Abstract
OBJECTIVE
To characterize use of uterine tamponade and interventional radiology procedures.
METHODS
This retrospective study analyzed uterine tamponade and interventional radiology procedures in a large administrative database. The primary outcomes were temporal trends in these procedures (i) during deliveries, (ii) by hospital volume, and (iii) prior to hysterectomy for uterine atony or delayed postpartum hemorrhage. Three three-year periods were analyzed: 2006–2008, 2009–2011, and 2012–2014. Risk for morbidity in the setting of hysterectomy with uterine tamponade and interventional radiology procedures as the primary exposures was additionally analyzed in adjusted models.
RESULTS
The study included 5,383,486 deliveries which involved 6,675 uterine tamponade procedures, 1,199 interventional radiology procedures, and 1,937 hysterectomies. Interventional radiology procedures increased from 16.4 to 25.7 per 100,000 delivery hospitalizations from 2006–2008 to 2012–2014 (p<0.01) while uterine tamponade increased from 86.3 to 158.1 (p<0.01). Interventional radiology procedures use was highest (45.0 per 100,000 deliveries, 95% CI 41.0–48.9) in the highest and lowest (8.9 per 100,000, 95% CI 7.1–10.7) in the lowest volume quintile. Uterine tamponade procedures were most common in the 4th (209.8 per 100,000, 95% CI 201.1–218.5) and lowest in the 3rd quintile (59.8 per 100,000, 95% CI 55.1–64.4). Interventional radiology procedures occurred prior to 3.3% of hysterectomies from 2006–2008 compared to 6.3% from 2012–2014 (p<0.05) while uterine tamponade procedures increased from 3.6% to 20.1% (p<0.01). Adjusted risks for morbidity in the setting of uterine tamponade and interventional radiology prior to hysterectomy were significantly higher (adjusted risk ratio [aRR] 1.63, 95% CI 1.47–1.81 and aRR 1.75 95% CI 1.51–2.03, respectively) compared to when these procedures were not performed.
CONCLUSION
This analysis found that uterine tamponade and interventional radiology procedures became increasingly common over the study period, are used across obstetric volume settings, and in the setting of hysterectomy may be associated with increased risk for morbidity, although this relationship is not necessarily causal.
Précis
Uterine tamponade and interventional radiology procedures were used increasingly during delivery hospitalizations over the study period.
INTRODUCTION
Hemorrhage remains a leading cause of severe maternal morbidity and mortality in the United States and is one of the most preventable causes of maternal death.1–5 Risk for postpartum hemorrhage (PPH) associated with severe morbidity such as transfusion or coagulopathy may be rising.4,6 Causes of postpartum hemorrhage include uterine atony, retained placenta, placental abnormalities, and trauma. Administration of uterotonics represents first-line treatment of excessive bleeding secondary to uterine atony.3,7 For women with bleeding refractory to uterotonics subsequent management options include: (i) uterine tamponade with devices such as the Bakri balloon, (ii) interventional radiology procedures including uterine artery embolization, and (iii) conservative surgical procedures such as the B-Lynch compression suture.7–12 For patients with acute, massive postpartum bleeding or persistent bleeding after other treatment options have failed, peripartum hysterectomy may be a required and life-saving intervention.13,14
While uterine tamponade and interventional radiology procedures may avert some cases of hysterectomy and a number of studies support favorable outcomes, large population data on use of these interventions is limited particularly in relation to hospital delivery volume.15–20 It is unclear to what degree uterine tamponade and interventional radiology procedures are being performed in hospitals with lower obstetric volumes that may have lesser resources which may affect outcomes. Use and characterization of uterine tamponade and interventional radiology procedures may be important in both in understanding overall practice patterns in PPH management and in quantifying complicated PPH requiring more intensive interventions.
Given that better understanding of these practice patterns and outcomes may be important in designing comparative effectiveness research and informing clinical guidelines and best practices, the purpose of this study was to characterize trends and outcomes related to use of uterine tamponade and interventional radiology procedures.
METHODS
This retrospective repeated cross sectional study21 used the 2006 to 2014 Perspective database, maintained by Premier Incorporated (Charlotte, NC). Cross sectional data from successive years of the database was analyzed; in contrast to retrospective cohort studies which follow patients longitudinally through time, repeated cross sectional studies evaluate multiple iterations of cross sectional data of similar populations over time.21 Perspective includes billing, device, procedure, and pharmacy data from over 600 hospitals across the United States. Hospitals in the database report 100% of hospitalizations at participating hospitals during a given year. Complete patient billing, hospital cost, and coding histories from hospitalizations are contained in the database. Ninety-five validation and quality assurance checks are performed prior to data being used for research. 22 Since 2011, over 6 million discharges annually have been captured by the Perspective database, representing approximately 15% of inpatient hospital stays.23 Upon receiving data from participating hospitals, Premier undertakes an extensive 7-part data validation and correction process that includes more than 95 quality assurance checks prior to being used for research.22 After validations are complete, the data are moved to the Perspective data warehouse to populate and maintain the databases for health services research.24 Because Perspective includes drug and device use, it provides additional information on postpartum hemorrhage management compared to discharge diagnoses alone. Perspective has previously been used in prior analyses of management of postpartum hemorrhage.25,26 Given that the data are de-identified the analysis was deemed exempt by the Columbia University Institutional Review Board.
The three primary objectives of this study were to characterize temporal trends in use of uterine tamponade and interventional radiology (i) in the general obstetric population by mode of delivery, (ii) by hospital delivery volume, and (iii) prior to hysterectomy for uterine atony or delayed postpartum hemorrhage (subsequently defined in this manuscript as PPH). Temporal trends for three three-year periods were analyzed: 2006–2008, 2009–2011, and 2012–2014. Secondary outcomes included determining: (i) risk for morbidity (as defined by a morbidity composite) in the setting of hysterectomy for PPH with interventional radiology and uterine tamponade interventions as primary exposures, (ii) use of uterine tamponade and interventional radiology procedures prior to hysterectomy for PPH by hospital delivery volume, and (iii) use of uterotonics including misoprostol, carboprost, and methylergonovine prior to peripartum hysterectomy for PPH. Uterotonics were queried in addition to uterine tamponade and interventional radiology procedures to determine trends in a baseline intervention for PPH. As an ancillary analysis, risks for (i) hysterectomy, and (ii) transfusion were determined by year individually for women undergoing uterine tamponade and interventional radiology procedures.
