankyo. The project described was supported by Grant Number T32GM086330 from the National Institute of General Medical Sciences. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of General Medical Sciences or the National Institutes of Health.
References
- 1.World Health Organization Definition of an Older or Elderly Person. http://www.who.int/healthinfo/survey/ageingdefnolder/en/index.html. Accessed December 5, 2012
- 2.Brubaker TH, Powers EA (1976) The stereotype of “old.” A review and alternative approach. J Gerontol 31 (4):441–447 [DOI] [PubMed] [Google Scholar]
- 3.Freund AM, Smith J (1999) Content and function of the self-definition in old and very old age. J Gerontol B Psychol Sci Soc Sci 54 (1):P55–67 [DOI] [PubMed] [Google Scholar]
- 4.Souare G, Lloyd L (2008) The History and Demography of Aging in the United States In: Loue S, Sajatovic M (eds) Encyclopedia of Aging and Public Health [Google Scholar]
- 5.Centers for Disease Control and Prevention, Merck Institute on Aging & Health, The Gerontological Society of America The State of Aging and Health in America; 2004. http://www.cdc.gov/aging/pdf/state_of_aging_and_health_in_america_2004.pdf. December 5, 2012] [Google Scholar]
- 6.Cerreta F, Eichler HG, Rasi G (2012) Drug policy for an aging population--the European Medicines Agency’s geriatric medicines strategy. N Engl J Med 367 (21):1972–1974. doi: 10.1056/NEJMp1209034 [DOI] [PubMed] [Google Scholar]
- 7.Heeringa J, van der Kuip DA, Hofman A, Kors JA, van Herpen G, Stricker BH, Stijnen T, Lip GY, Witteman JC (2006) Prevalence, incidence and lifetime risk of atrial fibrillation: the Rotterdam study. Eur Heart J 27 (8):949–953. doi: 10.1093/eurheartj/ehi825 [DOI] [PubMed] [Google Scholar]
- 8.Wolf PA, Abbott RD, Kannel WB (1987) Atrial fibrillation: a major contributor to stroke in the elderly. The Framingham Study. Arch Intern Med 147 (9):1561–1564 [PubMed] [Google Scholar]
- 9.Sellers MB, Newby LK (2011) Atrial fibrillation, anticoagulation, fall risk, and outcomes in elderly patients. Am Heart J 161 (2):241–246. doi: 10.1016/j.ahj.2010.11.002 [DOI] [PubMed] [Google Scholar]
- 10.Hart RG, Pearce LA, Aguilar MI (2007) Meta-analysis: antithrombotic therapy to prevent stroke in patients who have nonvalvular atrial fibrillation. Ann Intern Med 146 (12):857–867 [DOI] [PubMed] [Google Scholar]
- 11.Go AS, Hylek EM, Borowsky LH, Phillips KA, Selby JV, Singer DE (1999) Warfarin use among ambulatory patients with nonvalvular atrial fibrillation: the anticoagulation and risk factors in atrial fibrillation (ATRIA) study. Ann Intern Med 131 (12):927–934 [DOI] [PubMed] [Google Scholar]
- 12.McLean AJ, Le Couteur DG (2004) Aging biology and geriatric clinical pharmacology. Pharmacol Rev 56 (2):163–184. doi: 10.1124/pr.56.2.4 [DOI] [PubMed] [Google Scholar]
- 13.Klotz U (2009) Pharmacokinetics and drug metabolism in the elderly. Drug Metab Rev 41 (2):67–76. doi: 10.1080/03602530902722679 [DOI] [PubMed] [Google Scholar]