The morbidity composite was based on criteria from the Centers for Disease Control and Prevention (CDC).27 CDC severe maternal morbidity criteria includes 18 diagnoses identified based on International Classification of Diseases codes. Hysterectomy was removed from the composite because it was an inclusion criterium for this analysis. Because the most common diagnosis in the composite is transfusion and transfusion is much less likely to be life threatening or result in long-term sequelae than other conditions, this diagnosis was also removed from the composite leaving 16 diagnoses remaining: acute myocardial infarction, aneurysm, acute renal failure, adult respiratory distress syndrome, amniotic fluid embolism, cardiac arrest or ventricular fibrillation, disseminated intravascular coagulation, eclampsia, heart failure or arrest during surgery or procedure, puerperal cerebrovascular disorders, pulmonary edema or acute heart failure, severe anesthesia complications, shock, sickle cell disease with crisis, air and thrombotic embolism, temporary tracheostomy, and ventilation.
Inclusion criteria for this analysis included women 15 to 54 years of age who underwent a delivery hospitalization from January 2006 through December 2014. Delivery hospitalizations were identified using International Classification of Diseases, Ninth Edition, Clinical Modification (ICD-9-CM) diagnosis codes 650 and V27.x which ascertain >95% of delivery hospitalizations.28 The transition to ICD-10-CM codes in the Perspective database occurred after the first quarter of 2015; for this reason data from 2015 on was not analyzed. The presence of uterine tamponade was determined by querying patient billing files for uterine balloons, specific tamponade devices (Bakri balloon, Sengstaken-Blakemore tube, Rusch balloon) and for ICD-9-CM procedure code 75.8 during delivery hospitalizations. Interventional radiology procedures were determined by querying patient billing files for embolization procedures and ICD-9-CM procedure codes (68.24, 68.25, 39.79, 99.29) during delivery hospitalizations.
Obstetric volume was categorized by creating five roughly equal quintiles in terms of overall number of delivery hospitalizations with hospitals in the lowest quintile having the lowest annual delivery volumes and the hospitals in the highest quintile having the highest annual delivery volumes. Hospitals contributing less than a full year of data were excluded. Mode of delivery was classified as vaginal, cesarean, or repeat cesarean based on ICD-9-CM diagnosis (654.2x, 669.70, 669.71) and procedure (74.0, 74.1, 74.2, 74.4, 74.9, 74.99) codes. Peripartum hysterectomy cases were identified by ICD-9- CM procedure codes 68.3x, 68.4x, and 68.9 in the setting of a delivery hospitalization (ICD-9-CM diagnosis codes v27.x and 650).29 Uterine atony and secondary PPH were identified based on ICD-9-CM diagnosis codes (666.1x and 666.2x, respectively).4,6,30 To determine use of uterotonics we queried patient drug files for methylergonovine, carboprost, and misoprostol.25 Given that low-dose misoprostol (oral 50 μg or intravaginal 25 μg to a cumulative dose <250 μg) is used for induction, we considered misoprostol as a uterotonic only if the total dose exceeded 400 μg.25
Hospital characteristics included location (urban versus rural), teaching status (teaching versus nonteaching), geographic region (Midwest, Northeast, South, West), annualized delivery volume quintile, and hospital bed volume (small, medium, large). Demographic characteristics included maternal age at delivery (15–24, 25–34, 35–39, or 40–54 years), maternal race (white, black, other/unknown), marital status (married, single, unknown), year of delivery (2006 to 2014), and insurance status (commercial, Medicare, Medicaid, and uninsured or unknown).
Temporal trends were analyzed with the Cochrane-Armitage test for trend. Patient demographic and hospital factors were characterized based on receipt of interventional radiology and uterine tamponade procedures and compared with the chi-squared or Fisher’s exact test as appropriate. The associations between these demographic and hospital factors and outcomes are reported as risk ratios (RR) with 95% confidence intervals (CIs)
Adjusted log-linear models including the above demographic and hospital factors were performed for the following outcomes: (i) receipt of uterine tamponade and interventional radiology procedures among the entire obstetric population and (ii) severe morbidity among women undergoing hysterectomy for atony or secondary PPH. For these outcomes, women with both interventional radiology and uterine tamponade procedures were classified as having undergone interventional radiology procedures. For the adjusted models, results are reported as adjusted risk ratios (aRR) with 95% CIs as measures of effect; unadjusted risks are also reported for these outcomes. For the severe morbidity model, preeclampsia was added as a risk factor given its association with adverse outcomes. All analyses were performed with SAS 9.4 (SAS Institute, Cary, NC).
RESULTS
After eliminating 161,753 (2.9%) deliveries that occurred at hospitals contributing less than a year of data, 5,388,486 deliveries including 6,676 uterine tamponade procedures and 1,199 interventional radiology procedures from 500 hospitals were included in the analysis. Of 4,811 hysterectomies, 1,937 were associated with a diagnosis of PPH and were included in this analysis (Appendix 1, available online at http://links.lww.com/xxx). Of women who underwent interventional radiology procedures, 228 underwent hysterectomy (19.0%), while 289 women (4.3%) who underwent uterine tamponade procedures underwent hysterectomy. Overall there were 3,585,786 vaginal, 996,625 primary cesarean, and 806,075 repeat cesarean deliveries. Annualized delivery volume ranged from ≤1,309 deliveries for the first quintile (n=1,078,132), 1,310 to 2,187 deliveries for the second quintile (n=1,073,018), 2,200, to 2,931 deliveries for the third quintile (n=1,081,127), 2,943 to 4,000 deliveries for the fourth quintile (n=1,066,234), and 4,041 to 9,363 deliveries for the fifth quintile (n=1,089,975) (Table 1). Hysterectomy was more common at the highest versus lowest volume quintiles (110.3 versus 63.1 per 100,000 delivery hospitalizations, p<0.01); hysterectomy associated with atony or secondary PPH was also more common at highest versus lowest volume hospitals (40.6 versus 29.3 per 100,000 delivery hospitalizations, p<0.01) (Appendix 2, available online at http://links.lww.com/xxx).
Table 1.
Demographics of patients undergoing uterine tamponade or interventional radiology procedures
| Uterine tamponade (N=6,676) | Interventional radiology procedures (N=1,199) | Neither (N=5,380,611) | ||||
|---|---|---|---|---|---|---|
| N | % | N | % | N | % | |
| Year | ||||||
| 2006 | 371 | 5.6 | 95 | 7.9 | 532,901 | 9.9 |
| 2007 | 488 | 7.3 | 81 | 6.8 | 551,068 | 10.2 |
| 2008 | 518 | 7.8 | 90 | 7.5 | 541,057 | 10.1 |
| 2009 | 635 | 9.5 | 116 | 9.7 | 562,132 | 10.4 |
| 2010 | 748 | 11.2 | 133 | 11.1 | 573,452 | 10.7 |
| 2011 | 886 | 13.3 | 173 | 14.4 | 636,569 | 11.8 |
| 2012 | 1,054 | 15.8 | 181 | 15.1 | 699,073 | 13.0 |
| 2013 | 1,049 | 15.7 | 183 | 15.3 | 672,339 | 12.5 |
| 2014 | 927 | 13.9 | 147 | 12.3 | 612,020 | 11.4 |
| Age in years | ||||||
| 15–24 | 2,144 | 32.1 | 183 | 15.3 | 1733340 | 32.2 |
| 25–34 | 3,456 | 51.8 | 651 | 54.3 | 2854217 | 53.0 |
| 35–39 | 832 | 12.5 | 255 | 21.3 | 643,010 | 12.0 |
| 40–54 | 244 | 3.7 | 110 | 9.2 | 150,044 | 2.8 |
| Marital Status | ||||||
| Married | 3,307 | 49.5 | 609 | 50.8 | 2640047 | 49.1 |
| Single | 2,757 | 41.3 | 415 | 34.6 | 2093463 | 38.9 |
| Other/Unknown | 612 | 9.2 | 175 | 14.6 | 647,101 | 12.0 |
| Race | ||||||
| White | 2,380 | 35.7 | 515 | 43.0 | 2879422 | 53.5 |
| Black | 1,153 | 17.3 | 234 | 19.5 | 750,927 | 14.0 |
| Other/Unknown | 3,143 | 47.1 | 450 | 37.5 | 1750262 | 32.5 |
| Payer | ||||||
| Medicare | 44 | 0.7 | 14 | 1.2 | 40,389 | 0.8 |
| Medicaid | 2,558 | 38.3 | 416 | 34.7 | 2288269 | 42.5 |
| Commercial | 3,646 | 54.6 | 708 | 59.0 | 2739612 | 50.9 |
| Uninsured/Unknown | 428 | 6.4 | 61 | 5.1 | 312,341 | 5.8 |
| Rurality | ||||||
| Urban | 6,374 | 95.5 | 1,171 | 97.7 | 4866006 | 90.4 |
| Rural | 302 | 4.5 | 28 | 2.3 | 514,605 | 9.6 |
| Teaching hospital | ||||||
| No | 3,369 | 50.5 | 482 | 40.2 | 3224965 | 59.9 |
| Yes | 3,307 | 49.5 | 717 | 59.8 | 2155646 | 40.1 |
| Hospital bed size | ||||||
| <400 | 3,350 | 50.2 | 461 | 38.4 | 2987205 | 55.5 |
| 400–600 | 2,471 | 37.0 | 401 | 33.4 | 1397217 | 26.0 |
| >600 | 855 | 12.8 | 337 | 28.1 | 996,189 | 18.5 |
| Hospital region | ||||||
| Northeastern | 718 | 10.8 | 345 | 28.8 | 863,652 | 16.1 |
| Midwest | 2,037 | 30.5 | 178 | 14.8 | 934,195 | 17.4 |
| South | 2,599 | 38.9 | 405 | 33.8 | 2452365 | 45.6 |
| West | 1,322 | 19.8 | 271 | 22.6 | 1130399 | 21.0 |
| Hospital volume quintile | ||||||
| 1st | 1,550 | 23.2 | 96 | 8.0 | 1076486 | 20.0 |
| 2nd | 844 | 12.6 | 174 | 14.5 | 1072000 | 19.9 |
| 3rd | 606 | 9.1 | 231 | 19.3 | 1080290 | 20.1 |
| 4th | 2,196 | 32.9 | 208 | 17.3 | 1063830 | 19.8 |
| 5th | 1,480 | 22.2 | 490 | 40.9 | 1088005 | 20.2 |
Comparisons across demographic categories were all significant with p<0.01.
Interventional radiology procedures were more common among women 35 and older, black compared to white women, women with commercial insurance and at urban hospitals, teaching hospitals, and hospitals in the Northeast (p<0.01 for all) (Table 1). Uterine tamponade was more common among black women and women of other or unknown race compared to white women, urban compared to rural hospitals, and in the Midwest compared to other geographic regions (p<0.01). In the adjusted analyses, interventional radiology procedures were significantly more common among women 35 and older, black compared to white women, and in urban and teaching hospitals and those in the highest volume quintile (p<0.01 for all). In the adjusted analysis, uterine tamponade procedures were more common in urban and teaching hospitals, among black compared to white women, and in the Midwest compared to other regions (p<0.01 for all) (Appendix 3, available online at http://links.lww.com/xxx).