- 14.Wiggins JE (2012) Aging in the glomerulus. J Gerontol A Biol Sci Med Sci 67 (12):1358–1364. doi: 10.1093/gerona/gls157 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Aymanns C, Keller F, Maus S, Hartmann B, Czock D (2010) Review on pharmacokinetics and pharmacodynamics and the aging kidney. Clinical journal of the American Society of Nephrology : CJASN 5 (2):314–327. doi: 10.2215/cjn.03960609 [DOI] [PubMed] [Google Scholar]
- 16.Lindeman RD, Tobin J, Shock NW (1985) Longitudinal studies on the rate of decline in renal function with age. J Am Geriatr Soc 33 (4):278–285 [DOI] [PubMed] [Google Scholar]
- 17.Schwartz JB (2007) The current state of knowledge on age, sex, and their interactions on clinical pharmacology. Clin Pharmacol Ther 82 (1):87–96. doi: 10.1038/sj.clpt.6100226 [DOI] [PubMed] [Google Scholar]
- 18.Turnheim K (2003) When drug therapy gets old: pharmacokinetics and pharmacodynamics in the elderly. Exp Gerontol 38 (8):843–853 [DOI] [PubMed] [Google Scholar]
- 19.Gomez A, Ingelman-Sundberg M (2009) Pharmacoepigenetics: its role in interindividual differences in drug response. Clin Pharmacol Ther 85 (4):426–430. doi: 10.1038/clpt.2009.2 [DOI] [PubMed] [Google Scholar]
- 20.McLachlan AJ, Hilmer SN, Le Couteur DG (2009) Variability in response to medicines in older people: phenotypic and genotypic factors. Clin Pharmacol Ther 85 (4):431–433. doi: 10.1038/clpt.2009.1 [DOI] [PubMed] [Google Scholar]
- 21.Noble RE (2003) Drug therapy in the elderly. Metabolism 52 (10 Suppl 2):27–30 [DOI] [PubMed] [Google Scholar]
- 22.Carbonin P, Pahor M, Bernabei R, Sgadari A (1991) Is age an independent risk factor of adverse drug reactions in hospitalized medical patients? J Am Geriatr Soc 39 (11):1093–1099 [DOI] [PubMed] [Google Scholar]
- 23.Walker J, Wynne H (1994) Review: the frequency and severity of adverse drug reactions in elderly people. Age Ageing 23 (3):255–259 [DOI] [PubMed] [Google Scholar]
- 24.Gnjidic D, Hilmer SN (2012) Potential contribution of medications to frailty. J Am Geriatr Soc 60 (2):401. doi: 10.1111/j.1532-5415.2011.03810.x [DOI] [PubMed] [Google Scholar]
- 25.Gnjidic D, Hilmer SN, Blyth FM, Naganathan V, Cumming RG, Handelsman DJ, McLachlan AJ, Abernethy DR, Banks E, Le Couteur DG (2012) High-risk prescribing and incidence of frailty among older community-dwelling men. Clin Pharmacol Ther 91 (3):521–528. doi: 10.1038/clpt.2011.258 [DOI] [PubMed] [Google Scholar]
- 26.Wynne HA, Yelland C, Cope LH, Boddy A, Woodhouse KW, Bateman DN (1993) The association of age and frailty with the pharmacokinetics and pharmacodynamics of metoclopramide. Age Ageing 22 (5):354–359 [DOI] [PubMed] [Google Scholar]
- 27.McLachlan AJ, Pont LG (2012) Drug metabolism in older people--a key consideration in achieving optimal outcomes with medicines. J Gerontol A Biol Sci Med Sci 67 (2):175–180. doi: 10.1093/gerona/glr118 [DOI] [PubMed] [Google Scholar]
- 28.Franchini M (2006) Hemostasis and aging. Crit Rev Oncol Hematol 60 (2):144–151. doi: 10.1016/j.critrevonc.2006.06.004 [DOI] [PubMed] [Google Scholar]
- 29.Hauel NH, Nar H, Priepke H, Ries U, Stassen JM, Wienen W (2002) Structure-based design of novel potent nonpeptide thrombin inhibitors. J Med Chem 45 (9):1757–1766 [DOI] [PubMed] [Google Scholar]