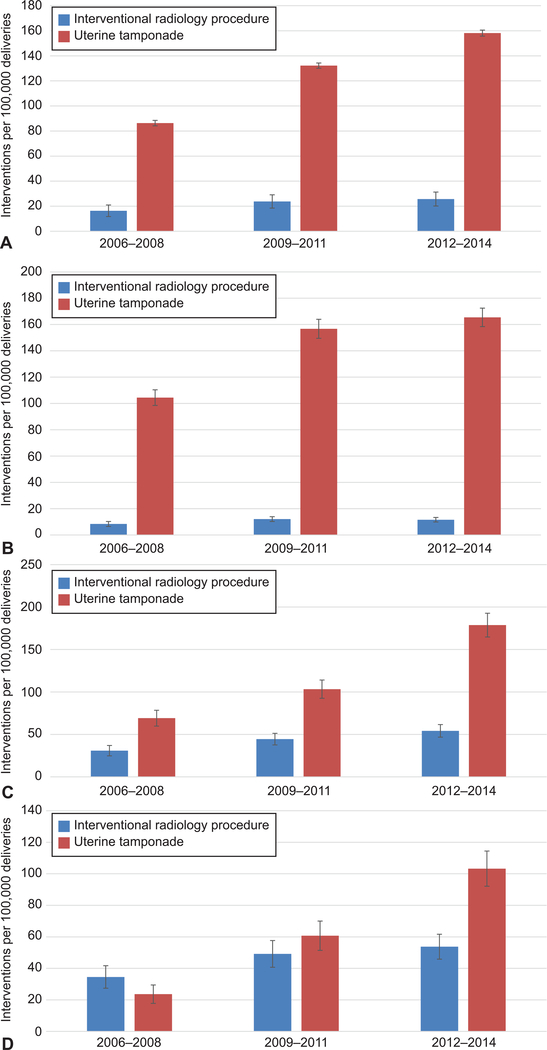
For the first primary objective, characterizing temporal trends, uterine tamponade and interventional radiology procedures both increased significantly over the study period. Interventional radiology procedures increased from 16.4 (95% CI 14.4, 18.3) to 23.8 (95% CI 21.5, 26.0) to 25.7 per 100,000 (95% CI 23.5, 27.9) delivery hospitalizations from 2006–2008 to 2009–2011 to 2012–2014 (p<0.05) (Figure 1A), while uterine tamponade increased from 86.3 (95% CI 81.8, 90.8) to 132.2 (95% CI 126.8, 137.5) to 158.1 per 100,000 (95% CI 152.6, 163.7) over the same period. In evaluating vaginal, primary cesarean, and repeat cesarean deliveries, (Figure 1B–1D) both interventions increased significantly for each type of delivery (p<0.05 for all analyses). From 2012–2014, uterine tamponade was most common during primary cesarean deliveries (178.7 per 100,000, 95% CI 164.8, 192.7) and least common during repeat cesarean deliveries (103.3 per 100,000, 95% CI 92.0, 114.6); for this period interventional radiology procedures were most common among repeat cesarean deliveries (53.7 per 100,000, 95% CI 45.6, 61.9) and least common among vaginal deliveries (11.6 per 100,000, 95% CI 9.8, 13.5).
Figure 1.
Temporal trends in uterine tamponade and embolization per 100,000 deliveries (A), per 100,000 vaginal deliveries (B), per 100,000 primary cesarean deliveries (C), and per 100,000 repeat cesarean deliveries (D) from 2006 through 2014. Trends for both interventions were statistically significant for all three types of delivery (P<.05). Error bars represent 95% CIs.
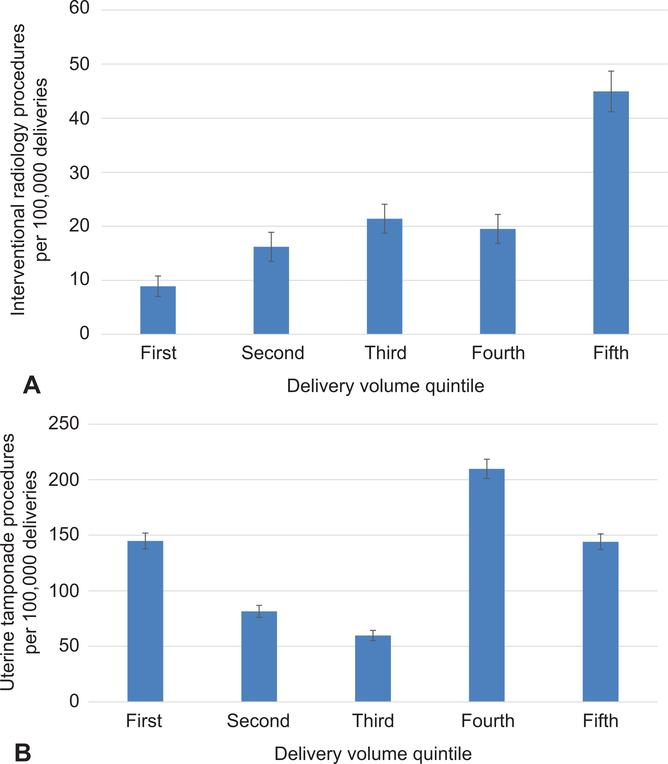
For the second primary objective, evaluation of uterine tamponade and interventional radiology procedures by hospital delivery volume quintile, use of interventional radiology procedures was highest (45.0 per 100,000, 95% CI 41.0–48.9) in the highest volume quintile, and lowest (8.9 per 100,000, 95% CI 7.1–10.7) in the lowest volume quintile; interventional radiology procedures were significantly more likely in the highest quintile than in any other quintile (p<0.01 for all comparisons) (Figure 2A). In comparison, uterine tamponade procedures were most common the 4th quintile (209.8 per 100,000, 95% CI 201.1–218.5), followed by the 1st volume quintile (144.9 per 100,000, 95% CI 137.7, 152.1) and lowest in the third quintile (59.8 per 100,000, 95% CI 55.1, 64.4 ) (Figure 2B); uterine tamponade procedures were significantly more common in the fourth quintile than any other quintile (p<0.01 for all comparisons).
Figure 2:
Likelihoods of interventional radiology (A) and uterine tamponade (B) procedures by hospital volume quintile. Each quintile accounts for approximately 20% of overall deliveries, with the first quintile composed of the lowest volume hospitals and the fifth quintiles composed of the highest volume hospitals (P<.01 for comparisons across both groups). Error bars represent 95% CI.