- 30.Connolly SJ, Ezekowitz MD, Yusuf S, Eikelboom J, Oldgren J, Parekh A, Pogue J, Reilly PA, Themeles E, Varrone J, Wang S, Alings M, Xavier D, Zhu J, Diaz R, Lewis BS, Darius H, Diener HC, Joyner CD, Wallentin L (2009) Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 361 (12):1139–1151. doi: 10.1056/NEJMoa0905561 [DOI] [PubMed] [Google Scholar]
- 31.Ezekowitz MD, Connolly S, Parekh A, Reilly PA, Varrone J, Wang S, Oldgren J, Themeles E, Wallentin L, Yusuf S (2009) Rationale and design of RE-LY: randomized evaluation of long-term anticoagulant therapy, warfarin, compared with dabigatran. Am Heart J 157 (5):805–810, 810 e801–802. doi: 10.1016/j.ahj.2009.02.005 [DOI] [PubMed] [Google Scholar]
- 32.Beasley BN, Unger EF, Temple R (2011) Anticoagulant options--why the FDA approved a higher but not a lower dose of dabigatran. N Engl J Med 364 (19):1788–1790. doi: 10.1056/NEJMp1103050 [DOI] [PubMed] [Google Scholar]
- 33.Eikelboom JW, Wallentin L, Connolly SJ, Ezekowitz M, Healey JS, Oldgren J, Yang S, Alings M, Kaatz S, Hohnloser SH, Diener HC, Franzosi MG, Huber K, Reilly P, Varrone J, Yusuf S (2011) Risk of bleeding with 2 doses of dabigatran compared with warfarin in older and younger patients with atrial fibrillation: an analysis of the randomized evaluation of long-term anticoagulant therapy (RE-LY) trial. Circulation 123 (21):2363–2372. doi: 10.1161/CIRCULATIONAHA.110.004747 [DOI] [PubMed] [Google Scholar]
- 34.Stangier J, Rathgen K, Stahle H, Mazur D (2010) Influence of renal impairment on the pharmacokinetics and pharmacodynamics of oral dabigatran etexilate: an open-label, parallel-group, single-centre study. Clin Pharmacokinet 49 (4):259–268. doi: 10.2165/11318170-000000000-00000 [DOI] [PubMed] [Google Scholar]
- 35.Lehr T, Haertter S, Liesenfeld KH, Staab A, Clemens A, Reilly PA, Friedman J (2012) Dabigatran etexilate in atrial fibrillation patients with severe renal impairment: dose identification using pharmacokinetic modeling and simulation. J Clin Pharmacol 52 (9):1373–1378. doi: 10.1177/0091270011417716 [DOI] [PubMed] [Google Scholar]
- 36.Liesenfeld KH, Lehr T, Dansirikul C, Reilly PA, Connolly SJ, Ezekowitz MD, Yusuf S, Wallentin L, Haertter S, Staab A (2011) Population pharmacokinetic analysis of the oral thrombin inhibitor dabigatran etexilate in patients with non-valvular atrial fibrillation from the RE-LY trial. Journal of thrombosis and haemostasis : JTH 9 (11):2168–2175. doi: 10.1111/j.1538-7836.2011.04498.x [DOI] [PubMed] [Google Scholar]
- 37.Health Canada (2008) Summary Basis of Decision (SBD) PrPRADAX™ Dabigatran etexilate, 75 mg and 110 mg capsules Boehringer Ingelheim Canada Ltd.. http://www.hc-sc.gc.ca/dhp-mps/prodpharma/sbd-smd/drug-med/sbd_smd_2008_pradax_114887-eng.php.
- 38.Stangier J, Stahle H, Rathgen K, Fuhr R (2008) Pharmacokinetics and pharmacodynamics of the direct oral thrombin inhibitor dabigatran in healthy elderly subjects. Clin Pharmacokinet 47 (1):47–59 [DOI] [PubMed] [Google Scholar]
- 39.European Medicines Agency (2009) Annex I Summary of Product Characteristics (Pradaxa). http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/000829/WC500041059.pdf.