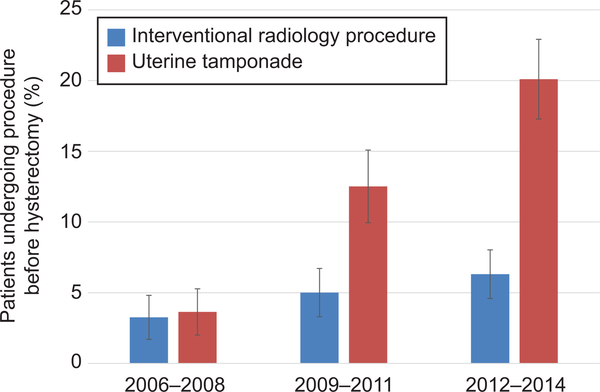
For the third primary objective, analysis of temporal trends in use of uterine tamponade and interventional radiology procedures prior to peripartum hysterectomy for PPH, both types of interventions increased temporally (Figure 3); interventional radiology occurred prior to 3.3% of hysterectomies from 2006 to 2008 compared to 6.3% in from 2012 to 2014 (p<0.05 for trend). Over this period, uterine tamponade procedures increased from 3.6% of hysterectomies to 20.1% (p<0.01 for trend).
Figure 3.
Temporal trends proportion of patients receiving uterine tamponade and embolization before peripartum hysterectomy for atony or delayed postpartum hemorrhage. These temporal trends were significant (P<.05).
A diagnosis from the morbidity composite in the setting of peripartum hysterectomy for PPH was more common in the setting of either uterine tamponade (160/233, 68.7%) or interventional radiology procedures (73/98, 74.5%) than no procedure (648/1606, 40.4%) (p<0.01). The risks for the morbidity composite with uterine tamponade versus no procedure and interventional radiology versus no procedure were significantly higher in both unadjusted (RR 1.70, 95% CI 1.54, 1.88 and RR 1.85, 95% CI 1.61, 2.12, respectively) and adjusted analyses (aRR 1.63, 95% CI 1.47, 1.81 and aRR 1.75 95% CI 1.51, 2.03, respectively) (Table 2).
Table 2.
Unadjusted and adjusted models for the morbidity composite among women undergoing peripartum hysterectomy with atony or secondary postpartum hemorrhage
| Unadjusted risk ratio (95%CI) | Adjusted risk ratio (95%CI) | |
|---|---|---|
| Procedure | ||
| Interventional radiology | 1.85 (1.61, 2.12)** | 1.75 (1.51, 2.03)** |
| Uterine tamponade | 1.70 (1.54, 1.88)** | 1.63 (1.47, 1.81)** |
| None | Referent | Referent |
| Year | ||
| 2006 | Referent | Referent |
| 2007 | 1.08 (0.80, 1.44) | 1.07 (0.80, 1.42) |
| 2008 | 1.34 (1.03, 1.73)* | 1.27 (1.00, 1.63) |
| 2009 | 1.36 (1.02, 1.81)* | 1.27 (0.96, 1.68) |
| 2010 | 1.16 (0.85, 1.59) | 1.08 (0.80, 1.46) |
| 2011 | 1.36 (1.05, 1.75)* | 1.29 (1.01, 1.65)* |
| 2012 | 1.50 (1.15, 1.95)* | 1.36 (1.06, 1.76)* |
| 2013 | 1.34 (1.03, 1.75)* | 1.20 (0.93, 1.56) |
| 2014 | 1.62 (1.26, 2.08)* | 1.43 (1.12, 1.82)* |
| Age in yearstam | ||
| 15–34 | Referent | Referent |
| 35–39 | 1.06 (0.91, 1.23) | 1.02 (0.86, 1.19) |
| 40–54 | 1.11 (0.95, 1.31) | 1.06 (0.89, 1.26) |
| Marital Status | ||
| Married | Referent | Referent |
| Single | 1.04 (0.93, 1.15) | 0.99 (0.87, 1.12) |
| Other/Unknown | 1.14 (0.99, 1.31) | 1.06 (0.93, 1.21) |
| Race | ||
| White | Referent | Referent |
| Black | 1.15 (0.98, 1.34) | 1.17 (1.01, 1.37)* |
| Other/Unknown | 1.17 (1.05, 1.30)* | 1.14 (1.02, 1.27)* |
| Payer | ||
| Medicaid | 1.01 (0.92, 1.12) | 1.00 (0.90, 1.11) |
| Commercial | Referent | Referent |
| Uninsured/unknown | 1.06 (0.87, 1.30) | 1.04 (0.86, 1.26) |
| Rurality | ||
| Urban | 1.33 (1.04, 1.70)* | 1.30 (1.03, 1.64)* |
| Rural | Referent | Referent |
| Teaching hospital | ||
| No | Referent | Referent |
| Yes | 0.99 (0.88, 1.12) | 0.89 (0.77, 1.03) |
| Hospital bed volume | ||
| <400 | Referent | Referent |
| 400–600 | 1.13 (0.99, 1.28) | 1.13 (0.99, 1.30) |
| >600 | 1.03 (0.86, 1.24) | 1.04 (0.85, 1.26) |
| Hospital region | ||
| Northeastern | Referent | Referent |
| Midwest | 1.04 (0.88, 1.24) | 1.07 (0.89, 1.28) |
| South | 0.96 (0.81, 1.13) | 0.99 (0.85, 1.16) |
| West | 1.10 (0.92, 1.30) | 1.05 (0.87, 1.27) |
| Preeclampsia | ||
| No | Referent | Referent |
| Yes | 1.23 (1.06, 1.41)* | 1.21 (1.05, 1.40)* |
CI, confidence interval.
P<0.05
p<0.01.
All variables in the table were included in the adjusted analyses.
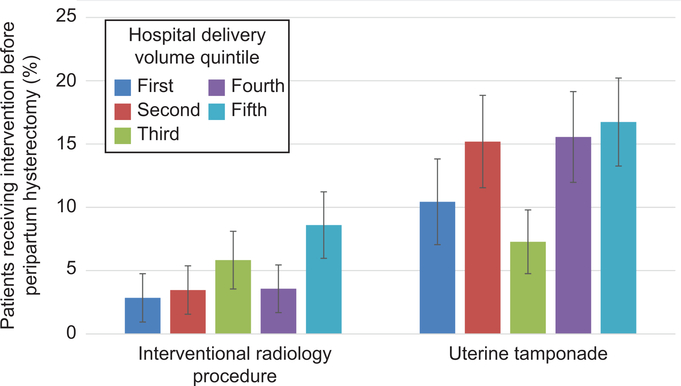
Evaluating uterine tamponade and interventional radiology procedures prior to peripartum hysterectomy for PPH by hospital volume quintile, both procedures were most common at the highest hospital volume quintile (8.6% and 16.7%, respectively) (Figure 4), while use of interventional radiology procedures were least common in the lowest volume quintile (2.9%) and uterine tamponade procedures were least common in the third (middle) volume quintile (7.3%) (p<0.01 for both comparisons of highest to lowest quintile).
Figure 4.
Proportion of patients receiving uterine tamponade and embolization before peripartum hysterectomy for atony by hospital volume quintile. Each quintile accounts for approximately 20% of overall deliveries, with the first quintile composed of the lowest volume hospitals and the fifth quintiles composed of the highest volume hospitals. Comparisons for both groups by hospital volume quintile were statistically significant (P<.01 for both groups).
Evaluation of the third secondary outcome, use of uterotonics, demonstrated that temporal trends were not statistically significant for use of methylergonovine (p=0.52) or carboprost (p=0.33) prior to hysterectomy but were significant for misoprostol increasing from 35.1% in 2006–2008 to 45.4% in 2012–2014 (p<0.01) (Appendix 4, available online at http://links.lww.com/xxx). Comparing use of uterotonics prior to hysterectomy based on hospital volume quintile, comparisons were significant for methylergonovine (p=0.01) and misoprostol (p<0.01) but not carboprost (p=0.27) (Appendix 5, available online at http://links.lww.com/xxx). For the ancillary analysis evaluating temporal trends in (i) transfusion, and (ii) hysterectomy, risk for these outcomes more than tripled in the setting of uterine tamponade procedures from 2006 to 2014 while differentials for interventional radiology procedures were modest (Table 3).
Table 3.
Risk for adverse outcomes among women with uterine tamponade and interventional radiology procedures
| Uterine tamponade procedures | |||||||||
| Morbidity composite | Transfusion | Hysterectomy | |||||||
| Absent | Present | Percent | Absent | Present | Percent | Absent | Present | Percent | |
| Year | |||||||||
| 2006 | n/a | ≤10 | n/a | 330 | 41 | 11.1% | n/a | ≤10 | n/a |
| 2007 | 473 | 15 | 3.1% | 431 | 57 | 11.7% | n/a | ≤10 | n/a |
| 2008 | 495 | 23 | 4.4% | 434 | 84 | 16.2% | 505 | 13 | 2.5% |
| 2009 | 599 | 36 | 5.7% | 509 | 126 | 19.8% | 611 | 24 | 3.8% |
| 2010 | 703 | 45 | 6.0% | 587 | 161 | 21.5% | 719 | 29 | 3.9% |
| 2011 | 806 | 80 | 9.0% | 663 | 223 | 25.2% | 845 | 41 | 4.6% |
| 2012 | 955 | 99 | 9.4% | 728 | 326 | 30.9% | 993 | 61 | 5.8% |
| 2013 | 952 | 97 | 9.2% | 740 | 309 | 29.5% | 994 | 55 | 5.2% |
| 2014 | 824 | 103 | 11.1% | 593 | 334 | 36.0% | 871 | 56 | 6.0% |
| Interventional radiology procedures | |||||||||
| Morbidity composite | Transfusion | Hysterectomy | |||||||
| Absent | Present | Percent | Absent | Present | Percent | Absent | Present | Percent | |
| Year | |||||||||
| 2006 | 67 | 28 | 29.5% | 28 | 67 | 70.5% | 80 | 15 | 15.8% |
| 2007 | 61 | 29 | 32.2% | 32 | 49 | 60.5% | n/a | ≤10 | n/a |
| 2008 | 60 | 30 | 33.3% | 28 | 62 | 68.9% | 76 | 14 | 15.6% |
| 2009 | 77 | 39 | 33.6% | 49 | 67 | 57.8% | 94 | 22 | 19.0% |
| 2010 | 88 | 45 | 33.8% | 49 | 84 | 63.2% | 103 | 30 | 22.6% |
| 2011 | 115 | 58 | 33.5% | 73 | 100 | 57.8% | 142 | 31 | 17.9% |
| 2012 | 108 | 73 | 40.3% | 71 | 110 | 60.8% | 146 | 35 | 19.3% |
| 2013 | 112 | 71 | 38.8% | 71 | 112 | 61.2% | 147 | 36 | 19.7% |
| 2014 | 93 | 54 | 36.7% | 66 | 81 | 55.1% | 111 | 36 | 24.5% |
Cell sizes ≤10 restricted to preserve patient confidentiality. Morbidity composite includes the 18 diagnoses in the CDC severe maternal morbidity composite excluding hysterectomy and transfusion. Temporal trends for each outcome for both UT and IR procedures were statistically significant (p<0.01).
DISCUSSION
This analysis found that use of both uterine tamponade and interventional radiology procedures increased significantly, supporting that these interventions became increasingly common in obstetric practice during the study period. In addition to use increasing among all deliveries, uterine tamponade and interventional radiology procedures were attempted prior to an increasing proportion of peripartum hysterectomies over the study period for PPH. In comparison, large differentials in use of uterotonics over the study period were not noted. Hospital volume appeared to be a significant factor in overall use of interventional radiology procedures. Use of interventional radiology procedures generally increased with hospital volume; in comparison, likelihood of use of tamponade did not appear to correlate with increased delivery volume. These latter findings related to interventional radiology procedures are intuitive given that higher volume centers may be more likely to have interventional radiology facilities and health care providers.