- 40.Eisenberg PR, Siegel JE, Abendschein DR, Miletich JP (1993) Importance of factor Xa in determining the procoagulant activity of whole-blood clots. J Clin Invest 91 (5):1877–1883. doi: 10.1172/jci116404 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Patel MR, Mahaffey KW, Garg J, Pan G, Singer DE, Hacke W, Breithardt G, Halperin JL, Hankey GJ, Piccini JP, Becker RC, Nessel CC, Paolini JF, Berkowitz SD, Fox KA, Califf RM (2011) Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med 365 (10):883–891. doi: 10.1056/NEJMoa1009638 [DOI] [PubMed] [Google Scholar]
- 42.Fox KA, Piccini JP, Wojdyla D, Becker RC, Halperin JL, Nessel CC, Paolini JF, Hankey GJ, Mahaffey KW, Patel MR, Singer DE, Califf RM (2011) Prevention of stroke and systemic embolism with rivaroxaban compared with warfarin in patients with non-valvular atrial fibrillation and moderate renal impairment. Eur Heart J 32 (19):2387–2394. doi: 10.1093/eurheartj/ehr342 [DOI] [PubMed] [Google Scholar]
- 43.Buller H, Deitchman D, Prins M, Segers A (2008) Efficacy and safety of the oral direct factor Xa inhibitor apixaban for symptomatic deep vein thrombosis. The Botticelli DVT dose-ranging study. Journal of thrombosis and haemostasis : JTH 6 (8):1313–1318. doi: 10.1111/j.1538-7836.2008.03054.x [DOI] [PubMed] [Google Scholar]
- 44.Kubitza D, Becka M, Mueck W, Zuehlsdorf M (2006) The effect of extreme age, and gender, on the pharmacology and tolerability of rivaroxaban - An oral, direct factor Xa inhibitor. Blood 108 (11):271A–272A [Google Scholar]
- 45.Kubitza D, Becka M, Roth A, Mueck W (2008) Dose-escalation study of the pharmacokinetics and pharmacodynamics of rivaroxaban in healthy elderly subjects. Curr Med Res Opin 24 (10):2757–2765. doi: 10.1185/03007990802361499 [DOI] [PubMed] [Google Scholar]
- 46.Mueck W, Eriksson BI, Bauer KA, Borris L, Dahl OE, Fisher WD, Gent M, Haas S, Huisman MV, Kakkar AK, Kalebo P, Kwong LM, Misselwitz F, Turpie AG (2008) Population pharmacokinetics and pharmacodynamics of rivaroxaban--an oral, direct factor Xa inhibitor--in patients undergoing major orthopaedic surgery. Clin Pharmacokinet 47 (3):203–216 [DOI] [PubMed] [Google Scholar]
- 47.Grillo JA, Zhao P, Bullock J, Booth BP, Lu M, Robie-Suh K, Berglund EG, Pang KS, Rahman A, Zhang L, Lesko LJ, Huang SM (2012) Utility of a physiologically-based pharmacokinetic (PBPK) modeling approach to quantitatively predict a complex drug-drug-disease interaction scenario for rivaroxaban during the drug review process: implications for clinical practice. Biopharm Drug Dispos 33 (2):99–110. doi: 10.1002/bdd.1771 [DOI] [PubMed] [Google Scholar]
- 48.Halperin JL, Hankey GJ, Wojdyla D, Piccini JP, Lokhnygina Y, Patel MR, Breithardt G, Singer DE, Becker RC, Hacke W, Paolini JF, Nessel CC, Mahaffey KW, Califf RM, Fox KA, ROCKET AF Steering Committee and Investigators (2012) Efficacy and Safety of Rivaroxaban Compared with Warfarin Among Elderly Patients with Nonvalvular Atrial Fibrillation in the ROCKET-AF Trial. PowerPoint Presentation [Google Scholar]