Use of uterine tamponade and interventional radiology procedures were both associated with increased risk for the morbidity composite in the setting of peripartum hysterectomy. In interpreting these findings an important consideration is that the degree to which peripartum hysterectomy was averted by uterine tamponade or interventional radiology procedures cannot be ascertained. While it may be possible that delaying peripartum hysterectomy in the setting of uterine tamponade or interventional radiology failure is associated with increased risk, it is not possible to determine to what degree these interventions prevented peripartum hysterectomy that otherwise would have occurred. Recent data from a hospital system in New York City found that adjusted peripartum hysterectomy risk decreased in the setting of increased balloon tamponade use as part of broader hemorrhage bundle implementation.31 Reduction of peripartum hysterectomy risk may be of particular importance in the United States. A recent analysis of administrative data in the US, England, and Australia found that risk of peripartum hysterectomy was significantly higher in the US compared to the other two countries.32 It is also possible that there is indication bias to some degree among patients who received uterine tamponade and interventional radiology interventions prior to hysterectomy compared to those who did not with patients at higher risk for severe morbidity receiving uterine tamponade and interventional radiology procedures. Decision making related to what clinical circumstances uterine tamponade and interventional radiology procedures were performed rather than initially performing hysterectomy cannot be gleaned from administrative data. A reasonable clinical inference from the study findings is that patients who fail uterine tamponade and interventional radiology interventions may be particularly high risk and more likely to require close surveillance and urgent intervention; preparation for emergent procedures including hysterectomy may be particularly important at smaller centers where surgical consultants and large blood volume may not be readily available.
In addition to not being able to estimate hysterectomy and other morbidity potentially averted by successful uterine tamponade and interventional radiology procedures, there are several important limitations to this analysis that should additionally be considered in interpreting the results. First, administrative data is collected primarily for billing and both misclassification and under-ascertainment are both potential concerns. Given that use of uterine tamponade procedures in particular increased several-fold over the study period, it is possible that these procedures could have been misclassified. Second and also related to the use of administrative data, this analysis does not allow for direct review of clinical records and we are not able to determine the sequence of clinical events including when medications were administered and whether interventional radiology procedures were performed after hysterectomy. It is possible that interventional radiology procedures were performed for delivery complications related to pelvic bleeding other than uterine atony, delayed postpartum hemorrhage, or placenta accreta; we cannot determine, for example, if some interventional radiology procedures occurred in the setting of bleeding after hysterectomy. For this reason, we may have overestimated the proportion of interventional radiology procedures occurring before hysterectomy and interventional radiology procedures may be a response to complicated peripartum hysterectomy, as opposed to a cause, of severe morbidity. Furthermore, we cannot determine to what degree health care providers used these procedures more frequently because of increasing availability and awareness of these clinical management options or because morbidity necessitating increased use, nor can we determine which specific device or intervention was used or whether procedures were performed optimally. Third, the sampling frame of hospitals included in this analysis changed over the study period and it is possible that estimates may be dependent to some degree on which centers were included; given the relatively modest number of uterine tamponade and interventional radiology interventions sensitivity analyses restricted to hospitals contributing data throughout the study period were not performed. Fourth, the hemorrhage bundle from the National Partnership for Maternal Safety was introduced after this study period and supported preparedness that could lead to more frequent and improved use of IT and interventional radiology procedures.3,33 Subsequent data is required to determine trends and outcomes in the context of these new clinical recommendations. Fifth, only a relatively small proportion of hysterectomies had a diagnosis of uterine atony or secondary postpartum hemorrhage. It is likely that a larger proportion of hysterectomies was performed for these indications and these results could be biased if uterine tamponade and interventional radiology procedures differ based on the presence of these secondary codes. Sixth, these data are now more than five years old and subsequent changes in clinical management may have transpired. Strengths of the study include the availability of a large sample over a nine-year period, the capability to query for both device use as well as procedure codes, and the ability to evaluate other interventions such as uteronic use.
In summary, this analysis found that uterine tamponade and interventional radiology procedures are becoming increasingly common, that they are used across obstetric volume settings, and that, although this relationship is not necessarily causal, peripartum hysterectomies in the setting uterine tamponade and interventional radiology procedures may be associated with higher risk.
Supplementary Material
Acknowledgments
Funding Dr. Friedman is supported by a career development award from the Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institutes of Health (K08HD082287).
Footnotes
Financial Disclosure Dr. Wright has served as a consultant for Tesaro and Clovis Oncology. Dr. D’Alton had a senior leadership role in ACOG II’s Safe Motherhood Initiative which received unrestricted funding from Merck for Mothers. The other authors did not report any potential conflicts of interest.
REFERENCES
- 1.Berg CJ, Harper MA, Atkinson SM, Bell EA, Hage ML. Preventability of pregnancy-related deaths - results of a state-wide review. Obstet Gynecol 2005;206:1228–34. [DOI] [PubMed] [Google Scholar]
- 2.Main EK. Decisions required for operating a maternal mortality review committee: the California experience. Semin Perinatol 2012;36:37–41. [DOI] [PubMed] [Google Scholar]
- 3.Main E, Goffman D, Scavone B, et al. National Partnership for Maternal Safety Consensus Bundle on Obstetric Hemorrhage. Obstet Gynecol 2015;126:155–62. [DOI] [PubMed] [Google Scholar]
- 4.Kramer MS, Berg C, Abenhaim H, et al. Incidence, risk factors, and temporal trends in severe postpartum hemorrhage. Am J Obstet Gynecol 2013;209:449 e1–7. [DOI] [PubMed] [Google Scholar]
- 5.Creanga AA, Syverson C, Seed K, Callaghan WM. Pregnancy-Related Mortality in the United States, 2011–2013. Obstet Gynecol 2017;130:366–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bateman BT, Berman MF, Riley LE, Leffert LR. The epidemiology of postpartum hemorrhage in a large, nationwide sample of deliveries. Anesth Analg 2010;110:1368–73. [DOI] [PubMed] [Google Scholar]
- 7.Committee on Practice Bulletins-Obstetrics. Practice Bulletin No. 183: Postpartum Hemorrhage. Obstet Gynecol 2017;130:e168–e86. [DOI] [PubMed] [Google Scholar]
- 8.Tindell K, Garfinkel R, Abu-Haydar E, et al. Uterine balloon tamponade for the treatment of postpartum haemorrhage in resource-poor settings: a systematic review. BJOG : an international journal of obstetrics and gynaecology 2013;120:5–14. [DOI] [PubMed] [Google Scholar]
- 9.Georgiou C Balloon tamponade in the management of postpartum haemorrhage: a review. BJOG : an international journal of obstetrics and gynaecology 2009;116:748–57. [DOI] [PubMed] [Google Scholar]
- 10.Vedantham S, Goodwin SC, McLucas B, Mohr G. Uterine artery embolization: an underused method of controlling pelvic hemorrhage. Am J Obstet Gynecol 1997;176:938–48. [DOI] [PubMed] [Google Scholar]
- 11.Kaya B, Tuten A, Daglar K, et al. B-Lynch uterine compression sutures in the conservative surgical management of uterine atony. Arch Gynecol Obstet 2015;291:1005–14. [DOI] [PubMed] [Google Scholar]
- 12.Burke TF, Thapa K, Shivkumar P, et al. Time for global scale-up, not randomized trials, of uterine balloon tamponade for postpartum hemorrhage. Int J Gynaecol Obstet 2018;142:115–8. [DOI] [PubMed] [Google Scholar]
- 13.Clark SL, Hankins GD. Preventing maternal death: 10 clinical diamonds. Obstet Gynecol 2012;119:360–4. [DOI] [PubMed] [Google Scholar]
- 14.Wright JD, Devine P, Shah M, et al. Morbidity and mortality of peripartum hysterectomy. Obstet Gynecol 2010;115:1187–93. [DOI] [PubMed] [Google Scholar]
- 15.Ruiz Labarta FJ, Pintado Recarte MP, Alvarez Luque A, et al. Outcomes of pelvic arterial embolization in the management of postpartum haemorrhage: a case series study and systematic review. European journal of obstetrics, gynecology, and reproductive biology 2016;206:12–21. [DOI] [PubMed] [Google Scholar]
- 16.Sathe NA, Likis FE, Young JL, Morgans A, Carlson-Bremer D, Andrews J. Procedures and Uterine-Sparing Surgeries for Managing Postpartum Hemorrhage: A Systematic Review. Obstetrical & gynecological survey 2016;71:99–113. [DOI] [PubMed] [Google Scholar]
- 17.Einerson BD, Son M, Schneider P, Fields I, Miller ES. The association between intrauterine balloon tamponade duration and postpartum hemorrhage outcomes. Am J Obstet Gynecol 2017;216:300 e1– e5. [DOI] [PubMed] [Google Scholar]
- 18.Revert M, Rozenberg P, Cottenet J, Quantin C. Intrauterine Balloon Tamponade for Severe Postpartum Hemorrhage. Obstetrics and gynecology 2018;131:143–9. [DOI] [PubMed] [Google Scholar]
- 19.Viteri OA, Sibai BM. Uterine balloon tamponade for the management of postpartum haemorrhage: A challenge and an opportunity for better evidence. BJOG : an international journal of obstetrics and gynaecology 2017. [DOI] [PubMed] [Google Scholar]
- 20.Hofmeyr GJ. Time to test tamponade. BJOG : an international journal of obstetrics and gynaecology 2017. [DOI] [PubMed] [Google Scholar]
- 21.Klebanoff MA, Snowden JM. Historical (retrospective) cohort studies and other epidemiologic study designs in perinatal research. Am J Obstet Gynecol 2018;219:447–50. [DOI] [PubMed] [Google Scholar]
- 22.Stulberg JJ, Delaney CP, Neuhauser DV, Aron DC, Fu P, Koroukian SM. Adherence to surgical care improvement project measures and the association with postoperative infections. Jama 2010;303:2479–85. [DOI] [PubMed] [Google Scholar]
- 23.About Premier healthcare database. 2016. (Accessed 10/24/2016, 2016, at https://www.premierinc.com/transforming-healthcare/healthcare-performance-improvement/premier-research-services/.)
- 24.Swanson SJ MB, Gunnarsson CL, Moore M, Howington JA, Maddaus MA, McKenna RJ, Miller DL. Video-assisted thoracoscopic lobectomy is less costly and morbid than open lobectomy: a retrospective multiinstitutional database analysis. Ann Thorac Surg 2012;93(4):1027–32. [DOI] [PubMed] [Google Scholar]
- 25.Bateman BT, Tsen LC, Liu J, Butwick AJ, Huybrechts KF. Patterns of second-line uterotonic use in a large sample of hospitalizations for childbirth in the United States: 2007–2011. Anesth Analg 2014;119:1344–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bateman BT, Huybrechts KF, Hernandez-Diaz S, Liu J, Ecker JL, Avorn J. Methylergonovine maleate and the risk of myocardial ischemia and infarction. Am J Obstet Gynecol 2013;209:459 e1– e13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.The Centers for Disease Control and Prevention. How does the CDC identify severe maternal morbidity. Accessed December 2, 2019 Available at: https://www.cdc.gov/reproductivehealth/maternalinfanthealth/smm/severe-morbidity-ICD.htm.
- 28.Kuklina E, Whiteman M, Hillis S, Jameieson D, Meikle S, Posner S. An enhanced method for identifying obstetric deliveries: implications for estimating maternal morbidity. Matern Child Health J 2008;12:469–77. [DOI] [PubMed] [Google Scholar]
- 29.Friedman AM, Wright JD, Ananth CV, Siddiq Z, D’Alton ME, Bateman BT. Population-based risk for peripartum hysterectomy during low- and moderate-risk delivery hospitalizations. Am J Obstet Gynecol 2016;215:640 e1– e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Callaghan WM, Kuklina EV, Berg CJ. Trends in postpartum hemorrhage: United States, 1994–2006. Am J Obstet Gynecol 2010;202:353 e1–6. [DOI] [PubMed] [Google Scholar]
- 31.Goffman D, Friedman AM, Sheen JJ, et al. A Framework for Improving Characterization of Obstetric Hemorrhage Using Informatics Data. Obstet Gynecol 2019;134:1317–25. [DOI] [PubMed] [Google Scholar]
- 32.Lipkind H, Zuckerwise L, Turner E, et al. Severe maternal morbidity during delivery hospitalisation in a large international administrative database, 2008–2013: a retrospective cohort. BJOG : an international journal of obstetrics and gynaecology 2019. [DOI] [PubMed] [Google Scholar]
- 33.D’Alton ME, Main EK, Menard MK, Levy BS. The National Partnership for Maternal Safety. Obstet Gynecol 2014;123:973–7. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.