- 49.Weinz C, Buetehorn U, Daehler HP, Kohlsdorfer C, Pleiss U, Sandmann S, Schlemmer KH, Schwarz T, Steinke W (2005) Pharmacokinetics of BAY 59–7939--an oral, direct Factor Xa inhibitor--in rats and dogs. Xenobiotica 35 (9):891–910. doi: 10.1080/00498250500250493 [DOI] [PubMed] [Google Scholar]
- 50.Hankey GJ, Patel MR, Stevens SR, Becker RC, Breithardt G, Carolei A, Diener HC, Donnan GA, Halperin JL, Mahaffey KW, Mas JL, Massaro A, Norrving B, Nessel CC, Paolini JF, Roine RO, Singer DE, Wong L, Califf RM, Fox KA, Hacke W, Investigators RASC (2012) Rivaroxaban compared with warfarin in patients with atrial fibrillation and previous stroke or transient ischaemic attack: a subgroup analysis of ROCKET AF. Lancet neurology 11 (4):315–322. doi: 10.1016/S1474-4422(12)70042-X [DOI] [PubMed] [Google Scholar]
- 51.Granger CB, Alexander JH, McMurray JJ, Lopes RD, Hylek EM, Hanna M, Al-Khalidi HR, Ansell J, Atar D, Avezum A, Bahit MC, Diaz R, Easton JD, Ezekowitz JA, Flaker G, Garcia D, Geraldes M, Gersh BJ, Golitsyn S, Goto S, Hermosillo AG, Hohnloser SH, Horowitz J, Mohan P, Jansky P, Lewis BS, Lopez-Sendon JL, Pais P, Parkhomenko A, Verheugt FW, Zhu J, Wallentin L, Committees A, Investigators (2011) Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med 365 (11):981–992. doi: 10.1056/NEJMoa1107039 [DOI] [PubMed] [Google Scholar]
- 52.Connolly SJ, Eikelboom J, Joyner C, Diener HC, Hart R, Golitsyn S, Flaker G, Avezum A, Hohnloser SH, Diaz R, Talajic M, Zhu J, Pais P, Budaj A, Parkhomenko A, Jansky P, Commerford P, Tan RS, Sim KH, Lewis BS, Van Mieghem W, Lip GY, Kim JH, Lanas-Zanetti F, Gonzalez-Hermosillo A, Dans AL, Munawar M, O’Donnell M, Lawrence J, Lewis G, Afzal R, Yusuf S (2011) Apixaban in patients with atrial fibrillation. N Engl J Med 364 (9):806–817. doi: 10.1056/NEJMoa1007432 [DOI] [PubMed] [Google Scholar]
- 53.Lassen MR, Davidson BL, Gallus A, Pineo G, Ansell J, Deitchman D (2007) The efficacy and safety of apixaban, an oral, direct factor Xa inhibitor, as thromboprophylaxis in patients following total knee replacement. Journal of thrombosis and haemostasis : JTH 5 (12):2368–2375. doi: 10.1111/j.1538-7836.2007.02764.x [DOI] [PubMed] [Google Scholar]
- 54.Hohnloser SH, Hijazi Z, Thomas L, Alexander JH, Amerena J, Hanna M, Keltai M, Lanas F, Lopes RD, Lopez-Sendon J, Granger CB, Wallentin L (2012) Efficacy of apixaban when compared with warfarin in relation to renal function in patients with atrial fibrillation: insights from the ARISTOTLE trial. Eur Heart J. doi: 10.1093/eurheartj/ehs274 [DOI] [PubMed] [Google Scholar]
- 55.Pedone C, Semeraro R, Chiurco D, D’Andria F, Gigante M, Coppola A, Corsonello A, Antonelli-Incalzi R (2008) Reliability of equations to estimate glomerular filtration rate in the very old. Aging clinical and experimental research 20 (6):496–502 [DOI] [PubMed] [Google Scholar]
- 56.Baker WL, Phung OJ (2012) Systematic Review and Adjusted Indirect Comparison Meta-Analysis of Oral Anticoagulants in Atrial Fibrillation. Circulation Cardiovascular quality and outcomes. doi: 10.1161/circoutcomes.112.966572 [DOI] [PubMed] [Google Scholar]
- 57.Wang L, Zhang D, Raghavan N, Yao M, Ma L, Frost CE, Maxwell BD, Chen SY, He K, Goosen TC, Humphreys WG, Grossman SJ (2010) In vitro assessment of metabolic drug-drug interaction potential of apixaban through cytochrome P450 phenotyping, inhibition, and induction studies. Drug Metab Dispos 38 (3):448–458. doi: 10.1124/dmd.109.029694 [DOI] [PubMed] [Google Scholar]
- 58.Furie KL, Goldstein LB, Alvers GW, Khateri P, Neyens R, Turakhia MP, Turan TN, Wood KA, on behalf of the American Heart Association Stroke Council, Council on Quality of Care and Outcomes Research, Council on Cardiovascular Nursing, Council on Clinical Cardiology, Council on Peripheral Vascular Disease, American Heart Association, American Stroke Association (2012) Oral Antithrombotic Agents for the Prevention of Stroke in Non-valvular Atrial Fibrillation; a Science Advisory for Healthcare Professionals from the American Heart Association / American Stroke Association. Stroke. doi: 10.1161/STR.0B013e318266722a [DOI] [PubMed] [Google Scholar]
- 59.Gharacholou SM, Becker RC (2009) Hemostasis and thrombosis in older adults. J Thromb Thrombolysis 27 (2):249–251. doi: 10.1007/s11239-009-0308-4 [DOI] [PubMed] [Google Scholar]
- 60.Yildiz O (2007) Vascular smooth muscle and endothelial functions in aging. Ann N Y Acad Sci 1100:353–360. doi: 10.1196/annals.1395.038 [DOI] [PubMed] [Google Scholar]
- 61.Puca AA, Carrizzo A, Ferrario A, Villa F, Vecchione C (2012) Endothelial nitric oxide synthase, vascular integrity and human exceptional longevity. Immunity & ageing : I & A 9 (1):26. doi: 10.1186/1742-4933-9-26 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Lane DA, Lip GY (2012) Use of the CHA(2)DS(2)-VASc and HAS-BLED scores to aid decision making for thromboprophylaxis in nonvalvular atrial fibrillation. Circulation 126 (7):860–865. doi: 10.1161/CIRCULATIONAHA.111.060061 [DOI] [PubMed] [Google Scholar]
- 63.Banerjee A, Lane DA, Torp-Pedersen C, Lip GY (2012) Net clinical benefit of new oral anticoagulants (dabigatran, rivaroxaban, apixaban) versus no treatment in a ‘real world’ atrial fibrillation population: a modelling analysis based on a nationwide cohort study. Thromb Haemost 107 (3):584–589. doi: 10.1160/TH11-11-0784 [DOI] [PubMed] [Google Scholar]
- 64.Cohen HJ, Harris T, Pieper CF (2003) Coagulation and activation of inflammatory pathways in the development of functional decline and mortality in the elderly. Am J Med 114 (3):180–187 [DOI] [PubMed] [Google Scholar]
- 65.Kanapuru B, Ershler WB (2009) Inflammation, coagulation, and the pathway to frailty. Am J Med 122 (7):605–613. doi: 10.1016/j.amjmed.2009.01.030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Reiner AP, Aragaki AK, Gray SL, Wactawski-Wende J, Cauley JA, Cochrane BB, Kooperberg CL, Woods NF, LaCroix AZ (2009) Inflammation and thrombosis biomarkers and incident frailty in postmenopausal women. Am J Med 122 (10):947–954. doi: 10.1016/j.amjmed.2009.04.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Perera V, Bajorek BV, Matthews S, Hilmer SN (2009) The impact of frailty on the utilisation of antithrombotic therapy in older patients with atrial fibrillation. Age Ageing 38 (2):156–162. doi: 10.1093/ageing/afn293 [DOI] [PubMed] [Google Scholar]
- 68.Pugh D, Pugh J, Mead GE (2011) Attitudes of physicians regarding anticoagulation for atrial fibrillation: a systematic review. Age Ageing 40 (6):675–683. doi: 10.1093/ageing/afr097 [DOI] [PubMed] [Google Scholar]
- 69.Friedman RJ, Kurth A, Clemens A, Noack H, Eriksson BI, Caprini JA (2012) Dabigatran etexilate and concomitant use of non-steroidal anti-inflammatory drugs or acetylsalicylic acid in patients undergoing total hip and total knee arthroplasty: no increased risk of bleeding. Thromb Haemost 108 (1):183–190. doi: 10.1160/th11-08-0589 [DOI] [PubMed] [Google Scholar]
- 70.Boehringer Ingelheim (2011) Dabigatran etexilate (Package Insert). Boehringer Ingelheim, Ridgefield, CT [Google Scholar]
- 71.The Henry J Kaiser Family Foundation (2012) Medicare. The Henry J. Kaiser Family Foundation; http://www.kff.org/medicare. [Google Scholar]
- 72.The Henry J Kaiser Family Foundation (2011) Summary of New Health Reform Law. The Henry J. Kaiser Family Foundation; http://www.kff.org/healthreform/8061.cfm. Accessed November 27 2012 [Google Scholar]
- 73.Hoadley J, Cubanski J, Hargrave E, Summer L, Huang J (2012) Medicare Part D: A First Look at Part D Plan Offerings in 2013. Kaiser Family Foundation; http://www.kff.org/medicare/upload/8375.pdf. December 14, 2012] [Google Scholar]
- 74.Manjunath G, Sarnak MJ, Levey AS (2001) Prediction equations to estimate glomerular filtration rate: an update. Curr Opin Nephrol Hypertens 10 (6):785–792 [DOI] [PubMed] [Google Scholar]
- 75.Cockcroft DW, Gault MH (1976) Prediction of creatinine clearance from serum creatinine. Nephron 16 (1):31–41 [DOI] [PubMed] [Google Scholar]
- 76.Mega JL, Braunwald E, Wiviott SD, Bassand JP, Bhatt DL, Bode C, Burton P, Cohen M, Cook-Bruns N, Fox KA, Goto S, Murphy SA, Plotnikov AN, Schneider D, Sun X, Verheugt FW, Gibson CM (2012) Rivaroxaban in patients with a recent acute coronary syndrome. N Engl J Med 366 (1):9–19. doi: 10.1056/NEJMoa1112277 [DOI] [PubMed] [Google Scholar]
- 77.Alexander JH, Lopes RD, James S, Kilaru R, He Y, Mohan P, Bhatt DL, Goodman S, Verheugt FW, Flather M, Huber K, Liaw D, Husted SE, Lopez-Sendon J, De Caterina R, Jansky P, Darius H, Vinereanu D, Cornel JH, Cools F, Atar D, Leiva-Pons JL, Keltai M, Ogawa H, Pais P, Parkhomenko A, Ruzyllo W, Diaz R, White H, Ruda M, Geraldes M, Lawrence J, Harrington RA, Wallentin L (2011) Apixaban with antiplatelet therapy after acute coronary syndrome. N Engl J Med 365 (8):699–708. doi: 10.1056/NEJMoa1105819 [DOI] [PubMed] [Google Scholar]
- 78.FDA expands use of Xarelto to treat, reduce recurrence of blood clots. Food and Drug Administration. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm326654.htm. Accessed December 31, 2012 2012